Penicitroamide, an Antimicrobial Metabolite with High Carbonylization from the Endophytic Fungus Penicillium sp. (NO. 24)
Abstract
:1. Introduction
2. Results
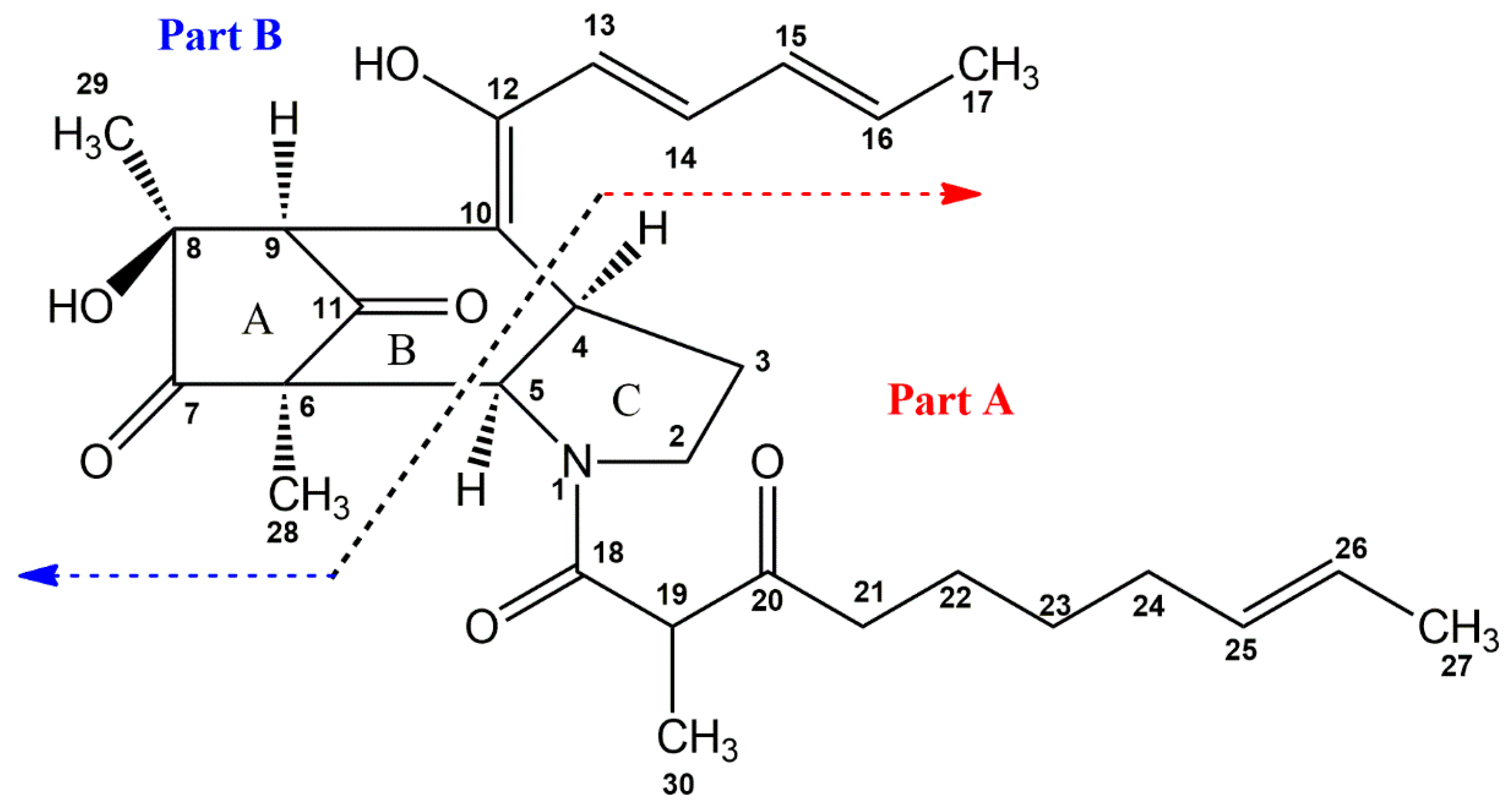
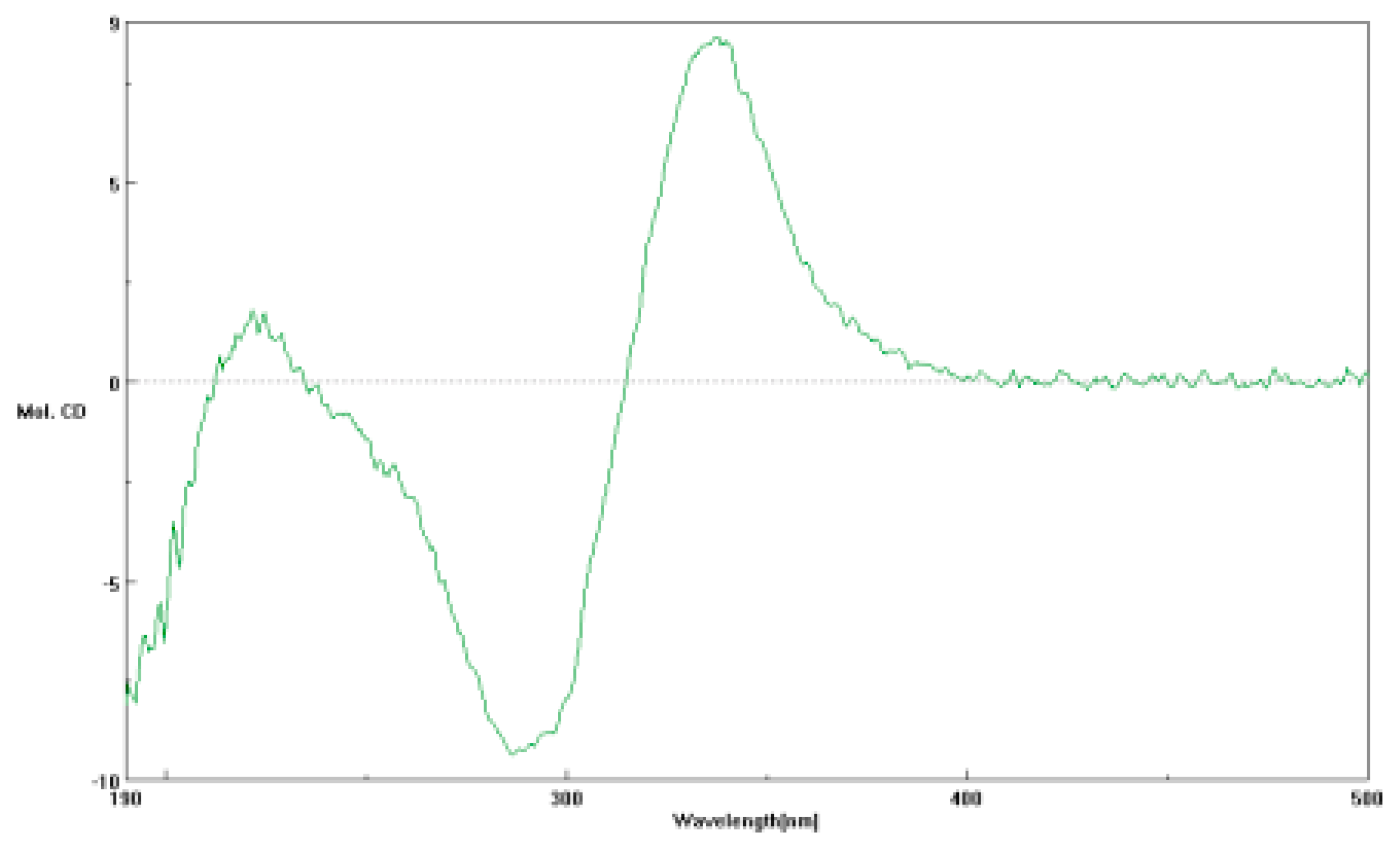
2.1. Structural Elucidationof Penicitroamide (1)
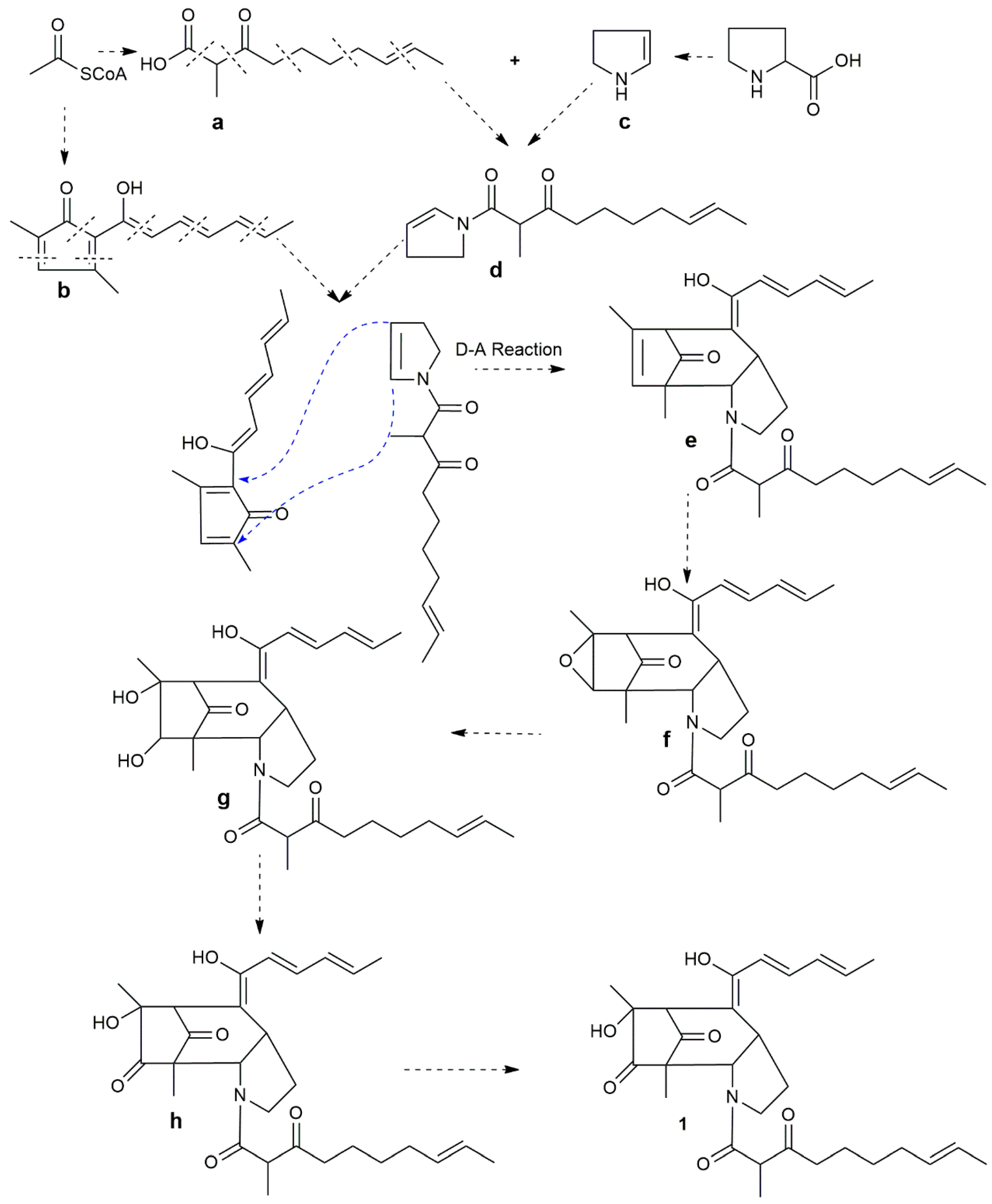
2.2. Possible Biosynthesis of Penicitroamide (1)
2.3. The Bioassay of Penicitroamide (1)
3. Experimental Section
3.1. General Procedures
3.2. Isolation and Identification of the Strain
3.3. The Isolation and Purification of Penicitroamide (1)
3.4. Bioactivity Assays
3.4.1. Cytotoxic Activity against Four Cancer Cell Lines In Vitro
3.4.2. Antimicrobial Assay
3.4.3. Brine Shrimp Lethality Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jiao, R.H.; Xu, S.; Liu, J.Y.; Ge, H.M.; Ding, H.; Xu, C.; Zhu, H.L.; Tan, R.X. Chaetominine, a Cytotoxic Alkaloid Produced by Endophytic Chaetomium sp. IFB-E015. Org. Lett. 2006, 8, 5709–5712. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Gessner, G.; Groth, I.; Lange, C.; Christner, A.; Bruhn, T.; Deng, Z.; Li, X.; Heinemann, S.H.; Grabley, S.; et al. Shearinines D–K, new indole triterpenoids from an endophytic Penicillium sp. (strain HKI0459) with blocking activity on large-conductance calcium-activated potassium channels. Tetrahedron 2007, 63, 435–444. [Google Scholar] [CrossRef]
- Ge, H.M.; Zhang, W.Y.; Ding, G.; Saparpakorn, P.; Song, Y.C.; Hannongbua, S.; Tan, R.X. Chaetoglobins A and B, two unusual alkaloids from endophytic Chaetomium globosum culture. Chem. Commun. 2008, 45, 5978–5980. [Google Scholar]
- Tsukamoto, S.; Umaoka, H.; Yoshikawa, K.; Ikeda, T.; Hirota, H. Notoamid O, a structurally unprecedented prenylated indole alkaloid, and notoamides P-R from a marine-derived fungus, Aspergillus sp. J. Nat. Prod. 2010, 73, 1438–1440. [Google Scholar] [CrossRef] [PubMed]
- Hua, C.; Yang, Y.; Sun, L.; Dou, H.; Tan, R.; Hou, Y. Chaetoglobosin F, a small molecule compound, possesses immunomodulatory properties on bone marrow-derived dendritic cells via TLR9 signaling pathway. Immunobiology 2013, 218, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Mándi, A.; Li, X.M.; Du, F.Y.; Wang, J.N.; Li, X.; Kurtán, T.; Wang, B.G. Varioxepine A, a 3H-oxepine-containing alkaloid with a new Oxa-cage from the marine algal-derived endophytic fungus Paecilomyces variotii. Org. Lett. 2014, 16, 4834–4837. [Google Scholar] [CrossRef] [PubMed]
- Lhamo, S.; Wang, X.B.; Li, T.X.; Wang, Y.; Li, Z.R.; Shi, Y.M.; Yang, M.H.; Kong, L.Y. Three new indole diketopiperazine alkaloids from a terrestrial-derived endophytic fungus Aspergillus sp. Tetrahedron Lett. 2015, 56, 2823–2826. [Google Scholar] [CrossRef]
- Fungi-1000-projects. Available online: http://genome.jgi.doe.gov/pages/fungi-1000-projects.jsf (accessed on 27 October 2016).
- Zhang, L.; Liu, Y.I.; Deng, Z.; Guo, Z.; Chen, J.; Tu, X.; Zou, K. Fusaodavinvin, a novel metabolite containing sulfur from the endophytic fungus Fusarium sp. (CTGU-ZL-34). Nat. Prod. Commun. 2013, 8, 83–84. [Google Scholar] [PubMed]
- Li, X.; Zhang, L.; Liu, Y.; Guo, Z.; Deng, Z.; Chen, J.; Tu, X.; Zou, K. A new metabolite from the endophytic fungus Penicillium citrinum. Nat. Prod. Commun. 2013, 8, 587–588. [Google Scholar]
- Wan, Q.; Feng, Z.; Li, X.; Lv, M.; Guo, Z.; Deng, Z.; Zou, K. Two new glycosidal metabolites of endophytic fungus Penicillium sp. (NO. 4) from Tapiscia sinensis. Z. Naturforsch. B 2016, 71, 283–286. [Google Scholar] [CrossRef]
- Zhang, H.; Deng, Z.; Guo, Z.; Tu, X.; Wang, J.; Zou, K. Pestalafuranones F–J, five new furanone analogues from the endophytic fungus Nigrospora sp. BM-2. Molecules 2014, 19, 819–825. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, M.; Tsuda, M.; Sekiguchi, M.; Mikami, Y.; Kobayashi, J. Perinadine A, a novel tetracyclic alkaloid from marine-derived fungus Penicillium citrinum. Org. Lett. 2005, 7, 4261–4264. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, M.; Sasaki, M.; Mugishima, T.; Komatsu, K.; Sone, T.; Tanaka, M.; Mikami, Y.; Kobayashi, J. Scalusamides A–C, new pyrrolidine alkaloids from the marine-derived fungus Penicillium citrinum. J. Nat. Prod. 2005, 68, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Kemble, A.R.; Macpherson, H.T. Determination of monoaminomonocarboxy-licacids by quantitative paper chromatography. Biochem. J. 1954, 56, 548–555. [Google Scholar] [CrossRef] [PubMed]
- Hien, D.T.; Jacobs, M.; Angenon, G.; Hermans, C.; Thu, T.T.; Son, L.V.; Roosens, N.H. Proline accumulation and Δ1-pyrroline-5-carboxylate synthetase gene properties in three rice cultivars differing in salinity and drought tolerance. Plant Sci. 2003, 165, 1059–1068. [Google Scholar] [CrossRef]
- Liu, J.; Zhu, J.K. Proline accumulation and salt-stress-induced gene expression in a salt-hyper sensitive mutant of Arabidopsis. Plant Physiol. 1997, 114, 591–596. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.; Yao, Y.N.; Yang, Y.Q.; Li, C.Y. Growth and physiological responses to supplemental UV-B radiation of two contrasting poplar species. Tree Physiol. 2006, 26, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Mehta, S.K.; Gaur, J.P. Heavy-metal-induced proline accumulation and its role in amelior ating metal toxicity in Chlorell avulgaris. New Phytol. 1999, 143, 253–259. [Google Scholar] [CrossRef]
- Kalamaki, M.S.; Merkouropoulos, G.; Kanellis, A.K. C anornithine accumulation modulate abiotic stress to lerance in Arabidopsis? Plant Signal Behav. 2009, 4, 1099–1101. [Google Scholar] [CrossRef] [PubMed]
- Wahid, A.; Gelani, S.; Ashraf, M.; Foolad, M.R. Heat tolerance in plants: An overview. Environ. Exp. Bot. 2007, 61, 199–223. [Google Scholar] [CrossRef]
- Yakushiji, F.; Miyamoto, Y.; Kunoh, Y.; Okamoto, R.; Nakaminami, H.; Yamazaki, Y.; Noguchi, N.; Hayashi, Y. Novel hybrid-type antimicrobial agents targeting the switch region of bacterial RNA polymerase. ACS Med. Chem. Lett. 2013, 4, 220–224. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are available from the authors.

| NO. | 13C (DEPT135) | 1H (J in Hz) | HMBC |
|---|---|---|---|
| 2 | 47.6 (CH2) | 3.71 (t, 9.5, 1H),2.75 (s, 1H) | 5 |
| 3 | 29.2 (CH2) | 2.10 (m, 1H), 1.50 (m, 1H) | 5 |
| 4 | 34.9 (CH) | 3.35 (m, 1H) d | 2, 5, 9, 23 |
| 5 | 61.2 (CH) | 4.65 (d, 8.6, 1H) | 2, 3, 9, 28 |
| 6 | 67.0 (C) | - | 5, 28 |
| 7 | 208.5 (C) | - | 5, 9, 8-OH, 28, 29 |
| 8 | 73.2 (C) | - | 8-OH, 9, 29 |
| 9 | 45.3 (CH) | 3.29 (m, 1H) d | 3, 5, 8-OH, 29 |
| 10 | 108.9 (C) | - | 13, 4, 9 |
| 11 | 197.3 (C) | - | 5, 9, 28 |
| 12 | 167.8 (C) | - | 13, 14 |
| 13 | 118.6 (CH) | 6.66 (d, 14.6, 1H) | 15 |
| 14 | 142.3 (CH) | 7.27 (dd, 11.0, 14.0, 1H) | 13, 15, 16 |
| 15 | 131.1 (CH) b | 6.39 (d, 11.4, 1H) | 13, 16, 17 |
| 16 | 139.7 (CH) | 6.28 (m, 1H) | 14, 15, 17 |
| 17 | 18.6 (CH3) | 1.85 (d, 6.0, 3H) | 15, 16 |
| 18 | 170.7 (C) | - | 2, 5, 19, 30 |
| 19 | 50.9 (CH) | 3.83 (br d, 6.8, 1H) | 30 |
| 20 | 206.8 (C) | - | 19, 21, 22, 30 |
| 21 | 40.0 (CH2) | 2.39 (m, 2H) | 22, 23 |
| 22 | 22.5 (CH2) | 1.39 (m, 2H) | 21, 23, 24 |
| 23 | 28.3 (CH2) | 1.23 (m, 2H) | 21, 22, 24, 27 |
| 24 | 31.8 (CH2) | 1.89 (m, 2H) | 22, 23, 25 |
| 25 | 131.1 (CH) | 5.36 (s, 1H) | 23, 24, 27 |
| 26 | 124.5 (CH) | 5.36 (s, 1H) c | 24, 27 |
| 27 | 17.7 (CH3) | 1.59 (br s, 3H) c | 25 |
| 28 | 10.4 (CH3) | 0.99 (s, 3H) | 5 |
| 29 | 23.4 (CH3) | 1.07 (s, 3H) | 8-OH, 9 |
| 30 | 13.3 (CH3) | 1.11 (d, 6.1, 3H) | 19 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, Z.-W.; Lv, M.-M.; Li, X.-S.; Zhang, L.; Liu, C.-X.; Guo, Z.-Y.; Deng, Z.-S.; Zou, K.; Proksch, P. Penicitroamide, an Antimicrobial Metabolite with High Carbonylization from the Endophytic Fungus Penicillium sp. (NO. 24). Molecules 2016, 21, 1438. https://doi.org/10.3390/molecules21111438
Feng Z-W, Lv M-M, Li X-S, Zhang L, Liu C-X, Guo Z-Y, Deng Z-S, Zou K, Proksch P. Penicitroamide, an Antimicrobial Metabolite with High Carbonylization from the Endophytic Fungus Penicillium sp. (NO. 24). Molecules. 2016; 21(11):1438. https://doi.org/10.3390/molecules21111438
Chicago/Turabian StyleFeng, Zi-Wei, Meng-Meng Lv, Xue-Shuang Li, Liang Zhang, Cheng-Xiong Liu, Zhi-Yong Guo, Zhang-Shuang Deng, Kun Zou, and Peter Proksch. 2016. "Penicitroamide, an Antimicrobial Metabolite with High Carbonylization from the Endophytic Fungus Penicillium sp. (NO. 24)" Molecules 21, no. 11: 1438. https://doi.org/10.3390/molecules21111438
APA StyleFeng, Z.-W., Lv, M.-M., Li, X.-S., Zhang, L., Liu, C.-X., Guo, Z.-Y., Deng, Z.-S., Zou, K., & Proksch, P. (2016). Penicitroamide, an Antimicrobial Metabolite with High Carbonylization from the Endophytic Fungus Penicillium sp. (NO. 24). Molecules, 21(11), 1438. https://doi.org/10.3390/molecules21111438






