Mentha spicata Essential Oil: Chemical Composition, Antioxidant and Antibacterial Activities against Planktonic and Biofilm Cultures of Vibrio spp. Strains
Abstract
:1. Introduction
2. Results and Discussion
2.1. Essential Oil Composition
| Components | I.r.i. a | Percent |
|---|---|---|
| α-pinene | 941 | 1.4 ± 0.17 |
| camphene | 955 | 0.2 ± 0.06 |
| sabinene | 977 | 1.4 ± 0.06 |
| β-pinene | 982 | 2.2 ± 0.25 |
| myrcene | 993 | 1.1 ± 0.15 |
| 3-octanol | 994 | 1.0 ± 0.21 |
| p-cymene | 1028 | 0.8 ± 0.06 |
| limonene | 1032 | 20.8 ± 1.12 |
| 1,8-cineole | 1034 | 17.0 ± 0.60 |
| (Z)-β-ocimene | 1042 | 0.2 ± 0.06 |
| cis-sabinene hydrate | 1070 | 1.6 ± 0.15 |
| linalool | 1101 | 0.4 ± 0.12 |
| cis-p-menth-2-en-1-ol | 1123 | 0.1 ± 0.00 |
| cis-limonene oxide | 1138 | 0.1 ± 0.06 |
| trans-limonene oxide | 1141 | 0.1 ± 0.00 |
| borneol | 1171 | 0.1 ± 0.06 |
| δ-terpineol | 1172 | 0.4 ± 0.12 |
| 4-terpineol | 1179 | 1.3 ± 0.26 |
| α-terpineol | 1191 | 0.5 ± 0.10 |
| dihydrocarveol | 1194 | 1.7 ± 0.31 |
| cis-dihydrocarvone | 1195 | 1.9 ± 0.49 |
| trans-carveol | 1219 | 0.4 ± 0.06 |
| cis-carveol | 1231 | 0.6 ± 0.15 |
| pulegone | 1239 | 0.3 ± 0.06 |
| carvone | 1244 | 40.8 ± 1.23 |
| isobornyl acetate | 1287 | 0.1 ± 0.00 |
| iso-dihydrocarveol acetate | 1327 | 0.2 ± 0.06 |
| β-bourbonene | 1385 | 0.9 ± 0.17 |
| β-elemene | 1392 | 0.3 ± 0.06 |
| β-caryophyllene | 1419 | 1.2 ± 0.25 |
| germacrene D | 1481 | 0.2 ± 0.06 |
| germacrene A | 1506 | 0.2 ± 0.15 |
| spathulenol | 1578 | 0.1 ± 0.00 |
| caryophyllene oxide | 1582 | 0.3 ± 0.06 |
| monoterpene hydrocarbons | 45.1 | |
| oxygenated monoterpenes | 50.6 | |
| sesquiterpene hydrocarbons | 2.8 | |
| oxygenated sesquiterpenes | 0.4 | |
| others | 1.0 | |
| total identified | 99.9 |
2.2. Antioxidant Activity
| Activity (µg/mL) | Spearmint Oil | BHT | BHA | Ascorbic Acid | EDTA |
|---|---|---|---|---|---|
| DPPH IC50 | 3.08 ± 0.07 | 11.48 ± 0.02 | - | - | - |
| Reducing Power EC50 | 2.49 ± 0.07 | - | - | 37.53 ± 0.39 | - |
| Chelating Power IC50 | 6.33 ± 0.12 | - | - | - | 32 ± 0 |
| β-Carotenes IC50 | 6.4 ± 0.07 | - | 48.00 ± 0.50 | - | - |
2.3. Anti-Vibrio spp. Activity
| Microorganisms | M. spicata Essential Oil | ||
|---|---|---|---|
| GIZ ± SD | MIC | MBC | |
| V. cholerae ATCC 9459 | 13.67 ± 0.58 f,g | 0.023 | 12 |
| V. cholerae (Granchi, Ancona) | 14.67 ± 0.58 f | 0.023 | 12 |
| V. vulnificus ATCC 27562 | 13.33 ± 0.58 g | 0.023 | 6 |
| V. vulnificus S5 (D. labrax, Chebba) | 14.67 ± 0.58 e,f | 0.047 | 6 |
| V. vulnificus V30 (S. aurata, Hergla) | 13.67 ± 0.58 f,g | 0.023 | 6 |
| V. parahaemolyticus ATCC 17802 | 14.67 ± 0.58 e,f | 0.023 | 12 |
| V. parahaemolyticus ATCC 43996 | 15.67 ± 0.58 d | 0.023 | 12 |
| V. parahaemolyticus I12 (Seawater, Italy) | 17.33 ± 0.58 c | 0.023 | 12 |
| V. parahaemolyticus I22 (Seawater, Italy) | 19.33 ± 0.58 b | 0.047 | 6 |
| V. parahaemolyticus (Malaga, Spain) | 14.33 ± 0.58 f,g | 0.047 | 3 |
| V. parahaemolyticus S949 (M. edulis, Bizerte) | 10 ± 0 i | 0.047 | 12 |
| V. parahaemolyticus S950 (M. edulis, Bizerte) | 11.67 ± 0.58 h | 0.023 | 12 |
| V. alginolyticus ATCC 33787 | 18.67 ± 0.58 b | 0.023 | 24 |
| V. alginolyticus ATCC 17749 | 7 ± 0 j | 0.047 | 12 |
| V. alginolyticus (Malaga, Spain) | 21.33 ± 0.58 a | 0.047 | 12 |
| V. alginolyticus S6 (D. labrax, Chebba) | 12.33 ± 0.58 h | 0.023 | 1.5 |
| V. alginolyticus S7 (M. edulis, Bizerte) | 13.67 ± 0.58 f,g | 0.023 | 0.75 |
| V. alginolyticus S8 (S. aurata, Hergla) | 14.33 ± 0.58 f,g | 0.023 | 6 |
| V. furnisii ATCC 35016 | 14.33 ± 0.58 f,g | 0.047 | 6 |
| V. cincinnatiensis ATCC 35912 | 13.67 ± 0.58 f,g | 0.023 | 12 |
| V. proteolyticus ATCC 15338 | 14.33 ± 0.58 f,g | 0.023 | 12 |
| V. natrigens ATCC 14048 | 13.67 ± 0.58 f,g | 0.023 | 12 |
| V. mimicus ATCC 33653 | 15.67 ± 0.58 d | 0.047 | 12 |
| V. fluvialis ATCC 33809 | 13.33 ± 0.58 g | 0.023 | 12 |
| V. anguillarum (Malaga, Spain) | 13.67 ± 0.58 f,g | 0.023 | 12 |
| V. carchariae ATCC 35084 | 9.67 ± 0.58 i | 0.023 | 12 |
| V. harveyii ATCC 18293 | 13.67 ± 0.58 f,g | 0.047 | 12 |
| V. diazotrophicus ATCC 33466 | 14.67 ± 0.58 e,f | 0.047 | 12 |
| V. tapetis CECT 4600T | 14.57 ± 0.58 e,f | 0.023 | 12 |
| V. splendidus ATCC 33125 | 17.67 ± 0.58 c | 0.047 | 12 |
| A. hydrophila ATCC 7966T | 14.67 ± 0.58 e,f | 0.023 | 12 |
| Vibrio strain | 1 | 2 | 3 | 4 | 5 | 6 | |
|---|---|---|---|---|---|---|---|
| E. caryophyllata * | GIZ | 11.33 ± 0.57 | 10.66 ± 0.57 | 13.66 ± 0.57 | 12.33 ± 0.57 | 11.66 ± 0.57 | 10 ± 0 |
| MIC | 0.15 | 0.31 | 0.156 | 0.156 | 0.156 | 0.156 | |
| MBC | 1.25 | 2.5 | >1.25 | 0.625 | 1.25 | 1.25 | |
| T. vulgaris * | GIZ | 13.33 ± 0.57 | 14 ± 1 | 14.66 ± 0.57 | 22.33 ± 0.57 | 12.66 ± 0.57 | 13 ± 1 |
| MIC | 0.078 | 0.156 | 0.156 | 0.156 | 0.156 | 0.156 | |
| MBC | 0.625 | 1.25 | 0.312 | 0.312 | 1.25 | 0.625 | |
| R. officinalis * | GIZ | - | 7 ± 0 | 9.33 ± 0.57 | 12.33 ± 0.57 | 14 ± 0 | 12 ± 1 |
| MIC | 0.31 | 0.195 | 0.625 | 1.25 | 0.156 | 0.156 | |
| MBC | 5 | >5 | >2.5 | 2.5 | 2.5 | 2.5 | |
| C. cyminum ** | GIZ | 11 ± 0 | 20.33 ± 0.58 | 15 ± 0 | 13.33 ± 0.58 | 12 ± 0 | 14.67 ± 0.58 |
| MIC | 0.312 | 0.156 | 0.156 | 0.078 | 0.156 | 0.312 | |
| MBC | 0.625 | 0.625 | 1.25 | 0.625 | 1.25 | 1.25 | |
| M. pulegium * | GIZ | 10 ± 1 | 13.33 ± 0.57 | 9 ± 0 | 11 ± 0 | 7 ± 0 | 8 ± 0 |
| MIC | 0.097 | 0.78 | 0.39 | 0.195 | 0.39 | 0.195 | |
| MBC | 3.125 | 6.25 | 3.125 | >1.56 | 3.125 | 3.125 | |
| M. longifolia * | GIZ | 9.33 ± 1.15 | 7.66 ± 0.57 | 8.66 ± 1.15 | 12 ± 0 | 11 ± 0 | 11 ± 0 |
| MIC | 0.78 | 0.78 | 0.195 | 0.39 | 0.39 | 0.39 | |
| MBC | >3.125 | 6.25 | 3.125 | 1.56 | 6.25 | 6.25 | |
| M. spicata *** | GIZ | 18.67 ± 0.58 | 17.67 ± 0.58 | 14.67 ± 0.58 | 15.67 ± 0.58 | 13.33 ± 0.58 | 13.33 ± 0.58 |
| MIC | 0.023 | 0.047 | 0.023 | 0.023 | 0.023 | 0.023 | |
| MBC | >24 | >12 | >12 | >12 | >6 | >12 | |
2.4. Biofilm Inhibition and Eradication
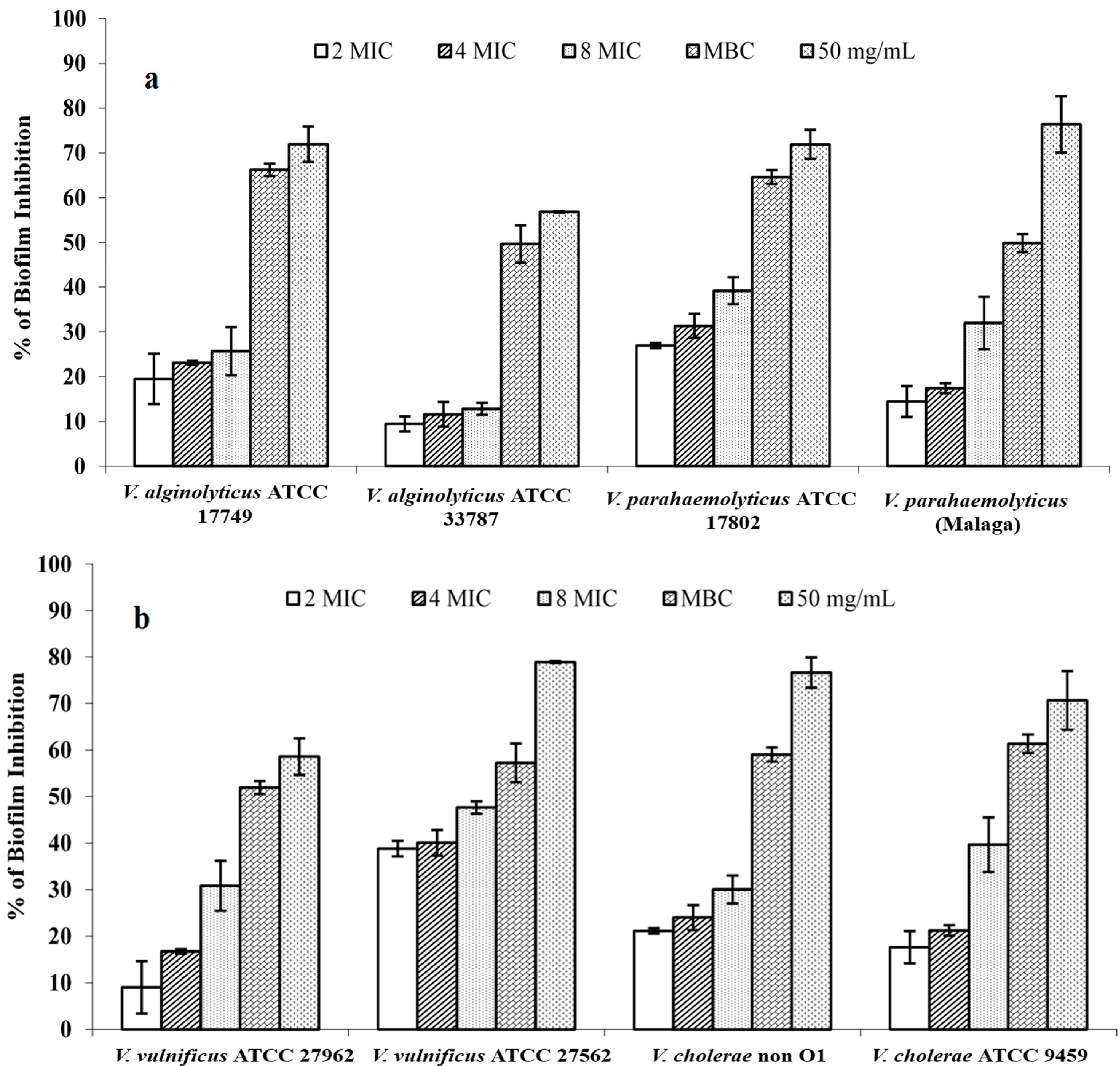
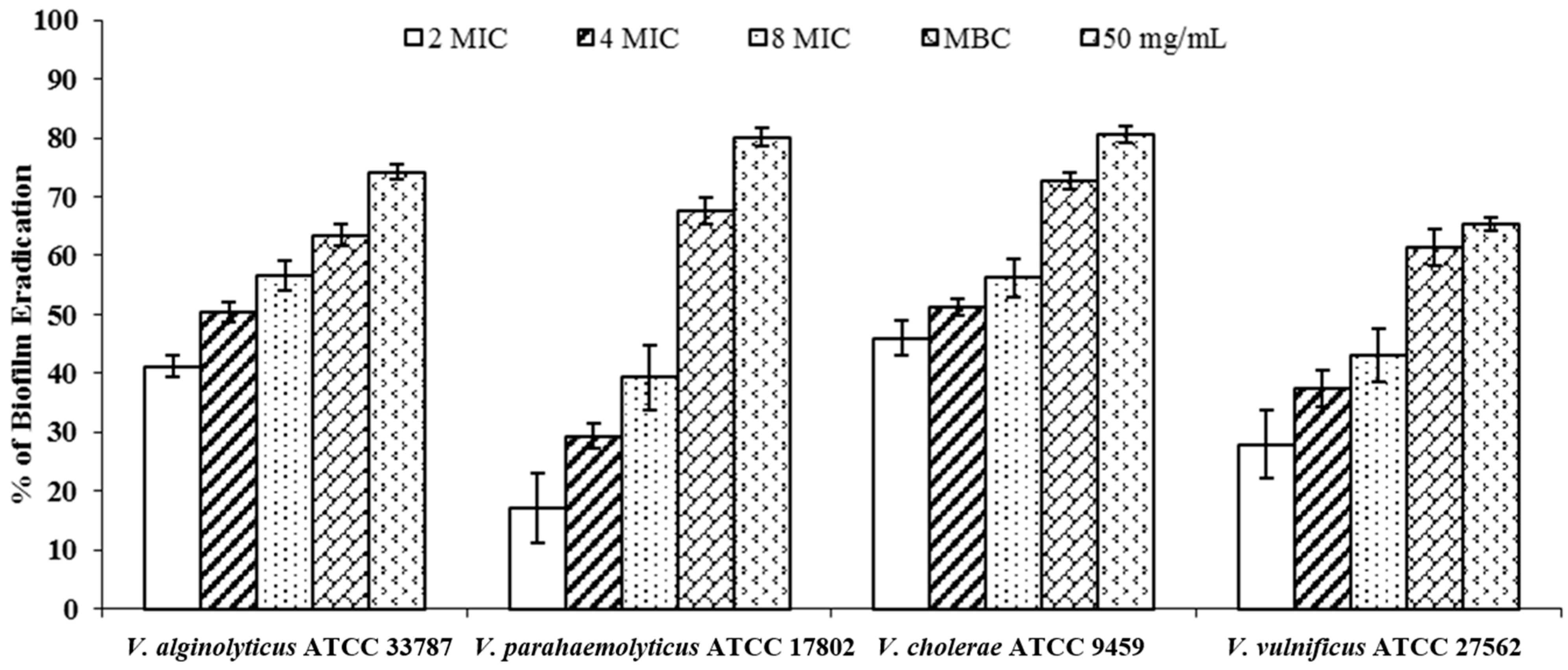
3. Experimental Section
3.1. Plant Material and Extraction of Essential Oil
3.2. GC-EIMS Analysis
3.3. Antioxidant Properties
3.3.1. DPPH Radical-Scavenging Activity.
3.3.2. Metal Chelating Activity.
3.3.3. Determination of Reducing Power
3.3.4. Anti-Peroxyl Radical Activity.
3.4. Antimicrobial Activity
3.4.1. Microorganisms
3.4.2. Disk-Diffusion Assay
3.4.3. Micro-Well Determination of MIC and MBC
3.5. Assessment of Vibrio spp. Biofilm Metabolic Activity Using XTT Reduction Assay
3.6. Effect of Essential Oils on Vibrio spp. Biofilm Formation
3.7. Effect on Established Biofilms
3.8. Statistical Analysis
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Pottier-Alapetite, G. Flore de la Tunisie, Angiospermes-Dicotylédones Gamopétales; Publication Scientifiques Tunisiennes: Tunis, Tunisie, 1961. [Google Scholar]
- Leporatti, M.L.; Ghedira, K. Comparative analysis of medicinal plants used in traditional medicine in Italy and Tunisia. J. Ethnobiol. Ethnomed. 2009, 5, 31–39. [Google Scholar]
- Lawrence, B.M. Mint: The Genus Mentha; Taylor and Francis Group: Boca Raton, FL, USA, 2007. [Google Scholar]
- Karousou, R.; Balta, M.; Hanlidou, E. Mints, smells and traditional uses in Thessaloniki (Greece) and other Mediterranean countries. J. Ethnopharmacol. 2007, 109, 248–257. [Google Scholar] [PubMed]
- Hajlaoui, H.; Snoussi, M.; Jannet, B.; Mighri, Z.; Bakhrouf, A. Comparison of chemical composition and antimicrobial activities of Mentha longifolia L. ssp. longifolia essential oil from two Tunisian localities (Gabes and Sidi Bouzid). Ann. Microbiol. 2008, 58, 513–520. [Google Scholar]
- Hajlaoui, H.; Trabelsi, N.; Noumi, E.; Snoussi, M.; Fallah, H.; Ksouri, R.; Bakhrouf, A. Biological activities of the essential oils and methanol extract of two cultivated mint species (Mentha longifolia and Mentha pulegium) used in the Tunisian folkloric medicine. World J. Microbiol. Biotechnol. 2009, 25, 2227–2238. [Google Scholar]
- Padalia, R.C.; Verma, R.S.; Chauhan, A.; Sundaresan, V.; Chandan, S.C. Essential oil composition of sixteen elite cultivars of Mentha from western Himalayan region, India. Maejo Int. J. Sci. Technol. 2013, 7, 83–93. [Google Scholar]
- Khanuja, S.P.S. Employ contract farming to boost area under cultivation for essential oil bearing crops. Chem. Wkly. 2005, 25, 180–181. [Google Scholar]
- Boukef, M.K. Traditional Medicine and Pharmacopoeia. In Plants in the Traditional Tunisian Medicine; Agency for Cultural and Technical Cooperation: Paris, France, 1986; p. 350. [Google Scholar]
- Bensabah, F.; Houbairi, S.; Essahli, M.; Lamiri, A.; Naja, J. Chemical Composition and Inhibitory Effect of the Essential Oil from Mentha spicata Irrigated by Wastewater on the Corrosion of Aluminum in 1 Molar Hydrochloric Acid. Port. Electrochim. Acta 2013, 31, 195–206. [Google Scholar]
- Tawaha, K.; Alali, F.Q.; Gharaibeh, M.; Mohamed, M.; El Elimat, T. Antioxidant activity and total phenolic content of selected Jordanian plant species. Food Chem. 2007, 104, 1372–1378. [Google Scholar]
- Kizil, S.; Hasimi, N.; Tolan, V.; Kilinç, E.; Yüksel, U. Mineral content, essential oil components and biological activity of two Mentha species (M. piperita L., M. spicata L.). Turk. J. Field Crops 2010, 15, 148–153. [Google Scholar]
- Tetika, F.; Civelek, S.; Cakilcioglu, U. Traditional uses of some medicinal plants in Malatya (Turkey). J. Ethnopharmacol. 2013, 146, 331–346. [Google Scholar]
- Beuchat, L.R. Sensitivity of Vibrio parahaemolyticus to spices and organic acids. J. Food Sci. 1976, 41, 899–902. [Google Scholar]
- Snoussi, M.; Hajlaoui, H.; Noumi, E.; Usai, D.; Sechi, L.A.; Zanetti, S.; Bakhrouf, A. In-vitro anti-Vibrio spp. activity and chemical composition of some Tunisian aromatic plants. World J. Microbiol. Biotechnol. 2008, 24, 3071–3076. [Google Scholar]
- Hajlaoui, H.; Mighri, H.; Noumi, E.; Snoussi, M.; Trabelsi, N.; Ksouri, R.; Bakhrouf, A. Chemical composition and biological activities of Tunisian Cuminum cyminum L. essential oil: A high effectiveness against Vibrio spp. strains. Food Chem. Toxicol. 2010, 48, 2186–2192. [Google Scholar] [PubMed]
- Birkenhauer, J.B.; Oliver, J.D. Use of diacetyl to reduce the load of Vibrio vulnificus in the Eastern oyster, Crassostrea virginica. J. Food Protect. 2003, 66, 38–43. [Google Scholar]
- Hasegawa, N.; Matsumoto, Y.; Hoshino, A.; Iwashita, K. Comparison of effects of Wasabia japonica and allyl isothiocyanate on the growth of four strains of Vibrio parahaemolyticus in lean and fatty tuna meat suspensions. Int. J. Food Microbiol. 1999, 49, 27–34. [Google Scholar] [PubMed]
- Baser, K.H.C.; Kürkçüoglu, M.; Tarimcilar, G.; Kaynak, G. Essential Oils of Mentha species from Northern Turkey. J. Essent. Oil Res. 1999, 11, 579–588. [Google Scholar]
- Telci, I.; Sahbaz, N.; Yilmaz, G.; Tugay, M.E. Agronomical and chemical characterization of spearmint (Mentha spicata L.) originating in Turkey. Econ. Bot. 2004, 58, 721–728. [Google Scholar]
- Telci, I.; Demirtas, I.; Bayram, E.; Arabaci, O.; Kacar, O. Environmental variation on aroma components of pulegone/piperitone rich spearmint (Mentha spicata L.). Ind. Crops Prod. 2010, 32, 588–592. [Google Scholar] [CrossRef]
- Kokkini, S.; Karousou, R.; Lanaras, T. Essential oils of spearmint (carvone-rich) plants from the island of Crete (Greece). Biochem. Syst. Ecol. 1995, 23, 425–430. [Google Scholar] [CrossRef]
- Mkaddem, M.; Bouajila, J.; Ennajar, M.; Lebrihi, A.; Mathieu, F.; Romdhane, M. Chemical composition and antimicrobial and antioxidant activities of Mentha (longifolia L. and viridis) essential oils. J. Food Sci. 2009, 74, 358–363. [Google Scholar] [CrossRef] [PubMed]
- Chou, G.X.; Zhou, R.H. The variation and chemotypes of the essential oil components in wild Mentha haplocalyx Briq. in China. J. Plant Resour. Environ. 1998, 7, 13–18. [Google Scholar]
- Bahl, J.R.; Bansal, R.P.; Garg, S.N.; Naqvi, A.A.; Luthra, R.; Kukreja, A.; Kumar, S. Qualitative evaluation of the essential oils of the prevalent cultivars of commercial mint species Mentha arvensis, M. spicata, M. piperita, M. cardiaca, M. citrata and M. viridis cultivated in indo-gangetic plains. J. Med. Arom. Plant Sci. 2000, 22, 787–797. [Google Scholar]
- El-Waheb, A.; Mohamed, A. Evaluation of spearmint (Mentha spicata L.) productivity grown in different locations under upper Egypt conditions. Res. J. Agric. Biol. Sci. 2009, 5, 250–254. [Google Scholar]
- Foda, M.I.; El-Sayed, M.A.; Hassan, A.A.; Rasmy, N.M.; El-Moghazy, M.M. Effect of spearmint essential oil on chemical composition and sensory properties of white cheese. J. Am. Sci. 2010, 6, 272–279. [Google Scholar]
- Zheljazkov, V.D.; Cantrell, C.L.; Astatkies, T. Yield and composition of oil from Japanese cornmint fresh and dry material harvested successively. Agron. J. 2010, 102, 1652–1656. [Google Scholar] [CrossRef]
- Roldán, L.P.; Díaz, G.J.; Duringer, J.M. Composition and antibacterial activity of essential oils obtained from plants of the Lamiaceae family against pathogenic and beneficial bacteria. Rev. Colomb. Cienc. Pec. 2010, 23, 451–461. [Google Scholar]
- Telci, I.; Sahbaz, N. Variations in yield, essential oil and carvone contents in clones selected from Carvone-scented landraces of Turkish Mentha species. J. Agron. 2005, 4, 96–102. [Google Scholar]
- Hua, C.X.; Wang, G.R.; Lei, Y. Evaluation of essential oil composition and DNA diversity of mint resources from China. Afr. J. Biotechnol. 2011, 10, 16740–16745. [Google Scholar]
- Chowdhury, J.U.; Nandi, N.C.; Uddin, M.; Rahman, M. Chemical constituents of essential oils from two types of spearmint (Mentha spicata L. and M. cardiaca L.) introduced in Bangladesh. Bangl. J. Sci. Ind. Res. 2007, 42, 79–82. [Google Scholar]
- Boukhebti, H.; Chaker, A.N.; Belhadj, H.; Sahli, F.; Ramdhani, M.; Laouer, H.; Harzallah, D. Chemical composition and antibacterial activity of Mentha pulegium L. and Mentha spicata L. essential oils. Der. Pharm. Lett. 2011, 3, 267–275. [Google Scholar]
- Znini, M.; Bouklah, M.; Majidi, L.; Kharchouf, S.; Aouniti, A.; Bouyanzer, A.; Hammouti, B.; Costa, J.; Al-Dyab, S.S. Chemical composition and inhibitory effect of Mentha spicata essential oil on the corrosion of steel in molar hydrochloric acid. Int. J. Electrochem. Sci. 2011, 6, 691–704. [Google Scholar]
- Hadjiakhoondi, A.; Aghel, N.; Zamanizadech-Nadgar, N.; Vatandoost, H. Chemical and biological study of Mentha spicata L. essential oil from Iran. DARU J. Pharm. Sci. 2000, 8, 19–21. [Google Scholar]
- Rasooli, I.; Gachkar, L.; Yadegarinia, D.; Bagher, M.R.; Astaneh, S.D.A. Antibacterial and antioxidative characterization of essential oils from Mentha piperita and Mentha spicata grown in Iran. Acta Aliment. 2008, 37, 41–52. [Google Scholar] [CrossRef]
- Sokovic, M.D.; Vukojevic, J.; Marin, P.D.; Brkic, D.D.; Vajs, V.; van Griensven, L.J.L.D. Chemical composition of essential oils of Thymus and Mentha species and their antifungal activities. Molecules 2009, 14, 238–249. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, R.S.; Nautiyal, M.C.; Tava, A. Essential oil composition from aerial parts of Mentha spicata L. J. Essent. Oil Bear. Pl. 2010, 13, 353–356. [Google Scholar] [CrossRef]
- Omarr, N.N.; El-Sayed, Z.I.A.; Romeh, A.A. Chemical constituent and biological activity of the essential oil of Mentha spicata L. grown in Zagazig region, Egypt. Res. J. Agric. Biol. Sci. 2009, 5, 1089–1097. [Google Scholar]
- Dhifi, W.; Jelali, N.; Mnif, W.; Litaiem, M.; Hamdi, N. Chemical composition of the essential oil of Mentha spicata from Tunisia and its biological activities. J. Food Biochem. 2013, 37, 362–368. [Google Scholar] [CrossRef]
- Zhao, D.; Xu, Y.W.; Yang, G.L.; Husaini, A.M.; Wu, W. Variation of essential oil of Mentha haplocalyx Briq. and Mentha spicata L. Ind. Crops Prod. 2013, 42, 251–260. [Google Scholar] [CrossRef]
- Arumugam, P.; Ramamurthy, P.; Santhiya, S.T.; Ramesh, A. Antioxidant activity measured in different solvent fractions obtained from Mentha spicata Linn.: An analysis by ABTS+ decolorization assay. Asia Pac. J. Clin. Nutr. 2006, 15, 119–124. [Google Scholar] [PubMed]
- Samarth, R.M.; Kumar, A. Mentha piperita (Linn.) leaf extract provides protection against radiation induced chromosomal damage in bone marrow of mice. Indian J. Exp. Biol. 2003, 41, 229–237. [Google Scholar] [PubMed]
- Ruberto, G.; Baratta, M.T.; Deans, S.G.; Dorman, H.J.D. Antioxidant and antimicrobial activity of Foeniculum vulgare and Crithmum maritimum essential oils. Planta Med. 2000, 66, 687–693. [Google Scholar] [CrossRef] [PubMed]
- Dorman, H.J.D.; Kosar, M.; Kahlos, K.; Holm, Y.; Hiltunen, R. Antioxidant properties and composition of aqueous extracts from Mentha species, hybrids, varieties, and cultivars. J. Agric. Food Chem. 2003, 51, 4563–4569. [Google Scholar] [CrossRef] [PubMed]
- Mata, A.T.; Proenca, C.; Ferreira, A.R.; Serralheiro, M.L.M.; Nougueira, J.M.F.; Arujo, E.M. Antioxidant and antiacetylcholinesterase activities of five plants used as Portuguese food species. Food Chem. 2007, 103, 778–786. [Google Scholar] [CrossRef]
- Nickavar, B.; Alinaghi, A.; Kamalinejad, M. Evaluation of the antioxidant properties of five Mentha species. Iran. J. Pharm. Res. 2008, 7, 203–209. [Google Scholar]
- Naidu, J.R.; Ismail, R.B.; Yeng, C.; Sasidharan, S.; Kumar, P. Chemical composition and antioxidant activity of the crude methanolic extracts of Mentha spicata. J. Phytol. 2012, 4, 13–18. [Google Scholar]
- Martins, M.R.; Tinoco, M.T.; Almeida, A.S.; Cruz-Morais, J. Chemical composition, antioxidant and antimicrobial properties of three essential oils from Portuguese flora. J. Pharmacogn. 2012, 3, 39–44. [Google Scholar]
- Yano, Y.; Satomi, M.; Oikawa, H. Antimicrobial effect of spices and herbs on Vibrio parahaemolyticus. Int. J. Food Microbiol. 2006, 111, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Shelef, L.A.; Naglik, O.A.; Bogen, D.W. Sensitivity of some common food-borne bacteria to the spices sage, rosemary and allspice. J. Food Sci. 1980, 45, 1042–1044. [Google Scholar] [CrossRef]
- Ghasemi Pirbalouti, A.; Hamedi, B.; Poor, F.M.; Rahimi, E.; Nejhad, R.N. Inhibitory activity of Iranian endemic medicinal plants against Vibrio parahaemolyticus and Vibrio harveyi. J. Med. Plants Res. 2011, 5, 7049–7053. [Google Scholar] [CrossRef]
- Lorenzi, V.; Muselli, A.; Bernardini, A.F.; Berti, L.; Pagès, J.M.; Amaral, L.; Bolla, J.M. Geraniol restores antibiotic activities against multidrug resistant isolates from gram-negative species. Antimicrob. Agents Chemother. 2009, 53, 2209–2211. [Google Scholar] [CrossRef] [PubMed]
- Sokovic, M.; Grubišic, D.; Ristic, M. Chemical composition and antifungal activity of the essential oils from leaves, calyx and corolla of Salvia brachyodon Vandas. J. Essent. Oil Res. 2005, 17, 227–229. [Google Scholar] [CrossRef]
- Sokovic, M.; van Griensven, L.J.L.D. Antimicrobial activity of essential oils and their components against the three major pathogens of the cultivated button mushroom, Agaricus bisporus. Eur. J. Plant Pathol. 2006, 116, 211–224. [Google Scholar] [CrossRef]
- Knobloch, K.; Weigand, H.; Weis, N.; Schwarm, H.M.; Vigenschow, H. Action of Terpenoids on Energy Metabolism. In Progress in Essential Oil Research; Brunke, E.J., Ed.; Walter de Gruyter: Berlin, Germany, 1986; pp. 429–445. [Google Scholar]
- Elmastaş, M.; Dermirtas, I.; Isildak, O.; Aboul-Enein, H.Y. Antioxidant Activity of S-Carvone Isolated from Spearmint (Mentha spicata L. Fam Lamiaceae). J. Liq. Chromatogr. Relat. Technol. 2006, 29, 1465–1475. [Google Scholar] [CrossRef]
- Helander, I.M.; Alakomi, H.L.; Latva-Kala, K.; Mattila-Sandholm, T.; Pol, I.; Smid, E.J.; Gorris, L.G.M.; von Wright, A. Characterization of the action of selected essential oil components on Gram-negative bacteria. J. Agric. Food Chem. 1998, 46, 3590–3595. [Google Scholar] [CrossRef]
- Gallucci, N.; Oliva, M.; Carezzano, E.; Zygadlo, J.; Demo, M. Terpenes antimicrobial activity against slime producing and non–producing staphylococci. Mol. Med. Chem. 2010, 21, 132–136. [Google Scholar]
- Smid, E.J.; de Witte, Y.; Gorris, L.G.M. Secondary plant metabolites as control agents of postharvest Penicillium rot on tulip bulb. Postharvest Biol. Technol. 1995, 6, 303–312. [Google Scholar] [CrossRef]
- Lee, S.K.; Tsao, R.; Peterson, C.; Coats, J.R. Insecticidal activity of monoterpenoids to western corn rootworm (Coleoptera: Chrysomelidae), two spotted spider mite (Acari: Tetranychidae), and house fly (Diptera: Muscidae). J. Econ. Entomol. 1997, 90, 883–892. [Google Scholar] [CrossRef] [PubMed]
- Kerstholt, R.P.V.; Ree, C.M.; Moll, M.C. Environmental life cycle analysis of potato sprout inhibitors. Ind. Crops Prod. 1997, 6, 187–194. [Google Scholar] [CrossRef]
- De Carvalho, C.C.C.R.; Manuela, M.; da Fonseca, R. Carvone: Why and how should one bother to produce this terpene. Food Chem. 2006, 95, 413–422. [Google Scholar] [CrossRef]
- Uribe, S.; Ramirez, J.; Peña, A. Effects of beta-pinene on yeast membrane functions. J. Bacteriol. 1985, 161, 1195–1200. [Google Scholar] [PubMed]
- Sikkema, J.; de Bont, J.A.; Poolman, B. Mechanisms of membrane toxicity of hydrocarbons. Microbiol. Rev. 1995, 59, 201–222. [Google Scholar] [PubMed]
- Mun, S.H.; Kang, O.H.; Joung, D.K.; Kim, S.B.; Choi, J.G.; Shin, D.W.; Kwon, D.E. In vitro anti-MRSA activity of carvone with gentamicin. Exp. Ther. Med. 2014, 7, 891–896. [Google Scholar] [PubMed]
- Naigre, R.; Kalck, P.; Rogues, C.; Roux, I.; Michel, G. Comparison of antimicrobial properties of monoterpenes and their carbonylated products. Planta Med. 1996, 62, 275–277. [Google Scholar] [CrossRef] [PubMed]
- Friedman, M.; Henika, P.R.; Mandrell, R.E. Bactericidal activities of plant essential oils and some of their isolated constituents against Campylobacter jejuni, Escherichia coli, Listeria monocytogenes, and Salmonella enterica. J. Food Protect. 2002, 65, 1545–1560. [Google Scholar]
- Aggarwal, K.K.; Khanuja, S.P.S.; Ahmad, A.; Kumar, T.R.S.; Gupta, V.K.; Kumar, S. Antimicrobial activity profiles of the two enantiomers of limonene and carvone isolated from the oils of Mentha spicata and Anethum sowa. Flavour Frag. J. 2002, 17, 59–63. [Google Scholar] [CrossRef]
- Projan, S.J.; Youngman, P.J. Antimicrobials: new solutions badly needed. Curr. Opin. Microbiol. 2002, 5, 463–465. [Google Scholar] [CrossRef] [PubMed]
- Leonard, C.M.; Virijevic, S.; Regnier, T.; Combrinck, S. Bioactivity of selected essential oils and some components on Listeria monocytogenes biofilms. S. Afr. J. Bot. 2010, 76, 676–680. [Google Scholar] [CrossRef]
- Sandasi, M.; Leonard, C.M.; Viljoen, A.M. The in vitro antibiofilm activity of selected culinary herbs and medicinal plants against Listeria monocytogenes. Lett. Appl. Microbiol. 2010, 50, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Saharkhiz, M.J.; Motamedi, M.; Zomorodian, K.; Pakshi, K.; Miri, R.; Hemyari, K. Chemical Composition, Antifungal and Antibiofilm Activities of the Essential Oil of Mentha piperita L. ISRN Pharm. 2012, 718645. [Google Scholar] [CrossRef]
- Quave, C.L.; Plano, L.R.W.; Pantuso, T.; Bennett, B.C. Effects of extracts from Italian medicinal plants on planktonic growth, biofilm formation and adherence of methicillin-resistant Staphylococcus aureus. J. Ethnopharmacol. 2008, 118, 418–428. [Google Scholar] [CrossRef] [PubMed]
- Rasooli, I.; Shayegh, S.; Astaneh, S. The effect of Mentha spicata and Eucalyptus camaldulensis essential oils on dental biofilm. Int. J. Dent. Hyg. 2009, 7, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, N.S.; Owlia, P.; Moein, N.L.; Rasooli, I.; Saderi, H.; Salari, M.H. Effects of sub-inhibitory concentrations of essential oils of Mentha spicata and Cuminum cyminum on virulence factors of Pseudomonas aeruginosa. J. Med. Plants 2010, 9, 124–130. [Google Scholar]
- Karthikeyan, A.; Rameshkumar, R.; Sivakumar, N.; Al Amri, I.S.; Karutha Pandian, S.; Ramesh, M. Antibiofilm activity of Dendrophthoe falcata against different bacterial pathogens. Planta Med. 2012, 78, 1918–1926. [Google Scholar] [CrossRef] [PubMed]
- Council of Europe. European Pharmacopoeia, 5th ed.; Council of Europe: Strasbourg Cedex, France, 2004; Volume I, pp. 217–218. [Google Scholar]
- Stenhagen, E.; Abrahamson, S.; Mc Lafferty, F.W. Registry of Mass Spectral Data; Wiley: New York, NY, USA, 1976. [Google Scholar]
- Massada, Y. Analysis of Essential Oils by Gas Chromatography and Mass Spectrometry; Wiley: New York, NY, USA, 1976. [Google Scholar]
- Jennings, W.; Shibamoto, T. Qualitative Analysis of Flavour and Fragrance Volatiles by Glass Capillary Chromatography; Academic Press: New York, NY, USA, 1980. [Google Scholar]
- Swigar, A.A.; Silverstein, R.M. Monoterpenes; Aldrich Chem Comp: Milwaukee, WI, USA, 1980. [Google Scholar]
- Davies, N.W. Gas chromatographic retention indices of monoterpenes and sesquiterpenes on methyl silicon and carbowax 20M phases. J. Chromatogr. 1990, 503, 1–24. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography-Mass Spectroscopy; Allured Co.: Carol Stream, IL, USA, 1995. [Google Scholar]
- Van den Dool, H.; Kratz, P.D. A generalization of the retention index system including linear temperature programmed gas-liquid partition chromatography. J. Chromatogr. A 1963, 11, 463–471. [Google Scholar] [CrossRef]
- Ksouri, R.; Megdiche, W.; Falleh, H.; Trabelsi, N.; Boulaaba, M.; Abdelly, C. Influence of biological, environmental and technical factors on phenolic content and antioxidant activities of Tunisian halophytes. Comptes Rendus Biol. 2008, 331, 865–873. [Google Scholar] [CrossRef] [PubMed]
- Ottaviani, D.; Santarelli, S.; Bacchiocchi, S.; Masini, L.; Ghittino, C.; Bacchiocchi, I. Presence of pathogenic Vibrio parahaemolyticus strains in mussels from the Adriatic Sea, Italy. Food Microbiol. 2005, 22, 585–590. [Google Scholar] [CrossRef]
- Gulluce, M.; Sahin, F.; Sokmen, M.; Ozer, H.; Daferera, D.; Sokmen, A.; Polissiou, M.; Adiguzel, A.; Ozkan, H. Antimicrobial and antioxidant properties of the essential oils and methanol extract from Mentha longifolia L ssp. longifolia. Food Chem. 2007, 103, 1449–1456. [Google Scholar] [CrossRef]
- Chaieb, K.; Kouidhi, B.; Jrah, H.; Mahdouani, K.; Bakhrouf, A. Antibacterial activity of Thymoquinone, an active principle of Nigella sativa and its potency to prevent bacterial biofilm formation. BMC Complment. Altern. Med. 2011, 11. [Google Scholar] [CrossRef] [PubMed]
- Nostro, A.; Sudano Roccaro, A.; Bisignano, G.; Marino, A.; Cannatelli, M.A.; Pizzimenti, F.C.; Cioni, P.L.; Procopio, F.; Blanco, A.R. Effects of oregano, carvacrol and thymol on Staphylococcus aureus and Staphylococcus epidermidis biofilms. J. Med. Microbiol. 2007, 56, 519–523. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Sample of the essential oil are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Snoussi, M.; Noumi, E.; Trabelsi, N.; Flamini, G.; Papetti, A.; De Feo, V. Mentha spicata Essential Oil: Chemical Composition, Antioxidant and Antibacterial Activities against Planktonic and Biofilm Cultures of Vibrio spp. Strains. Molecules 2015, 20, 14402-14424. https://doi.org/10.3390/molecules200814402
Snoussi M, Noumi E, Trabelsi N, Flamini G, Papetti A, De Feo V. Mentha spicata Essential Oil: Chemical Composition, Antioxidant and Antibacterial Activities against Planktonic and Biofilm Cultures of Vibrio spp. Strains. Molecules. 2015; 20(8):14402-14424. https://doi.org/10.3390/molecules200814402
Chicago/Turabian StyleSnoussi, Mejdi, Emira Noumi, Najla Trabelsi, Guido Flamini, Adele Papetti, and Vincenzo De Feo. 2015. "Mentha spicata Essential Oil: Chemical Composition, Antioxidant and Antibacterial Activities against Planktonic and Biofilm Cultures of Vibrio spp. Strains" Molecules 20, no. 8: 14402-14424. https://doi.org/10.3390/molecules200814402
APA StyleSnoussi, M., Noumi, E., Trabelsi, N., Flamini, G., Papetti, A., & De Feo, V. (2015). Mentha spicata Essential Oil: Chemical Composition, Antioxidant and Antibacterial Activities against Planktonic and Biofilm Cultures of Vibrio spp. Strains. Molecules, 20(8), 14402-14424. https://doi.org/10.3390/molecules200814402










