Phenolic Melatonin-Related Compounds: Their Role as Chemical Protectors against Oxidative Stress
Abstract
:1. Introduction
2. Oxidative Stress and Free Radicals
3. Chemical Antioxidants
3.1. Mechanisms of Action
Main Reaction Mechanisms Involved in AOC Assays
3.2. Melatonin (MLT)
3.3. Phenolic Compounds
4. Phenolic Melatonin-Related Compounds
4.1. Location and Sources
4.2. Functions and Toxicity
4.3. Antioxidant Activity
4.3.1. 5-Hydroxytryptophan (5-HTP)
4.3.2. Serotonin (5-HT)
4.3.3. N-Acetylserotonin (NAS)
4.3.4. 6-Hydroxymelatonin (6-HMLT)
4.3.5. Computationally-Designed Molecules
4.3.6. Chemical Pathways
4.4. Comparisons with Other Antioxidants
5. Concluding Remarks
Acknowledgments
Conflicts of Interest
References
- Davies, K.J. Oxidative stress: The paradox of aerobic life. Biochem. Soc. Symp. 1995, 61, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Pham-Huy, L.A.; He, H.; Pham-Huy, C. Free radicals, antioxidants in disease and health. Int. J. Biomed. Sci. 2008, 4, 89–96. [Google Scholar] [PubMed]
- Pacher, P.; Beckman, J.S.; Liaudet, L. Nitric oxide and peroxynitrite in health and disease. Physiol. Rev. 2007, 87, 315–424. [Google Scholar] [CrossRef] [PubMed]
- Genestra, M. Oxyl radicals, redox-sensitive signalling cascades and antioxidants. Cell. Signal. 2007, 19, 1807–1819. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.D.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Rhodes, C.J.; Moncol, J.; Izakovic, M.; Mazur, M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem. Biol. Interact. 2006, 160, 1–40. [Google Scholar] [CrossRef] [PubMed]
- Droge, W. Free radicals in the physiological control of cell function. Physiol. Rev. 2002, 82, 47–95. [Google Scholar] [CrossRef] [PubMed]
- Young, I.S.; Woodside, J.V. Antioxidants in health and disease. J. Clin. Pathol. 2001, 54, 176–186. [Google Scholar] [PubMed]
- Halliwell, B. Biochemistry of oxidative stress. Biochem. Soc. Trans. 2007, 35, 1147–1150. [Google Scholar] [CrossRef] [PubMed]
- Willcox, J.K.; Ash, S.L.; Catignani, G.L. Antioxidants and prevention of chronic disease. Crit. Rev. Food Sci. Nutr. 2004, 44, 275–295. [Google Scholar] [CrossRef] [PubMed]
- Parthasarathy, S.; Santanam, N.; Ramachandran, S.; Meilhac, O. Oxidants and antioxidants in atherogenesis: An appraisal. J. Lipid Res. 1999, 40, 2143–2157. [Google Scholar] [PubMed]
- Lowe, F.J.; Cemeli, E. Biomarkers of oxidative stress and the relationship to cigarette smoking. Mini Rev. Org. Chem. 2011, 8, 377–386. [Google Scholar] [CrossRef]
- Valko, M.; Morris, H.; Cronin, M.T.D. Metals, toxicity and oxidative stress. Curr. Med. Chem. 2005, 12, 1161–1208. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Sainz, R.M.; Mayo, J.C.; Lopez-Burillo, S. Melatonin: Reducing the toxicity and increasing the efficacy of drugs. J. Pharm. Pharmacol. 2002, 54, 1299–1321. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Manchester, L.C.; Tan, D.X. Neurotoxins: Free radical mechanisms and melatonin protection. Curr. Neuropharmacol. 2010, 8, 194–210. [Google Scholar] [CrossRef] [PubMed]
- Thanan, R.; Oikawa, S.; Hiraku, Y.; Ohnishi, S.; Ma, N.; Pinlaor, S.; Yongvanit, P.; Kawanishi, S.; Murata, M. Oxidative stress and its significant roles in neurodegenerative diseases and cancer. Int. J. Mol. Sci. 2014, 16, 193–217. [Google Scholar] [CrossRef] [PubMed]
- Filaire, E.; Dupuis, C.; Galvaing, G.; Aubreton, S.; Laurent, H.; Richard, R.; Filaire, M. Lung cancer: What are the links with oxidative stress, physical activity and nutrition. Lung Cancer 2013, 82, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Paschos, A.; Pandya, R.; Duivenvoorden, W.C.M.; Pinthus, J.H. Oxidative stress in prostate cancer: Changing research concepts towards a novel paradigm for prevention and therapeutics. Prostate Cancer Prostatic Dis. 2013, 16, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Siti, H.N.; Kamisah, Y.; Kamsiah, J. The role of oxidative stress, antioxidants and vascular inflammation in cardiovascular disease (a review). Vasc. Pharmacol. 2015, 71, 40–56. [Google Scholar] [CrossRef] [PubMed]
- Popolo, A.; Autore, G.; Pinto, A.; Marzocco, S. Oxidative stress in patients with cardiovascular disease and chronic renal failure. Free Radic. Res. 2013, 47, 346–356. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.R.; Shaw, C.A.; Langrish, J.P. From particles to patients: Oxidative stress and the cardiovascular effects of air pollution. Future Cardiol. 2012, 8, 577–602. [Google Scholar] [CrossRef] [PubMed]
- Blesa, J.; Trigo-Damas, I.; Quiroga-Varela, A.; Jackson-Lewis, V.R. Oxidative stress and Parkinson’s disease. Front. Neuroanat. 2015, 9, 91. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.; Nehru, B. Characterization of the lipopolysaccharide induced model of Parkinson’s disease: Role of oxidative stress and neuroinflammation. Neurochem. Int. 2015, 87, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Gaki, G.S.; Papavassiliou, A.G. Oxidative stress-induced signaling pathways implicated in the pathogenesis of Parkinson’s disease. Neuromol. Med. 2014, 16, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Swomley, A.M.; Butterfield, D.A. Oxidative stress in Alzheimer disease and mild cognitive impairment: Evidence from human data provided by redox proteomics. Arch. Toxicol. 2015, 89, 1669–1680. [Google Scholar] [CrossRef] [PubMed]
- Mota, S.I.; Costa, R.O.; Ferreira, I.L.; Santana, I.; Caldeira, G.L.; Padovano, C.; Fonseca, A.C.; Baldeiras, I.; Cunha, C.; Letra, L.; et al. Oxidative stress involving changes in Nrf2 and ER stress in early stages of Alzheimer’s disease. Biochim. Biophys. Acta 2015, 1852, 1428–1441. [Google Scholar] [CrossRef] [PubMed]
- Meraz-Ríos, M.A.; Franco-Bocanegra, D.; Toral Rios, D.; Campos-Peña, V. Early onset Alzheimer’s disease and oxidative stress. Oxid. Med. Cell. Longev. 2014, 2014, 375968. [Google Scholar] [CrossRef] [PubMed]
- Tousoulis, D.; Psaltopoulou, T.; Androulakis, E.; Papageorgiou, N.; Papaioannou, S.; Oikonomou, E.; Synetos, A.; Stefanadis, C. Oxidative stress and early atherosclerosis: Novel antioxidant treatment. Cardiovasc. Drugs Ther. 2015, 29, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Horke, S.; Förstermann, U. Vascular oxidative stress, nitric oxide and atherosclerosis. Atherosclerosis 2014, 237, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Peluso, I.; Morabito, G.; Urban, L.; Ioannone, F.; Serafini, M. Oxidative stress in atherosclerosis development: The central role of LDL and oxidative burst. Endocr. Metab. Immune Disord. 2012, 12, 351–360. [Google Scholar] [CrossRef]
- Rosales-Corral, S.; Tan, D.X.; Manchester, L.; Reiter, R.J. Diabetes and Alzheimer disease, two overlapping pathologies with the same background: Oxidative stress. Oxid. Med. Cell. Longev. 2015, 2015, 985845. [Google Scholar] [CrossRef] [PubMed]
- Zephy, D.; Ahmad, J. Type 2 diabetes mellitus: Role of melatonin and oxidative stress. Diabetes Metab. Syndr. 2015, 9, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Maiese, K. New insights for oxidative stress and diabetes mellitus. Oxid. Med. Cell. Longev. 2015, 2015, 875961. [Google Scholar] [CrossRef] [PubMed]
- Ramis, M.R.; Esteban, S.; Miralles, A.; Tan, D.X.; Reiter, R.J. Protective effects of melatonin and mitochondria-targeted antioxidants against oxidative stress: A review. Curr. Med. Chem. 2015, 22, 2690–2711. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Tan, D.X.; Reiter, R.J. Melatonin as a natural ally against oxidative stress: A physicochemical examination. J. Pineal Res. 2011, 51, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R. Antioxidative protection by melatonin: Multiplicity of mechanisms from radical detoxification to radical avoidance. Endocrine 2005, 27, 119–130. [Google Scholar] [CrossRef]
- Jahnke, G.; Marr, M.; Myers, C.; Wilson, R.; Travlos, G.; Price, C. Maternal and developmental toxicity evaluation of melatonin administered orally to pregnant Sprague-Dawley rats. Toxicol. Sci. 1999, 50, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Ceraulo, L.; Ferrugia, M.; Tesoriere, L.; Segreto, S.; Livrea, M.A.; Turco Liveri, V. Interactions of melatonin with membrane models: Portioning of melatonin in AOT and lecithin reversed micelles. J. Pineal Res. 1999, 26, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Bonnefont-Rousselot, D.; Collin, F. Melatonin: Action as antioxidant and potential applications in human disease and aging. Toxicology 2010, 278, 55–67. [Google Scholar] [CrossRef] [PubMed]
- Gurer-Orhan, H.; Suzen, S. Melatonin, its metabolites and its synthetic analogs as multi-faceted compounds: Antioxidant, prooxidant and inhibitor of bioactivation reactions. Curr. Med. Chem. 2015, 22, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Tan, D.X.; Reiter, R.J. On the free radical scavenging activities of melatonin’s metabolites, AFMK and AMK. J. Pineal Res. 2013, 54, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Jou, M.J.; Korkmaz, A.; Manchester, L.C.; Paredes, S.D. Biogenic amines in the reduction of oxidative stress: Melatonin and its metabolites. Neuro Endocrinol. Lett. 2008, 29, 391–398. [Google Scholar] [PubMed]
- Álvarez-Diduk, R.; Galano, A.; Tan, D.X.; Reiter, R.J. N-Acetylserotonin and 6-hydroxymelatonin against oxidative stress: Implications for the overall protection exerted by melatonin. J. Phys. Chem. B 2015, 119, 8535–8543. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Hardeland, R.; Manchester, L.C.; Galano, A.; Reiter, R.J. Cyclic-3-hydroxymelatonin (C3HOM), a potent antioxidant, scavenges free radicals and suppresses oxidative reactions. Curr. Med. Chem. 2014, 21, 1557–1565. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Tan, D.X.; Reiter, R.J. Cyclic 3-hydroxymelatonin, a key metabolite enhancing the peroxyl radical scavenging activity of melatonin. RSC Adv. 2014, 4, 5220–5227. [Google Scholar] [CrossRef]
- Martins, N.; Barros, L.; Ferreira, I.C.F.R. In vivo antioxidant activity of phenolic compounds: Facts and gaps. Trends Food Sci. Technol. 2016, 48, 1–12. [Google Scholar] [CrossRef]
- Achat, S.; Rakotomanomana, N.; Madani, K.; Dangles, O. Antioxidant activity of olive phenols and other dietary phenols in model gastric conditions: Scavenging of the free radical DPPH and inhibition of the haem-induced peroxidation of linoleic acid. Food Chem. 2016, 213, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects—A review. J. Funct. Foods 2015, 18, 820–897. [Google Scholar] [CrossRef]
- Galano, A. Free Radicals Induced Oxidative Stress at a Molecular Level: The Current Status, Challenges and Perspectives of Computational Chemistry Based Protocols. J. Mex. Chem. Soc. 2015, 59, 231–262. [Google Scholar]
- Araújo, M.; Pimentel, F.B.; Alves, R.C.; Oliveira, M.B.P.P. Phenolic compounds from olive mill wastes: Health effects, analytical approach and application as food antioxidants. Trends Food Sci. Technol. 2015, 45, 200–211. [Google Scholar] [CrossRef]
- Liu, L.; Jin, C.; Zhang, Y. Lipophilic phenolic compounds (Lipo-PCs): Emerging antioxidants applied in lipid systems. RSC Adv. 2014, 4, 2879–2891. [Google Scholar] [CrossRef]
- Ho, C.T.; Wang, M. Dietary phenolics as reactive carbonyl scavengers: Potential impact on human health and mechanism of action. J. Tradit. Complement. Med. 2013, 3, 139–141. [Google Scholar] [CrossRef] [PubMed]
- Galano, A. Computational-aided design of melatonin analogues with outstanding multifunctional antioxidant capacity. RSC Adv. 2016, 6, 22951–22963. [Google Scholar] [CrossRef]
- Gerschman, R.; Gilbert, D.L.; Nye, S.W.; Dwyer, P.; Fenn, W.O. Oxygen poisoning and X-irradiation: A mechanism in common. Science 1954, 119, 623–626. [Google Scholar] [CrossRef] [PubMed]
- Kaminskyy, V.O.; Zhivotovsky, B. Free radicals in cross talk between autophagy and apoptosis. Antioxid. Redox Signal. 2014, 21, 86–102. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Zhu, M. Free radical oxidation of cardiolipin: Chemical mechanisms, detection and implication in apoptosis, mitochondrial dysfunction and human diseases. Free Radic. Res. 2012, 46, 959–974. [Google Scholar] [CrossRef] [PubMed]
- Sayre, L.M.; Perry, G.; Smith, M.A. Oxidative stress and neurotoxicity. Chem. Res. Toxicol. 2008, 21, 172–188. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.; Walder, K.; Puri, B.K.; Berk, M.; Maes, M. The Deleterious Effects of Oxidative and Nitrosative Stress on Palmitoylation, Membrane Lipid Rafts and Lipid-Based Cellular Signalling: New Drug Targets in Neuroimmune Disorders. Mol. Neurobiol. 2016, 53, 4638–4658. [Google Scholar] [CrossRef] [PubMed]
- Niedzielska, E.; Smaga, I.; Gawlik, M.; Moniczewski, A.; Stankowicz, P.; Pera, J.; Filip, M. Oxidative Stress in Neurodegenerative Diseases. Mol. Neurobiol. 2016, 53, 4094–4125. [Google Scholar] [CrossRef] [PubMed]
- Daulatzai, M.A. Fundamental role of pan-inflammation and oxidative-nitrosative pathways in neuropathogenesis of Alzheimer’s disease. Am. J. Neurodegener. Dis. 2016, 5, 1–28. [Google Scholar] [PubMed]
- Ljubisavljevic, S. Oxidative Stress and Neurobiology of Demyelination. Mol. Neurobiol. 2016, 53, 744–758. [Google Scholar] [CrossRef] [PubMed]
- Cobb, C.A.; Cole, M.P. Oxidative and nitrative stress in neurodegeneration. Neurobiol. Dis. 2015, 84, 4–21. [Google Scholar] [CrossRef] [PubMed]
- Bhat, A.H.; Dar, K.B.; Anees, S.; Zargar, M.A.; Masood, A.; Sofi, M.A.; Ganie, S.A. Oxidative stress, mitochondrial dysfunction and neurodegenerative diseases; a mechanistic insight. Biomed. Pharmacother. 2015, 74, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Fischer, R.; Maier, O. Interrelation of oxidative stress and inflammation in neurodegenerative disease: Role of TNF. Oxid. Med. Cell. Longev. 2015, 2015, 610813. [Google Scholar] [CrossRef] [PubMed]
- Hecht, F.; Pessoa, C.F.; Gentile, L.B.; Rosenthal, D.; Carvalho, D.P.; Fortunato, R.S. The role of oxidative stress on breast cancer development and therapy. Tumor Biol. 2016, 37, 4281–4291. [Google Scholar] [CrossRef] [PubMed]
- Zuo, T.; Zhu, M.; Xu, W. Roles of oxidative stress in polycystic ovary syndrome and cancers. Oxid. Med. Cell. Longev. 2016, 2016, 8589318. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Li, S.; Cao, Y.; Tian, X.; Zeng, R.; Liao, D.F.; Cao, D. Oxidative stress and carbonyl lesions in ulcerative colitis and associated colorectal cancer. Oxid. Med. Cell. Longev. 2016, 2016, 9875298. [Google Scholar] [CrossRef] [PubMed]
- Dizdaroglu, M. Oxidatively induced DNA damage and its repair in cancer. Mutat. Res. Rev. Mutat. Res. 2015, 763, 212–245. [Google Scholar] [CrossRef] [PubMed]
- Choudhari, S.K.; Chaudhary, M.; Gadbail, A.R.; Sharma, A.; Tekade, S. Oxidative and antioxidative mechanisms in oral cancer and precancer: A review. Oral Oncol. 2014, 50, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Kayama, Y.; Raaz, U.; Jagger, A.; Adam, M.; Schellinger, I.N.; Sakamoto, M.; Suzuki, H.; Toyama, K.; Spin, J.M.; Tsao, P.S. Diabetic cardiovascular disease induced by oxidative stress. Int. J. Mol. Sci. 2015, 16, 25234–25263. [Google Scholar] [CrossRef] [PubMed]
- Roul, D.; Recchia, F.A. Metabolic Alterations Induce Oxidative Stress in Diabetic and Failing Hearts: Different Pathways, Same Outcome. Antioxid. Redox Signal. 2015, 22, 1502–1514. [Google Scholar] [CrossRef] [PubMed]
- Marseglia, L.; D’Angelo, G.; Manti, S.; Arrigo, T.; Barberi, I.; Reiter, R.J.; Gitto, E. Oxidative stress-mediated aging during the fetal and perinatal periods. Oxid. Med. Cell. Longev. 2014, 2014, 358375. [Google Scholar] [CrossRef] [PubMed]
- Wells, P.G.; Bhatia, S.; Drake, D.M.; Miller-Pinsler, L. Fetal oxidative stress mechanisms of neurodevelopmental deficits and exacerbation by ethanol and methamphetamine. Birth Defects Res. C Embryo Today 2016, 108, 108–130. [Google Scholar] [CrossRef] [PubMed]
- Hansson, S.R.; Nääv, A.; Erlandsson, L. Oxidative stress in preeclampsia and the role of free fetal hemoglobin. Front. Physiol. 2015, 6, 516. [Google Scholar] [CrossRef] [PubMed]
- Perrone, S.; Tataranno, M.L.; Stazzoni, G.; Buonocore, G. Biomarkers of oxidative stress in fetal and neonatal diseases. J. Matern. Fetal Neonatal Med. 2012, 25, 2575–2578. [Google Scholar] [CrossRef] [PubMed]
- Correia-Costa, L.; Sousa, T.; Morato, M.; Cosme, D.; Afonso, J.; Areias, J.C.; Schaefer, F.; Guerra, A.; Afonso, A.C.; Azevedo, A.; et al. Oxidative stress and nitric oxide are increased in obese children and correlate with cardiometabolic risk and renal function. Br. J. Nutr. 2016, 116, 805–815. [Google Scholar] [CrossRef] [PubMed]
- Di Pietro, M.; Filardo, S.; de Santis, F.; Mastromarino, P.; Sessa, R. Chlamydia pneumoniae and oxidative stress in cardiovascular disease: State of the art and prevention strategies. Int. J. Mol. Sci. 2015, 16, 724–735. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Xia, S.; Kalionis, B.; Wan, W.; Sun, T. The Role of Oxidative Stress and Inflammation in Cardiovascular Aging. Biomed. Res. Int. 2014, 2014, 615312. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.Y.; Xu, X.; Li, X.C. Cardiovascular diseases: Oxidative damage and antioxidant protection. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 3091–3096. [Google Scholar] [PubMed]
- Ferrari, R.S.; Andrade, C.F. Oxidative Stress and Lung Ischemia-Reperfusion Injury. Oxid. Med. Cell. Longev. 2015, 2015, 590987. [Google Scholar] [CrossRef] [PubMed]
- Cheresh, P.; Kim, S.J.; Tulasiram, S.; Kamp, D.W. Oxidative stress and pulmonary fibrosis. Biochim. Biophys. Acta 2013, 1832, 1028–1040. [Google Scholar] [CrossRef] [PubMed]
- Pandey, R.; Singh, M.; Singhal, U.; Gupta, K.B.; Aggarwal, S.K. Oxidative/nitrosative stress and the pathobiology of chronic obstructive pulmonary disease. J. Clin. Diagn. Res. 2013, 7, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Ratliff, B.B.; Abdulmahdi, W.; Pawar, R.; Wolin, M.S. Oxidant mechanisms in renal injury and disease. Antioxid. Redox Signal. 2016, 25, 119–146. [Google Scholar] [CrossRef] [PubMed]
- Tamay-Cach, F.; Quintana-Pérez, J.C.; Trujillo-Ferrara, J.G.; Cuevas-Hernández, R.I.; Del Valle-Mondragón, L.; García-Trejo, E.M.; Arellano-Mendoza, M.G. A review of the impact of oxidative stress and some antioxidant therapies on renal damage. Ren. Fail. 2016, 38, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Nita, M.; Grzybowski, A. The Role of the Reactive Oxygen Species and Oxidative Stress in the Pathomechanism of the Age-Related Ocular Diseases and Other Pathologies of the Anterior and Posterior Eye Segments in Adults. Oxid. Med. Cell. Longev. 2016, 2016, 3164734. [Google Scholar] [CrossRef] [PubMed]
- Kruk, J.; Kubasik-Kładna, K.; Aboul-Enein, H.Y. The role oxidative stress in the pathogenesis of eye diseases: Current status and a dual role of physical activity. Mini-Rev. Med. Chem. 2016, 16, 241–257. [Google Scholar] [CrossRef]
- Saccà, S.C.; Izzotti, A. Oxidative stress and glaucoma: Injury in the anterior segment of the eye. Prog. Brain Res. 2008, 173, 385–407. [Google Scholar] [PubMed]
- Griffiths, H.R.; Dunston, C.R.; Bennett, S.J.; Grant, M.M.; Phillips, D.C.; Kitas, G.D. Free radicals and redox signalling in T-cells during chronic inflammation and ageing. Biochem. Soc. Trans. 2011, 39, 1273–1278. [Google Scholar] [CrossRef] [PubMed]
- Schetter, A.J.; Heegaard, N.H.H.; Harris, C.C. Inflammation and cancer: Interweaving microRNA, free radical, cytokine and p53 pathways. Carcinogenesis 2009, 31, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Giannapas, M.; Karnis, L.; Dailianis, S. Generation of free radicals in haemocytes of mussels after exposure to low molecular weight PAH components: Immune activation, oxidative and genotoxic effects. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2012, 155, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Lantow, M.; Schuderer, J.; Hartwig, C.; Simkó, M. Free radical release and HSP70 expression in two human immune-relevant cell lines after exposure to 1800 MHz radiofrequency radiation. Radiat. Res. 2006, 165, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Wei, B.; Song, X.; An, N.; Zhou, Y.; Jin, X.; Zhang, Y. Edaravone’s free radical scavenging mechanisms of neuroprotection against cerebral ischemia: Review of the literature. Int. J. Neurosci. 2015, 125, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Niatsetskaya, Z.V.; Sosunov, S.A.; Matsiukevich, D.; Utkina-Sosunova, I.V.; Ratner, V.I.; Starkov, A.A.; Ten, V.S. The oxygen free radicals originating from mitochondrial complex I contribute to oxidative brain injury following hypoxia-ischemia in neonatal mice. J. Neurosci. 2012, 32, 3235–3244. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H. The link between infection and cancer: Tumor vasculature, free radicals, and drug delivery to tumors via the EPR effect. Cancer Sci. 2013, 104, 779–789. [Google Scholar] [CrossRef] [PubMed]
- Guha, M.; Maity, P.; Choubey, V.; Mitra, K.; Reiter, R.J.; Bandyopadhyay, U. Melatonin inhibits free radical-mediated mitochondrial-dependent hepatocyte apoptosis and liver damage induced during malarial infection. J. Pineal Res. 2007, 43, 372–381. [Google Scholar] [CrossRef] [PubMed]
- De Luca, C.; Deeva, I.; Mariani, S.; Maiani, G.; Stancato, A.; Korkina, L. Monitoring antioxidant defenses and free radical production in space-flight, aviation and railway engine operators, for the prevention and treatment of oxidative stress, immunological impairment, and pre-mature cell aging. Toxicol. Ind. Health 2009, 25, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Morales-Alamo, D.; Calbet, J.A.L. Free radicals and sprint exercise in humans. Free Radic. Res. 2014, 48, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Benameur, L.; Charif, N.; Li, Y.; Stoltz, J.F.; de Isla, N. Toward an understanding of mechanism of aging-induced oxidative stress in human mesenchymal stem cells. Bio-Med. Mater. Eng. 2015, 25, S41–S46. [Google Scholar]
- Shiraiwa, M.; Selzle, K.; Pöschl, U. Hazardous components and health effects of atmospheric aerosol particles: Reactive oxygen species, soot, polycyclic aromatic compounds and allergenic proteins. Free Radic. Res. 2012, 46, 927–939. [Google Scholar] [CrossRef] [PubMed]
- Valencia-Islas, N.; Zambrano, A.; Rojas, J.L. Ozone reactivity and free radical scavenging behavior of phenolic secondary metabolites in lichens exposed to chronic oxidant air pollution from Mexico City. J. Chem. Ecol. 2007, 33, 1619–1634. [Google Scholar] [CrossRef] [PubMed]
- Kamiński, P.; Kurhalyuk, N.; Szady-Grad, M. Heavy metal-induced oxidative stress and changes in physiological process of free radicals in the blood of white stork (Ciconia ciconia) chicks in polluted areas. Pol. J. Environ. Stud. 2007, 16, 555–562. [Google Scholar]
- Vejerano, E.; Lomnicki, S.; Dellinger, B. Lifetime of combustion-generated environmentally persistent free radicals on Zn(II)O and other transition metal oxides. J. Environ. Monit. 2012, 14, 2803–2806. [Google Scholar] [CrossRef] [PubMed]
- Robinson, E.A.; Johnson, J.D. Methods for analysis of free radicals in cigarette smoke. Mini Rev. Org. Chem. 2011, 8, 401–411. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Wu, D.; Liu, B.; Xie, W. Electron paramagnetic resonance spin-trapping study of free radicals in gas phase of mainstream cigarette smoke using two spin traps. Tobacco Sci. Technol. 2014, 47, 47–53. [Google Scholar]
- Michail, K.; Baghdasarian, A.; Narwaley, M.; Aljuhani, N.; Siraki, A.G. Scavenging of free-radical metabolites of aniline xenobiotics and drugs by amino acid derivatives: Toxicological implications of radical-transfer reactions. Chem. Res. Toxicol. 2013, 26, 1872–1883. [Google Scholar] [CrossRef] [PubMed]
- Aleryani, S.L.; Aleryani, R.A.; Al-Akwa, A.A. Khat a drug of abuse: Roles of free radicals and antioxidants. Drug Test. Anal. 2011, 3, 548–551. [Google Scholar] [CrossRef] [PubMed]
- Narwaley, M.; Michail, K.; Arvadia, P.; Siraki, A.G. Drug-induced protein free radical formation is attenuated by unsaturated fatty acids by scavenging drug-derived phenyl radical metabolites. Chem. Res. Toxicol. 2011, 24, 1031–1039. [Google Scholar] [CrossRef] [PubMed]
- Karadayian, A.G.; Bustamante, J.; Czerniczyniec, A.; Lombardi, P.; Cutrera, R.A.; Lores-Arnaiz, S. Alcohol hangover induces mitochondrial dysfunction and free radical production in mouse cerebellum. Neuroscience 2015, 304, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Albano, E. Alcohol, oxidative stress and free radical damage. Proc. Nutr. Soc. 2006, 65, 278–290. [Google Scholar] [CrossRef] [PubMed]
- Spitz, D.R.; Hauer-Jensen, M. Ionizing radiation-induced responses: Where free radical chemistry meets redox biology and medicine. Antioxid. Redox Signal. 2014, 20, 1407–1409. [Google Scholar] [CrossRef] [PubMed]
- Rancan, F.; Nazemi, B.; Rautenberg, S.; Ryll, M.; Hadam, S.; Gao, Q.; Hackbarth, S.; Haag, S.F.; Graf, C.; Rühl, E.; et al. Ultraviolet radiation and nanoparticle induced intracellular free radicals generation measured in human keratinocytes by electron paramagnetic resonance spectroscopy. Skin Res. Technol. 2014, 20, 182–193. [Google Scholar] [CrossRef] [PubMed]
- Burlaka, A.; Tsybulin, O.; Sidorik, E.; Lukin, S.; Polishuk, V.; Tsehmistrenko, S.; Yakymenko, I. Overproduction of free radical species in embryonal cells exposed to low intensity radiofrequency radiation. Exp. Oncol. 2013, 35, 219–225. [Google Scholar] [PubMed]
- Milnerowicz, H.; Ściskalska, M.; Dul, M. Molecular mechanisms of the impact of smoke-oxidants. Exp. Toxicol. Pathol. 2015, 67, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Buxton, G.V.; Greenstock, C.L.; Helman, W.P.; Ross, A.B. Critical Review of rate constants for reactions of hydrated electrons, hydrogen atoms and hydroxyl radicals (•OH/•O− in aqueous solution. J. Phys. Chem. Ref. Data 1988, 17, 513–886. [Google Scholar] [CrossRef]
- Rose, R.C.; Bode, A.M. Biology of free radical scavengers: An evaluation of ascorbate. FASEB J. 1993, 7, 1135–1142. [Google Scholar] [PubMed]
- Reiter, R.J.; Tan, D.X.; Herman, T.S.; Thomas, C.R., Jr. Melatonin as a radioprotective agent: A review. Int. J. Radiat. Oncol. Biol. Phys. 2004, 59, 639–653. [Google Scholar]
- Candeias, L.P.; Steenken, S. Reaction of HO• with guanine derivatives in aqueous solution: Formation of two different redox-active OH-adduct radicals and their unimolecular transformation reactions. Properties of G(-H)•. Chem. Eur. J. 2000, 6, 475–484. [Google Scholar] [CrossRef]
- Chatgilialoglu, C.; D’Angelantonio, M.; Guerra, M.; Kaloudis, P.; Mulazzani, Q.G. A reevaluation of the ambident reactivity of the guanine moiety towards hydroxyl radicals. Angew. Chem. Int. Ed. Engl. 2009, 48, 2214–2217. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Alvarez-Idaboy, J.R. Guanosine + OH radical reaction in aqueous solution: A reinterpretation of the UV-vis data based on thermodynamic and kinetic calculations. Org. Lett. 2009, 11, 5114–5117. [Google Scholar] [CrossRef] [PubMed]
- León-Carmona, J.R.; Galano, A. Is caffeine a good scavenger of oxygenated free radicals? J. Phys. Chem. B 2011, 115, 4538–4546. [Google Scholar] [CrossRef] [PubMed]
- Galano, A. Relative antioxidant efficiency of a large series of carotenoids in terms of one electron transfer reactions. J. Phys. Chem. B 2007, 111, 12898–12908. [Google Scholar] [CrossRef] [PubMed]
- Martinez, A.; Vargas, R.; Galano, A. What is important to prevent oxidative stress? A theoretical study on electron-transfer reactions between carotenoids and free radicals. J. Phys. Chem. B 2009, 113, 12113–12120. [Google Scholar] [CrossRef] [PubMed]
- Martínez, A.; Vargas, R.; Galano, A. Theoretical study on the chemical fate of adducts formed through free radical addition reactions to carotenoids. Theor. Chem. Acc. 2010, 127, 595–603. [Google Scholar] [CrossRef]
- Martínez, A.; Galano, A. Free radical scavenging activity of ultrashort single-walled carbon nanotubes with different structures through electron transfer reactions. J. Phys. Chem. C 2010, 114, 8184–8191. [Google Scholar] [CrossRef]
- Marnett, L.J. Peroxyl free radicals: Potential mediators of tumor initiation and promotion. Carcinogenesis 1987, 8, 1365–1373. [Google Scholar] [CrossRef] [PubMed]
- Terpinc, P.; Abramovič, H. A kinetic approach for evaluation of the antioxidant activity of selected phenolic acids. Food Chem. 2010, 121, 366–371. [Google Scholar] [CrossRef]
- Sies, H. Oxidative stress: Oxidants and antioxidants. Exp. Physiol. 1997, 82, 291–295. [Google Scholar] [CrossRef] [PubMed]
- De Grey, A.D.N.J. HO2•: The forgotten radical. DNA Cell Biol. 2002, 21, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Bielski, B.H.J.; Arudi, R.L.; Sutherland, M.W. A study of the reactivity of HO2/O2—With unsaturated fatty acids. J. Biol. Chem. 1983, 258, 4759–4761. [Google Scholar] [PubMed]
- Aikens, J.; Dix, T.A. Perhydroxyl radical (HOO•) initiated lipid peroxidation: The role of fatty acid hydroperoxides. J. Biol. Chem. 1991, 266, 15091–15098. [Google Scholar] [PubMed]
- Galano, A. On the direct scavenging activity of melatonin towards hydroxyl and a series of peroxyl radicals. Phys. Chem. Chem. Phys. 2011, 13, 7178–7188. [Google Scholar] [CrossRef] [PubMed]
- Neta, P.; Huie, R.E.; Mosseri, S.; Shastri, L.V.; Mittal, J.P.; Maruthamuthu, P.; Steenken, S. Rate constants for reduction of substituted methylperoxyl radicals by ascorbate ions and N,N,N′,N′,-Tetramethyl-p-phenylenediamine. J. Phys. Chem. 1989, 93, 4099–4104. [Google Scholar] [CrossRef]
- Brunelli, L.; Crow, J.P.; Beckman, J.S. The comparative toxicity of nitric oxide and peroxynitrite to Escherichia coli. Arch. Biochem. Biophys. 1995, 316, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Squadrito, G.L.; Pryor, W.A. Oxidative chemistry of nitric oxide: The roles of superoxide, peroxynitrite, and carbon dioxide. Free Radic. Biol. Med. 1998, 25, 392–403. [Google Scholar] [CrossRef]
- Radi, R.; Peluffo, G.; Alvarez, M.N.; Naviliat, M.; Cayota, A. Unraveling peroxynitrite formation in biological systems. Free Radic. Biol. Med. 2001, 30, 463–488. [Google Scholar] [CrossRef]
- Douki, T.; Cadet, J. Peroxynitrite mediated oxidation of purine bases of nucleosides and isolated DNA. Free Radic. Res. 1996, 24, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Wiseman, H.; Halliwell, B. Damage to DNA by reactive oxygen and nitrogen species: Role in inflammatory disease and progression to cancer. Biochem. J. 1996, 313, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Koppal, T.; Drake, J.; Yatin, S.; Jordan, B.; Varadarajan, S.; Bettenhausen, L.; Butterfield, D.A. Peroxynitrite-induced alterations in synaptosomal membrane proteins: Insight into oxidative stress in Alzheimer’s disease. J. Neurochem. 1999, 72, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Prütz, W.A.; Mönig, H.; Butler, J.; Land, E.J. Reactions of nitrogen dioxide in aqueous model systems: Oxidation of tyrosine units in peptides and proteins. Arch. Biochem. Biophys. 1985, 243, 125–134. [Google Scholar] [CrossRef]
- Abedinzadeh, Z. Sulfur-centered reactive intermediates derived from the oxidation of sulfur compounds of biological interest. Can. J. Physiol. Pharmacol. 2001, 79, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Giles, G.I.; Tasker, K.M.; Jacob, C. Hypothesis: The role of reactive sulfur species in oxidative stress. Free Radic. Biol. Med. 2001, 31, 1279–1283. [Google Scholar] [CrossRef]
- Giles, G.I.; Tasker, K.M.; Jacob, C. Oxidation of biological thiols by highly reactive disulfide-S-oxides. Gen. Physiol. Biophys. 2002, 21, 65–72. [Google Scholar] [PubMed]
- Mishanina, T.V.; Libiad, M.; Banerjee, R. Biogenesis of reactive sulfur species for signaling by hydrogen sulfide oxidation pathways. Nat. Chem. Biol. 2015, 11, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Rao, A.V.; Agarwal, S. Role of lycopene as antioxidant carotenoid in the prevention of chronic diseases: A review. Nutr. Res. 1999, 19, 305–323. [Google Scholar] [CrossRef]
- Sita, G.; Hrelia, P.; Tarozzi, A.; Morroni, F. Isothiocyanates are promising compounds against oxidative stress, neuroinflammation and cell death that may benefit neurodegeneration in Parkinson’s disease. Int. J. Mol. Sci. 2016, 17, 1454. [Google Scholar] [CrossRef] [PubMed]
- Skibska, B.; Goraca, A. The protective effect of lipoic acid on selected cardiovascular diseases caused by age-related oxidative stress. Oxid. Med. Cell. Longev. 2015, 2015, 313021. [Google Scholar] [CrossRef] [PubMed]
- Lançon, A.; Frazzi, R.; Latruffe, N. Anti-oxidant, anti-inflammatory and anti-angiogenic properties of resveratrol in ocular diseases. Molecules 2016, 21, 304. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tsao, R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Niki, E. Antioxidant capacity of foods for scavenging reactive oxidants and inhibition of plasma lipid oxidation induced by multiple oxidants. Food Funct. 2016, 7, 2156–2168. [Google Scholar] [CrossRef] [PubMed]
- Amir Aslani, B.; Ghobadi, S. Studies on oxidants and antioxidants with a brief glance at their relevance to the immune system. Life Sci. 2016, 146, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Kovacic, P.; Somanathan, R.; Abadjian, M.C.Z. Natural monophenols as therapeutics, antioxidants and toxins; electron transfer, radicals and oxidative stress. Nat. Prod. J. 2015, 5, 142–151. [Google Scholar] [CrossRef]
- Chaiyasit, W.; Elias, R.J.; McClements, D.J.; Decker, E.A. Role of physical structures in bulk oils on lipid oxidation. Crit. Rev. Food Sci. Nutr. 2007, 47, 299–317. [Google Scholar] [CrossRef] [PubMed]
- Litwinienko, G.; Ingold, K.U. Abnormal solvent effects on hydrogen atom abstractions. 1. The reactions of phenols with 2,2-diphenyl-1-picrylhydrazyl (dpph•) in alcohols. J. Org. Chem. 2003, 68, 3433–3438. [Google Scholar] [CrossRef] [PubMed]
- Weinstein, J.; Bielski, B.H.J. Kinetics of the interaction of HO2 and O2—Radicals with hydrogen peroxide. The Haber-Weiss reaction. J. Am. Chem. Soc. 1979, 101, 58–62. [Google Scholar] [CrossRef]
- Rodriguez-Rodriguez, C.; Sanchez de Groot, N.; Rimola, A.; Alvarez-Larena, A.; Lloveras, V.; Vidal-Gancedo, J.; Ventura, S.; Vendrell, J.; Sodupe, M.; Gonzalez-Duarte, P. Design, selection, and characterization of thioflavin-based intercalation compounds with metal chelating properties for application in alzheimer’s disease. J. Am. Chem. Soc. 2009, 131, 1436–1451. [Google Scholar] [CrossRef] [PubMed]
- Francisco-Marquez, M.; Aguilar-Fernández, M.; Galano, A. Anthranilic acid as a secondary antioxidant: Implications to the inhibition of OH production and the associated oxidative stress. Comput. Theor. Chem. 2016, 1077, 18–24. [Google Scholar] [CrossRef]
- Miche, H.; Brumas, V.; Berthon, G. Copper(II) interactions with nonsteroidal antiinflammatory agents. II. Anthranilic acid as a potential OH-inactivating ligand. J. Inorg. Biochem. 1997, 68, 27–38. [Google Scholar] [CrossRef]
- Gaubert, S.; Bouchaut, M.; Brumas, V.; Berthon, G. Copper-ligand interactions and physiological free radical processes. Part 3. Influence of histidine, salicylic acid and anthranilic acid on copper-driven Fenton chemistry in vitro. Free Radic. Res. 2000, 32, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Berthon, G. Is copper pro- or anti-inflammatory? A reconciling view and a novel approach for the use of copper in the control of inflammation. Agents Actions 1993, 39, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Terao, J.; Minami, Y.; Bando, N. Singlet molecular oxygen-quenching activity of carotenoids: Relevance to protection of the skin from photoaging. J. Clin. Biochem. Nutr. 2011, 48, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, F.; Zhong, Y. Measurement of antioxidant activity. J. Funct. Foods 2015, 18, 757–781. [Google Scholar] [CrossRef]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized Methods for the Determination of Antioxidant Capacity and Phenolics in Foods and Dietary Supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- Frankel, E.N.; Meyer, A.S. The problems of using one-dimensional methods to evaluate multifunctional food and biological antioxidants. J. Sci. Food Agric. 2000, 80, 1925–1941. [Google Scholar] [CrossRef]
- Antolovich, M.; Prenzler, P.D.; Patsalides, E.; McDonald, S.; Robards, K. Methods for testing antioxidant activity. Analyst 2002, 127, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Boxin, O.U.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef] [PubMed]
- Donno, D.; Cavanna, M.; Beccaro, G.L.; Mellano, M.G.; Torello Marinoni, D.; Cerutti, A.K.; Bounous, G. Currants and strawberries as bioactive compound sources: Determination of antioxidant profiles with HPLC-DAD/MS. J. Appl. Bot. Food Qual. 2013, 86, 1–10. [Google Scholar]
- Huang, D.; Ou, B.; Hampsch-Woodill, M.; Flanagan, J.A.; Deemer, E.K. Development and validation of oxygen radical absorbance capacity assay for lipophilic antioxidants using randomly methylated β-cyclodextrin as the solubility enhancer. J. Agric. Food Chem. 2002, 50, 1815–1821. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, A.; Selga, A.; Torres, J.L.; Julià, L. Reducing Activity of Polyphenols with Stable Radicals of the TTM Series. Electron Transfer versus H-Abstraction Reactions in Flavan-3-ols. Org. Lett. 2004, 6, 4583–4586. [Google Scholar] [CrossRef] [PubMed]
- Benzie, I.F.F.; Strain, J.J. Ferric reducing/antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Methods Enzymol. 1998, 299, 15–27. [Google Scholar]
- Xie, J.; Schaich, K.M. Re-evaluation of the 2,2-diphenyl-1-picrylhydrazyl free radical (DPPH) assay for antioxidant activity. J. Agric. Food Chem. 2014, 62, 4251–4260. [Google Scholar] [CrossRef] [PubMed]
- Arnao, M.B. Some methodological problems in the determination of antioxidant activity using chromogen radicals: A practical case. Trends Food Sci. Technol. 2001, 11, 419–421. [Google Scholar] [CrossRef]
- Berker, K.I.; Demirata, B.; Apak, R. Determination of Total Antioxidant Capacity of Lipophilic and Hydrophilic Antioxidants in the Same Solution by Using Ferric-Ferricyanide Assay. Food Anal. Methods 2012, 5, 1150–1158. [Google Scholar] [CrossRef]
- Galano, A.; Alvarez-Idaboy, J.R. A computational methodology for accurate predictions of rate constants in solution: Application to the assessment of primary antioxidant activity. J. Comput. Chem. 2013, 34, 2430–2445. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Alvarez-Idaboy, J.R. Kinetics of radical-molecule reactions in aqueous solution: A benchmark study of the performance of density functional methods. J. Comput. Chem. 2014, 35, 2019–2026. [Google Scholar] [CrossRef] [PubMed]
- Mahal, H.S.; Sharma, H.S.; Mukherjee, T. Antioxidant properties of melatonin: A pulse radiolysis study. Free Radic. Biol. Med. 1999, 26, 557–565. [Google Scholar] [CrossRef]
- Wardman, P. Reduction Potentials of One Electron Couples Involving Free Radicals in Aqueous Solution. J. Phys. Chem. Ref. Data 1989, 18, 1637–1755. [Google Scholar] [CrossRef]
- Paradies, G.; Petrosillo, G.; Paradies, V.; Reiter, R.J.; Ruggiero, F.M. Melatonin, cardiolipin and mitochondrial bioenergetics in health and disease. J. Pineal Res. 2010, 48, 297–310. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Reiter, R.J.; Qi, W.B.; Karbownik, M.; Calvo, J.R. Significance of melatonin in antioxidative defense system: Reactions and products. Biol. Signals Recept. 2000, 9, 137–159. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Reiter, R.J.; Manchester, L.C.; Yan, M.T.; El-Sawi, M.; Sainz, R.M.; Mayo, J.C.; Kohen, R.; Allegra, M.; Hardeland, R. Chemical and physical properties and potential mechanisms: Melatonin as a broad spectrum antioxidant and free radical scavenger. Curr. Top. Med. Chem. 2002, 2, 181–197. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R.; Reiter, R.J.; Poeggeler, B.; Tan, D.X. The significance of the metabolism of the neurohormone melatonin: Antioxidative protection and formation of bioactive substances. Neurosci. Biobehav. Rev. 1993, 17, 347–357. [Google Scholar] [CrossRef]
- Tan, D.X.; Hardeland, R.; Manchester, L.C.; Paredes, S.D.; Korkmaz, A.; Sainz, R.M.; Mayo, J.C.; Fuentes-Broto, L.; Reiter, R.J. The changing biological roles of melatonin during evolution: From an antioxidant to signals of darkness, sexual selection and fitness. Biol. Rev. 2010, 85, 607–623. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, M.; Fukuda, A.; Hara, M.; Terada, A.; Kitanaka, Y.; Owada, S. Melatonin prevents the increase in hydroxyl radical-spin trap adduct formation caused by the addition of cisplatin in vitro. Life Sci. 2003, 72, 1773–1780. [Google Scholar] [CrossRef]
- Scaiano, J.C. Exploratory laser flash photolysis study of free radical reactions and magnetic field effects in melatonin chemistry. J. Pineal Res. 1995, 19, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Acuña-Castroviejo, D.; Tan, D.X.; Burkhardt, S. Free radical-mediated molecular damage: Mechanisms for the protective actions of melatonin in the central nervous system. Ann. N. Y. Acad. Sci. 2001, 939, 200–215. [Google Scholar] [CrossRef] [PubMed]
- Cagnoli, C.M.; Atabay, C.; Kharlamova, E.; Manev, H. Melatonin protects neurons from singlet oxygen-induced apoptosis. J. Pineal Res. 1995, 18, 222–226. [Google Scholar] [CrossRef] [PubMed]
- Matuszak, Z.; Bilska, M.A.; Reszkat, K.J.; Chignell, C.F.; Bilski, P. Interaction of Singlet Molecular Oxygen with Melatonin and Related Indoles. Photochem. Photobiol. 2003, 78, 449–455. [Google Scholar] [CrossRef]
- Escames, G.; Guerrero, J.M.; Reiter, R.J.; Garcia, J.J.; Munoz-Hoyos, A.; Ortiz, G.G.; Oh, C.S. Melatonin and vitamin E limit nitric oxide-induced lipid peroxidation in rat brain homogenates. Neurosci. Lett. 1997, 230, 147–150. [Google Scholar] [CrossRef]
- Siu, A.W.; Ortiz, G.G.; Benitez-King, G.; To, C.H.; Reiter, R.J. Effect of melatonin on the nitric oxide treated retina. Br. J. Ophthalmol. 2004, 88, 1078–1081. [Google Scholar] [CrossRef] [PubMed]
- Melchiorri, D.; Reiter, R.J.; Attia, A.M.; Hara, M.; Burgos, A.; Nistico, G. Potent protective effect of melatonin on in vivo paraquat-induced oxidative damage in rats. Life Sci. 1994, 56, 83–89. [Google Scholar] [CrossRef]
- Zavodnik, I.B.; Domanski, A.V.; Lapshina, E.A.; Bryszewska, M.; Reiter, R.J. Melatonin directly scavenges free radicals generated in red blood cells and a cell-free system: Chemiluminescence measurements and theoretical calculations. Life Sci. 2006, 79, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Gitto, E.; Karbownik, M.; Reiter, R.J.; Xian Tan, D.; Cuzzocrea, S.; Chiurazzi, P.; Cordaro, S.; Corona, G.; Trimarchi, G.; Barberi, I. Effects of melatonin treatment in septic newborns. Pediatr. Res. 2001, 50, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Longoni, B.; Salgo, M.G.; Pryor, W.A.; Marchiafava, P.L. Effects of melatonin on lipid peroxidation induced by oxygen radicals. Life Sci. 1998, 62, 853–859. [Google Scholar] [CrossRef]
- Taysi, S.; Koc, M.; Büyükokuroǧlu, M.E.; Altinkaynak, K.; Şahin, Y.N. Melatonin reduces lipid peroxidation and nitric oxide during irradiation-induced oxidative injury in the rat liver. J. Pineal Res. 2003, 34, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Wakatsuki, A.; Okatani, Y.; Ikenoue, N.; Izumiya, C.; Kaneda, C. Melatonin inhibits oxidative modification of low-density lipoprotein particles in normolipidemic post-menopausal women. J. Pineal Res. 2000, 28, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Rosen, J.; Than, N.N.; Koch, D.; Poeggeler, B.; Laatsch, H.; Hardeland, R. Interactions of melatonin and its metabolites with the ABTS cation radical: Extension of the radical scavenger cascade and formation of a novel class of oxidation products, C2-substituted 3-indolinones. J. Pineal Res. 2006, 41, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Terron, M.P.; Flores, L.J.; Reiter, R.J. One molecule, many derivatives: A never-ending interaction of melatonin with reactive oxygen and nitrogen species? J. Pineal Res. 2007, 42, 28–42. [Google Scholar] [CrossRef] [PubMed]
- Joshi, N.; Biswas, J.; Nath, C.; Singh, S. Promising Role of Melatonin as Neuroprotectant in Neurodegenerative Pathology. Mol. Neurobiol. 2015, 52, 330–340. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.; Morel, A.; Saso, L.; Saluk, J. Melatonin redox activity. Its potential clinical applications in neurodegenerative disorders. Curr. Top. Med. Chem. 2015, 15, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Olcese, J.M.; Cao, C.; Mori, T.; Mamcarz, M.B.; Maxwell, A.; Runfeldt, M.J.; Wang, L.; Zhang, C.; Lin, X.; Zhang, G.; et al. Protection against cognitive deficits and markers of neurodegeneration by long-term oral administration of melatonin in a transgenic model of Alzheimer disease. J. Pineal Res. 2009, 47, 82–96. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.; Huang, F.; Fan, W.; Cheng, S.; Chen, Y.; Zhang, W.; Shi, H.; He, H. Differential effects of melatonin on amyloid-β peptide 25–35-induced mitochondrial dysfunction in hippocampal neurons at different stages of culture. J. Pineal Res. 2010, 48, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Stefanova, N.A.; Maksimova, K.Y.; Kiseleva, E.; Rudnitskaya, E.A.; Muraleva, N.A.; Kolosova, N.G. Melatonin attenuates impairments of structural hippocampal neuroplasticity in OXYS rats during active progression of Alzheimer’s disease-like pathology. J. Pineal Res. 2015, 59, 163–177. [Google Scholar] [CrossRef] [PubMed]
- Rudnitskaya, E.A.; Muraleva, N.A.; Maksimova, K.Y.; Kiseleva, E.; Kolosova, N.G.; Stefanova, N.A. Melatonin Attenuates Memory Impairment, Amyloid-β Accumulation, and Neurodegeneration in a Rat Model of Sporadic Alzheimer’s Disease. J. Alzheimer’s Dis. 2015, 47, 103–116. [Google Scholar] [CrossRef] [PubMed]
- Ali, T.; Kim, M.O. Melatonin ameliorates amyloid beta-induced memory deficits, tau hyperphosphorylation and neurodegeneration via PI3/Akt/GSk3β pathway in the mouse hippocampus. J. Pineal Res. 2015, 59, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Brusco, L.I.; Márquez, M.; Cardinali, D.P. Monozygotic twins with Alzheimer’s disease treated with melatonin: Case report. J. Pineal Res. 1998, 25, 260–263. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.H.; Swaab, D.F. The human pineal gland and melatonin in aging and Alzheimer’s disease. J. Pineal Res. 2005, 38, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Carriere, C.H.; Kang, N.H.; Niles, L.P. Chronic low-dose melatonin treatment maintains nigrostriatal integrity in an intrastriatal rotenone model of Parkinson’s disease. Brain Res. 2016, 1633, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Yildirim, F.B.; Ozsoy, O.; Tanriover, G.; Kaya, Y.; Ogut, E.; Gemici, B.; Dilmac, S.; Ozkan, A.; Agar, A.; Aslan, M. Mechanism of the beneficial effect of melatonin in experimental Parkinson’s disease. Neurochem. Int. 2014, 79, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bush, S.H.; Lacaze-Masmonteil, N.; McNamara-Kilian, M.T.; MacDonald, A.R.; Tierney, S.; Momoli, F.; Agar, M.; Currow, D.C.; Lawlor, P.G. The preventative role of exogenous melatonin administration to patients with advanced cancer who are at risk of delirium: Study protocol for a randomized controlled trial. Trials 2016, 17, 399. [Google Scholar] [CrossRef] [PubMed]
- Innominato, P.F.; Lim, A.S.; Palesh, O.; Clemons, M.; Trudeau, M.; Eisen, A.; Wang, C.; Kiss, A.; Pritchard, K.I.; Bjarnason, G.A. The effect of melatonin on sleep and quality of life in patients with advanced breast cancer. Support. Care Cancer 2016, 24, 1097–1105. [Google Scholar] [CrossRef] [PubMed]
- Nooshinfar, E.; Bashash, D.; Safaroghli-Azar, A.; Bayati, S.; Rezaei-Tavirani, M.; Ghaffari, S.H.; Akbari, M.E. Melatonin promotes ATO-induced apoptosis in MCF-7 cells: Proposing novel therapeutic potential for breast cancer. Biomed. Pharmacother. 2016, 83, 456–465. [Google Scholar] [CrossRef] [PubMed]
- Sabzichi, M.; Samadi, N.; Mohammadian, J.; Hamishehkar, H.; Akbarzadeh, M.; Molavi, O. Sustained release of melatonin: A novel approach in elevating efficacy of tamoxifen in breast cancer treatment. Colloids Surf. B Biointerfaces 2016, 145, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Koşar, P.A.; Nazıroğlu, M.; Övey, İ.S.; Çiğ, B. Synergic Effects of Doxorubicin and Melatonin on Apoptosis and Mitochondrial Oxidative Stress in MCF-7 Breast Cancer Cells: Involvement of TRPV1 Channels. J. Membr. Biol. 2016, 249, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Yang, Y.; Fan, C.; Han, J.; Wang, D.; Di, S.; Hu, W.; Liu, D.; Li, X.; Reiter, R.J.; et al. Melatonin as a potential anticarcinogen for non-small-cell lung cancer. Oncotarget 2016, 7, 46768–46784. [Google Scholar] [CrossRef] [PubMed]
- Akbarzadeh, M.; Nouri, M.; Banekohal, M.V.; Cheraghi, O.; Tajalli, H.; Movassaghpour, A.; Soltani, S.; Cheraghi, H.; Feizy, N.; Montazersaheb, S.; et al. Effects of combination of melatonin and laser irradiation on ovarian cancer cells and endothelial lineage viability. Lasers Med. Sci. 2016, 31, 1565–1572. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.V.; Narechania, M.B. The genotoxic effects of anti-cancer drug gossypol on human lymphocytes and its mitigation by melatonin. Drug Chem. Toxicol. 2016, 39, 357–361. [Google Scholar] [CrossRef] [PubMed]
- Borin, T.F.; Arbab, A.S.; Gelaleti, G.B.; Ferreira, L.C.; Moschetta, M.G.; Jardim-Perassi, B.V.; Iskander, A.; Varma, N.R.S.; Shankar, A.; Coimbra, V.B.; et al. Melatonin decreases breast cancer metastasis by modulating Rho-associated kinase protein-1 expression. J. Pineal Res. 2016, 60, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Paterniti, I.; Cordaro, M.; Esposito, E.; Cuzzocrea, S. The antioxidative property of melatonin against brain ischemia. Expert Rev. Neurother. 2016, 16, 841–848. [Google Scholar] [CrossRef] [PubMed]
- Aydemir, S.; Dogan, D.; Kocak, A.; Dilsiz, N. The effect of melatonin on spinal cord after ischemia in rats. Spinal Cord 2016, 54, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, P.; Pandey, A.K.; Paul, S.; Patnaik, R. Melatonin renders neuroprotection by protein kinase C mediated aquaporin-4 inhibition in animal model of focal cerebral ischemia. Life Sci. 2014, 100, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Kilic, U.; Yilmaz, B.; Reiter, R.J.; Yüksel, A.; Kilic, E. Effects of memantine and melatonin on signal transduction pathways vascular leakage and brain injury after focal cerebral ischemia in mice. Neuroscience 2013, 237, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Sun, Y.; Yi, W.; Li, Y.; Fan, C.; Xin, Z.; Jiang, S.; Di, S.; Qu, Y.; Reiter, R.J.; et al. A review of melatonin as a suitable antioxidant against myocardial ischemia-reperfusion injury and clinical heart diseases. J. Pineal Res. 2014, 57, 357–366. [Google Scholar] [CrossRef] [PubMed]
- De Souza, A.V.G.; Golim, M.A.; Deffune, E.; Domingues, M.A.C.; de Carvalho, L.R.; Vianna, I.G.; Castiglia, Y.M.M.; Vianna, P.T.G. Evaluation of renal protection from high doses of melatonin in an experimental model of renal ischemia and reperfusion in hyperglycemic rats. Transpl. Proc. 2014, 46, 1591–1593. [Google Scholar] [CrossRef] [PubMed]
- Deng, W.S.; Xu, Q.; Liu, Y.; Jiang, C.H.; Zhou, H.; Gu, L. Effects of melatonin on liver function and lipid peroxidation in a rat model of hepatic ischemia/reperfusion injury. Exp. Ther. Med. 2016, 11, 1955–1960. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yang, Y.; Feng, Y.; Yan, J.; Fan, C.; Jiang, S.; Qu, Y. A review of melatonin in hepatic ischemia/reperfusion injury and clinical liver disease. Ann. Med. 2014, 46, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Kireev, R.; Bitoun, S.; Cuesta, S.; Tejerina, A.; Ibarrola, C.; Moreno, E.; Vara, E.; Tresguerres, J.A.F. Melatonin treatment protects liver of Zucker rats after ischemia/reperfusion by diminishing oxidative stress and apoptosis. Eur. J. Pharmacol. 2013, 701, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Yip, H.K.; Chang, Y.C.; Wallace, C.G.; Chang, L.T.; Tsai, T.H.; Chen, Y.L.; Chang, H.W.; Leu, S.; Zhen, Y.Y.; Tsai, C.Y.; et al. Melatonin treatment improves adipose-derived mesenchymal stem cell therapy for acute lung ischemia-reperfusion injury. J. Pineal Res. 2013, 54, 207–221. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Liu, X.; Rosales-Corral, S.A.; Acuna-Castroviejo, D.; Reiter, R.J. Mitochondria and chloroplasts as the original sites of melatonin synthesis: A hypothesis related to melatonin’s primary function and evolution in eukaryotes. J. Pineal Res. 2013, 54, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.T.; Kleszczyński, K.; Semak, I.; Janjetovic, Z.; Żmijewski, M.A.; Kim, T.K.; Slominski, R.M.; Reiter, R.J.; Fischer, T.W. Local melatoninergic system as the protector of skin integrity. Int. J. Mol. Sci. 2014, 15, 17705–17732. [Google Scholar] [CrossRef] [PubMed]
- Poeggeler, B.; Reiter, R.J.; Hardeland, R.; Sewerynek, E.; Melchiorri, D.; Barlow-Walden, L.R. Melatonin, a mediator of electron transfer and repair reactions, acts synergistically with the chain-breaking antioxidants ascorbate, trolox and glutathione. Neuroendocrinol. Lett. 1995, 17, 87–92. [Google Scholar]
- Gitto, E.; Tan, D.X.; Reiter, R.J.; Karbownik, M.; Manchester, L.C.; Cuzzocrea, S.; Fulia, F.; Barberi, I. Individual and synergistic antioxidative actions of melatonin: Studies with vitamin E, vitamin C, glutathione and desferrrioxamine (desferoxamine) in rat liver homogenates. J. Pharm. Pharmacol. 2001, 53, 1393–1401. [Google Scholar] [CrossRef] [PubMed]
- Sliwinski, T.; Rozej, W.; Morawiec-Bajda, A.; Morawiec, Z.; Reiter, R.; Blasiak, J. Protective action of melatonin against oxidative DNA damage-Chemical inactivation versus base-excision repair. Mutat. Res. 2007, 634, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Fischer, T.W.; Slominski, A.; Zmijewski, M.A.; Reiter, R.J.; Paus, R. Melatonin as a major skin protectant: From free radical scavenging to DNA damage repair. Exp. Dermatol. 2008, 17, 713–730. [Google Scholar] [CrossRef] [PubMed]
- Davanipour, Z.; Poulsen, H.E.; Weimann, A.; Sobel, E. Endogenous melatonin and oxidatively damaged guanine in DNA. BMC Endocr. Disord. 2009, 9, 22. [Google Scholar] [CrossRef] [PubMed]
- De Almeida, E.A.; Martinez, G.R.; Klitzke, C.F.; de Medeiros, M.H.G.; Di Mascio, P. Oxidation of melatonin by singlet molecular oxygen (O2(1Δg)) produces N1-acetyl-N2-formyl-5-methoxykynurenine. J. Pineal Res. 2003, 35, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Limson, J.; Nyokong, T.; Daya, S. The interaction of melatonin and its precursors with aluminium, cadmium, copper, iron, lead, and zinc: An adsorptive voltammetric study. J. Pineal Res. 1998, 24, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Zatta, P.; Tognon, G.; Carampin, P. Melatonin prevents free radical formation due to the interaction between β-amyloid peptides and metal ions [Al(III), Zn(II), Cu(II), Mn(II), Fe(II)]. J. Pineal Res. 2003, 35, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Mayo, J.C.; Tan, D.X.; Sainz, R.M.; Natarajan, M.; Lopez-Burillo, S.; Reiter, R.J. Protection against oxidative protein damage induced by metal-catalyzed reaction or alkylperoxyl radicals: Comparative effects of melatonin and other antioxidants. Biochim. Biophys. Acta 2003, 1620, 139–150. [Google Scholar] [CrossRef]
- Parmar, P.; Limson, J.; Nyokong, T.; Daya, S. Melatonin protects against copper-mediated free radical damage. J. Pineal Res. 2002, 32, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Romero, A.; Ramos, E.; de Los Ríos, C.; Egea, J.; del Pino, J.; Reiter, R.J. A review of metal-catalyzed molecular damage: Protection by melatonin. J. Pineal Res. 2014, 56, 343–370. [Google Scholar] [CrossRef] [PubMed]
- Moudache, M.; Colon, M.; Nerín, C.; Zaidi, F. Phenolic content and antioxidant activity of olive by-products and antioxidant film containing olive leaf extract. Food Chem. 2016, 212, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Saxena, M.; Saxena, J.; Pradhan, A. Flavonoids and phenolic acids as antioxidants in plants and human health. Int. J. Pharm. Sci. Rev. Res. 2012, 16, 130–134. [Google Scholar]
- Fernandez-Panchon, M.S.; Villano, D.; Troncoso, A.M.; Garcia-Parrilla, M.C. Antioxidant activity of phenolic compounds: From in vitro results to in vivo evidence. Crit. Rev. Food Sci. Nutr. 2008, 48, 649–671. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Mazzone, G.; Alvarez-Diduk, R.; Marino, T.; Alvarez-Idaboy, J.R.; Russo, N. Food Antioxidants: Chemical Insights at the Molecular Level. Annu. Rev. Food Sci. Technol. 2016, 7, 335–352. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Yu, H.; Shao, S.; Wang, S.; Guo, Y. Evaluating the antioxidant capacity of polyphenols with an off-on fluorescence probe and the mechanism study. Anal. Methods 2014, 6, 7149–7153. [Google Scholar] [CrossRef]
- Mendoza-Wilson, A.M.; Castro-Arredondo, S.I.; Balandrán-Quintana, R.R. Computational study of the structure-free radical scavenging relationship of procyanidins. Food Chem. 2014, 161, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Gao, Y.; Li, F.; Liang, A.; Xu, Z.; Bai, Y.; Mai, W.; Han, L.; Chen, D. Maclurin protects against hydroxyl radical-induced damages to mesenchymal stem cells: Antioxidant evaluation and mechanistic insight. Chem. Biol. Interact. 2014, 219, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Xue, Y.; An, L.; Zheng, Y.; Dou, Y.; Zhang, L.; Liu, Y. Theoretical study on the structural and antioxidant properties of some recently synthesised 2,4,5-trimethoxy chalcones. Food Chem. 2014, 171, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Praveena, R.; Sadasivam, K.; Deepha, V.; Sivakumar, R. Antioxidant potential of orientin: A combined experimental and DFT approach. J. Mol. Struct. 2014, 1061, 114–123. [Google Scholar] [CrossRef]
- Mikulski, D.; Eder, K.; Molski, M. Quantum-chemical study on relationship between structure and antioxidant properties of hepatoprotective compounds occurring in Cynara scolymus and Silybum marianum. J. Theor. Comput. Chem. 2014, 13, 1450004. [Google Scholar] [CrossRef]
- Galano, A.; Martínez, A. Capsaicin, a tasty free radical scavenger: Mechanism of action and kinetics. J. Phys. Chem. B 2012, 116, 1200–1208. [Google Scholar] [CrossRef] [PubMed]
- Martínez, A.; Galano, A.; Vargas, R. Free radical scavenger properties of α-mangostin: Thermodynamics and kinetics of HAT and RAF mechanisms. J. Phys. Chem. B 2011, 115, 12591–12598. [Google Scholar] [CrossRef] [PubMed]
- Dimitrić Marković, J.M.; Milenković, D.; Amić, D.; Mojović, M.; Pašti, I.; Marković, Z.S. The preferred radical scavenging mechanisms of fisetin and baicalein towards oxygen-centred radicals in polar protic and polar aprotic solvents. RSC Adv. 2014, 4, 32228–32236. [Google Scholar] [CrossRef]
- Mazzone, G.; Toscano, M.; Russo, N. Density functional predictions of antioxidant activity and UV spectral features of nasutin A, isonasutin, ellagic acid, and one of its possible derivatives. J. Agric. Food Chem. 2013, 61, 9650–9657. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Zheng, Y.; An, L.; Zhang, L.; Qian, Y.; Yu, D.; Gong, X.; Liu, Y. A theoretical study of the structure-radical scavenging activity of hydroxychalcones. Comput. Theor. Chem. 2012, 982, 74–83. [Google Scholar] [CrossRef]
- Jeremić, S.; Filipović, N.; Peulić, A.; Marković, Z. Thermodynamical aspect of radical scavenging activity of alizarin and alizarin red S. Theoretical comparative study. Comput. Theor. Chem. 2014, 1047, 15–21. [Google Scholar] [CrossRef]
- Xue, Y.; Zheng, Y.; An, L.; Dou, Y.; Liu, Y. Density functional theory study of the structure-antioxidant activity of polyphenolic deoxybenzoins. Food Chem. 2014, 151, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Medina, M.E.; Galano, A.; Alvarez-Idaboy, J.R. Theoretical study on the peroxyl radicals scavenging activity of esculetin and its regeneration in aqueous solution. Phys. Chem. Chem. Phys. 2014, 16, 1197–1207. [Google Scholar] [CrossRef] [PubMed]
- Marković, Z.; Crossed D Signorović, J.; Dimitrić Marković, J.M.; Živić, M.; Amić, D. Investigation of the radical scavenging potency of hydroxybenzoic acids and their carboxylate anions. Monatsh. Chem. 2014, 145, 953–962. [Google Scholar] [CrossRef]
- Pérez-González, A.; Galano, A.; Alvarez-Idaboy, J.R. Dihydroxybenzoic acids as free radical scavengers: Mechanisms, kinetics, and trends in activity. New J. Chem. 2014, 38, 2639–2652. [Google Scholar] [CrossRef]
- Medina, M.E.; Iuga, C.; Álvarez-Idaboy, J.R. Antioxidant activity of fraxetin and its regeneration in aqueous media. A density functional theory study. RSC Adv. 2014, 4, 52920–52932. [Google Scholar] [CrossRef]
- Caicedo, C.; Iuga, C.; Castañeda-Arriaga, R.; Alvarez-Idaboy, J.R. Antioxidant activity of selected natural polyphenolic compounds from soybean via peroxyl radical scavenging. RSC Adv. 2014, 4, 38918–38930. [Google Scholar] [CrossRef]
- Iuga, C.; Alvarez-Idaboy, J.R.; Russo, N. Antioxidant activity of trans-resveratrol toward hydroxyl and hydroperoxyl radicals: A quantum chemical and computational kinetics study. J. Org. Chem. 2012, 77, 3868–3877. [Google Scholar] [CrossRef] [PubMed]
- Cordova-Gomez, M.; Galano, A.; Alvarez-Idaboy, J.R. Piceatannol, a better peroxyl radical scavenger than resveratrol. RSC Adv. 2013, 3, 20209–20218. [Google Scholar] [CrossRef]
- Shang, Y.J.; Qian, Y.P.; Liu, X.D.; Dai, F.; Shang, X.L.; Jia, W.Q.; Liu, Q.; Fang, J.G.; Zhou, B. Radical-scavenging activity and mechanism of resveratrol-oriented analogues: Influence of the solvent, radical, and substitution. J. Org. Chem. 2009, 74, 5025–5031. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Zhang, L.; Li, Y.; Yu, D.; Zheng, Y.; An, L.; Gong, X.; Liu, Y. A DFT study on the structure and radical scavenging activity of newly synthesized hydroxychalcones. J. Phys. Org. Chem. 2013, 26, 240–248. [Google Scholar] [CrossRef]
- Marković, Z.; Milenković, D.; Orović, J.; Dimitrić Marković, J.M.; Stepanić, V.; Lučić, B.; Amić, D. Free radical scavenging activity of morin 2′-O-phenoxide anion. Food Chem. 2012, 135, 2070–2077. [Google Scholar] [CrossRef] [PubMed]
- Di Meo, F.; Lemaur, V.; Cornil, J.; Lazzaroni, R.; Duroux, J.L.; Olivier, Y.; Trouillas, P. Free radical scavenging by natural polyphenols: Atom versus electron transfer. J. Phys. Chem. A 2013, 117, 2082–2092. [Google Scholar] [CrossRef] [PubMed]
- Mazzone, G.; Galano, A.; Alvarez-Idaboy, J.R.; Russo, N. Coumarin-Chalcone Hybrids as Peroxyl Radical Scavengers: Kinetics and Mechanisms. J. Chem. Inf. Model. 2016, 56, 662–670. [Google Scholar] [CrossRef] [PubMed]
- Villuendas-Rey, Y.; Alvarez-Idaboy, J.R.; Galano, A. Assessing the protective activity of a recently discovered phenolic compound against oxidative stress using computational chemistry. J. Chem. Inf. Model. 2015, 55, 2552–2561. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Sánchez, R.D.; Mendoza-Wilson, A.M.; Torrescano-Urrutia, G.R.; Sánchez-Escalante, A. Antiradical potential of phenolic compounds fingerprints of propolis extracts: DFT approach. Comp. Theor. Chem. 2015, 1066, 7–13. [Google Scholar] [CrossRef]
- Kicel, A.; Michel, P.; Owczarek, A.; Marchelak, A.; Zyzelewicz, D.; Budryn, G.; Oracz, J.; Anna Olszewska, M. Phenolic profile and antioxidant potential of leaves from selected Cotoneaster Medik. Species. Molecules 2016, 21, 688. [Google Scholar] [CrossRef] [PubMed]
- Medina, M.E.; Iuga, C.; Alvarez-Idaboy, J.R. Antioxidant activity of propyl gallate in aqueous and lipid media: A theoretical study. Phys. Chem. Chem. Phys. 2013, 15, 13137–13146. [Google Scholar] [CrossRef] [PubMed]
- Dimitrić Marković, J.M.; Milenković, D.; Amić, D.; Popović-Bijelić, A.; Mojović, M.; Pašti, I.A.; Marković, Z.S. Energy requirements of the reactions of kaempferol and selected radical species in different media: Towards the prediction of the possible radical scavenging mechanisms. Struct. Chem. 2014, 25, 1795–1804. [Google Scholar] [CrossRef]
- Inagaki, T.; Yamamoto, T. Critical role of deep hydrogen tunneling to accelerate the antioxidant reaction of ubiquinol and vitamin e. J. Phys. Chem. B 2014, 118, 937–950. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Liu, W.; Peng, C.; Ren, Q.; Lu, W.; Deng, W. A DFT study on reaction of eupatilin with hydroxyl radical in solution. Int. J. Quantum Chem 2013, 113, 966–974. [Google Scholar] [CrossRef]
- Amorati, R.; Baschieri, A.; Morroni, G.; Gambino, R.; Valgimigli, L. Peroxyl Radical Reactions in Water Solution: A Gym for Proton-Coupled Electron-Transfer Theories. Chem. Eur. J. 2016, 22, 7924–7934. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, T.; Uno, B. Importance of proton-coupled electron transfer from natural phenolic compounds in superoxide scavenging. Chem. Pharm. Bull. 2015, 63, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Urbaniak, A.; Szelag, M.; Molski, M. Theoretical investigation of stereochemistry and solvent influence on antioxidant activity of ferulic acid. Comput. Theor. Chem. 2013, 1012, 33–40. [Google Scholar] [CrossRef]
- Fifen, J.J.; Nsangou, M.; Dhaouadi, Z.; Motapon, O.; Jaidane, N. Solvent effects on the antioxidant activity of 3,4-dihydroxyphenylpyruvic acid: DFT and TD-DFT studies. Comput. Theor. Chem. 2011, 966, 232–243. [Google Scholar] [CrossRef]
- Benayahoum, A.; Amira-Guebailia, H.; Houache, O. Homolytic and heterolytic O-H bond cleavage in trans-resveratrol and some phenantrene analogs: A theoretical study. Comput. Theor. Chem. 2014, 1037, 1–9. [Google Scholar] [CrossRef]
- Qian, Y.P.; Shang, Y.J.; Teng, Q.F.; Chang, J.; Fan, G.J.; Wei, X.; Li, R.R.; Li, H.P.; Yao, X.J.; Dai, F.; et al. Hydroxychalcones as potent antioxidants: Structure-activity relationship analysis and mechanism considerations. Food Chem. 2011, 126, 241–248. [Google Scholar] [CrossRef]
- Musialik, M.; Kuzmicz, R.; Pawlowski, T.S.; Litwinienko, G. Acidity of hydroxyl groups: An overlooked influence on antiradical properties of flavonoids. J. Org. Chem. 2009, 74, 2699–2709. [Google Scholar] [CrossRef] [PubMed]
- Martínez, A.; Hernández-Marin, E.; Galano, A. Xanthones as antioxidants: A theoretical study on the thermodynamics and kinetics of the single electron transfer mechanism. Food Funct. 2012, 3, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Najafi, M. On the antioxidant activity of ortho- and meta-substituted indolin-2-one derivatives. Monatsh. Chem. 2014, 145, 291–299. [Google Scholar] [CrossRef]
- Najafi, M. On the antioxidant activity of the Ortho and Meta substituted Daidzein derivatives in the gas phase and solvent environment. J. Mex. Chem. Soc. 2014, 58, 36–45. [Google Scholar]
- Dorović, J.; Marković, J.M.D.; Stepanić, V.; Begović, N.; Amić, D.; Marković, Z. Influence of different free radicals on scavenging potency of gallic acid. J. Mol. Model. 2014, 20, 2345. [Google Scholar] [CrossRef] [PubMed]
- Marković, Z.; Đorović, J.; Dekić, M.; Radulović, M.; Marković, S.; Ilić, M. DFT study of free radical scavenging activity of erodiol. Chem. Pap. 2013, 67, 1453–1461. [Google Scholar] [CrossRef]
- Van Wenum, E.; Jurczakowski, R.; Litwinienko, G. Media effects on the mechanism of antioxidant action of silybin and 2,3-dehydrosilybin: Role of the enol group. J. Org. Chem. 2013, 78, 9102–9112. [Google Scholar] [CrossRef] [PubMed]
- Farmanzadeh, D.; Najafi, M. Antioxidant activity of aminothiazol hydroxycoumarin derivatives. J. Theor. Comput. Chem. 2013, 12, 1350058. [Google Scholar] [CrossRef]
- Lengyel, J.; Rimarčík, J.; Vagánek, A.; Klein, E. On the radical scavenging activity of isoflavones: Thermodynamics of O-H bond cleavage. Phys. Chem. Chem. Phys. 2013, 15, 10895–10903. [Google Scholar] [CrossRef] [PubMed]
- Senthil kumar, K.; Kumaresan, R. A DFT study on the structural, electronic properties and radical scavenging mechanisms of calycosin, glycitein, pratensein and prunetin. Comput. Theor. Chem. 2012, 985, 14–22. [Google Scholar] [CrossRef]
- Alberto, M.E.; Russo, N.; Grand, A.; Galano, A. A physicochemical examination of the free radical scavenging activity of Trolox: Mechanism, kinetics and influence of the environment. Phys. Chem. Chem. Phys. 2013, 15, 4642–4650. [Google Scholar] [CrossRef] [PubMed]
- Marković, Z.S.; Dimitrić Marković, J.M.; Milenković, D.; Filipović, N. Mechanistic study of the structure-activity relationship for the free radical scavenging activity of baicalein. J. Mol. Model. 2011, 17, 2575–2584. [Google Scholar] [CrossRef] [PubMed]
- Jeremić, S.R.; Šehović, S.F.; Manojlović, N.T.; Marković, Z.S. Antioxidant and free radical scavenging activity of purpurin. Monatsh. Chem. 2012, 143, 427–435. [Google Scholar] [CrossRef]
- Mendoza-Sarmiento, G.; Rojas-Hernández, A.; Galano, A.; Gutiérrez, A. A combined experimental-theoretical study of the acid-base behavior of mangiferin: Implications for its antioxidant activity. RSC Adv. 2016, 6, 51171–51182. [Google Scholar] [CrossRef]
- Papadopoulos, A.G.; Nenadis, N.; Sigalas, M.P. DFT study of radical scavenging activity of sesame oil lignans and selected in vivo metabolites of sesamin. Comput. Theor. Chem. 2016, 1077, 125–132. [Google Scholar] [CrossRef]
- Marković, Z.; Đorović, J.; Petrović, Z.D.; Petrović, V.P.; Simijonović, D. Investigation of the antioxidant and radical scavenging activities of some phenolic Schiff bases with different free radicals. J. Mol. Model. 2015, 21, 293. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Alvarez-Idaboy, J.R.; Francisco-Márquez, M. Physicochemical insights on the free radical scavenging activity of sesamol: Importance of the acid/base equilibrium. J. Phys. Chem. B 2011, 115, 13101–13109. [Google Scholar] [CrossRef] [PubMed]
- Ouchi, A.; Nagaoka, S.I.; Abe, K.; Mukai, K. Kinetic study of the aroxyl radical-scavenging reaction of α-tocopherol in methanol solution: Notable effect of the alkali and alkaline earth metal salts on the reaction rates. J. Phys. Chem. B 2009, 113, 13322–13331. [Google Scholar] [CrossRef] [PubMed]
- Marković, Z.S.; Marković, S.; Dimitrić Marković, J.M.; Milenković, D. Structure and reactivity of baicalein radical cation. Int. J. Quantum Chem. 2012, 112, 2009–2017. [Google Scholar] [CrossRef]
- Marković, Z.; Amić, D.; Milenković, D.; Dimitrić-Marković, J.M.; Marković, S. Examination of the chemical behavior of the quercetin radical cation towards some bases. Phys. Chem. Chem. Phys. 2013, 15, 7370–7378. [Google Scholar] [CrossRef] [PubMed]
- Joshi, R.; Gangabhagirathi, R.; Venu, S.; Adhikari, S.; Mukherjee, T. Antioxidant activity and free radical scavenging reactions of gentisic acid: In Vitro and pulse radiolysis studies. Free Radic. Res. 2012, 46, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, I.; Ohkubo, K.; Miyazaki, K.; Hakamata, W.; Urano, S.; Ozawa, T.; Okuda, H.; Fukuzumi, S.; Ikota, N.; Fukuhara, K. A planar catechin analogue having a more negative oxidation potential than (+)-catechin as an electron transfer antioxidant against a peroxyl radical. Chem. Res. Toxicol. 2004, 17, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, I.; Shimada, T.; Ohkubo, K.; Manda, S.; Shimizu, T.; Urano, S.; Okuda, H.; Miyata, N.; Ozawa, T.; Anzai, K.; et al. Involvement of electron transfer in the radical-scavenging reaction of resveratrol. Chem. Lett. 2007, 36, 1276–1277. [Google Scholar] [CrossRef]
- Sueishi, Y.; Hori, M.; Kita, M.; Kotake, Y. Nitric oxide (NO) scavenging capacity of natural antioxidants. Food Chem. 2011, 129, 866–870. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Uribe, C.E.; Vallejo, W.; Castellar, W.; Trilleras, J.; Ortiz, S.; Rodriguez-Serrano, A.; Zarate, X.; Quiroga, J. Novel (E)-1-(pyrrole-2-yl)-3-(aryl)-2-(propen-1-one) derivatives as efficient singlet oxygen quenchers: Kinetics and quantum chemical calculations. RSC Adv. 2015, 5, 71565–71572. [Google Scholar] [CrossRef]
- Asano, M.; Iwahashi, H. Caffeic acid inhibits the formation of 7-carboxyheptyl radicals from oleic acid under flavin mononucleotide photosensitization by scavenging singlet oxygen and quenching the excited state of flavin mononucleotide. Molecules 2014, 19, 12486–12499. [Google Scholar] [CrossRef] [PubMed]
- Ohara, K.; Doi, K.; Niizaki, Y.; Nagaoka, S.I. A time-resolved luminescence study on singlet oxygen quenching by hydroxycinnamic acids under acidic, neutral and basic conditions. J. Photochem. Photobiol. A Chem. 2012, 249, 1–8. [Google Scholar] [CrossRef]
- Mukai, K.; Ishikawa, E.; Ouchi, A.; Nagaoka, S.I.; Suzuki, T.; Izumisawa, K.; Koike, T. Kinetic study of the quenching reaction of singlet oxygen by α-, β-, γ-, δ-tocotrienols, and palm oil and soybean extracts in solution. Biosci. Biotechnol. Biochem. 2014, 78, 2089–2101. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.S.; Jung, M.Y. Protective activities of catechins on singlet oxygen induced photooxidation of α-terpinene in methanol: Structure and singlet oxygen quenching activity relationship. Food Sci. Biotechnol. 2013, 22, 249–256. [Google Scholar] [CrossRef]
- Jung, M.Y.; Choi, D.S. Electron spin resonance and luminescence spectroscopic observation and kinetic study of chemical and physical singlet oxygen quenching by resveratrol in methanol. J. Agric. Food Chem. 2010, 58, 11888–11895. [Google Scholar] [CrossRef] [PubMed]
- Chadha, R.; Mahal, H.S.; Mukherjee, T.; Kapoor, S. Evidence for a possible role of 3-hydroxyanthranilic acid as an antioxidant. J. Phys. Org. Chem. 2009, 22, 349–354. [Google Scholar] [CrossRef]
- Sitarek, P.; Skała, E.; Wysokińska, H.; Wielanek, M.; Szemraj, J.; Toma, M.; Śliwiński, T. The effect of leonurus sibiricus plant extracts on stimulating repair and protective activity against oxidative DNA damage in CHO cells and content of phenolic compounds. Oxid. Med. Cell. Longev. 2016, 2016, 5738193. [Google Scholar] [CrossRef] [PubMed]
- Chaudhari, G.M.; Mahajan, R.T. Comparative antioxidant activity of twenty traditional Indian medicinal plants and its correlation with total flavonoid and phenolic content. Int. J. Pharm. Sci. Rev. Res. 2015, 30, 105–111. [Google Scholar]
- Vasantha Rupasinghe, H.P.; Nair, S.V.G.; Robinson, R.A. Chemopreventive properties of fruit phenolic compounds and their possible mode of actions. Stud. Nat. Prod. Chem. 2014, 42, 229–266. [Google Scholar]
- Kabanda, M.M. A theoretical study of the antioxidant properties of phenolic acid amides investigated through the radical-scavenging and metal chelation mechanisms. Eur. Food Res. Technol. 2015, 241, 553–572. [Google Scholar] [CrossRef]
- Liu, H.; Cao, J.; Jiang, W. Evaluation and comparison of vitamin C, phenolic compounds, antioxidant properties and metal chelating activity of pulp and peel from selected peach cultivars. Food Sci. Technol. 2015, 63, 1042–1048. [Google Scholar] [CrossRef]
- Działo, M.; Mierziak, J.; Korzun, U.; Preisner, M.; Szopa, J.; Kulma, A. The potential of plant phenolics in prevention and therapy of skin disorders. Int. J. Mol. Sci. 2016, 17, 160. [Google Scholar] [CrossRef] [PubMed]
- Rangel-Huerta, O.D.; Pastor-Villaescusa, B.; Aguilera, C.M.; Gil, A. A systematic review of the efficacy of bioactive compounds in cardiovascular disease: Phenolic compounds. Nutrients 2015, 7, 5177–5216. [Google Scholar] [CrossRef] [PubMed]
- Bulotta, S.; Celano, M.; Lepore, S.M.; Montalcini, T.; Pujia, A.; Russo, D. Beneficial effects of the olive oil phenolic components oleuropein and hydroxytyrosol: Focus on protection against cardiovascular and metabolic diseases. J. Transl. Med. 2014, 12, 219. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Mateos, A.; Heiss, C.; Borges, G.; Crozier, A. Berry (poly)phenols and cardiovascular health. J. Agric. Food Chem. 2014, 62, 3842–3851. [Google Scholar] [CrossRef] [PubMed]
- Panda, V.; Laddha, A.; Nandave, M.; Srinath, S. Dietary Phenolic Acids of Macrotyloma uniflorum (Horse Gram) Protect the Rat Heart Against Isoproterenol-Induced Myocardial Infarction. Phytother. Res. 2016, 1146–1155. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Morató, J.; Xicota, L.; Fitó, M.; Farré, M.; Dierssen, M.; de La Torre, R. Potential role of olive oil phenolic compounds in the prevention of neurodegenerative diseases. Molecules 2015, 20, 4655–4680. [Google Scholar] [CrossRef] [PubMed]
- Rigacci, S.; Stefani, M. Nutraceuticals and amyloid neurodegenerative diseases: A focus on natural phenols. Expert Rev. Neurother. 2014, 15, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Ai, G.; Huang, Z.M.; Liu, Q.C.; Han, Y.Q.; Chen, X. The protective effect of total phenolics from Oenanthe Javanica on acute liver failure induced by d-galactosamine. J. Ethnopharmacol. 2016, 186, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Razavi-Azarkhiavi, K.; Iranshahy, M.; Sahebkar, A.; Shirani, K.; Karimi, G. The Protective Role of Phenolic Compounds Against Doxorubicin-induced Cardiotoxicity: A Comprehensive Review. Nutr. Cancer 2016, 68, 892–917. [Google Scholar] [CrossRef] [PubMed]
- Parkinson, L.; Keast, R. Oleocanthal, a Phenolic Derived from Virgin Olive Oil: A Review of the Beneficial Effects on Inflammatory Disease. Int. J. Mol. Sci. 2014, 15, 12323–12334. [Google Scholar] [CrossRef] [PubMed]
- Perron, N.R.; Brumaghim, J.L. A review of the antioxidant mechanisms of polyphenol compounds related to iron binding. Cell Biochem. Biophys. 2009, 53, 75–100. [Google Scholar] [CrossRef] [PubMed]
- Scalbert, A.; Williamson, G. Dietary intake and bioavailability of polyphenols. J. Nutr. 2000, 130 (Suppl. S8), 2073S–2085S. [Google Scholar]
- D’Archivio, M.; Filesi, C.; Varì, R.; Scazzocchio, B.; Masella, R. Bioavailability of the polyphenols: Status and controversies. Int. J. Mol. Sci. 2010, 11, 1321–1342. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Esteban-Zubero, E.; Zhou, Z.; Reiter, R.J. Melatonin as a potent and inducible endogenous antioxidant: Synthesis and metabolism. Molecules 2015, 20, 18886–18906. [Google Scholar] [CrossRef] [PubMed]
- Fischer, T.W.; Sweatman, T.W.; Semak, I.; Sayre, R.M.; Wortsman, J.; Slominski, A. Constitutive and UV-induced metabolism of melatonin in keratinocytes and cell-free systems. FASEB J. 2006, 20, E897–E907. [Google Scholar] [CrossRef] [PubMed]
- Byeon, Y.; Tan, D.X.; Reiter, R.J.; Back, K. Predominance of 2-hydroxymelatonin over melatonin in plants. J. Pineal Res. 2015, 59, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Ximenes, V.F.; Padovan, C.Z.; Carvalho, D.A.; Fernandes, J.R. Oxidation of melatonin by taurine chloramine. J. Pineal Res. 2010, 49, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Watts, S.W.; Morrison, S.F.; Davis, R.P.; Barman, S.M. Serotonin and blood pressure regulation. Pharmacol. Rev. 2012, 64, 359–388. [Google Scholar] [CrossRef] [PubMed]
- Turner, E.H.; Blackwell, A.D. 5-Hydroxytryptophan plus SSRIs for interferon-induced depression: Synergistic mechanisms for normalizing synaptic serotonin. Med. Hypotheses 2005, 65, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Birdsall, T.C. 5-Hydroxytryptophan: A Clinically-Effective Serotonin Precursor. Altern. Med. Rev. 1998, 3, 271–280. [Google Scholar] [PubMed]
- Turner, E.H.; Loftis, J.M.; Blackwell, A.D. Serotonin a la carte: Supplementation with the serotonin precursor 5-hydroxytryptophan. Pharmacol. Ther. 2006, 109, 325–338. [Google Scholar] [CrossRef] [PubMed]
- Kema, I.P.; De Vries, E.G.E.; Muskiet, F.A.J. Clinical chemistry of serotonin and metabolites. J. Chromatogr. B Biomed. Sci. Appl. 2000, 747, 33–48. [Google Scholar] [CrossRef]
- González-Gómez, D.; Lozano, M.; Fernández-León, M.F.; Ayuso, M.C.; Bernalte, M.J.; Rodríguez, A.B. Detection and quantification of melatonin and serotonin in eight Sweet Cherry cultivars (Prunus avium L.). Eur. Food Res. Technol. 2009, 229, 223–229. [Google Scholar] [CrossRef]
- Ramakrishna, A.; Giridhar, P.; Sankar, K.U.; Ravishankar, G.A. Melatonin and serotonin profiles in beans of Coffea species. J. Pineal Res. 2012, 52, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Ly, D.; Kang, K.; Choi, J.Y.; Ishihara, A.; Back, K.; Lee, S.G. HPLC analysis of serotonin, tryptamine, tyramine, and the hydroxycinnamic acid amides of serotonin and tyramine in food vegetables. J. Med. Food 2008, 11, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Kang, K.; Lee, K.; Back, K. Characterization of tryptamine 5-hydroxylase and serotonin synthesis in rice plants. Plant Cell Rep. 2007, 26, 2009–2015. [Google Scholar] [CrossRef] [PubMed]
- Olivier, B. Serotonin: A never-ending story. Eur. J. Pharmacol. 2015, 753, 2–18. [Google Scholar] [CrossRef] [PubMed]
- Gershon, M.D. 5-Hydroxytryptamine (serotonin) in the gastrointestinal tract. Curr. Opin. Endocrinol. Diabetes Obes. 2013, 20, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Nausheen, F.; Mohsin, H.; Lakhan, S.E. Neurotransmitters in hiccups. SpringerPlus 2016, 5, 1357. [Google Scholar] [CrossRef] [PubMed]
- Byeon, Y.; Back, K. Melatonin synthesis in rice seedlings in vivo is enhanced at high temperatures and under dark conditions due to increased serotonin N-acetyltransferase and N-acetylserotonin methyltransferase activities. J. Pineal Res. 2014, 56, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Chan, Z. The cysteine2/histidine2-type transcription factor ZINC FINGER of ARABIDOPSIS THALIANA 6-activated C-REPEAT-BINDING FACTOR pathway is essential for melatonin-mediated freezing stress resistance in Arabidopsis. J. Pineal Res. 2014, 57, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Byeon, Y.; Lee, H.Y.; Back, K. Chloroplastic and cytoplasmic overexpression of sheep serotonin N-acetyltransferase in transgenic rice plants is associated with low melatonin production despite high enzyme activity. J. Pineal Res. 2015, 58, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.Y.; Byeon, Y.; Tan, D.X.; Reiter, R.J.; Back, K. Arabidopsis serotonin N-acetyltransferase knockout mutant plants exhibit decreased melatonin and salicylic acid levels resulting in susceptibility to an avirulent pathogen. J. Pineal Res. 2015, 58, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Young, I.M.; Leone, R.M.; Francis, P.; Stovell, P.; Silman, R.E. Melatonin is metabolized to N-acetyl serotonin and 6-hydroxymelatonin in man. J. Clin. Endocrinol. Metab. 1985, 60, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Dellegar, S.M.; Murphy, S.A.; Bourne, A.E.; Dicesare, J.C.; Purser, G.H. Identification of the factors affecting the rate of deactivation of hypochlorous acid by melatonin. Biochem. Biophys. Res. Commun. 1999, 257, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Agozzino, P.; Avellone, G.; Bongiorno, D.; Ceraulo, L.; Filizzola, F.; Natoli, M.C.; Livrea, M.A.; Tesoriere, L. Melatonin: Structural characterization of its non-enzymatic mono-oxygenate metabolite. J. Pineal Res. 2003, 35, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Horstman, J.A.; Wrona, M.Z.; Dryhurst, G. Further insights into the reaction of melatonin with hydroxyl radical. Bioorg. Chem. 2002, 30, 371–382. [Google Scholar] [CrossRef]
- Semak, I.; Naumova, M.; Korik, E.; Terekhovich, V.; Wortsman, J.; Slominski, A. A novel metabolic pathway of melatonin: Oxidation by cytochrome C. Biochemistry 2005, 44, 9300–9307. [Google Scholar] [CrossRef] [PubMed]
- Byeon, Y.; Back, K. Molecular cloning of melatonin 2-hydroxylase responsible for 2-hydroxymelatonin production in rice (Oryza sativa). J. Pineal Res. 2015, 58, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Lahiri, D.K.; Ge, Y.W.; Sharman, E.H.; Bondy, S.C. Age-related changes in serum melatonin in mice: Higher levels of combined melatonin and 6-hydroxymelatonin sulfate in the cerebral cortex than serum, heart, liver and kidney tissues. J. Pineal Res. 2004, 36, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.K.; Lin, Z.; Tidwell, W.J.; Li, W.; Slominski, A.T. Melatonin and its metabolites accumulate in the human epidermis in vivo and inhibit proliferation and tyrosinase activity in epidermal melanocytes in vitro. Mol. Cell. Endocrinol. 2015, 404, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.K.; Kleszczynśki, K.; Janjetovic, Z.; Sweatman, T.; Lin, Z.; Li, W.; Reiter, R.J.; Fischer, T.W.; Slominski, A.T. Metabolism of melatonin and biological activity of intermediates of melatoninergic pathway in human skin cells. FASEB J. 2013, 27, 2742–2755. [Google Scholar] [CrossRef] [PubMed]
- Wiechmann, A.F.; Sherry, D.M. Role of Melatonin and its Receptors in the Vertebrate Retina. Int. Rev. Cell Mol. Biol. 2013, 300, 211–242. [Google Scholar] [PubMed]
- Pinato, L.; da Silveira Cruz-Machado, S.; Franco, D.G.; Campos, L.M.G.; Cecon, E.; Fernandes, P.A.C.M.; Bittencourt, J.C.; Markus, R.P. Selective protection of the cerebellum against intracerebroventricular LPS is mediated by local melatonin synthesis. Brain Struct. Funct. 2013, 220, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Cruz, M.H.C.; Leal, C.L.V.; Cruz, J.F.; Tan, D.X.; Reiter, R.J. Essential actions of melatonin in protecting the ovary from oxidative damage. Theriogenology 2014, 82, 925–932. [Google Scholar] [CrossRef] [PubMed]
- Acuña-Castroviejo, D.; Escames, G.; Venegas, C.; Díaz-Casado, M.E.; Lima-Cabello, E.; López, L.C.; Rosales-Corral, S.; Tan, D.X.; Reiter, R.J. Extrapineal melatonin: Sources, regulation, and potential functions. Cell. Mol. Life Sci. 2014, 71, 2997–3025. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Zhou, Z.; Cruz, M.H.C.; Fuentes-Broto, L.; Galano, A. Phytomelatonin: Assisting plants to survive and thrive. Molecules 2015, 20, 7396–7437. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.; McKain, B.W.; Madan-Khetarpal, S.; McGuire, M.; Diler, R.S.; Perel, J.M.; Vockley, J.; Brent, D.A. GTP-cyclohydrolase deficiency responsive to sapropterin and 5-HTP supplementation: Relief of treatment-refractory depression and suicidal behaviour. BMJ Case Rep. 2011, 2011, bcr0320113927. [Google Scholar] [PubMed]
- Dell’Osso, L.; Carmassi, C.; Mucci, F.; Marazziti, D. Depression, serotonin and tryptophan. Curr. Pharm. Des. 2016, 22, 949–954. [Google Scholar] [CrossRef] [PubMed]
- Abdala-Valencia, H.; Berdnikovs, S.; McCary, C.A.; Urick, D.; Mahadevia, R.; Marchese, M.E.; Swartz, K.; Wright, L.; Mutlu, G.M.; Cook-Mills, J.M. Inhibition of allergic inflammation by supplementation with 5-hydroxytryptophan. Am. J. Physiol. Lung Cell Mol. Physiol. 2012, 303, L642–L660. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhao, H.; Yang, X.; Xue, Q.; Cotten, J.F.; Feng, H.J. 5-Hydroxytryptophan, a precursor for serotonin synthesis, reduces seizure-induced respiratory arrest. Epilepsia 2016, 57, 1228–1235. [Google Scholar] [CrossRef] [PubMed]
- Bocchio, M.; McHugh, S.B.; Bannerman, D.M.; Sharp, T.; Capogna, M. Serotonin, amygdala and fear: Assembling the puzzle. Front. Neural Circuits 2016, 10, 24. [Google Scholar] [CrossRef] [PubMed]
- Lesch, K.P.; Waider, J. Serotonin in the Modulation of Neural Plasticity and Networks: Implications for Neurodevelopmental Disorders. Neuron 2012, 76, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Patrick, R.P.; Ames, B.N. Vitamin D and the omega-3 fatty acids control serotonin synthesis and action, part 2: Relevance for ADHD, bipolar disorder, schizophrenia, and impulsive behavior. FASEB J. 2015, 29, 2207–2222. [Google Scholar] [CrossRef] [PubMed]
- Lesch, K.P.; Araragi, N.; Waider, J.; van den Hove, D.; Gutknecht, L. Targeting brain serotonin synthesis: Insights into neurodevelopmental disorders with long-term outcomes related to negative emotionality, aggression and antisocial behaviour. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2012, 367, 2426–2443. [Google Scholar] [CrossRef] [PubMed]
- Sanfey, A.G. Social decision-making: Insights from game theory and neuroscience. Science 2007, 318, 598–602. [Google Scholar] [CrossRef] [PubMed]
- Way, B.M.; Laćan, G.; Fairbanks, L.A.; Melega, W.P. Architectonic distribution of the serotonin transporter within the orbitofrontal cortex of the vervet monkey. Neuroscience 2007, 148, 937–948. [Google Scholar] [CrossRef] [PubMed]
- Pagan, C.; Delorme, R.; Callebert, J.; Goubran-Botros, H.; Amsellem, F.; Drouot, X.; Boudebesse, C.; Dudal, K.L.; Ngo-Nguyen, N.; Laouamri, H.; et al. The serotonin-N-acetylserotonin-melatonin pathway as a biomarker for autism spectrum disorders. Transl. Psychiatry 2014, 4, e479. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.Y.; Byeon, Y.; Back, K. Melatonin as a signal molecule triggering defense responses against pathogen attack in Arabidopsis and tobacco. J. Pineal Res. 2014, 57, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Sun, Q.; Zhang, H.; Cao, Y.; Weeda, S.; Ren, S.; Guo, Y.D. Roles of melatonin in abiotic stress resistance in plants. J. Exp. Bot. 2015, 66, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Janjetovic, Z.; Nahmias, Z.P.; Hanna, S.; Jarrett, S.G.; Kim, T.K.; Reiter, R.J.; Slominski, A.T. Melatonin and its metabolites ameliorate ultraviolet B-induced damage in human epidermal keratinocytes. J. Pineal Res. 2014, 57, 90–102. [Google Scholar] [CrossRef] [PubMed]
- Calvo, J.R.; Reiter, R.J.; Garca, J.J.; Ortiz, G.G.; Tan, D.X.; Karbownik, M. Characterization of the protective effects of melatonin and related indoles against α-naphthylisothiocyanate-induced liver injury in rats. J. Cell. Biochem. 2001, 80, 461–470. [Google Scholar] [CrossRef]
- García, J.J.; Reiter, R.J.; Karbownik, M.; Calvo, J.R.; Ortiz, G.G.; Tan, D.X.; Martínez-Ballarín, E.; Acua-Castroviejo, D. N-acetylserotonin suppresses hepatic microsomal membrane rigidity associated with lipid peroxidation. Eur. J. Pharmacol. 2001, 428, 169–175. [Google Scholar] [CrossRef]
- García, J.J.; Lõpez-Pingarrõn, L.; Almeida-Souza, P.; Tres, A.; Escudero, P.; García-Gil, F.A.; Tan, D.X.; Reiter, R.J.; Ramírez, J.M.; Bernal-Pérez, M. Protective effects of melatonin in reducing oxidative stress and in preserving the fluidity of biological membranes: A review. J. Pineal Res. 2014, 56, 225–237. [Google Scholar] [CrossRef] [PubMed]
- García, J.J.; Reiter, R.J.; Guerrero, J.M.; Escames, G.; Yu, B.P.; Oh, C.S.; Muñoz-Hoyos, A. Melatonin prevents changes in microsomal membrane fluidity during induced lipid peroxidation. FEBS Lett. 1997, 408, 297–300. [Google Scholar] [CrossRef]
- Anderson, G.; Rodriguez, M. Multiple sclerosis: The role of melatonin and N-acetylserotonin. Mult. Scler. Relat. Disord. 2015, 4, 112–123. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Zheng, J.; Jiang, Z.; Shi, C.; Li, J.; Du, X.; Wang, H.; Jiang, J.; Wang, X. Protective effect of N-acetylserotonin against acute hepatic ischemia-reperfusion injury in mice. Int. J. Mol. Sci. 2013, 14, 17680–17693. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Medina, M.E.; Tan, D.X.; Reiter, R.J. Melatonin and its metabolites as copper chelating agents and their role in inhibiting oxidative stress: A physicochemical analysis. J. Pineal Res. 2015, 58, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Christen, S.; Peterhans, E.; Stocker, R. Antioxidant activities of some tryptophan metabolites: Possible implication for inflammatory diseases. Proc. Natl. Acad. Sci. USA 1990, 87, 2506–2510. [Google Scholar] [CrossRef] [PubMed]
- Cadenas, E.; Simic, M.G.; Sies, H. Antioxidant activity of 5-hydroxytryptophan, 5-hydroxyindole, and dopa against microsomal lipid peroxidation and its dependence on vitamin E. Free Radic. Res. 1989, 6, 11–17. [Google Scholar] [CrossRef]
- Millán-Plano, S.; Piedrafita, E.; Miana-Mena, F.J.; Fuentes-Broto, L.; Martínez-Ballarín, E.; López-Pingarrón, L.; Sáenz, M.A.; García, J.J. Melatonin and structurally-related compounds protect synaptosomal membranes from free radical damage. Int. J. Mol. Sci. 2010, 11, 312–328. [Google Scholar] [CrossRef] [PubMed]
- Weber, O.A.; Simeon, V. Tryptamine, 5-hydroxytryptamine and 5-hydroxytryptophan complexes of proton and some divalent metal ions. J. Inorg. Nucl. Chem. 1971, 33, 2097–2101. [Google Scholar] [CrossRef]
- Lysek, N.; Kinscherf, R.; Claus, R.; Lindel, T. l-5-hydroxytryptophan: Antioxidant and anti-apoptotic principle of the intertidal sponge Hymeniacidon heliophila. Z. Naturforsch. C 2003, 58, 568–572. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.J.; Lee, J.S.; Kim, J.M.; Lee, E.K.; Han, Y.K.; Kim, H.J.; Choi, J.; Ha, Y.M.; No, J.K.; Kim, Y.H.; et al. 5-hydroxytrytophan inhibits tert-butylhydroperoxide (t-BHP)-induced oxidative damage via the suppression of reactive species (RS) and nuclear factor-κB (NF-κB) activation on human fibroblast. J. Agric. Food Chem. 2010, 58, 6387–6394. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Gonzales, M.C.; Fuentes-Broto, L.; Martínez-Ballarín, E.; Miana-Mena, F.J.; Berzosa, C.; García-Gil, F.A.; Aranda, M.; García, J.J. Effects of tryptophan and 5-hydroxytryptophan on the hepatic cell membrane rigidity due to oxidative stress. J. Membr. Biol. 2009, 231, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.H.; Hsu, P.Y.; Meng, M.; Su, C.C. Supplement of 5-hydroxytryptophan before induction suppresses inflammation and collagen-induced arthritis. Arthritis Res. Ther. 2015, 17, 364. [Google Scholar] [CrossRef] [PubMed]
- Keithahn, C.; Lerchl, A. 5-Hydroxytryptophan is a more potent in vitro hydroxyl radical scavenger than melatonin or vitamin C. J. Pineal Res. 2005, 38, 62–66. [Google Scholar] [CrossRef] [PubMed]
- Herraiz, T.; Galisteo, J. Endogenous and dietary indoles: A class of antioxidants and radical scavengers in the ABTS assay. Free Radic. Res. 2004, 38, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Siddhuraju, P.; Becker, K. Studies on antioxidant activities of mucuna seed (Mucuna pruriens var utilis) extract and various non-protein amino/imino acids through in vitro models. J. Sci. Food Agric. 2003, 83, 1517–1524. [Google Scholar] [CrossRef]
- Noda, Y.; Mori, A.; Liburdy, R.; Packer, L. Melatonin and its precursors scavenge nitric oxide. J. Pineal Res. 1999, 27, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Derlacz, R.A.; Sliwinska, M.; Piekutowska, A.; Winiarska, K.; Drozak, J.; Bryla, J. Melatonin is more effective than taurine and 5-hydroxytryptophan against hyperglycemia-induced kidney-cortex tubules injury. J. Pineal Res. 2007, 42, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Daniels, W.M.U.; van Rensburg, S.J.; van Zyl, J.M.; van Der Walt, B.J.; Taljaard, J.J.F. Free radical scavenging effects of melatonin and serotonin: Possible mechanism. Neuroreport 1996, 7, 1593–1596. [Google Scholar] [CrossRef] [PubMed]
- Huether, G.; Fettkötter, I.; Keilhoff, G.; Wolf, G. Serotonin acts as a radical scavenger and is oxidized to a dimer during the respiratory burst of activated microglia. J. Neurochem. 1997, 69, 2096–2101. [Google Scholar] [CrossRef] [PubMed]
- Schuff-Werner, P.; Splettstoesser, W. Antioxidative properties of serotonin and the bactericidal function of polymorphonuclear phagocytes. Adv. Exp. Med. Biol. 2000, 467, 321–325. [Google Scholar]
- Sarikaya, S.B.Ö.; Gülçin, I. Radical scavenging and antioxidant capacity of serotonin. Curr. Bioact. Comp. 2013, 9, 143–152. [Google Scholar] [CrossRef]
- Gülçin, I. Measurement of antioxidant ability of melatonin and serotonin by the DMPD and CUPRAC methods as trolox equivalent. J. Enzyme Inhib. Med. Chem. 2008, 23, 871–876. [Google Scholar] [CrossRef] [PubMed]
- Betten, Å.; Dahlgren, C.; Hermodsson, S.; Hellstrand, K. Serotonin protects NK cells against oxidatively induced functional inhibition and apoptosis. J. Leukoc. Biol. 2001, 70, 65–72. [Google Scholar] [PubMed]
- Sang Soo, H.; Dong Hyun, K.; Suk Ha, L.; Yun Sang, K.; Chung Soo, L. Antioxidant effects of serotonin and l-DOPA on oxidative damages of brain synaptosomes. Korean J. Physiol. Pharmacol. 1999, 3, 147–155. [Google Scholar]
- Longoni, B.; Pryor, W.A.; Marchiafava, P. Inhibition of lipid peroxidation by N-acetylserotonin and its role in retinal physiology. Biochem. Biophys. Res. Commun. 1997, 233, 778–780. [Google Scholar] [CrossRef] [PubMed]
- Wölfler, A.; Abuja, P.M.; Schauenstein, K.; Liebmann, P.M. N-acetylserotonin is a better extra- and intracellular antioxidant than melatonin. FEBS Lett. 1999, 449, 206–210. [Google Scholar] [CrossRef]
- Zhou, H.; Wang, J.; Jiang, J.; Stavrovskaya, I.G.; Li, M.; Li, W.; Wu, Q.; Zhang, X.; Luo, C.; Zhou, S.; et al. N-acetyl-serotonin offers neuroprotection through inhibiting mitochondrial death pathways and autophagic activation in experimental models of ischemic injury. J. Neurosci. 2014, 34, 2967–2978. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Yu, S.; Jiang, Z.; Liang, C.; Yu, W.; Li, J.; Du, X.; Wang, H.; Gao, X.; Wang, X. N-acetyl-serotonin protects HepG2 cells from oxidative stress injury induced by hydrogen peroxide. Oxid. Med. Cell. Longev. 2014, 2014, 310504. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, L.M.; Macedo, D.S.; de Freitas, R.M.; de Albuquerque Oliveira, A.; Vasconcelos, S.M.M.; de Sousa, F.C.F.; de Barros Viana, G.S. Protective effects of N-acetylserotonin against 6-hydroxydopamine-induced neurotoxicity. Life Sci. 2005, 76, 2193–2202. [Google Scholar] [CrossRef] [PubMed]
- Bachurin, S.; Oxenkrug, G.F.; Lermontova, N.; Afanasiev, A.; Beznosko, B.; Vankin, G.; Shevtzova, E.; Mukhina, T.; Serkova, T. N-acetylserotonin, melatonin and their derivatives improve cognition and protect against β-amyloid-induced neurotoxicity. Ann. N. Y. Acad. Sci. 1999, 890, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Oxenkrug, G.; Requintina, P.; Bachurin, S. Antioxidant and antiaging activity of N-acetylserotonin and melatonin in the in vivo models. Ann. N. Y. Acad. Sci. 2001, 939, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Tosini, G.; Ye, K.; Iuvone, P.M. N-acetylserotonin: Neuroprotection, neurogenesis, and the sleepy brain. Neuroscientist 2012, 18, 645–653. [Google Scholar] [CrossRef] [PubMed]
- Chan, T.Y.; Tang, P.L. Characterization of the antioxidant effects of melatonin and related indoleamines in vitro. J. Pineal Res. 1996, 20, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Lezoualc’h, F.; Sparapani, M.; Behl, C. N-acetyl-serotonin (normelatonin) and melatonin protect neurons against oxidative challenges and suppress the activity of the transcription factor NF-κB. J. Pineal Res. 1998, 24, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Tang, G.Y.; Ip, A.K.; Siu, A.W. Pinoline and N-acetylserotonin reduce glutamate-induced lipid peroxidation in retinal homogenates. Neurosci. Lett. 2007, 412, 191–194. [Google Scholar] [CrossRef] [PubMed]
- Seeger, H.; Mueck, A.O.; Lippert, T.H. Effect of melatonin and metabolites on copper-mediated oxidation of low density lipoprotein. Br. J. Clin. Pharmacol. 1997, 44, 283–284. [Google Scholar] [CrossRef]
- Gozzo, A.; Lesieur, D.; Duriez, P.; Fruchart, J.C.; Teissier, E. Structure-activity relationships in a series of melatonin analogues with the low-density lipoprotein oxidation model. Free Radic. Biol. Med. 1999, 26, 1538–1543. [Google Scholar] [CrossRef]
- Siu, A.W.; Cheung, J.P.; To, C.H.; Chan, E.K.; Chan, J.K.; Cheung, J.C. N-acetyl-serotonin reduces copper(I) ion-induced lipid peroxidation in bovine retinal homogenates. Acta Ophthalmol. Scand. 2001, 79, 69–71. [Google Scholar] [CrossRef] [PubMed]
- Qi, W.; Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Siu, A.W.; Garcia, J.J. Increased levels of oxidatively damaged DNA induced by chromium(III) and H2O2: Protection by melatonin and related molecules. J. Pineal Res. 2000, 29, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Guajardo, M.H.; Terrasa, A.M.; Catalá, A. Protective effect of indoleamines on in vitro ascorbate-Fe 2+dependent lipid peroxidation of rod outer segment membranes of bovine retina. J. Pineal Res. 2003, 35, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Karbownik, M.; Gitto, E.; Lewiñski, A.; Reiter, R.J. Relative efficacies of indole antioxidants in reducing autoxidation and iron-induced lipid peroxidation in hamster testes. J. Cell. Biochem. 2001, 81, 693–699. [Google Scholar] [CrossRef] [PubMed]
- Oxenkrug, G. Antioxidant effects of N-acetylserotonin: Possible mechanisms and clinical implications. Ann. N. Y. Acad. Sci. 2005, 1053, 334–347. [Google Scholar] [CrossRef] [PubMed]
- Pierrefiche, G.; Topall, G.; Courboin, G.; Henriet, I.; Laborit, H. Antioxidant activity of melatonin in mice. Res. Commun. Chem. Pathol. Pharmacol. 1993, 80, 211–224. [Google Scholar] [PubMed]
- Maharaj, D.S.; Walker, R.B.; Glass, B.D.; Daya, S. 6-Hydroxymelatonin protects against cyanide induced oxidative stress in rat brain homogenates. J. Chem. Neuroanat. 2003, 26, 103–107. [Google Scholar] [CrossRef]
- Maharaj, D.S.; Limson, J.L.; Daya, S. 6-Hydroxymelatonin converts Fe(III) to Fe(II) and reduces iron-induced lipid peroxidation. Life Sci. 2003, 72, 1367–1375. [Google Scholar] [CrossRef]
- Maharaj, D.S.; Maharaj, H.; Antunes, E.M.; Maree, D.M.; Nyokong, T.; Glass, B.D.; Daya, S. 6-Hydroxymelatonin protects against quinolinic-acid-induced oxidative neurotoxicity in the rat hippocampus. J. Pharm. Pharmacol. 2005, 57, 877–881. [Google Scholar] [CrossRef] [PubMed]
- Maharaj, D.S.; Anoopkumar-Dukie, S.; Glass, B.D.; Antunes, E.M.; Lack, B.; Walker, R.B.; Daya, S. The identification of the UV degradants of melatonin and their ability to scavenge free radicals. J. Pineal Res. 2002, 32, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Maharaj, D.S.; Maharaj, H.; Daya, S.; Glass, B.D. Melatonin and 6-hydroxymelatonin protect against iron-induced neurotoxicity. J. Neurochem. 2006, 96, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Marenich, A.V.; Cramer, C.J.; Truhlar, D.G. Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J. Phys. Chem. B 2009, 113, 6378–6396. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Schultz, N.E.; Truhlar, D.G. Design of density functionals by combining the method of constraint satisfaction with parametrization for thermochemistry, thermochemical kinetics, and noncovalent interactions. J. Chem. Theory Comput. 2006, 2, 364–382. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Pérez-González, A. On the free radical scavenging mechanism of protocatechuic acid, regeneration of the catechol group in aqueous solution. Theor. Chem. Acc. 2012, 131, 1–13. [Google Scholar] [CrossRef]
- Galano, A.; Francisco Marquez, M.; Pérez-González, A. Ellagic acid: An unusually versatile protector against oxidative stress. Chem. Res. Toxicol. 2014, 27, 904–918. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Diduk, R.; Galano, A. Adrenaline and noradrenaline: Protectors against oxidative stress or molecular targets? J. Phys. Chem. B 2015, 119, 3479–3491. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Del. Rev. 2001, 46, 3–26. [Google Scholar] [CrossRef]
- Ghose, A.K.; Viswanadhan, V.N.; Wendoloski, J.J. A knowledge-based approach in designing combinatorial or medicinal chemistry libraries for drug discovery. 1. A qualitative and quantitative characterization of known drug databases. J. Comb. Chem. 1999, 1, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Sverdlov, R.L.; Khrishchanovich, A.V.; Brinkevich, S.D.; Shadyro, O.I. Interaction of tryptophan and its derivatives with oxygen-and nitrogen-centered radicals. High Energy Chem. 2015, 49, 83–91. [Google Scholar] [CrossRef]
- Sverdlov, R.L.; Brinkevich, S.D.; Shadyro, O.I. Interaction of tryptophan and related compounds with oxygen- and carbon-centered radicals. Free Radic. Res. 2014, 48, 1200–1205. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Naranjo, M.I.; Moyá, M.L.; Cantos-Villar, E.; Garcia-Parrilla, M.C. Comparative evaluation of the antioxidant activity of melatonin and related indoles. J. Food Compos. Anal. 2012, 28, 16–22. [Google Scholar] [CrossRef]
- Najafi, M.; Farmanzadeh, D.; Klein, E.; Zahedi, M. A Theoretical study on the enthalpies of homolytic and heterolytic N-H bond cleavage in substituted melatonins in the gas-phase and aqueous solution. Acta Chim. Slov. 2013, 60, 43–55. [Google Scholar] [PubMed]
- Pérez-González, A.; Alvarez-Idaboy, J.R.; Galano, A. Free-radical scavenging by tryptophan and its metabolites through electron transfer based processes. J. Mol. Model. 2015, 21, 213. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Diduk, R.; Galano, A.; Tan, D.X.; Reiter, R.J. The key role of the sequential proton loss electron transfer mechanism on the free radical scavenging activity of some melatonin-related compounds. Theor. Chem. Acc. 2016, 135, 1–5. [Google Scholar] [CrossRef]
- Frankel, E.N.; Huang, S.-W.; Kanner, J.; German, J.B. Interfacial Phenomena in the Evaluation of Antioxidants: Bulk Oils vs. Emulsions. J. Agric. Food Chem. 1994, 42, 1054–1059. [Google Scholar] [CrossRef]
- Frankel, E.N.; Huang, S.-W.; Aeschbach, R.; Prior, E. Antioxidant Activity of a Rosemary Extract and Its Constituents, Carnosic Acid, Carnosol, and Rosmarinic Acid, in Bulk Oil and Oil-in-Water Emulsion. J. Agric. Food Chem. 1996, 44, 131–135. [Google Scholar] [CrossRef]
- Simko, F.; Bednarova, K.R.; Krajcirovicova, K.; Hrenak, J.; Celec, P.; Kamodyova, N.; Gajdosechova, L.; Zorad, S.; Adamcova, M. Melatonin reduces cardiac remodeling and improves survival in rats with isoproterenol-induced heart failure. J. Pineal Res. 2014, 57, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Fagali, N.; Catalá, A. The antioxidant behaviour of melatonin and structural analogues during lipid peroxidation depends not only on their functional groups but also on the assay system. Biochem. Biophys. Res. Commun. 2012, 423, 873–877. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Murcia, M.A.; Chirico, S.; Aruoma, O.I. Free radicals and antioxidants in food and in vivo: What they do and how they work. Crit. Rev. Food Sci. Nutr. 1995, 35, 7–20. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Whiteman, M. Measuring reactive species and oxidative damage in vivo and in cell culture: How should you do it and what do the results mean? Br. J. Pharmacol. 2004, 142, 231–255. [Google Scholar] [CrossRef] [PubMed]
- Kawanishi, S.; Sakurai, H. Differential anti-lipid peroxidative activity of melatonin. Naturwissenschaften 2002, 89, 31–33. [Google Scholar] [CrossRef] [PubMed]
- Gaikwad, P.; Naik, G.H.; Priyadarsini, K.I.; Mohan, H.; Rao, B.S.M. Radiation induced oxidation of hydroxy indoles by NO•2 and Br•−2-radicals: Effect of pH. J. Phys. Org. Chem. 2011, 24, 657–662. [Google Scholar] [CrossRef]
- Matuszak, Z.; Reszka, K.J.; Chignell, C.F. Reaction of melatonin and related indoles with hydroxyl radicals: EPR and spin trapping investigations. Free Radic. Biol. Med. 1997, 23, 367–372. [Google Scholar] [CrossRef]
- Roberts, J.E.; Hu, D.N.; Wishart, J.F. Pulse radiolysis studies of melatonin and chloromelatonin. J. Photochem. Photobiol. B Biol. 1998, 42, 125–132. [Google Scholar] [CrossRef]
- Stasica, P.; Ulanski, P.; Rosiak, J.M. Reactions of melatonin with radicals in deoxygenated aqueous solution. J. Radioanal. Nucl. Chem. 1998, 232, 107–113. [Google Scholar] [CrossRef]
- Poeggeler, B.; Reiter, R.J.; Hardeland, R.; Tan, D.X.; Barlow-Walden, L.R. Melatonin and structurally-related, endogenous indoles act as potent electron donors and radical scavengers in vitro. Redox Rep. 1996, 2, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Chyan, Y.J.; Poeggeler, B.; Omar, R.A.; Chain, D.G.; Frangione, B.; Ghiso, J.; Pappolla, M.A. Potent neuroprotective properties against the Alzheimer β-amyloid by an endogenous melatonin-related indole structure, indole-3-propionic acid. J. Biol. Chem. 1999, 274, 21937–21942. [Google Scholar] [CrossRef] [PubMed]
- León-Carmona, J.R.; Alvarez-Idaboy, J.R.; Galano, A. On the peroxyl scavenging activity of hydroxycinnamic acid derivatives: Mechanisms, kinetics, and importance of the acid-base equilibrium. Phys. Chem. Chem. Phys. 2012, 14, 12534–12543. [Google Scholar] [CrossRef] [PubMed]
- Dorfman, L.M.; Adams, G.E. Reactivity of the Hydroxyl Radical in Aqueous Solutions; U.S. Department of Commerce, National Bureau of Standards: Washington, DC, USA, 1973.
- Wilson, W.E. A critical review of the gas phase reaction kinetics of the hydroxyl radical. J. Phys. Chem. Ref. Data 1972, 1, 535–573. [Google Scholar] [CrossRef]
- Gulcin, I.; Buyukokuroglu, M.E.; Kufrevioglu, O.I. Metal chelating and hydrogen peroxide scavenging effects of melatonin. J. Pineal Res. 2003, 34, 278–281. [Google Scholar] [CrossRef] [PubMed]
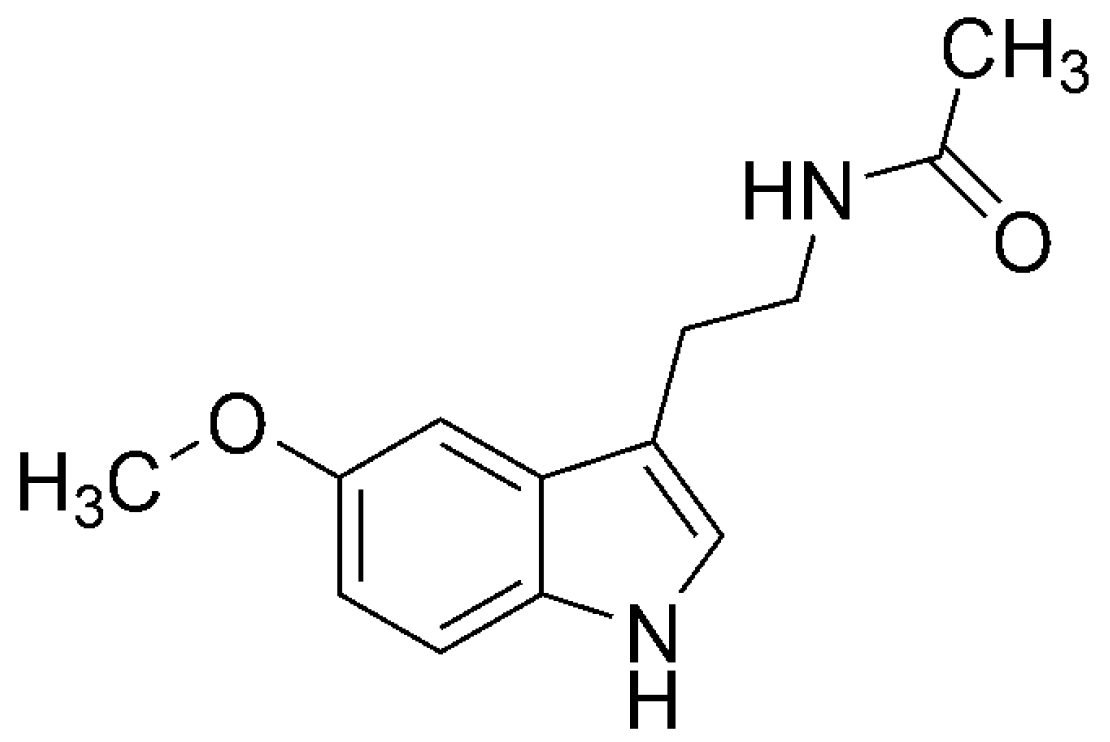
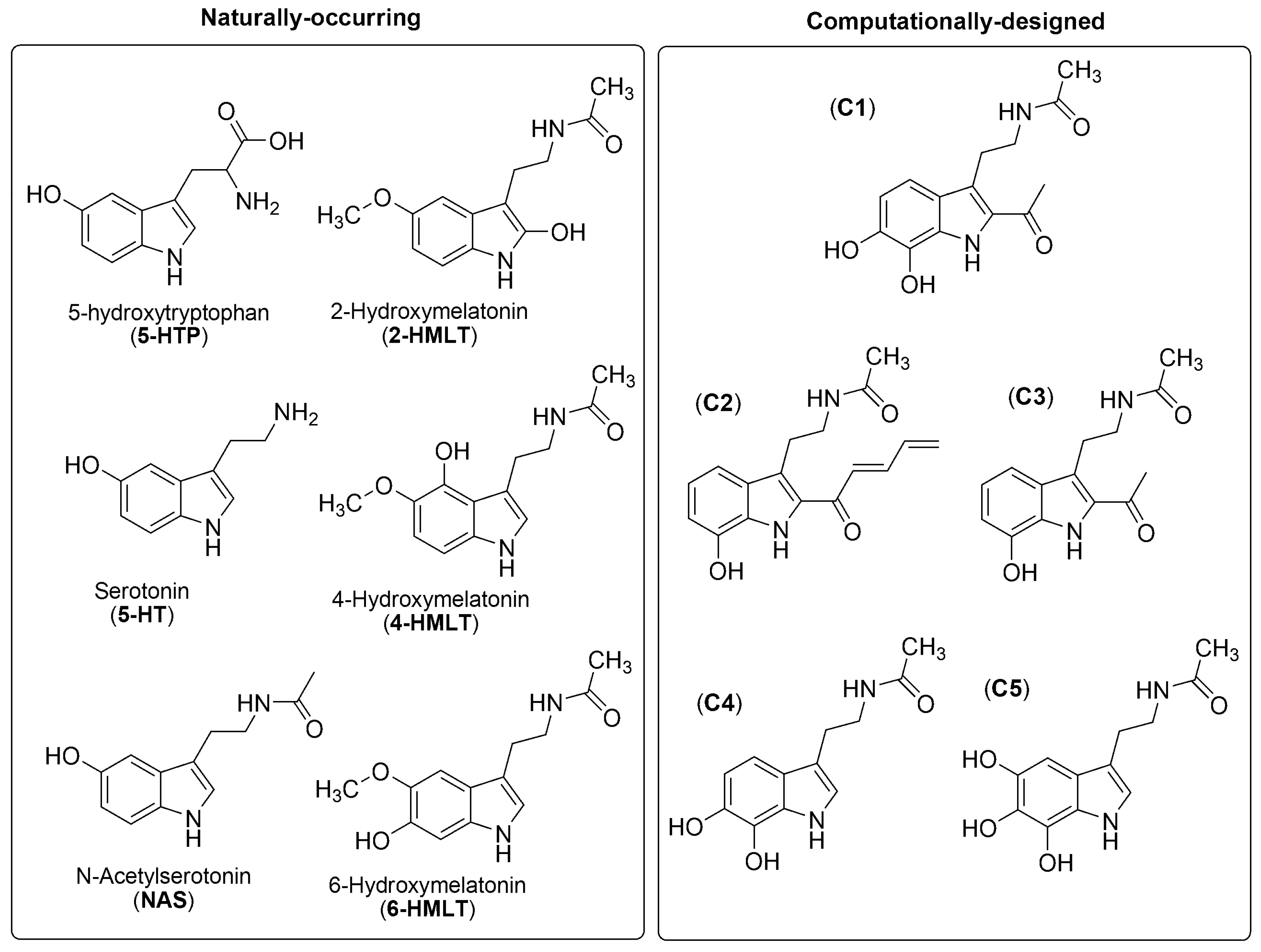
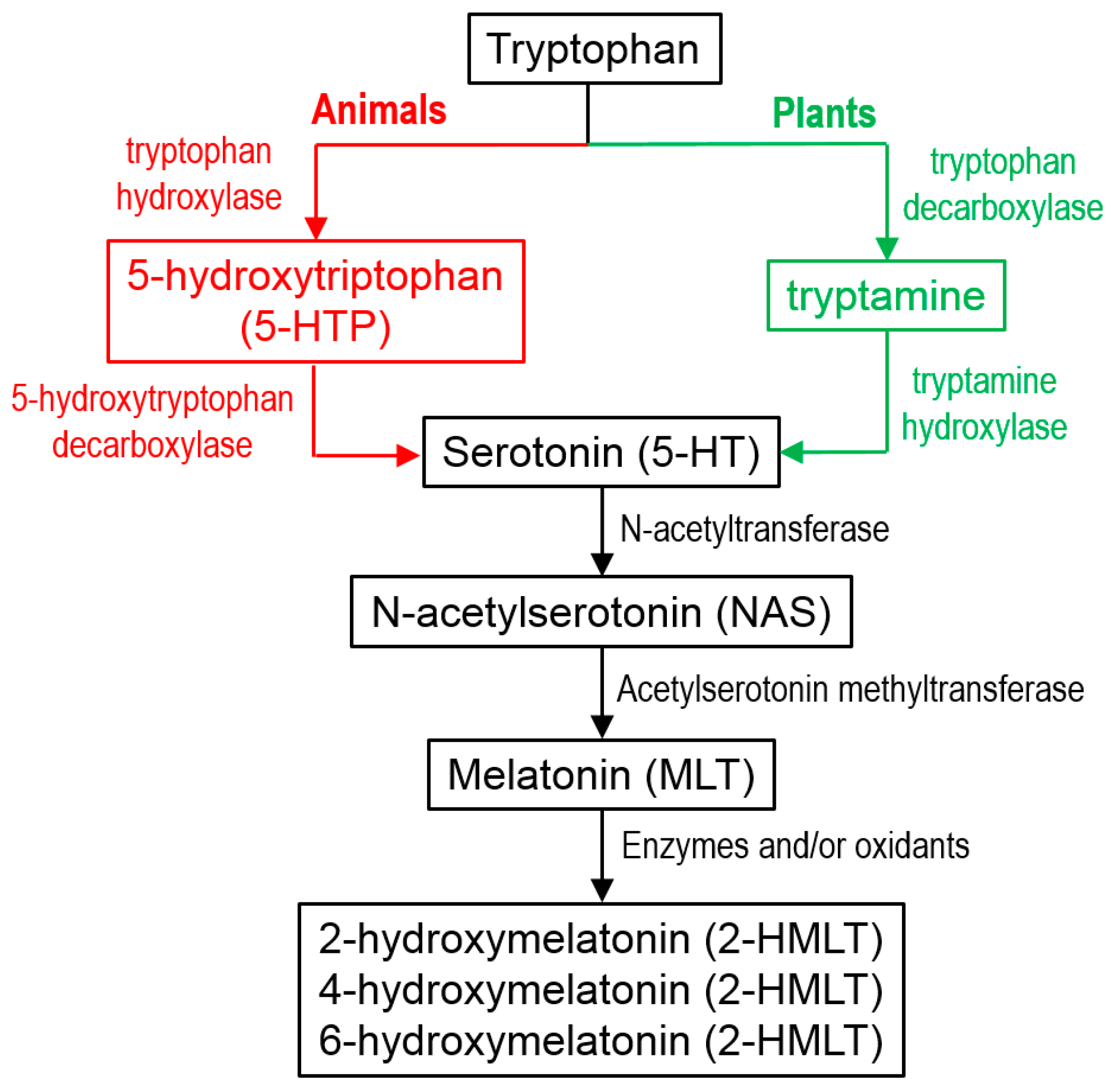
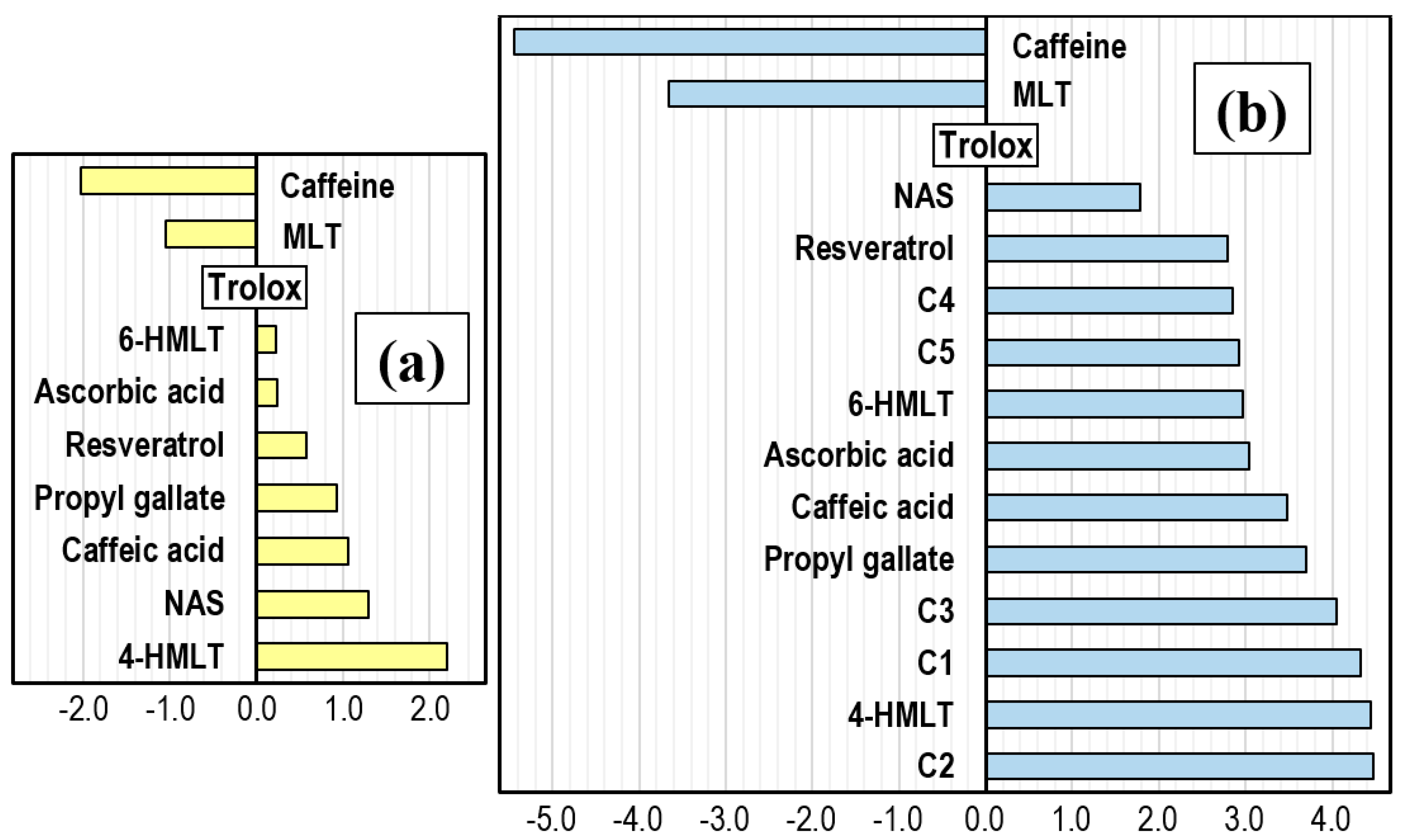
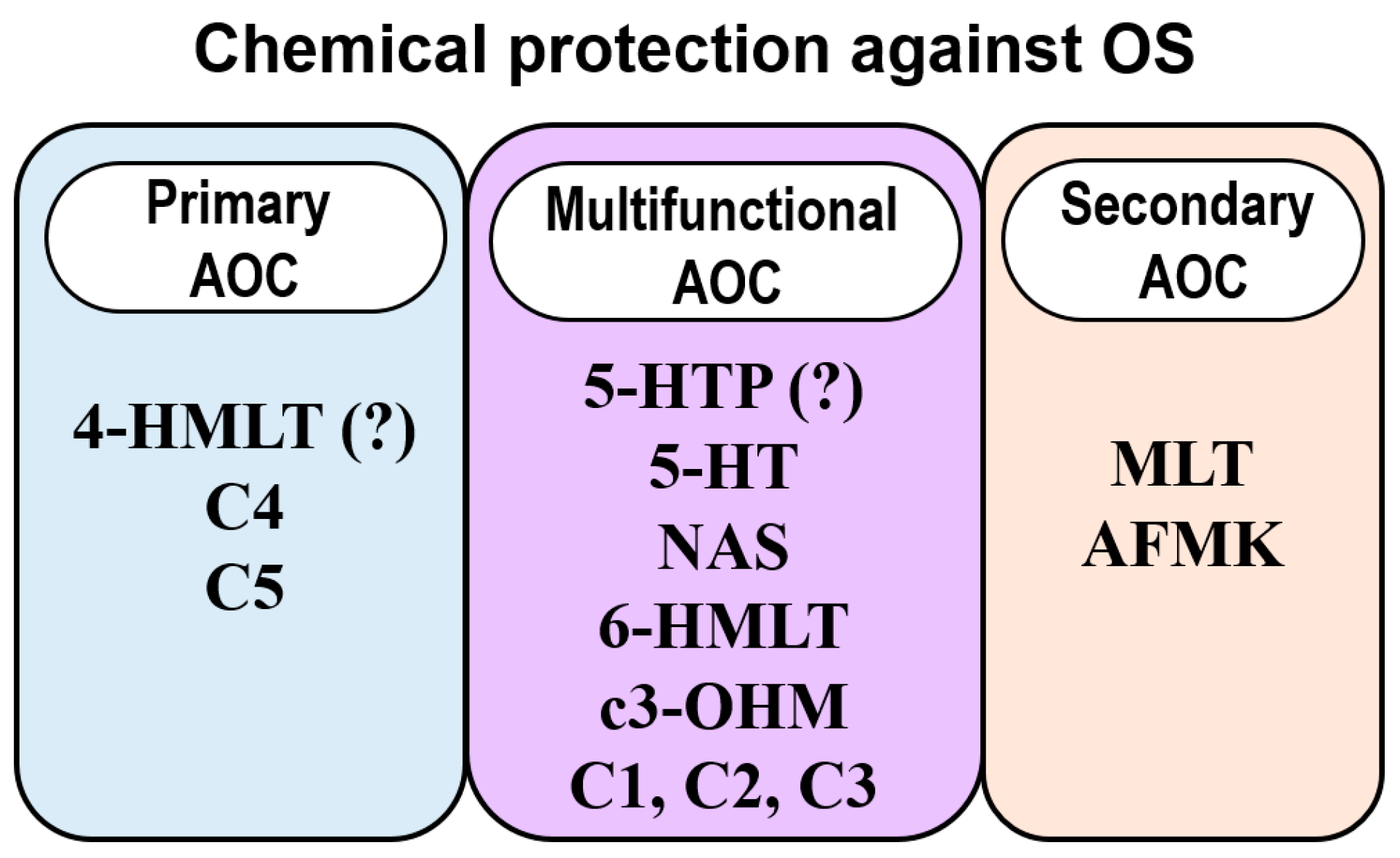
| ROS | RNS | RSS | |||
|---|---|---|---|---|---|
| Name | Acronym/Formulae | Name | Acronym/Formulae | Name | Acronym/Formulae |
| superoxide radical anion | O2•− | peroxynitrite | ONOO− | thiyl radicals | RS• |
| hydroxyl radical | •OH | nitric oxide | •NO | sulfenic acids | RSOH |
| alkoxyl radicals | RO• | nitrogen dioxide | •NO2 | disulfide-S-oxides | RS(O)2SR |
| peroxyl radicals | ROO• | ||||
| Name | Acronym | Chemical Reaction * |
|---|---|---|
| Radical Adduct Formation | RAF | HnAtx + •R → [HnAtx-R]• |
| Single Electron Transfer | SET | HnAtx + •R → HnAtx+• + R− (I) HnAtx + •R → HnAtx−• + R+ (II) |
| Hydrogen Atom Transfer | HAT | HnAtx + •R → Hn−1Atx• + HR |
| Proton Coupled Electron Transfer | PCET | HnAtx + •R → Hn−1Atx• + HR |
| Sequential Proton Loss Electron Transfer | SPLET | HnAtx → Hn−1Atx− + H+ Hn−1Atx− + •R → Hn−1Atx• + R− |
| Sequential Electron Proton Transfer | SEPT | HnAtx + •R → Hn−1Atx•+ + R− Hn−1Atx•+ → Hn−1Atx• + H+ |
| Antioxidant | FR | k (M−1·s−1) | Assay | Main Solvent | Reference |
|---|---|---|---|---|---|
| 5-HTP | •OH | 1.2 × 1010 | Experimental | not specified | [449] |
| 5-HTP | •NO2 | 9.0 × 105 | Experimental | water, pH = 5 | [450] |
| 5-HTP | •NO2 | 5.6 × 107 | Experimental | water, pH = 9 | [450] |
| 5-HTP | O2•− | 1.2 × 104 | Experimental | not specified | [449] |
| 5-HTP | DPPH• | ~7 × 10−1 | Experimental | methanol | [439] |
| NAS | DPPH• | ~2 × 100 | Experimental | methanol | [439] |
| NAS | HOO• | 6.70 × 104 | Theoretical | lipid | [43] |
| NAS | HOO• | 1.17 × 106 | Theoretical | water, pH = 7.4 | [53] |
| 6-HMLT | •OH | 1.1 × 1010 | Experimental | water, pH = 7 | [451] |
| 6-HMLT | O2•− | 2.7 × 104 | Experimental | not specified | [449] |
| 6-HMLT | HOO• | 5.81 × 103 | Theoretical | lipid | [43] |
| 6-HMLT | HOO• | 3.62 × 106 | Theoretical | water, pH = 7.4 | [53] |
| 4-HMLT | HOO• | 5.48 × 105 | Theoretical | lipid | This work |
| 4-HMLT | HOO• | 6.21 × 106 | Theoretical | water, pH = 7.4 | This work |
| C1 | HOO• | 4.79 × 106 | Theoretical | water, pH = 7.4 | [53] |
| C2 | HOO• | 6.79 × 106 | Theoretical | water, pH = 7.4 | [53] |
| C3 | HOO• | 2.54 × 106 | Theoretical | water, pH = 7.4 | [53] |
| C4 | HOO• | 1.61 × 105 | Theoretical | water, pH = 7.4 | [53] |
| C5 | HOO• | 1.89 × 105 | Theoretical | water, pH = 7.4 | [53] |
| MLT | •OH | 2.23 × 1010 | Theoretical | benzene | [131] |
| MLT | •OH | 2.57 × 1010 | Experimental | water | (a) |
| MLT | •OOH | 3.11 × 102 | Theoretical | benzene | [131] |
| MLT | •OOH | 1.99 × 101 | Theoretical | water, pH = 7.4 | [131] |
| MLT | O2•− | <1.0 × 104 | Experimental | water | [452] |
| MLT | •NO2 | 3.7 × 106 | Experimental | water, pH = 7 | [175] |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galano, A.; Castañeda-Arriaga, R.; Pérez-González, A.; Tan, D.-X.; Reiter, R.J. Phenolic Melatonin-Related Compounds: Their Role as Chemical Protectors against Oxidative Stress. Molecules 2016, 21, 1442. https://doi.org/10.3390/molecules21111442
Galano A, Castañeda-Arriaga R, Pérez-González A, Tan D-X, Reiter RJ. Phenolic Melatonin-Related Compounds: Their Role as Chemical Protectors against Oxidative Stress. Molecules. 2016; 21(11):1442. https://doi.org/10.3390/molecules21111442
Chicago/Turabian StyleGalano, Annia, Romina Castañeda-Arriaga, Adriana Pérez-González, Dun-Xian Tan, and Russel J. Reiter. 2016. "Phenolic Melatonin-Related Compounds: Their Role as Chemical Protectors against Oxidative Stress" Molecules 21, no. 11: 1442. https://doi.org/10.3390/molecules21111442
APA StyleGalano, A., Castañeda-Arriaga, R., Pérez-González, A., Tan, D.-X., & Reiter, R. J. (2016). Phenolic Melatonin-Related Compounds: Their Role as Chemical Protectors against Oxidative Stress. Molecules, 21(11), 1442. https://doi.org/10.3390/molecules21111442








