Antiprotozoal and Antiglycation Activities of Sesquiterpene Coumarins from Ferula narthex Exudate
Abstract
:1. Introduction
2. Results and Discussion
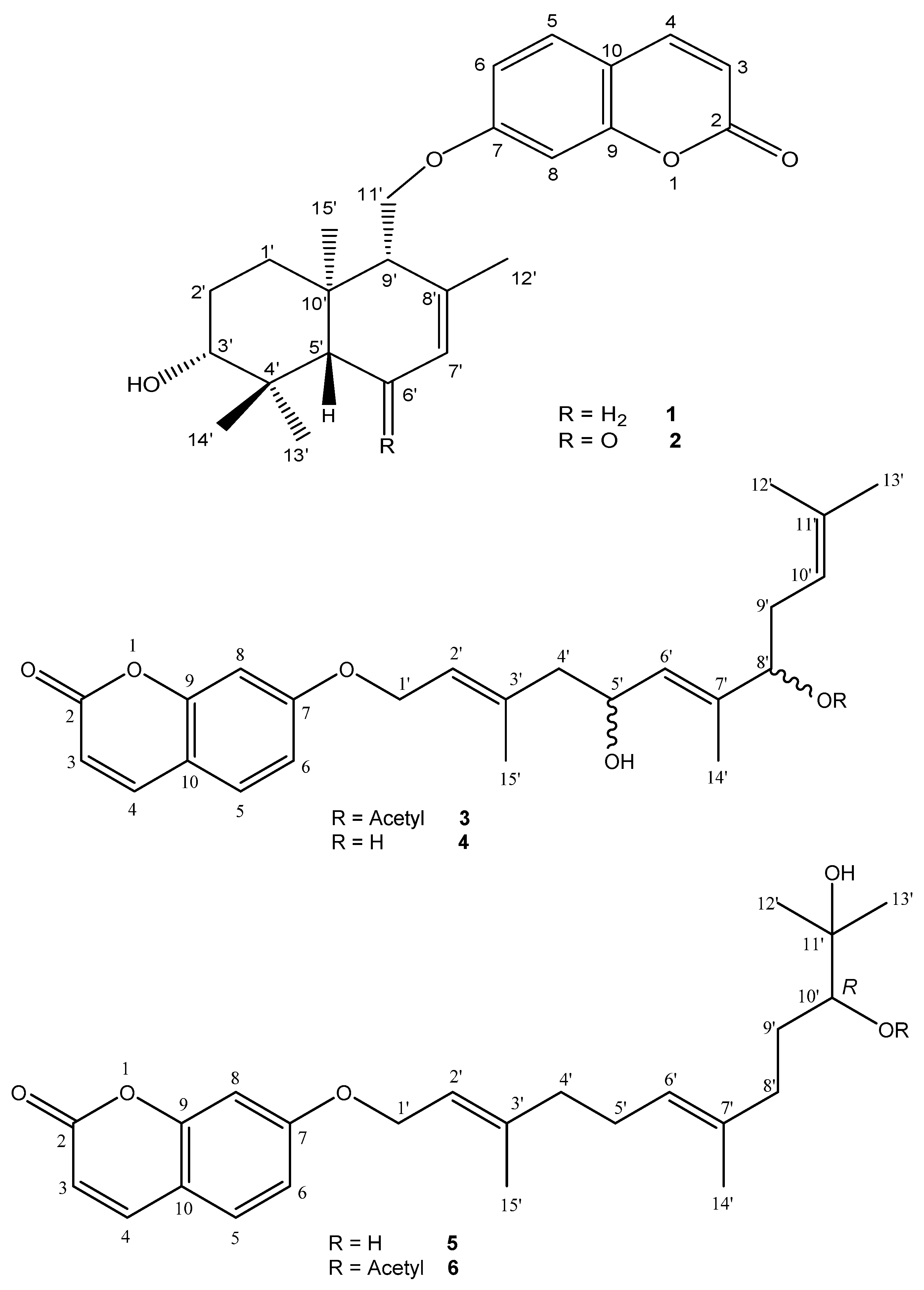
2.1. Structure Elucidation
2.2. Antimicrobial Activity
2.3. Antiglycation Activity
3. Materials and Methods
3.1. General Experimetal Procedures
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Antimicrobial Activity
3.5. Antiglycation Activity
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pimenov, M.G.; Leonov, M.V. The Asian Umbelliferae biodiversity database (ASIUM) with particular reference to South-West Asian taxa. Turk. J. Bot. 2004, 28, 139–145. [Google Scholar]
- Khan, N.; Ahmed, N.; Ahmed, A.; Shaukat, S.S.; Wahab, M.; Ajaib, M.; Siddiqui, M.F.; Nasir, M. Important medicinal plants of Chitral Gol National Park (CGNP) Pakistan. Pak. J. Bot. 2011, 43, 797–809. [Google Scholar]
- Mahendra, P.; Bisht, S. Ferula asafoetida: Traditional uses and pharmacological activity. Pharmacog. Rev. 2012, 6, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Shinwari, Z.K.; Gilani, S.S. Sustainable harvest of medicinal plants at Bulashbar Nullah, Astore (Northern Pakistan). J. Ethnopharmacol. 2003, 84, 289–298. [Google Scholar] [CrossRef]
- Srinivasan, K. Spices as influencers of body metabolism: An overview of three decades of research. Food Res. Int. 2005, 38, 77–86. [Google Scholar] [CrossRef]
- Bashir, S.; Alam, M.; Ahmad, B.; Aman, A.; Ali, J. Screening of Ferula narthex Boiss. crude methanolic extract for analgesic, gastrointestinal motility and insecticidal activity. Middle-East. J. Sci. Res. 2013, 14, 471–475. [Google Scholar]
- Bashir, S.; Alam, M.; Ahmad, B.; Aman, A. Antibacterial, anti-fungal and phytotoxic activities of Ferula narthex Boiss. Pak. J. Pharm. Sci. 2014, 27, 1819–1825. [Google Scholar] [PubMed]
- Iranshahy, M.; Iranshahi, M. Traditional uses, phytochemistry and pharmacology of asafoetida (Ferula assa-foetida oleo-gum-resin)—A review. J. Ethnopharmacol. 2011, 134, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Appendino, G.; Tagliapietra, S.; Nano, G.M.; Jakupovic, J. Sesquiterpene coumarin ethers from asafetida. Phytochemistry 1994, 35, 183–186. [Google Scholar] [CrossRef]
- Bandyopadhyay, D.; Basak, B.; Chatterjee, A.; Lai, T.K.; Banerji, A.; Banerji, J.; Neuman, A.; Prange, T. Saradaferin, a new sesquiterpenoid coumarin from Ferula assa-foetida. Nat. Prod. Res. 2006, 20, 961–965. [Google Scholar] [CrossRef] [PubMed]
- Iranshahi, M.; Masullo, M.; Asili, A.; Hamedzadeh, A.; Janabin, B.; Festa, M.; Capasso, A.; Piacente, S. Sesquiterpene coumarins from Ferula gumosa. J. Nat. Prod. 2010, 73, 1958–1962. [Google Scholar] [CrossRef] [PubMed]
- Iranshahi, M.; Kalategi, F.; Shebkar, A.; Sardashti, A.; Schneider, B. New sesquiterpene coumarins from roots of Ferula flabelliloba. Pharm. Biol. 2010, 48, 217–220. [Google Scholar] [CrossRef] [PubMed]
- Nazari, Z.E.; Iranshahi, M. Biologically active sesquiterpene coumarins from Ferula species. Phytother. Res. 2011, 25, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Iranshahi, M.; Barthomeuf, C.; Bayet-Robert, M.; Chollet, P.; Davoodi, D.; Piacente, S.; Rezaee, R.; Shebkar, A. Drimane-type sesquiterpene coumarines from Ferula gummosa fruits enhance doxorubicin uptake in doxorubincin-resistant human breast cancer cell line. J. Tradit. Complement. Med. 2014, 4, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.A. Sesquiterpene coumarins and sesquiterpenes from Ferula sinaica. Phytochemistry. 1999, 50, 109–112. [Google Scholar] [CrossRef]
- Marco, J.A.; Sanz, J.F.; Yuste, A.; Rustaiyan, A. New umbelliferon sesquiterpene ethers from roots of Ligularia persica. Liebigs. Ann. Chem. 1991, 9, 929–931. [Google Scholar] [CrossRef]
- Saidkhodzhaev, A.I.; Malikov, V.M. The stereochemistry of terpenoid coumarins. Chem. Nat. Prod. (Engl. Transl.) 1978, 14, 601–605. [Google Scholar] [CrossRef]
- Su, B.-N.; Takaishi, Y.; Honda, G.; Itoh, M.; Takeda, Y.; Kodzhimatov, O.K.; Ashurmetov, O. Sesquiterpene coumarins and related derivatives from Ferula pallida. J. Nat. Prod. 2000, 63, 436–440. [Google Scholar] [CrossRef] [PubMed]
- Bashir, S.; Alam, M.; Adhikari, A.; Shrestha, R.L. (Swagat); Yousuf, S.; Ahmad, B.; Parveen, S.; Aman, A.; Choudhary, M.I. New antileishmanial sesquiterpene coumarins from Ferula narthex Boiss. Phytochem. Lett. 2014, 9, 46–50. [Google Scholar] [CrossRef]
- Kajimoto, T.; Yahiro, K.; Nohara, T. Sesquiterpenoid and disulphide derivatives from Ferula assa-foetida. Phytochemistry 1989, 28, 1761–1763. [Google Scholar] [CrossRef]
- Abd EL-Razek, M.H.; Wu, Y.C.; Chang, F.R. Sesquiterpene coumarins from Ferula foetida. J. Chin. Chem. Soc. 2007, 54, 235–238. [Google Scholar] [CrossRef]
- Lee, C.L.; Chiang, L.C.; Cheng, L.H.; Liaw, C.C.; Abd El-Razek, M.H.; Chang, F.R.; Wu, Y.C. Influenza A (H1N1) antiviral and cytotoxic agents from Ferula assa-foetida. J. Nat. Prod. 2009, 72, 1568–1572. [Google Scholar] [CrossRef] [PubMed]
- Kiryalov, N.P.; Bagirov, V.Y. The structure of karatavicinol. Chem. Nat. Prod. (Engl. Transl.) 1979, 15, 191–192. [Google Scholar] [CrossRef]
- Al-Yahya, M.A.; Muhammad, I.; Mirza, H.H.; El-Feraly, F.S. Antibacterial Constituents from the Rhizomes of Ferula communis. Phytother. Res. 1998, 12, 335–339. [Google Scholar] [CrossRef]
- Dastan, D.; Salehi, P.; Gohari, A.R.; Ebrahimi, S.N.; Aliahmadi, A.; Hamburger, M. New coumarin derivatives from Ferula pseudalliacea with antibacterial activity. Planta Med. 2015, 80, 1118–1123. [Google Scholar]
- Iranshahi, M.; Arfa, P.; Ramezani, M.; Jaafiri, M.R.; Sadeghian, H.; Bassarello, C.; Piacente, S.; Pizza, C. Sesquiterpene coumarins from Ferula szowitsiana and in vitro antileishmanial activity of 7-prenyloxycoumarins against promastogotes. Phytochemistry 2007, 68, 554–561. [Google Scholar] [CrossRef] [PubMed]
- Barthomeuf, C.; Lim, S.; Iranshahi, M.; Chollet, P. Umbelliprenin from Ferula szowitsiana inhibits the growth of human M4Beu metastatic pigmented malignant melanoma cells through cell-cycle arrest in G1 and induction of caspase-dependent apoptosis. Phytomedicine 2008, 15, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Shakeri, A.; Iranshahy, M.; Iranshahi, M. Biological properties and molecular targets of umbelliprenin—A mini-review. J. Asian Nat. Prod. Res. 2014, 16, 884–889. [Google Scholar] [CrossRef] [PubMed]
- Rahbar, S. Novel inhibitors of glycation and AGE formation. Cell. Biochem. Biophys. 2007, 48, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Capistrano, I.R.; Wouters, A.; Foubert, K.; Baldé, A.M.; Apers, S.; Lardon, F.; Pieters, L.; Exarchou, V. Phytochemical characterisation of a cytotoxic stem bark extract of Steganotaenia araliacea and identification of a protoflavanone by LC-SPE-NMR. Phytochem. Lett. 2015, 12, 119–124. [Google Scholar] [CrossRef]
- Amin, A.; Tuenter, E.; Exarchou, V.; Upadhyay, A.; Cos, P.; Maes, L.; Apers, S.; Pieters, L. Phytochemical and pharmacological investigations on Nymphoides indica leaf extracts. Phytother. Res. 2016. [Google Scholar] [CrossRef] [PubMed]
- Baldé, E.S.; Megalizzi, V.; Traore, M.S.; Cos, P.; Maes, L.; Decaestecker, C.; Pieters, L.; Baldé, A.M. Antiprotozoal, antimicrobial and antitumor activities in vitro of Pavetta crassipes K. Schum leaves. J. Ethnopharmacol. 2010, 130, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Cos, P.; Vlietinck, A.J.; Vanden Berghe, D.; Maes, L. Anti-infective potential of natural products: How to develop a stronger in vitro “proof-of-concept”. J. Ethnopharmacol. 2006, 106, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Mesia, G.K.; Tona, G.L.; Nanga, T.H.; Cimanga, R.K.; Apers, S.; Cos, P.; Maes, L.; Pieters, L.; Vlietinck, A.J. Antiprotozoal and cytotoxic screening of 45 plant extracts from Democratic Republic of Congo. J. Ethnopharmacol. 2008, 115, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Rubiolo, P.; Matteodo, M.; Riccio, G.; Ballero, M.; Christen, P.; Fleury-Souverain, S.; Veuthey, J.L.; Bicchi, C. Analytical discrimination of poisonous and nonpoisonous chemotypes of giant fennel (Ferula communis L.) through their biologically active and volatile fractions. J. Agric. Food Chem. 2006, 54, 7556–7563. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are not available from the authors.
| Sample | IC50 μg/mL | ||||
|---|---|---|---|---|---|
| S. aureus | E. coli | C. albicans | M. canis | MRC-5 | |
| MeOH 90% | 19.9 | >64 | >64 | 22.0 | 36.5 |
| Chloroform | >64 | >64 | >64 | >64 | 49.8 |
| Ethyl acetate | >64 | >64 | >64 | >64 | >64 |
| n-Hexane | >64 | >64 | >64 | 8.3 | >64 |
| n-Butanol | >64 | >64 | >64 | >64 | >64 |
| Aqueous | >64 | >64 | >64 | >64 | >64 |
| Compound No. | IC50 (μM) | |||||
|---|---|---|---|---|---|---|
| MRC5 | Pf-K1 | T. bruc. | T. cruz. | L. inf. | PMM | |
| 1 | 8.0 | 22.4 | 8.1 | 8.6 | 6.8 | 8 |
| 3 | 31.7 | 7.4 | 32.4 | 19.1 | 12.7 | 32 |
| 4 | 11.7 | 1.3 | 32.6 | 10.5 | 12.7 | 32 |
| 5 | 20.4 | 16.0 | 32.4 | 9.4 | 32.4 | 32 |
| Compound No. | BSA-Glucose BSA-MGO | |||
|---|---|---|---|---|
| % Inhibition a | IC50 (mM) | % Inhibition b | IC50 (mM) | |
| 1 | 47 | - | 1.71 | |
| 2 | 0.41 | 40 | - | |
| 3 | 44 | - | 1.03 | |
| 4 | 1.83 | 35 | - | |
| 5 | 32 | - | - | 1.86 |
| 6 | 36 | - | 20 | - |
| Aminoguanidine | 1.75 | 0.15 | ||
| Quercetin | 0.23 | 0.35 | ||
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amin, A.; Tuenter, E.; Cos, P.; Maes, L.; Exarchou, V.; Apers, S.; Pieters, L. Antiprotozoal and Antiglycation Activities of Sesquiterpene Coumarins from Ferula narthex Exudate. Molecules 2016, 21, 1287. https://doi.org/10.3390/molecules21101287
Amin A, Tuenter E, Cos P, Maes L, Exarchou V, Apers S, Pieters L. Antiprotozoal and Antiglycation Activities of Sesquiterpene Coumarins from Ferula narthex Exudate. Molecules. 2016; 21(10):1287. https://doi.org/10.3390/molecules21101287
Chicago/Turabian StyleAmin, Adnan, Emmy Tuenter, Paul Cos, Louis Maes, Vassiliki Exarchou, Sandra Apers, and Luc Pieters. 2016. "Antiprotozoal and Antiglycation Activities of Sesquiterpene Coumarins from Ferula narthex Exudate" Molecules 21, no. 10: 1287. https://doi.org/10.3390/molecules21101287
APA StyleAmin, A., Tuenter, E., Cos, P., Maes, L., Exarchou, V., Apers, S., & Pieters, L. (2016). Antiprotozoal and Antiglycation Activities of Sesquiterpene Coumarins from Ferula narthex Exudate. Molecules, 21(10), 1287. https://doi.org/10.3390/molecules21101287