Inhibition of Advanced Glycation End-Product Formation and Antioxidant Activity by Extracts and Polyphenols from Scutellaria alpina L. and S. altissima L.
Abstract
:1. Introduction
2. Results
3. Discussion
4. Experimental Section
4.1. Plant Material
4.2. Preparation of Extracts
4.3. Chemicals
4.4. Total Flavonoid Content
4.5. DPPH Radical Scavenging Assay
4.6. FRAP Assay
4.7. Linoleic Acid Peroxidation Inhibition
4.8. Antiglycation Assay
4.9. Statistical Analysis
5. Conclusion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Séro, L.; Sanguinet, L.; Blanchard, P.; Dang, B.T.; Morel, S.; Richomme, P.; Seraphin, D.; Derbré, S. Tuning a 96-well microtiter plate fluorescence-based assay to identify AGE inhibitors in crude plant extracts. Molecules 2013, 18, 14320–14339. [Google Scholar] [CrossRef] [PubMed]
- Fukami, K.; Yamagishi, S.; Ueda, S.; Okuda, S. Role of AGEs in diabetic nephropathy. Curr. Pharm. Des. 2008, 14, 946–952. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, K.; Yasujima, M.; Yagihashi, S. Role of advanced glycation end products in diabetic neuropathy. Curr. Pharm. Des. 2008, 14, 953–961. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, S.I.; Nakamura, K.; Matsui, T.; Ueda, S.; Noda, Y.; Imaizumi, T. Inhibitors of advanced glycation end products (AGEs): Potential utility for the treatment of cardiovascular disease. Cardiovasc. Ther. 2008, 26, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, G.; Satya, S.; Khandelwal, R.K.; Naik, S.N. Commonly consumed Indian plant food materials in the management of diabetes mellitus. Diabetes Metab. Syndr. Clin. Res. Rev. 2010, 4, 21–40. [Google Scholar] [CrossRef]
- Jung, H.A.; Kim, Y.S.; Choi, J.S. Quantitative HPLC analysis of two key flavonoids and inhibitory activities against aldose reductase from different parts of the Korean thistle, Cirsium maackii. Food Chem. Toxicol. 2009, 47, 2790–2797. [Google Scholar] [CrossRef] [PubMed]
- Tsuji-Naito, K.; Saeki, H.; Hamano, M. Inhibitory effects of Chrysanthemum species extracts on formation of advanced glycation end products. Food Chem. 2009, 116, 854–859. [Google Scholar] [CrossRef]
- Reddy, V.P.; Beyaz, A. Inhibitors of the Maillard reaction and AGE breakers as therapeutics for multiple diseases. Drug Discov. Today 2006, 11, 646–654. [Google Scholar] [CrossRef] [PubMed]
- McDougall, G.J.; Shapiro, F.; Dobson, P.; Smith, P.; Blake, A.; Stewart, D. Different polyphenolic components of soft fruits inhibit α-amylase and α-glucosidase. J. Agr. Food Chem. 2005, 53, 2760–2766. [Google Scholar] [CrossRef] [PubMed]
- Pinto, M.D.S.; Kwon, Y.I.; Apostolidis, E.; Lajolo, F.M.; Genovese, M.I.; Shetty, K. Potential of Ginkgo biloba L. leaves in the management of hyperglycemia and hypertension using in vitro models. Bioresource Technol. 2009, 100, 6599–6609. [Google Scholar] [CrossRef] [PubMed]
- Shang, X.; He, X.; He, X.; Li, M.; Zhang, R.; Fan, P.; Zhang, Q.; Jia, Z. The genus Scutellaria an ethnopharmacological and phytochemical review. J. Ethnopharmacol. 2010, 128, 279–313. [Google Scholar] [CrossRef] [PubMed]
- Hou, G.Y.; Wang, L.; Liu, S.; Song, F.R.; Liu, Z.Q. Inhibitory effect of eleven herbal extracts on advanced glycation end-products formation and aldose reductase activity. Chinese Chem. Lett. 2014, 25, 1039–1043. [Google Scholar] [CrossRef]
- Grzegorczyk-Karolak, I.; Wysokińska, H.; Olas, B. Studies on the antioxidant properties of extracts from the roots and shoots of two Scutellaria species in human blood plasma. Acta. Biochim. Pol. 2015, 62, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Grzegorczyk-Karolak, I.; Kuźma, Ł.; Wysokińska, H. The use of long-term Scutellaria altissima callus cultures for shoot regeneration, production of bioactive metabolites and micropropagation. J. Med. Plants Res. 2013, 7, 3003–3313. [Google Scholar]
- Grzegorczyk-Karolak, I.; Kuźma, Ł.; Wysokińska, H. Study on the chemical composition and antioxidant activity of extracts from shoot culture and regenerated plants of Scutellaria altissima L. Acta. Physiol. Plant 2015, 37, 1736–1744. [Google Scholar] [CrossRef]
- Kazeem, M.I.; Akanji, M.A.; Hafizur, R.M.; Choudhary, M.I. Antiglycation, antioxidant and toxicological potential of polyphenol extracts of alligator pepper, ginger and nutmeg from Nigeria. Asian Pac. J. Trop. Biomem. 2012, 2, 727–732. [Google Scholar] [CrossRef]
- Povichit, N.; Phrutivorapongkul, A.; Suttajit, M.; Chaiyasut, C.; Leelapornpisid, P. Phenolic content and in vitro inhibitory effects on oxidation and protein glycation of some Thai medicinal plants. Pak. J. Pharm. Sci. 2010, 23, 403–408. [Google Scholar] [PubMed]
- Procházková, D.; Boušová, I.; Wilhelmová, N. Antioxidant and prooxidant properties of flavonoids. Fitoterapia 2011, 82, 513–523. [Google Scholar] [CrossRef] [PubMed]
- Kyriakopoulou, I.; Magiatis, P.; Skaltsounis, A.L.; Aligiannis, N.; Harvala, C. Samioside, a new phenylethanoid glycoside with free-radical scavenging and antimicrobial activities from Phlomis samia. J. Nat. Prod. 2001, 64, 1095–1097. [Google Scholar] [CrossRef] [PubMed]
- Thornalley, P.J. Use of aminoguanidine (Pimagedine) to prevent the formation of advanced glycation endproducts. Arch. Biochem. Biophys. 2003, 419, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Parengkuan, L.; Yagi, M.; Matsushima, M.; Ogura, M.; Hamada, U.; Yonei, Y. Anti-glycation activity of various fruits. Anti-Aging Med. 2013, 10, 70–76. [Google Scholar]
- Rahbar, S.; Figarola, J.L. Novel inhibitors of advanced glycation endproducts. Arch. Biochem. Biophys. 2003, 419, 63–79. [Google Scholar] [CrossRef]
- Nakagawa, T.; Yokozawa, T.; Terasawa, K.; Shu, S.; Juneja, L.R. Protective activity of green tea against free radical-and glucose-mediated protein damage. J. Agr. Food Chem. 2002, 50, 2418–2422. [Google Scholar] [CrossRef]
- Chen, Y.F.; Roan, H.Y.; Li, C.K.; Huang, Y.C.; Wang, T.S. Relationship between antioxidant and antiglycation ability of saponins, polyphenols, and polysaccharides in Chinese herbal medicines used to treat diabetes. J. Med. Plants Res. 2011, 5, 2322–2331. [Google Scholar]
- Harris, C.S.; Cuerrier, A.; Lamont, E.; Haddad, P.S.; Arnason, J.T.; Bennett, S.A.; Johns, T. Investigating wild berries as a dietary approach to reducing the formation of advanced glycation endproducts: Chemical correlates of in vitro antiglycation activity. Plant Foods Hum. Nutr. 2014, 69, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Grzegorczyk, I.; Matkowski, A.; Wysokińska, H. Antioxidant activity of extracts from in vitro cultures of Salvia officinalis L. Food Chem. 2007, 104, 536–541. [Google Scholar] [CrossRef]
- Rice-Evans, C.; Miller, N.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Wu, C.H.; Yen, G.C. Inhibitory effect of naturally occurring flavonoids on the formation of advanced glycation endproducts. J. Agr. Food Chem. 2005, 53, 3167–3173. [Google Scholar] [CrossRef] [PubMed]
- Dearlove, R.P.; Greenspan, P.; Hartle, D.K.; Swanson, R.B.; Hargrove, J.L. Inhibition of protein glycation by extracts of culinary herbs and spices. J. Med. Food 2008, 11, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Ramkissoon, J.S.; Mahomoodally, M.F.; Ahmed, N.; Subratty, A.H. Relationship between total phenolic content, antioxidant potential, and antiglycation abilities of common culinary herbs and spices. J. Med. Food 2012, 15, 1116–1123. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, H.; Wang, T.; Managi, H.; Yoshikawa, M. Structural requirements of flavonoids for inhibition of protein glycation and radical scavenging activities. Bioorgan. Med. Chem. 2003, 11, 5317–5323. [Google Scholar] [CrossRef]
- Yu, S.Y.; Lee, I.S.; Jung, S.H.; Lee, Y.M.; Lee, Y.R.; Kim, J.H.; Sun, H.; Kim, J.S. Caffeoylated phenylpropanoid glycosides from Brandisia hancei inhibit advanced glycation end product formation and aldose reductase in vitro and vessel dilation in larval zebrafish in vivo. Planta Med. 2013, 79, 1705–1709. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, T.; Ninomiya, K.; Imamura, M.; Akaki, J.; Fujikura, S.; Pan, Y.; Yuan, D.; Yoshikawa, M.; Jia, X.; Li, Z.; et al. Acylated phenylethanoid glycosides, echinacoside and acteoside from Cistanche tubulosa, improve glucose tolerance in mice. J. Nat. Med. 2014, 68, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Lamaison, J.L.; Carnat, A. Levels of principal flavonoids in flowers and leaves of Crataegus monogyna Jacq and Crataegus laevigata (Poiret) Dc (Rosaceae). Pharm. Acta Helv. 1990, 65, 315–320. [Google Scholar]
- Weremczuk-Jeżyna, I.; Grzegorczyk-Karolak, I.; Frydrych, B.; Królicka, A.; Wysokińska, H. Hairy roots of Dracocephalum moldavica: Rosmarinic acid content and antioxidant potential. Acta. Physiol. Plant 2013, 35, 2095–2103. [Google Scholar] [CrossRef]
- Pulido, R.; Bravo, L.; Saura-Calixto, F. Antioxidant activity of dietary polyphenols as determined by a modified ferric reducing/antioxidant power assay. J. Agr. Food Chem. 2000, 48, 3396–3402. [Google Scholar] [CrossRef]
- Choi, C.W.; Kim, S.C.; Hwang, S.S.; Choi, B.K.; Ahn, H.J.; Lee, M.Y.; Park, S.H.; Kim, S.K. Antioxidant activity and free radical scavenging capacity between Korean medicinal plants and flavonoids by assay-guided comparison. Plant Sci. 2002, 163, 1161–1168. [Google Scholar] [CrossRef]
- Bhatwadekar, A.D.; Ghole, V.S. Rapid method for the preparation of an AGE-BSA standard calibrator using thermal glycation. J. Clin. Lab. Anal. 2005, 19, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are available from the authors.
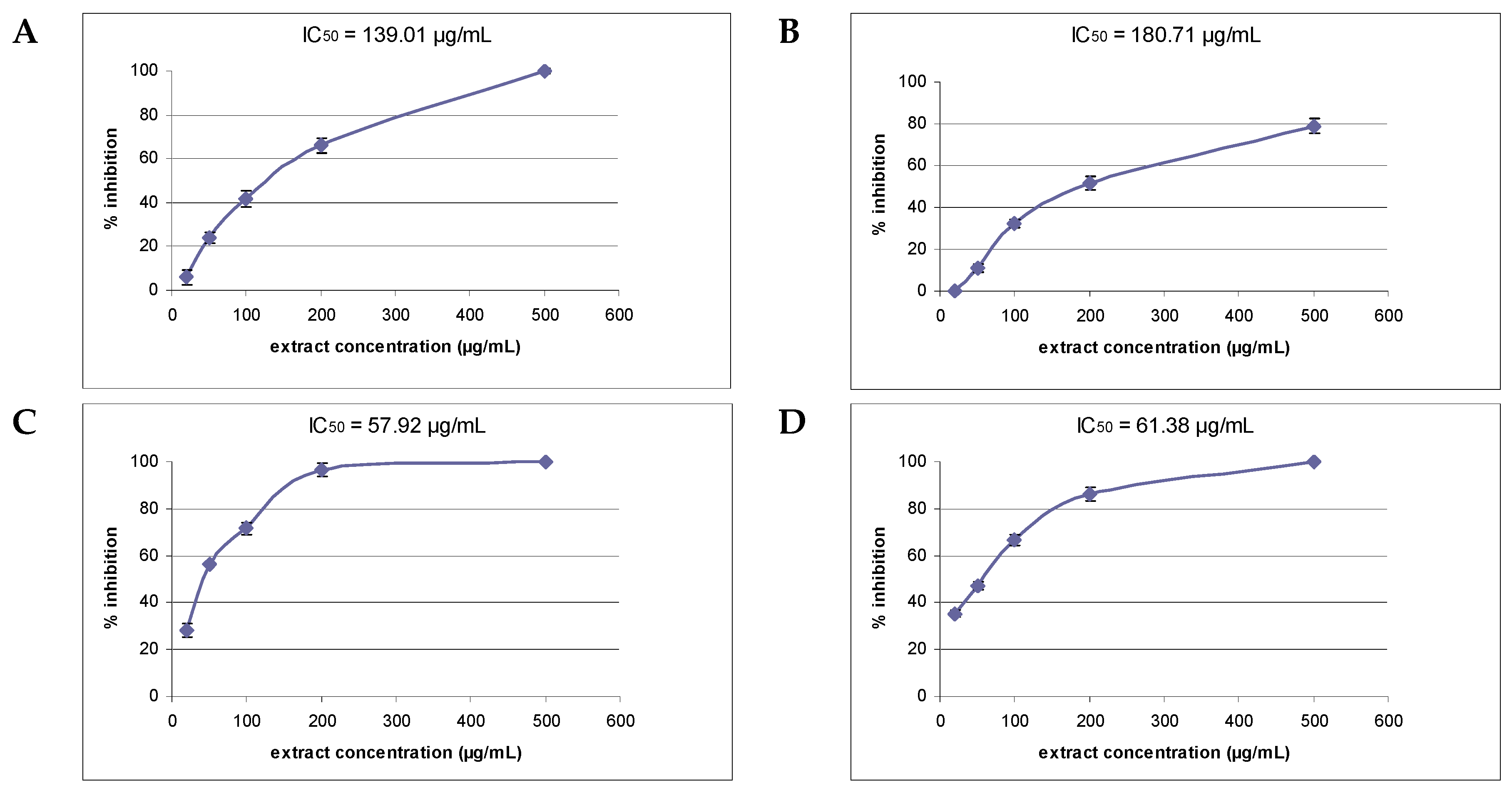
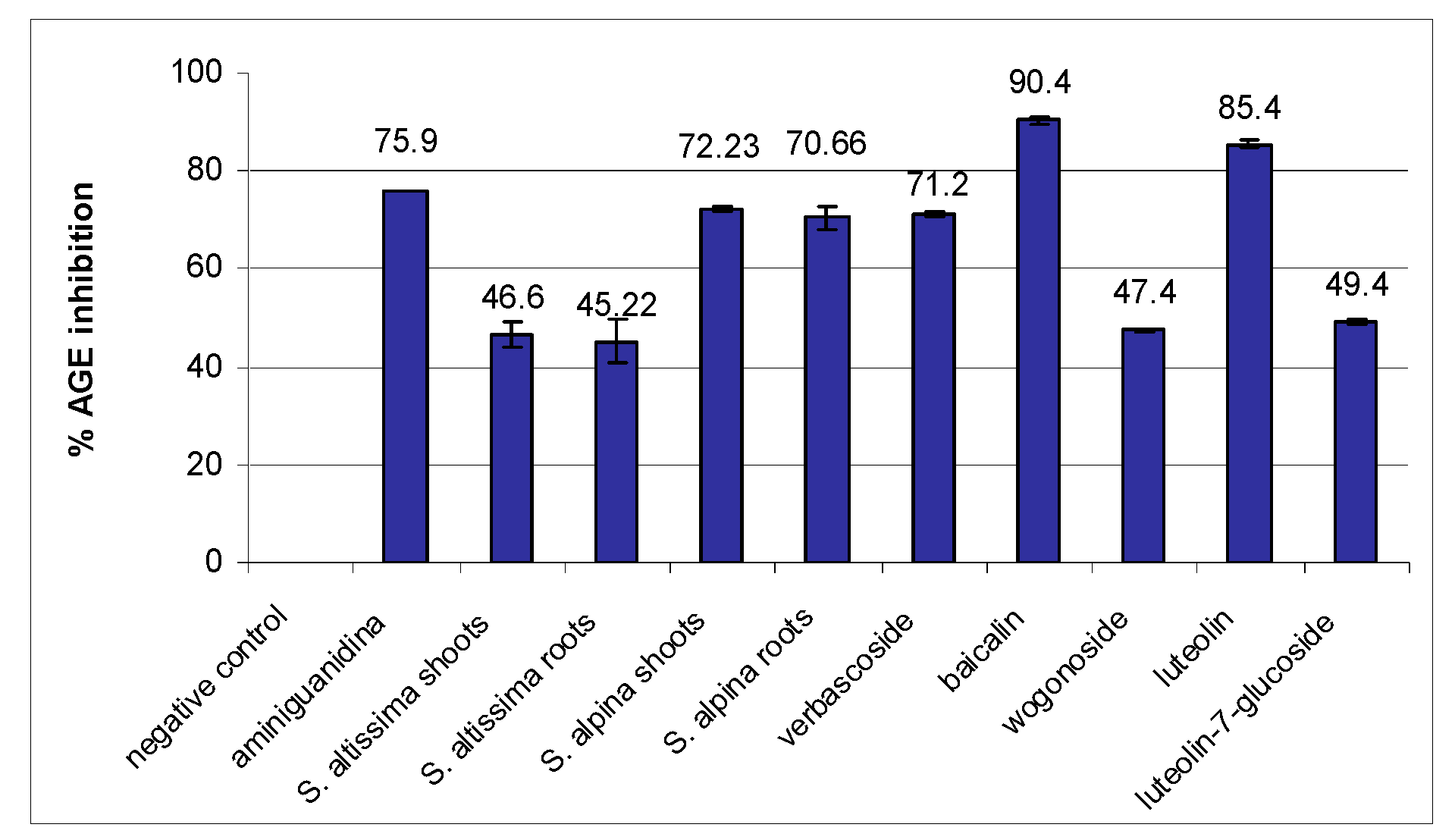
| Plant Material | Total Flavonoid Content * | FRAP µM Fe(II)/g Dry Extract | DPPH Assay EC50 (µg/mL) ** | % Inhibition Lipid Peroxidation |
|---|---|---|---|---|
| S. altissima shoots | 14.01 ± 0.15 a | 391.66 ± 6.13 c | 102.68 ± 3.91 d | 29.76 ± 1.19 d |
| S. altissima roots | 13.83 ± 0.13 a | 368.37 ± 7.23 d | 82.94 ± 0.45 c | 36.90 ± 0.84 c |
| S. alpina shoots | 25.05 ± 0.43 b | 669.63 ± 11.07 b | 71.26 ± 0.34 b | 51.85 ± 3.75 b |
| S. alpina roots | 27.16 ± 0.87 b | 717.60 ± 1.35 a | 68. 98 ±0.30 a | 60.32 ± 1.61 a |
| Correlation Coefficient (r) | Flavonoid Content | Verbascoside Content | FRAP | DPPH (EC50) | % Inhibition LPO |
|---|---|---|---|---|---|
| % inhibition AGE formation | 0.99 | 0.73 | 0.99 | −0.83 | 0.93 |
| Linear regression | y = 2.05x + 17.57 | y = 3.85x + 44.92 | y = 0.08x + 15.71 | y = −0.79x + 123.18 | y = 0.98x + 14.72 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grzegorczyk-Karolak, I.; Gołąb, K.; Gburek, J.; Wysokińska, H.; Matkowski, A. Inhibition of Advanced Glycation End-Product Formation and Antioxidant Activity by Extracts and Polyphenols from Scutellaria alpina L. and S. altissima L. Molecules 2016, 21, 739. https://doi.org/10.3390/molecules21060739
Grzegorczyk-Karolak I, Gołąb K, Gburek J, Wysokińska H, Matkowski A. Inhibition of Advanced Glycation End-Product Formation and Antioxidant Activity by Extracts and Polyphenols from Scutellaria alpina L. and S. altissima L. Molecules. 2016; 21(6):739. https://doi.org/10.3390/molecules21060739
Chicago/Turabian StyleGrzegorczyk-Karolak, Izabela, Krzysztof Gołąb, Jakub Gburek, Halina Wysokińska, and Adam Matkowski. 2016. "Inhibition of Advanced Glycation End-Product Formation and Antioxidant Activity by Extracts and Polyphenols from Scutellaria alpina L. and S. altissima L." Molecules 21, no. 6: 739. https://doi.org/10.3390/molecules21060739
APA StyleGrzegorczyk-Karolak, I., Gołąb, K., Gburek, J., Wysokińska, H., & Matkowski, A. (2016). Inhibition of Advanced Glycation End-Product Formation and Antioxidant Activity by Extracts and Polyphenols from Scutellaria alpina L. and S. altissima L. Molecules, 21(6), 739. https://doi.org/10.3390/molecules21060739






