Induction of Mitochondrial Dependent Apoptosis in Human Leukemia K562 Cells by Meconopsis integrifolia: A Species from Traditional Tibetan Medicine
Abstract
:1. Introduction
2. Results and Discussion
2.1. Cytotoxicity of M. integrifolia
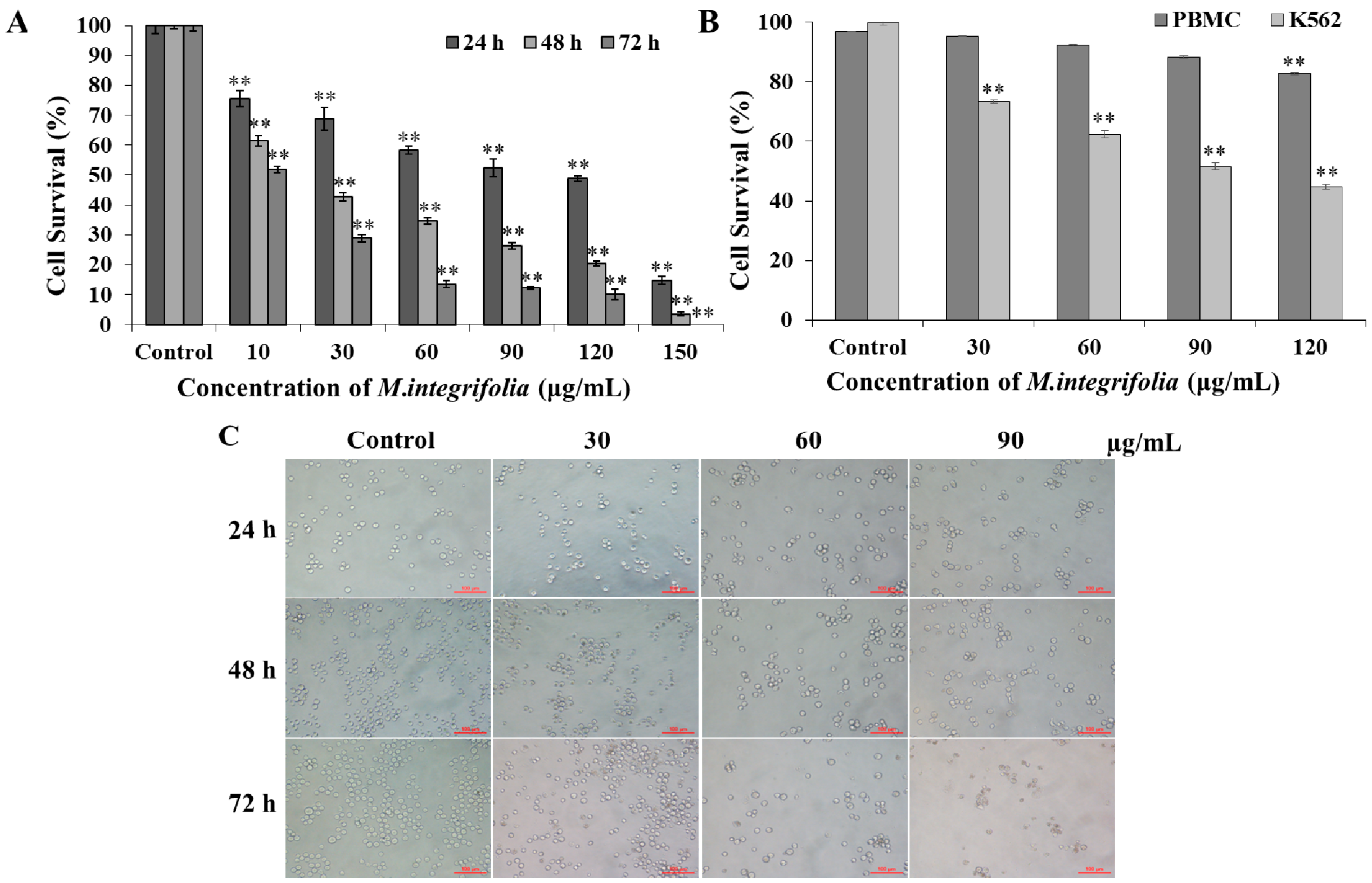
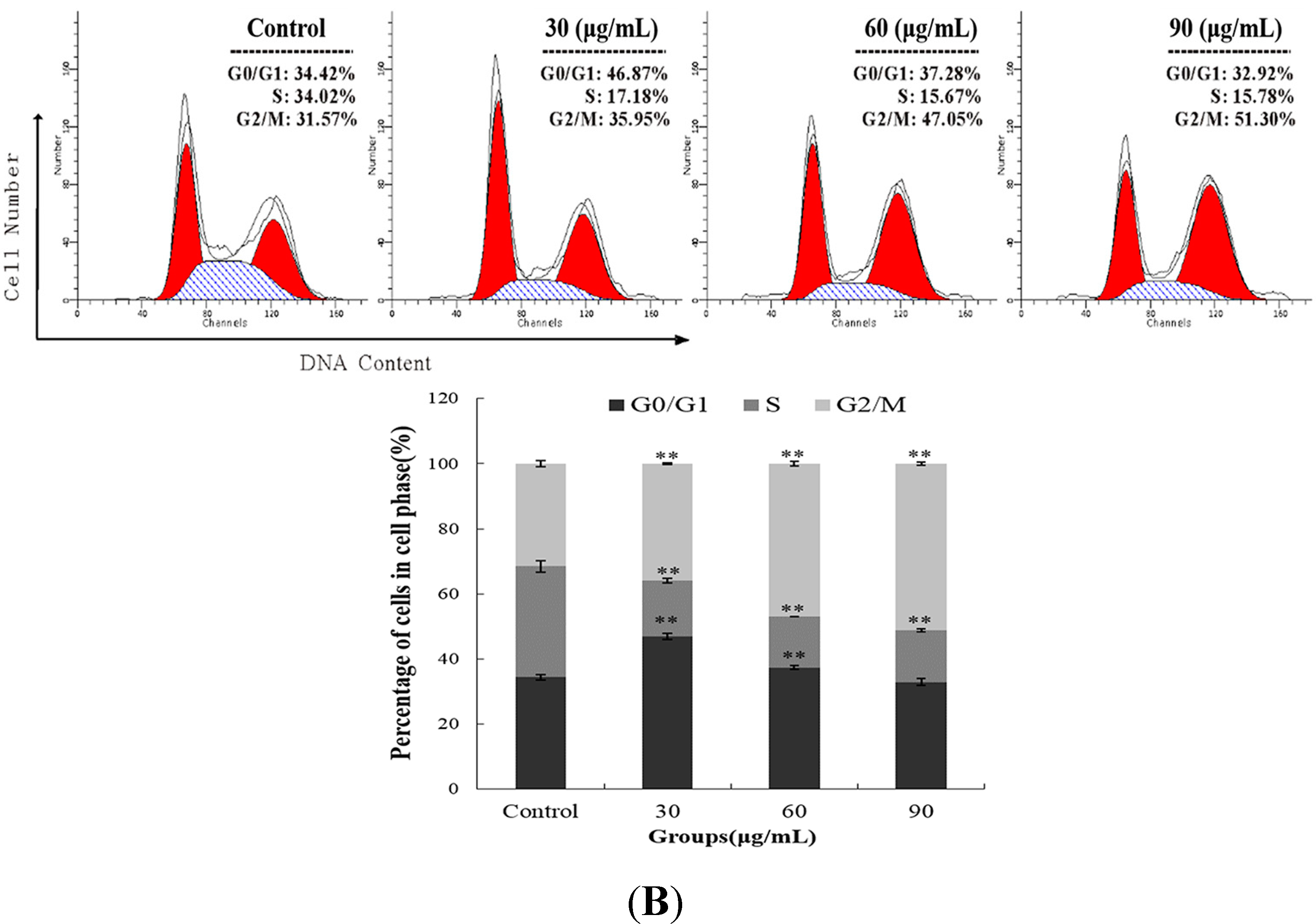
2.2. M. integrifolia Causes Cell Cycle Arrest in K562 Cells
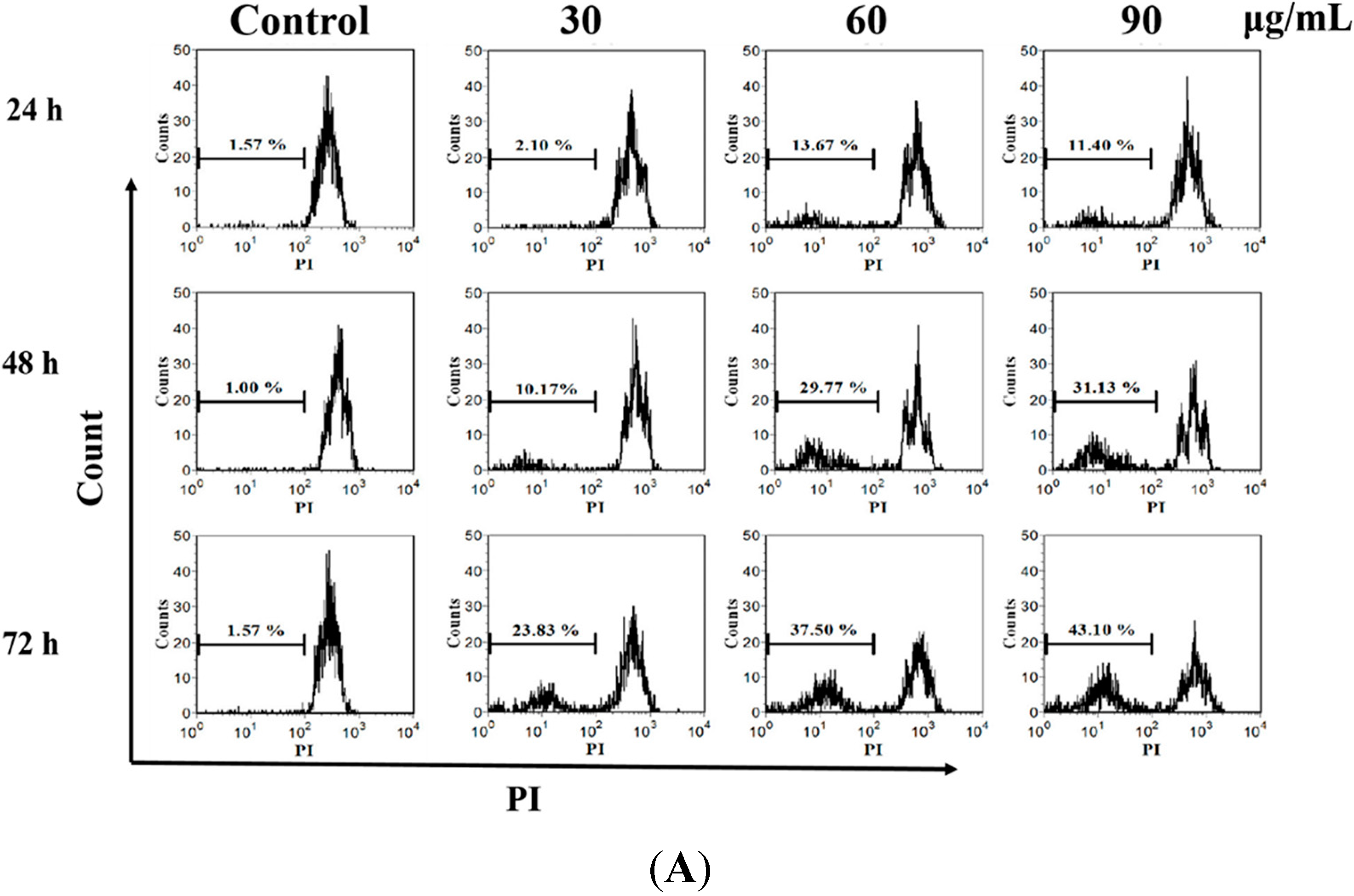
2.3. M. integrifolia Triggers Cell Apoptosis in K562 Cells
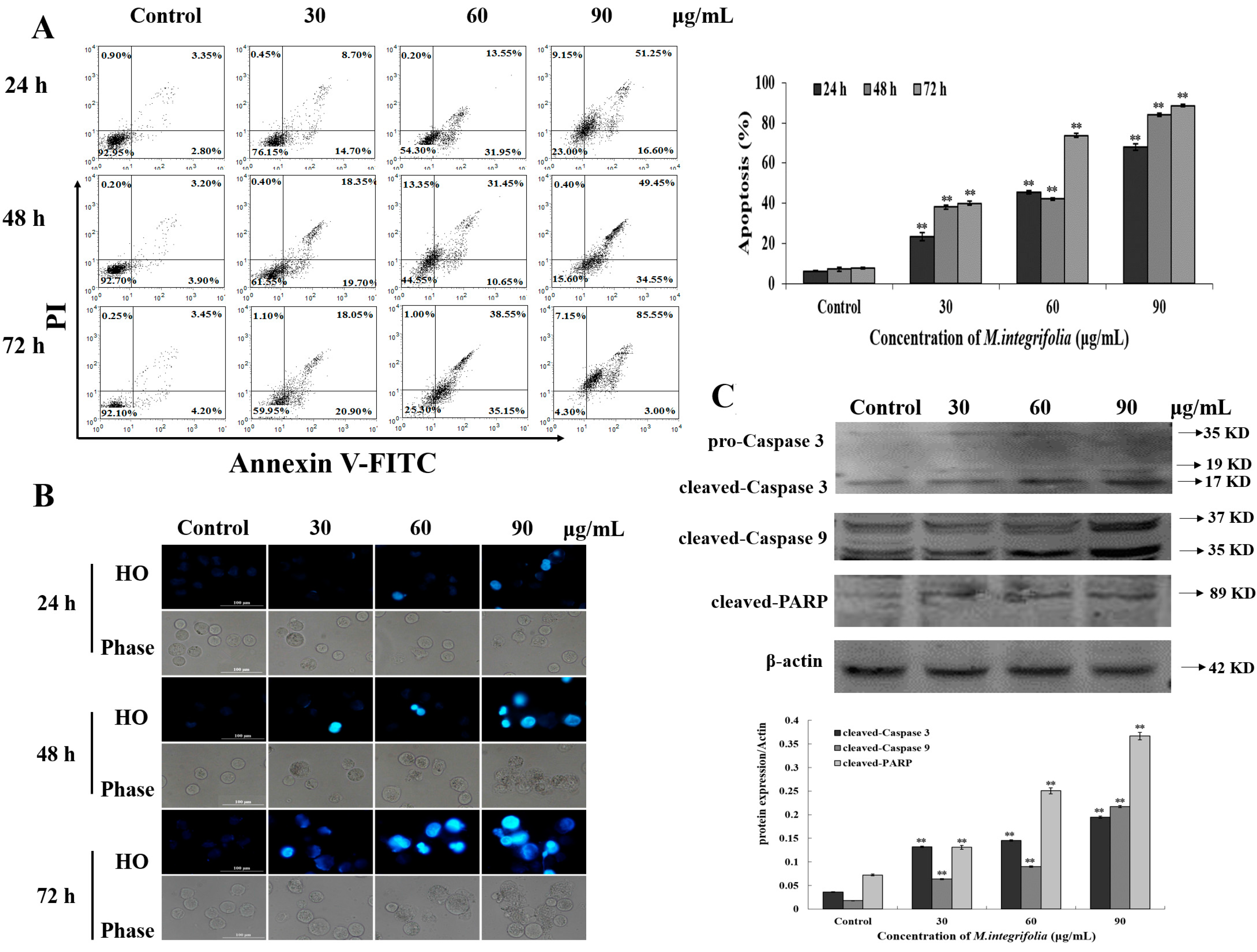
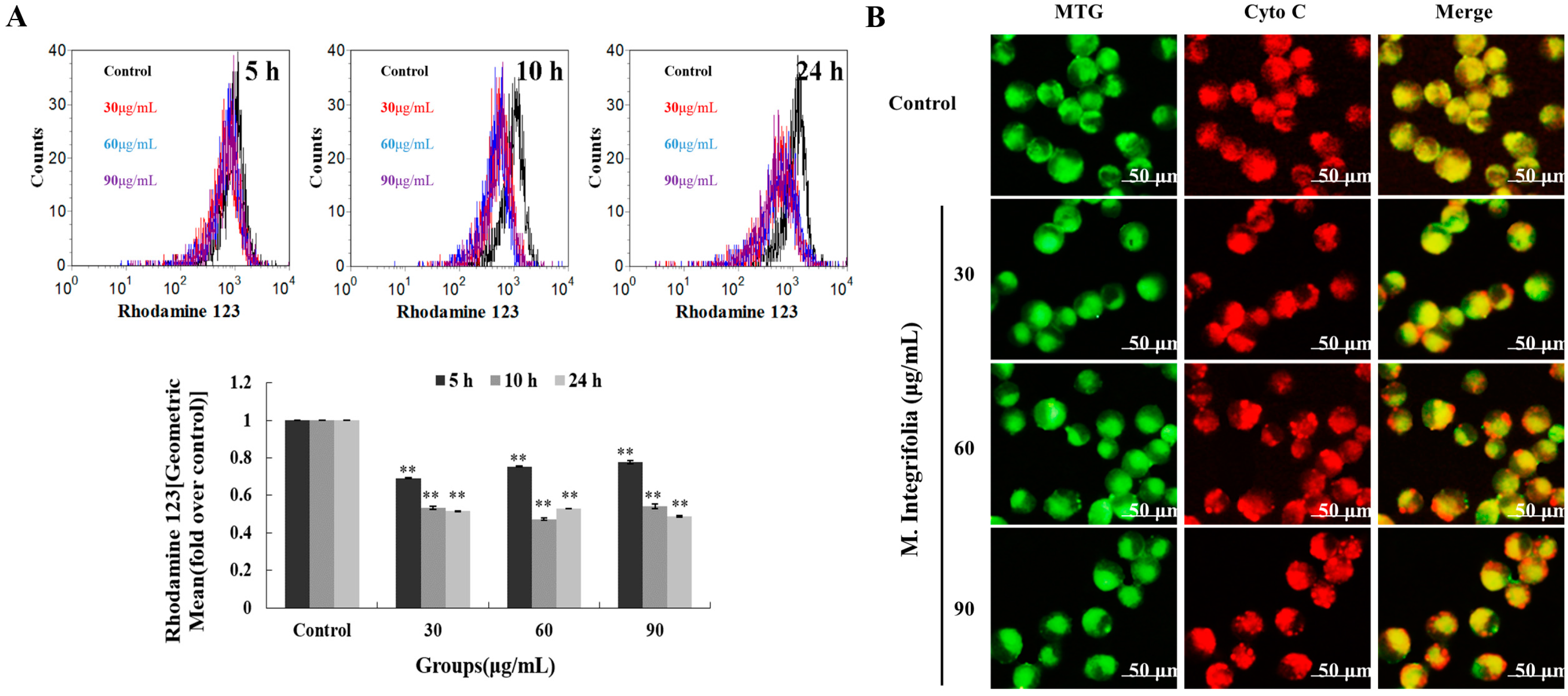
2.4. Mitochondria Play a Key Role Following M. integrifolia Incubation
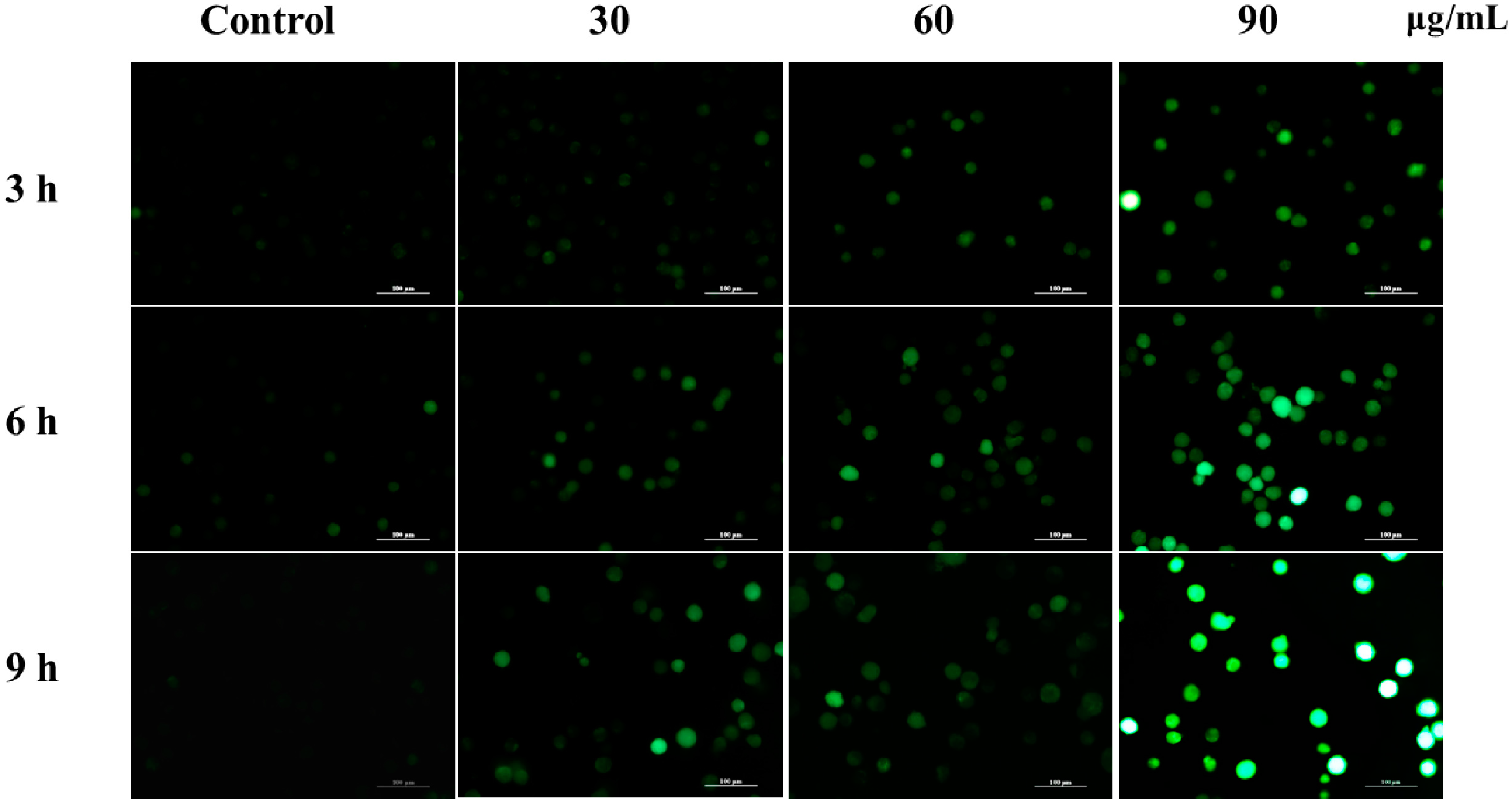
2.5. M. integrifolia Initiates ROS Accumulation in K562 Cells
3. Experimental Section
3.1. Preparation for the M. integrifolia Extract
3.2. Cell Cultures
3.3. Cell Viability Tests
3.4. Phase-Contrast Microscopy Observation
3.5. Apoptosis Detection by Flow Cytometry
3.6. Hoechst 33342 Staining
3.7. Cell Cycle Analysis
3.8. DNA Fragmentation Assay
3.9. Intracellular Reactive Oxygen Species Detection
3.10. Measurement of Mitochondrial Membrane Potential (MMP)
3.11. Cytochrome C Detection
3.12. Western Blotting Staining
3.13. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Carolan, J.C.; Hook, I.L.; Chase, M.W.; Kadereit, J.W.; Hodkinson, T.R. Phylogenetics of Papaver and related genera based on DNA sequences from ITS nuclear ribosomal DNA and plastid trnL intron and trnL-F intergenic spacers. Ann. Bot. 2006, 98, 141–155. [Google Scholar] [CrossRef]
- Kapoor, L.D. Opium Poppy: Botany, Chemistry and Pharmacology; Food Products Press: New York, NY, USA, 1995. [Google Scholar]
- Sulaiman, I.M.; Hasnain, S.E. Randomamplified polymorphic DNA (RAPD) markers reveal genetic homogeneity in the endangered Himalayan species Meconopsis paniculata and M. simplicifolia. Theor. Appl. Genet. 1996, 93, 91–96. [Google Scholar] [CrossRef]
- Wu, C.; Chuang, H. A study on the taxonomic system of the genus Meconopsis. Acta Bot. Yunnanica 1980, 2, 371–381. [Google Scholar]
- Yang, F.S.; Qin, A.L.; Li, Y.F.; Wang, X.Q. Great Genetic Differentiation among Populations of Meconopsis integrifolia and its Implication for Plant Speciation in the Qinghai-Tibetan Plateau. PLoS ONE 2012, 7, e37196. [Google Scholar] [CrossRef]
- Wu, H.F.; Ding, I.S.; Wang, H.; Zhang, X.F. Advances in the research of phytochemistry and pharmacology of Meconopsis Vig. Nat. Prod. Res. Dev. 2011, 23, 163–168. [Google Scholar]
- Samant, S.S. Diversity, endemism and socio-economic values of the Indian Himalayan Papaveraceae and Fumariaceae. J. Indian Bot. Soc. 2005, 84, 33–44. [Google Scholar]
- Zhou, G.; Chen, Y.X.; Liu, S.; Yao, X.C.; Wang, Y.W. In vitro and in vivo hepatoprotective and antioxidant activity of ethanolic extract from Meconopsis integrifolia (Maxim.) Franch. J. Ethnopharmacol. 2013, 148, 664–670. [Google Scholar]
- Wu, H.F.; Pan, L.; Zou, D.S.; Yang, S.J.; Zhang, X.F. Analysis on volatile oils from three species of Meconopsis by GC-MS. Chin. Pharm. J. 2006, 41, 1298–1300. [Google Scholar]
- Zhou, Y.; Song, J.Z.; Choi, F.F.K.; Wu, H.F.; Qiao, C.F.; Ding, L.S.; Gesang, S.L.; Xu, H.X. An experimental design approach using response surface techniques to obtain optimal liquid chromatography and mass spectrometry conditions to determine the alkaloids in Meconopsis species. J. Chromatogr. A 2009, 1216, 7013–7023. [Google Scholar] [CrossRef]
- Ding, L.; Li, J.P. Study on protective effect of Meconopsis quintuplinervia Regel on experimental hepatic injury in mice. Chin. Qinghai J. Anim. Vet. 2007, 37, 7–8. [Google Scholar]
- Brisdelli, F.; Coccia, C.; Cinque, B.; Cifone, M.G.; Bozzi, A. Induction of apoptosis by quercetin: different response of human chronic myeloid (K562) and acute lymphoblastic (HSB-2) leukemia cells. Mol. Cell. Biochem. 2007, 296, 137–149. [Google Scholar] [CrossRef]
- Li-Weber, M. Targeting apoptosis pathways in cancer by Chinese medicine. Cancer Lett. 2013, 332, 304–312. [Google Scholar] [CrossRef]
- Choedon, T.; Dolma, D.; Mathan, G.; Kumar, V. Molecular insights into the anti-cancer properties of traditional Tibetan medicine Yukyung Karne. BMC Complement. Altern. Med. 2014, 14, 380. [Google Scholar] [CrossRef]
- Giono, L.E.; Manfredi, J.J. Mdm2 is required for inhibition of CDK2 activity by p21, thereby contributing to p53-dependent cell cycle arrest. Mol. Cell. Biol. 2007, 27, 4166–4178. [Google Scholar] [CrossRef]
- Leventis, P.A.; Grinstein, S. The distribution and function of phosphatidylserine in cellular membranes. Ann. Rev. Biophys. 2010, 39, 407–427. [Google Scholar] [CrossRef]
- Hafeez, B.B.; Siddiqui, I.A.; Asim, M.; Malik, A.; Afaq, F.; Adhami, V.M.; Saleem, M.; Din, M.; Mukhtar, H. A dietary anthocyanidin delphinidin induces apoptosis of human prostate cancer PC3 cells in vitro and in vivo: Involvement of nuclear factor-κB signaling. Cancer Res. 2008, 68, 8564–8572. [Google Scholar] [CrossRef]
- Ndozangue-Tou riguine, O.; Hamelin, J.; Brйard, J. Cytoskeleton and apoptosis. Biochem. Pharmacol. 2008, 76, 11–18. [Google Scholar] [CrossRef]
- Naziroglu, M.; Cihangir Uğuz, A.; Koçak, A.; Bal, R. Acetaminophen at different doses protects brain microsomal Ca2+ ATPase and the antioxidant redox system in rats. J. Membr. Biol. 2009, 231, 57–64. [Google Scholar] [CrossRef]
- Lewinska, A.; Siwak, J.; Rzeszutek, I.; Wnuk, M. Diosmin induces genotoxicity and apoptosis in DU145 prostate cancer cell line. Toxicol. in Vitro 2014, 29, 417–425. [Google Scholar] [CrossRef]
- Song, H.; Bao, J.; Wei, Y.; Chen, Y.; Mao, X.; Li, J.; Yang, Z.; Xue, Y. Kaempferol inhibits gastric cancer tumor growth: An in vitro and in vivo study. Oncol. Rep. 2015, 33, 868–874. [Google Scholar] [CrossRef]
- Gerald, M. Caspases: The executioners of apoptosis. Biochem. J. 1997, 326, 1–16. [Google Scholar]
- He, N.; Shi, X.; Zhao, Y.; Tian, L.; Wang, D. Inhibitory effects and molecular mechanisms of selenium-containing tea polysaccharides on human breast cancer mcf-7 cells. J. Agric. Food Chem. 2013, 61, 579–588. [Google Scholar] [CrossRef]
- Fulda, S.; Galluzzi, L.; Kroemer, G. Targeting mitochondria for cancer therapy. Nat. Rev. Drug Discov. 2010, 9, 447–464. [Google Scholar] [CrossRef]
- Chidambara Murthy, K.N.; Jayaprakasha, G.K.; Kumar, V.; Rathore, K.S.; Patil, B.S. Citrus limonin and its glucoside inhibit colon adenocarcinoma cell proliferation through apoptosis. J. Agric. Food Chem. 2011, 59, 2314–2323. [Google Scholar] [CrossRef]
- Hengartner, M.O. The biochemistry of apoptosis. Nature 2000, 407, 770–776. [Google Scholar] [CrossRef]
- Marzo, I.; Brenner, C.; Zamzami, N.; Jurgensmeier, J.M.; Susin, S.A. Bax and adenine nucleotide translocator cooperate in the mitochondrial control of apoptosis. Science 1998, 281, 2027–2031. [Google Scholar] [CrossRef]
- Indran, I.R.; Tufo, G.; Pervaiz, S.; Brenner, C. Recent advances in apoptosis, mitochondria and drug resistance in cancer cells. Bba-biomembranes 2011, 1807, 735–745. [Google Scholar] [CrossRef]
- Krysko, D.V. Apoptosis and necrosis: Detection, discrimination and phagocytosis. Methods 2008, 44, 205–221. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, J.; Wang, P.; Wang, X.; Tang, W.; Liu, C.; Wang, Y.; Yuan, W.; Kong, L.; Liu, Q. Induction of Mitochondrial Dependent Apoptosis in Human Leukemia K562 Cells by Meconopsis integrifolia: A Species from Traditional Tibetan Medicine. Molecules 2015, 20, 11981-11993. https://doi.org/10.3390/molecules200711981
Fan J, Wang P, Wang X, Tang W, Liu C, Wang Y, Yuan W, Kong L, Liu Q. Induction of Mitochondrial Dependent Apoptosis in Human Leukemia K562 Cells by Meconopsis integrifolia: A Species from Traditional Tibetan Medicine. Molecules. 2015; 20(7):11981-11993. https://doi.org/10.3390/molecules200711981
Chicago/Turabian StyleFan, Jianping, Pan Wang, Xiaobing Wang, Wei Tang, Chunliang Liu, Yaqin Wang, Wenjuan Yuan, Lulu Kong, and Quanhong Liu. 2015. "Induction of Mitochondrial Dependent Apoptosis in Human Leukemia K562 Cells by Meconopsis integrifolia: A Species from Traditional Tibetan Medicine" Molecules 20, no. 7: 11981-11993. https://doi.org/10.3390/molecules200711981
APA StyleFan, J., Wang, P., Wang, X., Tang, W., Liu, C., Wang, Y., Yuan, W., Kong, L., & Liu, Q. (2015). Induction of Mitochondrial Dependent Apoptosis in Human Leukemia K562 Cells by Meconopsis integrifolia: A Species from Traditional Tibetan Medicine. Molecules, 20(7), 11981-11993. https://doi.org/10.3390/molecules200711981




