Mimicking the Lipid Peroxidation Inhibitory Activity of Phospholipid Hydroperoxide Glutathione Peroxidase (GPx4) by Using Fatty Acid Conjugates of a Water-Soluble Selenolane
Abstract
:1. Introduction
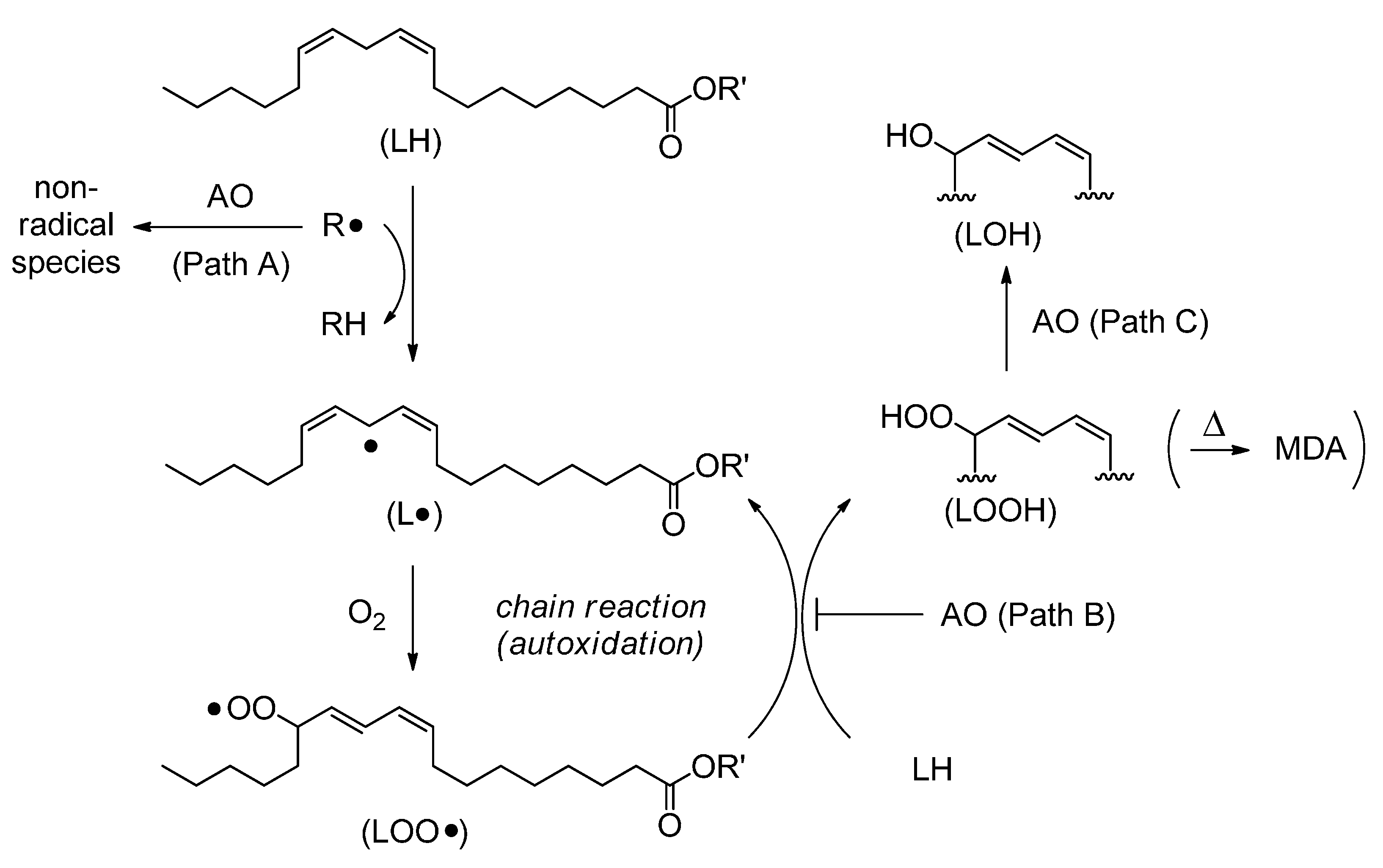
2. Results and Discussion
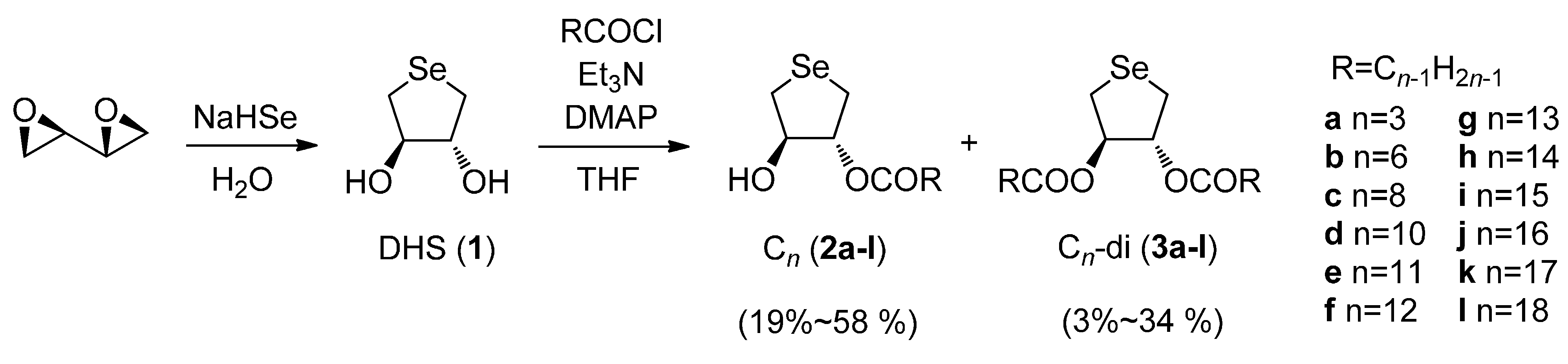
2.1. Synthesis of DHS-Fatty Acid Conjugates 2
2.2. Lipid Peroxidation Assay Using Lecithin/Cholesterol Liposome

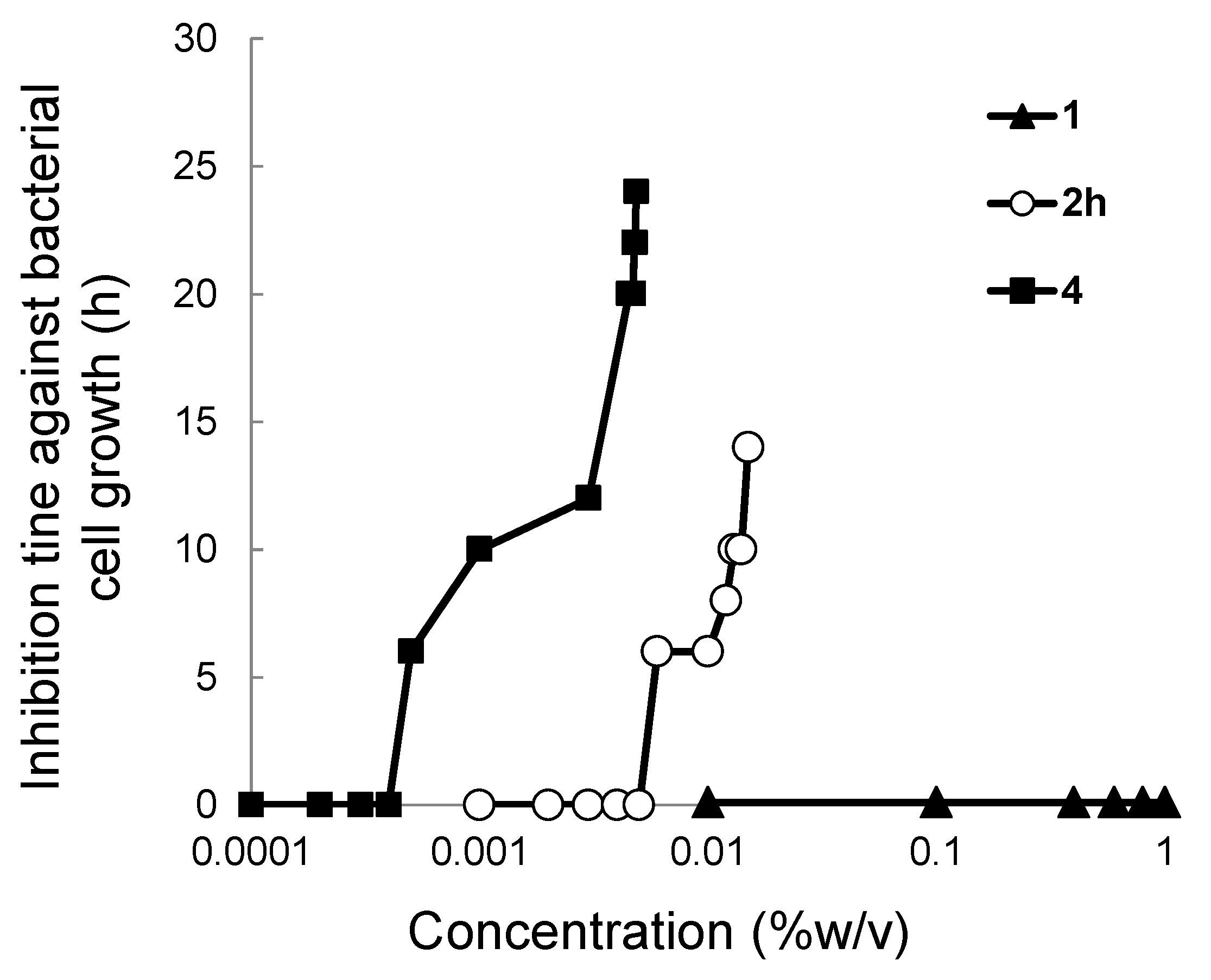
2.3. Colony Formation Assay
2.4. Antioxidant Function as Possible GPx4 Mimics

3. Experimental Section
3.1. General Information
3.2. Synthesis of DHS-Fatty Acid Conjugates 2a–l
3.3. Lipid Peroxidation Assay
3.4. Colony Formation Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Margis, R.; Dunand, C.; Teixeira, F.K.; Margis-Pinheiro, M. Glutathione peroxidase family—An evolutionary overview. FEBS J. 2008, 275, 3959–3970. [Google Scholar] [CrossRef] [PubMed]
- Brigelius-Flohé, R.; Maiorino, M. Glutathione peroxidases. Biochim. Biophys. Acta 2013, 1830, 3289–3303. [Google Scholar] [CrossRef] [PubMed]
- Ursini, F.; Heim, S.; Kiess, M.; Maiorino, M.; Roveri, A.; Wissing, J.; Flohé, L. Dual function of the selenoprotein PHGPx during sperm maturation. Science 1999, 285, 1393–1396. [Google Scholar] [CrossRef] [PubMed]
- Ufer, C.; Wang, C.C.; Fähling, M.; Schiebel, H.; Thiele, B.J.; Billett, E.E.; Kuhn, H.; Borchert, A. Translational regulation of glutathione peroxidase 4 expression through guanine-rich sequence-binding factor 1 is essential for embryonic brain development. Genes Dev. 2008, 22, 1838–1850. [Google Scholar] [CrossRef] [PubMed]
- Schneider, M.; Förster, H.; Boersma, A.; Seiler, A.; Wehnes, H.; Sinowatz, F.; Neumüller, C.; Deutsch, M.J.; Walch, A.; Hrabé de Angelis, M.; et al. Mitochondrial glutathione peroxidase 4 disruption causes male infertility. FASEB J. 2009, 23, 3233–3242. [Google Scholar] [CrossRef] [PubMed]
- Mugesh, G.; Singh, H.B. Synthetic organoselenium compounds as antioxidants: glutathione peroxidase activity. Chem. Soc. Rev. 2000, 29, 347–357. [Google Scholar] [CrossRef]
- Mugesh, G.; du Mont, W.W.; Sies, H. Chemistry of biologically important synthetic organoselenium compounds. Chem. Rev. 2001, 101, 2125–2179. [Google Scholar] [CrossRef] [PubMed]
- Bhabak, K.P.; Mugesh, G. Functional mimics of glutathione peroxidase: Bioinspired synthetic antioxidants. Acc. Chem. Res. 2010, 43, 1408–1419. [Google Scholar] [CrossRef] [PubMed]
- Raneva, V.; Shimasaki, H.; Furukawa, Y.; Ueta, N.; Yanishlieva, N.; Aaseng, J.E.; Partali, V.; Sliwka, H.R.; Yoshida, Y.; Niki, E. Action of 1-(11-selenadodecyl)-glycerol and 1-(11-selenadodecyl)-3-trolox-glycerol against lipid peroxidation. Lipids 2002, 37, 633–640. [Google Scholar] [CrossRef] [PubMed]
- Shanks, D.; Amorati, R.; Fumo, M.G.; Pedulli, G.F.; Valgimigli, L.; Engman, L. Synthesis and antioxidant profile of all-rac-α-selenotocopherol. J. Org. Chem. 2006, 71, 1033–1038. [Google Scholar] [CrossRef] [PubMed]
- Kumakura, F.; Mishra, B.; Priyadarsini, K.I.; Iwaoka, M. A water-soluble cyclic selenide with enhanced glutathione peroxidase-like catalytic activities. Eur. J. Org. Chem. 2010, 440–445. [Google Scholar] [CrossRef]
- Iwaoka, M.; Sano, N.; Lin, Y.Y.; Katakura, A.; Noguchi, M.; Takahashi, K.; Kumakura, F.; Arai, K.; Singh, B.G.; Kunwar, A.; et al. Fatty acid conjugates of water-soluble (±)-trans-selenolane-3,4-diol: Effects of alkyl chain length on the antioxidant capacity. ChemBioChem 2015, 16, 1226–1234. [Google Scholar] [CrossRef] [PubMed]
- Phadnis, P.P.; Wadawale, A.; Priyadarsini, K.I.; Jain, V.K.; Iwaoka, M. Synthesis, characterization, and structure of trans-3,4-dihydroxy-1-selenolane {DHS(OH)2} substituted derivatives. Tetrahedron Lett 2015, 56, 2293–2296. [Google Scholar] [CrossRef]
- Iwaoka, M.; Takahashi, T.; Tomoda, S. Syntheses and structural characterization of water-soluble selenium reagents for the redox control of protein disulfide bonds. Heteroat. Chem. 2001, 12, 293–299. [Google Scholar] [CrossRef]
- Meotti, F.C.; Stangherlin, E.C.; Zeni, G.; Nogueira, C.W.; Rocha, J.B.T. Protective role of aryl and alkyl diselenides on lipid peroxidation. Environ. Res. 2004, 94, 276–282. [Google Scholar] [CrossRef]
- Santos, F.W.; Zeni, G.; Rocha, J.B.T.; Weis, S.N.; Fachinetto, J.M.; M. Favero, A.; Nogueira, C.W. Diphenyl diselenide reverses cadmium-induced oxidative damage on mice tissues. Chem. Biol. Interact. 2005, 151, 159–165. [Google Scholar] [CrossRef] [PubMed]
- De Freitas, A.S.; de Souza Prestes, A.; Wagner, C.; Sudati, J.H.; Alves, D.; Porciúncula, L.O.; Kade, I.J.; Rocha, J.B.T. Reduction of diphenyl diselenide and analogs by mammalian thioredoxin reductase is independent of their gluthathione peroxidase-like activity: A possible novel pathway for their antioxidant activity. Molecules 2010, 15, 7699–7714. [Google Scholar] [CrossRef]
- Da Rocha, J.T.; Pinton, S.; Mazzanti, A.; Mazzanti, C.M.; Beckemann, D.V.; Nogueira, C.W.; Zeni, G. Effects of diphenyl diselenide on lipid profile and hepatic oxidative stress parameters in ovariectomized female rats. J. Pharm. Pharmacol. 2011, 63, 663–669. [Google Scholar] [CrossRef] [PubMed]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Cai, Y.J.; Fang, J.G.; Ma, L.P.; Yang, L.; Liu, Z.L. Inhibition of free radical-induced peroxidation of rat liver microsomes by resveratrol and its analogues. Biochim. Biophys. Acta 2003, 1637, 31–38. [Google Scholar] [CrossRef]
- Michener, H.D. The action of subtilin on heated bacterial spores. J. Bacteriol. 1955, 70, 192–200. [Google Scholar] [PubMed]
- Sakamoto, T.; Hours, R.A.; Sakai, T. Purification, characterization, and production of two pectic transeliminases with protopectinase activity from Bacillus subtilis. Biosci. Biotech. Biochem. 1994, 58, 353–358. [Google Scholar] [CrossRef]
- Auclair, C.; Voisin, E.; Banoun, H.; Paoletti, C.; Bernadou, J.; Meunier, B. Potential antitumor agents: Synthesis and biological properties of aliphatic amino acid 9-hydroxyellipticinium derivatives. J. Med. Chem. 1984, 27, 1161–1166. [Google Scholar] [CrossRef] [PubMed]
- Russell, A.D. Bacterial spores and chemical sporicidal agents. Clin. Microbiol. Rev. 1990, 3, 99–119. [Google Scholar] [PubMed]
- Arai, K.; Dedachi, K.; Iwaoka, M. Rapid and quantitative disulfide bond formation for a polypeptide chain using a cyclic selenoxide reagent in an aqueous medium. Chem. Eur. J. 2011, 17, 481–485. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of compounds 1, 2a–l and 3a–l are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iwaoka, M.; Katakura, A.; Mishima, J.; Ishihara, Y.; Kunwar, A.; Priyadarsini, K.I. Mimicking the Lipid Peroxidation Inhibitory Activity of Phospholipid Hydroperoxide Glutathione Peroxidase (GPx4) by Using Fatty Acid Conjugates of a Water-Soluble Selenolane. Molecules 2015, 20, 12364-12375. https://doi.org/10.3390/molecules200712364
Iwaoka M, Katakura A, Mishima J, Ishihara Y, Kunwar A, Priyadarsini KI. Mimicking the Lipid Peroxidation Inhibitory Activity of Phospholipid Hydroperoxide Glutathione Peroxidase (GPx4) by Using Fatty Acid Conjugates of a Water-Soluble Selenolane. Molecules. 2015; 20(7):12364-12375. https://doi.org/10.3390/molecules200712364
Chicago/Turabian StyleIwaoka, Michio, Arisa Katakura, Jun Mishima, Yoshimi Ishihara, Amit Kunwar, and Kavirayani Indira Priyadarsini. 2015. "Mimicking the Lipid Peroxidation Inhibitory Activity of Phospholipid Hydroperoxide Glutathione Peroxidase (GPx4) by Using Fatty Acid Conjugates of a Water-Soluble Selenolane" Molecules 20, no. 7: 12364-12375. https://doi.org/10.3390/molecules200712364
APA StyleIwaoka, M., Katakura, A., Mishima, J., Ishihara, Y., Kunwar, A., & Priyadarsini, K. I. (2015). Mimicking the Lipid Peroxidation Inhibitory Activity of Phospholipid Hydroperoxide Glutathione Peroxidase (GPx4) by Using Fatty Acid Conjugates of a Water-Soluble Selenolane. Molecules, 20(7), 12364-12375. https://doi.org/10.3390/molecules200712364







