Natural Products for the Prevention and Treatment of Hangover and Alcohol Use Disorder
Abstract
1. Introduction
2. Natural Products with Anti-Hangover Properties
2.1. Pueraria Lobata
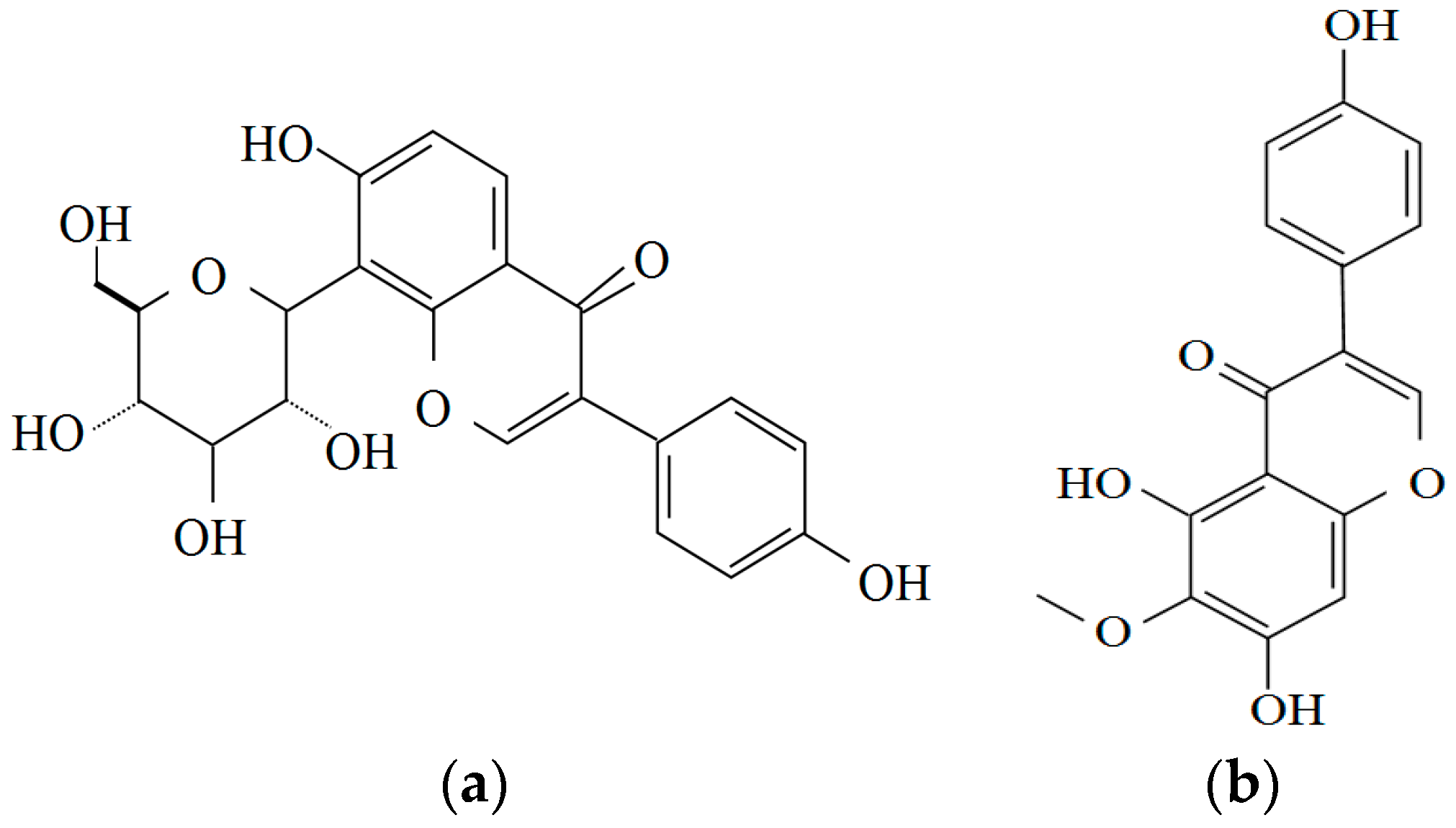
2.2. Fructus Evodiae
2.3. Trigonela Foenum-Graecum
2.4. Hovenia Dulcis
2.5. Pyrus Pyrifolia
2.6. Mangifera Indica L.
2.7. Diospyros Kaki Thunb.
2.8. Thymus Vulgaris
2.9. Zingiber Officinale
2.10. Asparagus Officinalis
2.11. Oenanthe Javanica
2.12. Opuntia Ficus-Indica
2.13. Panax Ginseng
3. Natural Plants for Alcohol Use Disorder
3.1. Hypericum Perforatum
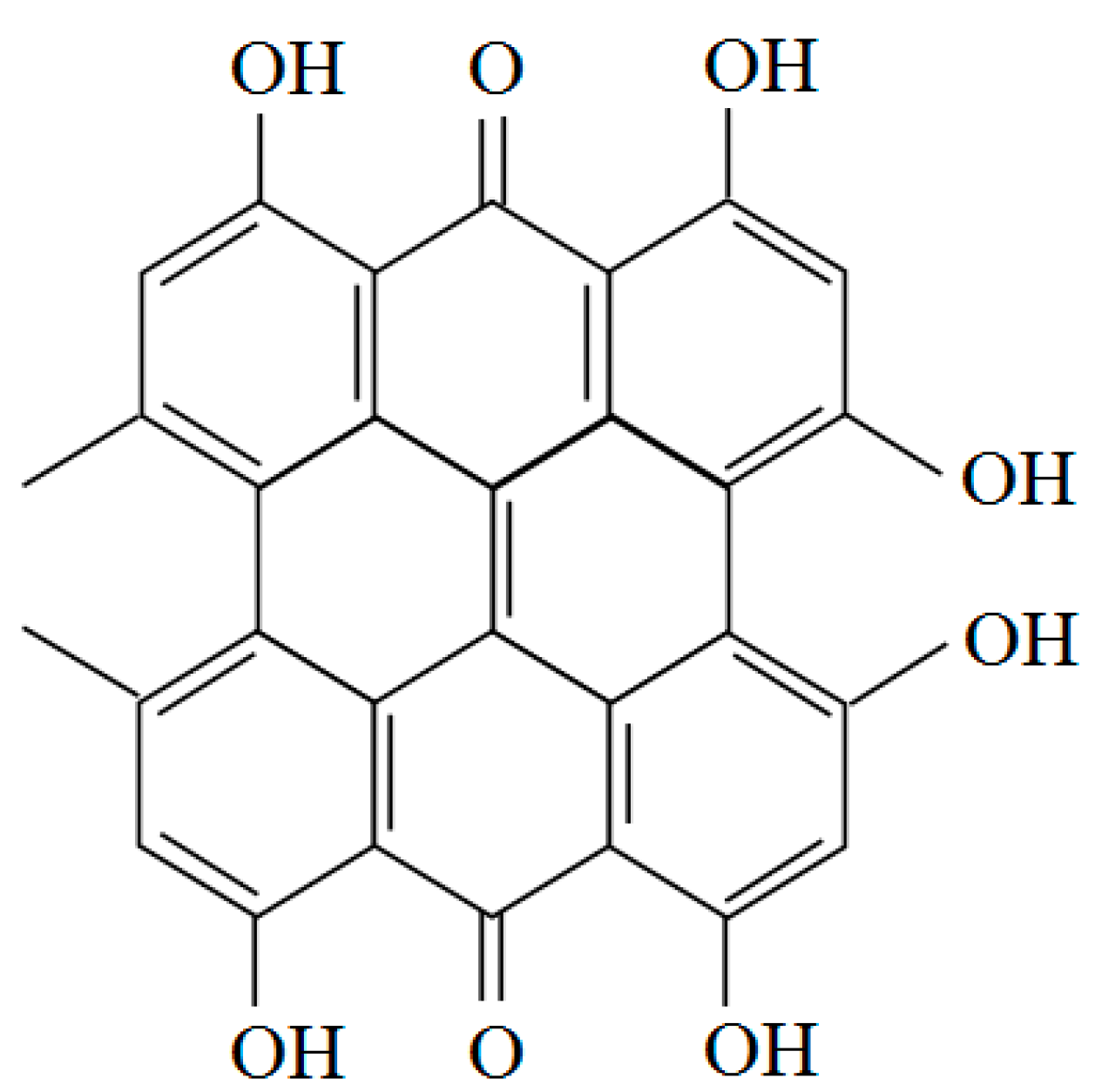
3.2. Salvia Miltiorrhiza
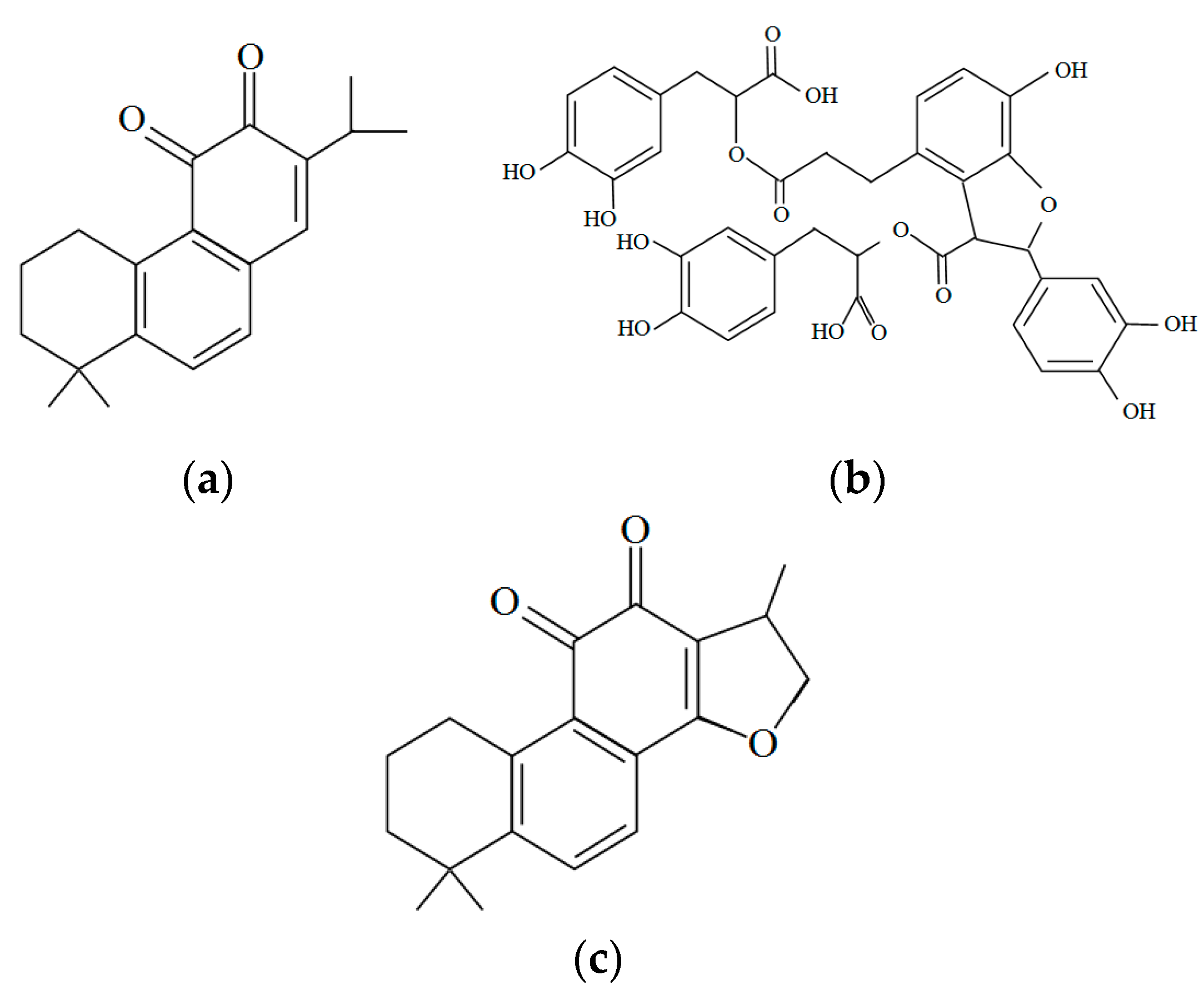
3.3. Scutellaria Baicalensis
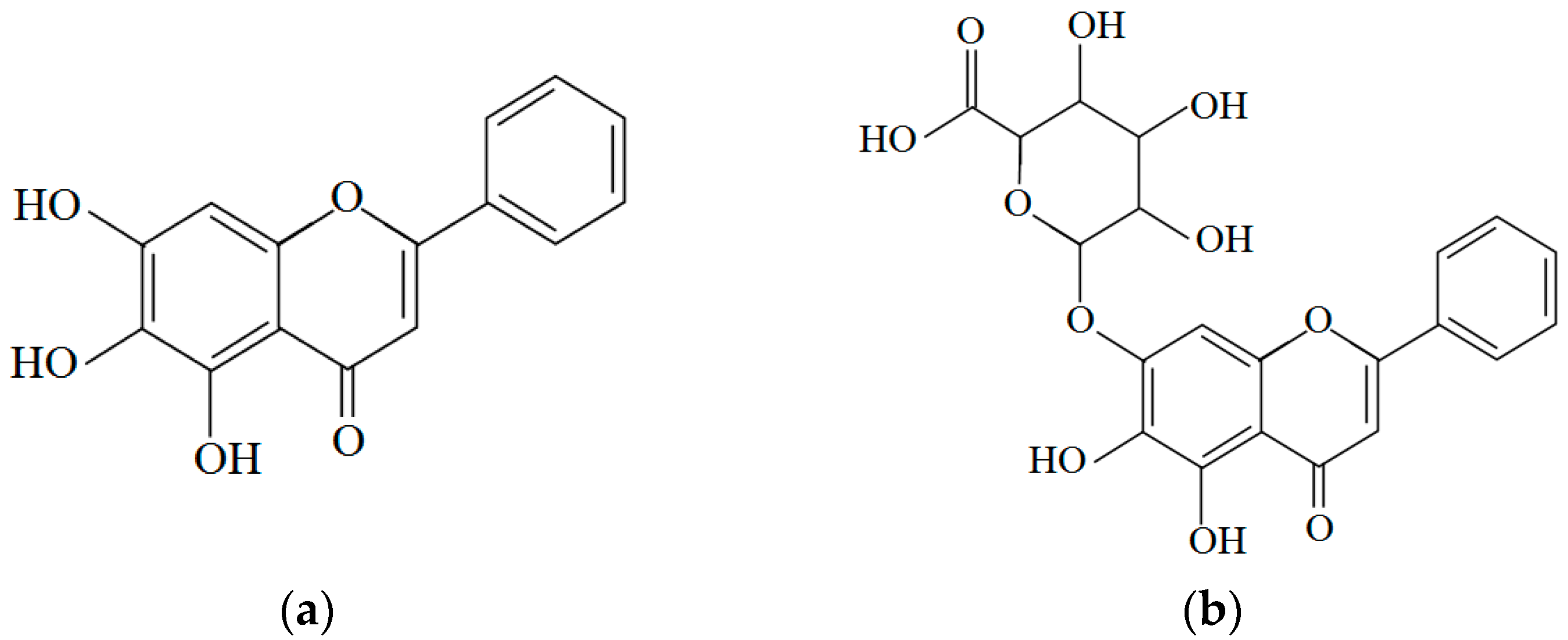
3.4. Rhizoma Coptidis
3.5. Other Natural Products for Alcohol Use Disorder
| Natural Products | Bioactive Components | Part of Plant Used | Subjects | Biological Effects and Molecular Mechanism(s) | Reference |
|---|---|---|---|---|---|
| Pueraria lobata | Tectoridin | Dried flower | Humans | Reduced hangover symptoms by promoting the elimination of blood acetaldehyde | [41] |
| Mice | Suppression of alcohol-induced liver steatosis by modulating the disturbance of the peroxisome proliferator-activated receptor α pathway and ameliorating mitochondrial function | [42] | |||
| Cells | Suppression of alcohol-induced apoptosis in human neuroblastoma cells | [43] | |||
| Puerarin and daidzein | Root | Humans | Reduced alcohol intake in a naturalistic setting | [45] | |
| Humans | Reduced alcohol consumption in a binge drinking paradigm | [46] | |||
| Rats | Reduced anxiogenic effects of alcohol withdrawal via increased social interaction and locomotor activity | [47] | |||
| Rats | Mitigation of liver damage (AST, ALT, GGT) and lipid deposition induced by chronic alcohol intake as well as TNF-α release, protein expression of endotoxin receptors | [48] | |||
| Fructus evodiae | Dehydroevodiamine, evodiamine and rutaecarpine | Dried and unripe fruit | Mice | Alleviation of hangover through stimulating the expression of hepatic alcohol metabolizing and antioxidant enzymes | [50] |
| Rats | Prevention of alcohol-induced gastric mucosal lesions by strengthening the mucosal barrier integrity and increasing gastric mucosal nitric oxide (NO) synthesis | [51] | |||
| Trigonela foenum-graecum | Polyphenols | Seeds | Cells | Prevention of the toxic effects of alcohol through increased cell viability, reduced lactate dehydrogenase leakage and normalized GSH/GSSG ratios | [53,55] |
| Rats | Suppressed alcohol-induced abnormalities in the liver through restoration of liver enzymes, ADH and ALDH activities | [54] | |||
| Rats | Suppression of alcohol toxicity through prevention of enzymatic leakage, and improved lipid profiles | [56,57,58] | |||
| Hovenia dulcis | Heteropolysaccharides | Peduncle | Mice | Suppression of acute alcohol-induced liver injury through decreased serum levels of AST, ALT and liver MDA, and restored liver SOD, GST and GSH | [63,64] |
| Dihydromyricetin | Rats | Reduced alcohol consumption in an intermittent voluntary alcohol intake paradigm | [65] | ||
| Pyrus pyrifolia | Polyphenols | Fruit | Humans | Alleviation of alcohol hangover through lowered blood alcohol levels and modifed genetic variation of ALDH2 | [67] |
| Mice | Alleviation of alcohol hangover through decreased blood alcohol levels | [68] | |||
| Mangifera indica L. | Polyphenols | Flesh and peel | Mice | Decreased plasma alcohol levels and increased activities of ADH and ALDH | [70] |
| Diospyros kaki Thunb. | Flavones and phenolics | Vinegar from fruit | Mice | Prevention of metabolic disorders induced by alcohol through blood alcohol clearance and decreased triglyceride and total cholesterol levels | [71] |
| Leave and fruit | Mice | Prevention of hepatic injury by accelerating alcohol metabolism, activating the antioxidative enzyme system and decreasing fat accumulation | [72] | ||
| Thymus vulgaris | Essential oil | Leave | Mice | Amelioration of liver and brain alcohol injuries through decreased NO and MDA levels and increased the total antioxidant capacity and GPX activity | [76] |
| Zingiber officinale | 6-gingerol | Rhizome | Mice | Amelioration of liver and brain alcohol injuries through decreasing L-γ-glutamyl transpeptidase and butyryl cholinesterase | [76] |
| Humans | Decreased signs and symptoms of alcohol hangover | [79] | |||
| Essential oil and citral | Mice | Hepatoprotective property against AFLD by decreasing levels of D-glucurono-6,3-lactone, glycerol-3-phosphate, and pyruvic acid in serum | [80] | ||
| Asparagus officinalis | Flavonoids | Shoots and the leaves | Cells | Alleviation of alcohol toxicity by upregulating the activities of ADH and ALDH | [82] |
| Oenanthe javanica | Caffeic acid | Leave and stem | Rats and mice | Alleviation of alcohol intoxication by accelerating alcohol metabolism | [84] |
| Opuntia ficus-indica | Flavones and phenolics | Cladode | Humans | Reduced hangover symptoms by inflammatory mediator production inhibition | [88,89] |
| Rats | Suppression of liver damage induced by alcohol through ending free radical chain reactions or enhancing the endogenous antioxidant activities | [90] | |||
| Panax ginseng | Ginsenosides | Root | Humans | Relief from hangover symptoms through reduced expiratory and plasma alcohol levels and hangover severity | [91] |
| Linoleic acid | Mice | Alleviation of hangover through reduced alcohol and acetaldehyde levels and enhanced ADH and ALDH activities | [92] | ||
| Mice | Suppressed alcohol-induced toxicity on male fertility | [93] | |||
| Ricefish | Suppressed alcohol-induced toxicity on embryogenesis | [94] | |||
| Mouse embryos | Suppressed alcohol-induced toxicity in the neurocranium through its effects on antioxidant activity | [95] | |||
| Rats | Suppressed alcohol-induced toxicity in the gastric system via the restoration of heat-shock proteins | [96] | |||
| Hypericum perforatum | Hypericin and hyperforin | Leave and flowering tops | Rats and mice | Reduced voluntary alcohol intake in acute and chronic alcohol treatment through neurochemical mechanisms | [101,104,105] |
| Rats | Attenuated alcohol withdrawal syndrome by inhibition of the effects on tremors and audiogenic seizures | [102] | |||
| Salvia miltiorrhiza | Miltirone | Root | Rats | Reduced alcohol intake and blood alcohol levels through curbing alcohol absorption | [110] |
| Rats | Reduced alcohol intake and delayed acquisition of alcohol-drinking behavior | [111] | |||
| Idn 5082 | Rats | Delayed acquisition of alcohol drinking behavior, and relapse prevention by suppressing the extra alcohol consumption after deprivation | [112,113] | ||
| salvianolic acid B | Rats | Attenuation of acute alcohol-induced hepatocyte apoptosis through SIRT1-mediated deacetylation of the p53 pathway | [115] | ||
| Scutellariae Radix | Baicalein, baicalin and wogonin | Root | Rats | Attenuated liver fibrosis through liver sinusoidal endothelial cell activation and HSC migration | [121] |
| Rhizoma coptidis | Berberine | Rhizome | Rats | Reduced alcohol intake and withdrawal induced hyperexcitability through its neuromodulatory action | [127,128] |
| Rats | Attenuated acute alcohol-induced gastrointestinal mucosa damage through regulation of cytokines | [129] | |||
| Stephania ambigua and Corydalis teranda | Levo-tetrahydro-palmatine | Rats | Reduced alcohol intake through dopamine D2 receptor-mediated PKA signaling in caudate-putamen | [130] | |
| Strychnos nux-vomica L. | Brucine | Fruit | Rats | Decreased alcohol consumption through glycine receptor antagonist | [131] |
| Tabernanthe iboga | Ibogaine | Root | Rats | Reduced alcohol intake through mediation of glial cell line-derived neurotrophic factor | [132] |
| Jodina rhombifolia | Unclear | Leave | Rats | Reduced alcohol intake without tolerance and apparent side-effects | [134,135] |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Costanzo, S.; di Castelnuovo, A.; Donati, M.B.; Iacoviello, L.; de Gaetano, G. Alcohol consumption and mortality in patients with cardiovascular disease a meta-analysis. J. Am. Coll. Cardiol. 2010, 55, 1339–1347. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Sola, J. Cardiovascular risks and benefits of moderate and heavy alcohol consumption. Nat. Rev. Cardiol. 2015, 12, 576–587. [Google Scholar] [CrossRef] [PubMed]
- Lippi, G.; Mattiuzzi, C.; Franchini, M. Alcohol consumption and venous thromboembolism: Friend or foe? Intern. Emerg. Med. 2015, 10, 907–913. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Drca, N.; Wolk, A. Alcohol consumption and risk of atrial fibrillation a prospective study and dose-response meta-analysis. J. Am. Coll. Cardiol. 2014, 64, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Holmes, M.V.; Dale, C.E.; Zuccolo, L.; Silverwood, R.J.; Guo, Y.; Ye, Z.; Prieto-Merino, D.; Dehghan, A.; Trompet, S.; Wong, A.; et al. Association between alcohol and cardiovascular disease: Mendelian randomisation analysis based on individual participant data. BMJ 2014, 349, g4164. [Google Scholar] [CrossRef] [PubMed]
- Woo, Y.S.; Yoon, S.J.; Lee, H.K.; Lee, C.U.; Chae, J.H.; Lee, C.T.; Kim, D.J. Concentration changes of malcohol in blood samples during an experimentally induced alcohol hangover state. Addict. Biol. 2005, 10, 351–355. [Google Scholar] [CrossRef] [PubMed]
- Dueland, A.N. Headache and alcohol. Headache 2015, 55, 1045–1049. [Google Scholar] [CrossRef] [PubMed]
- Hartung, B.; Schwender, H.; Mindiashvili, N.; Ritz-Timme, S.; Malczyk, A.; Daldrup, T. The effect of alcohol hangover on the ability to ride a bicycle. Int. J. Legal. Med. 2015, 129, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Wiese, J.G.; Shlipak, M.G.; Browner, W.S. The alcohol hangover. Ann. Intern. Med. 2000, 132, 897–902. [Google Scholar] [CrossRef] [PubMed]
- Rosa-e-Silva, L.; Troncon, L.E.; Oliveira, R.B.; Gallo, L.; Foss, M.C. Fecal parameters and gastrointestinal transit in patients with alcohol related chronic pancreatitis with and without chronic diarrhea: Factors associated with this symptom. Gastroenterology 2013, 1441, S457. [Google Scholar] [CrossRef]
- Chiang, C.P.; Wu, C.W.; Lee, S.P.; Ho, J.L.; Lee, S.L.; Nieh, S.; Yin, S.J. Expression pattern, alcohol-metabolizing activities, and cellular localization of alcohol and aldehyde dehydrogenases in human small intestine. Alcohol. Clin. Exp. Res. 2012, 36, 2047–2058. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, L.M.; Persson, E.C.; Weinstein, S.J.; Graubard, B.I.; Freedman, N.D.; Mannisto, S.; Albanes, D.; McGlynn, K.A. Alcohol consumption, one-carbon metabolites, liver cancer and liver disease mortality. PLoS ONE 2013, 8, e78156. [Google Scholar] [CrossRef] [PubMed]
- Kouda, K.; Iki, M.; Fujita, Y.; Tamaki, J.; Yura, A.; Kadowaki, E.; Sato, Y.; Moon, J.S.; Morikawa, M.; Tomioka, K.; et al. Alcohol intake and bone status in elderly Japanese men: Baseline data from the Fujiwara-kyo osteoporosis risk in men (Formen) study. Bone 2011, 49, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Handing, E.P.; Andel, R.; Kadlecova, P.; Gatz, M.; Pedersen, N.L. Midlife alcohol consumption and risk of dementia over 43 years of follow-up: A population-based study from the Swedish twin registry. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2015, 70, 1248–1254. [Google Scholar] [CrossRef] [PubMed]
- Langballe, E.M.; Ask, H.; Holmen, J.; Stordal, E.; Saltvedt, I.; Selbaek, G.; Fikseaunet, A.; Bergh, S.; Nafstad, P.; Tambs, K. Alcohol consumption and risk of dementia up to 27 years later in a large, population-based sample: the HUNT study, Norway. Eur. J. Epidemiol. 2015, 30, 1049–1056. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y. Alcohol and neurological diseases. Nichidai Igaku Zasshi 2011, 70, 150–154. [Google Scholar] [CrossRef]
- Scoccianti, C.; Straif, K.; Romieu, I. Recent evidence on alcohol and cancer epidemiology. Future Oncol. 2013, 9, 1315–1322. [Google Scholar] [CrossRef] [PubMed]
- Barker, J.M.; Taylor, J.R. Habitual alcohol seeking: Modeling the transition from casual drinking to addiction. Neurosci. Biobehav. Rev. 2014, 47, 281–294. [Google Scholar] [CrossRef] [PubMed]
- Rehm, J.; Mathers, C.; Popova, S.; Thavorncharoensap, M.; Teerawattananon, Y.; Patra, J. Alcohol and global health 1 global burden of disease and injury and economic cost attributable to alcohol use and alcohol-use disorders. Lancet 2009, 373, 2223–2233. [Google Scholar] [CrossRef]
- Li, S.; Gan, L.Q.; Li, S.K.; Zheng, J.C.; Xu, D.P.; Li, H.B. Effects of herbal infusions, tea and carbonated beverages on alcohol dehydrogenase and aldehyde dehydrogenase activity. Food Funct. 2014, 5, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, C.J.P. Genetic-epidemiological evidence for the role of acetaldehyde in cancers related to alcohol drinking. Adv. Exp. Med. Biol. 2015, 815, 41–58. [Google Scholar]
- Brooks, P.J.; Zakhari, S. Acetaldehyde and the genome: Beyond nuclear DNA adducts and carcinogenesis. Environ. Mol. Mutagen. 2014, 55, 77–91. [Google Scholar] [CrossRef] [PubMed]
- McCarty, M.F. Nutraceutical strategies for ameliorating the toxic effects of alcohol. Med. Hypotheses 2013, 80, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Seth, D.; Haber, P.S.; Syn, W.K.; Diehl, A.M.; Day, C.P. Pathogenesis of alcohol-induced liver disease: Classical concepts and recent advances. J. Gastroen. Hepatol. 2011, 26, 1089–1105. [Google Scholar] [CrossRef] [PubMed]
- Karadayian, A.G.; Bustamante, J.; Czerniczyniec, A.; Lombardi, P.; Cutrera, R.A.; Lores-Arnaiz, S. Alcohol hangover induces mitochondrial dysfunction and free radical production in mouse cerebellum. Neuroscience 2015, 304, 47–59. [Google Scholar] [CrossRef]
- Park, S.K.; Qi, X.F.; Song, S.B.; Kim, D.H.; Teng, Y.C.; Yoon, Y.S.; Kim, K.Y.; Li, J.H.; Jin, D.; Lee, K.J. Electrolyzed-reduced water inhibits acute alcohol-induced hangovers in Sprague-Dawley rats. Biomed. Res. 2009, 30, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Li, A.N.; Li, S.; Zhang, Y.J.; Xu, X.R.; Chen, Y.M.; Li, H.B. Resources and biological activities of natural polyphenols. Nutrients 2014, 6, 6020–6047. [Google Scholar] [CrossRef] [PubMed]
- Maher, J.J. Treatment of alcoholic hepatitis. J. Gastroen. Hepatol. 2002, 17, 448–455. [Google Scholar] [CrossRef] [Green Version]
- Li, A.N.; Li, S.; Li, H.B.; Xu, D.P.; Xu, X.R.; Chen, F. Total phenolic contents and antioxidant capacities of 51 edible and wild flowers. J. Funct. Foods 2014, 6, 319–330. [Google Scholar] [CrossRef]
- Van de Loo, A.J.A.E.; Hogewoning, A.; Raasveld, S.J.; de Zeeuw, R.; Bosma, E.R.; Bouwmeester, N.H.; Lukkes, M.; Brookhuis, K.A.; Knipping, K.; Garssen, J.; et al. Saliva cytokine concentrations the day after heavy alcohol consumption in drinkers suffering from a hangover versus those who claim to be hangover resistant. Alcohol Alcohol. 2015, 50 (Suppl. 1), FOC7–2. [Google Scholar]
- Rosenthal, R.N. Current and future drug therapies for alcohol dependence. J. Clin. Psychopharmacol. 2006, 26 (Suppl. 1), 20–29. [Google Scholar] [CrossRef]
- Carai, M.A.M.; Agabio, R.; Bombardelli, E.; Bourov, I.; Gessa, G.L.; Lobina, C.; Morazzoni, P.; Pani, M.; Reali, R.; Vacca, G.; Colombo, G. Potential use of medicinal plants in the treatment of alcoholism. Fitoerapia 2000, 71 (Suppl. 1), 38–42. [Google Scholar] [CrossRef]
- Okoli, C.O.; Akah, P.A. A pilot evaluation of the anti-inflammatory activity of Culcasia scandens, a traditional antirheumatic agent. J. Altern. Complement. Med. 2000, 6, 423–427. [Google Scholar] [CrossRef]
- Xu, B.J.; Zheng, Y.N.; Sung, C.K. Natural medicines for alcoholism treatment: a review. Drug Alcohol Rev. 2005, 24, 525–536. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.; Xu, B.T.; Gan, R.Y.; Zhang, Y.; Xu, X.R.; Xia, E.Q.; Li, H.B. Total phenolic contents and antioxidant capacities of herbal and tea infusions. Int. J. Mol. Sci. 2011, 12, 2112–2124. [Google Scholar] [CrossRef] [PubMed]
- Deng, G.F.; Lin, X.; Xu, X.R.; Gao, L.L.; Xie, J.F.; Li, H.B. Antioxidant capacities and total phenolic contents of 56 vegetables. J. Funct. Foods 2013, 5, 260–266. [Google Scholar] [CrossRef]
- Fu, L.; Xu, B.T.; Xu, X.R.; Gan, R.Y.; Zhang, Y.; Xia, E.Q.; Li, H.B. Antioxidant capacities and total phenolic contents of 62 fruits. Food Chem. 2011, 129, 345–350. [Google Scholar] [CrossRef]
- Li, S.; Li, S.K.; Gan, R.Y.; Song, F.L.; Kuang, L.; Li, H.B. Antioxidant capacities and total phenolic contents of infusions from 223 medicinal plants. Ind. Crop. Prod. 2013, 51, 289–298. [Google Scholar] [CrossRef]
- Marino, M.D.; Aksenov, M.Y.; Kelly, S.J. Vitamin E protects against alcohol-induced cell loss and oxidative stress in the neonatal rat hippocampus. Int. J. Dev. Neurosci. 2004, 22, 363–377. [Google Scholar] [CrossRef] [PubMed]
- McGregor, N.R. Pueraria lobata (Kudzu root) hangover remedies and acetaldehyde-associated neoplasm risk. Alcohol 2007, 41, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, T.; Hosono, T.; Matsushita, Y.; Kawashima, K.; Someya, M.; Nakajima, Y.; Narui, K.; Hibi, Y.; Ishizaki, M.; Kinjo, J.; et al. Pharmacological studies on Puerariae Flos IV: Effects of Pueraria thomsonii dried flower extracts on blood alcohol and acetaldehyde levels in humans. Int. J. Clin. Pharmacol. Res. 2002, 22, 23–28. [Google Scholar] [PubMed]
- Xiong, Y.; Yang, Y.Q.; Yang, J.; Chai, H.Y.; Li, Y.; Yang, J.; Jia, Z.M.; Wang, Z.R. Tectoridin, an isoflavone glycoside from the flower of Pueraria lobata, prevents acute alcohol-induced liver steatosis in mice. Toxicology 2010, 276, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Jang, M.H.; Shin, M.C.; Kim, Y.J.; Chung, J.H.; Yim, S.V.; Kim, E.H.; Kim, Y.; Kim, C.J. Protective effects of Puerariae flos against alcohol-induced apoptosis on human neuroblastoma cell line SK-N-MC. Jpn. J. Pharmacol. 2001, 87, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Olsen, R.W. Alcohol use disorders and current pharmacological therapies: the role of GABAA receptors. Acta Pharmacol. Sin. 2014, 8, 981–993. [Google Scholar] [CrossRef] [PubMed]
- Lukas, S.E.; Penetar, D.; Berko, J.; Vicens, L.; Palmer, C.; Mallya, G.; Macklin, E.A.; Lee, D.Y.W. An extract of the Chinese herbal root Kudzu reduces alcohol drinking by heavy drinkers in a naturalistic setting. Alcohol Clin. Exp. Res. 2005, 29, 756–762. [Google Scholar] [CrossRef] [PubMed]
- Penetar, D.M.; Toto, L.H.; Lee, D.Y.W.; Lukas, S.E. A single dose of kudzu extract reduces alcohol consumption in a binge drinking paradigm. Drug Alcohol Depen. 2015, 153, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Overstreet, D.H.; Kralic, J.E.; Morrow, A.L.; Ma, Z.Z.; Zhang, Y.W.; Lee, D.Y. NPI-031G (puerarin) reduces anxiogenic effects of alcohol withdrawal or benzodiazepine inverse or 5-HT2C agonists. Pharmacol. Biochem. Behav. 2003, 75, 619–625. [Google Scholar] [CrossRef]
- Li, R.; Xu, L.Y.; Liang, T.; Li, Y.W.; Zhang, S.J.; Duan, X.Q. Puerarin mediates hepatoprotection against CCl4-induced hepatic fibrosis rats through attenuation of inflammation response and amelioration of metabolic function. Food Chem. Toxicol. 2013, 52, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.H.; Cui, T.; Sun, Z.L.; Huang, F.; Chen, L.; Xu, L.; Feng, Q.; Hu, Y.Y. Effects of Puerariae Radix extract on endotoxin receptors and TNF-α expression induced by gut-derived endotoxin in chronic alcoholic liver injury. Evid. Based Complement. Altern. Med. 2012, 2012. Article ID 234987. [Google Scholar] [CrossRef] [PubMed]
- Schramm, A.; Hamburger, M. Gram-scale purification of dehydroevodiamine from Evodia rutaecarpa fruits and a procedure for selective removal of quaternary indoloquinazoline alkaloids from Evodia extracts. Fitoterapia 2014, 94, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.H.; Shim, S.M.; Lee, S.R.; Mar, W.; Kim, G.H. Effect of Evodiae fructus extracts on gene expressions related with alcohol metabolism and antioxidation in alcohol-loaded mice. Food Chem. Toxicol. 2005, 43, 1365–1371. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Wu, D.Z.; Yuan, J.Y.; Zhang, R.R.; Hu, Z.B. Gastroprotective effect of Fructus Evodiae water extract on alcohol-induced gastric lesions in rats. Am. J. Chin. Med. 2006, 34, 1027–1035. [Google Scholar] [CrossRef] [PubMed]
- Kaviarasan, S.; Ramamurty, N.; Gunasekaran, P.; Varalakshmi, E.; Anuradha, C.V. Fenugreek (Trigonella foenum graecum) seed extract prevents alcohol-induced toxicity and apoptosis in chang liver cells. Alcohol Alcohol. 2006, 41, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Kaviarasan, S.; Anuradha, C.V. Fenugreek (Trigonella foenum graecum) seed polyphenols protect liver from alcohol toxicity: A role on hepatic detoxification system and apoptosis. Pharmazie 2007, 62, 299–304. [Google Scholar] [PubMed]
- Kaviarasan, S.; Ramamurthy, N.; Gunasekaran, P.; Varalakshmi, E.; Anuradha, C.V. Induction of alcohol-metabolizing enzymes and heat shock protein expression by alcohol and modulation by fenugreek seed polyphenols in chang liver cells. Toxicol. Mech. Methods 2009, 19, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Kaviarasan, S.; Viswanathan, P.; Anuradha, C.V. Fenugreek seed (Trigonella foenum graecum) polyphenols inhibit alcohol-induced collagen and lipid accumulation in rat liver. Cell Biol. Toxicol. 2007, 23, 373–383. [Google Scholar] [CrossRef] [PubMed]
- Thirunavukkarasu, V.; Anuradha, C.V.; Viswanathan, P. Protective effect of fenugreek (Trigonella foenum graecum) seeds in experimental alcohol toxicity. Phytother. Res. 2003, 17, 737–743. [Google Scholar] [CrossRef] [PubMed]
- Kaviarasan, S.; Sundarapandiyan, R.; Anuradha, C.V. Protective action of fenugreek (Trigonella foenum graecum) seed polyphenols against alcohol-induced protein and lipid damage in rat liver. Cell Biol. Toxicol. 2008, 24, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Pribac, G.C.; Sferdian, M.F.; Neamtu, C.; Craciun, C.; Rosioru, C.L.; Ardelean, A.; Totolici, B.D. Fenugreek powder exerts protective effects on alcoholised rats' kidney, highlighted using ultrastructural studies. Rom. J. Morphol. Embryol. 2015, 56, 445–451. [Google Scholar]
- Wang, M.C.; Jiang, C.X.; Ma, L.P.; Zhang, Z.J.; Cao, L.; Liu, J.; Zeng, X.X. Preparation, preliminary characterization and immunostimulatory activity of polysaccharide fractions from the peduncles of Hovenia dulcis. Food Chem. 2013, 138, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Hyun, T.K.; Eom, S.H.; Yu, C.Y.; Roitsch, T. Hovenia dulcis—An Asian traditional herb. Planta Med. 2010, 76, 943–949. [Google Scholar] [CrossRef] [PubMed]
- Na, C.S.; Yoon, S.Y.; Kim, J.B.; Na, D.S.; Dong, M.S.; Lee, M.Y.; Hong, C.Y. Anti-fatigue activity of Hovenia dulcis on a swimming mouse model through the inhibition of stress hormone expression and antioxidation. Am. J. Chin. Med. 2013, 41, 945–955. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.C.; Zhu, P.L.; Jiang, C.X.; Ma, L.P.; Zhang, Z.J.; Zeng, X.X. Preliminary characterization, antioxidant activity in vitro and hepatoprotective effect on acute alcohol-induced liver injury in mice of polysaccharides from the peduncles of Hovenia dulcis. Food Chem. Toxicol. 2012, 50, 2964–2970. [Google Scholar] [CrossRef] [PubMed]
- Du, J.A.; He, D.; Sun, L.N.; Han, T.; Zhang, H.; Qin, L.P.; Rahman, K. Semen Hoveniae extract protects against acute alcohol-induced liver injury in mice. Pharm. Biol. 2010, 48, 953–958. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Lindemeyer, A.K.; Gonzalez, C.; Shao, X.S.M.; Spigelman, I.; Olsen, R.W.; Liang, J. Dihydromyricetin as a novel anti-alcohol intoxication medication. J. Neurosci. 2012, 32, 390–401. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.B.; Choi, H.A.; Han, H.S.; Park, J.H.; Kim, S.; Bae, J.H.; Kim, H.K.; Choi, C. Polyphenolic compounds from Korean pear and their biological activities. Food Sci. Biotechnol. 2003, 12, 262–267. [Google Scholar]
- Lee, H.S.; Isse, T.; Kawamoto, T.; Baik, H.W.; Park, J.Y.; Yang, M. Effect of Korean pear (Pyruspyrifolia cv. Shingo) juice on hangover severity following alcohol consumption. Food Chem. Toxicol. 2013, 58, 101–106. [Google Scholar] [PubMed]
- Lee, H.S.; Isse, T.; Kawamoto, T.; Woo, H.S.; Kim, A.K.; Park, J.Y.; Yang, M. Effects and action mechanisms of Korean pear (Pyrus pyrifolia cv. Shingo) on alcohol detoxification. Phytother. Res. 2012, 26, 1753–1758. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M.; Poovarodom, S.; Leontowicz, H.; Leontowicz, M.; Vearasilp, S.; Trakhtenberg, S.; Gorinstein, S. The multiple nutrition properties of some exotic fruits: Biological activity and active metabolites. Food Res. Int. 2011, 44, 1671–1701. [Google Scholar] [CrossRef]
- Kim, S.H.; Cho, S.K.; Min, T.S.; Kim, Y.; Yang, S.O.; Kim, H.S.; Hyun, S.H.; Kim, H.; Kim, Y.S.; Choi, H.K. Ameliorating effects of Mango (Mangifera indica L.) fruit on plasma alcohol level in a mouse model assessed with H-1-NMR based metabolic profiling. J. Clin. Biochem. Nutr. 2011, 48, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Moon, Y.J.; Cha, Y.S. Effects of persimmon-vinegar on lipid metabolism and alcohol clearance in chronic alcohol-fed rats. J. Med. Food 2008, 11, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.S.; Kim, M.J.; Ma, J. Protective effects of persimmon leaf and fruit extracts against acute alcohol-induced hepatotoxicity. Prev. Nutr. Food Sci. 2007, 12, 202–208. [Google Scholar]
- Undeger, U.; Basaran, A.; Degen, G.H.; Basaran, N. Antioxidant activities of major thyme ingredients and lack of (oxidative) DNA damage in V79 Chinese hamster lung fibroblast cells at low levels of carvacrol and thymol. Food Chem. Toxicol. 2009, 47, 2037–2043. [Google Scholar] [CrossRef] [PubMed]
- Asbaghian, S.; Shafaghat, A.; Zarea, K.; Kasimov, F.; Salimi, F. Comparison of volatile constituents, and antioxidant and antibacterial activities of the essential oils of Thymus caucasicus, T. kotschyanus and T. vulgaris. Nat. Prod. Commun. 2011, 6, 137–140. [Google Scholar] [PubMed]
- Baranauskiene, R.; Venskutonis, P.R.; Viskelis, P.; Dambrauskiene, E. Influence of nitrogen fertilizers on the yield and composition of thyme (Thymus vulgaris). J. Agric. Food Chem. 2003, 51, 7751–7758. [Google Scholar] [CrossRef] [PubMed]
- Shati, A.A.; Elsaid, F.G. Effects of water extracts of thyme (Thymus vulgaris) and ginger (Zingiber officinale Roscoe) on alcohol abuse. Food Chem. Toxicol. 2009, 47, 1945–1949. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Li, X.C.; Chen, L.; Lu, W.Z.; Chen, X.W.; Han, L.; Chen, D.F. Protective effect against hydroxyl radical-induced DNA damage and antioxidant mechanism of 6-gingerol: A chemical study. Bull. Korean Chem. Soc. 2014, 35, 1633–1638. [Google Scholar] [CrossRef]
- Shanmugam, K.R.; Ramakrishna, C.H.; Mallikarjuna, K.; Reddy, K.S. Protective effect of ginger against alcohol-induced renal damage and antioxidant enzymes in male albino rats. Indian J. Exp. Biol. 2010, 48, 143–149. [Google Scholar] [PubMed]
- Takahashi, M.; Li, W.; Koike, K.; Sadamoto, K. Clinical effectiveness of KSS formula, a traditional folk remedy for alcohol hangover symptoms. J. Nat. Med. 2010, 64, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.T.; Raghu, R.; Lin, S.H.; Wang, S.Y.; Kuo, C.H.; Tseng, Y.F.J.; Sheen, L.Y. Metabolomics of ginger essential oil against alcoholic fatty liver in mice. J. Agric. Food Chem. 2013, 61, 11231–11240. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Castilla, S.; de la Puerta, R.; Gimenez, M.D.G.; Fernandez-Arche, M.A.; Guillen-Bejarano, R. Bioactive constituents from “Triguero” asparagus improve the plasma lipid profile and liver antioxidant status in hypercholesterolemic rats. Int. J. Mol. Sci. 2013, 14, 21227–21239. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.Y.; Cui, Z.G.; Lee, S.R.; Kim, S.J.; Kang, H.K.; Lee, Y.K.; Park, D.B. Effects of Asparagus officinalis extracts on liver cell toxicity and alcohol metabolism. J. Food Sci. 2009, 74, H204–H208. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.A.; Jung, Y.S.; Lee, S.J.; Park, S.C.; Kim, M.J.; Lee, E.J.; Byun, H.J.; Jhee, K.H.; Lee, S.P. Hepatoprotective effects of fermented field water-dropwort (Oenanthe javanica) extract and its major constituents. Food Chem. Toxicol. 2014, 67, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Kim, K.H.; Lee, Y.J.; Lee, S.H.; Park, J.C.; Nam, D.H. Oenanthe javanica extract accelerates alcohol metabolism in alcohol-treated animals. BMB Rep. 2009, 42, 482–485. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; You, Y.; Hwang, K.; Lee, J.; Chun, J.; Chung, J.W.; Shim, S.; Park, C.S.; Jun, W. Isolation and identification of compound from dropwort (Oenanthe javanica) with protective potential against oxidative stress in HepG2 cells. Food Sci. Biotechnol. 2011, 20, 1743–1746. [Google Scholar] [CrossRef]
- Lim, H.K.; Kim, H.S.; Kim, S.H.; Chang, M.J.; Rhee, G.S.; Choi, J. Protective effects of acetylbergenin against carbon tetrachloride-induced hepatotoxicity in rats. Arch. Pharm. Res. 2001, 24, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Teresa Sumaya-Martinez, M.T.; Cruz-Jaime, S.; Madrigal-Santillan, E.; Diego Garcia-Paredes, J.D.; Carino-Cortes, R.; Cruz-Cansino, N.; Valadez-Vega, C.; Martinez-Cardenas, L.; Alanis-Garcia, E. Betalain, acid ascorbic, phenolic contents and antioxidant properties of purple, red, yellow and white cactus pears. Int. J. Mol. Sci. 2011, 12, 6452–6468. [Google Scholar] [CrossRef] [PubMed]
- Wiese, J.; McPherson, S.; Odden, M.C.; Shlipak, M.G. Effect of Opuntia ficus indica on symptoms of the alcohol hangover. Arch. Intern. Med. 2004, 164, 1334–1340. [Google Scholar] [CrossRef] [PubMed]
- Wiese, J.; Mcpherson, S.; Shlipak, M. The effect of Opuntia ficus indica on the severity of the alcohol hangover. J. Gen. Intern. Med. 2002, 171, 134. [Google Scholar]
- Alimi, H.; Hfaeidh, N.; Mbarki, S.; Bouoni, Z.; Sakly, M.; Ben Rouma, K. Evaluation of Opuntia ficus indica f. inermis fruit juice hepatoprotective effect upon alcohol toxicity in rats. Gen. Physiol. Biophys. 2012, 31, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.H.; Kwak, J.H.; Jeon, G.; Lee, J.W.; Seo, J.H.; Lee, H.S.; Lee, J.H. Red ginseng relieves the effects of alcohol consumption and hangover symptoms in healthy men: A randomized crossover study. Food Funct. 2014, 5, 528–534. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.I.; Kim, S.T.; Lee, D.H.; Yu, J.M.; Jang, S.K.; Joo, S.S. Ginsenoside-free molecules from steam-dried ginseng berry promote alcohol metabolism: An alternative choice for an alcohol hangover. J. Food Sci. 2014, 79, C1323–C1330. [Google Scholar] [CrossRef] [PubMed]
- Jang, M.; Min, J.W.; In, J.G.; Yang, D.C. Effects of red ginseng extract on the epididymal sperm motility of mice exposed to alcohol. Int. J. Toxicol. 2011, 30, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Haron, M.H.; Avula, B.; Khan, I.A.; Mathur, S.K.; Dasmahapatra, A.K. Modulation of alcohol toxicity by Asian ginseng (Panax ginseng) in Japanese ricefish (Oryzias latipes) embryogenesis. Comp. Biochem. Phys. C 2013, 157, 287–297. [Google Scholar]
- Lee, S.R.; Kim, M.R.; Yon, J.M.; Baek, I.J.; Park, C.G.; Lee, B.J.; Yun, Y.W.; Nam, S.Y. Black ginseng inhibits alcohol-induced teratogenesis in cultured mouse embryos through its effects on antioxidant activity. Toxicol. Vitro 2009, 23, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Yeo, M.; Kim, D.K.; Cho, S.W.; Do Hong, H. Ginseng, the root of Panax ginseng C.A. Meyer, protects alcohol-induced gastric damages in rat through the induction of cytoprotective heat-shock protein 27. Dig. Dis. Sci. 2008, 53, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Arias, A.J.; Gelernter, J.; Gueorguieva, R.; Ralevski, E.; Petrakis, I.L. Pharmacogenetics of naltrexone and disulfiram in alcohol dependent, dually diagnosed veterans. Am. J. Addict. 2014, 23, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Stromberg, M.F.; Mackler, S.A.; Volpicelli, J.R.; O’Brien, C.P. Effect of acamprosate and naltrexone, alone or in combination, on alcohol consumption. Alcohol 2001, 23, 109–116. [Google Scholar] [CrossRef]
- Johnson, B.A.; Ait-Daoud, N.; Akhtar, F.Z.; Ma, J.Z. Oral topiramate reduces the consequences of drinking and improves the quality of life of alcohol-dependent individuals—A randomized controlled trial. Arch. Gen. Psychiatry 2004, 61, 905–912. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Mdzinarishvili, A.; Kiewert, C.; Abbruscato, T.; Bickel, U.; van der Schyf, C.J.; Klein, J. NMDA receptor-antagonistic properties of hyperforin, a constituent of St. John’s wort. J. Pharm. Sci. 2006, 102, 47–54. [Google Scholar] [CrossRef]
- Perfumi, M.; Mattioli, L.; Cucculelli, M.; Massi, M. Reduction of alcohol intake by chronic treatment with Hypericum perforatum, alone or combined with naltrexone in rats. J. Psychopharmacol. 2005, 19, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Coskun, I.; Uzbay, I.T.; Ozturk, N.; Ozturk, Y. Attenuation of alcohol withdrawal syndrome by extract of Hypericum perforatum in Wistar rats. Fund. Clin. Pharmacol. 2006, 20, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Bozin, B.; Kladar, N.; Grujic, N.; Anackov, G.; Samojlik, I.; Gavaric, N.; Conic, B.S. Impact of origin and biological source on chemical composition, anticholinesterase and antioxidant properties of some St. John’s Wort species (Hypericum spp.; Hypericaceae) from the Central Balkans. Molecules 2013, 18, 11733–11750. [Google Scholar] [CrossRef] [PubMed]
- Wright, C.W.; Gott, M.; Grayson, B.; Hanna, M.; Smith, A.G.; Sunter, A.; Neill, J.C. Correlation of hyperforin content of Hypericum perforatum (St. John’s Wort) extracts with their effects on alcohol drinking in C57BL/6J mice: A preliminary study. J. Psychopharmacol. 2003, 17, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Perfumi, M.; Panocka, I.; Ciccocioppo, R.; Vitali, D.; Froldi, R.; Massi, M. Effects of a malcoholic extract and a hyperforin-enriched CO2 extract of Hypericum perforatum on alcohol intake in rats. Alcohol Alcohol. 2001, 36, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Perfumi, M.; Santoni, M.; Ciccocioppo, R.; Massi, M. Blockade of γ-aminobutyric acid receptors does not modify the inhibition of alcohol intake induced by Hypericum perforatum in rats. Alcohol Alcohol. 2002, 37, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Perfumi, M.; Santoni, M.; Cippitelli, A.; Ciccocioppo, R.; Froldi, R.; Massi, M. Hypericum perforatum CO2 extract and opioid receptor antagonists act synergistically to reduce alcohol intake in alcohol-preferring rats. Alcohol Clin. Exp. Res. 2003, 27, 1554–1562. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.P.; Guo, J.J.; Bao, J.L.; Lu, J.J.; Wang, Y.T. The anticancer properties of Salvia Miltiorrhiza Bunge (Danshen): A systematic review. Med. Res. Rev. 2014, 34, 768–794. [Google Scholar] [CrossRef] [PubMed]
- De Palma, A.; Rossi, R.; Carai, M.; Cabras, C.; Colombo, G.; Arnoldi, L.; Fuzzati, N.; Riva, A.; Morazzoni, P.; Mauri, P.L. Pharmaceutical and biomedical analysis of terpene constituents in Salvia miltiorrhiza. Curr. Pharm. Anal. 2008, 4, 249–257. [Google Scholar] [CrossRef]
- Colombo, G.; Agabio, R.; Lobina, C.; Reali, R.; Morazzoni, P.; Bombardelli, E.; Gessa, G.L. Salvia miltiorrhiza extract inhibits alcohol absorption, preference, and discrimination in sP rats. Alcohol 1999, 18, 65–70. [Google Scholar] [CrossRef]
- Colombo, G.; Serra, S.; Vacca, G.; Orru, A.; Maccioni, P.; Morazzoni, P.; Bombardelli, E.; Riva, A.; Gessa, G.L.; Carai, M.A.M. Identification of miltirone as active ingredient of Salvia miltiorrhiza responsible for the reducing effect of root extracts on alcohol intake in rats. Alcohol. Clin. Exp. Res. 2006, 30, 754–762. [Google Scholar] [CrossRef] [PubMed]
- Brunetti, G.; Serra, S.; Vacca, G.; Lobina, C.; Morazzoni, P.; Bombardelli, E.; Colombo, G.; Gessa, G.L.; Carai, M.A.M. IDN 5082, a standardized extract of Salvia miltiorrhiza, delays acquisition of alcohol drinking behavior in rats. J. Ethnopharmacol. 2003, 85, 93–97. [Google Scholar] [CrossRef]
- Serra, S.; Vacca, G.; Tumatis, S.; Carrucciu, A.; Morazzoni, P.; Bombardelli, E.; Colombo, G.; Gessa, G.L.; Carai, M.A.M. Anti-relapse properties of IDN 5082, a standardized extract of Salvia miltiorrhiza, in alcohol-preferring rats. J. Ethnopharmacol. 2003, 88, 249–252. [Google Scholar] [CrossRef]
- Vacca, G.; Colombo, G.; Brunetti, G.; Melis, S.; Molinari, D.; Serra, S.; Seghizzi, R.; Morazzoni, P.; Bombardelli, E.; Gessa, G.L.; Carai, M.A.M. Reducing effect of Salvia miltiorrhiza extracts on alcohol intake: Influence of vehicle. Phytother. Res. 2003, 17, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Li, M.Z.; Lu, Y.; Hu, Y.; Zhai, X.H.; Xu, W.; Jing, H.R.; Tian, X.F.; Lin, Y.; Gao, D.Y.; Yao, J.H. Salvianolic acid B protects against acute alcohol-induced liver injury through SIRT1-mediated deacetylation of p53 in rats. Toxicol. Lett. 2014, 228, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Song, S.H.; Liu, W.Y.; Liu, F.; Wang, Z.X.; Ding, G.S.; Guo, W.Y.; Fu, Z.R. Magnesium lithospermate B reduces inflammatory response in a mouse model of hepatic ischemia-reperfusion injury. Cell Biochem. Biophys. 2014, 69, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Jin, Q.; Jiang, S.; Wu, Y.L.; Bai, T.; Yang, Y.; Jin, X.J.; Lian, L.H.; Nan, J.X. Hepatoprotective effect of cryptotanshinone from Salvia miltiorrhiza in D-galactosamine/lipopolysaccharide-induced fulminant hepatic failure. Phytomedicine 2014, 21, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.P.; Rui, W.J.; Wu, C.; He, S.F.; Jiang, J.M.; Zhang, X.X.; Yang, Y. Compound astragalus and Salvia miltiorrhiza extracts suppress hepatocarcinogenesis by modulating transforming growth factor-β/Smad signaling. J. Gastroenterol. Hepatol. 2014, 29, 1284–1291. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Yang, Y.; Zhang, X.X.; Xu, S.X.; He, S.F.; Huang, W.J.; Roberts, M.S. Compound astragalus and Salvia miltiorrhiza extract inhibits cell invasion by modulating transforming growth factor-β/Smad in HepG2 cell. J. Gastroenterol. Hepatol. 2010, 25, 420–426. [Google Scholar] [CrossRef] [PubMed]
- Rui, W.J.; Xie, L.; Liu, X.; He, S.F.; Wu, C.; Zhang, X.X.; Zhang, L.J.; Yang, Y. Compound astragalus and Salvia miltiorrhiza extract suppresses hepatocellular carcinoma progression by inhibiting fibrosis and PAI-1 mRNA transcription. J. Ethnopharmacol. 2014, 151, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Talbi, A.; Zhao, D.; Liu, Q.W.; Li, J.X.; Fan, A.; Yang, W.; Han, X.; Chen, X.J. Pharmacokinetics, tissue distribution, excretion and plasma protein binding studies of wogonin in rats. Molecules 2014, 19, 5538–5549. [Google Scholar] [CrossRef] [PubMed]
- Pan, T.L.; Wang, P.W.; Leu, Y.L.; Wu, T.H.; Wu, T.S. Inhibitory effects of Scutellaria baicalensis extract on hepatic stellate cells through inducing G2/M cell cycle arrest and activating ERK-dependent apoptosis via Bax and caspase pathway. J. Ethnopharmacol. 2012, 139, 829–837. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Park, W. Anti-inflammatory effect of wogonin on RAW 264.7 mouse macrophages induced with polyinosinic-polycytidylic acid. Molecules 2015, 20, 6888–6900. [Google Scholar] [CrossRef] [PubMed]
- Liang, R.R.; Zhang, S.; Qi, J.A.; Wang, Z.D.; Li, J.; Liu, P.J.; Huang, C.; Le, X.F.; Yang, J.; Li, Z.F. Preferential inhibition of hepatocellular carcinoma by the flavonoid Baicalein through blocking MEK-ERK signaling. Int. J. Oncol. 2012, 41, 969–978. [Google Scholar] [PubMed]
- Kim, S.J.; Lee, S.M. Effect of baicalin on toll-like receptor 4-mediated ischemia/reperfusion inflammatory responses in alcoholic fatty liver condition. Toxicol. Appl. Pharm. 2012, 258, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.Y.; Gong, X.; Zhang, L.; Li, H.Z.; Zhou, Y.F.; Zhou, Q.X. Protective effect of baicalin against lipopolysaccharide/d-galactosamine-induced liver injury in mice by up-regulation of heme oxygenase-1. Eur. J. Pharmacol. 2008, 587, 302–308. [Google Scholar] [CrossRef] [PubMed]
- Bhutada, P.; Mundhada, Y.; Bansod, K.; Hiware, R.; Rathod, S.; Dixit, P.; Mundhada, D. Berberine protects C57BL/6J mice against alcohol withdrawal-induced hyperexcitability. Phytother. Res. 2011, 25, 302–307. [Google Scholar] [PubMed]
- Bhutada, P.; Mundhada, Y.; Bansod, K.; Rathod, S.; Hiware, R.; Dixit, P.; Umathe, S.; Mundhada, D. Inhibitory effect of berberine on the motivational effects of alcohol in mice. Prog. Neuropsychopharmacol. Biol. Psychiatry 2010, 34, 1472–1479. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.P.; Lei, F.; Du, F.; Chai, Y.S.; Jiang, J.F.; Wang, Y.G.; Yu, X.; Yan, X.J.; Xing, D.M.; Du, L.J. Protection of gastrointestinal mucosa from acute heavy alcohol consumption: The effect of berberine and its correlation with TLR2, 4/IL1β-TNFα signaling. PLoS ONE 2015, 10, e0134044. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.; Hinton, D.J.; Johng, S.; Wang, J.B.; Choi, D.S. Levo-tetrahydropalmatine decreases alcohol drinking and antagonizes dopamine D2 receptor-mediated signaling in the mouse dorsal striatum. Behav. Brain Res. 2013, 244, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.L.; Liu, Q.; Gong, Q.; Li, J.X.; Wei, S.P.; Wang, Y.T.; Liang, H.; Zhang, M.; Jing, L.; Yong, Z.; Lawrence, A.J.; Liang, J.H. Brucine suppresses alcohol intake and preference in alcohol-preferring Fawn-Hooded rats. Acta Pharmacol. Sin. 2014, 35, 853–861. [Google Scholar] [CrossRef] [PubMed][Green Version]
- He, D.Y.; McGough, N.N.; Ravindranathan, A.; Jeanblanc, J.; Logrip, M.L.; Phamluong, K.; Janak, P.H.; Ron, D. Glial cell line-derived neurotrophic factor mediates the desirable actions of the anti-addiction drug ibogaine against alcohol consumption. J. Neurosci. 2005, 25, 619–628. [Google Scholar] [CrossRef] [PubMed]
- Carnicella, S.; Kharazia, V.; Jeanblanc, J.; Janak, P.H.; Ron, D. GDNF is a fast-acting potent inhibitor of alcohol consumption and relapse. Proc. Natl. Acad. Sci. USA 2008, 105, 8114–8119. [Google Scholar] [CrossRef] [PubMed]
- Teves, M.R.; Wendel, G.H.; Pelzer, L.E. Jodina rhombifolia leaves lyophilized aqueous extract decreases alcohol intake and preference in adolescent male Wistar rats. J. Ethnopharmacol. 2015, 174, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Teves, M.R.; Wendel, G.H.; Pelzer, L.E. Reduction in voluntary alcohol intake following repeated oral administration of Jodina rhombifolia lyophilized aqueous extract in male Wistar rats. J. Ethnopharmacol. 2015, 161, 170–174. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, F.; Li, Y.; Zhang, Y.-J.; Zhou, Y.; Li, S.; Li, H.-B. Natural Products for the Prevention and Treatment of Hangover and Alcohol Use Disorder. Molecules 2016, 21, 64. https://doi.org/10.3390/molecules21010064
Wang F, Li Y, Zhang Y-J, Zhou Y, Li S, Li H-B. Natural Products for the Prevention and Treatment of Hangover and Alcohol Use Disorder. Molecules. 2016; 21(1):64. https://doi.org/10.3390/molecules21010064
Chicago/Turabian StyleWang, Fang, Ya Li, Yu-Jie Zhang, Yue Zhou, Sha Li, and Hua-Bin Li. 2016. "Natural Products for the Prevention and Treatment of Hangover and Alcohol Use Disorder" Molecules 21, no. 1: 64. https://doi.org/10.3390/molecules21010064
APA StyleWang, F., Li, Y., Zhang, Y.-J., Zhou, Y., Li, S., & Li, H.-B. (2016). Natural Products for the Prevention and Treatment of Hangover and Alcohol Use Disorder. Molecules, 21(1), 64. https://doi.org/10.3390/molecules21010064







