Anti-Protozoal Activities of Cembrane-Type Diterpenes from Vietnamese Soft Corals
Abstract
:1. Introduction
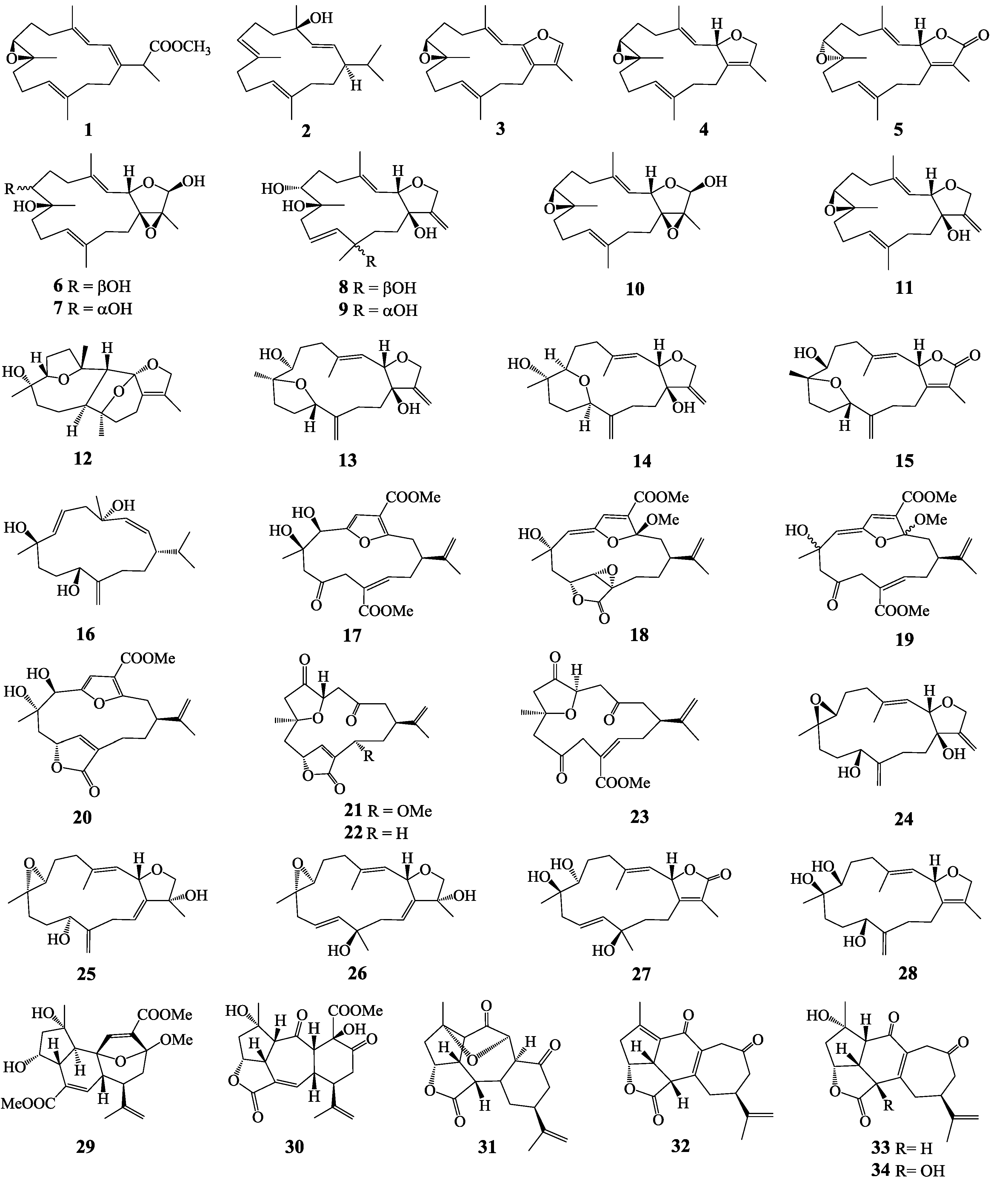
2. Results and Discussion
| Compounds | IC50 Values (μM) a | |||
|---|---|---|---|---|
| T. b. rhodesiense | L. donovani | P. falciparum | Cytotoxicity L6 | |
| 7 S,8S-Epoxy-1,3,11-cembratriene-16-oic methyl ester (1) | 1.00 ± 0.19 | ND | 22.95 ± 0.15 | >54 |
| (1 R,4R,2E,7E,11E)-Cembra-2,7,11-trien-4-ol (2) | 1.14 ± 0.50 | ND | 14.80 ± 0.90 | 49.40 ± 5.20 |
| Lobocrasol A ( 6) | 9.97 ± 0.64 | 0.18 ± 0.02 | ND | 55.95 ± 3.65 |
| Lobocrasol C ( 8) | 9.37 ± 0.74 | 0.17 ± 0.05 | 7.37 ± 0.35 | 40.45 ± 7.75 |
| Laevigatol A ( 10) | >20 | ND | 3.02 ± 0.50 | 28.75 ± 10.35 |
| Crassumol D ( 12) | 0.61 ± 0.01 | ND | ND | 58.70 ± 1.00 |
| Crassumol E ( 13) | 0.72 ± 0.04 | ND | 7.43 ± 0.86 | 43.00 ± 2.60 |
| Crassumol G ( 15) | 12.20 ± 0.10 | ND | 7.57 ± 0.59 | 33.20 ± 13.50 |
| (1 S,2E,4S,6E,8S,11S)-2,6,12(20)-Cembrantriene-4,8,11-triol (16) | 0.65 ± 0.01 | ND | ND | 48.65 ± 3.45 |
| Sinumaximol A ( 17) | >20 | ND | 7.20 ± 0.71 | 45.00 ± 2.90 |
| Sinumaximol C ( 18) | 9.88 ± 1.93 | ND | ND | 28.65 ± 9.95 |
| 13- Epi-scabrolide C (21) | 11.76 ± 4.46 | ND | ND | 47.50 ± 6.00 |
| Melarsoprol b | 0.005 ± 0.00 | NT | NT | 28.90 ± 9.30 |
| Miltefosine b | NT | 0.14 ± 0.05 | NT | 143.10 ± 35.30 |
| Chloroquine b | NT | NT | 0.004 ± 0.00 | 120.30 ± 27.50 |
| Podophyllotoxin b | NT | NT | NT | 0.014 ± 0.002 |
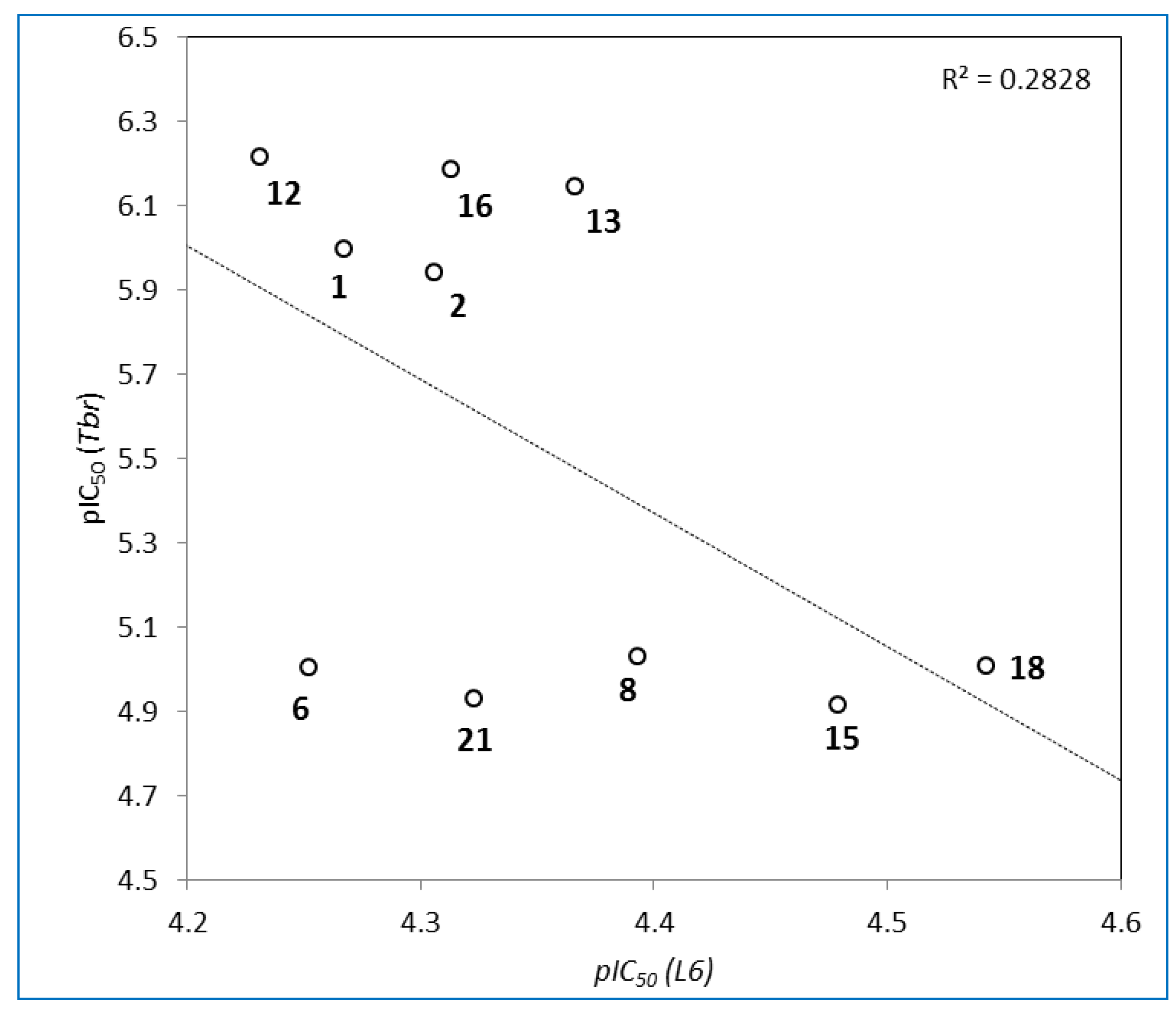
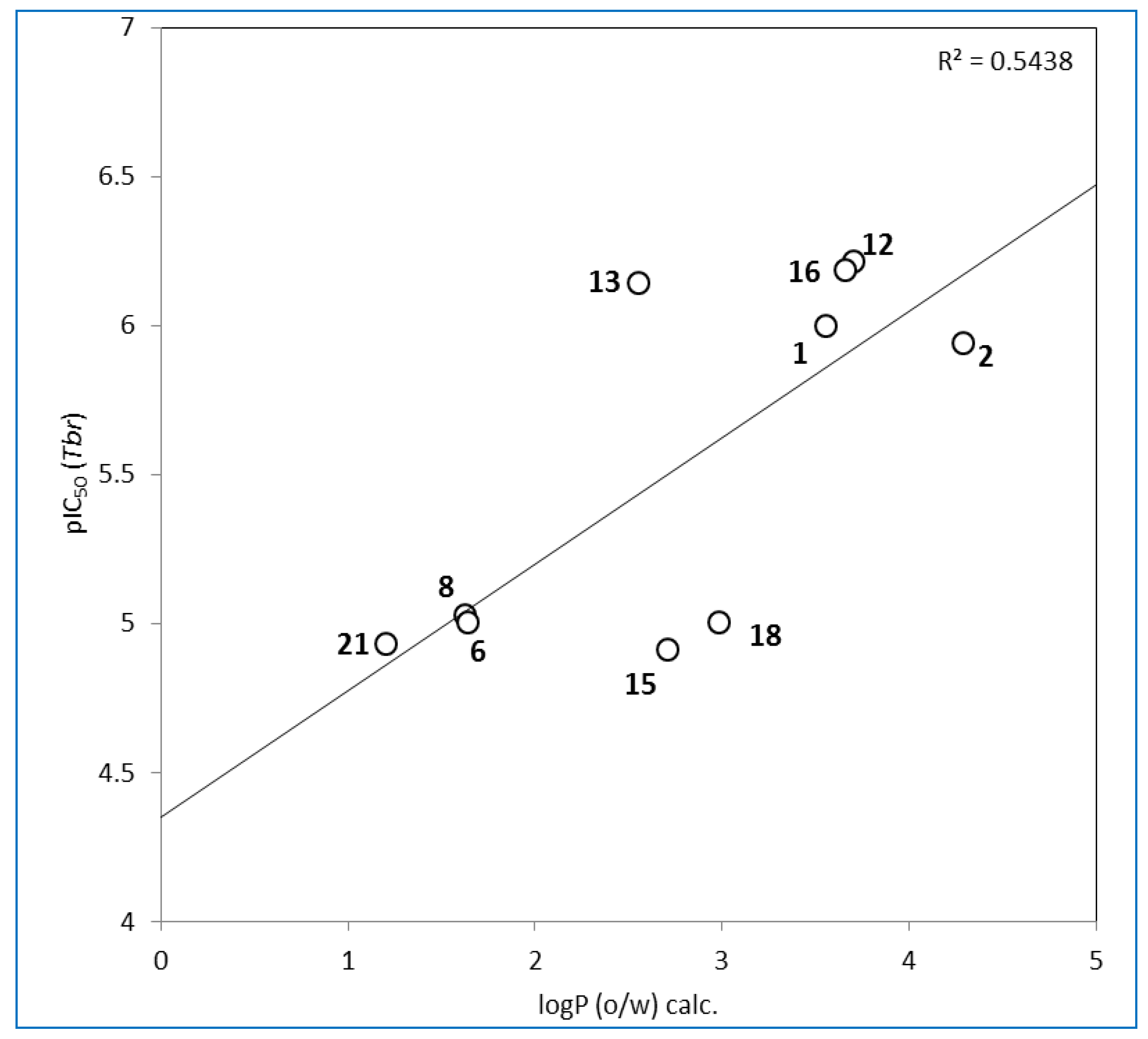
3. Experimental Section
3.1. Biological Material, Isolation and Structural Characterization of the Tested Compounds
3.2. Samples for Biological Tests
3.3. Anti-Trypanosomal Activity on T. brucei rhodesiense (Tbr)
3.4. Antileishmanial Activity on L. donovani (Ld; Axenic Amastigote Assay)
3.5. Antiplasmodial Activity on P. falciparum (Pf) NF54 Strain
3.6. Cytotoxicity Assay against L6 Cells
3.7. Positive Controls
3.8. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schmidt, T.J.; Khalid, S.A.; Romanha, A.J.; Alves, T.M.A.; Biavatti, M.W.; Brun, R.; Costa, F.B.D.; Castro, S.L.D.; Ferreira, V.F.; Lacerda, M.V.G.D.; et al. The potential of secondary metabolites from plants as drugs or leads against protozoan neglected diseases-Part II. Curr. Med. Chem. 2012, 19, 2176–2228. [Google Scholar] [CrossRef] [PubMed]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2013, 30, 237–323. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.J.; Grkovic, T.; Sykes, M.L.; Avery, V.M. Trypanocidal activity of marine natural products. Mar. Drugs 2013, 11, 4058–4082. [Google Scholar] [CrossRef] [PubMed]
- Rubio, B.K.; Tenney, K.; Ang, K.H.; Abdulla, M.; Arkin, M.; McKerrow, J.H.; Crews, P. The marine sponge Diacarnus bismarckensis as a source of peroxiterpene inhibitors of Trypanosoma brucei, the causative agent of sleeping sickness. J. Nat. Prod. 2009, 72, 218–222. [Google Scholar] [CrossRef] [PubMed]
- Watts, K.R.; Tenney, K.; Crews, P. The structural diversity and promise of antiparasitic marine invertebrate-derived small molecules. Curr. Opin. Biotechnol. 2010, 21, 808–818. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, L.M.; Knudsen, G.M.; Helbig, C.; Muylder, G.D.; Mascuch, S.M.; Mackey, Z.B.; Gerwick, L.; Clayton, C.; McKerrow, J.H.; Linington, R.G. Examination of the mode of action of the almiramide family of natural products against the kinetoplastid parasite Trypanosoma brucei. J. Nat. Prod. 2013, 76, 630–641. [Google Scholar] [CrossRef] [PubMed]
- Zofou, D.; Kang, F.N.; Sippl, W.; Efange, S.M.N. Bioactive natural products derived from the Central Africanflora against neglected tropical diseases and HIV. Nat. Prod. Rep. 2013, 30, 1098–1120. [Google Scholar] [CrossRef] [PubMed]
- Manilal, A.; Thajuddin, N.; Selvin, J.; Idhayadhulla, A.; Kumar, R.S.; Sujith, S. In vitro larvicidal activity of marine algae against the human vectors Culexquinque fasciatus (Say) and Aedes aegypti (Linnaeus) (Diptera: Culicidae). Int. J. Zool. Res. 2011, 7, 272–278. [Google Scholar] [CrossRef]
- Thao, N.P.; No, J.H.; Luyen, B.T.T.; Yang, G.; Byun, S.Y.; Goo, J.; Kim, K.T.; Cuong, N.X.; Nam, N.H.; Minh, C.V.; et al. Secondary metabolites from Vietnamese marine invertebrates with activity against Trypanosoma brucei and T. cruzi. Molecules 2014, 19, 7869–7880. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.J.; Kaiser, M.; Brun, R. Complete structural assignment of serratol, a cembrane-type diterpene from Boswellia serrata, and evaluation of its antiprotozoal activity. Planta Med. 2011, 77, 849–850. [Google Scholar] [CrossRef] [PubMed]
- Cuong, N.X.; Thao, N.P.; Luyen, B.T.T.; Ngan, N.T.T.; Thuy, D.T.T.; Song, S.B.; Nam, N.H.; Kiem, P.V.; Kim, Y.H.; Minh, C.V. Cembranoid diterpenes from the soft coral Lobophytum crassum and their anti-inflammatory activities. Chem. Pharm. Bull. 2014, 62, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Thao, N.P.; Luyen, B.T.T.; Ngan, N.T.T.; Song, S.B.; Cuong, N.X.; Nam, N.H.; Kiem, P.V.; Kim, Y.H.; Minh, C.V. New anti-inflammatory cembranoid diterpenoids from the Vietnamese soft coral Lobophytum crassum. Bioorg. Med. Chem. Lett. 2014, 24, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Quang, T.H.; Ha, T.T.; Minh, C.V.; Kiem, P.V.; Huong, H.T.; Ngan, N.T.T.; Nhiem, N.X.; Tung, N.H.; Tai, B.H.; Thuy, D.T.T.; et al. Cytotoxic and anti-inflammatory cembranoids from the Vietnamese soft coral Lobophytum laevigatum. Bioorg. Med. Chem. 2011, 19, 2625–2632. [Google Scholar] [CrossRef] [PubMed]
- Thao, N.P.; Nam, N.H.; Cuong, N.X.; Quang, T.H.; Tung, P.T.; Dat, L.D.; Chae, D.; Kim, S.; Koh, Y.S.; Kiem, P.V.; et al. Anti-inflammatory norditerpenoids from the soft coral Sinularia maxima. Bioorg. Med. Chem. Lett. 2013, 23, 228–231. [Google Scholar] [CrossRef] [PubMed]
- Thao, N.P.; Nam, N.H.; Cuong, N.X.; Quang, T.H.; Tung, P.T.; Tai, B.H.; Luyen, B.T.T.; Chae, D.; Kim, S.; Koh, Y.S.; et al. Diterpenoids from the soft coral Sinularia maxima and their inhibitory effects on lipopolysaccharide-stimulated production of proinflammatory cytokines in bone marrow-derived dendritic cells. Chem. Pharm. Bull. 2012, 60, 1581–1589. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.J.; Nour, A.M.M.; Khalid, S.A.; Kaiser, M.; Brun, R. Quantitative structure—Antiprotozoal activity relationships of sesquiterpene lactones. Molecules 2009, 14, 2062–2076. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.J.; Costa, F.B.D.; Lopes, N.P.; Kaiser, M.; Brun, R. In silico prediction and experimental evaluation of furanoheliangolide sesquiterpene lactones as potent agents against Trypanosoma brucei rhodesiense. Antimicrob. Agents Chemother. 2014, 58, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Räz, B.; Iten, M.; Grether-Bühler, Y.; Kaminsky, R.; Brun, R. The Alamar Blue® assay to determine drug sensitivity of African trypanosomes (T.b. rhodesiense and T.b. gambiense) in vitro. Acta Trop. 1997, 68, 139–147. [Google Scholar] [CrossRef]
- Cunningham, I. New culture medium for maintenance of tsetse tissues and growth of trypanosomatids. J. Protozool. 1977, 23, 325–329. [Google Scholar] [CrossRef]
- Matile, H.; Pink, J.R.L. Plasmodium falciparum malaria parasite cultures and their use in immunology. In Immunological Methods; Lefkovits, I., Pernis, B., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 221–234. [Google Scholar]
- Sample Availability: Samples of the compounds are not available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thao, N.P.; Luyen, B.T.T.; Brun, R.; Kaiser, M.; Van Kiem, P.; Van Minh, C.; Schmidt, T.J.; Kang, J.S.; Kim, Y.H. Anti-Protozoal Activities of Cembrane-Type Diterpenes from Vietnamese Soft Corals. Molecules 2015, 20, 12459-12468. https://doi.org/10.3390/molecules200712459
Thao NP, Luyen BTT, Brun R, Kaiser M, Van Kiem P, Van Minh C, Schmidt TJ, Kang JS, Kim YH. Anti-Protozoal Activities of Cembrane-Type Diterpenes from Vietnamese Soft Corals. Molecules. 2015; 20(7):12459-12468. https://doi.org/10.3390/molecules200712459
Chicago/Turabian StyleThao, Nguyen Phuong, Bui Thi Thuy Luyen, Reto Brun, Marcel Kaiser, Phan Van Kiem, Chau Van Minh, Thomas J. Schmidt, Jong Seong Kang, and Young Ho Kim. 2015. "Anti-Protozoal Activities of Cembrane-Type Diterpenes from Vietnamese Soft Corals" Molecules 20, no. 7: 12459-12468. https://doi.org/10.3390/molecules200712459
APA StyleThao, N. P., Luyen, B. T. T., Brun, R., Kaiser, M., Van Kiem, P., Van Minh, C., Schmidt, T. J., Kang, J. S., & Kim, Y. H. (2015). Anti-Protozoal Activities of Cembrane-Type Diterpenes from Vietnamese Soft Corals. Molecules, 20(7), 12459-12468. https://doi.org/10.3390/molecules200712459







