2.1. Statistical Analysis and the Model Fitting
Response surface optimization is more advantageous than the traditional single parameter optimization in that it saves time and raw material [
23,
24]. There were a total of 20 runs for optimizing the three individual parameters in the current CCD.
Three independent parameters in the study were namely extraction time, extraction pressure and extraction temperature as response (dependent variable). The ranges and the levels of the independent variables are given in
Table 1. The extraction yields of
D. sophia seed oil obtained under the twenty different testing conditions are shown in
Table 2.
Table 1.
Experimental range and levels of independent test variables.
Table 1.
Experimental range and levels of independent test variables.
| Variables | Ranges and Levels |
|---|
| −1.68 | −1 | 0 | 1 | 1.68 |
|---|
| A (Pressure, MPa) | 4.89 | 10 | 17.50 | 25 | 30 |
| B (Temperature, °C) | 43.18 | 50 | 60 | 70 | 76.82 |
| C (Time, min) | 2.50 | 40 | 95 | 150 | 187.50 |
Table 2 shows the experimental conditions and the results of extraction yield of
D. sophia seed oil according to the factorial design. By applying multiple regression analysis on the experimental data, the response variable and the test variables were related by the following second-order polynomial equation:
Extraction yield was the yield of
D. sophia seed oil (%). A, B and C were the actual values of the variables for extraction pressure (MPa), temperature (°C) and time (min), respectively. The results of the analysis of variance, goodness-of-fit and the adequacy of the models were summarized in
Table 3. The determination coefficient (R
2 = 0.9836) was showed by ANOVA of the quadratic regression model, indicating that only 1.64% of the total variations were not explained by the model. The value of the adjusted determination coefficient (Adjusted R
2 = 0.9689) also confirmed that the model was highly significant. At the same time, a very low value 2.52 of coefficient of the variation (CV) clearly indicated a very high degree of precision and a good deal of reliability of the experimental values. The model was found to be adequate for prediction within the range of experimental variables. The
p-values were used as a tool to check the significance of each coefficient, which in turn may indicate the pattern of the interactions between the variables. The smaller was the value of P, the more significant was the corresponding coefficient [
25,
26]. It can be seen from this table that the linear coefficients (A, B, C), a quadratic term coefficient (A
2, C
2) and cross product coefficients (AB) were significant, with very small
p-values (
p < 0.05). The other term coefficients (AC, BC, B
2) were not significant (
p > 0.05).
The “Lack of Fit F-value” of 1.46 and the probability (p) value of 0.3443 (p > 0.05) imply that the Lack of Fit is not significant relative to the pure error, which measured the fitness of models, so the results indicated that the model was accurate for predicting response variations. In brief, the responses were explained well by the regression equation, and this allowed it to establish response surfaces and it was feasible to use the regression models to predict the yields of D. sophia seed oil. The full model developed from Equation (1) was used to make three-dimensional and contour plots to predict the relationships between the independent variables and the dependent variables.
Table 2.
Experimental program and results for SFE–CO2 of D. sophia seed oil.
Table 2.
Experimental program and results for SFE–CO2 of D. sophia seed oil.
| No | A Pressure | B Temperature | C Time | Pressure (MPa) | Temperature (°C) | Time (min) | Extraction Yield (%) |
|---|
| 1 | 0 | 0 | 0 | 17.5 | 60 | 95 | 26.48 |
| 2 | 1.68 | 0 | 0 | 30 | 60 | 95 | 27.32 |
| 3 | 1 | –1 | –1 | 25 | 50 | 40 | 25.30 |
| 4 | 0 | 0 | 0 | 18 | 60 | 95 | 27.01 |
| 5 | 1 | 1 | –1 | 25 | 70 | 40 | 25.69 |
| 6 | 0 | 0 | 0 | 18 | 60 | 95 | 26.49 |
| 7 | 0 | 0 | 0 | 18 | 60 | 95 | 27.25 |
| 8 | –1 | 1 | –1 | 10 | 70 | 40 | 22.25 |
| 9 | 0 | 1.68 | 0 | 18 | 77 | 95 | 28.78 |
| 10 | 0 | 0 | 0 | 18 | 60 | 95 | 28.01 |
| 11 | 0 | 0 | –1.68 | 18 | 60 | 3 | 21.15 |
| 12 | –1 | –1 | 1 | 10 | 50 | 150 | 21.40 |
| 13 | 0 | 0 | 0 | 18 | 60 | 95 | 27.40 |
| 14 | –1 | –1 | –1 | 10 | 50 | 40 | 18.15 |
| 15 | 1 | 1 | 1 | 25 | 70 | 150 | 29.17 |
| 16 | –1.68 | 0 | 0 | 5 | 60 | 95 | 18.14 |
| 17 | –1 | 1 | 1 | 10 | 70 | 150 | 26.26 |
| 18 | 1 | –1 | 1 | 25 | 50 | 150 | 32.10 |
| 19 | 0 | –1.68 | 0 | 18 | 43 | 95 | 26.13 |
| 20 | 0 | 0 | 1.68 | 18 | 60 | 187 | 29.13 |
Table 3.
D. sophia seed oil extraction yield the regression equation coefficient and significant testing.
Table 3.
D. sophia seed oil extraction yield the regression equation coefficient and significant testing.
| Source | Sum of Squares | df | Mean Square | F Value | p-Value Prob > F | |
|---|
| Model | 252.16 | 9 | 28.02 | 66.83 | <0.0001 | significant |
| A-Pressure | 115.05 | 1 | 115.05 | 274.42 | <0.0001 | |
| B-Temperature | 8.66 | 1 | 8.66 | 20.66 | 0.0011 | |
| C-Time | 70.19 | 1 | 70.19 | 167.41 | <0.0001 | |
| AB | 16.53 | 1 | 16.53 | 39.43 | <0.0001 | |
| AC | 1.14 | 1 | 1.14 | 2.72 | 0.1302 | |
| BC | 0.82 | 1 | 0.82 | 1.95 | 0.1924 | |
| A2 | 34.01 | 1 | 34.01 | 81.12 | <0.0001 | |
| B2 | 0.26 | 1 | 0.26 | 0.62 | 0.4493 | |
| C2 | 6.75 | 1 | 6.75 | 16.09 | 0.0025 | |
| Residual | 4.19 | 10 | 0.42 | | | |
| Lack of Fit | 2.49 | 5 | 0.50 | 1.46 | 0.3443 | not significant |
| Pure Error | 1.70 | 5 | 0.34 | | | |
| Cor Total | 256.36 | 19 | | | | |
2.2. Optimization of SFE Experimental Conditions
The graphical representations of the regression Equation (1), called the response surfaces and the contour plots, were obtained using Design-export 8.0, and the results of extraction yield of
D. sophia seed oil affected by extraction pressure, extraction temperature and extraction time are presented in
Figure 1 and
Figure 2. Response surface methodology plays a key role in identifying the optimum values of the independent variables efficiently, under which dependent variable could arrive the maximum response [
16]. In the response surface plot and contour plot, the extraction yield of
D. sophia seed oil was obtained along with two continuous variables, while the other variable was fixed constant at its zero level (center value of the testing ranges). In the two figures, the maximum predicted value indicated by the surface was confined in the smallest ellipse in the contour diagram. Elliptical contours are obtained when there is a perfect interaction between the independent variables. The independent variables and maximum predicted values from the figures corresponded with the optimum values of the dependent variables (responses) obtained by the equations [
27,
28,
29].
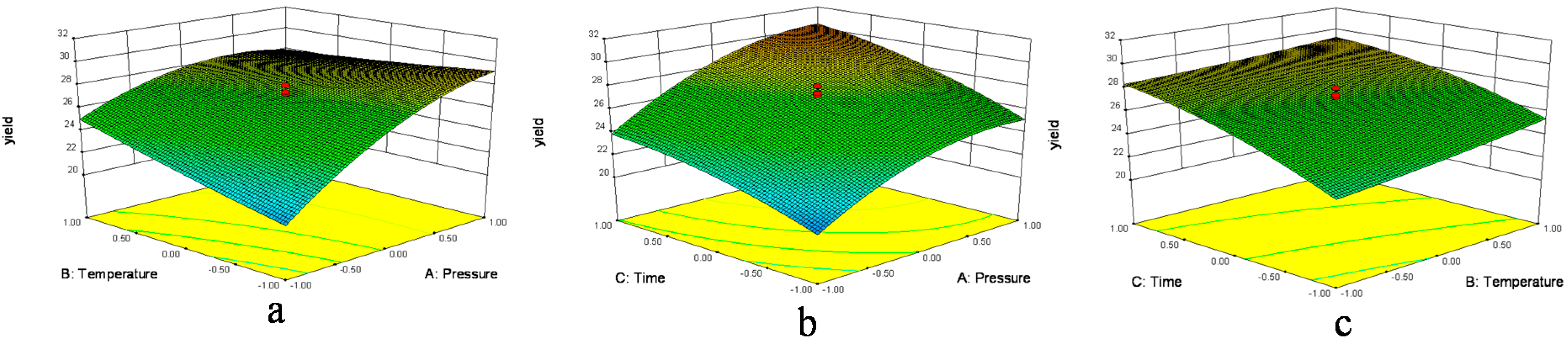
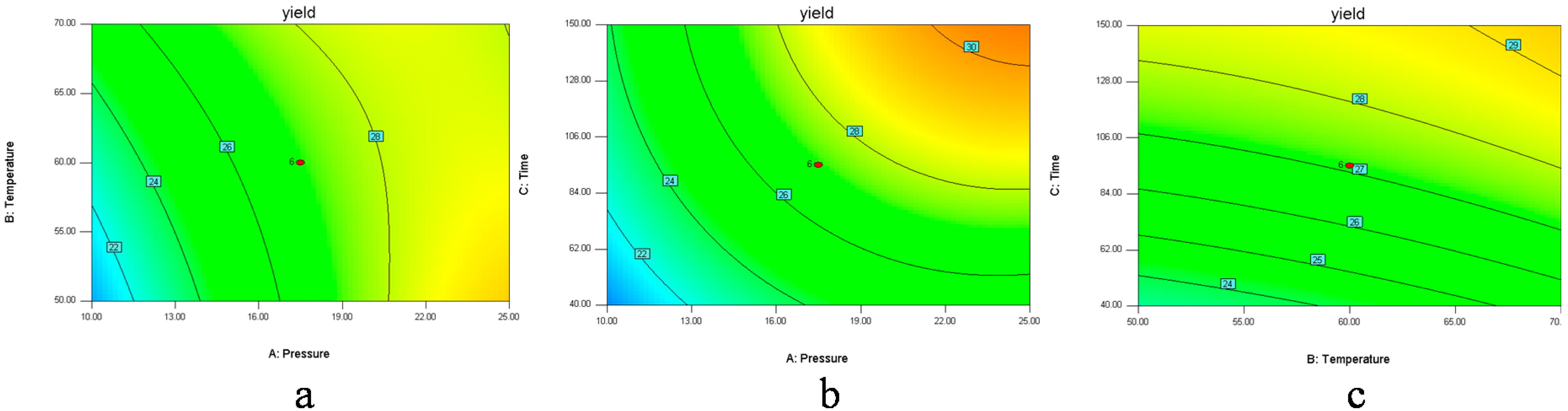
In
Figure 1a and
Figure 2a, when the 3D response surface plot and the contour plot were developed for the extraction yield of
D. sophia seed oil with varying extraction temperature and extraction pressure at fixed extraction time, the extraction yield of
D. sophia seed oil increased with the increasing extraction temperature, and increased rapidly with increase of extraction pressure from 10 to 17.5 MPa, then gradually decreased from 17.5 to 25 MPa. The
Figure 1b and
Figure 2b showed the 3D response surface plot and the contour plot at varying extraction pressure and extraction time at fixed extraction temperature. As in the case of the extraction yield of
D. sophia seed oil, extraction pressure had a positive impact on the extraction yield of
D. sophia seed oil. There was an increase in the extraction yield of
D. sophia seed oil with increase in the extraction time. However, the extraction yield of
D. sophia seed oil was found to increase rapidly with increase of extraction time from 40 to 95 min, but beyond 95 min, the yield increased slowly with increasing extraction. The results are in accord with the data in
Table 3, which showed that the interactive effect of extraction pressure with extraction time on the yield of
D. sophia seed oil was not very weak (
p = 0.1302). The 3D response surface plot and the contour plot based on independent extraction temperature and extraction time are shown in
Figure 1c and
Figure 2c, while the other independent variable, extraction pressure at a zero level. An increase in the extraction yield of
D. sophia seed oil could be evidently achieved with the increases of extraction temperature or extraction time. It was obvious that the extraction yield of
D. sophia seed oil was almost proportional to extraction temperature and extraction time in certain range of variable.
Figure 1.
Response surface plots (3D) showing the effects of variables for the yield of D. sophia seed oil. (a) Effect of extraction pressure (A) and extraction temperature (B) under extraction time (C) 95 min; (b) effect of A and C at B = 60 °C and (c) effect of C and B at A = 17.5 MPa.
Figure 1.
Response surface plots (3D) showing the effects of variables for the yield of D. sophia seed oil. (a) Effect of extraction pressure (A) and extraction temperature (B) under extraction time (C) 95 min; (b) effect of A and C at B = 60 °C and (c) effect of C and B at A = 17.5 MPa.
Figure 2.
Contour plots (2D) showing the effects of variables (a) extraction pressure (A) and temperature (B); (b) extraction pressure (A) and time (C); (c) extraction temperature (B) and time (C) on the yield of D. sophia seed oil.
Figure 2.
Contour plots (2D) showing the effects of variables (a) extraction pressure (A) and temperature (B); (b) extraction pressure (A) and time (C); (c) extraction temperature (B) and time (C) on the yield of D. sophia seed oil.
Figure 1 and
Figure 2 show the effects of pressure on the yield with each of the other two factors held constant. Pressure has the largest influence on the extraction yield. The extraction yields were increased with an increase in pressure, because raising the extraction pressure leads to a higher fluid density, which increases the solubility of the analytes. The solubility of oil in supercritical fluid directly affects the extraction yield and a balance between the SFE-CO
2 density and the oil vapor pressure controls it. At high pressure, the influence of temperature on the solubility of oil is predominated by the oil vapor pressure effect, and then the solubility of oil increases with the increase of temperature. While at low pressure, SFE-CO
2 density has a pronounced effect on the solubility of oil and the solubility decreases with the increase of temperature [
10,
30]. In
Figure 1 and
Figure 2, the graphs also show the effects of time with each of the two other factors on the yield. The trend was similar to the pressure. It may be that when the time reached a certain level the solubility reached saturation; the influence of the other factor temperature was not as significant as that of the pressure and time.
Optimization of extraction condition was achieved. It could be seen that the best extraction yield was reached at extraction pressure of 25 MPa, extraction temperature of 50 °C and extraction time of 150 min for the extractor and 3 MPa/40 °C for the separators. Among the three extraction parameters studied, extraction pressure was the most significant factor to affect the extraction yield of
D. sophia seed oil, followed by extraction temperature, and extraction time according to the regression coefficients significance of the quadratic polynomial model (
Table 3) and gradient of slope in the 3D response surface plot (
Figure 1).
2.4. Chemical Compositions
GC-MS was used to analyze the chemical composition of
D. sophia seed oil after methyl esterification which was found to have a very complex composition with compound identifications reported in
Table 4. A total of approximately 67 compounds were separated in
D. sophia seed oil, of which 51 compounds, representing 98.21% of the total oils, were positively identified by comparison of their retention indexes and the mass spectra of each GC component with those of standards and with reported data [
31,
32]. From GC analysis, 9, 12-octadecadiynoic acid methyl ester and methyl 10, 13, 16-docosatrienoate were found to represent 21.58% and 19.39% of the extracted oil, respectively. Other compounds identified in a large amount were γ-linolenic acid methyl ester (15.87%), isonicotinic acid, cyclohexyl ester (13.35%), dioctyl succinate (4.12%), 9-octadecynoic acid methyl ester (4.84%) and methyl 11,14-eicosadienoate (4.25%), and the remaining compounds, which number over 50 (15.06%).
There have been no reports regarding chemical composition analysis of
D. sophia seed oil extracted by SFE-CO
2. There have been a paper regarding GC analysis of oil composition of
D. sophia seed oil which reported that 24 components were identified in
D. sophia seed oil extracted exhaustively with benzine (70–80 °C) for extraction to afford a yellowish-brown oil in 22% yield [
19]. Many of the fatty acids and other compounds present in oils have long been known to benefit our health. There is clearly great potential for developing functional oils. According to the results of this study,
D. sophia seed oil has the amount of unsaturated fatty acid in the present study and
D. sophia seed is an inexpensive source of essential fatty acid. For example, γ-linolenic acid (GLA) has been promoted as medication for a variety of ailments including breast pain and eczema [
31]. GLA is also sometimes promoted as an anti-cancer agent. Neither GLA nor other GLA-rich supplements have been convincingly shown to be useful in preventing or treating any other health conditions [
32,
33,
34,
35]. 9-octadecynoic acid (oleic acid) is the most abundant fatty acid in human adipose tissue [
36], and second in abundance in human tissues overall only to palmitic acid [
37]. Oleic acid as its sodium salt is a major component of soap as an emulsifying agent. It is also used as an emollient [
38]. Small amounts of oleic acid are used as an excipient in pharmaceuticals, and it is used as an emulsifying or solubilizing agent in aerosol products [
39]. Oleic acid is also used to induce lung damage in certain types of animals, for the purpose of testing new drugs and other means to treat lung diseases [
40]. Oleic acid is used as a soldering flux in stained glass work for joining lead came [
41]. Therefore,
D. sophia seed oil obtained with SFE method may have potential for use as a kind of specialty oil in various product applications. It is a cost effective technique at the laboratory scale and it seems to be applicable for industrial oil extraction.
Table 4.
Identification and quantification of compounds contained in D. sophia seed oil after derivatisation compared with literature data.
Table 4.
Identification and quantification of compounds contained in D. sophia seed oil after derivatisation compared with literature data.
| No. | Compound | Relative Amount (%) a | Molecular Formula | RI b | RI c |
|---|
| 1 | cis-1,2-Dihydrocatechol | 0.01 | C6H8O2 | 1060 | 1029 |
| 2 | Carvol | 0.01 | C10H14O | 1190 | 1154 |
| 3 | 1-Tridecene | 0.01 | C13H26 | 1292 | 1307 |
| 4 | exo-2-Hydroxycineole acetate | 0.01 | C12H20O3 | 1344 | 1331 |
| 5 | 3-Dodecanone | 0.01 | C12H24O | 1350 | 1331 |
| 6 | Hydrocoumarin | 0.03 | C9H8O2 | - d | 1370 |
| 7 | Eugenol | 0.03 | C10H12O2 | 1362 | 1370 |
| 8 | 2-Dodecenal | 0.01 | C12H22O | 1410 | 1432 |
| 9 | Tetrahydroionol | 0.01 | C13H26O4 | 1432 | 1444 |
| 10 | α-Ionone | 0.01 | C13H20O | 1456 | 1444 |
| 11 | trans-α-Bergamotene | 0.01 | C15H24 | 1430 | 1460 |
| 12 | β-Sesquiphellandrene | 0.01 | C15H24 | 1446 | 1478 |
| 13 | α-Farnesene | 0.01 | C15H24 | 1458 | 1478 |
| 14 | Octylcyclohexane | 0.03 | C14H28 | 1476 | 1485 |
| 15 | β-Bisabolene | 0.01 | C15H24 | 1500 | 1500 |
| 16 | trans-Calamenene | 0.02 | C15H22 | 1537 | 1524 |
| 17 | 2,12-Dimethyltridecan-4-one | 0.01 | C15H30O | 1589 | 1531 |
| 18 | Prenyl salicylate | 0.01 | C12H14O3 | - | 1531 |
| 19 | Vanillic acid | 0.01 | C8H8O4 | 1566 | 1547 |
| 20 | Acetisoeugenol | 0.01 | C12H14O | 1569 | 1599 |
| 21 | 7-Hexadecene | 0.02 | C16H32 | 1620 | 1633 |
| 22 | 1-Tetradecanol | 0.01 | C14H30O | 1656 | 1637 |
| 23 | β-Nootkatol | 0.01 | C15H24O | 1662 | 1670 |
| 24 | Benzoic acid, heptyl ester | 0.03 | C14H20O2 | 1682 | 1695 |
| 25 | 1-Nonylcycloheptane | 0.01 | C16H32 | 1696 | 1700 |
| 26 | Heptadecane | 0.19 | C17H36 | 1711 | 1725 |
| 27 | n-Pentadecanol | 0.01 | C14H30O2 | 1755 | 1739 |
| 28 | (9E)-9-Octadecene | 0.03 | C18H36 | 1818 | 1825 |
| 29 | Thymyl angelate | 0.01 | C15H20O2 | - | 1830 |
| 30 | 1-Hexadecanol | 0.01 | C16H34O | 1854 | 1840 |
| 31 | (Z)-11-Hexadecen-1-ol | 0.01 | C16H32O | - | 1892 |
| 32 | Costunlide | 0.42 | C15H20O2 | 1897 | 1901 |
| 33 | Isonicotinic acid, cyclohexyl ester | 13.35 | C12H15NO2 | - | 1926 |
| 34 | Z-11-Hexadecenoic acid | 0.01 | C16H30O2 | 1976 | 1948 |
| 35 | Sulfurous acid dicyclohexyl ester | 0.02 | C12H22O3S | 1964 | 1961 |
| 36 | Methyl 9,12-heptadecadienoate | 0.02 | C18H32O2 | 1994 | 1998 |
| 37 | 3β,17β-Androstanediol | 0.02 | C19H32O2 | - | 2025 |
| 38 | (Z,Z)-9,12-Octadecadien-1-ol | 0.02 | C18H34O | 2069 | 2060 |
| 39 | γ-Linolenic acid methyl ester | 15.87 | C19H32O2 | 2101 | 2091 |
| 40 | 9,12-Octadecadiynoic acid methyl ester | 21.58 | C19H30O2 | 2112 | 2097 |
| 41 | 9-Octadecynoic acid methyl ester | 4.84 | C19H34O2 | 2095 | 2099 |
| 42 | Methyl 11,14-eicosadienoate | 4.25 | C22H40O2 | 2118 | 2125 |
| 43 | cis-11, 14-Eicosadienoic acid methyl ester | 10.11 | C21H38O2 | 2292 | 2299 |
| 44 | Methyl 7, 11, 14-eicosatrienoate | 1.6 | C21H36O2 | 2300 | 2307 |
| 45 | Dioctyl succinate | 4.12 | C20H38O4 | - | 2335 |
| 46 | Butyl 9,12,15-octadecatrienoate | 0.01 | C17H23NO4 | 2399 | 2386 |
| 47 | Hydrastininic acid | 0.04 | C11H9NO6 | 2342 | 2400 |
| 48 | 2-Chloroethyl linoleate | 0.03 | C20H35ClO2 | 2418 | 2432 |
| 49 | Methyl 10,13,16-docosatrienoate | 19.39 | C23H42O2 | 2499 | 2491 |
| 50 | Pentacosanal | 1.18 | C25H50O | - | 2493 |
| 51 | cis-13,16-Docasadienoic acid | 0.72 | C22H40O2 | 2580 | 2531 |