Attenuated UV Radiation Alters Volatile Profile in Cabernet Sauvignon Grapes under Field Conditions
Abstract
:1. Introduction
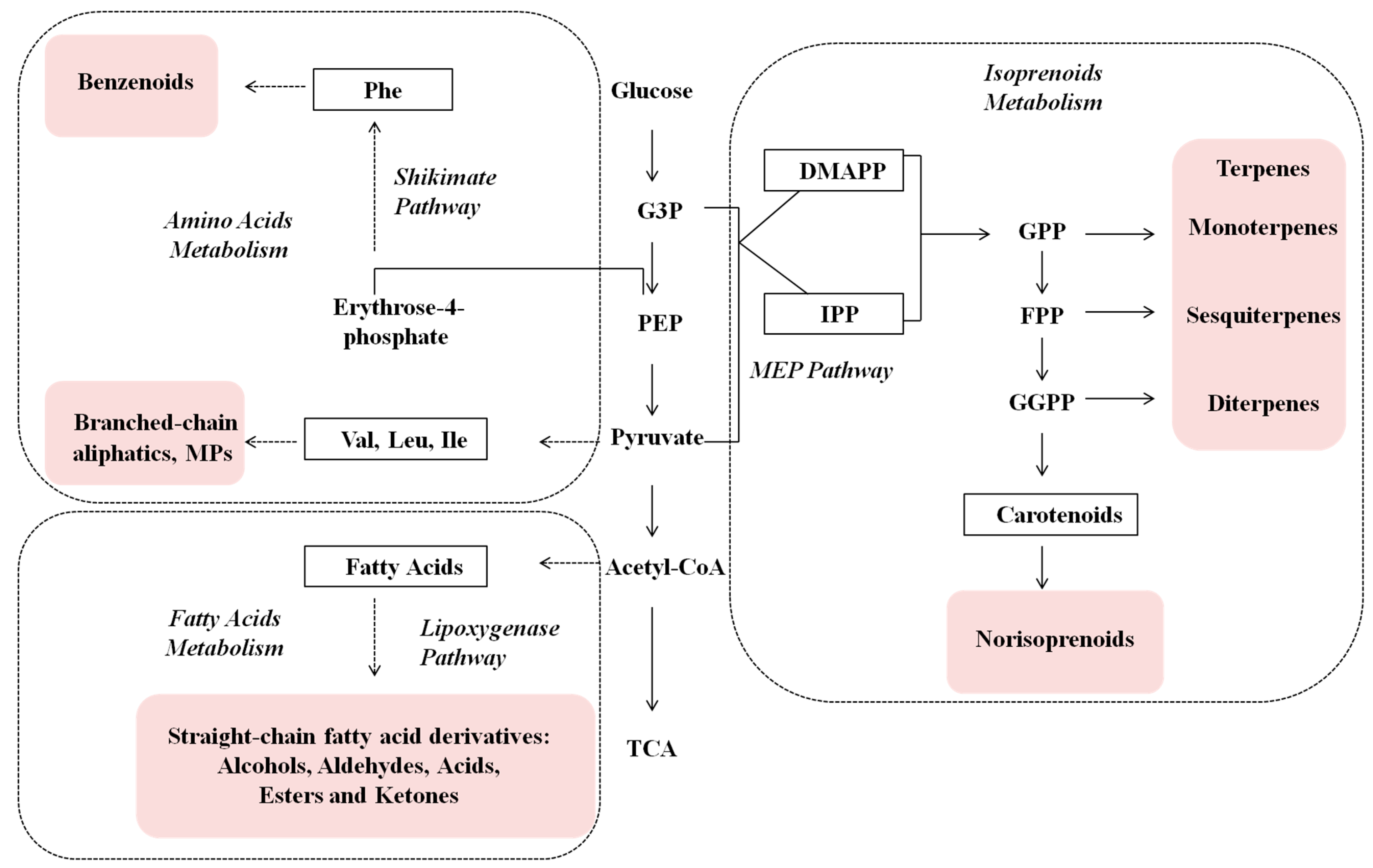
2. Results and Discussion
2.1. Effect of Attenuated UV Radiation on Three Major Classes of Volatiles
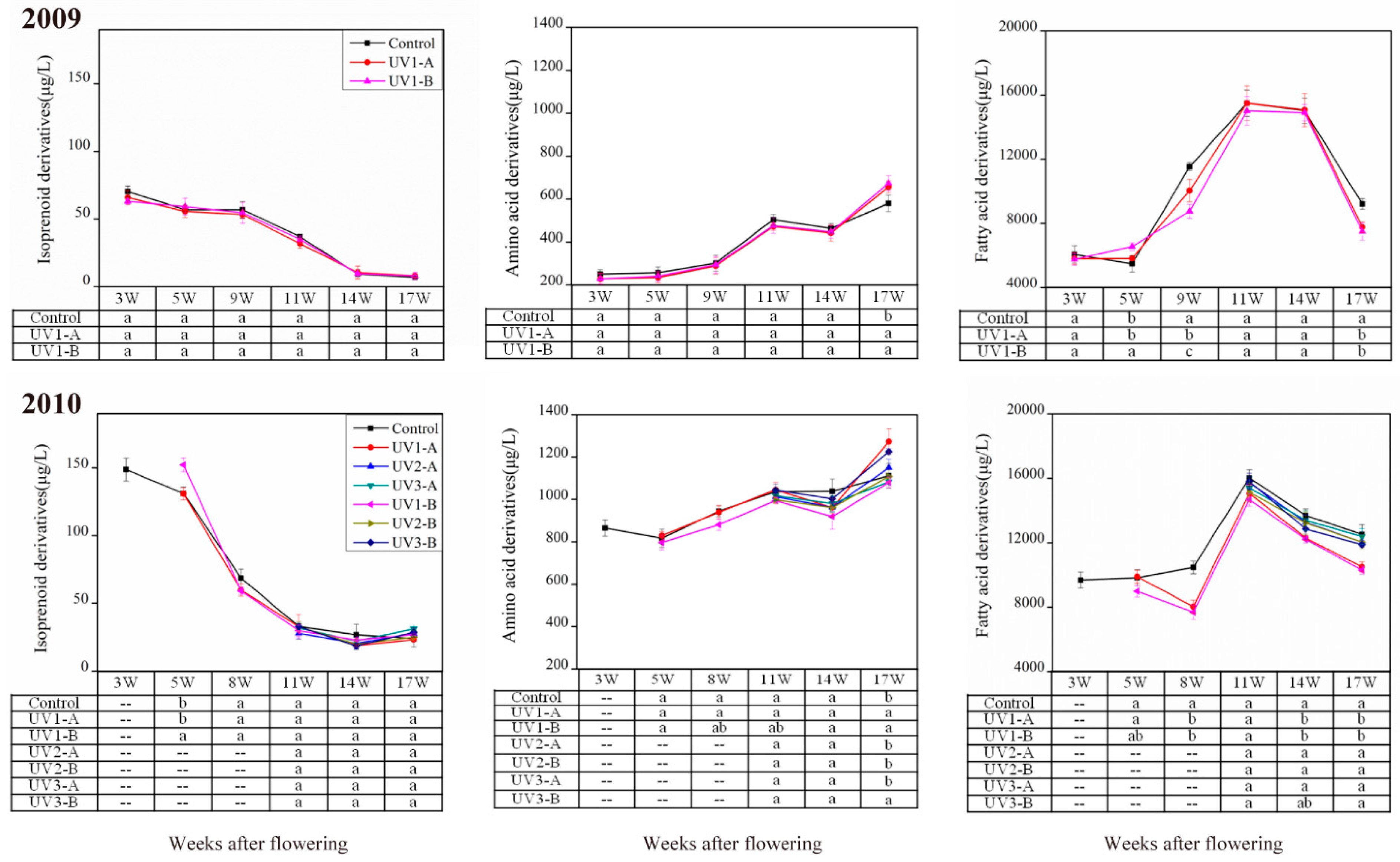
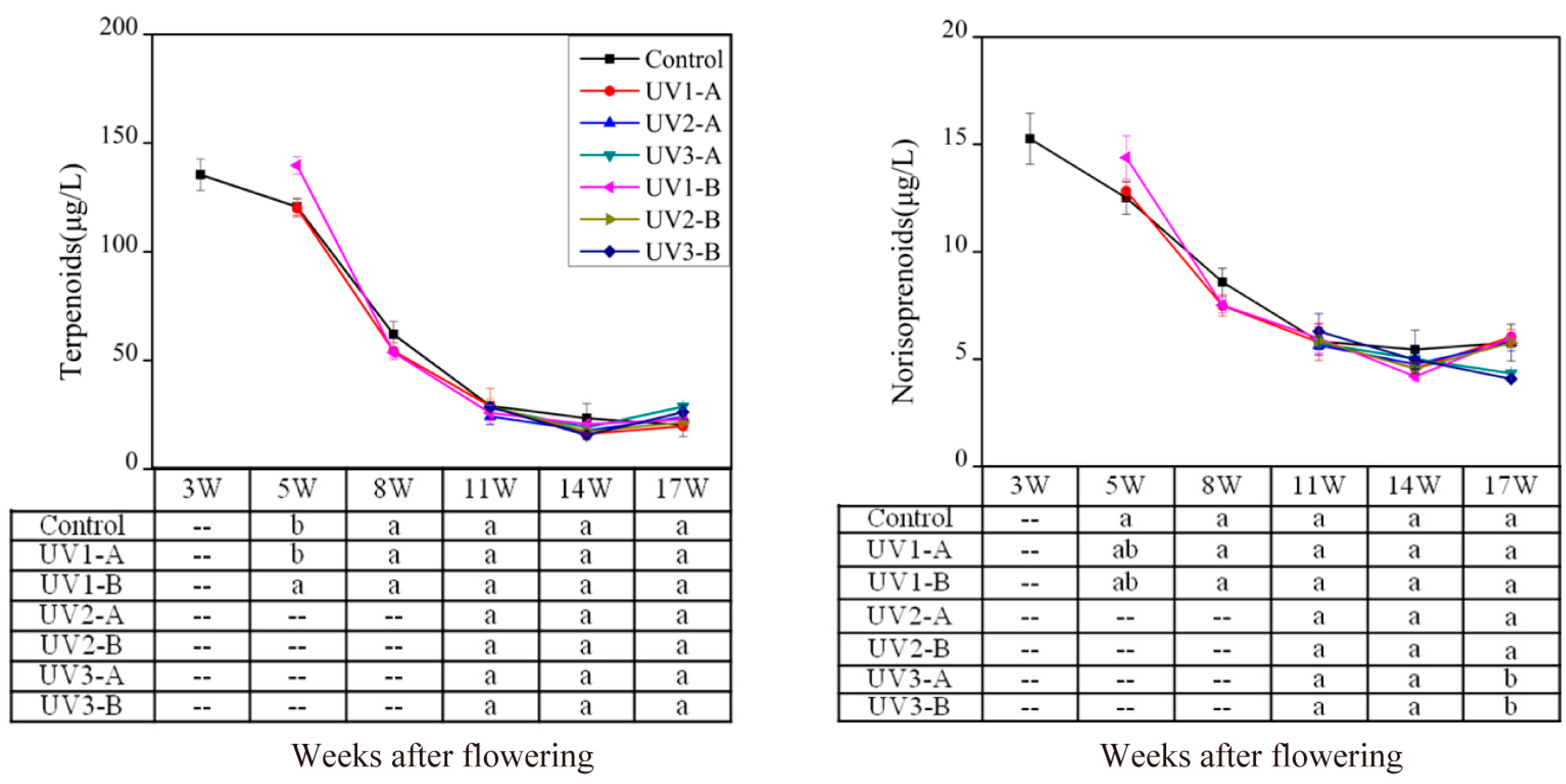
2.1.1. Effect on Isoprenoid Volatile Compounds
2.1.2. Effect on Amino Acid-Derived Volatiles
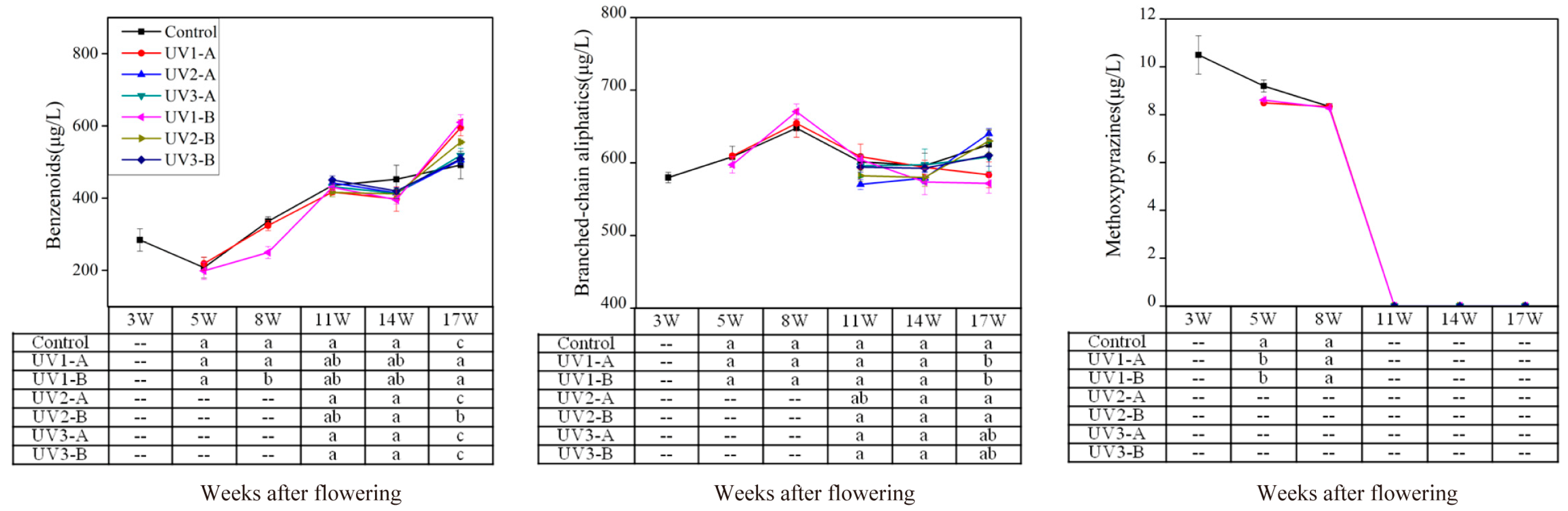
2.1.3. Effect on Fatty Acid-Derived Volatiles

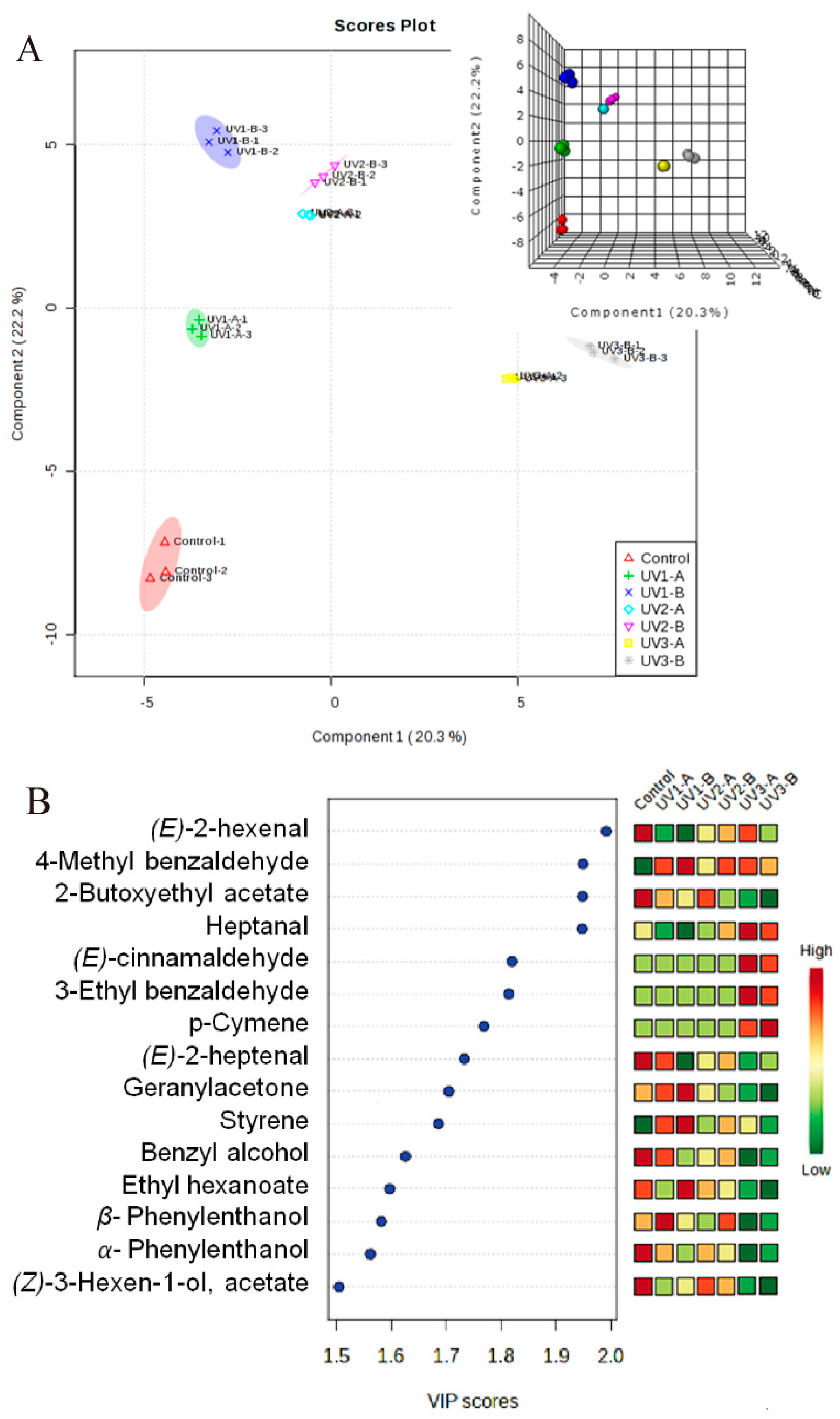
2.2. Partial Least Square Discriminant Analysis
| No. | Class | Calibration Curve | R2 Value | Linear Range (μg/L) | Concentration (μg/L FW) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | UV1-A | UV1-B | UV2-A | UV2-B | UV3-A | UV3-B | |||||
| Isoprene-derived | |||||||||||
| Terpenes | |||||||||||
| 1 | β-Myrcene | y = 1.572x + 0.0010 | 0.9885 | 0.1–3 | tr | tr | tr | tr | tr | tr | tr |
| 2 | Limonene | y = 4.105x + 0.0012 | 0.9795 | 0.1–3 | tr | tr | tr | tr | tr | tr | tr |
| 3 | Eucalyptol | y = 3.1992x − 0.0003 | 0.9580 | 0.1–3 | tr | tr | tr | tr | tr | tr | tr |
| 4 | γ-Terpinene | y = 3.09x + 0.0017 | 0.9879 | 0.1–40 | tr | tr | tr | tr | tr | tr | tr |
| 5 | Terpinolene | y = 3.8618x + (2 × 10−5) | 0.9639 | 0.1–20 | tr | tr | tr | tr | tr | tr | tr |
| 6 | cis-Furan linalool oxide | y = 1.403x − 0.0008 | 0.9890 | 0.1–2 | tr | tr | tr | tr | tr | tr | tr |
| 7 | Dihydro myrcenol | y = 1.7082x + 0.0026 | 0.9763 | 0.1–20 | 0.34 ± 0.03c | tr | 0.55 ± 0.03a | 0.50 ± 0.03a | tr | 0.41 ± 0.05b | 0.25 ± 0.03d |
| 8 | trans-Furan linalool oxide | y = 0.9459x − 0.0028 | 0.9890 | 0.1–2 | tr | tr | tr | tr | tr | tr | tr |
| 9 | Camphor | y = 13.113x − 0.0019 | 0.9643 | 0.1–20 | 2.75 ± 0.32a | 2.35 ± 0.28ab | 2.10 ± 0.22b | 2.60 ± 0.25a | 2.50 ± 0.35a | 2.93 ± 0.38a | 2.53 ± 0.22a |
| 10 | Linalool | y = 0.2918x + 0.0036 | 0.9937 | 0.1–40 | 3.87 ± 0.43a | 3.92 ± 0.25a | 3.83 ± 0.31a | 3.72 ± 0.14a | 3.74 ± 0.10a | 3.82 ± 0.45a | 3.76 ± 0.21a |
| 11 | Hotrienol | y = 0.433x − 0.0006 | 0.9971 | 0.1–40 | tr | tr | tr | tr | tr | tr | tr |
| 12 | β-Cyclocitral | y = 0.0906x + 0.0009 | 0.9635 | 0.1–20 | 0.24 ± 0.03a | 0.24 ± 0.02a | tr | 0.23 ± 0.03a | tr | 0.22 ± 0.02a | 0.23 ± 0.01a |
| 13 | α-Terpineol | y = 0.1792x + 0.0017 | 0.9940 | 0.1–20 | 1.26 ± 0.12c | 2.93 ± 0.13b | 4.40 ± 0.35a | 4.43 ± 0.25a | 3.94 ± 0.50a | 3.00 ± 0.21b | 3.03 ± 0.23b |
| 14 | Borneol | y = 4.0332x + 0.0024 | 0.9496 | 0.1–10 | 2.33 ± 0.22b | 2.50 ± 0.22ab | 2.87 ± 0.22a | 2.95 ± 0.22a | 2.53 ± 0.34ab | 2.68 ± 0.28a | 2.32 ± 0.22b |
| 15 | Citral | y = 0.2763x – (8 × 10−6) | 0.9951 | 0.1–5 | tr | tr | tr | tr | tr | tr | tr |
| 16 | β-Citronellol | y = 0.4061x + 0.0002 | 0.9972 | 0.1–4 | tr | tr | tr | tr | tr | tr | tr |
| 17 | Geraniol | y = 0.4406x + 0.0075 | 0.9944 | 0.3–50 | 9.07 ± 1.02a | 9.02 ± 0.81a | 9.13 ± 0.92a | 9.10 ± 0.61a | 9.09 ± 0.50a | 8.75 ± 0.64a | 8.83 ± 0.73a |
| 18 | α-Calacorene | y = 0.375x + 0.0002 | 0.9795 | 0.3–20 | tr | tr | tr | tr | tr | tr | tr |
| Norisoprenoids | |||||||||||
| 19 | Cyclohexanone, 2,2,6-trimethyl | y = 7.8699x − 0.0021 | 0.9623 | 0.1–20 | 1.22 ± 0.11b | 1.36 ± 0.07a | 1.47 ± 0.07a | 1.34 ± 0.08a | 1.35 ± 0.03a | 0.77 ± 0.03c | 0.69 ± 0.04c |
| 20 | 5-Hepten-2-one, 6-methyl- | y = 0.2047x + 0.0007 | 0.9886 | 0.1–40 | 1.34 ± 0.28a | 1.54 ± 0.13a | 1.30 ± 0.16a | 1.52 ± 0.22a | 1.43 ± 0.10a | 1.13 ± 0.05b | 1.07 ± 0.01b |
| 21 | Dihydroedulan I | y = 0.0893x + 0.0003 | 0.9958 | 0.1–20 | tr | tr | tr | tr | tr | tr | tr |
| 22 | cis-Theaspirane | y = 0.2012x + 0.0012 | 0.9906 | 0.1–40 | tr | tr | tr | tr | tr | tr | tr |
| 23 | β-Damascenone | y = 0.2377x + (7 × 10−5) | 0.9952 | 0.1–10 | 0.40 ± 0.03a | 0.28 ± 0.01bc | 0.21 ± 0.01d | 0.31 ± 0.02b | 0.35 ± 0.03ab | 0.26 ± 0.01c | 0.26 ± 0.01c |
| 24 | Geranylacetone | y = 0.2036x + 0.0007 | 0.9965 | 0.1–20 | 1.92 ± 0.15a | 1.95 ± 0.18a | 2.25 ± 0.25a | 1.85 ± 0.23a | 1.81 ± 0.17ab | 1.15 ± 0.01b | 1.05 ± 0.09b |
| 25 | Nerylactone | y = 0.1002x + 0.0023 | 0.9950 | 1.0–50 | 0.80 ± 0.05a | 0.90 ± 0.06a | 0.75 ± 0.04b | 0.76 ± 0.06b | 0.82 ± 0.03ab | 0.78 ± 0.04b | 0.77 ± 0.03b |
| 26 | β-Ionone | y = 0.1391x + 0.0002 | 0.9785 | 0.05–10 | 0.09 ± 0.01b | tr | tr | tr | tr | 0.24 ± 0.01a | 0.23 ± 0.01a |
| Amino acid-derived | |||||||||||
| Benzenoids | |||||||||||
| 27 | Styrene | y = 3.7779x + 0.0127 | 0.9850 | 1–500 | 53.95 ± 3.36d | 182.79 ± 5.25a | 200.60 ± 4.12a | 99.69 ± 5.12c | 140.24 ± 3.52b | 133.04 ± 2.15b | 96.68 ± 2.25c |
| 28 | p-Cymene | y = 3.0572x + 0.003 | 0.9812 | 0.1–50 | tr | tr | tr | tr | tr | 2.70 ± 0.12 | 2.76 ± 0.16 |
| 29 | Benzaldehyde | y = 0.1682x + 0.0036 | 0.9907 | 1–50 | 5.58 ± 0.10d | 10.22 ± 0.82a | 10.44 ± 0.21a | 7.07 ± 0.43b | 9.47 ± 0.21ab | 7.50 ± 0.55b | 6.61 ± 0.11c |
| 30 | 4-Methyl benzaldehyde | y = 0.1716x + 0.0003 | 0.9998 | 0.1–10 | tr | 2.98 ± 0.01 | 2.99 ± 0.01 | 2.83± 0.01 | 2.89 ± 0.01 | 2.86 ± 0.01 | 2.89 ± 0.01 |
| 31 | Benzenacetaldehyde | y = 0.321x + 0.0026 | 0.9786 | 1–100 | 16.63 ± 1.23b | 19.29 ± 0.82a | 20.55 ± 2.22a | 19.15 ± 1.21a | 19.60 ± 0.56a | 16.67 ± 0.34b | 16.89 ± 0.78b |
| 32 | Acetophenone | y = 0.7011x + 0.0133 | 0.9814 | 1–50 | 13.05 ± 0.97b | 15.16 ± 1.01a | 15.02 ± 1.17a | 13.67 ± 1.11ab | 14.44 ± 1.38a | 14.24 ± 1.02a | 13.90 ± 1.27a |
| 33 | Ethyl benzoate | y = 0.0793x + 0.0016 | 0.9927 | 0.1–20 | tr | 1.64 ± 0.25 | 1.65 ± 0.34 | 0.81 ± 0.05 | 1.63 ± 0.03 | 1.63 ± 0.14 | 1.62 ± 0.08 |
| 34 | 3-Ethyl benzaldehyde | y = 0.1195x – (7 × 10−5) | 0.9640 | 0.1–20 | tr | tr | tr | nd | nd | 2.77 ± 0.01 | 2.75 ± 0.01 |
| 35 | 1-(4-Methylphenyl)-ethanone | y = 0.9436x + 0.0075 | 0.9687 | 0.1–50 | tr | 2.61 ± 0.15 | 2.61 ± 0.10 | tr | tr | 2.61 ± 0.04 | 2.60 ± 0.01 |
| 36 | Naphthalene | y = 7.1162x − 0.0197 | 0.9849 | 0.1–100 | 1.65 ± 0.13a | 1.75 ± 0.12a | 1.59 ± 0.10a | 1.23 ± 0.12c | 1.34 ± 0.10ab | 1.37 ± 0.10b | 1.44 ± 0.10b |
| 37 | Methyl salicylate | y = 0.1535x + 0.005 | 0.9916 | 0.1–40 | 15.05 ± 0.60a | 15.13 ± 0.42a | 15.08 ± 0.21a | 15.05 ± 0.29a | 15.12 ± 0.37a | 15.07 ± 0.23a | 15.05 ± 0.21a |
| 38 | α-Phenylethanol | y = 4.9179x − 0.0146 | 0.9984 | 0.1–40 | 1.69 ± 0.12a | 1.54 ± 0.13a | 1.07 ± 0.08c | 1.30 ± 0.05b | 1.08 ± 0.05c | 0.85 ± 0.06d | 1.00 ± 0.10cd |
| 39 | 3,4-Dimethylbenzaldehyde | y = 0.4247x + 0.0027 | 0.9697 | 0.1–100 | 27.31 ± 1.52a | 24.93 ± 1.84b | 26.01 ± 1.56a | 23.12 ± 2.52b | 23.49 ± 1.23b | 25.12 ± 2.43ab | 23.49 ± 1.57b |
| 40 | Guaiacol | y = 1.2271x + 0.0008 | 0.9916 | 0.05–50 | tr | 3.17 ± 0.20 | 6.34 ± 0.14 | tr | tr | 6.31 ± 0.20 | 6.28 ± 0.12 |
| 41 | Benzyl alcohol | y = 16.596x + 0.0065 | 0.9894 | 2–500 | 182.24 ± 9.80a | 180.57 ± 10.50a | 160.29 ± 10.01ab | 161.98 ± 9.48ab | 179.23 ± 5.26a | 138.28 ± 10.20b | 145.29 ± 6.25b |
| 42 | 2,6-Diterbutyl-4-methyl phenol | y = 0.0922x − 0.0048 | 0.9612 | 0.01–8 | 0.61 ± 0.02b | 0.47 ± 0.01c | 0.55 ± 0.01b | 0.54 ± 0.01b | 0.46 ± 0.02c | 1.14 ± 0.02a | 0.58 ± 0.01b |
| 43 | β-Phenylethanol | y = 4.8719x − 0.0061 | 0.9856 | 10–500 | 89.03 ± 3.25a | 90.14 ± 3.56a | 84.84 ± 4.14a | 83.84 ± 3.07a | 89.65 ± 3.03a | 72.03 ± 2.02b | 73.87 ± 2.03b |
| 44 | 4-Methyl phenol | y = 0.5652x + 0.0062 | 0.9920 | 0.6–200 | 16.41 ± 0.59a | 16.43 ± 0.66a | 16.40 ± 0.42a | 16.39 ± 0.41a | 16.40 ± 0.51a | 16.37 ± 0.41a | 16.35 ± 0.35a |
| 45 | Phenol | y = 2.1793x − 0.0024 | 0.9986 | 0.2–100 | 23.98 ± 0.52bc | 26.58 ± 0.23b | 31.30 ± 0.82a | 22.49 ± 1.13c | 25.81 ± 0.63b | 23.96 ± 1.02bc | 22.58 ± 0.58c |
| 46 | (E)-Cinnamaldehyde | y = 0.7087x + 0.012 | 0.9774 | 0.1–10 | tr | tr | tr | nd | nd | 2.65 ± 0.23 | 2.60 ± 0.16 |
| Branched-chain aliphatics | |||||||||||
| 47 | Methyl isobutyl ketone | y = 11.85x − 0.0019 | 0.9987 | 0.5–120 | 616.00 ± 14.26a | 571.33 ± 16.20b | 559.61 ± 11.06b | 630.00 ± 26.55a | 620.00 ± 15.15a | 600.57 ± 13.25a | 600.00 ± 12.15a |
| 48 | 4,6-Dimethyl-2-heptanone | y = 0.4184x − 0.0004 | 0.9855 | 0.1–10 | tr | tr | tr | tr | tr | tr | tr |
| 49 | 3-Methyl-2-buten-1-ol | y = 2.0949x − 0.0003 | 0.9882 | 0.05–20 | 1.41 ± 0.13c | 2.65 ± 0.15a | 2.48 ± 0.16a | 2.10 ± 0.18b | 1.93 ± 0.11b | 1.80 ± 0.14b | 2.55 ± 0.13a |
| 50 | 1-Hexanol,2-ethyl- | y = 0.073x − 0.0024 | 0.9987 | 0.1–26 | 0.79 ± 0.06b | 1.07 ± 0.11a | 0.69 ± 0.02c | 1.12 ± 0.03a | 0.85 ± 0.05b | 0.74 ± 0.01b | 0.58 ± 0.02c |
| 51 | (S)-3-ethyl-4-methylpentanol | y = 2.0815x − 0.0003 | 0.9989 | 0.4–5 | 6.75 ± 0.47c | 8.33 ± 0.32a | 8.70 ± 0.45a | 7.12 ± 0.33b | 8.25 ± 0.54a | 6.34 ± 0.34c | 6.17 ± 0.11c |
| Methoxypyrazine | |||||||||||
| 52 | 3-Isobutyl-2-methoxypyrazine | y = 106.98x − 0.0012 | 0.9996 | 0.1–50 | tr | tr | tr | tr | tr | tr | tr |
| Fatty acid-derived | |||||||||||
| Straight-chain alcohols | |||||||||||
| 53 | 1-Butanol | y = 33.703x − 0.0019 | 0.9996 | 0.3–200 | 24.71 ± 1.20b | 25.95 ± 1.31ab | 27.26 ± 1.21a | 26.60 ± 1.54a | 28.66 ± 1.62a | 26.79 ± 1.10a | 27.26 ± 1.00a |
| 54 | (E)-2-penten-1-ol | y = 2.6285x − 0.0006 | 0.9635 | 0.1–50 | 10.00 ± 0.82a | 9.56 ± 0.46a | 9.55 ± 0.56a | 9.50 ± 0.34a | 9.68 ± 0.72a | 9.81 ± 0.61a | 9.63 ± 0.50a |
| 55 | 2-Heptanol | y = 0.3165x − 0.0002 | 0.9942 | 0.1–100 | 1.69 ± 0.11a | 1.16 ± 0.07ab | 1.04 ± 0.02ab | 1.42 ± 0.09a | 1.81 ± 0.05a | 1.32 ± 0.53 | 1.51 ± 0.01a |
| 56 | (Z)-2-penten-1-ol | y=2.9381x − 0.0018 | 0.9954 | 1–120 | 23.89 ± 1.21a | 21.26 ± 1.39a | 21.52± 1.08a | 19.76 ± 1.34b | 22.85 ± 1.38a | 18.76 ± 1.43b | 22.99 ± 1.07a |
| 57 | 1-Hexanol | y = 0.4184x-0.015 | 0.9937 | 0.3–500 | 481.83 ± 7.24a | 432.20 ± 7.12b | 418.50 ± 7.56b | 470.00 ± 14.97a | 468.23 ± 16.66a | 467.48 ± 15.30a | 464.76 ± 11.77a |
| 58 | (E)-3-hexen-1-ol | y = 0.3519x + 0.0001 | 0.9982 | 0.2–200 | 21.31 ± 1.20a | 20.60 ± 1.04a | 21.32 ± 1.10a | 22.58 ± 1.14a | 21.76 ± 1.24a | 20.94 ± 1.85a | 21.17 ± 1.02a |
| 59 | (Z)-3-hexen-1-ol | y = 4.2084x − 0.0042 | 0.9990 | 1–500 | 30.79 ± 2.32b | 39.47 ± 1.89a | 29.64 ± 2.12b | 29.50 ± 1.92b | 28.73 ± 2.51b | 31.23 ± 3.13b | 33.23 ± 3.68b |
| 60 | (E)-2-hexen-1-ol | y = 2.1862x − 0.0605 | 0.9627 | 1–500 | 29.19 ± 2.70a | 20.67 ± 0.88b | 20.14 ± 1.88b | 22.00 ± 1.56b | 24.00 ± 2.70b | 24.41 ± 1.23b | 25.48 ± 1.23b |
| 61 | (Z)-2-hexen-1-ol | y = 3.0267x − 0.0027 | 0.9983 | 0.3–200 | 5.34 ± 0.84bc | 2.47 ± 0.12d | 2.38 ± 0.22d | 9.02 ± 0.58a | 6.35 ± 0.23b | 3.94 ± 0.38c | 4.43 ± 0.35bc |
| 62 | 2-Octanol | y = 0.1804x + 0.0002 | 0.9947 | 0.1–120 | 0.26 ± 0.01a | 0.25 ± 0.01a | 0.23 ± 0.01a | 0.28 ± 0.02a | 0.27 ± 0.01a | 0.27 ± 0.01a | 0.29 ± 0.01a |
| 63 | 1-Octen-3-ol | y = 0.1343x-0.0004 | 0.9967 | 0.1–50 | 1.55 ± 0.12a | 1.47 ± 0.10b | 1.27 ± 0.08b | 1.55 ± 0.06a | 1.62 ± 0.13a | 1.36 ± 0.12ab | 1.75 ± 0.15a |
| 64 | 1-Heptanol | y = 0.2501x − 0.0001 | 0.9888 | 0.04–300 | 0.21 ± 0.01b | 0.20 ± 0.01b | 0.18 ± 0.01b | 0.30 ± 0.02a | 0.26 ± 0.02a | 0.20 ± 0.01b | 0.29 ± 0.01a |
| 65 | 2-Nonanol | y = 0.047x + 0.0008 | 0.9985 | 0.1–100 | 0.87 ± 0.06a | 0.84 ± 0.05a | 0.84 ± 0.05a | 0.86 ± 0.01a | 0.87 ± 0.02a | 0.85 ± 0.02a | 0.86 ± 0.02a |
| 66 | 1-Octanol | y = 0.2199x + 0.0003 | 0.9980 | 0.3–200 | 0.63 ± 0.05b | 0.75 ± 0.02a | 0.64 ± 0.02b | 0.74 ± 0.12a | 0.70 ± 0.04ab | 0.62 ± 0.05b | 0.74 ± 0.06a |
| 67 | (E)-2-octen-1-ol | y = 0.1313x − 0.0002 | 0.9885 | 0.05–10 | 0.42 ± 0.02b | 0.35 ± 0.02cd | 0.33 ± 0.02d | 0.41 ± 0.01b | 0.43 ± 0.01b | 0.37 ± 0.03c | 0.47 ± 0.01a |
| 68 | 1-Nonanol | y = 0.0572x + 0.0002 | 0.9857 | 0.1–20 | 0.85 ± 0.03a | 0.84 ± 0.02a | 0.83 ± 0.04a | 0.85 ± 0.08a | 0.84 ± 0.02a | 0.83 ± 0.05a | 0.84 ± 0.05a |
| 69 | 1-Decanol | y = 0.2717x + 0.0045 | 0.9950 | 0.1–20 | 0.98 ± 0.05a | 1.00 ± 0.14a | 1.12 ± 0.11a | 0.95 ± 0.06a | 1.05 ± 0.08a | 1.08 ± 0.04a | 0.97± 0.04a |
| 70 | 1-Dodecanol | y = 0.4996x + 0.0018 | 0.9987 | 0.1–100 | 1.99 ± 0.10a | 2.06 ± 0.02a | 2.00 ± 0.04a | 2.13 ± 0.25a | 1.96 ± 0.04a | 2.28 ± 0.22a | 1.97 ± 0.05a |
| Straight-chain aldehydes | |||||||||||
| 71 | Butanal | y = 5.9567x + 0.043 | 0.9983 | 1–200 | 61.83 ± 2.51a | 61.51 ± 2.13a | 59.35 ± 3.95a | 60.36 ± 2.32a | 60.50 ± 2.00a | 56.57 ± 4.03a | 53.15 ± 2.25b |
| 72 | Pentanal | y = 5.5978x + 0.1834 | 0.9668 | 10–500 | 56.14 ± 2.26a | 56.73 ± 1.56a | 56.39 ± 2.13a | 56.33 ± 2.23a | 56.02 ± 2.55a | 53.05 ± 2.13b | 50.55 ± 1.87b |
| 73 | Hexanal | y = 5.7615x − 0.1338 | 0.9999 | 100–5000 | 4625.22 ± 50.20a | 4218.57 ± 78.30b | 4199.11 ± 85.30b | 4578.71 ± 64.00a | 4727.43 ± 155.20a | 4500.00 ± 82.30a | 4600.00 ± 73.20a |
| 74 | (Z)-3-hexenal | y = 5.095x + 0.0644 | 0.9958 | 5–300 | 168.85 ± 4.68a | 155.88 ± 8.82a | 142.83 ± 5.70b | 150.00 ± 3.60b | 159.88 ± 5.30a | 136.50 ± 5.60c | 118.79 ± 4.53d |
| 75 | Heptanal | y = 3.3543x − 0.0042 | 0.9908 | 4–200 | 22.51 ± 1.02a | 14.49 ± 0.89b | 10.99 ± 0.85b | 21.27 ± 1.23a | 23.00 ± 0.98a | 24.11 ± 1.78a | 23.56 ± 0.56a |
| 76 | (E)-2-hexenal | y = 5.915x − 0.5077 | 0.9994 | 650–20000 | 6000.00 ± 83.20a | 5275.21 ± 65.30c | 5174.30 ± 64.30c | 5399.94 ± 95.30b | 5496 ± 62.30b | 5838.32 ± 184.30a | 5299.82 ± 72.50c |
| 77 | Octanal | y = 5.0692x − 0.0017 | 0.9839 | 0.1–20 | 4.07 ± 0.12a | 3.59 ± 0.20b | 4.13 ± 0.08a | 3.36 ± 0.15c | 3.52 ± 0.10b | 3.75 ± 0.10b | 2.99 ± 0.10d |
| 78 | (E)-2-heptenal | y = 0.3201x − 0.0002 | 0.9984 | 0.1–10 | 0.48 ± 0.04a | 0.38 ± 0.02b | 0.25± 0.01d | 0.37± 0.01b | 0.37 ± 0.03b | 0.28 ± 0.02cd | 0.32 ± 0.03c |
| 79 | Nonanal | y = 1.1837x − 0.0042 | 0.9862 | 0.1–100 | 2.86 ± 0.16a | 1.15 ± 0.11c | 3.06 ± 0.30a | 2.50 ± 0.19b | 2.59 ± 0.15b | 3.04 ± 0.13a | 2.56 ± 0.13b |
| 80 | (E,E)-2,4-hexadienal | y = 4.8522x + 0.2187 | 0.9837 | 5–1000 | 158.97 ± 7.58a | 146.65 ± 11.37a | 139.37 ± 4.60b | 154.21 ± 5.30a | 162.13 ± 4.50a | 124.47 ± 3.40c | 112.78 ± 2.30d |
| 81 | (E)-2-octenal | y = 0.1313x + 0.0007 | 0.9883 | 0.1–20 | 0.42 ± 0.03a | 0.37 ± 0.02ab | 0.37 ± 0.03ab | 0.36 ± 0.02b | 0.48 ± 0.04a | 0.35 ± 0.02b | 0.35 ± 0.03b |
| 82 | (E,E)-2,4-heptadienal | y = 0.5649x − 0.0015 | 0.9979 | 1–300 | 85.50 ± 5.40a | 81.39 ± 1.10a | 81.40 ± 1.79a | 81.49 ± 2.80a | 79.01 ± 3.56a | 83.97 ± 5.23a | 83.18 ± 3.53a |
| 83 | Decanal | y = 3.5466x − 0.0206 | 0.9967 | 0.1–100 | 6.37 ± 0.16a | 4.84 ± 0.23bc | 4.44 ± 0.11c | 5.38 ± 0.13b | 4.26 ± 0.23c | 3.91 ± 0.19c | 3.81 ± 0.21c |
| 84 | (E)-2-nonenal | y = 0.0709x – (4 × 10−5) | 0.9881 | 0.01–10 | 0.69 ± 0.04a | 0.74 ± 0.05a | 0.69 ± 0.02a | 0.64 ± 0.01a | 0.68 ± 0.01a | 0.68 ± 0.01a | 0.63 ± 0.05a |
| 85 | (E,Z)-2,6-nonadienal | y = 0.0462x + 0.0009 | 0.9937 | 0.1–4 | 0.35 ± 0.01b | 0.32 ± 0.01c | 0.36 ± 0.02b | 0.35 ± 0.02b | 0.28 ± 0.02d | 0.44 ± 0.04a | 0.38 ± 0.03a |
| 86 | (E,E)-2,4-nonadienal | y = 0.0572x + 0.0002 | 0.9857 | 0.1–4 | 0.34 ± 0.01a | 0.30 ± 0.03ab | 0.25 ± 0.01c | 0.34 ± 0.01a | 0.32 ± 0.01a | 0.28 ± 0.02b | 0.23 ± 0.01c |
| 87 | Dodecanal | y = 0.0353x – (4 × 10−5) | 0.9867 | 0.1–5 | 0.49 ± 0.03b | 0.57 ± 0.04a | 0.55 ± 0.03a | 0.42 ± 0.01c | 0.40 ± 0.02c | 0.45 ± 0.03b | 0.46 ± 0.02b |
| Straight-chain esters | |||||||||||
| 88 | Ethyl acetate | y = 3.3663x + 0.1055 | 0.9959 | 1–60 | 4.21 ± 0.20b | 4.66 ± 0.25b | 6.39 ± 0.10a | 4.81 ± 0.20b | 6.86 ± 0.50a | 6.57 ± 0.30a | 6.20 ± 0.48a |
| 89 | Ethyl butanoate | y = 0.5709x – (8 × 10−5) | 0.9811 | 0.1–5 | tr | tr | tr | tr | tr | tr | tr |
| 90 | 2-Butoxyethyl acetate | y = 1.1038x − 0.0008 | 0.9959 | 0.1–20 | 3.25 ± 0.08a | 1.37 ± 0.10b | 1.04 ± 0.06bc | 1.54 ± 0.07b | 1.04 ± 0.08c | 0.89 ± 0.03d | 0.40 ± 0.01e |
| 91 | Hexyl acetate | y = 0.5956x + 0.0037 | 0.9867 | 1–50 | 4.17 ± 0.10a | 4.52 ± 0.25a | 4.76 ± 0.18a | 4.70 ± 0.31a | 4.63 ± 0.15a | 3.91 ± 0.17a | 3.82 ± 0.03a |
| 92 | Ethyl hexanoate | y = 2.1749x – 0.1021 | 0.9956 | 0.1–30 | 1.59 ± 0.15a | 1.42 ± 0.07a | 1.64 ± 0.05a | 1.55 ± 0.11a | 1.54 ± 0.15a | 1.13 ± 0.07b | 1.03 ± 0.09b |
| 93 | (Z)-3-hexen-1-ol, acetate | y = 5.1939x − 0.0037 | 0.9980 | 0.1–100 | 5.37 ± 0.19a | 1.16 ± 0.11e | 1.64 ± 0.04d | 4.32 ± 0.29b | 2.45 ± 0.10c | 0.90 ± 0.11e | 0.33 ± 0.05f |
| 94 | Ethyl octanoate | y = 0.6077x + 0.0006 | 0.9899 | 0.5–50 | 1.21 ± 0.04a | 1.17 ± 0.09a | 1.27 ± 0.10a | 1.24 ± 0.09a | 1.13 ± 0.23a | 0.96 ± 0.01a | 1.00 ± 0.09a |
| Straight-chain acids | |||||||||||
| 95 | Hexanoic acid | y = 2.7019x + 0.0793 | 0.9931 | 1–700 | 101.93 ± 10.11a | 98.18 ± 2.21a | 102.63 ± 3.21a | 96.32 ± 2.13a | 93.52 ± 3.25a | 84.33 ± 2.12b | 83.57 ± 3.13b |
| 96 | (E)-3-Hexenoic acid | y = 5.1482x − 0.0028 | 0.9976 | 0.1–5 | tr | tr | tr | tr | tr | tr | tr |
| 97 | Nonanoic acid | y = 0.3257x − 0.0066 | 0.9884 | 0.1–5 | tr | tr | tr | tr | tr | tr | tr |
| Straight-chain ketones | |||||||||||
| 98 | 1-Penten-3-one | y = 12.315x − 0.0004 | 0.9685 | 0.1–120 | 29.95 ± 1.35a | 27.00 ± 1.85a | 28.00 ± 1.35a | 33.30 ± 2.68 | 27.73 ± 2.12 | 30.00 ± 2.38 | 28.60 ± 2.02 |
| 99 | 3-Octanone | y = 0.0725x – (2 × 10−5) | 0.9608 | 0.1–10 | 0.91 ± 0.06b | 1.00 ± 0.04b | 1.58 ± 0.08a | 1.03 ± 0.05b | 0.86 ± 0.06b | 1.23 ± 0.10b | 1.07 ± 0.07b |
| 100 | 2-Octanone | y = 4.7791x − 0.0019 | 0.8967 | 0.1–20 | 1.65 ± 0.12b | 1.80 ± 0.14b | 1.63 ± 0.15b | 1.55 ± 0.10b | 1.39 ± 0.08c | 2.50 ± 0.13a | 1.68 ± 0.12b |
| 101 | 1-Octen-3-one | y = 2.008x + 0.0032 | 0.9058 | 0.1–40 | 3.47 ± 0.32ab | 3.69 ± 0.30ab | 4.24 ± 0.53a | 3.27 ± 0.32b | 3.13 ± 0.28b | 3.48 ± 0.33ab | 3.75 ± 0.25ab |
| 102 | 2,3-Octanedione | y = 3.355x-0.4503 | 0.9873 | 0.1–5 | 0.50 ± 0.04a | 0.53 ± 0.05a | 0.45 ± 0.05a | 0.55 ± 0.05a | 0.42 ± 0.08b | 0.52 ± 0.05a | 0.48 ± 0.05a |
3. Experimental Section
3.1. Plant Materials
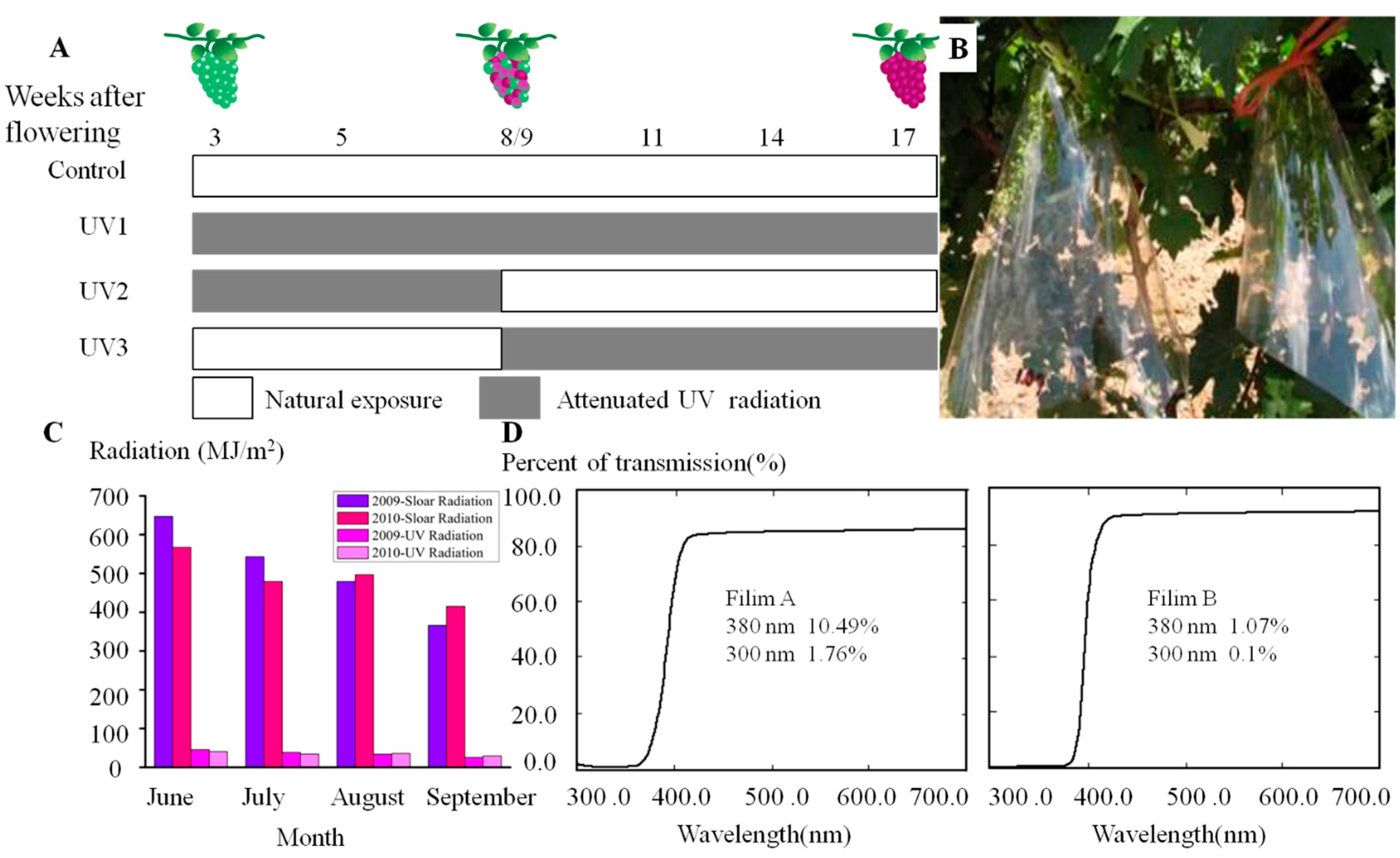
3.2. Field Treatments and Sampling
3.3. Extraction of Volatile Compounds
3.4. GC-MS Analysis
3.5. Qualitative and Quantitative Analysis
3.6. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Knudsen, J.T.; Eriksson, R.; Gershenzon, J.; Stahl, B. Diversity and distribution of floral scent. Bot. Rev. 2006, 72, 1–120. [Google Scholar] [CrossRef]
- Newman, J.D.; Chappell, J. Isoprenoid biosynthesis in plants: Carbon partitioning within the cytoplasmic pathway. Crit. Rev. Biochem. Mol. Biol. 1999, 34, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Rohmer, M. The discovery of a mevalonate-independent pathway for isoprenoid biosynthesis in bacteria, algae and higher plants. Nat. Prod. Rep. 1999, 16, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Dunlevy, J.D.; Kalua, C.M.; Keyzers, R.A.; Boss, P.K. The production of flavour & aroma compounds in grape berries. In Grapevine Molecular Physiology & Biotechnology, 2nd ed.; Roubelakis-Angelakis, K.A., Ed.; Springer Netherlands: Dordrecht, The Netherlands, 2009; pp. 293–330. [Google Scholar]
- González-Barreiro, C.; Rial-Otero, R.; Cancho-Grande, B.; Simal-Gándara, J. Wine aroma compounds in grapes: A critical review. Crit. Rev. Food Sci. Nutr. 2015, 55, 202–218. [Google Scholar] [CrossRef] [PubMed]
- Buttery, R.G.; Seifert, R.M.; Guadagni, D.G.; Ling, L.C. Characterization of some volatile constituents of bell peppers. J. Agric. Food Chem. 1969, 17, 1322–1327. [Google Scholar] [CrossRef] [PubMed]
- Parr, W.V.; Green, J.A.; White, K.G.; Sherlock, R.R. The distinctive flavour of New Zealand Sauvignon blanc: Sensory characterisation by wine professionals. Food Qual. Prefer. 2007, 18, 849–861. [Google Scholar] [CrossRef]
- Salas, J.J.; Sánchez, J.; Ramli, U.S.; Manaf, A.M.; Williams, M.; Harwood, J.L. Biochemistry of lipid metabolism in olive and other oil fruits. Prog. Lipid Res. 2000, 39, 151–180. [Google Scholar] [CrossRef]
- Kalua, C.M.; Boss, P.K. Evolution of volatile compounds during the development of Cabernet Sauvignon grapes (Vitis vinifera L.). J. Agric. Food Chem. 2009, 57, 3818–3830. [Google Scholar] [CrossRef] [PubMed]
- Robinson, A.L.; Boss, P.K.; Solomon, P.S.; Trengove, R.D.; Heymann, H.; Ebeler, S.E. Origins of grape and wine aroma. Part 1. Chemical components and viticultural impacts. Am. J. Enol. Vitic. 2014, 65, 1–24. [Google Scholar] [CrossRef]
- Mckenzie, R.; Bodeker, G.; Scott, G.; Slusser, J.; Lantz, K. Geographical differences in erythemally-weighted uv measured at mid-latitude usda sites. Photochem. Photobiol. Sci. 2006, 5, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Seckmeyer, G.; Mayer, B.; Bernhard, G.; Mckenzie, R.L.; Johnston, P.V.; Kotkamp, M. Geographical differences in the uv measured by intercompared spectroradiometers. Geophys. Res. Lett. 1995, 22, 1889–1892. [Google Scholar] [CrossRef]
- Caldwell, M.M.; Teramura, A.H.; Tevini, M. The changing solar ultraviolet climate and the ecological consequences for higher plants. Trends Ecol. Evol. 1989, 4, 363–367. [Google Scholar] [CrossRef]
- McKenzie, R.L.; Björn, L.O.; Bais, A.; Ilyasd, M. Changes in biologically active ultraviolet radiation reaching the Earth’s surface. Photochem. Photobiol. Sci. 2003, 2, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Schultz, H.R.; Lohnertz, O.; Bettner, W.; Balo, B.; Linsenmeier, A.; Jahnisch, A. Is grape composition affected by current levels of UV-B radiation? Vitis 1998, 37, 191–192. [Google Scholar]
- Núñez-Olivera, E.; Martínez-Abaigar, J.; Tomás, R.; Otero, S.; Arróniz-Crespo, M. Physiological effects of solar ultraviolet-B exclusion on two cultivars of Vitis vinifera L. from La Rioja, Spain. Am. J. Enol. Vitic. 2006, 57, 441–448. [Google Scholar]
- Kolb, C.A.; Kopecký, J.; Riederer, M.; Pfündel, E.E. UV screening by phenolics in berries of grapevine (Vitis vinifera). Funct. Plant Biol. 2003, 30, 1177–1186. [Google Scholar] [CrossRef]
- Zhang, Z.Z.; Li, X.X.; Chu, Y.N.; Zhang, M.X.; Wen, Y.Q.; Duan, C.Q.; Pan, Q.H. Three types of ultraviolet irradiation differentially promote expression of shikimate pathway genes and production of anthocyanins in grape berries. Plant Physiol. Biochem. 2012, 57, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Z.; Che, X.N.; Pan, Q.H.; Li, X.X.; Duan, C.Q. Transcriptional activation of flavan-3-ols biosynthesis in grape berries by UV irradiation depending on developmental stage. Plant Sci. 2013, 208, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Smart, R.; Wang, H.; Dambergs, B.; Sparrow, A.; Qian, M.C. Effect of grape bunch sunlight exposure and UV radiation on phenolics and volatile composition of Vitis vinifera L. cv. Pinot noir wine. Food Chem. 2015, 173, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Šuklje, K.; Antalick, G.; Coetzee, Z.; Schmidtke, L.M.; Baša Česnik, H.; Brandt, J.; Deloire, A. Effect of leaf removal and ultraviolet radiation on the composition and sensory perception of Vitis vinifera L. cv. Sauvignon Blanc wine. Aust. J. Grape Wine Res. 2014, 20, 223–233. [Google Scholar] [CrossRef]
- Zavala, J.A.; Ravetta, D.A. The effect of solar UV-B radiation on terpenes and biomass production in Grindelia chiloensis (Asteraceae), a woody perennial of Patagonia, Argentina. Plant Ecol. 2002, 161, 185–191. [Google Scholar] [CrossRef]
- Lafontaine, M.; Schultz, H.R.; Lopes, C.; Bá1o, B.; Varadi, G. Leaf and fruit responses of Riesling grapevines to UV-radiation in the field. In Proceedings of the VII International Symposium on Grapevine Physiology and Biotechnology, Davis, CA, USA, 21–25 June 2004; Williams, L.E., Ed.; ISHS: Davis, CA, USA, 2005. [Google Scholar]
- Gregan, S.M.; Wargent, J.J.; Liu, L.; Shinkle, J.; Hofmann, R.; Winefield, C.; Jordan, B. Effects of solar ultraviolet radiation and canopy manipulation on the biochemical composition of Sauvignon Blanc grapes. Aust. J. Grape Wine Res. 2012, 18, 227–238. [Google Scholar] [CrossRef]
- Ohlsson, A.B.; Segerfeldt, P.; Lindstrom, A.; Borg-Karlson, A.K.; Berglund, T. UV-B exposure of indoor-grown Picea abies seedlings causes an epigenetic effect and selective emission of terpenes. Z. Naturforsch. C 2013, 68, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Eichholz, I.; Huyskens-Keil, S.; Keller, A.; Ulrich, D.; Kroh, L.W.; Rohn, S. UV-B-induced changes of volatile metabolites and phenolic compounds in blueberries (Vaccinium corymbosum L.). Food Chem. 2011, 126, 60–64. [Google Scholar] [CrossRef]
- Beaulieu, J.C. Effect of UV irradiation on cut cantaloupe: Terpenoids and esters. J. Food Sci. 2007, 72, S272–S281. [Google Scholar] [CrossRef] [PubMed]
- Gil, M.; Bottini, R.; Berli, F.; Pontin, M.; Silva, M.F.; Piccoli, P. Volatile organic compounds characterized from grapevine (Vitis vinifera L. cv. Malbec) berries increase at pre-harvest and in response to UV-B radiation. Phytochemistry 2013, 96, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Llusia, J.; Llorens, L.; Bernal, M.; Verdaguer, D.; Peñuelas, J. Effects of UV radiation and water limitation on the volatile terpene emission rates, photosynthesis rates, and stomatal conductance in four Mediterranean species. Acta Physiol. Plant. 2012, 34, 757–769. [Google Scholar] [CrossRef]
- Keller, M.; Torres-Martinez, N. Does UV radiation affect wine grape composition? In Proceedings of the XXVI International Horticultural Congress: Viticulture-Living with Limitations, Toronto, ON, Canada, 11–17 August 2002; Reynolds, A.G., Bowen, P., Eds.; ISHS: Toronto, ON, Canada, 2004. [Google Scholar]
- Strauss, C.R.; Wilson, B.; Gooley, P.R.; Williams, P.J. Role of monoterpenes in grape and wineflavor. ACS Symp. Ser. 1986, 317, 222–242. [Google Scholar]
- Mathieu, S.; Terrier, N.; Procureur, J.; Bigey, F.; Günata, Z. A carotenoid cleavage dioxygenase from Vitis vinifera L.: Functional characterization and expression during grape berry development in relation to C13-norisoprenoid accumulation. J. Exp. Bot. 2005, 56, 2721–2731. [Google Scholar] [CrossRef] [PubMed]
- Conde1, C.; Silva1, P.; Fontes1, N.; Dias1, A.C.P.; Tavares1, R.M.; Sousa1, M.J.; Agasse1, A.; Delrot, S.; Gerós1, H. Biochemical changes throughout grape berry development and fruit and wine quality. Food 2007, 1, 1–22. [Google Scholar]
- Karapinar, M.; Aktug, S.E. Inhibition of foodborne pathogens by thymol eugenol menthol and anethole. Int. J. Food Microbiol. 1987, 4, 161–166. [Google Scholar] [CrossRef]
- Herrmann, K.M.; Weaver, L.M. The shikimate pathway. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1999, 50, 473–503. [Google Scholar] [CrossRef] [PubMed]
- Logemann, E.; Tavernaro, A.; Schulz, W.; Somssich, I.E.; Hahlbrock, K. UV light selectively coinduces supply pathways from primary metabolism and flavonoid secondary product formation in parsley. Proc. Natl. Acad. Sci. USA 2000, 97, 1903–1907. [Google Scholar] [CrossRef] [PubMed]
- Schwab, W.; Davidovich-Rikanati, R.; Lewinsohn, E. Biosynthesis of plant-derived flavor compounds. Plant J. 2008, 54, 712–732. [Google Scholar] [CrossRef] [PubMed]
- Blee, E. Impact of phyto-oxylipins in plant defense. Trends Plant Sci. 2002, 7, 315–321. [Google Scholar] [CrossRef]
- Howe, G.A.; Schilmiller, A.L. Oxylipin metabolism in response to stress. Curr. Opin. Plant Biol. 2002, 5, 230–236. [Google Scholar] [CrossRef]
- Weber, H. Fatty acid-derived signals in plants. Trends Plant Sci. 2002, 7, 217–224. [Google Scholar] [CrossRef]
- Kobayashi, H.; Takase, H.; Suzuki, Y.; Tanzawa, F.; Takata, R.; Fujita, K.; Kohno, M.; Mochizuki, M.; Suzuki, S.; Konno, T. Environmental stress enhances biosynthesis of flavor precursors, S-3-(hexan-1-ol)-glutathione and S-3-(hexan-1-ol)-l-cysteine, in grapevine through glutathione S-transferase activation. J. Exp. Bot. 2011, 62, 1325–1336. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Sinelnikov, I.V.; Han, B.; Wishart, D.S. MetaboAnalyst 3.0—Making metabolomics more meaningful. Nucleic Acids Res. 2015, 1. [Google Scholar] [CrossRef] [PubMed]
- Sefton, M.A. Hydrolytically-released volatile secondary metabolites from a juice sample of Vitis vinifera grape cvs Merlot and Cabernet Sauvignon. Aust. J. Grape Wine Res. 1998, 4, 30–38. [Google Scholar] [CrossRef]
- Pineau, B.; Barbe, J.C.; Leeuwen, C.V.; Dubourdieu, D. Which impact for bdamascenone on red wines aroma. J. Agric. Food Chem. 2007, 55, 4103–4108. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.Q.; Cheng, G.; Duan, L.L.; Jiang, R.; Pan, Q.H.; Duan, C.Q.; Wang, J. Effect of training systems on fatty acids and their derived volatiles in Cabernet Sauvignon grapes and wines of the north foot of Mt. Tianshan. Food Chem. 2015, 181, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not available.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, D.; Gao, Y.; Li, X.-X.; Li, Z.; Pan, Q.-H. Attenuated UV Radiation Alters Volatile Profile in Cabernet Sauvignon Grapes under Field Conditions. Molecules 2015, 20, 16946-16969. https://doi.org/10.3390/molecules200916946
Liu D, Gao Y, Li X-X, Li Z, Pan Q-H. Attenuated UV Radiation Alters Volatile Profile in Cabernet Sauvignon Grapes under Field Conditions. Molecules. 2015; 20(9):16946-16969. https://doi.org/10.3390/molecules200916946
Chicago/Turabian StyleLiu, Di, Yuan Gao, Xiao-Xi Li, Zheng Li, and Qiu-Hong Pan. 2015. "Attenuated UV Radiation Alters Volatile Profile in Cabernet Sauvignon Grapes under Field Conditions" Molecules 20, no. 9: 16946-16969. https://doi.org/10.3390/molecules200916946







