Regulation of Obesity and Metabolic Complications by Gamma and Delta Tocotrienols
Abstract
:1. Introduction
2. Rationale for the Anti-Obesity Property of T3s
3. Bioavailability and Accumulation of T3s
4. Effects of T3s on Obesity
4.1. Influences of T3s on Body Weight, Food Intake and Blood Profiles
4.2. Epidemiological Evidence; Observational and Clinical Trials
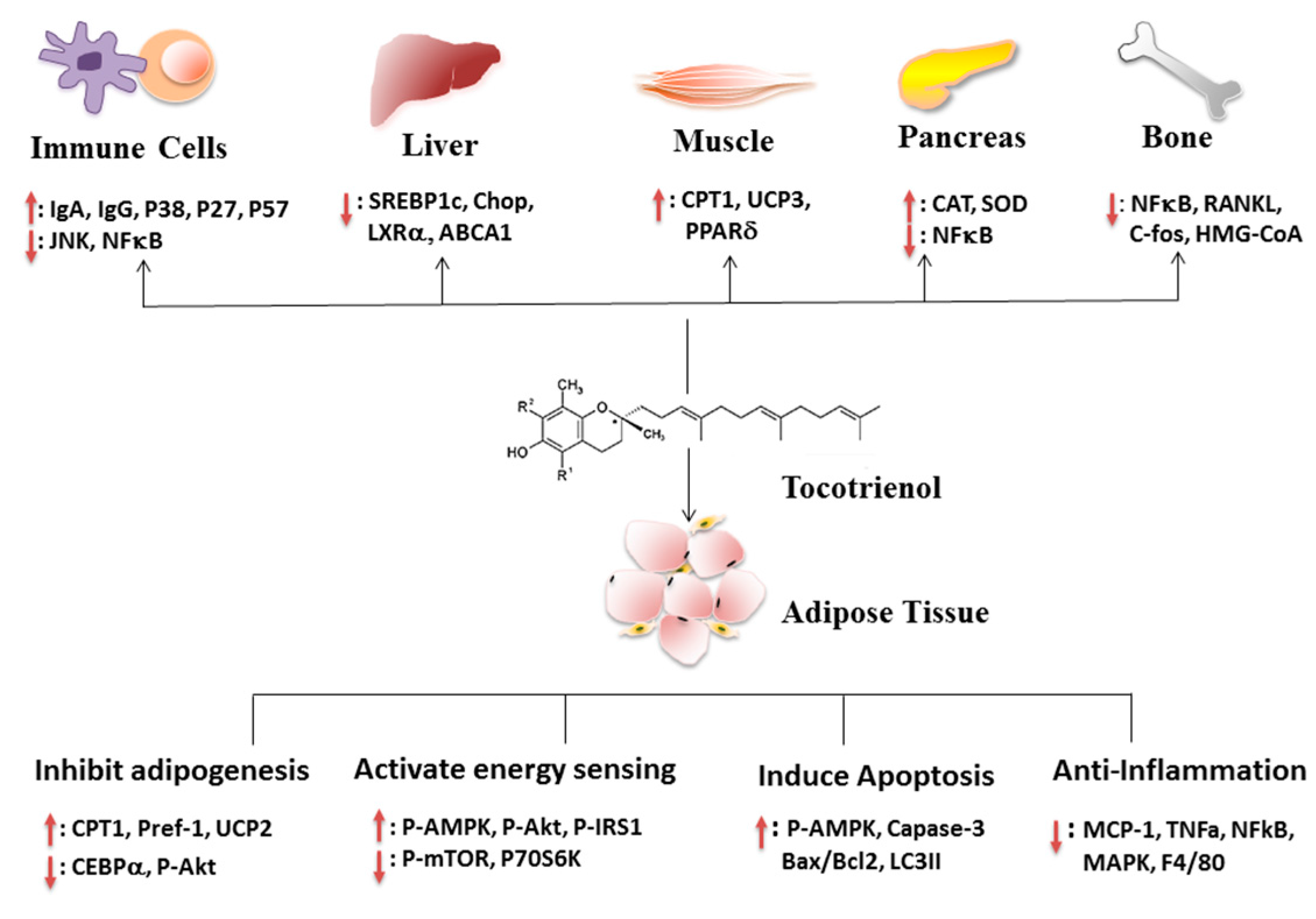
5. Molecular Mechanisms of the Anti-Obesity Effects of T3s
5.1. Effect of T3s on Adipose Tissue
5.1.1. Modulation of Fat Cell Adipogenesis
5.1.2. Modulation of Energy Sensing
5.1.3. Induction of Apoptosis in Preadipocytes
5.1.4. Modulation of Inflammation
5.2. Effects of T3s on Immune System
5.3. Effects of T3s on Liver
5.4. Effects of T3s on Other Tissues
6. Limitations and Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Freedman, D.S.; Horlick, M.; Berenson, G.S. A comparison of the Slaughter skinfold-thickness equations and BMI in predicting body fatness and cardiovascular disease risk factor levels in children. Am. J. Clin. Nutr. 2013, 98, 1417–1424. [Google Scholar] [CrossRef] [PubMed]
- Wohlfahrt-Veje, C.; Tinggaard, J.; Winther, K.; Mouritsen, A.; Hagen, C.P.; Mieritz, M.G.; de Renzy-Martin, K.T.; Boas, M.; Petersen, J.H.; Main, K.M. Body fat throughout childhood in 2647 healthy Danish children: Agreement of BMI, waist circumference, skinfolds with dual X-ray absorptiometry. Eur. J. Clin. Nutr. 2014, 68, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; van Dam, R.M.; Spiegelman, D.; Heymsfield, S.B.; Willett, W.C.; Hu, F.B. Comparison of dual-energy X-ray absorptiometric and anthropometric measures of adiposity in relation to adiposity-related biologic factors. Am. J. Epidemiol. 2010, 172, 1442–1454. [Google Scholar] [CrossRef] [PubMed]
- Willett, K.; Jiang, R.; Lenart, E.; Spiegelman, D.; Willett, W. Comparison of bioelectrical impedance and BMI in predicting obesity-related medical conditions. Obesity 2006, 14, 480–490. [Google Scholar] [CrossRef] [PubMed]
- Ogden, C.; Carroll, M.; Fryar, C.; Flegal, K. Prevalence of obesity among adults and youth: United States, 2011–2014. NCHS Data Brief 2015, 219, 1–8. [Google Scholar] [PubMed]
- Wang, Y.C.; McPherson, K.; Marsh, T.; Gortmaker, S.L.; Brown, M. Health and economic burden of the projected obesity trends in the USA and the UK. Lancet 2011, 378, 815–825. [Google Scholar] [CrossRef]
- Sun, K.; Kusminski, C.M.; Scherer, P.E. Adipose tissue remodeling and obesity. J. Clin. Investig. 2011, 121, 2094–2101. [Google Scholar] [CrossRef] [PubMed]
- Flodmark, C.-E.; Marcus, C.; Britton, M. Interventions to prevent obesity in children and adolescents: A systematic literature review. Int. J. Obes. 2006, 30, 579–589. [Google Scholar] [CrossRef] [PubMed]
- Piironen, V.; Syvaoja, E.L.; Varo, P.; Salminen, K.; Koivistoinen, P. Tocopherols and tocotrienols in Finnish foods: Vegetables, fruits, and berries. J. Agric. Food Chem. 1986, 34, 742–746. [Google Scholar] [CrossRef]
- Aggarwal, B.B.; Sundaram, C.; Prasad, S.; Kannappan, R. Tocotrienols, the vitamin E of the 21st century: Its potential against cancer and other chronic diseases. Biochem. Pharmacol. 2010, 80, 1613–1631. [Google Scholar] [CrossRef] [PubMed]
- Singh, U.; Devaraj, S.; Jialal, I. Vitamin E, oxidative stress, and inflammation. Annu. Rev. Nutr. 2005, 25, 151–174. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, A.A.; Qureshi, N.; Wright, J.; Shen, Z.; Kramer, G.; Gapor, A.; Chong, Y.; DeWitt, G.; Ong, A.; Peterson, D. Lowering of serum cholesterol in hypercholesterolemic humans by tocotrienols (palmvitee). Am. J. Clin. Nutr. 1991, 53, 1021S–1026S. [Google Scholar] [PubMed]
- Manu, K.A.; Shanmugam, M.K.; Ramachandran, L.; Li, F.; Fong, C.W.; Kumar, A.P.; Tan, P.; Sethi, G. First evidence that γ-tocotrienol inhibits the growth of human gastric cancer and chemosensitizes it to capecitabine in a xenograft mouse model through the modulation of NF-κB pathway. Clin. Cancer Res. 2012, 18, 2220–2229. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Xiao, H.; Jin, H.; Koo, P.T.; Tsang, D.J.; Yang, C.S. Synergistic actions of atorvastatin with γ-tocotrienol and celecoxib against human colon cancer HT29 and HCT116 cells. Int. J. Cancer 2010, 126, 852–863. [Google Scholar] [CrossRef] [PubMed]
- Fang, F.; Kang, Z.; Wong, C. Vitamin E tocotrienols improve insulin sensitivity through activating peroxisome proliferator-activated receptors. Mol. Nutr. Food Res. 2010, 54, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Ima-Nirwana, S.; Suhaniza, S. Effects of tocopherols and tocotrienols on body composition and bone calcium content in adrenalectomized rats replaced with dexamethasone. J. Med. food 2004, 7, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Magosso, E.; Ansari, M.A.; Gopalan, Y.; Shuaib, I.L.; Wong, J.-W.; Khan, N.A.K.; Bakar, M.R.A.; Ng, B.-H.; Yuen, K.-H. Tocotrienols for normalisation of hepatic echogenic response in nonalcoholic fatty liver: A randomised placebo-controlled clinical trial. Nutr. J. 2013, 12. [Google Scholar] [CrossRef] [PubMed]
- Weng-Yew, W.; Brown, L. Nutrapharmacology of tocotrienols for metabolic syndrome. Curr. Pharm. Des. 2011, 17, 2206–2214. [Google Scholar] [CrossRef] [PubMed]
- Peh, H.Y.; Tan, W.S.; Liao, W.; Wong, W.S. Vitamin E therapy beyond cancer: Tocopherol versus tocotrienol. Pharmacol. Ther. 2015. [Google Scholar] [CrossRef] [PubMed]
- Ayoub, N.; Bachawal, S.; Sylvester, P. γ-Tocotrienol inhibits HGF-dependent mitogenesis and Met activation in highly malignant mammary tumour cells. Cell Prolif. 2011, 44, 516–526. [Google Scholar] [CrossRef] [PubMed]
- Campbell, S.E.; Rudder, B.; Phillips, R.B.; Whaley, S.G.; Stimmel, J.B.; Leesnitzer, L.M.; Lightner, J.; Dessus-Babus, S.; Duffourc, M.; Stone, W.L. γ-Tocotrienol induces growth arrest through a novel pathway with TGFβ2 in prostate cancer. Free Radic. Biol. Med. 2011, 50, 1344–1354. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.; Wang, Z.; Geamanu, A.; Sarkar, F.H.; Gupta, S.V. Inhibition of cell growth and induction of apoptosis in non-small cell lung cancer cells by delta-tocotrienol is associated with notch-1 down-regulation. J. Cell. Biochem. 2011, 112, 2773–2783. [Google Scholar] [CrossRef] [PubMed]
- Patacsil, D.; Tran, A.T.; Cho, Y.S.; Suy, S.; Saenz, F.; Malyukova, I.; Ressom, H.; Collins, S.P.; Clarke, R.; Kumar, D. γ-Tocotrienol induced apoptosis is associated with unfolded protein response in human breast cancer cells. J. Nutr. Biochem. 2012, 23, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Shin-Kang, S.; Ramsauer, V.P.; Lightner, J.; Chakraborty, K.; Stone, W.; Campbell, S.; Reddy, S.A.; Krishnan, K. Tocotrienols inhibit AKT and ERK activation and suppress pancreatic cancer cell proliferation by suppressing the ErbB2 pathway. Free Radic. Biol. Med. 2011, 51, 1164–1174. [Google Scholar] [CrossRef] [PubMed]
- Wilankar, C.; Khan, N.M.; Checker, R.; Sharma, D.; Patwardhan, R.; Gota, V.; Sandur, S.K.; Devasagayam, T.P. γ-Tocotrienol induces apoptosis in human T cell lymphoma through activation of both intrinsic and extrinsic pathways. Curr. Pharm. Des. 2011, 17, 2176–2189. [Google Scholar] [CrossRef] [PubMed]
- Wilankar, C.; Sharma, D.; Checker, R.; Khan, N.M.; Patwardhan, R.; Patil, A.; Sandur, S.K.; Devasagayam, T.P. Role of immunoregulatory transcription factors in differential immunomodulatory effects of tocotrienols. Free Radic. Biol. Med. 2011, 51, 129–143. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, N.; Khalid, B.; Luke, D.; Ima Nirwana, S. Tocotrienol offers better protection than tocopherol from free radical-induced damage of rat bone. Clin. Exp. Pharmacol. Physiol. 2005, 32, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.R.; Siddiqui, S.; Parveen, K.; Javed, S.; Diwakar, S.; Siddiqui, W.A. Nephroprotective action of tocotrienol-rich fraction (TRF) from palm oil against potassium dichromate (K2Cr2O7)-induced acute renal injury in rats. Chem. Biol. Interact. 2010, 186, 228–238. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Khan, M.; Siddiqui, M.; Arif, J. An in vivo and in silico approach to elucidate the Tocotrienol-mediated fortification against infection and inflammation induced alterations in antioxidant defense system. Eur. Rev. Med. Pharmacol. Sci. 2011, 15, 916–930. [Google Scholar] [PubMed]
- Maniam, S.; Mohamed, N.; Shuid, A.N.; Soelaiman, I.N. Palm tocotrienol exerted better antioxidant activities in bone than α-tocopherol. Basic Clin. Pharmacol. Toxicol. 2008, 103, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Taridi, N.; Yahaya, M.F.; Teoh, S.L.; Latiff, A.A.; Ngah, W.; Das, S.; Mazlan, M. Tocotrienol rich fraction (TRF) supplementation protects against oxidative DNA damage and improves cognitive functions in Wistar rats. Clin. Ter. 2010, 162, 93–98. [Google Scholar]
- Vraka, P.S.; Drouza, C.; Rikkou, M.P.; Odysseos, A.D.; Keramidas, A.D. Synthesis and study of the cancer cell growth inhibitory properties of α-, γ-tocopheryl and γ-tocotrienyl 2-phenylselenyl succinates. Bioorg. Med. Chem. 2006, 14, 2684–2696. [Google Scholar] [CrossRef] [PubMed]
- Shrader, W.D.; Amagata, A.; Barnes, A.; Enns, G.M.; Hinman, A.; Jankowski, O.; Kheifets, V.; Komatsuzaki, R.; Lee, E.; Mollard, P. α-Tocotrienol quinone modulates oxidative stress response and the biochemistry of aging. Bioorg. Med. Chem. Lett. 2011, 21, 3693–3698. [Google Scholar] [CrossRef] [PubMed]
- Matringe, M.; Ksas, B.; Rey, P.; Havaux, M. Tocotrienols, the unsaturated forms of vitamin E, can function as antioxidants and lipid protectors in tobacco leaves. Plant Physiol. 2008, 147, 764–778. [Google Scholar] [CrossRef] [PubMed]
- Chin, S.-F.; Hamid, N.A.A.; Latiff, A.A.; Zakaria, Z.; Mazlan, M.; Yusof, Y.A.M.; Karim, A.A.; Ibahim, J.; Hamid, Z.; Ngah, W.Z.W. Reduction of DNA damage in older healthy adults by Tri E® Tocotrienol supplementation. Nutrition 2008, 24, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Schwedhelm, E.; Maas, R.; Troost, R.; Böger, R.H. Clinical pharmacokinetics of antioxidants and their impact on systemic oxidative stress. Clin. Pharmacokinet. 2003, 42, 437–459. [Google Scholar] [CrossRef] [PubMed]
- Prasad, K. Tocotrienols and cardiovascular health. Curr. Pharm. Des. 2011, 17, 2147–2154. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Tan, W.; Kang, Z.; Wong, C.W. Tocotrienol enriched palm oil prevents atherosclerosis through modulating the activities of peroxisome proliferators-activated receptors. Atherosclerosis 2010, 211, 278–282. [Google Scholar] [CrossRef] [PubMed]
- Nafeeza, M.; Norzana, A.; Jalaluddin, H.; Gapor, M. The effects of a tocotrienol-rich fraction on experimentally induced atherosclerosis in the aorta of rabbits. Malays. J. Pathol. 2001, 23, 17–25. [Google Scholar] [PubMed]
- Baliarsingh, S.; Beg, Z.H.; Ahmad, J. The therapeutic impacts of tocotrienols in type 2 diabetic patients with hyperlipidemia. Atherosclerosis 2005, 182, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Black, T.M.; Wang, P.; Maeda, N.; Coleman, R.A. Palm tocotrienols protect ApoE +/− mice from diet-induced atheroma formation. J. Nutr. 2000, 130, 2420–2426. [Google Scholar] [PubMed]
- Kannappan, R.; Gupta, S.C.; Kim, J.H.; Aggarwal, B.B. Tocotrienols fight cancer by targeting multiple cell signaling pathways. Genes Nutr. 2012, 7, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.-C.; Chung, Y.-F.; Yeh, Y.-T.; Chaung, H.-C.; Kuo, F.-C.; Fu, O.-Y.; Chen, H.-Y.; Hou, M.-F.; Yuan, S.-S.F. Serum adiponectin and leptin levels in Taiwanese breast cancer patients. Cancer Lett. 2006, 237, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Mantzoros, C.; Petridou, E.; Dessypris, N.; Chavelas, C.; Dalamaga, M.; Alexe, D.M.; Papadiamantis, Y.; Markopoulos, C.; Spanos, E.; Chrousos, G. Adiponectin and breast cancer risk. J. Clin. Endocrinol. Metab. 2004, 89, 1102–1107. [Google Scholar] [CrossRef] [PubMed]
- Malaviya, A.; Parajuli, P.; Sylvester, P.W. Anticancer effects of combined γ-tocotrienol and PPARγ antagonist treatment are associated with a suppression in adipogenic factor expression. J. Pharm. Nutr. Sci. 2014, 4, 43–56. [Google Scholar]
- Yap, S.; Yuen, K.; Wong, J. Pharmacokinetics and bioavailability of α-, γ- and δ-tocotrienols under different food status. J. Pharm. Pharmacol. 2001, 53, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Kang, I.; Fang, X.; Wang, W.; Lee, M.; Hollins, R.; Marshall, M.; Chung, S. γ-Tocotrienol attenuates high-fat diet-induced obesity and insulin resistance by inhibiting adipose inflammation and M1 macrophage recruitment. Int. J. Obes. 2015, 39, 438–446. [Google Scholar] [CrossRef] [PubMed]
- Yap, S.P.; Yuen, K.H.; Lim, A.B. Influence of route of administration on the absorption and disposition of α-, γ- and δ-tocotrienols in rats. J. Pharm. Pharmacol. 2003, 55, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Sontag, T.J.; Parker, R.S. Influence of major structural features of tocopherols and tocotrienols on their ω-oxidation by tocopherol-ω-hydroxylase. J. Lipid Res. 2007, 48, 1090–1098. [Google Scholar] [CrossRef] [PubMed]
- Brigelius-Flohé, R. Vitamin E and drug metabolism. Biochem. Biophys. Res. Commun. 2003, 305, 737–740. [Google Scholar] [CrossRef]
- Fu, J.-Y.; Che, H.-L.; Tan, D.M.-Y.; Teng, K.-T. Bioavailability of tocotrienols: Evidence in human studies. Nutr. Metab. 2014, 11, 1. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, A.A.; Khan, D.A.; Saleem, S.; Silswal, N.; Trias, A.M.; Tan, B.; Qureshi, N. Pharmacokinetics and bioavailability of annatto d-tocotrienol in healthy fed subjects. J. Clin. Exp. Cardiol. 2015, 6. [Google Scholar] [CrossRef]
- Meganathan, P.; Jabir, R.S.; Fuang, H.G.; Bhoo-Pathy, N.; Choudhury, R.B.; Taib, N.A.; Nesaretnam, K.; Chik, Z. A new formulation of Gamma Delta Tocotrienol has superior bioavailability compared to existing Tocotrienol-Rich Fraction in healthy human subjects. Sci. Rep. 2015, 5, 13550. [Google Scholar] [CrossRef] [PubMed]
- Patel, V.; Rink, C.; Gordillo, G.M.; Khanna, S.; Gnyawali, U.; Roy, S.; Shneker, B.; Ganesh, K.; Phillips, G.; More, J.L. Oral tocotrienols are transported to human tissues and delay the progression of the model for end-stage liver disease score in patients. J. Nutr. 2012, 142, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Shibata, A.; Nakagawa, K.; Shirakawa, H.; Kobayashi, T.; Kawakami, Y.; Takashima, R.; Ohashi, A.; Sato, S.; Ohsaki, Y.; Kimura, F. Physiological effects and tissue distribution from large doses of tocotrienol in rats. Biosci. Biotechnol. Biochem. 2012, 76, 1805–1808. [Google Scholar] [CrossRef] [PubMed]
- Uchida, T.; Nomura, S.; Sakuma, E.; Hanzawa, F.; Ikeda, S. α-Tocopherol does not accelerate depletion of γ-tocopherol and tocotrienol or excretion of their metabolites in rats. Lipids 2013, 48, 687–695. [Google Scholar] [CrossRef] [PubMed]
- Burdeos, G.C.; Nakagawa, K.; Kimura, F.; Miyazawa, T. Tocotrienol attenuates triglyceride accumulation in HepG2 cells and F344 rats. Lipids 2012, 47, 471–481. [Google Scholar] [CrossRef] [PubMed]
- Wong, W.Y.; Ward, L.C.; Fong, C.W.; Yap, W.N.; Brown, L. Anti-inflammatory γ- and δ-tocotrienols improve cardiovascular, liver and metabolic function in diet-induced obese rats. Eur. J. Nutr. 2015. [Google Scholar] [CrossRef] [PubMed]
- Wong, W.-Y.; Poudyal, H.; Ward, L.C.; Brown, L. Tocotrienols reverse cardiovascular, metabolic and liver changes in high carbohydrate, high fat diet-fed rats. Nutrients 2012, 4, 1527–1541. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.S.; Radhakrishnan, A.K.; Ibrahim, T.A.; Cheong, S.K. δ- and γ-Tocotrienols induce classical ultrastructural apoptotic changes in human T lymphoblastic leukemic cells. Microsc. Microanal. 2012, 18, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Candiracci, M.; Justo, M.L.; Castano, A.; Rodriguez-Rodriguez, R.; Herrera, M.D. Rice bran enzymatic extract-supplemented diets modulate adipose tissue inflammation markers in Zucker rats. Nutrition 2014, 30, 466–472. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Mukherjee, S.; Lekli, I.; Gurusamy, N.; Bardhan, J.; Raychoudhury, U.; Chakravarty, R.; Banerji, S.; Knowlton, A.A.; Das, D.K. Tocotrienols confer resistance to ischemia in hypercholesterolemic hearts: Insight with genomics. Mol. Cell. Biochem. 2012, 360, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Watkins, T.; Lenz, P.; Gapor, A.; Struck, M.; Tomeo, A.; Bierenbaum, M. γ-Tocotrienol as a hypocholesterolemic and antioxidant agent in rats fed atherogenic diets. Lipids 1993, 28, 1113–1118. [Google Scholar] [CrossRef] [PubMed]
- Chou, T.W.; Ma, C.Y.; Cheng, H.H.; Chen, Y.Y.; Lai, M.H. A rice bran oil diet improves lipid abnormalities and suppress hyperinsulinemic responses in rats with streptozotocin/nicotinamide-induced type 2 diabetes. J. Clin. Biochem. Nutr. 2009, 45, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, A.A.; Qureshi, N.; Hasler-Rapacz, J.O.; Weber, F.E.; Chaudhary, V.; Crenshaw, T.D.; Gapor, A.; Ong, A.S.; Chong, Y.H.; Peterson, D.; et al. Dietary tocotrienols reduce concentrations of plasma cholesterol, apolipoprotein B, thromboxane B2, and platelet factor 4 in pigs with inherited hyperlipidemias. Am. J. Clin. Nutr. 1991, 53, 1042S–1046S. [Google Scholar]
- Budin, S.B.; Othman, F.; Louis, S.R.; Bakar, M.A.; Das, S.; Mohamed, J. The effects of palm oil tocotrienol-rich fraction supplementation on biochemical parameters, oxidative stress and the vascular wall of streptozotocin-induced diabetic rats. Clinics 2009, 64, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, A.A.; Bradlow, B.A.; Brace, L.; Manganello, J.; Peterson, D.M.; Pearce, B.C.; Wright, J.J.; Gapor, A.; Elson, C.E. Response of hypercholesterolemic subjects to administration of tocotrienols. Lipids 1995, 30, 1171–1177. [Google Scholar] [CrossRef] [PubMed]
- Zaiden, N.; Yap, W.N.; Ong, S.; Xu, C.H.; Teo, V.H.; Chang, C.P.; Zhang, X.W.; Nesaretnam, K.; Shiba, S.; Yap, Y.L. Gamma delta tocotrienols reduce hepatic triglyceride synthesis and VLDL secretion. J. Atheroscler. Thromb. 2010, 17, 1019–1032. [Google Scholar] [CrossRef] [PubMed]
- Heng, E.C.; Karsani, S.A.; Abdul Rahman, M.; Abdul Hamid, N.A.; Hamid, Z.; Wan Ngah, W.Z. Supplementation with tocotrienol-rich fraction alters the plasma levels of Apolipoprotein A-I precursor, Apolipoprotein E precursor, and C-reactive protein precursor from young and old individuals. Eur. J. Nutr. 2013, 52, 1811–1820. [Google Scholar] [CrossRef] [PubMed]
- Haghighat, N.; Vafa, M.; Eghtesadi, S.; Heidari, I.; Hosseini, A.; Rostami, A. The effects of tocotrienols added to canola oil on microalbuminuria, inflammation, and nitrosative stress in patients with type 2 diabetes: A randomized, double-blind, placebo-controlled trial. Int. J. Prev. Med. 2014, 5, 617–623. [Google Scholar] [PubMed]
- Irandoost, P.; Ebrahimi-Mameghani, M.; Pirouzpanah, S. Does grape seed oil improve inflammation and insulin resistance in overweight or obese women? Int. J. Food Sci. Nutr. 2013, 64, 706–710. [Google Scholar] [CrossRef] [PubMed]
- Uto-Kondo, H.; Ohmori, R.; Kiyose, C.; Kishimoto, Y.; Saito, H.; Igarashi, O.; Kondo, K. Tocotrienol suppresses adipocyte differentiation and Akt phosphorylation in 3T3-L1 preadipocytes. J. Nutr. 2009, 139, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Burdeos, G.C.; Nakagawa, K.; Abe, T.; Kimura, F.; Miyazawa, T. Tocotrienol modulates crucial lipid metabolism-related genes in differentiated 3T3-L1 preadipocytes. Food Funct. 2014, 5, 2221–2227. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.J.; Huang, G.Y.; Ng, L.T. γ-Tocotrienol induced cell cycle arrest and apoptosis via activating the Bax-mediated mitochondrial and AMPK signaling pathways in 3T3-L1 adipocytes. Food Chem. Toxicol. 2013, 59, 501–513. [Google Scholar] [CrossRef] [PubMed]
- Zuk, P.A.; Zhu, M.; Ashjian, P.; de Ugarte, D.A.; Huang, J.I.; Mizuno, H.; Alfonso, Z.C.; Fraser, J.K.; Benhaim, P.; Hedrick, M.H. Human adipose tissue is a source of multipotent stem cells. Mol. Biol. Cell 2002, 13, 4279–4295. [Google Scholar] [CrossRef] [PubMed]
- Gimble, J.M.; Katz, A.J.; Bunnell, B.A. Adipose-derived stem cells for regenerative medicine. Circ. Res. 2007, 100, 1249–1260. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Ha, J.H.; Okla, M.; Chung, S. Activation of autophagy and AMPK by γ-tocotrienol suppresses the adipogenesis in human adipose derived stem cells. Mol. Nutr. Food Res. 2014, 58, 569–579. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.W. Possible anti-obesity therapeutics from nature—A review. Phytochemistry 2010, 71, 1625–1641. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Y.; Lee, M.H.; Hsu, C.C.; Wei, C.L.; Tsai, Y.C. Methyl cinnamate inhibits adipocyte differentiation via activation of the CaMKK2-AMPK pathway in 3T3-L1 preadipocytes. J. Agric. Food Chem. 2012, 60, 955–963. [Google Scholar] [CrossRef] [PubMed]
- Ono, M.; Fujimori, K. Antiadipogenic effect of dietary apigenin through activation of AMPK in 3T3-L1 cells. J. Agric. Food Chem. 2011, 59, 13346–13352. [Google Scholar] [CrossRef] [PubMed]
- Dave, S.; Kaur, N.J.; Nanduri, R.; Dkhar, H.K.; Kumar, A.; Gupta, P. Inhibition of adipogenesis and induction of apoptosis and lipolysis by stem bromelain in 3T3-L1 adipocytes. PLoS ONE 2012, 7, e30831. [Google Scholar] [CrossRef] [PubMed]
- Baur, J.A.; Pearson, K.J.; Price, N.L.; Jamieson, H.A.; Lerin, C.; Kalra, A.; Prabhu, V.V.; Allard, J.S.; Lopez-Lluch, G.; Lewis, K.; et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature 2006, 444, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Laplante, M.; Sabatini, D.M. An emerging role of mTOR in lipid biosynthesis. Curr. Biol. 2009, 19, R1046–R1052. [Google Scholar] [CrossRef] [PubMed]
- Hresko, R.C.; Mueckler, M. mTOR·RICTOR is the Ser473 kinase for Akt/protein kinase B in 3T3-L1 adipocytes. J. Biol. Chem. 2005, 280, 40406–40416. [Google Scholar] [CrossRef] [PubMed]
- Roh, C.; Han, J.; Tzatsos, A.; Kandror, K.V. Nutrient-sensing mTOR-mediated pathway regulates leptin production in isolated rat adipocytes. Am. J. Physiol. Endocrinol. Metab. 2003, 284, E322–E330. [Google Scholar] [CrossRef] [PubMed]
- Li, X.H.; Fu, D.; Latif, N.H.; Mullaney, C.P.; Ney, P.H.; Mog, S.R.; Whitnall, M.H.; Srinivasan, V.; Xiao, M. δ-Tocotrienol protects mouse and human hematopoietic progenitors from γ-irradiation through extracellular signal-regulated kinase/mammalian target of rapamycin signaling. Haematologica 2010, 95, 1996–2004. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.J.; Sylvester, P.W. δ-Tocotrienol inhibits neoplastic mammary epithelial cell proliferation by decreasing Akt and nuclear factor kappaB activity. Exp. Biol. Med. 2005, 230, 235–241. [Google Scholar]
- Kim, J.Y.; van de Wall, E.; Laplante, M.; Azzara, A.; Trujillo, M.E.; Hofmann, S.M.; Schraw, T.; Durand, J.L.; Li, H.; Li, G.; et al. Obesity-associated improvements in metabolic profile through expansion of adipose tissue. J. Clin. Investig. 2007, 117, 2621–2637. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Simmons-Menchaca, M.; Gapor, A.; Sanders, B.G.; Kline, K. Induction of apoptosis in human breast cancer cells by tocopherols and tocotrienols. Nutr. Cancer 1999, 33, 26–32. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, B.S.; Briski, K.P.; Gapor, A.; Sylvester, P.W. Antiproliferative and apoptotic effects of tocopherols and tocotrienols on preneoplastic and neoplastic mouse mammary epithelial cells. Proc. Soc. Exp. Biol. Med. 2000, 224, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, J.K.; Gupta, S. Tocotrienol-rich fraction of palm oil induces cell cycle arrest and apoptosis selectively in human prostate cancer cells. Biochem. Biophys. Res. Commun. 2006, 346, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Ahn, K.S.; Sethi, G.; Krishnan, K.; Aggarwal, B.B. γ-Tocotrienol inhibits nuclear factor-κB signaling pathway through inhibition of receptor-interacting protein and TAK1 leading to suppression of antiapoptotic gene products and potentiation of apoptosis. J. Biol. Chem. 2007, 282, 809–820. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, A.S.; Obin, M.S. Obesity and the role of adipose tissue in inflammation and metabolism. Am. J. Clin. Nutr. 2006, 83, 461S–465S. [Google Scholar]
- Wisse, B.E. The inflammatory syndrome: The role of adipose tissue cytokines in metabolic disorders linked to obesity. J. Am. Soc. Nephrol. 2004, 15, 2792–2800. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, A.A.; Reis, J.C.; Papasian, C.J.; Morrison, D.C.; Qureshi, N. Tocotrienols inhibit lipopolysaccharide-induced pro-inflammatory cytokines in macrophages of female mice. Lipids Health Dis. 2010, 9, 143. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, T.; Shoji, A.; Gu, N.; Joo, E.; Li, S.; Adachi, T.; Yamazaki, H.; Yasuda, K.; Kondoh, T.; Tsuda, K. γ-Tocotrienol attenuates TNF-α-induced changes in secretion and gene expression of MCP-1, IL-6 and adiponectin in 3T3-L1 adipocytes. Mol. Med. Rep. 2012, 5, 905–909. [Google Scholar] [PubMed]
- Kaileh, M.; Sen, R. Role of NF-κB in the anti-inflammatory effects of tocotrienols. J. Am. Coll. Nutr. 2010, 29, 334S–339S. [Google Scholar] [CrossRef]
- Justo, M.L.; Candiracci, M.; Dantas, A.P.; de Sotomayor, M.A.; Parrado, J.; Vila, E.; Herrera, M.D.; Rodriguez-Rodriguez, R. Rice bran enzymatic extract restores endothelial function and vascular contractility in obese rats by reducing vascular inflammation and oxidative stress. J. Nutr. Biochem. 2013, 24, 1453–1461. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q. Natural forms of vitamin E: Metabolism, antioxidant, and anti-inflammatory activities and their role in disease prevention and therapy. Free Radic. Biol. Med. 2014, 72, 76–90. [Google Scholar] [CrossRef] [PubMed]
- Yam, M.L.; Abdul Hafid, S.R.; Cheng, H.M.; Nesaretnam, K. Tocotrienols suppress proinflammatory markers and cyclooxygenase-2 expression in RAW264.7 macrophages. Lipids 2009, 44, 787–797. [Google Scholar] [CrossRef] [PubMed]
- Biron, C.A.; Nguyen, K.B.; Pien, G.C.; Cousens, L.P.; Salazar-Mather, T.P. Natural killer cells in antiviral defense: Function and regulation by innate cytokines. Annu. Rev. Immunol. 1999, 17, 189–220. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.Y.; Wakizono, Y.; Sunada, Y.; Hung, P.; Nonaka, M.; Sugano, M.; Yamada, K. Dietary effect of tocopherols and tocotrienols on the immune function of spleen and mesenteric lymph node lymphocytes in Brown Norway rats. Biosci. Biotechnol. Biochem. 1999, 63, 1697–1702. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; Pae, M.; Dao, M.C.; Smith, D.; Meydani, S.N.; Wu, D. Dietary supplementation with tocotrienols enhances immune function in C57BL/6 mice. J. Nutr. 2010, 140, 1335–1341. [Google Scholar] [CrossRef] [PubMed]
- Munteanu, A.; Ricciarelli, R.; Massone, S.; Zingg, J.M. Modulation of proteasome activity by vitamin E in THP-1 monocytes. IUBMB Life 2007, 59, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Weisberg, S.P.; McCann, D.; Desai, M.; Rosenbaum, M.; Leibel, R.L.; Ferrante, A.W., Jr. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Investig. 2003, 112, 1796–1808. [Google Scholar] [CrossRef] [PubMed]
- Theriault, A.; Chao, J.T.; Gapor, A. Tocotrienol is the most effective vitamin E for reducing endothelial expression of adhesion molecules and adhesion to monocytes. Atherosclerosis 2002, 160, 21–30. [Google Scholar] [CrossRef]
- Wu, S.J.; Liu, P.L.; Ng, L.T. Tocotrienol-rich fraction of palm oil exhibits anti-inflammatory property by suppressing the expression of inflammatory mediators in human monocytic cells. Mol. Nutr. Food Res. 2008, 52, 921–929. [Google Scholar] [CrossRef] [PubMed]
- Chao, J.T.; Gapor, A.; Theriault, A. Inhibitory effect of delta-tocotrienol, a HMG CoA reductase inhibitor, on monocyte-endothelial cell adhesion. J. Nutr. Sci. Vitaminol. 2002, 48, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Jiang, Q. γ-Tocotrienol inhibits lipopolysaccharide-induced interlukin-6 and granulocyte colony-stimulating factor by suppressing C/EBPβ and NF-κB in macrophages. J. Nutr. Biochem. 2013, 24, 1146–1152. [Google Scholar] [CrossRef] [PubMed]
- Tres, A.; Nuchi, C.D.; Magrinya, N.; Guardiola, F.; Bou, R.; Codony, R. Use of palm-oil by-products in chicken and rabbit feeds: Effect on the fatty acid and tocol composition of meat, liver and plasma. Animal 2012, 6, 1005–1017. [Google Scholar] [CrossRef] [PubMed]
- Yachi, R.; Igarashi, O.; Kiyose, C. Protective effects of vitamin E analogs against carbon tetrachloride-induced fatty liver in rats. J. Clin. Biochem. Nutr. 2010, 47, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Muto, C.; Yachi, R.; Aoki, Y.; Koike, T.; Igarashi, O.; Kiyose, C. γ-Tocotrienol reduces the triacylglycerol level in rat primary hepatocytes through regulation of fatty acid metabolism. J. Clin. Biochem. Nutr. 2013, 52, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Burdeos, G.C.; Nakagawa, K.; Watanabe, A.; Kimura, F.; Miyazawa, T. γ-Tocotrienol attenuates triglyceride through effect on lipogenic gene expressions in mouse hepatocellular carcinoma Hepa 1–6. J. Nutr. Sci. Vitaminol. 2013, 59, 148–151. [Google Scholar] [CrossRef] [PubMed]
- Ebong, P.E.; Owu, D.U.; Isong, E.U. Influence of palm oil (Elaesis guineensis) on health. Plant Foods Hum. Nutr. 1999, 53, 209–222. [Google Scholar] [CrossRef] [PubMed]
- Budin, S.B.; Han, C.M.; Jayusman, P.A.; Taib, I.S. Tocotrienol rich fraction prevents fenitrothion induced pancreatic damage by restoring antioxidant status. Pak. J. Biol. Sci. 2012, 15, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Kunnumakkara, A.B.; Sung, B.; Ravindran, J.; Diagaradjane, P.; Deorukhkar, A.; Dey, S.; Koca, C.; Yadav, V.R.; Tong, Z.; Gelovani, J.G.; Guha, S.; Krishnan, S.; Aggarwal, B.B. γ-Tocotrienol inhibits pancreatic tumors and sensitizes them to gemcitabine treatment by modulating the inflammatory microenvironment. Cancer Res. 2010, 70, 8695–8705. [Google Scholar] [CrossRef] [PubMed]
- Janicka, A.; Wren, T.A.; Sanchez, M.M.; Dorey, F.; Kim, P.S.; Mittelman, S.D.; Gilsanz, V. Fat mass is not beneficial to bone in adolescents and young adults. J. Clin. Endocrinol. Metab. 2007, 92, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.J.; Liu, Y.J.; Liu, P.Y.; Hamilton, J.; Recker, R.R.; Deng, H.W. Relationship of obesity with osteoporosis. J. Clin. Endocrinol. Metab. 2007, 92, 1640–1646. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.J. Effects of obesity on bone metabolism. J. Orthop. Surg. Res. 2011, 6, 30. [Google Scholar] [CrossRef] [PubMed]
- Nazrun, A.S.; Norazlina, M.; Norliza, M.; Nirwana, S.I. The anti-inflammatory role of vitamin E in prevention of osteoporosis. Adv. Pharmacol. Sci. 2012, 2012, 142702. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Tandon, V.R.; Mahajan, S.; Mahajan, V.; Mahajan, A. Obesity: Friend or foe for osteoporosis. J. Midlife Health 2014, 5, 6–9. [Google Scholar] [CrossRef] [PubMed]
- Colaianni, G.; Brunetti, G.; Faienza, M.F.; Colucci, S.; Grano, M. Osteoporosis and obesity: Role of Wnt pathway in human and murine models. World J. Orthop. 2014, 5, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Rosen, C.J.; Bouxsein, M.L. Mechanisms of disease: Is osteoporosis the obesity of bone? Nat. Clin. Pract. Rheumatol. 2006, 2, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Ilich, J.Z.; Kelly, O.J.; Kim, Y.; Spicer, M.T. Low-grade chronic inflammation perpetuated by modern diet as a promoter of obesity and osteoporosis. Arh. Hig. Rada Toksikol. 2014, 65, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Soelaiman, I.N.; Ming, W.; Abu Bakar, R.; Hashnan, N.A.; Mohd Ali, H.; Mohamed, N.; Muhammad, N.; Shuid, A.N. Palm tocotrienol supplementation enhanced bone formation in oestrogen-deficient rats. Int. J. Endocrinol. 2012. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, N.; Luke, D.A.; Shuid, A.N.; Mohamed, N.; Soelaiman, I.N. Two different isomers of vitamin E prevent bone loss in postmenopausal osteoporosis rat model. Evid. Based Complement. Altern. Med. 2012. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, N.; Luke, D.A.; Shuid, A.N.; Mohamed, N.; Soelaiman, I.N. Tocotrienol supplementation in postmenopausal osteoporosis: Evidence from a laboratory study. Clinics 2013, 68, 1338–1343. [Google Scholar] [CrossRef]
- Shuid, A.N.; Mehat, Z.; Mohamed, N.; Muhammad, N.; Soelaiman, I.N. Vitamin E exhibits bone anabolic actions in normal male rats. J. Bone Miner. Metab. 2010, 28, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Abd Manan, N.; Mohamed, N.; Shuid, A.N. Effects of low-dose versus high-dose γ-tocotrienol on the bone cells exposed to the hydrogen peroxide-induced oxidative stress and apoptosis. Evid. Based Complement. Altern. Med. 2012. [Google Scholar] [CrossRef] [PubMed]
- Ha, H.; Lee, J.H.; Kim, H.N.; Lee, Z.H. alpha-Tocotrienol inhibits osteoclastic bone resorption by suppressing RANKL expression and signaling and bone resorbing activity. Biochem. Biophys. Res. Commun. 2011, 406, 546–551. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Ding, Y.; Peng, Y.; Wu, Y.; Fan, J.; Li, W.; Yang, R.; Yang, M.; Fu, Q. γ-Tocotrienol protects against ovariectomy-induced bone loss via mevalonate pathway as HMG-CoA reductase inhibitor. Bone 2014, 67, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Wang, W.; Okla, M.; Kang, I.; Moreau, R.; Chung, S. Suppression of NLRP3 inflammasome by γ-tocotrienol ameliorates type 2 diabetes. J. Lipid Res. 2016, 57, 66–76. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Effects | Dose/Isomers/Duration | Models | Ref. |
|---|---|---|---|---|
| Body weight | ― | 120 mg/kg/TRF 1/8 weeks | Wistar Rat | [59] |
| ― | 60 mg/kg/γT3 2/8 weeks | SD Rat | [16] | |
| ↓ | 10 mg RBT 3/3 weeks | F334 Rat | [57] | |
| ↓ | 0.05% γT3 4 in diet/4 weeks | C57BL/6J | [47] | |
| ― | 85 mg/kg/αT3 5, or γT3 6 or δT3 7/8 weeks | Wistar Rat | [58] | |
| Food intake | ↑ | 120 mg/kg/TRF 1/8 weeks | Wistar Rat | [59] |
| ― | 0.05% γT3 4 in diet/4 weeks | C57BL/6J | [47] | |
| ― | 85 mg/kg/αT3 5, or γT3 6 or δT3 7/8 weeks | Wistar Rat | [58] | |
| Mesenteric fat | ↓ | 120 mg/kg/TRF 1/8 weeks | Wistar Rat | [59] |
| ↓ | 10 mg RBT 3/3 weeks | F334 Rat | [57] | |
| ↓ | 0.05% γT3 4 in diet/4 weeks | C57BL/6J | [47] | |
| Perirenal fat | ― | 120 mg/kg/TRF 1/8 weeks | Wistar Rat | [59] |
| Epididymal fat | ― | 120 mg/kg/TRF 1/8 weeks | Wistar Rat | [59] |
| ↓ | 10 mg RBT 3/3 weeks | F334 Rat | [57] | |
| ↓ | 0.05% γT3 4 in diet/4 weeks | C57BL/6J | [47] | |
| ― | 85 mg/kg/αT3 5, or γT3 6/8 weeks | Wistar Rat | [58] | |
| ↓ | 85 mg/kg/δT3 7/8 weeks | Wistar Rat | [58] | |
| Body fat | ↓ | 60 mg/kg/γT3 2/8 weeks | SD Rats | [16] |
| ↓ | 120 mg/kg/TRF 1/8 weeks | Wistar Rat | [59] | |
| ↓ | 5% rice bran extract 8/20 weeks | Zucker rice | [61] | |
| ― | 85 mg/kg/αT3 5, or γT3 6/8 weeks | Wistar Rat | [58] | |
| ↓ | 85 mg/kg/ δT3 7/8 weeks | Wistar Rat | [58] | |
| Serum or plasma total cholesterol | ― | 120 mg/kg/TRF 1/8 weeks | Wistar Rat | [59] |
| ↓ | 20 μM/kg/γT3 9/4 weeks | New Zealand rabbit | [62] | |
| ↓ | 0.09% γT3 10 in diet/5 weeks | Wistar Rat | [63] | |
| ↓ | 50 μg/g TRF 11 in diet/6 weeks | Wistar Rat; Cholesterolemic Pigs | [64,65] | |
| ― | 85 mg/kg/αT3 5, or γT3 6/8 weeks | Wistar Rat | [58] | |
| ↓ | 85 mg/kg/δT3 7/8 weeks | Wistar Rat | [58] | |
| Fasting plasma glucose | ↓ | 120 mg/kg/TRF 1/8 weeks | Wistar Rat | [59] |
| ↓ | 200 mg/kg BW/TRF 12/8 weeks | SD Rats | [66] | |
| ― | 85 mg/kg/αT3 5, or γT3 6/8 weeks | Wistar Rat | [58] | |
| ↓ | 85 mg/kg/δT37/8 weeks | Wistar Rat | [58] | |
| Plasma free fatty acid | ↓ | 120 mg/kg/TRF 1/8 weeks | Wistar Rat | [59] |
| ↓ | 0.09% γT3 9 in diet/5 weeks | Wistar Rat | [63] | |
| ― | 85 mg/kg/αT3 5 | Wistar Rat | [58] | |
| ↓ | 85 mg/kg/γT3 6 or δT3 7/8 weeks | Wistar Rat | [58] | |
| Plasma triglyceride | ↓ | 120 mg/kg/TRF 1/8 weeks | Wistar Rat | [59] |
| ↓ | 10 mg RBT 3/3 weeks | F334 Rat | [57] | |
| ― | 85 mg/kg/αT3 5, or γT3 6/8 weeks | Wistar Rat | [58] | |
| ↓ | 85 mg/kg/δT3 7/8 weeks | Wistar Rat | [58] |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, L.; Fang, X.; Marshall, M.R.; Chung, S. Regulation of Obesity and Metabolic Complications by Gamma and Delta Tocotrienols. Molecules 2016, 21, 344. https://doi.org/10.3390/molecules21030344
Zhao L, Fang X, Marshall MR, Chung S. Regulation of Obesity and Metabolic Complications by Gamma and Delta Tocotrienols. Molecules. 2016; 21(3):344. https://doi.org/10.3390/molecules21030344
Chicago/Turabian StyleZhao, Lu, Xiefan Fang, Maurice R. Marshall, and Soonkyu Chung. 2016. "Regulation of Obesity and Metabolic Complications by Gamma and Delta Tocotrienols" Molecules 21, no. 3: 344. https://doi.org/10.3390/molecules21030344
APA StyleZhao, L., Fang, X., Marshall, M. R., & Chung, S. (2016). Regulation of Obesity and Metabolic Complications by Gamma and Delta Tocotrienols. Molecules, 21(3), 344. https://doi.org/10.3390/molecules21030344






