Antiproliferative and Structure Activity Relationships of Amaryllidaceae Alkaloids
Abstract
:1. Introduction
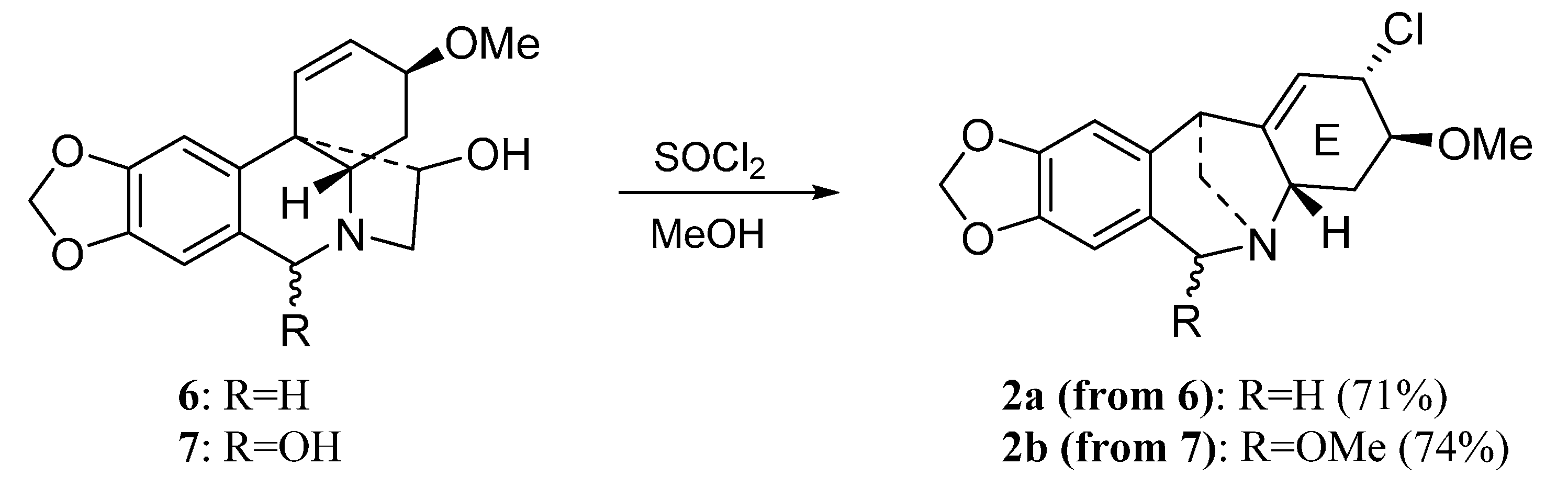
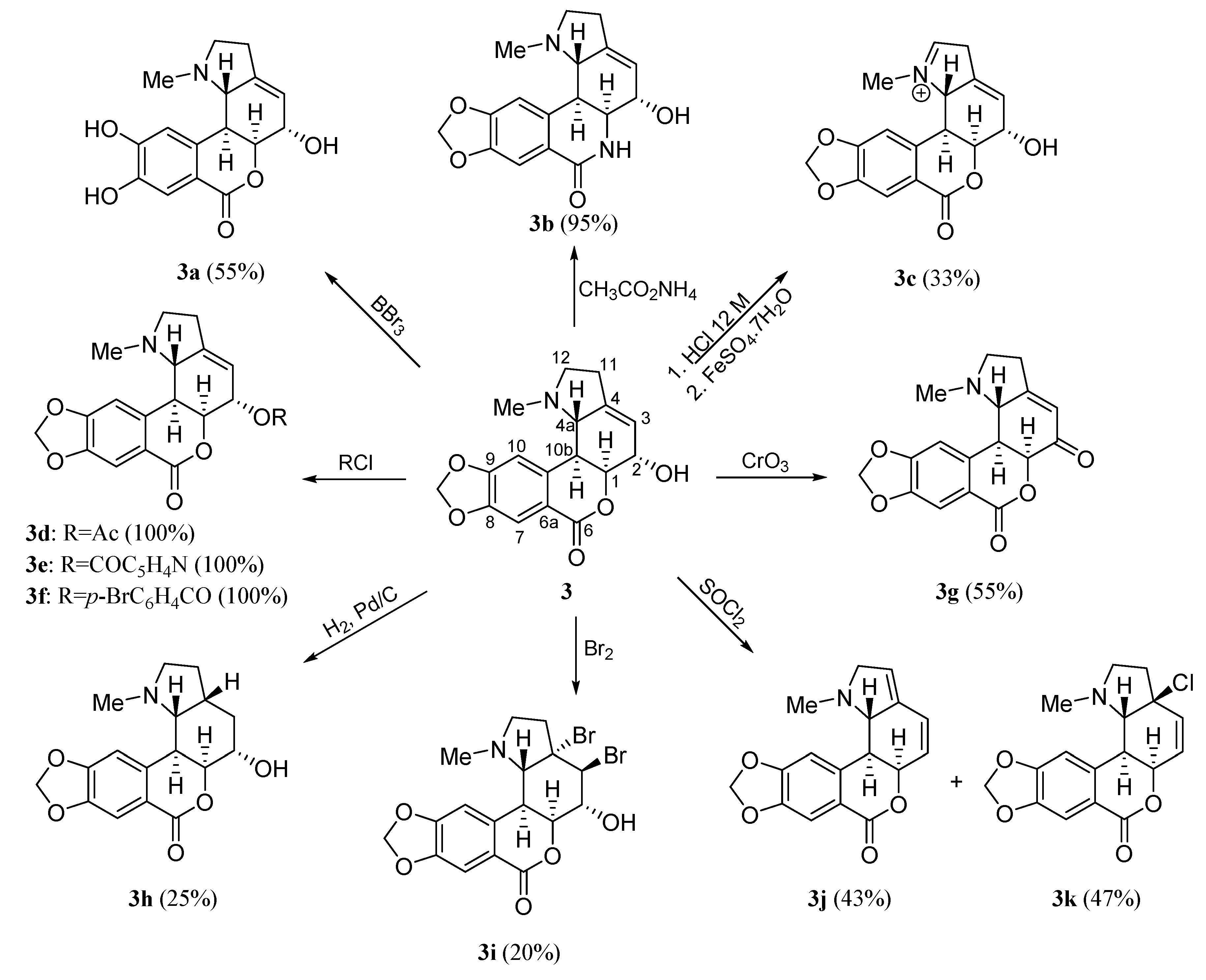
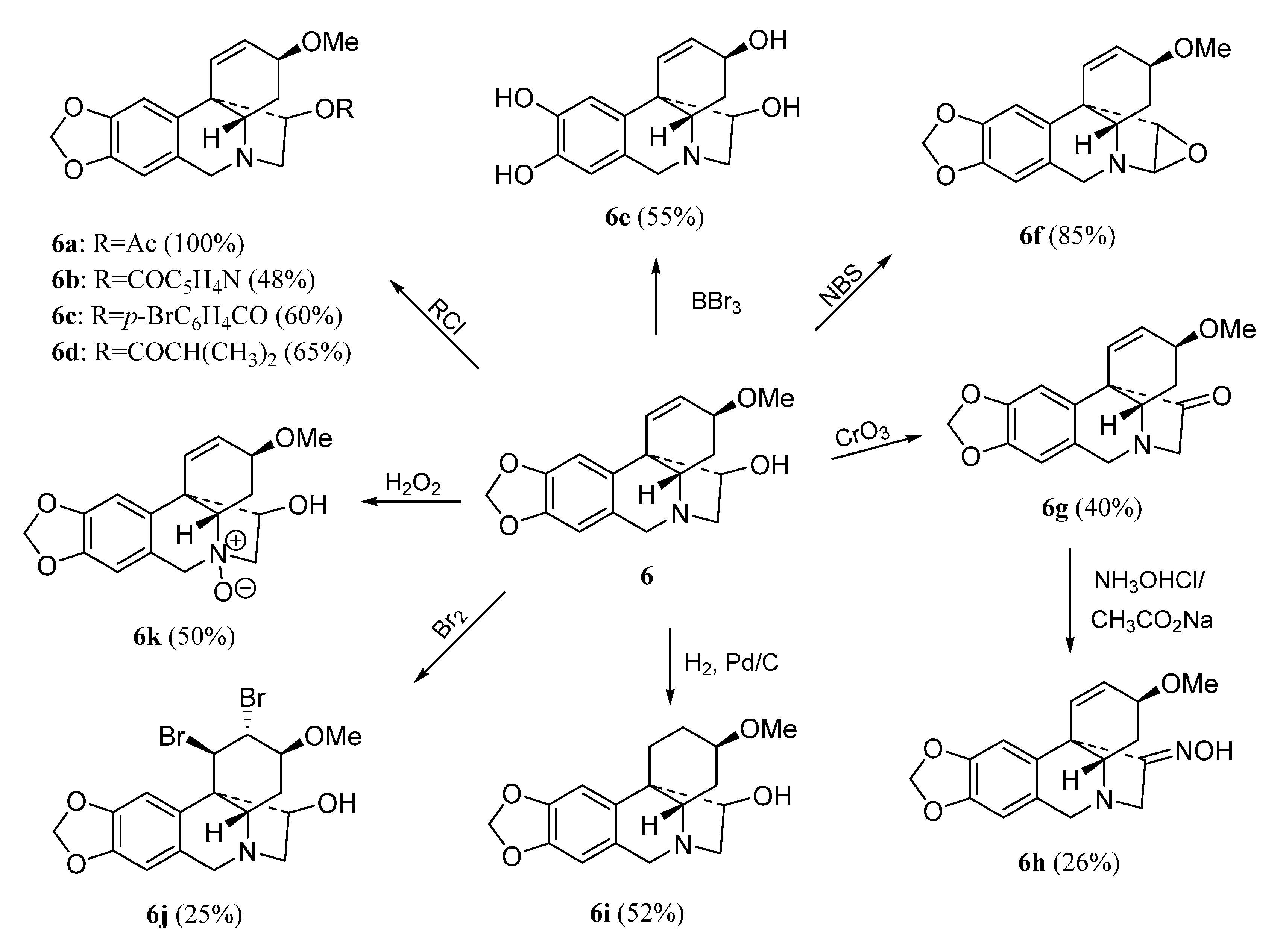
2. Results and Discussion


| Compound | A2780 | SW1573 | T47-D | WiDr |
|---|---|---|---|---|
| 1 | ≥100 | ≥100 | ≥100 | ≥100 |
| 2 | 8.3 ± 0.5 | 4.3 ± 0.7 | 6.5 ± 2.0 | 9.1 ± 1.0 |
| 2a | 3.4 ± 1.0 | 3.9 ± 0.7 | 8.8 ± 1.0 | 7.5 ± 2.0 |
| 2b | 75.2 ± 25.0 | ≥100 | ≥100 | ≥100 |
| 3 | 16.5 ± 10.0 | 12.5 ± 7.0 | 52.9 ± 14.0 | 39.1 ± 20.0 |
| 3a | 41.1 ± 3.0 | 90.3 ± 11.0 | ≥ 100 | ≥100 |
| 3b | 16.8 ± 7.0 | 17.3 ± 9.0 | 26.7 ± 10.0 | 29.1 ± 11.0 |
| 3c | ≥100 | ≥100 | ≥100 | ≥100 |
| 3d | 58.7 ± 7.0 | 91.5 ± 10.0 | ≥100 | ≥100 |
| 3e | ≥100 | ≥100 | ≥100 | ≥100 |
| 3f | ≥100 | ≥100 | ≥100 | ≥100 |
| 3g | 14.6 ± 8.0 | 25.0 ± 6.0 | ≥100 | ≥100 |
| 3h | ≥100 | ≥100 | ≥100 | ≥100 |
| 3i | 67.2 ± 13.0 | ≥100 | ≥100 | ≥100 |
| 3j | ≥100 | ≥100 | ≥100 | ≥100 |
| 3k | 54.9 ± 24.0 | ≥100 | ≥100 | ≥100 |
| 4 | ≥100 | ≥100 | ≥100 | ≥100 |
| 5 | 21.0 ± 2.0 | 16.9 ± 4.0 | 12.5 ± 9.0 | 21.1 ± 6.0 |
| 5a | ≥100 | ≥100 | ≥100 | ≥100 |
| 5b | 35.9 ± 5.0 | 34.8 ± 4.0 | 51.1 ± 3.0 | 53.8 ± 3.0 |
| 5c | 100 | 100 | 100 | 100 |
| 6 | 0.68 ± 0.2 | 2.1 ± 2.0 | 0.87 ± 0.4 | 1.2 ± 0.5 |
| 6a | ≥100 | ≥100 | ≥100 | ≥100 |
| 6b | 27.2 ± 10.0 | 29.5 ± 12.0 | 72.3 ± 24.0 | 63.3 ± 35.0 |
| 6c | 19.1 ± 1.0 | 21.9 ± 4.0 | 46.1 ± 30.0 | 32.8 ± 22.0 |
| 6d | ≥100 | ≥100 | ≥100 | ≥100 |
| 6e | ≥100 | ≥100 | ≥100 | ≥100 |
| 6f | ≥100 | ≥100 | ≥100 | ≥100 |
| 6g | 1.5 ± 0.1 | 2.7 ± 0.1 | 4.4 ± 1.5 | 3.5 ± 2.0 |
| 6h | 33.2 ± 2.0 | 39.1 ± 20.0 | 78.6 ± 24.0 | 67.4 ± 34.0 |
| 6i | 31.4 ± 7.0 | 29.8 ± 3.0 | ≥100 | 58.9 ± 12.0 |
| 6j | ≥100 | ≥100 | ≥100 | ≥100 |
| 6k | 27.2 ± 5.0 | 22.0 ± 20.0 | ≥100 | ≥100 |
| 7 | 1.5 ± 0.1 | 2.0 ± 1.0 | 1.8 ± 1.0 | 2.7 ± 2.0 |
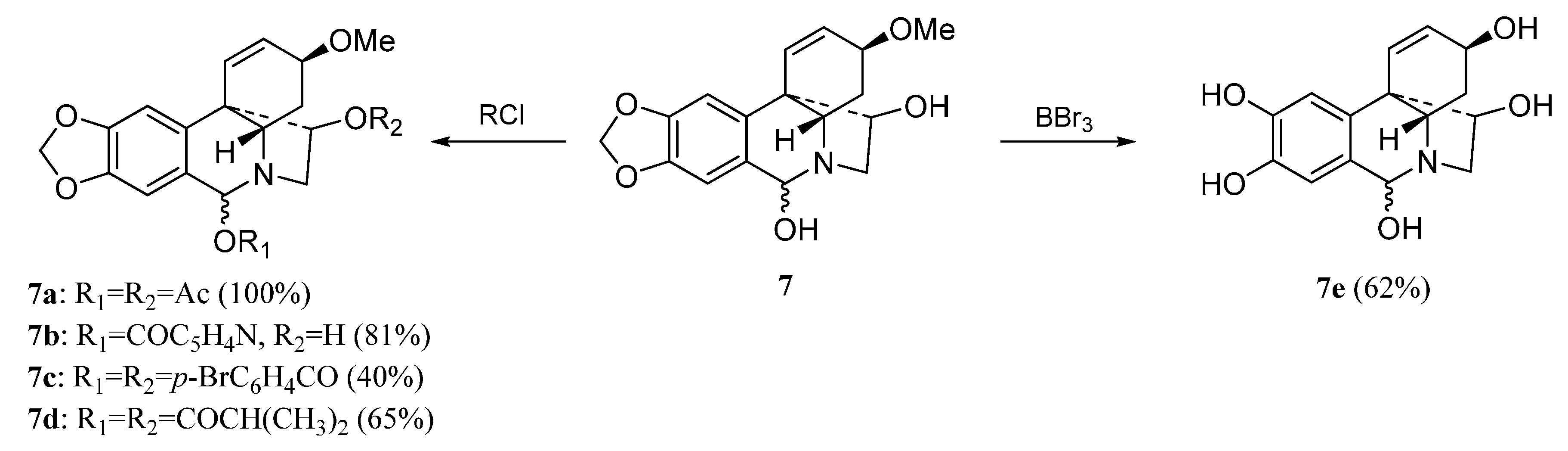
| 7a | ≥100 | ≥100 | ≥100 | ≥100 |
| 7b | 6.9 ± 1.0 | 4.5 ± 0.7 | 8.2 ± 1.2 | 10.1 ± 0.5 |
| 7c | ≥100 | ≥100 | ≥100 | ≥100 |
| 7d | ≥100 | ≥100 | ≥100 | ≥100 |
| 7e | ≥100 | ≥100 | ≥100 | ≥100 |
| Compound | MW | LogP | H-Bond Donors | H-Bond Acceptors | Rotable Bonds | TPSA |
|---|---|---|---|---|---|---|
| 1 | 331 | 1.53 | 1 | 6 | 1 | 60.40 |
| 2 | 287 | 0.54 | 2 | 5 | 1 | 62.16 |
| 2a | 319 | 2.39 | 0 | 4 | 1 | 30.94 |
| 2b | 349 | 2.55 | 0 | 5 | 2 | 40.17 |
| 3 | 315 | 1.23 | 1 | 6 | 0 | 68.24 |
| 3a | 303 | 0.37 | 3 | 6 | 0 | 90.23 |
| 3b | 314 | 0.59 | 2 | 6 | 0 | 71.03 |
| 3c | 314 | −2.91 | 1 | 6 | 0 | 68.01 |
| 3d | 357 | 1.93 | 0 | 7 | 2 | 74.32 |
| 3e | 420 | 2.37 | 0 | 8 | 3 | 87.21 |
| 3f | 498 | 4.47 | 0 | 7 | 3 | 74.32 |
| 3g | 313 | 1.04 | 0 | 6 | 0 | 65.08 |
| 3h | 317 | 1.41 | 1 | 6 | 0 | 68.24 |
| 3i | 475 | 2.29 | 1 | 6 | 0 | 68.24 |
| 3j | 297 | 2.12 | 0 | 5 | 0 | 48.01 |
| 3k | 333 | 2.60 | 0 | 5 | 0 | 48.01 |
| 4 | 271 | 1.59 | 1 | 4 | 0 | 41.93 |
| 5 | 287 | 0.67 | 2 | 5 | 0 | 62.16 |
| 5a | 371 | 2.08 | 0 | 7 | 4 | 74.32 |
| 5b | 497 | 2.38 | 0 | 9 | 6 | 100.10 |
| 5c | 653 | 7.15 | 0 | 7 | 6 | 74.32 |
| 6 | 301 | 1.29 | 1 | 5 | 1 | 51.17 |
| 6a | 343 | 1.99 | 0 | 6 | 3 | 57.25 |
| 6b | 406 | 2.14 | 0 | 7 | 4 | 70.14 |
| 6c | 484 | 4.52 | 0 | 6 | 4 | 57.25 |
| 6d | 383 | 3.46 | 0 | 6 | 4 | 57.25 |
| 6e | 275 | −0.19 | 4 | 5 | 0 | 84.15 |
| 6f | 299 | 1.74 | 0 | 5 | 1 | 43.47 |
| 6g | 299 | 1.10 | 0 | 5 | 1 | 48.01 |
| 6h | 314 | 1.55 | 1 | 6 | 1 | 63.53 |
| 6i | 301 | 1.29 | 1 | 5 | 1 | 51.17 |
| 6j | 461 | 2.22 | 1 | 5 | 1 | 51.17 |
| 6k | 317 | 1.25 | 1 | 6 | 1 | 65.00 |
| 7 | 317 | 0.83 | 2 | 6 | 1 | 71.40 |
| 7a | 401 | 2.23 | 0 | 8 | 5 | 83.55 |
| 7b | 422 | 1.68 | 1 | 8 | 4 | 90.37 |
| 7c | 683 | 7.30 | 0 | 8 | 7 | 83.55 |
| 7d | 481 | 5.17 | 0 | 8 | 7 | 83.55 |
| 7e | 291 | −0.65 | 5 | 6 | 0 | 104.38 |
3. Experimental Section
3.1. Natural and Semisynthetic Amaryllidaceae Alkaloids
3.2. Antiproliferative Assay
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Siegel, R.; Ma, J.; Zou, Z.; Jemal, A. Cancer Statistics, 2014. CA Cancer J. Clin. 2014, 64, 9–29. [Google Scholar] [CrossRef] [PubMed]
- Cragg, G.M.; Newman, D.J. Plants as source of anticancer agents. J. Ethnopharmacol. 2005, 100, 72–79. [Google Scholar] [CrossRef] [PubMed]
- McNulty, J.; Nair, J.; Little, J.; Brennan, D.; Bastida, J. Structure-activity studies on acetylcholinesterase inhibition in the lycorine series of Amaryllidaceae alkaloids. Bioorg. Med. Chem. Lett. 2010, 20, 5290–5294. [Google Scholar] [CrossRef] [PubMed]
- Citoglu, G.; Acikara, O.; Yilmaz, B.; Özbek, H. Evaluation of analgesic, anti-inflammatory and hepatoprotective effects of lycorine from Sternbergia fisheriana (Herbert) Rupr. Fitoterapia 2012, 83, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Evidente, A.; Andolfi, A.; Abou-Donia, A.H.; Touema, S.M.; Hammoda, H.M.; Shawky, E.; Motta, A. (−)-Amarbellisine, a lycorine-type alkaloid from Amaryllis belladonna L. growing in Egypt. Phytochemistry 2004, 65, 2113–2118. [Google Scholar] [CrossRef] [PubMed]
- Cedrón, J.; Gutiérrez, L.; Flores, N.; Ravelo, A.; Estévez-Braun, A. Synthesis and antimalarial activity of new haemanthamine-type derivatives. Bioorg. Med. Chem. 2012, 20, 5464–5472. [Google Scholar] [CrossRef] [PubMed]
- Cedrón, J.; Gutiérrez, L.; Flores, N.; Ravelo, A.; Estévez-Braun, A. Preparation and antimalarial activity of semisynthetic lycorenine derivative. Eur. J. Med. Chem. 2013, 63, 722–730. [Google Scholar] [CrossRef] [PubMed]
- Cedrón, J.; Gutiérrez, L.; Flores, N.; Ravelo, A.; Estévez-Braun, A. Synthesis and antiplasmodial activity of lycorine derivatives. Bioorg. Med. Chem. 2010, 18, 4694–4701. [Google Scholar] [CrossRef] [PubMed]
- Pettit, G.R.; Gaddamidi, V.; Herald, D.L.; Singh, S.B.; Cragg, G.M.; Schmidt, J.M.; Boettner, F.E.; Williams, M.; Sagawa, Y. Antineoplastic Agents, 120. Pancratium littorale. J. Nat. Prod. 1986, 49, 995–1002. [Google Scholar] [CrossRef] [PubMed]
- Kornienko, A.; Evidente, A. Chemistry, Biology, and Medicinal Potential of Narciclasine and its Congeners. Chem. Rev. 2008, 108, 1982–2014. [Google Scholar] [CrossRef] [PubMed]
- Cedrón, J.; Del Arco-Aguilar, M.; Estévez-Braun, A.; Ravelo, A. Chemistry and biology of Pancratium alkaloids. Alkaloids Chem. Biol. 2010, 68, 1–37. [Google Scholar] [PubMed]
- Wang, P.; Yuan, H.; Zhang, X.; Li, Y.; Shang, L.; Yin, Z. Novel Lycorine Derivatives as Anticancer Agents: Synthesis and in Vitro Biological Evaluation. Molecules 2014, 19, 2469–2480. [Google Scholar] [CrossRef] [PubMed]
- Ingrassia, L.; Lefranc, F.; Dewelle, J.; Pottier, L.; Mathieu, V.; Spiegl-Kreinecker, S.; Sauvage, S.; El Yazidi, M.; Dehoux, M.; Berger, W.; et al. Structure-Activity Relationship Analysis of Novel Derivatives of Narciclasine (an Amaryllidaceae Isocarbostyril Derivative) as Potential Anticancer Agent. J. Med. Chem. 2009, 52, 1100–1114. [Google Scholar] [CrossRef] [PubMed]
- Evidente, A.; Kireev, A.; Jenkins, A.; Romero, A.; Steelant, W.; Van Slambrouck, S.; Kornienko, A. Biological Evaluation of Structurally Diverse Amaryllidaceae Alkaloids and their Synthetic Derivatives: Discovery of Novel Leads for Anticancer Drug Design. Planta Med. 2009, 75, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Cedrón, J.; Oberti, J.; Estévez-Braun, A.; Ravelo, A.; del Arco-Aguilar, M.; López, M. Pancratium canariense as an important source of Amaryllidaceae alkaloids. J. Nat. Prod. 2009, 72, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Van Goietsenoven, G.; Andolfi, A.; Lallemand, B.; Cimmino, A.; Lamoral-Theys, D.; Gras, T.; Abou-Donia, A.; Dubois, J.; Lefranc, F.; Mathieu, V.; et al. Amaryllidaceae alkaloids belonging to different structural subgroups display activity against apoptosis-resistant cancer cells. J. Nat. Prod. 2010, 73, 1223–1227. [Google Scholar] [CrossRef] [PubMed]
- Cedrón, J.; Estévez-Braun, A.; Ravelo, A.; Gutiérrez, D.; Flores, N.; Bucio, M.; Pérez-Hernández, N.; Joseph-Nathan, P. Bioactive montanine derivatives from halide-induced rearrangements of haemanthamine-type alkaloids. Absolute configuration by VCD. Org. Lett. 2009, 11, 1491–1494. [Google Scholar] [CrossRef] [PubMed]
- Miranda, P.; Padrón, J.; Padrón, J.; Villar, J.; Martín, V. Synthesis and SAR Study of Cytotoxic Alkyl Chloro Dihydropyrans. Chem. Med. Chem. 2006, 1, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not available.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cedrón, J.C.; Ravelo, Á.G.; León, L.G.; Padrón, J.M.; Estévez-Braun, A. Antiproliferative and Structure Activity Relationships of Amaryllidaceae Alkaloids. Molecules 2015, 20, 13854-13863. https://doi.org/10.3390/molecules200813854
Cedrón JC, Ravelo ÁG, León LG, Padrón JM, Estévez-Braun A. Antiproliferative and Structure Activity Relationships of Amaryllidaceae Alkaloids. Molecules. 2015; 20(8):13854-13863. https://doi.org/10.3390/molecules200813854
Chicago/Turabian StyleCedrón, Juan C., Ángel G. Ravelo, Leticia G. León, José M. Padrón, and Ana Estévez-Braun. 2015. "Antiproliferative and Structure Activity Relationships of Amaryllidaceae Alkaloids" Molecules 20, no. 8: 13854-13863. https://doi.org/10.3390/molecules200813854
APA StyleCedrón, J. C., Ravelo, Á. G., León, L. G., Padrón, J. M., & Estévez-Braun, A. (2015). Antiproliferative and Structure Activity Relationships of Amaryllidaceae Alkaloids. Molecules, 20(8), 13854-13863. https://doi.org/10.3390/molecules200813854