Curcumin Shows Antiviral Properties against Norovirus
Abstract
:1. Introduction
2. Results
2.1. Evaluation of Phytochemical Cytotoxicity
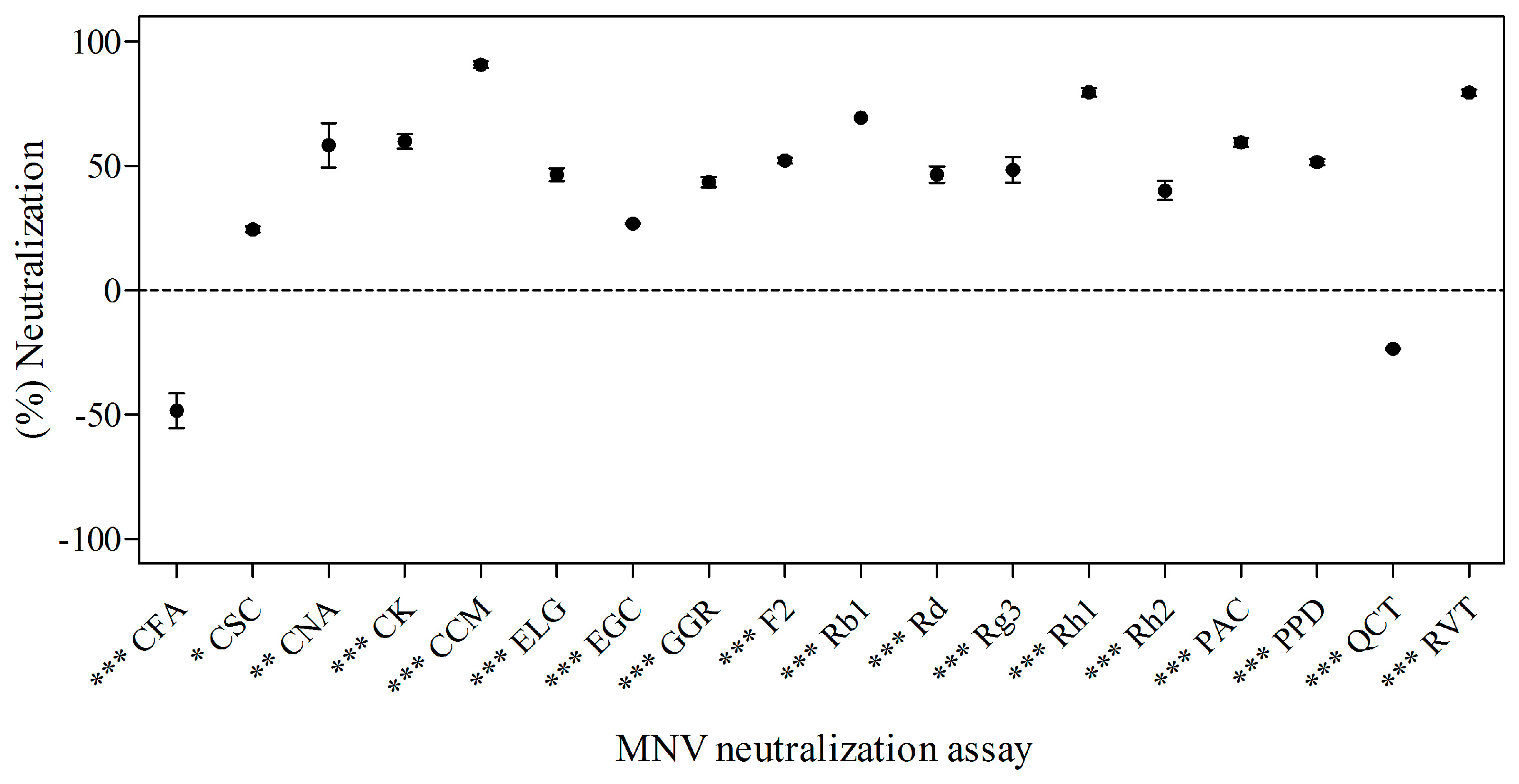
2.2. Effects of Phytochemicals on Neutralization of MNV
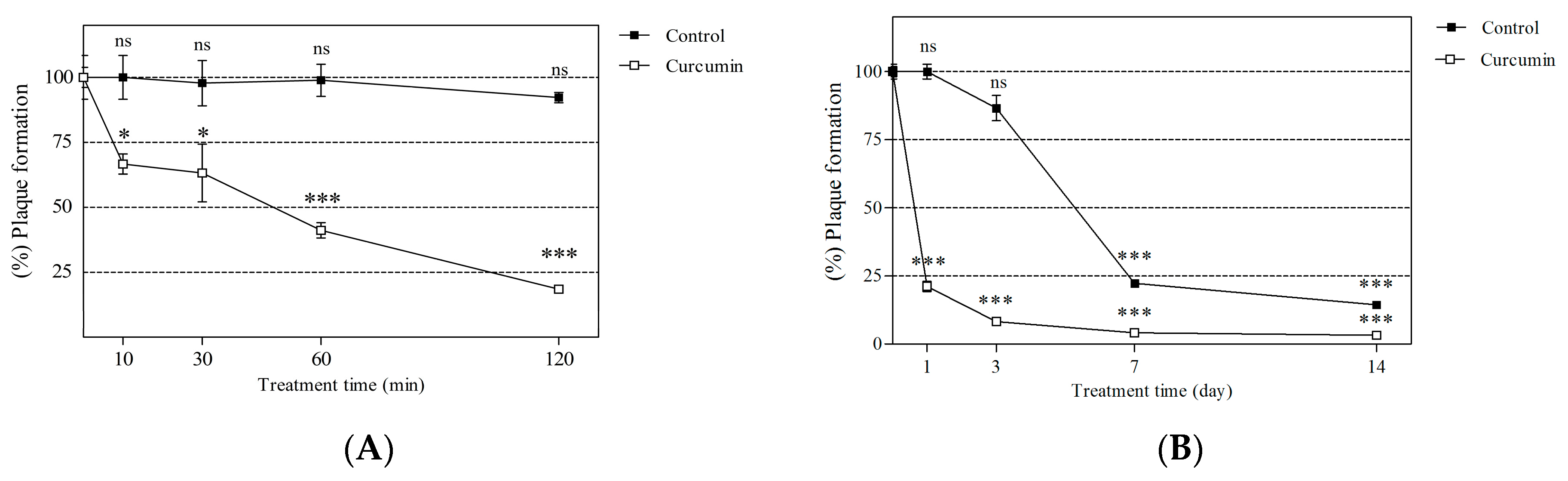
2.3. Dose-Response Relationship between CCM Exposure and Inactivation of MNV
2.4. Short- and Long-Term Effects of Curcumin on Viral Neutralization
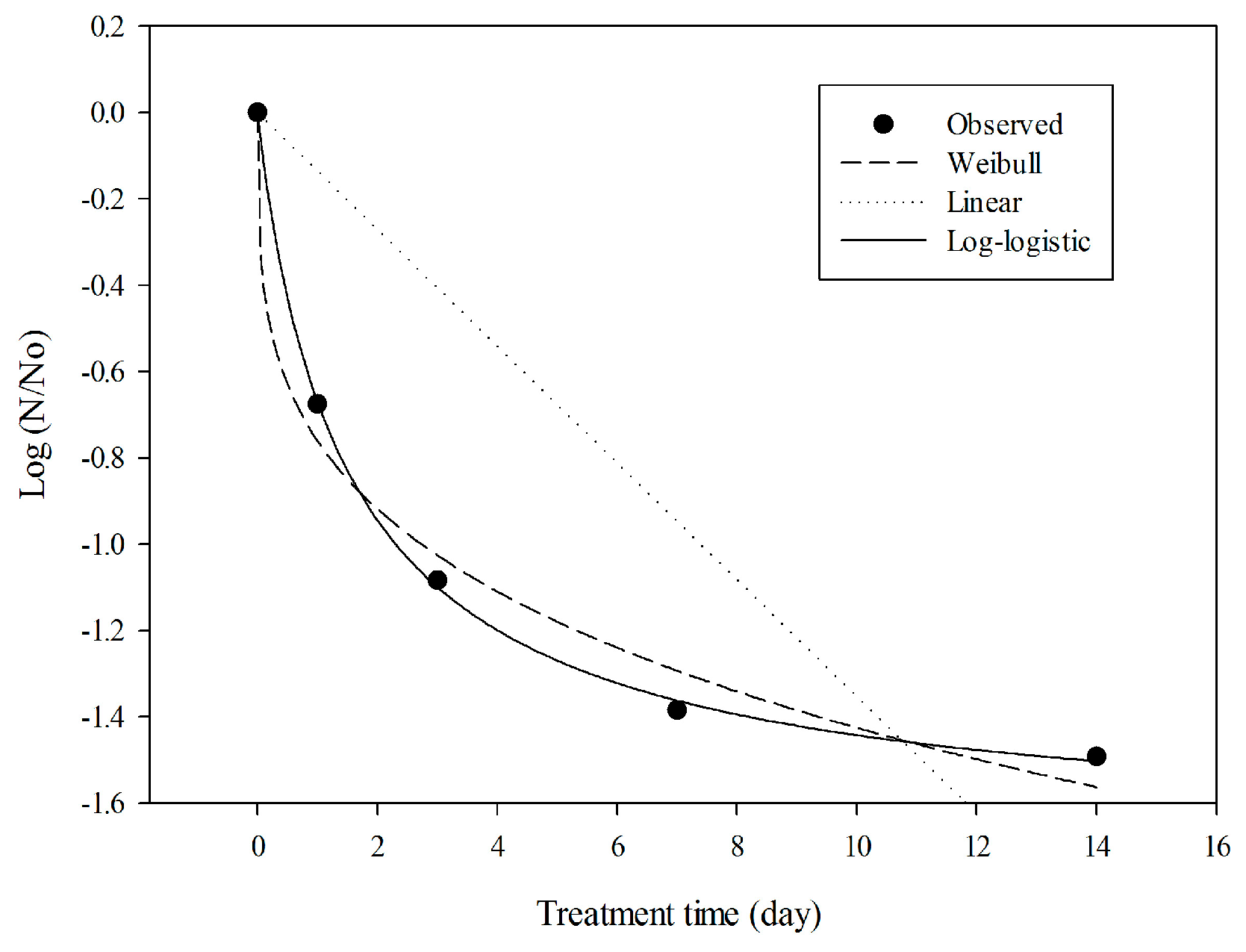
2.5. Model Evaluation with Experimental Data
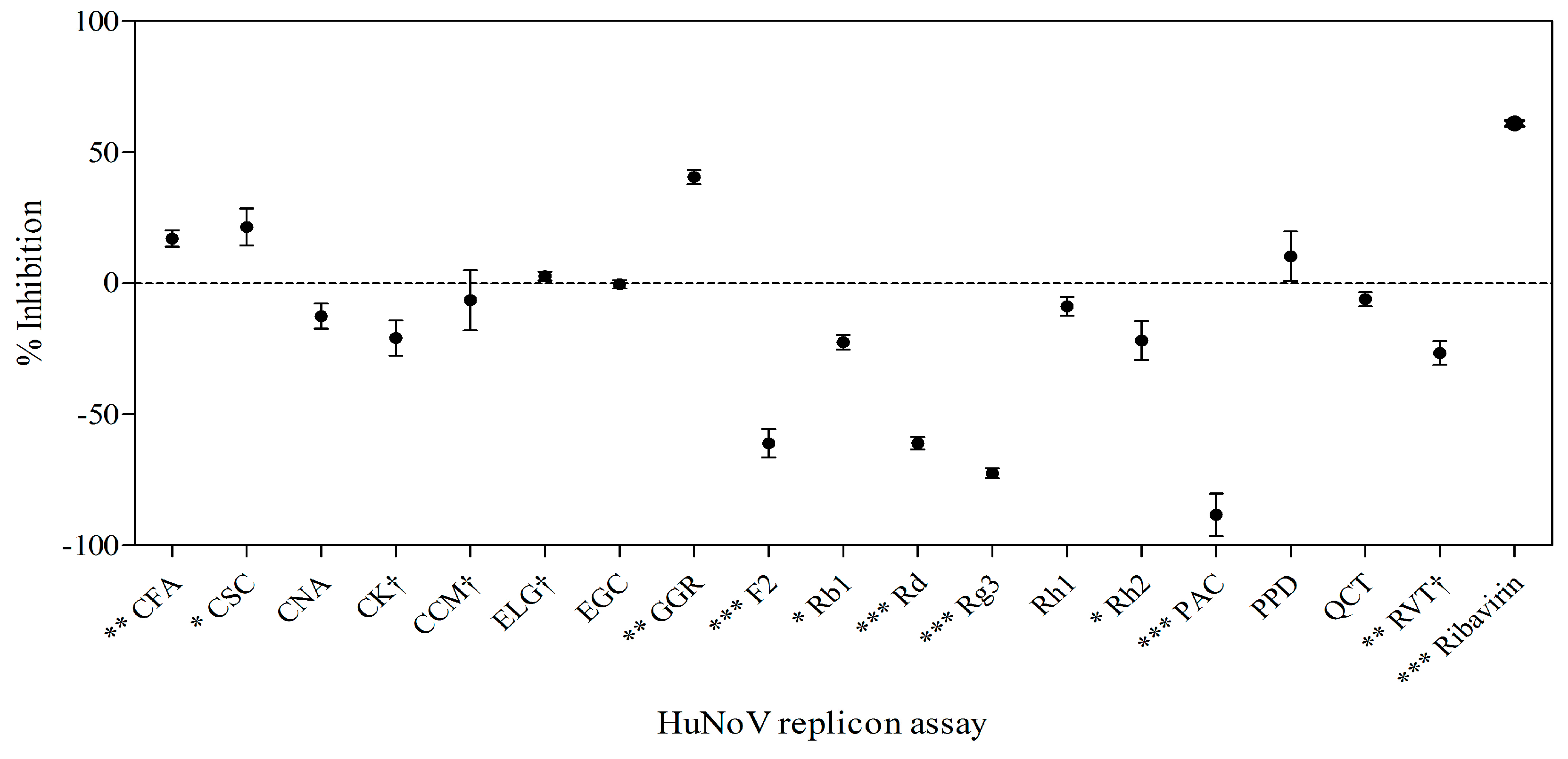
2.6. Effects of Phytochemicals on Replication of HuNoV in Replicon-Bearing Cells
3. Discussion
4. Materials and Methods
4.1. Tested Phytochemicals
4.2. Virus and Cell Lines
4.3. Cultivation of MNV and Preparation of Viral Stock
4.4. Cytotoxicity Assay
4.5. Quantification of MNV by Plaque Assay
4.6. MNV Neutraization Asay
4.7. HuNoV Replication Assay Using HuNoV Replicon-Bearing HG23 Cells
4.8. RNA Extraction and Real-time RT-PCR
4.9. Fitting the Experimental Data to Three Different Models
4.10. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix
| Name of Phytochemical | Abbreviation | Molecular Weight | Sources |
|---|---|---|---|
| Protopanaxadiol | PPD | 460.73 | ginseng |
| Compound K | CK | 653.81 | ginseng |
| Ginsenoside Rh2 | Rh2 | 622.87 | ginseng |
| Ginsenoside Rg3 | Rg3 | 785.01 | ginseng |
| Ginsenoside F2 | F2 | 785.01 | ginseng |
| Ginsenoside Rd | Rd | 947.15 | ginseng |
| Ginsenoside Rb1 | Rb1 | 1109.30 | ginseng |
| Ginsenoside Rh1 | Rh1 | 638.87 | ginseng |
| Curcumin | CCM | 368.38 | turmeric |
| Resveratrol | RVT | 228.24 | grape and berries |
| Cinnamic acid | CNA | 148.16 | cinnamon |
| Caffeic acid | CFA | 180.16 | coffee |
| Epigallocatechin gallate | EGC | 458.37 | green tea |
| Capsaicin | CSC | 305.41 | red pepper |
| Quercetin | QCT | 302.24 | onions and apples |
| 10-Gingerol | GGR | 350.50 | ginger |
| Proanthocyanidin | PAC | 578.52 | fruits and berries |
| Ellagic acid | ELG | 302.19 | fruits and berries |
References
- Ike, A.C.; Brockmann, S.O.; Hartelt, K.; Marschang, R.E.; Contzen, M.; Oehme, R.M. Molecular epidemiology of norovirus in outbreaks of gastroenteritis in Southwest Germany from 2001 to 2004. J. Clin. Microbiol. 2006, 44, 1262–1267. [Google Scholar] [CrossRef] [PubMed]
- Lopman, B.; Reacher, M.; van Duijnhoven, Y.; Hanon, F.-X.; Brown, D.; Koopmans, M. Viral gastroenteritis outbreaks in Europe, 1995–2000. Emerg. Infect. Dis. 2003, 9, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Payne, D.C.; Vinjé, J.; Szilagyi, P.G.; Edwards, K.M.; Staat, M.A.; Weinberg, G.A.; Hall, C.B.; Chappell, J.; Bernstein, D.I.; Curns, A.T. Norovirus and medically attended gastroenteritis in us children. N. Engl. J. Med. 2013, 368, 1121–1130. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, J.E.; Feldman, R.; Campbell, D.S.; Lookabaugh, C.; Gary, G.W. The frequency of a norwalk-like pattern of illness in outbreaks of acute gastroenteritis. Am. J. Public Health 1982, 72, 1329–1332. [Google Scholar] [CrossRef] [PubMed]
- Glass, R.I.; Parashar, U.D.; Estes, M.K. Norovirus gastroenteritis. N. Engl. J. Med. 2009, 361, 1776–1785. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.P.; Edmunds, W.J.; Pebody, R.; Brown, D.W.; Lopman, B.A. Deaths from norovirus among the elderly, England and Wales. Emerg. Infect. Dis. 2008, 14, 1546–1552. [Google Scholar] [CrossRef] [PubMed]
- Isakbaeva, E.T.; Widdowson, M.-A.; Beard, R.S.; Bulens, S.N.; Mullins, J.; Monroe, S.S.; Bresee, J.; Sassano, P.; Cramer, E.H.; Glass, R.I. Norovirus transmission on cruise ship. Emerg. Infect. Dis. 2005, 11, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Lopman, B.A.; Adak, G.K.; Reacher, M.H.; Brown, D. Two epidemiologic patterns of norovirus outbreaks: Surveillance in England and Wales, 1992–2000. Emerg. Infect. Dis. 2003, 9, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Norovirus outbreak in an elementary school—District of Columbia, February 2007. Morb. Mortal. Wkly. Rep. 2008, 56, 1340. [Google Scholar]
- Lee, J.; Zoh, K.; Ko, G. Inactivation and UV disinfection of murine norovirus with TiO2 under various environmental conditions. Appl. Environ. Microbiol. 2008, 74, 2111–2117. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; D’Souza, D.H. Naturally occurring flavonoids against human norovirus surrogates. Food Environ. Virol. 2013, 5, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Fino, V.R.; Kniel, K.E. UV light inactivation of hepatitis A virus, Aichi virus, and feline calicivirus on strawberries, green onions, and lettuce. J. Food Prot. 2008, 71, 908–913. [Google Scholar] [PubMed]
- Li, D.; Baert, L.; Uyttendaele, M. Inactivation of food-borne viruses using natural biochemical substances. Food Microbiol. 2013, 35, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, J.; Rivera-Aban, M.; Greening, G. Evaluation of murine norovirus as a surrogate for human norovirus and hepatitis a virus in heat inactivation studies. J. Appl. Microbiol. 2009, 107, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Yuen, Y.; Hsiao, H.-M.; Jaykus, L.-A.; Moe, C. Effectiveness of liquid soap and hand sanitizer against norwalk virus on contaminated hands. Appl. Environ. Microbiol. 2010, 76, 394–399. [Google Scholar] [CrossRef] [PubMed]
- Park, G.; Linden, K.; Sobsey, M. Inactivation of murine norovirus, feline calicivirus and echovirus 12 as surrogates for human norovirus (nov) and coliphage (F+) MS2 by ultraviolet light (254 nm) and the effect of cell association on UV inactivation. Lett. Appl. Microbiol. 2011, 52, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Gilling, D.H.; Kitajima, M.; Torrey, J.R.; Bright, K.R. Antiviral efficacy and mechanisms of action of oregano essential oil and its primary component carvacrol against murine norovirus. J. Appl. Microbiol. 2014, 116, 1149–1163. [Google Scholar] [CrossRef] [PubMed]
- Iwasawa, A.; Niwano, Y.; Mokudai, T.; Kohno, M. Antiviral activity of proanthocyanidin against feline calicivirus used as a surrogate for noroviruses, and coxsackievirus used as a representative enteric virus. Biocontrol Sci. 2009, 14, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, X.; Wang, Y.; Chen, F.; Yu, Z.; Wang, L.; Chen, S.; Guo, M. Effect of citrus lemon oil on growth and adherence of streptococcus mutans. World J. Microbiol. Biotechnol. 2013, 29, 1161–1167. [Google Scholar] [CrossRef] [PubMed]
- Gilling, D.H.; Kitajima, M.; Torrey, J.R.; Bright, K.R. Mechanisms of antiviral action of plant antimicrobials against murine norovirus. Appl. Environ. Microbiol. 2014, 80, 4898–4910. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.K.; Watanabe, M.; Zhu, S.; Graves, C.L.; Keyes, L.R.; Grau, K.R.; Gonzalez-Hernandez, M.B.; Iovine, N.M.; Wobus, C.E.; Vinje, J.; et al. Enteric bacteria promote human and mouse norovirus infection of B cells. Science 2014, 346, 755–759. [Google Scholar] [CrossRef] [PubMed]
- Ettayebi, K.; Crawford, S.E.; Murakami, K.; Broughman, J.R.; Karandikar, U.; Tenge, V.R.; Neill, F.H.; Blutt, S.E.; Zeng, X.-L.; Qu, L. Replication of human noroviruses in stem cell-derived human enteroids. Science 2016, 353, 1387–1393. [Google Scholar] [CrossRef] [PubMed]
- Kolawole, A.O.; Rocha-Pereira, J.; Elftman, M.D.; Neyts, J.; Wobus, C.E. Inhibition of human norovirus by a viral polymerase inhibitor in the b cell culture system and in the mouse model. Antiviral Res. 2016, 132, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Wobus, C.E.; Thackray, L.B.; Virgin, H.W. Murine norovirus: A model system to study norovirus biology and pathogenesis. J. Virol. 2006, 80, 5104–5112. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.O.; Sosnovtsev, S.V.; Belliot, G.; King, A.D.; Green, K.Y. Stable expression of a Norwalk virus RNA replicon in a human hepatoma cell line. Virology 2006, 353, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Oh, M.; Lee, J.-H.; Bae, S.Y.; Seok, J.H.; Kim, S.; Chung, Y.B.; Han, K.R.; Kim, K.H.; Chung, M.S. Protective effects of red wine and resveratrol for foodborne virus surrogates. Food Control 2015, 47, 502–509. [Google Scholar] [CrossRef]
- Corbo, M.R.; Bevilacqua, A.; Campaniello, D.; D’Amato, D.; Speranza, B.; Sinigaglia, M. Prolonging microbial shelf life of foods through the use of natural compounds and non-thermal approaches—A review. Int. J. Food Sci. Technol. 2009, 44, 223–241. [Google Scholar] [CrossRef]
- D’Souza, D.H. Phytocompounds for the control of human enteric viruses. Curr. Opin. Virol. 2014, 4, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-F.; Dai, Y.-C.; Zhong, W.; Tan, M.; Lv, Z.-P.; Zhou, Y.-C.; Jiang, X. Tannic acid inhibited norovirus binding to HBGA receptors, a study of 50 Chinese medicinal herbs. Bioorg. Med. Chem. 2012, 20, 1616–1623. [Google Scholar] [CrossRef] [PubMed]
- Prasad, S.; Gupta, S.C.; Tyagi, A.K.; Aggarwal, B.B. Curcumin, a component of golden spice: From bedside to bench and back. Biotechnol. Adv. 2014, 32, 1053–1064. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.M.; Lopes, T.; Oleastro, M.; Gato, I.V.; Floch, P.; Benejat, L.; Chaves, P.; Pereira, T.; Seixas, E.; Machado, J.; et al. Curcumin inhibits gastric inflammation induced by helicobacter pylori infection in a mouse model. Nutrients 2015, 7, 306–320. [Google Scholar] [CrossRef] [PubMed]
- Zandi, K.; Ramedani, E.; Mohammadi, K.; Tajbakhsh, S.; Deilami, I.; Rastian, Z.; Fouladvand, M.; Yousefi, F.; Farshadpour, F. Evaluation of antiviral activities of curcumin derivatives against HSV-1 in vero cell line. Nat. Prod. Commun. 2010, 5, 1935–1938. [Google Scholar] [PubMed]
- Commandeur, J.; Vermeulen, N. Cytotoxicity and cytoprotective activities of natural compounds. The case of curcumin. Xenobiotica 1996, 26, 667–680. [Google Scholar] [CrossRef] [PubMed]
- Asher, G.N.; Xie, Y.; Moaddel, R.; Sanghvi, M.; Dossou, K.S.; Kashuba, A.D.M.; Sandler, R.S.; Hawke, R.L. Randomized pharmacokinetic crossover study comparing 2 curcumin preparations in plasma and rectal tissue of healthy human volunteers. J. Clin. Pharmacol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Colpitts, C.C.; Schang, L.M.; Rachmawati, H.; Frentzen, A.; Pfaender, S.; Behrendt, P.; Brown, R.J.; Bankwitz, D.; Steinmann, J.; Ott, M. Turmeric curcumin inhibits entry of all hepatitis c virus genotypes into human liver cells. Gut 2014. [Google Scholar] [CrossRef]
- Kim, K.; Kim, K.H.; Kim, H.Y.; Cho, H.K.; Sakamoto, N.; Cheong, J. Curcumin inhibits hepatitis c virus replication via suppressing the akt-srebp-1 pathway. FEBS Lett. 2010, 584, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Zorofchian Moghadamtousi, S.; Abdul Kadir, H.; Hassandarvish, P.; Tajik, H.; Abubakar, S.; Zandi, K. A review on antibacterial, antiviral, and antifungal activity of curcumin. BioMed Res. Int. 2014, 2014, 186864. [Google Scholar] [CrossRef] [PubMed]
- Randazzo, W.; Aznar, R.; Sánchez, G. Curcumin-mediated photodynamic inactivation of norovirus surrogates. Food Environ. Virol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Hou, W.; Cao, B.; Zuo, T.; Xue, C.; Leung, A.W.; Xu, C.; Tang, Q.-J. Virucidal efficacy of treatment with photodynamically activated curcumin on murine norovirus bio-accumulated in oysters. Photodiagn. Photodyn. Ther. 2015, 12, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Si, J.; Yun, H.S.; Ko, G. Effect of temperature and relative humidity on the survival of foodborne viruses during food storage. Appl. Environ. Microbiol. 2015, 81, 2075–2081. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.M.; Lopman, B.A.; Levy, K. A systematic review and meta-analysis of the global seasonality of norovirus. PLoS ONE 2013, 8, e75922. [Google Scholar] [CrossRef] [PubMed]
- Horm, K.M.; D’Souza, D.H. Survival of human norovirus surrogates in milk, orange, and pomegranate juice, and juice blends at refrigeration (4 °C). Food Microbiol. 2011, 28, 1054–1061. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; Howell, A.B.; D’Souza, D.H. The effect of cranberry juice and cranberry proanthocyanidins on the infectivity of human enteric viral surrogates. Food Microbiol. 2010, 27, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; Howell, A.B.; D’Souza, D.H. Antiviral effects of cranberry juice and cranberry proanthocyanidins on foodborne viral surrogates—A time dependence study in vitro. Food Microbiol. 2010, 27, 985–991. [Google Scholar] [CrossRef] [PubMed]
- Jones, N.L.; Shabib, S.; Sherman, P.M. Capsaicin as an inhibitor of the growth of the gastric pathogen helicobacter pylori. FEMS Microbiol. Lett. 1997, 146, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Molina-Torres, J.; Garcia-Chavez, A.; Ramirez-Chavez, E. Antimicrobial properties of alkamides present in flavouring plants traditionally used in Mesoamerica: Affinin and capsaicin. J. Ethnopharmacol. 1999, 64, 241–248. [Google Scholar] [CrossRef]
- Kalia, N.P.; Mahajan, P.; Mehra, R.; Nargotra, A.; Sharma, J.P.; Koul, S.; Khan, I.A. Capsaicin, a novel inhibitor of the nora efflux pump, reduces the intracellular invasion of staphylococcus aureus. J. Antimicrob. Chemother. 2012, 67, 2401–2408. [Google Scholar] [CrossRef] [PubMed]
- Ljungdahl, A.; Kristensson, K.; Lundberg, J.M.; Lycke, E.; Svennerholm, B.; Ziegler, R. Herpes simplex virus infection in capsaicin-treated mice. J. Neurol. Sci. 1986, 72, 223–230. [Google Scholar] [CrossRef]
- Stanberry, L.R. Capsaicin interferes with the centrifugal spread of virus in primary and recurrent genital herpes simplex virus infections. J. Infect. Dis. 1990, 162, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Mahady, G.B.; Pendland, S.L.; Yun, G.S.; Lu, Z.Z.; Stoia, A. Ginger (zingiber officinale roscoe) and the gingerols inhibit the growth of cag a+ strains of helicobacter pylori. Anticancer Res. 2003, 23, 3699–3702. [Google Scholar] [PubMed]
- Park, M.; Bae, J.; Lee, D.S. Antibacterial activity of [10]-gingerol and [12]-gingerol isolated from ginger rhizome against periodontal bacteria. Phytother. Res. 2008, 22, 1446–1449. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.O. Role of cholesterol pathways in norovirus replication. J. Virol. 2009, 83, 8587–8595. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, A.A.; Abuirmeileh, N.; Din, Z.Z.; Ahmad, Y.; Burger, W.C.; Elson, C.E. Suppression of cholesterogenesis and reduction of LDL cholesterol by dietary ginseng and its fractions in chicken liver. Atherosclerosis 1983, 48, 81–94. [Google Scholar] [CrossRef]
- Wobus, C.E.; Karst, S.M.; Thackray, L.B.; Chang, K.-O.; Sosnovtsev, S.V.; Belliot, G.; Krug, A.; Mackenzie, J.M.; Green, K.Y.; Virgin, H.W. Replication of norovirus in cell culture reveals a tropism for dendritic cells and macrophages. PLoS Biol. 2004, 2, e432. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-J.; Shin, Y.; Lee, K.H.; Kim, T.-J. Anethum graveloens flower extracts inhibited a lipopolysaccharide-induced inflammatory response by blocking inos expression and NF-κb activity in macrophages. Biosci. Biotechnol. Biochem. 2012, 76, 1122–1127. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Hernandez, M.B.; Cunha, J.B.; Wobus, C.E. Plaque assay for murine norovirus. J. Vis. Exp. 2012, 22, e4297. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.-O.; George, D.W. Interferons and ribavirin effectively inhibit norwalk virus replication in replicon-bearing cells. J. Virol. 2007, 81, 12111–12118. [Google Scholar] [CrossRef] [PubMed]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29. [Google Scholar] [CrossRef]
- Park, Y.; Cho, Y.-H.; Ko, G. A duplex real-time RT-PCR assay for the simultaneous genogroup-specific detection of noroviruses in both clinical and environmental specimens. Virus Genes 2011, 43, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Spann, K.M.; Tran, K.-C.; Chi, B.; Rabin, R.L.; Collins, P.L. Suppression of the induction of alpha, beta, and lambda interferons by the ns1 and ns2 proteins of human respiratory syncytial virus in human epithelial cells and macrophages. J. Virol. 2004, 78, 4363–4369. [Google Scholar] [CrossRef] [PubMed]
- Peleg, M.; Cole, M.B. Reinterpretation of microbial survival curves. Crit. Rev. Food Sci. Nutr. 1998, 38, 353–380. [Google Scholar] [CrossRef] [PubMed]
- Cole, M.B.; Davies, K.W.; Munro, G.; Holyoak, C.D.; Kilsby, D.C. A vitalistic model to describe the thermal inactivation of Listeria monocytogenes. J. Ind. Microbiol. 1993, 12, 232–239. [Google Scholar] [CrossRef]
- Chen, H.; Hoover, D.G. Modeling the combined effect of high hydrostatic pressure and mild heat on the inactivation kinetics of listeria monocytogenes scott a in whole milk. Innov. Food Sci. Emerg. Technol. 2003, 4, 25–34. [Google Scholar] [CrossRef]
- Sample Availability: Not Available.

| Common Name | Abbreviation | RAW 264.7 a | HG23 b |
|---|---|---|---|
| % Cell Viability | |||
| Caffeic acid | CFA | 101.14 ± 6.00 | 92.62 ± 3.87 |
| Capsaicin | CSC | 119.18 ± 2.83 | 82.92 ± 0.58 |
| Cinnamic acid | CNA | 102.25 ± 4.11 | 93.87 ± 6.75 |
| Compound K | CK | 91.89 ± 1.10 | 80.67 ± 3.07 † |
| Curcumin | CCM | 102.14 ± 1.89 | 92.57 ± 2.41 † |
| Ellagic acid | ELG | 80.32 ± 1.68 | 89.91 ± 7.50 † |
| Epigallocatechin gallate | EGC | 94.44 ± 5.55 | 87.44 ± 3.09 |
| 10-Gingerol | GGR | 83.68 ± 3.03 | 86.07 ± 7.02 |
| Ginsenoside F2 | F2 | 88.30 ± 3.65 | 101.52 ± 4.22 |
| Ginsenoside Rb1 | Rb1 | 95.27 ± 5.31 | 101.48 ± 4.61 |
| Ginsenoside Rd | Rd | 87.17 ± 0.79 | 102.11 ± 2.80 |
| Ginsenoside Rg3 | Rg3 | 84.75 ± 2.10 | 95.29 ± 3.47 |
| Ginsenoside Rh1 | Rh1 | 98.54 ± 2.94 | 94.38 ± 4.51 |
| Ginsenoside Rh2 | Rh2 | 95.32 ± 2.00 | 94.18 ± 4.86 |
| Proanthocyanidin | PAC | 87.47 ± 7.31 | 102.40 ± 0.57 |
| Protopanaxadiol | PPD | 97.57 ± 1.80 | 99.03 ± 3.17 |
| Quercetin | QCT | 127.26 ± 3.65 | 88.50 ± 7.23 |
| Resveratrol | RVT | 84.46 ± 2.03 | 80.07 ± 5.94 † |
| Fitting Model | Curcumin Inactivation | |||
|---|---|---|---|---|
| R2 | RMSE | AIC | D-value | |
| Linear distribution | 0.25 | 0.53 | 2.40 | 7.39 |
| Weibull distribution | 0.98 | 0.09 | −10.94 | 2.72 |
| Log-logistic distribution | 0.99 | 0.02 | −22.05 | 2.30 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, M.; Lee, G.; Si, J.; Lee, S.-J.; You, H.J.; Ko, G. Curcumin Shows Antiviral Properties against Norovirus. Molecules 2016, 21, 1401. https://doi.org/10.3390/molecules21101401
Yang M, Lee G, Si J, Lee S-J, You HJ, Ko G. Curcumin Shows Antiviral Properties against Norovirus. Molecules. 2016; 21(10):1401. https://doi.org/10.3390/molecules21101401
Chicago/Turabian StyleYang, Minji, GilJae Lee, Jiyeon Si, Sung-Joon Lee, Hyun Ju You, and GwangPyo Ko. 2016. "Curcumin Shows Antiviral Properties against Norovirus" Molecules 21, no. 10: 1401. https://doi.org/10.3390/molecules21101401
APA StyleYang, M., Lee, G., Si, J., Lee, S.-J., You, H. J., & Ko, G. (2016). Curcumin Shows Antiviral Properties against Norovirus. Molecules, 21(10), 1401. https://doi.org/10.3390/molecules21101401






