Nanofibers of Cellulose and Its Derivatives Fabricated Using Direct Electrospinning
Abstract
:1. Introduction
2. ES Processes of Cellulose and Cellulose-Derivatives
2.1. Cellulose-Derivatives
| Matrix Polymers | Solvents (Concentrations) | Fiber Diameters in Averages (nm) | Post-Spun Treatments and Focus of Research | References |
|---|---|---|---|---|
| CA (1) | Acetone/DW = 5:1 (8%) | 300–500 | Deacetylation using 50 mM NaOH for conversion to RC-ESNWs; Detection of metal ions | [5] |
| CA: PVA | CA, Acetone/DMF = 2:1 (17 wt %) PVA, DW (10 or 15 wt %) | CA, 740 nm PVA, 340 or 710 nm | Independent nozzles for CA and PVA solutions to prepare composite ESNWs; Deacetylation using 50 mM NaOH for conversion to RC-ESNWs; Evaluation of hydrophilic properties before and after deacetylation. | [6] |
| CAP (2) (Mw ~60,000 g/mol) | Aceton/DMF = 3: 1 (25 wt/v %) | 500–800 | Sustainable release of low molecular weight substances having anti-virus activity, suppression of infective activity of the virus, and so on. | [7] |
| CA (Mw, 25,000) | Acetone/DMF = 1: 4 (14 wt %) | 170 ± 40 nm | Antibacterial activity expression by fabrication of nano-composite with ZnO. | [8] |
| CA (Dp, 200; DA, 2.5)/RSF (3) = 1:9 | Formic acid (12 wt %) | 142 nm ± 32% | Changes in mechanical properties of ESNWs though composite fabrication and metal ion adsorption. | [9,10] |
| PMIA (4)/CA (1:2) or cellulose (6:1) | PMIA, 8 wt % LiCl/DMAc (15%) Cellulose, swelled in 8 wt % LiCl/DMAc (10%) CA, MDAc (15 wt %) | PMIA/CA, 250–300 nm PMIA/cellulose, 250–350 nm | Development of ES process and reinforcement of mechanical properties of ESNWs. | [11] |
| CA (Mw, 61,000 g/mol, acetylation 40%) | Acetone/DMAc = 2:1 (20 wt %) | 750 nm (500–1500 nm) | Graft polymerization of methacrylic acid on CA and evaluation meal ion adsorption. | [12] |
| CA (Mw, 30,000, actylation 39.8%) | Acetone/DMAc = 2:1 (16 wt/v %) | 385 nm | Deacetylation using 0.3 M NaOH, then condensation of Oxolane-2,5-dione (succinic anhydride), then evaluation of metal ion adsorption. | [13] |
| CA (Mw, 30,000 g/mol, acetylation 39.8%)/PEO (5) (Mn, 300,000 g/mol) | CA, 99.9% AcOH (20 wt/v %) PEO 90% EtOH (3 wt/v %) | CA soln/POE soln = 99.6/0.4–91.9, 950–1170 nm | Composite ESNWs with ZnO nano-particles, changes in mechanical properties, and application for food wrapping technology. | [14] |
| CA (Mw, 30,000 Da, actylation 39.7 wt %) | Acetone/DMAc = 2:1 v/v% (17 w/w %) | 701–1057 nm | Development of ES process, and in vitro evaluations on sustained release of gallic acid (3,4,5-trihydroxy benzoic acid) | [15,16] |
| Cellulose (Dp, 1100) | [EMIM] [OAc] (6) (1.75 wt %) * | 1000–2500 nm ** | A fundamental study on fabrication of CB(7) -binding ESNWs for application as affinity ligand carrier. | [17] |
| Cellulose (Dp, 1100)/chitosan (800 cP) or/PMMA(8) (120 kDa) | [EMIM] [OAc] (Cellulose, 2 w/w%)/chitosan, 0.4 w/w %) [EMIM] [OAc]/DMSO/TritonX100 (cellulose, 2.25 w/w %/PMMA 0.5 w/w %) * | Cellulose/chitosan, 150–450 nm Cellulose/PMMA, 150–350 nm | Partial oxidative cleavage using periodates to generate aldehyde groups to bind Lysostaohin in a fundamental study for application as antibacterial wound dressing materials. | [18] |
| Cellulose/Cellulose nanocrystal CNC) | Cellulose, NMMO(9)/DW = 4: 1 wt (1.5 wt %), Shell solution CNC, DMSO (0.42–3.28 wt %), core suspension | ~100 nm | Development of ES process using layered nozzles having shell (cellulose in NMMO) and core (CNC dispersion in DMSO); Relationship between crystalinities of CNC and modulus of ESNWs. | [19,20] |
| Cellulose (cotton linter, Dp, 12,000) | 8.5 wt % LiCl/DMAc (1.0–1.35 wt %) | 80–100 nm | Immersion in water to revome residual LiCl and development of ES process for high molecular weight cellulose preparation. | [21] |
| polyquaternium-4 cellulose (PQ-4) (N, 1.5%–2.3%)/PVA (Dp, 1750 ± 50, DA, 98%) = 1:3, 1:2, 1:1 | PQ-4, DW (2–8 wt %) PVA, DW (1, 10 wt %) PQ-4/PVA, DW (7 wt %) | PQ-4, no-spun PQ-4/PVA 1:3, 202–217 (±38–59) 1:2, 157–246 (±46–66) 1:3, 211–249 (±60–71) | Antibacterial activity expressed by blend with PQ-4. | [22] |
| CA (acetylation, 29.6%) | Acetone/DMAc = 3:2 (15 wt %) | 200 nm | Deacetylation using 0.5M KOH/EtOH solution, then partial oxidative cleavage with periodate to generate aldehyde groups to immobilize an enzyme, lipase, and evaluation of immobilized enzyme activity. | [23] |
| CA | TFE (10) (150 g/L) | 200 nm–3000 nm | Deacetylation using 0.1 N NaOH in EtOH/DW = 1:4, then partial oxidative cleavage using sodium iodate to generate aldehyde groups to bind 2-aminoethyl sulfate and evaluation of scaffolding properties for cell culture. | [24] |
| CA (Mw, 30,000) | DCM (11)/Acetone = 1:1–3:1 (v:v) (5.0, 7.5, 10 w.v %) | DCM/Acetone = 1:1, CA, 10 wt %, 300–1000 2:1, CA, 7.5 wt %, 750–1500 3:1, CA 5 wt %, 1500–3500 | Development of CA fine fibers having porous structures. | [25] |
| CA (Mw, 61,000, acetylation 40%) | AcCN (12)/0–30w/w %EtOH (15–21 w/w %) | Sub-micrometric fibers | Vacuum deposition of Al, Cu, and Ag onto CA-ESNWs and evaluation of electrochemical properties for application as bio-battery. | [26] |
| CA (Mw, 30,000 g/mol)/PBA (13) (610,000 g/mol) = 90:10, 80:20, 70:30 (w/w) | Acetone//DMF = 8:2 (w/w)/0.2 wt % isocyanate | CA/PBA = 90:10, 510 80:20, 1700 70:30, 3980 | Curing using isocyanate and effect of the treatment on the fin fiber morphologies of ESNWs. | [27] |
| CA (Mw, ~30 kDa, acetylation 39.7 wt %)/EA (14) (14–76 kDa) = 91:9, 77:23, 66:34 (w/w) | AcOH/formic acid/DW/tween40 (CA/EA = 91:9, 19.25wt%; 77:23, 18.25 wt %, 66:34, 17.25 wt %) | CA/EA = 91:9, 242 ± 32 77:23, 384 ± 54 66:34, 410 ± 38 | A fundamental study on development of eatable fine fiber ESNWs for application as in vivo sustainable release mateials. | [28] |
| CA (d, 1.184 g/cm3; Tm, 280 °C)/PAN (15) (d, 1.184 g/cm3, Tm, 317 °C)/MWCNT (16) (d, 1.2–1.7 g/cm3) = | CA, Acetone/DMAc =1:2 (15 wt %/10 wt %), core soln. PAN/MWCNT, DMF (10 wt %/35 wt %), shell susp. | 150 (core, ~60; shell, ~50) | Development of ES process for highly loaded CNT composite ESNWs, and evaluation of thermostabilities of ESNWs for application of electronic devises. | [29] |
| CA | Acetone/BzOH (17) = 2:1 (14 wt %) MEK (18)/BzOH = 4:1 (14 wt %) Acetone/DMSO =2:1 (18 wt %), others | 3410 ± 1780 2030 ± 660 650 ± 130 | A fundamental investigation of processing condition, including solubilization parameters of solutes and solvents, viscosity, and correlation with the fibrous structures. | [30] |
| CA (Mn, 30,000, acetylation 39.8) | Acetone/DMF/DW = 85%/10%/5% (v/v/v) (17 v/v %) | 500 (100–1000) | Development of Quillaja sapnin-loaded ESNWs then deacetylatioin using 50mM NaOH aq/EtOH and evaluation of sub-component yields in RC-ESNW and antimycotic activities. | [31] |
| CA (Mn, 30,000, acetylation 39.8%, DS, 2.46)/POSS (19) | Acetone/DMAc = 2:1 (CA, 15 wt %/) POSS, 3 or 5 wt %CA | POSS3 wt %, 262 ± 59 POSS5 wt %, 269 ± 50 | Development of organic/inorganic composite ESNWs having nano-scaled dispersion of the inorganic sub-components. | [32] |
2.2. ‘Direct’ ES Process of Cellulose Solutions

3. Applied Research Studies for the Functionalization of Cellulose- or Cellulose-Composite-ESNWs
3.1. Chemical Modification of Cellulose-ESNWs
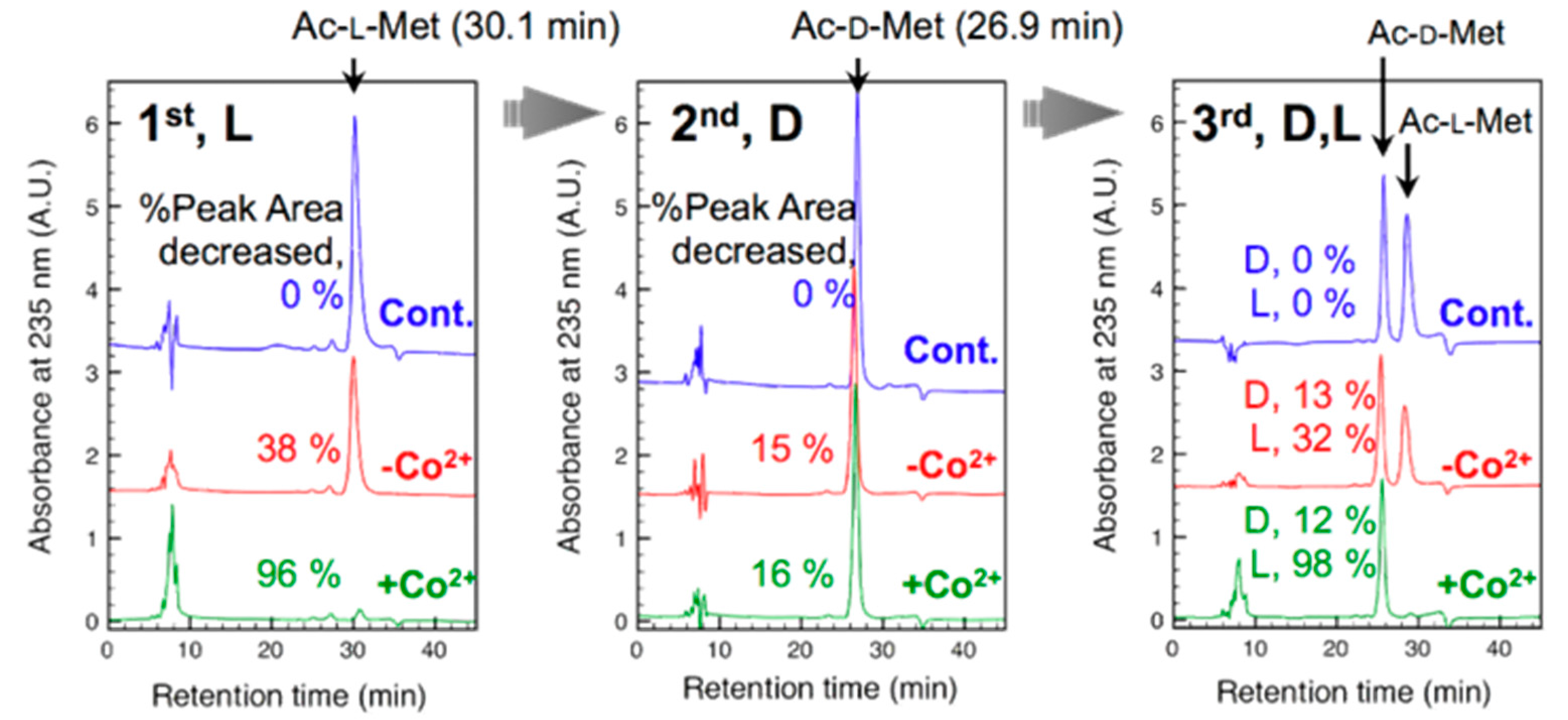
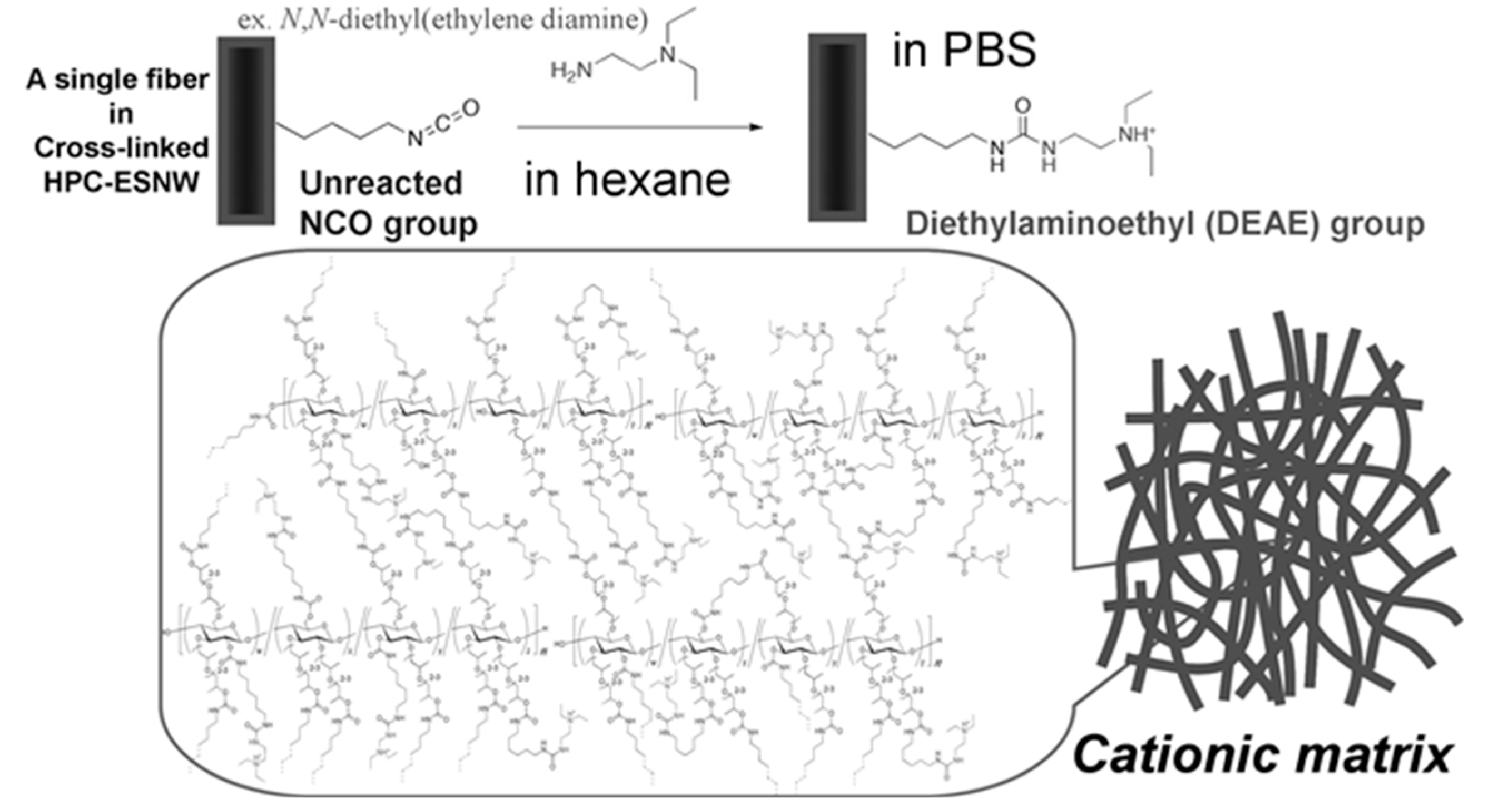
3.2. Cellulose-Composite-ESNWs

4. On a Specific Issue of Cellulose-ESNWs Prepared from TFA Solutions
5. Concluding Remarks
Acknowledgments
Conflicts of Interest
References
- Formhals, A. Nozzle for the Electrical Spinning of Artificial Fibers from Materials such as Cellulose Acetate. Application 1936–96226. patent 2077373, 18 August 1936, 1937. [Google Scholar]
- Reneker, D.H.; Hou, H. Electrospinning. Encycl. Biomater. Biomed. Eng. 2004, 1, 543–550. [Google Scholar]
- Lyons, J.; Ko, F.K. Nanofibers. Encycl. Nanosci. Nanotechnol. 2004, 6, 727–738. [Google Scholar]
- Huang, Z.-M.; Zhang, Y.Z.; Kotaki, M.; Ramakrishna, S. A review on polymer nanofibers by electrospinning and their applications in nanocomposites. Compos. Sci. Technol. 2003, 63, 2223–2253. [Google Scholar] [CrossRef]
- Wang, M.; Meng, G.; Huang, Q.; Qian, Y. Electrospun 1,4-DHAQ-doped cellulose nanofiber films for reusable fluorescence detection of trace Cu2+ and Further for Cr3+. Environ. Sci. Technol. 2012, 46, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Khatri, Z.; Wei, K.; Kim, B.-S.; Kim, I.-S. Effect of deacetylation on wicking behavior of co-electrospun cellulose acetate/polyvinyl alcohol nanofibers blend. Carbohydr. Polym. 2012, 87, 2183–2188. [Google Scholar] [CrossRef]
- Huang, C.; Soenen, S.J.; van, G.E.; Vanham, G.; Rejman, J.; Van, C.S.; Vervaet, C.; Coenye, T.; Verstraelen, H.; Temmerman, M.; et al. Electrospun cellulose acetate phthalate fibers for semen induced anti-HIV vaginal drug delivery. Biomaterials 2012, 33, 962–969. [Google Scholar] [CrossRef] [PubMed]
- Anitha, S.; Brabu, B.; Thiruvadigal, D.J.; Gopalakrishnan, C.; Natarajan, T.S. Optical, bactericidal and water repellent properties of electrospun nano-composite membranes of cellulose acetate and ZnO. Carbohydr. Polym. 2012, 87, 1065–1072. [Google Scholar] [CrossRef]
- Zhou, W.; He, J.; Cui, S.; Gao, W. Preparation of electrospun silk fibroin/Cellulose Acetate blend nanofibers and their applications to heavy metal ions adsorption. Fibers Polym. 2011, 12, 431–437. [Google Scholar] [CrossRef]
- Zhao, D.; Feng, Q.; Lv, L.; Li, J. Fabrication and characterization of cellulose acetate ultrafine fiber containing silver nanoparticles by electrospinning. Adv. Mater. Res. 2011, 337, 116–119. [Google Scholar] [CrossRef]
- Yao, L.; Lee, C.; Kim, J. Electrospun meta-aramid/cellulose acetate and meta-aramid/cellulose composite nanofibers. Fibers Polym. 2011, 12, 197–206. [Google Scholar] [CrossRef]
- Tian, Y.; Wu, M.; Liu, R.; Li, Y.; Wang, D.; Tan, J.; Wu, R.; Huang, Y. Electrospun membrane of cellulose acetate for heavy metal ion adsorption in water treatment. Carbohydr. Polym. 2011, 83, 743–748. [Google Scholar] [CrossRef]
- Stephen, M.; Catherine, N.; Brenda, M.; Andrew, K.; Leslie, P.; Corrine, G. Oxolane-2,5-dione modified electrospun cellulose nanofibers for heavy metals adsorption. J. Hazard. Mater. 2011, 192, 922–927. [Google Scholar] [CrossRef] [PubMed]
- Pittarate, C.; Yoovidhya, T.; Srichumpuang, W.; Intasanta, N.; Wongsasulak, S. Effects of poly(ethylene oxide) and ZnO nanoparticles on the morphology, tensile and thermal properties of cellulose acetate nanocomposite fibrous film. Polym. J. 2011, 43, 978–986. [Google Scholar] [CrossRef]
- Phiriyawirut, M.; Phachamud, T. Suitable electrospinning condition for gallic acid-loaded cellulose acetate fiber. Res. J. Pharm. Biol. Chem. Sci. 2011, 2, 210–220. [Google Scholar]
- Phachamud, T.; Phiriyawirut, M. In vitro cytotoxicity and degradability tests of gallic acid-loaded cellulose acetate electrospun fiber. Res. J. Pharm. Biol. Chem. Sci. 2011, 2, 85–98. [Google Scholar]
- Miyauchi, M.; Miao, J.; Simmons, T.J.; Dordick, J.S.; Linhardt, R.J. Flexible electrospun cellulose fibers as an affinity packing material for the separation of bovine serum albumin. J. Chromatogr. Sep. Tech. 2011, 2. [Google Scholar] [CrossRef]
- Miao, J.; Pangule, R.C.; Paskaleva, E.E.; Hwang, E.E.; Kane, R.S.; Linhardt, R.J.; Dordick, J.S. Lysostaphin-functionalized cellulose fibers with antistaphylococcal activity for wound healing applications. Biomaterials 2011, 32, 9557–9567. [Google Scholar] [CrossRef] [PubMed]
- Magalhaes, W.L.E.; Cao, X.; Ramires, M.A.; Lucia, L.A. Novel all-cellulose composite displaying aligned cellulose nanofibers reinforced with cellulose nanocrystals. Tappi J. 2011, 10, 19–25. [Google Scholar]
- Esteves, M.W.L.; Cao, X.; Lucia, L.A. Cellulose Nanocrystals/Cellulose Core-in-Shell Nanocomposite Assemblies. Langmuir 2009, 25, 13250–13257. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Chen, R.; Zhang, X.; Xiong, J.; Zheng, Y.; Dong, W. Fabrication and characterization of electrospun nanofibers of high DP natural cotton lines cellulose. Fibers Polym. 2011, 12, 345–351. [Google Scholar] [CrossRef]
- Jia, B.; Zhou, J.; Zhang, L. Electrospun nano-fiber mats containing cationic cellulose derivatives and poly (vinyl alcohol) with antibacterial activity. Carbohydr. Res. 2011, 346, 1337–1341. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.-J.; Chen, P.-C.; Huang, F.; Ou, Y.; Chen, M.-R.; Xu, Z.-K. Immobilization of Candida rugosa lipase on electrospun cellulose nanofiber membrane. J. Mol. Catal. B Enzym. 2011, 70, 95–100. [Google Scholar] [CrossRef]
- Filion, T.M.; Kutikov, A.; Song, J. Chemically modified cellulose fibrous meshes for use as tissue engineering scaffolds. Bioorg. Med. Chem. Lett. 2011, 21, 5067–5070. [Google Scholar] [CrossRef] [PubMed]
- Celebioglu, A.; Uyar, T. Electrospun porous cellulose acetate fibers from volatile solvent mixture. Mater. Lett. 2011, 65, 2291–2294. [Google Scholar] [CrossRef]
- Baptista, A.C.; Martins, J.I.; Fortunato, E.; Martins, R.; Borges, J.P.; Ferreira, I. Thin and flexible bio-batteries made of electrospun cellulose-based membranes. Biosens. Bioelectron. 2011, 26, 2742–2745. [Google Scholar] [CrossRef] [PubMed]
- Baek, W.-I.; Pant, H.R.; Nam, K.-T.; Nirmala, R.; Oh, H.-J.; Kim, I.; Kim, H.-Y. Effect of adhesive on the morphology and mechanical properties of electrospun fibrous mat of cellulose acetate. Carbohydr. Res. 2011, 346, 1956–1961. [Google Scholar] [CrossRef] [PubMed]
- Wongsasulak, S.; Patapeejumruswong, M.; Weiss, J.; Supaphol, P.; Yoovidhya, T. Electrospinning of food-grade nanofibers from cellulose acetate and egg albumen blends. J. Food Eng. 2010, 98, 370–376. [Google Scholar] [CrossRef]
- Lisunova, M.; Hildmann, A.; Hatting, B.; Datsyuk, V.; Reich, S. Nanofibres of CA/PAN with high amount of carbon nanotubes by core-shell electrospinning. Compos. Sci. Technol. 2010, 70, 1584–1588. [Google Scholar] [CrossRef]
- Haas, D.; Heinrich, S.; Greil, P. Solvent control of cellulose acetate nanofibre felt structure produced by electrospinning. J. Mater. Sci. 2010, 45, 1299–1306. [Google Scholar] [CrossRef]
- Dixit, V.; Tewari, J.; Obendorf, S.K. Fungal Growth Inhibition of Regenerated Cellulose Nanofibrous Membranes Containing Quillaja Saponin. Arch. Environ. Contam. Toxicol. 2010, 59, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Cozza, E.S.; Monticelli, O.; Marsano, E. Electrospinning: A Novel Method to Incorporate POSS into a Polymer Matrix. Macromol. Mater. Eng. 2010, 295, 791–795. [Google Scholar] [CrossRef]
- Formhals, A. Electrical Spinning of Fibers such as Those Produced from Cellulose Acetate. Application 1936–88429. patent 2116942, 1 July 1936, 1938. [Google Scholar]
- Liu, H.; Hsieh, Y.-L. Ultrafine fibrous cellulose membranes from electrospinning of cellulose acetate. J. Polym. Sci. B Polyme Phys. 2002, 40, 2119–2129. [Google Scholar] [CrossRef]
- Nartker, S.; Drzal, L.T. Electrospun cellulose nitrate nanofibers. J. Nanosci. Nanotechnol. 2010, 10, 5810–5813. [Google Scholar] [CrossRef] [PubMed]
- Ohkawa, K.; Cha, D.; Kim, H.; Nishida, A.; Yamamoto, H. Electrospinning of chitosan. Macromol. Rapid Commun. 2004, 25, 1600–1605. [Google Scholar] [CrossRef]
- Ohkawa, K.; Minato, K.-I.; Kumagai, G.; Hayashi, S.; Yamamoto, H. Chitosan nanofiber. Biomacromolecules 2006, 7, 3291–3294. [Google Scholar] [CrossRef] [PubMed]
- Ohkawa, K.; Yamamoto, H. Electro-spun Finer Finers of Cellulose and Chitosan. Jpn. Patent 2008–308780; 4992024, 13 June 2007, 2012. [Google Scholar]
- Ohkawa, K. Electrospinning of natural and bio-related polymeric materials. Sen’i Gakkaishi 2008, 64, 36–44. [Google Scholar] [CrossRef]
- Kulpinski, P. Cellulose nanofibers prepared by the N-methylmorpholine-N-oxide method. J. Appl. Polym. Sci. 2005, 98, 1855–1859. [Google Scholar] [CrossRef]
- Freire, M.G.; Teles, A.R.R.; Ferreira, R.A.S.; Carlos, L.D.; Lopes-da-Silva, J.A.; Coutinho, J.A.P. Electrospun nanosized cellulose fibers using ionic liquids at room temperature. Green Chem. 2011, 13, 3173–3180. [Google Scholar] [CrossRef]
- Quan, S.-L.; Kang, S.-G.; Chin, I.-J. Characterization of cellulose fibers electrospun using ionic liquid. Cellulose 2010, 17, 223–230. [Google Scholar] [CrossRef]
- Park, T.-J.; Jung, Y.J.; Choi, S.-W.; Park, H.; Kim, H.; Kim, E.; Lee, S.H.; Kim, J.H. Native chitosan/cellulose composite fibers from an ionic liquid via electrospinning. Macromol. Res. 2011, 19, 213–215. [Google Scholar] [CrossRef]
- Ohkawa, K.; Hayashi, S.; Nishida, A.; Yamamoto, H.; Ducreux, J. Preparation of pure cellulose nanofiber via electrospinning. Text. Res. J. 2009, 79, 1396–1401. [Google Scholar] [CrossRef]
- Devarayan, K.; Periasamy, V.; Nishida, A.; Araki, J.; Ohkawa, K. Preparation of electrospun non-woven fabrics from cationic derivative of hydroxypropyl cellulose and immobilization of enzyme. Polym. Prepr. Jpn. 2010, 59, 4862. [Google Scholar]
- Chibata, I.; Tosa, T.; Sato, T.; Mori, T. Production of L-amino acids by aminoacylase adsorbed on DEAE-Sephadex. Methods Enzymol. 1976, 44, 746–759. [Google Scholar] [PubMed]
- Hayashi, S.; Ohkawa, K.; Yamamoto, H.; Yamaguchi, M.; Kimoto, S.; Kurata, S.; Shinji, H. Calcium phosphate crystallization on electrospun cellulose non-woven fabrics containing synthetic phosphorylated polypeptides. Macromol. Mater. Eng. 2009, 294, 315–322. [Google Scholar] [CrossRef]
- Ohkawa, K.; Hayashi, S.; Kameyama, N.; Yamamoto, H.; Yamaguchi, M.; Kimoto, S.; Kurata, S.; Shinji, H. Synthesis of collagen-like sequential polypeptides containing O-phospho-L-hydroxyproline and preparation of electrospun composite fibers for possible dental application. Macromol. Biosci. 2009, 9, 79–92. [Google Scholar] [CrossRef] [PubMed]
- Ohkawa, K. Preparation of natural and bio-related polymeric micro-fiber materials by electrospinning. In Kinosei Sen’i no Saishin Gijutsu; Shirai, H., Ed.; CMC Publishing: Tokyo, Japan, 2009; Chapter 4; pp. 37–50. [Google Scholar]
- James, R.; Toti, U.S.; Laurencin, C.T.; Kumbar, S.G. Electrospun nanofibrous scaffolds for engineering soft connective tissues. In Biomedical Nanotechnology; Hurst, S.J., Ed.; Humana Press: New York, NY, USA, 2011; Chapter 16; pp. 243–258. [Google Scholar]
- Min, B.M.; Lee, S.W.; Lim, J.N.; You, Y.; Lee, T.S.; Kang, P.H.; Park, W.H. Chitin and chitosan nanofibers: Electrospinning of chitin and deacetylation of chitin nanofibers. Polymer 2004, 45, 7137–7142. [Google Scholar] [CrossRef]
- Nada, A.A.; James, R.; Shelke, N.B.; Harmon, M.D.; Awad, H.M.; Nagarale, R.K.; Kumbar, S.G. A smart methodology to fabricate electrospun chitosan nanofiber matrices for regenerative engineering applications. Poylm. Adv. Technol. 2014, 35, 507–515. [Google Scholar] [CrossRef]
- Ohkawa, K.; Nishibayashi, M.; Devarayan, K.; Hachisu, M.; Araki, J. Synthesis of peptide-cellulose conjugate mediated by a soluble cellulose derivative having β-Ala esters. Int. J. Biol. Macromol. 2013, 53, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Devarayan, K.; Hayashi, T.; Hachisu, M.; Araki, J.; Ohkawa, K. Correlations between steric/thermochemical parameters and O-/N-acylation reactions of cellulose. Carbohydr. Polym. 2013, 94, 468–478. [Google Scholar] [CrossRef] [PubMed]
- Devarayan, K.; Hachisu, M.; Araki, J.; Ohkawa, K. Synthesis of peptide-cellulose conjugate mediated by a soluble cellulose derivative having β-Ala esters (II): Conjugates with O-phospho-L-serine-containing peptides. Cellulose 2013, 20, 365–378. [Google Scholar] [CrossRef]
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ohkawa, K. Nanofibers of Cellulose and Its Derivatives Fabricated Using Direct Electrospinning. Molecules 2015, 20, 9139-9154. https://doi.org/10.3390/molecules20059139
Ohkawa K. Nanofibers of Cellulose and Its Derivatives Fabricated Using Direct Electrospinning. Molecules. 2015; 20(5):9139-9154. https://doi.org/10.3390/molecules20059139
Chicago/Turabian StyleOhkawa, Kousaku. 2015. "Nanofibers of Cellulose and Its Derivatives Fabricated Using Direct Electrospinning" Molecules 20, no. 5: 9139-9154. https://doi.org/10.3390/molecules20059139
APA StyleOhkawa, K. (2015). Nanofibers of Cellulose and Its Derivatives Fabricated Using Direct Electrospinning. Molecules, 20(5), 9139-9154. https://doi.org/10.3390/molecules20059139





