RETRACTED: The Aminopyridinol Derivative BJ-1201 Protects Murine Hippocampal Cells against Glutamate-Induced Neurotoxicity via Heme Oxygenase-1
Abstract
:1. Introduction
2. Results
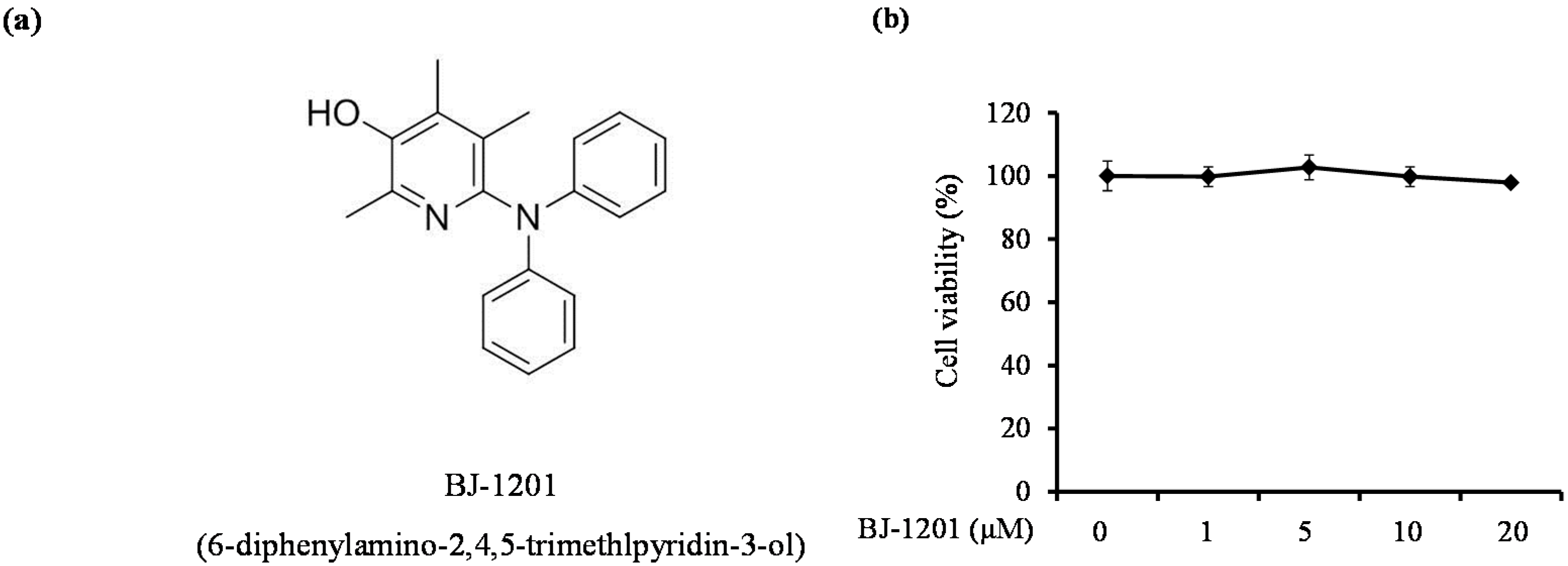
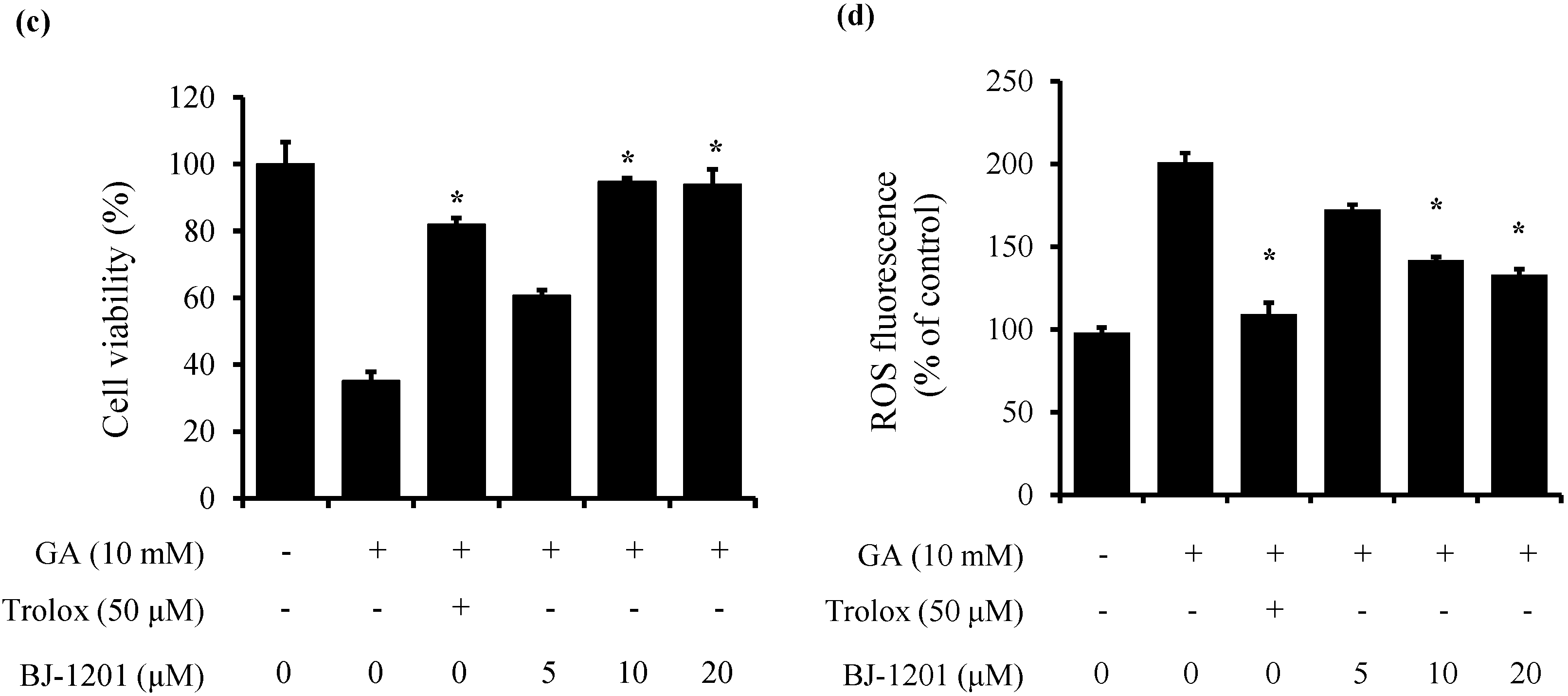
2.1. BJ-1201 Inhibits Glutamate-Induced Oxidative Neurotoxicity
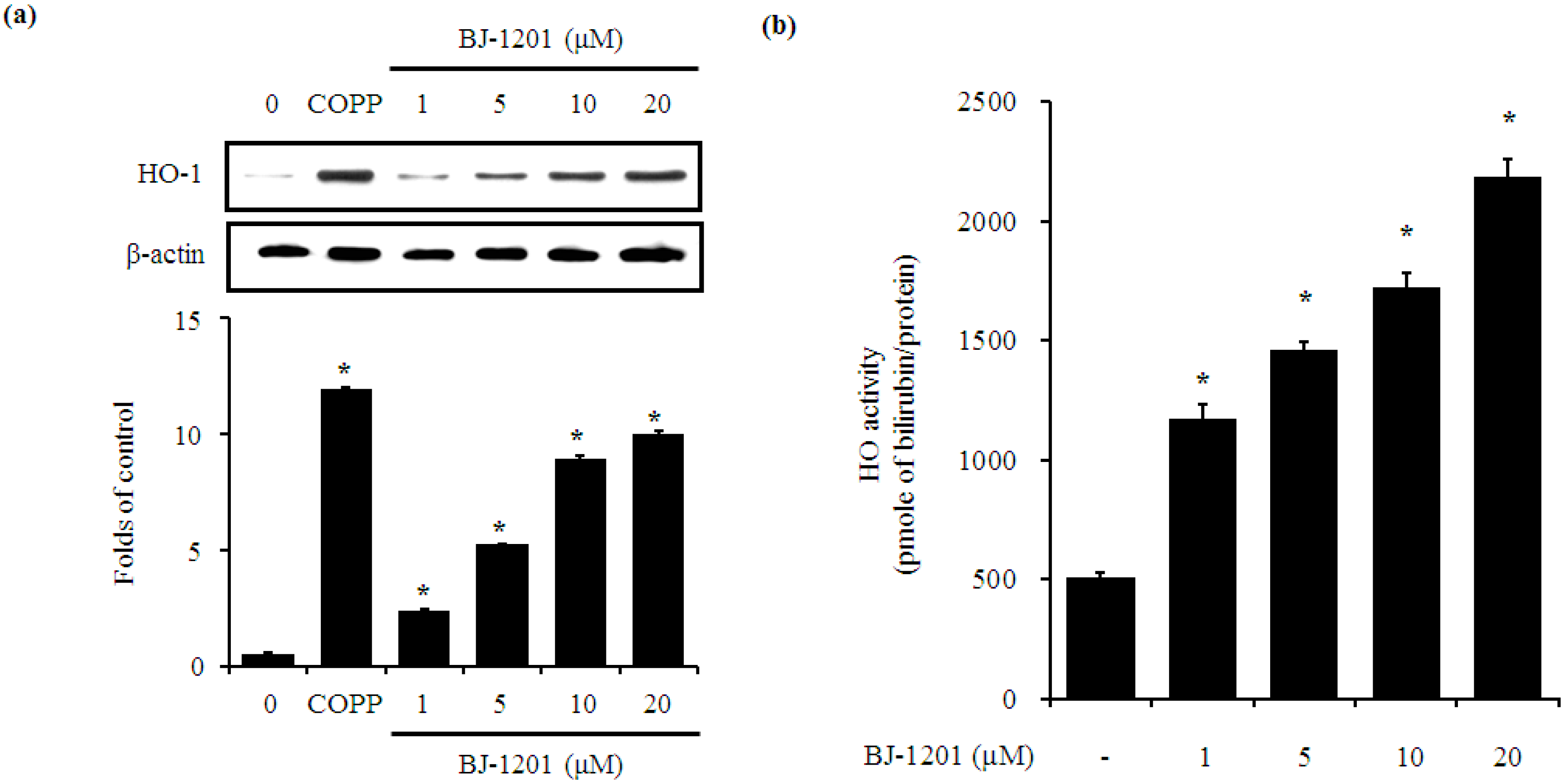
2.2. BJ-1201 Upregulates HO-1 Expression
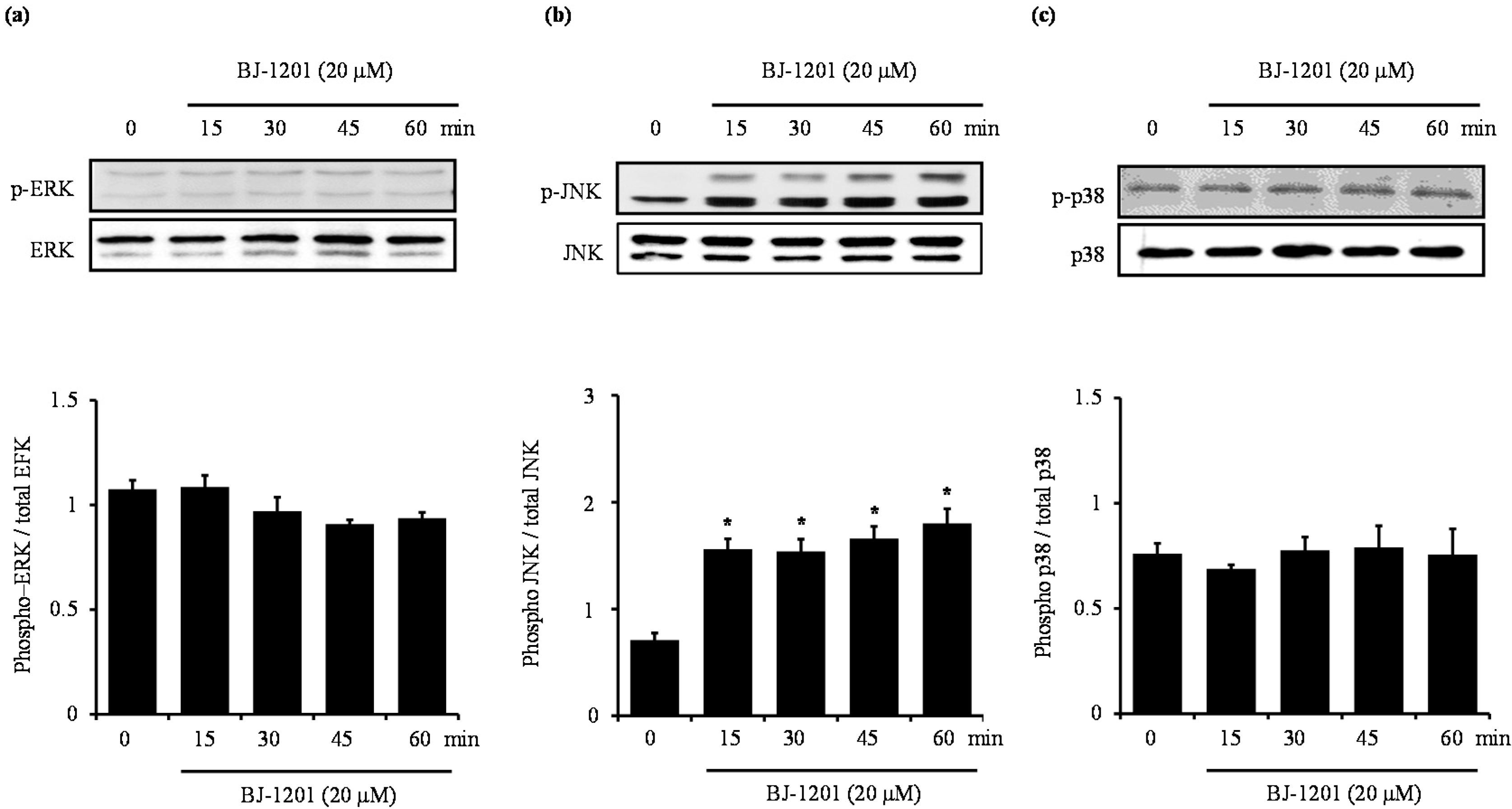
2.3. BJ-1201 Activates MAPK in HT22 Cells
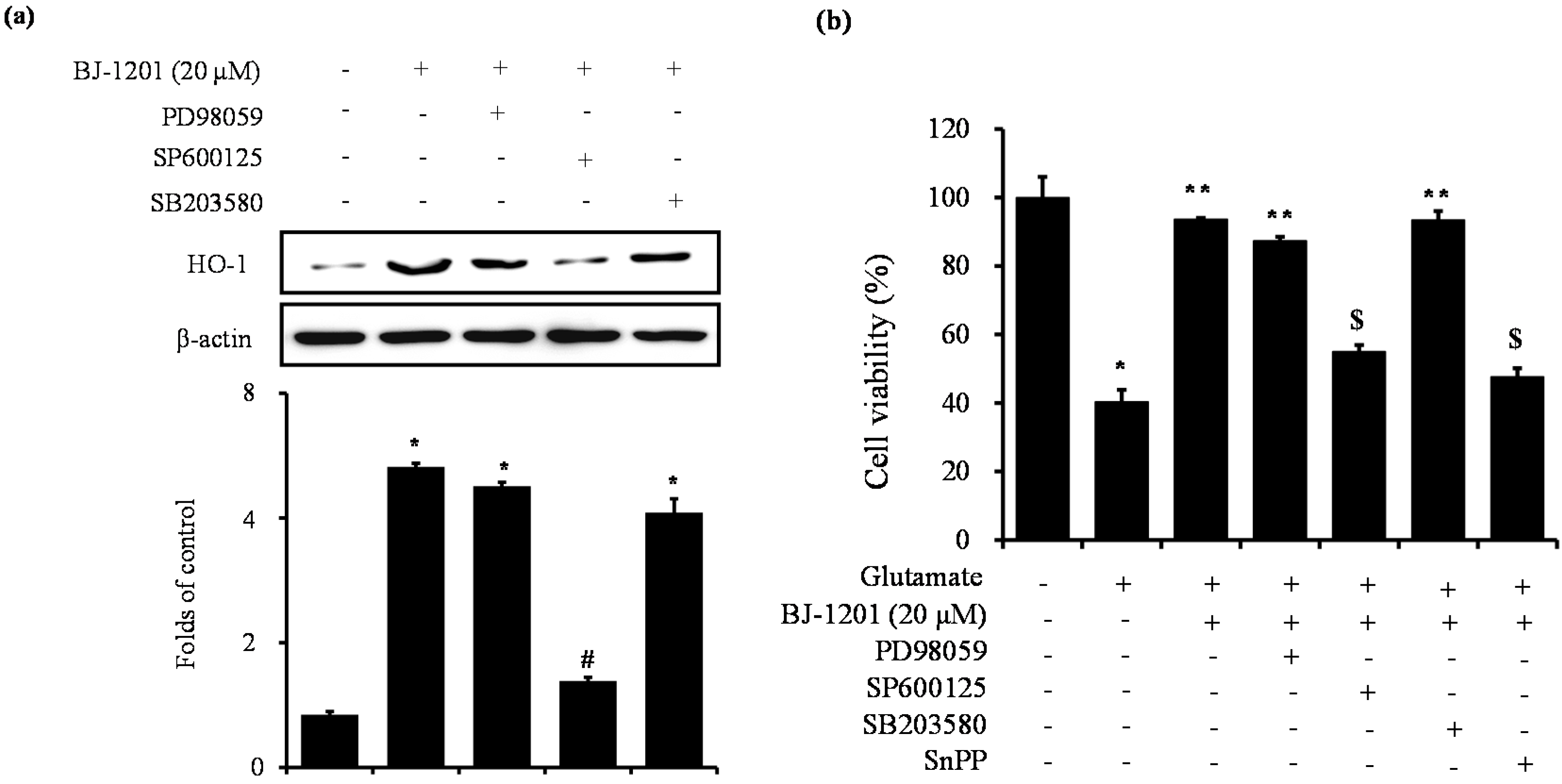
2.4. Involvement of JNK in BJ-1201-Inducing HO-1 Expression and Glutamate-Induced Neurotoxicity
2.5. BJ-1201 Induced Upregulation and Nrf-2 Nuclear Translocation in HT22 Cells
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Culture
4.3. Cell Viability
4.4. Measurement of Reactive Oxygen Species
4.5. HO-1 Activity
4.6. Preparation of Nuclear and Cytosolic Extraction
4.7. Western Blot Analysis
4.8. Luciferase Assay
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| HO-1 | heme oxygenase-1 |
| Nrf2 | nuclear transcription factor erythroid-2 related factor 2 |
| AREs | antioxidant response elements |
| MAPKs | mitogen-activated protein kinases |
References
- Jazwa, A.; Cuadrado, A. Targeting heme oxygenase-1 for neuroprotection and neuroinflammation in neurodegenerative diseases. Curr. Drug Targets 2010, 11, 1517–1531. [Google Scholar] [CrossRef] [PubMed]
- Markesbery, W.R. Oxidative stress hypothesis in Alzheimer’s disease. Free Radic. Biol. Med. 1997, 23, 134–147. [Google Scholar] [CrossRef]
- Maher, P.; Davis, J.B. The role of monoamine metabolism in oxidative glutamate toxicity. J. Neurosci. 1996, 16, 6394–6401. [Google Scholar] [PubMed]
- Flora, S.J. Structural, chemical and biological aspects of antioxidants for strategies against metal and metalloid exposure. Oxid. Med. Cell Longev. 2009, 2, 191–206. [Google Scholar] [CrossRef] [PubMed]
- Youdim, K.A.; Martin, A.; Joseph, J.A. Incorporation of the elderberry anthocyanins by endothelial cells increases protection against oxidative stress. Free Radic. Biol. Med. 2000, 29, 51–60. [Google Scholar] [CrossRef]
- Padgaonkar, V.A.; Leverenz, V.R.; Dang, L.; Chen, S.C.; Pelliccia, S.; Giblin, F.J. Thioredoxin reductase may be essential for the normal growth of hyperbaric oxygen-treated human lens epithelial cells. Exp. Eye Res. 2004, 79, 847–857. [Google Scholar] [CrossRef] [PubMed]
- Farombi, E.O.; Surh, Y.J. Heme oxygenase-1 as a potential therapeutic target for hepatoprotection. J. Biochem. Mol. Biol. 2006, 39, 479–491. [Google Scholar] [CrossRef] [PubMed]
- Chodorowski, Z.; Sein Anand, J.; Rybakowska, I.; Kaletha, K. Heme oxygenase as a potential therapeutic target for hepatoprotection. Prz. Lek. 2007, 64, 360–362. [Google Scholar] [PubMed]
- Rossler, O.G.; Bauer, I.; Chung, H.Y.; Thiel, G. Glutamate-induced cell death of immortalized murine hippocampal neurons: Neuroprotective activity of heme oxygenase-1, heat shock protein 70, and sodium selenite. Neurosci. Lett. 2004, 362, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Immenschuh, S.; Ramadori, G. Gene regulation of heme oxygenase-1 as a therapeutic target. Biochem. Pharm. 2000, 60, 1121–1128. [Google Scholar] [CrossRef]
- Yang, H.J.; Weon, J.B.; Lee, B.; Ma, C.J. The alteration of components in the fermented Hwangryunhaedok-tang and its neuroprotective activity. Pharmacogn. Mag. 2011, 7, 207–212. [Google Scholar] [PubMed]
- Cho, N.; Choi, J.H.; Yang, H.; Jeong, E.J.; Lee, K.Y.; Kim, Y.C.; Sung, S.H. Neuroprotective and anti-inflammatory effects of flavonoids isolated from Rhus verniciflua in neuronal HT22 and microglial BV2 cell lines. Food Chem. Toxicol. 2012, 50, 1940–1945. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.S.; Li, B.; Kim, K.S.; Jeong, G.S.; Kim, E.C.; Kim, Y.C. Butein protects human dental pulp cells from hydrogen peroxide-induced oxidative toxicity via Nrf2 pathway-dependent heme oxygenase-1 expressions. Toxicol. In Vitro 2013, 27, 874–881. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.Q.; Ding, J.; Xiao, Z.H.; Liu, C.M. Puerarin ameliorates carbon tetrachloride-induced oxidative DNA damage and inflammation in mouse kidney through ERK/Nrf2/ARE pathway. Food Chem. Toxicol. 2014, 71, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Wijtmans, M.; Pratt, D.A.; Valgimigli, L.; DiLabio, G.A.; Pedulli, G.F.; Porter, N.A. 6-Amino-3-pyridinols: Towards diffusion-controlled chain-breaking antioxidants. Angew. Chem. Int. Ed. Engl. 2003, 42, 4370–4373. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.G.; Kang, Y.; Lee, H.; Lee, E.K.; Nam, T.G.; Kim, J.A.; Jeong, B.S. 6-Amino-2,4,5-trimethylpyridin-3-ols: A new general synthetic route and antiangiogenic activity. Eur. J. Med. Chem. 2014, 78, 126–139. [Google Scholar] [CrossRef] [PubMed]
- Kaizaki, A.; Tanaka, S.; Ishige, K.; Numazawa, S.; Yoshida, T. The neuroprotective effect of heme oxygenase (HO) on oxidative stress in HO-1 siRNA-transfected HT22 cells. Brain Res. 2006, 1108, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Ishii, T.; Itoh, K.; Takahashi, S.; Sato, H.; Yanagawa, T.; Katoh, Y.; Bannai, S.; Yamamoto, M. Transcription factor Nrf2 coordinately regulates a group of oxidative stress-inducible genes in macrophages. J. Biol. Chem. 2000, 275, 16023–16029. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Calkins, M.J.; Chan, K.; Kan, Y.W.; Johnson, J.A. Identification of the NF-E2-related factor-2-dependent genes conferring protection against oxidative stress in primary cortical astrocytes using oligonucleotide microarray analysis. J. Biol. Chem. 2003, 278, 12029–12038. [Google Scholar] [CrossRef] [PubMed]
- Fransen, M.; Nordgren, M.; Wang, B.; Apanasets, O. Role of peroxisomes in ROS/RNS-metabolism: Implications for human disease. Biochim. Biophys. Acta 2012, 1822, 1363–1373. [Google Scholar] [CrossRef] [PubMed]
- Michaelis, E.K. Molecular biology of glutamate receptors in the central nervous system and their role in excitotoxicity, oxidative stress and aging. Prog. Neurobiol. 1998, 54, 369–415. [Google Scholar] [CrossRef]
- Deecher, D.C.; Daoud, P.; Bhat, R.A.; O’Connor, L.T. Endogenously expressed estrogen receptors mediate neuroprotection in hippocampal cells (HT22). J. Cell Biochem. 2005, 95, 302–312. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Li, L.; Suo, W.Z. HT22 hippocampal neuronal cell line possesses functional cholinergic properties. Life Sci. 2009, 84, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.W. Glutamate neurotoxicity and diseases of the nervous system. Neuron 1988, 1, 623–634. [Google Scholar] [CrossRef]
- Lipton, S.A. Pathologically activated therapeutics for neuroprotection. Nat. Rev. Neurosci. 2007, 8, 803–808. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.; Schubert, D.; Maher, P. Oxytosis: A novel form of programmed cell death. Curr. Top. Med. Chem. 2001, 1, 497–506. [Google Scholar] [PubMed]
- Mattson, M.P. Apoptosis in neurodegenerative disorders. Nat. Rev. Mol. Cell Biol. 2000, 1, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Jeong, G.S.; An, R.B.; Pae, H.O.; Chung, H.T.; Yoon, K.H.; Kang, D.G.; Lee, H.S.; Kim, Y.C. Cudratricusxanthone A protects mouse hippocampal cells against glutamate-induced neurotoxicity via the induction of heme oxygenase-1. Planta Med. 2008, 74, 1368–1373. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Lu, R.; Zhang, B.; Shen, J.; Yang, L.; Xiao, S.; Liu, J.; Suo, W.Z. Differentiation of HT22 neurons induces expression of NMDA receptor that mediates homocysteine cytotoxicity. Neurol. Res. 2012, 34, 38–43. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Liu, J.; Cheng, S.; Xing, Y.; Suo, W.Z. Differentiation renders susceptibility to excitotoxicity in HT22 neurons. Neural Regen. Res. 2013, 8, 1297–1306. [Google Scholar] [PubMed]
- Syapin, P.J. Regulation of haeme oxygenase-1 for treatment of neuroinflammation and brain disorders. Br. J. Pharmacol. 2008, 155, 623–640. [Google Scholar] [CrossRef] [PubMed]
- Sarady-Andrews, J.K.; Liu, F.; Gallo, D.; Nakao, A.; Overhaus, M.; Ollinger, R.; Choi, A.M.; Otterbein, L.E. Biliverdin administration protects against endotoxin-induced acute lung injury in rats. Am. J. Phys. Lung Cell Mol. Phys. 2005, 289, L1131–L1137. [Google Scholar] [CrossRef] [PubMed]
- Foresti, R.; Bains, S.K.; Pitchumony, T.S.; de Castro Bras, L.E.; Drago, F.; Dubois-Rande, J.L. Bucolo, C.; Motterlini, R. Small molecule activators of the Nrf2-HO-1 antioxidant axis modulate heme metabolism and inflammation in BV2 microglia cells. Pharm. Res. 2013, 76, 132–148. [Google Scholar] [CrossRef] [PubMed]
- Johnen, G.; Kaufman, S. Studies on the enzymatic and transcriptional activity of the dimerization cofactor for hepatocyte nuclear factor 1. Proc. Natl. Acad. Sci. USA 1997, 94, 13469–13474. [Google Scholar] [CrossRef] [PubMed]
- Itoh, K.; Mochizuki, M.; Ishii, Y.; Ishii, T.; Shibata, T.; Kawamoto, Y.; Kelly, V.; Sekizawa, K.; Uchida, K.; Yamamoto, M. Transcription factor Nrf2 regulates inflammation by mediating the effect of 15-deoxy-Δ(12,14)-prostaglandin J(2). Mol. Cell Biol. 2004, 24, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Wang, M.; Jing, X.; Shi, H.; Ren, M.; Lou, H. (−)-Epigallocatechin gallate protects against cerebral ischemia-induced oxidative stress via Nrf2/ARE signaling. Neurochem. Res. 2014, 39, 1292–1299. [Google Scholar] [CrossRef] [PubMed]
- Ishii, T.; Itoh, K.; Sato, H.; Bannai, S. Oxidative stress-inducible proteins in macrophages. Free Radic. Res. 1999, 31, 351–355. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the BJ-1201 is available from the authors.

© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, D.-S.; Nam, T.-G.; Jeong, B.-S.; Jeong, G.-S. RETRACTED: The Aminopyridinol Derivative BJ-1201 Protects Murine Hippocampal Cells against Glutamate-Induced Neurotoxicity via Heme Oxygenase-1. Molecules 2016, 21, 594. https://doi.org/10.3390/molecules21050594
Lee D-S, Nam T-G, Jeong B-S, Jeong G-S. RETRACTED: The Aminopyridinol Derivative BJ-1201 Protects Murine Hippocampal Cells against Glutamate-Induced Neurotoxicity via Heme Oxygenase-1. Molecules. 2016; 21(5):594. https://doi.org/10.3390/molecules21050594
Chicago/Turabian StyleLee, Dong-Sung, Tae-Gyu Nam, Byeong-Seon Jeong, and Gil-Saeng Jeong. 2016. "RETRACTED: The Aminopyridinol Derivative BJ-1201 Protects Murine Hippocampal Cells against Glutamate-Induced Neurotoxicity via Heme Oxygenase-1" Molecules 21, no. 5: 594. https://doi.org/10.3390/molecules21050594
APA StyleLee, D.-S., Nam, T.-G., Jeong, B.-S., & Jeong, G.-S. (2016). RETRACTED: The Aminopyridinol Derivative BJ-1201 Protects Murine Hippocampal Cells against Glutamate-Induced Neurotoxicity via Heme Oxygenase-1. Molecules, 21(5), 594. https://doi.org/10.3390/molecules21050594




