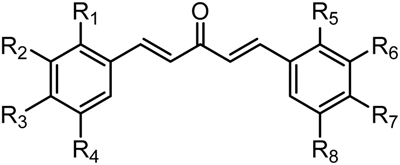
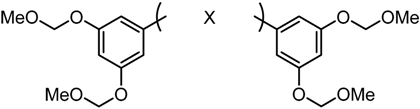Structure-Activity Relationships of the Antitumor C5-Curcuminoid GO-Y030
Abstract
:1. Introduction
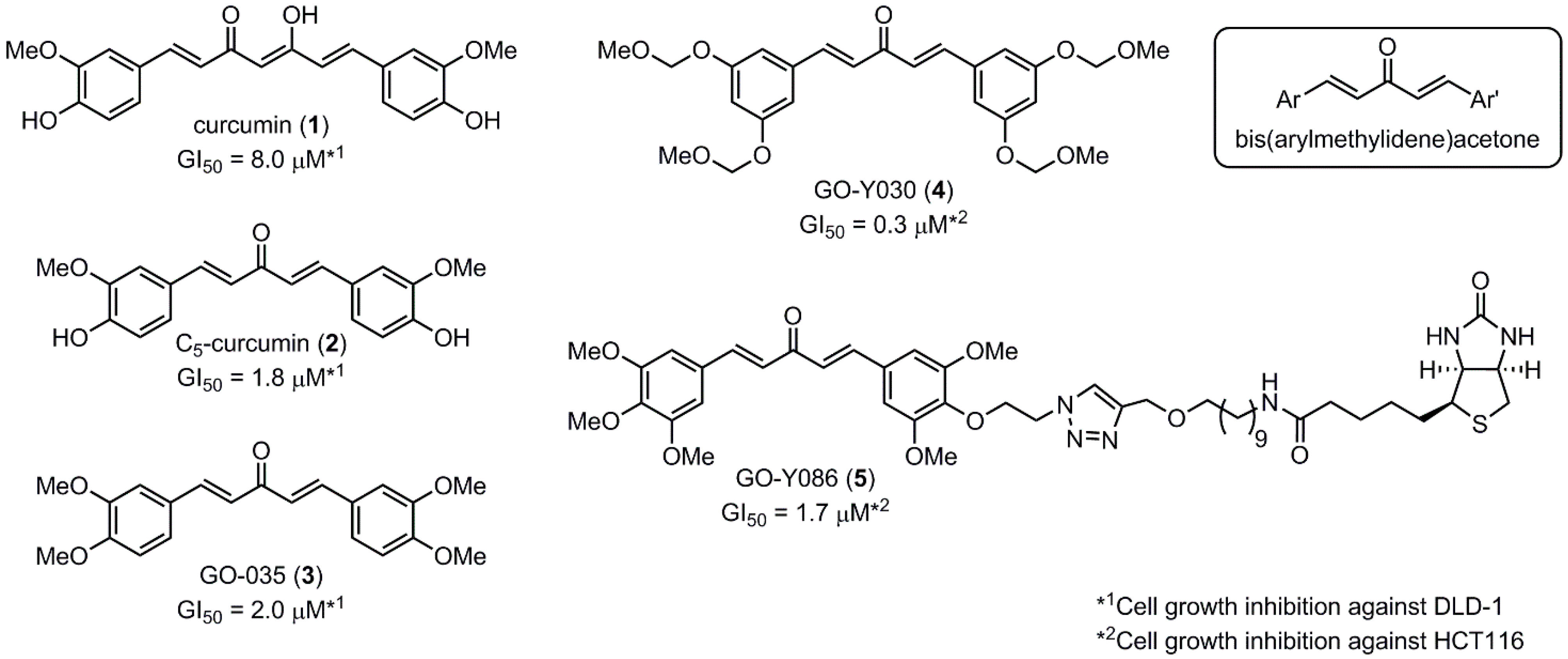
2. Results and Discussion
2.1. Chemistry
2.2. Pharmacological Evaluation
2.3. SAR of C5-Curcuminoid GO-Y030
2.3.1. Roles of the 3,5-Bismethoxymethoxy Substituent
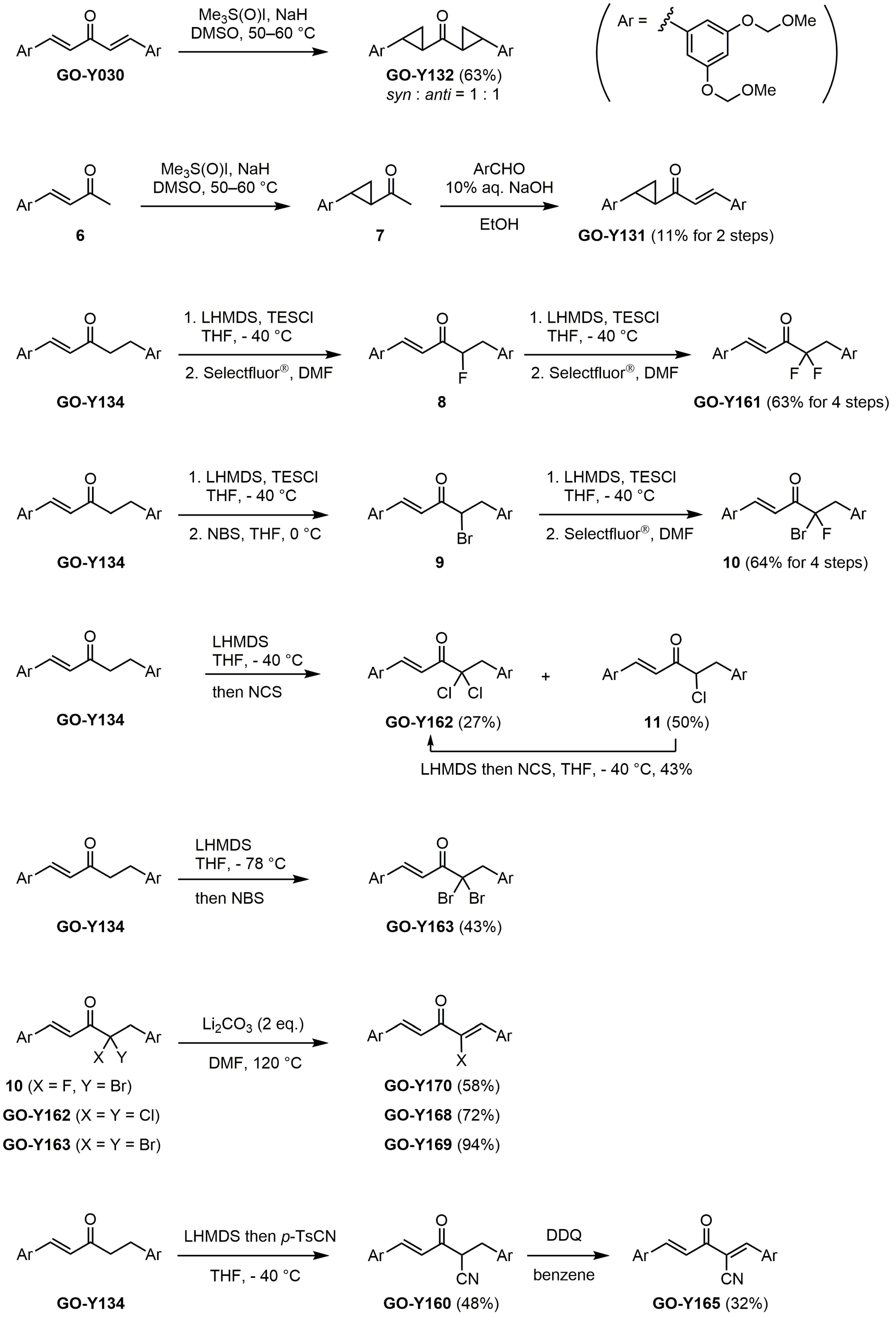
| Compound | R | GI50 (μM) *2 | |
|---|---|---|---|
| GO-949 | R2 = R3 = OCH2O, R6 = R7 = OCH2O | R1 = R4 = R5 = R8 = H | >50 *1 |
| GO-Y030 | R2 = R4 = R6 = R8 = OCH2OMe | R1 = R3 = R5 = R7 = H | 0.3 |
| GO-Y038 | R2 = R4 = R6 = R8 = OH | R1 = R3 = R5 = R7 = H | 1.5 |
| GO-Y067 | R2 = R4 = R6 = R8 = OMe | R1 = R3 = R5 = R7 = H | 2.0 |
| GO-Y129 | R2 = R4 = R6 = R8 = OCH2CH2OMe | R1 = R3 = R5 = R7 = H | 0.2 |
| GO-Y130 | R2 = R4 = R6 = R8 = OCH2CH2OH | R1 = R3 = R5 = R7 = H | 6.4 |
| GO-Y147 | R2 = R4 = R6 = R8 = OCH2OCH2CH2OMe | R1 = R3 = R5 = R7 = H | 2.0 |
| GO-Y156 | R2 = R4 = OCH2OMe, R6 = R7 = OCH2O | R1 = R3 = R5 = R8 = H | 7.9 |
| GO-Y164 | R1 = R4 = R5 = R8 = OCH2OMe | R2 = R3 = R6 = R7 = H | 1.7 |
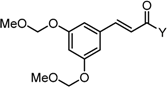
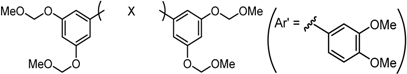
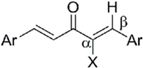
2.3.2. Roles of the Central Tether Moiety
| X Compound GI50 (μM) *2 | 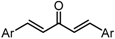 | 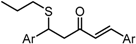 |  |  |
| GO-Y030 | GO-Y077 | GO-Y131 | GO-Y134 | |
| 0.3 | 0.9 | 6.8 | 18 | |
| X Compound GI50 (μM) *2 | 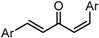 | 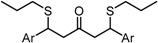 |  | 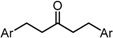 |
| GO-Y095 | GO-Y075 | GO-Y132 | GO-Y133 | |
| 0.9 | 2.1 | >50 | >50 |
2.3.3. Effect of Two Aromatic Rings
| Compound | Y | GI50 (μM) *2 |
|---|---|---|
| GO-Y148 | OMe | >50 |
| GO-Y149 | H | 49 |
| GO-Y150 | SCH2CH2CH3 | >50 |
| GO-Y151 | NHMe | >50 |
| GO-Y157 | OH | >50 |
| GO-Y171 | C(CH3)3 | 36 |
| Compound | X | GI50 (μM) *2 |
|---|---|---|
| GO-Y154 | 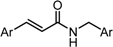 | >50 |
| GO-Y158 | 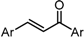 | 7.3 |
| GO-Y159 |  | >50 |
| GO-Y025 |  | 2.0 |
2.3.4. Effects of Introduction of Electron Withdrawing Group and Amino Group at C-2
| Type 1 Analogues | ||||||
| X Compound GI50 (μM) *2 |  |  | 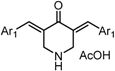 | 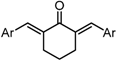 | 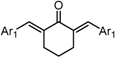 | |
| GO-Y166 | GO-Y167 | EF-24 | GO-Y152 | GO-Y153 | ||
| 1.9 | 2.8 | 2.3 | 3.9 | 18 | ||
| X Compound GI50 (μM) *2 | 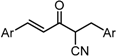 | 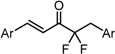 | 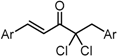 | 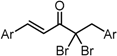 | ||
| GO-Y160 | GO-Y161 | GO-Y162 | GO-Y163 | |||
| >50 | >50 | 19 | 3.6 | |||
| Type 2 Analogues | ||||||
| X Compound GI50 (μM) *2 | 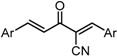 | 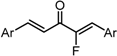 | 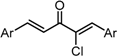 | 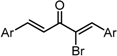 | 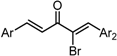 | |
| GO-Y165 | GO-Y170 | GO-Y168 | GO-Y169 | GO-Y172 | ||
| 33 | 3.6 | 1.0 | 1.8 | 1.3 | ||
| Compound | X | Chemical Shift of Cβ-H (ppm) | GI50 (μM) *2 |
|---|---|---|---|
| GO-Y030 | H | 7.64 | 0.3 |
| GO-Y165 | CN | 8.22 | 3.3 |
| GO-Y170 | F | 6.89 | 3.6 |
| GO-Y168 | Cl | 7.77 | 1.0 |
| GO-Y169 | Br | 7.99 | 1.8 |
3. Experimental Section
3.1. General Information
3.2. Chemical Synthesis
3.3. Cell Growth Suppression Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Minassi, A.; Sánchez-Duffhues, G.; Collado, J.A.; Muñoz, E.; Appendino, G. Dissecting the pharmacophore of curcumin. Which structural element is critical for which action? J. Nat. Prod. 2013, 76, 1105–1112. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Thomas, S.G.; Kunnumakkara, A.B.; Sundaram, C.; Harikumar, K.B.; Sung, B.; Tharakan, S.T.; Misra, K.; Priyadarsini, I.K.; Rajasekharan, K.N.; et al. Biological activities of curcumin and its analogues (Congeners) made by man and Mother Nature. Biochem. Pharmacol. 2008, 76, 1590–1611. [Google Scholar] [CrossRef] [PubMed]
- Youssef, K.M.; El-Sherbeny, M.A.; El-Shafie, F.S.; Farag, H.A.; Al-Deeb, O.A.; Awadalla, S.A.A. Synthesis of curcumin analogues as potential antioxidant, cancer chemopreventive agents. Arch. Pharm. (Weinheim) 2004, 337, 42–54. [Google Scholar] [CrossRef] [PubMed]
- Sardjiman, S.S.; Reksohadiprodjo, M.S.; Hakim, L.; van der Goot, H.; Timmerman, H. 1,5-Diphenyl-1,4-pentadiene-3-ones and cyclic analogues as antioxidative agents. Synthesis and structure-activity relationship. Eur. J. Med. Chem. 1997, 32, 625–630. [Google Scholar] [CrossRef]
- Liang, G.; Li, X.; Chen, L.; Yang, S.; Wu, X.; Studer, E.; Gurley, E.; Hylemon, P.B.; Ye, F.; Li, Y.; et al. Synthesis and anti-inflammatory activities of mono-carbonyl analogues of curcumin. Bioorg. Med. Chem. Lett. 2008, 18, 1525–1529. [Google Scholar] [CrossRef] [PubMed]
- Adams, B.K.; Ferstl, E.M.; Davis, M.C.; Herold, M.; Kurtkaya, S.; Camalier, R.F.; Hollingshead, M.G.; Kaur, G.; Sausville, E.A.; Rickles, F.R.; et al. Synthesis and biological evaluation of novel curcumin analogs as anti-cancer and anti-angiogenesis agents. Bioorg. Med. Chem. 2004, 12, 3871–3883. [Google Scholar] [CrossRef] [PubMed]
- Adams, B.K.; Cai, J.; Armstrong, J.; Herold, M.; Lu, Y.J.; Sun, A.; Snyder, J.P.; Liotta, D.C.; Jones, D.P.; Shoji, M. EF24, a novel synthetic curcumin analog, induces apoptosis in cancer cells via a redox-dependent mechanism. Anticancer Drugs 2005, 16, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.L.; Ali, A.; Du, Y.; Fu, H.; Jin, H.X.; Chin, T.M.; Khan, M.; Go, M.L. Synthesis and evaluation of bisbenzylidenedioxotetrahydrothiopranones as activators of endoplasmic reticulum (ER) stress signaling pathways and apoptotic cell death in acute promyelocytic leukemic cells. J. Med. Chem. 2014, 57, 5904–5918. [Google Scholar] [CrossRef] [PubMed]
- Liang, G.; Shao, L.; Wang, Y.; Zhao, C.; Chu, Y.; Xiao, J.; Zhao, Y.; Li, X.; Yang, S. Exploration and synthesis of curcumin analogues with improved structural stability both in vitro and in vivo as cytotoxic agents. Bioorg. Med. Chem. 2009, 17, 2623–2631. [Google Scholar] [CrossRef] [PubMed]
- Quincoces Suarez, J.A.; Rando, D.G.; Santos, R.P.; Gonçalves, C.P.; Ferreira, E.; de Carvalho, J.E.; Kohn, L.; Maria, D.A.; Faião-Flores, F.; Michalik, D.; et al. New antitumoral agents I: In vitro anticancer activity and in vivo acute toxicity of synthetic 1,5-bis(4-hydroxy-3-methoxyphenyl)-1,4-pentadien-3-one and derivatives. Bioorg. Med. Chem. 2010, 18, 6275–6281. [Google Scholar] [CrossRef] [PubMed]
- Ohori, H.; Yamakoshi, H.; Tomizawa, M.; Shibuya, M.; Kakudo, Y.; Takahashi, A.; Takahashi, S.; Kato, S.; Suzuki, T.; Ishioka, C.; et al. Synthesis and biological analysis of new curcumin analogues bearing an enhanced potential for the medicinal treatment of cancer. Mol. Cancer Ther. 2006, 5, 2563–2571. [Google Scholar] [CrossRef] [PubMed]
- Yamakoshi, H.; Ohori, H.; Kudo, C.; Sato, A.; Kanoh, N.; Ishioka, C.; Shibata, H.; Iwabuchi, Y. Structure-activity relationship of C5-curcuminoids and synthesis of their molecular probes thereof. Bioorg. Med. Chem. 2010, 18, 1083–1092. [Google Scholar] [CrossRef] [PubMed]
- Masuda, T.; Jitoe, A.; Isobe, J.; Nakatani, N.; Yonemori, S. Anti-oxidative and anti-inflammatory curcumin-related phenolics from rhizomes of Curcuma domestica. Phytochemistry 1993, 32, 1557–1560. [Google Scholar] [CrossRef]
- Jiang, J.L.; Jin, X.L.; Zhang, H.; Su, X.; Qiao, B.; Yuan, Y.J. Identification of antitumor constituents in curcuminoids from Curcuma longa L. based on the composition-activity relationship. J. Pharm. Biomed. Anal. 2012, 70, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Shibata, H.; Yamakoshi, H.; Sato, A.; Ohori, H.; Kakudo, Y.; Kudo, C.; Takahashi, Y.; Watanabe, M.; Takano, H.; Ishioka, C.; et al. Newly synthesized curcumin analog has improved potential to prevent colorectal carcinogenesis in vivo. Cancer Sci. 2009, 100, 956–960. [Google Scholar] [CrossRef] [PubMed]
- Kudo, C.; Yamakoshi, H.; Sato, A.; Ohori, H.; Ishioka, C.; Iwabuchi, Y.; Shibata, H. Novel curcumin analogs, GO-Y030 and GO-Y078, are multi-targeted agents with enhanced abilities for multiple myeloma. Anticancer Res. 2011, 31, 3719–3726. [Google Scholar] [PubMed]
- Lin, L.; Liu, Y.; Li, H.; Li, P.-K.; Fuchs, J.; Shibata, H.; Iwabuchi, Y.; Lin, J. Targeting colon cancer stem cells using a new curcumin analogue, GO-Y030. Br. J. Cancer 2011, 105, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Yamakoshi, H.; Kanoh, N.; Kudo, C.; Sato, A.; Ueda, K.; Muroi, M.; Kon, S.; Satake, M.; Ohori, H.; Ishioka, C.; et al. KSRP/FUBP2 is a binding protein of GO-Y086, a cytotoxic curcumin analogue. ACS Med. Chem. Lett. 2010, 1, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Guengerich, F.P.; MacDonald, J.S. Applying mechanisms of chemical toxicity to predict drug safety. Chem. Res. Toxicol. 2007, 20, 344–369. [Google Scholar] [CrossRef] [PubMed]
- Park, B.K.; Boobis, A.; Clarke, S.; Goldring, C.E.P.; Jones, D.; Kenna, J.G.; Lambert, C.; Laverty, H.G.; Naisbitt, D.J.; Nelson, S.; et al. Managing the challenge of chemically reactive metabolites in drug development. Nat. Rev. Drug Discov. 2011, 10, 292–306. [Google Scholar] [CrossRef] [PubMed]
- Park, B.K.; Pirmohamed, M.; Kitteringham, N.R. Role of drug disposition in drug hypersensitivity: A chemical, molecular, and clinical perspective. Chem. Res. Toxicol. 1998, 11, 969–988. [Google Scholar] [CrossRef] [PubMed]
- Dahmke, I.N.; Boettcher, S.P.; Groh, M.; Mahlknecht, U. Cooking enhances curcumin anti-cancerogenic activity through pyrolytic formation of “deketene curcumin”. Food Chem. 2014, 151, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Ciaccio, J.A.; Aman, C.E. “Instant Methylide” modification of the corey-chaykovsky cyclopropanation reaction. Synth. Commun. 2006, 36, 1333–1341. [Google Scholar] [CrossRef]
- Baskin, J.M.; Prescher, J.A.; Laughlin, S.T.; Agard, N.J.; Chang, P.V.; Miller, I.A.; Lo, A.; Codelli, J.A.; Bertozzi, C.R. Copper-free click chemistry for dynamic in vivo imaging. Proc. Natl. Acad. Sci. USA 2007, 104, 16793–16797. [Google Scholar] [CrossRef] [PubMed]
- Karimi, S.; Grohmann, K.G.; Todaro, L. Intramolecular ring-opening of cyclopropanones by enolate anions. J. Org. Chem. 1995, 60, 554–559. [Google Scholar] [CrossRef]
- Zheng, S.; Laxmi, Y.R.S.; David, E.; Dinkova-Kostova, A.T.; Shiavoni, K.H.; Ren, Y.; Zheng, Y.; Trevino, I.; Bumeister, R.; Ojima, I.; et al. Synthesis, chemical reactivity as Michael acceptors, and biological potency of monocyclic cyanoenones, novel and highly potent anti-inflammatory and cytoprotective agents. J. Med. Chem. 2012, 55, 4837–4846. [Google Scholar] [CrossRef] [PubMed]
- Parrish, J.P.; Kastrinsky, D.B.; Wolkenberg, S.E.; Igarashi, Y.; Boger, D.L. DNA alkylation properties of yatakemycin. J. Am. Chem. Soc. 2003, 125, 10971–10976. [Google Scholar] [CrossRef] [PubMed]
- MacMillan, K.S.; Boger, D.L. Fundamental relationships between structure, reactivity, and biological activity for the duocarmycins and CC-1065. J. Med. Chem. 2009, 52, 5771–5780. [Google Scholar] [CrossRef] [PubMed]
- Serafimova, I.M.; Pufall, M.A.; Krishnan, S.; Duda, K.; Cohen, M.S.; Maglathlin, R.L.; McFarland, J.M.; Miller, R.M.; Frödin, M.; Taunton, J. Reversible targeting of noncatalytic cysteines with chemically tuned electrophiles. Nat. Chem. Biol. 2012, 8, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Wissner, A.; Mansour, T.S. The development of HKI-272 and related compounds for the treatment of cancer. Arch. Pharm. (Weinheim) 2008, 341, 465–477. [Google Scholar] [CrossRef] [PubMed]
- Honda, T.; Rounds, B.V.; Gribble, G.W.; Suh, N.; Wang, Y.; Sporn, M.B. Design and synthesis of 2-cyano-3,12-dioxoolean-1,9-dien-28-oic acid, a novel and highly active inhibitor of nitric oxide production in mouse macrophages. Bioorg. Med. Chem. Lett. 1998, 8, 2711–2714. [Google Scholar] [CrossRef]
- Al-Rifai, N.; Rücker, H.; Amslinger, S. Opening or closing the lock? When reactivity is the key to biological activity. Chem. Eur. J. 2013, 19, 15384–15395. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Ab Aziz, F.H.; Syahida, A.; Abas, F.; Shaari, K.; Israf, D.A.; Lajis, N.H. Synthesis and biological evaluation of curcumin-like diarylpentanoid analogues for anti-inflammatory, antioxidant and anti-tyrosinase activities. Eur. J. Med. Chem. 2009, 44, 3195–3200. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: GO-Y030 is available from Tokyo Chemical Industry (http://www.tcichemicals.com/eshop/en/gb/commodity/B4823/).
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kohyama, A.; Yamakoshi, H.; Hongo, S.; Kanoh, N.; Shibata, H.; Iwabuchi, Y. Structure-Activity Relationships of the Antitumor C5-Curcuminoid GO-Y030. Molecules 2015, 20, 15374-15391. https://doi.org/10.3390/molecules200815374
Kohyama A, Yamakoshi H, Hongo S, Kanoh N, Shibata H, Iwabuchi Y. Structure-Activity Relationships of the Antitumor C5-Curcuminoid GO-Y030. Molecules. 2015; 20(8):15374-15391. https://doi.org/10.3390/molecules200815374
Chicago/Turabian StyleKohyama, Aki, Hiroyuki Yamakoshi, Shoko Hongo, Naoki Kanoh, Hiroyuki Shibata, and Yoshiharu Iwabuchi. 2015. "Structure-Activity Relationships of the Antitumor C5-Curcuminoid GO-Y030" Molecules 20, no. 8: 15374-15391. https://doi.org/10.3390/molecules200815374
APA StyleKohyama, A., Yamakoshi, H., Hongo, S., Kanoh, N., Shibata, H., & Iwabuchi, Y. (2015). Structure-Activity Relationships of the Antitumor C5-Curcuminoid GO-Y030. Molecules, 20(8), 15374-15391. https://doi.org/10.3390/molecules200815374