Effects of Two Different Rhodiola rosea Extracts on Primary Human Visceral Adipocytes
Abstract
:1. Introduction
2. Results and Discussion
2.1. Cell Viability
| Dose (µg/mL) | P10 | P20 | A7 | |||
|---|---|---|---|---|---|---|
| RS | RR | RS | RR | RS | RR | |
| 5 | 91.83 A ± 7.55 | 91.51 A ± 7.58 | 91.34 A ± 3.26 | 89.41 A ± 3.42 | 96.32 A ± 1.71 | 96.74 A ± 1.83 |
| 10 | 80.26 B ± 6.84 | 80.17 B ± 7.01 | 78.52 B ± 2.43 | 79.02 B ± 3.13 | 95.81 A ± 2.05 | 96.29 A ± 1.92 |
| 30 | 76.75 C ± 6.43 | 79.95 B ± 6.78 | 66.85 C ± 2.88 | 67.77 C ± 3.33 | 96.09 A ± 2.25 | 93.84 A ± 2.25 |
| 70 | 53.58 D ± 7.15 | 55.30 C ± 4.23 | 36.26 D ± 2.06 | 42.28 D ± 4.33 | 89.81 B ± 1.91 | 84.34 B ± 3.06 |
2.1.1. RR and RS Extracts Decrease Triglyceride Accumulation
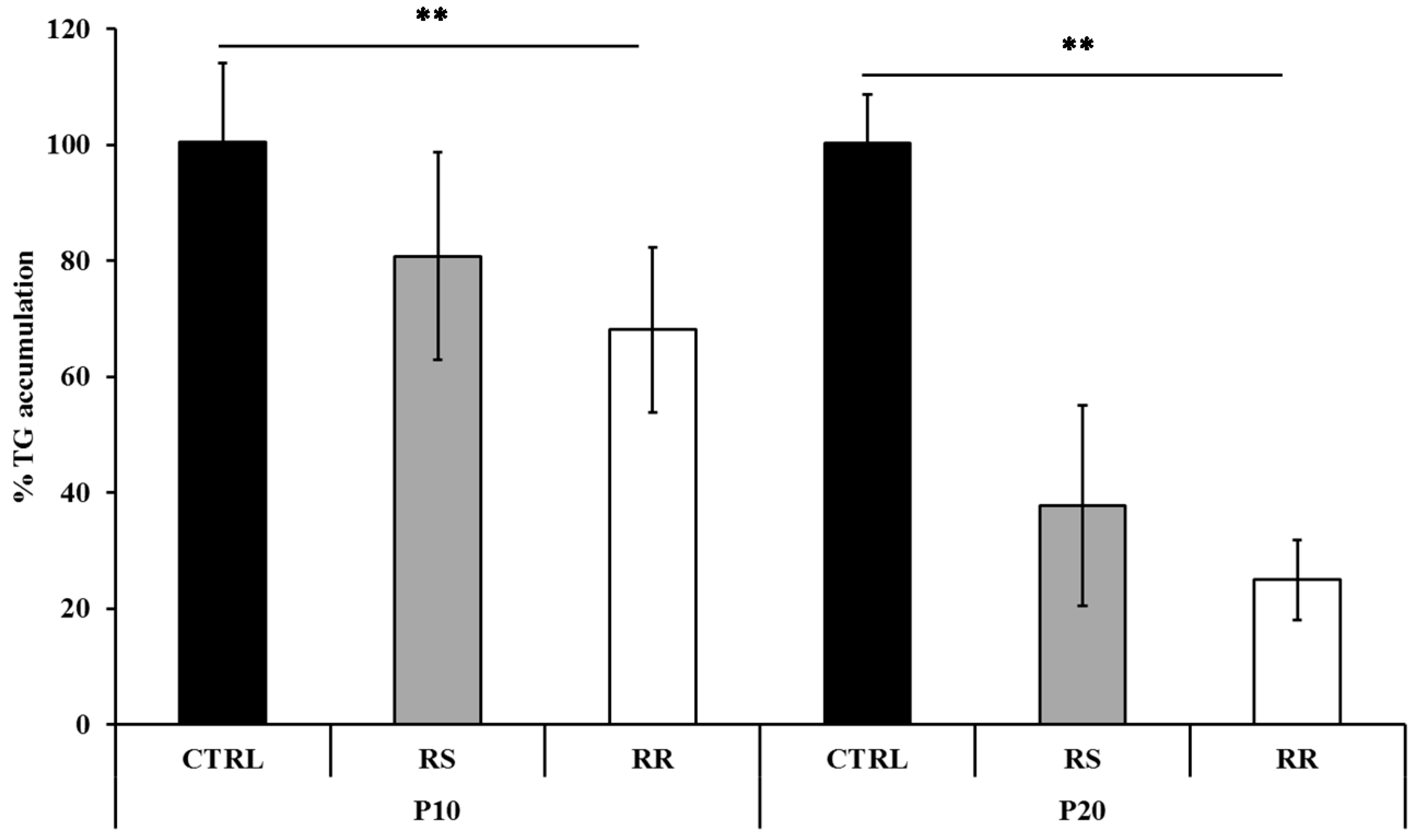
2.1.2. RR and RS Extracts Increase Glycerol Release
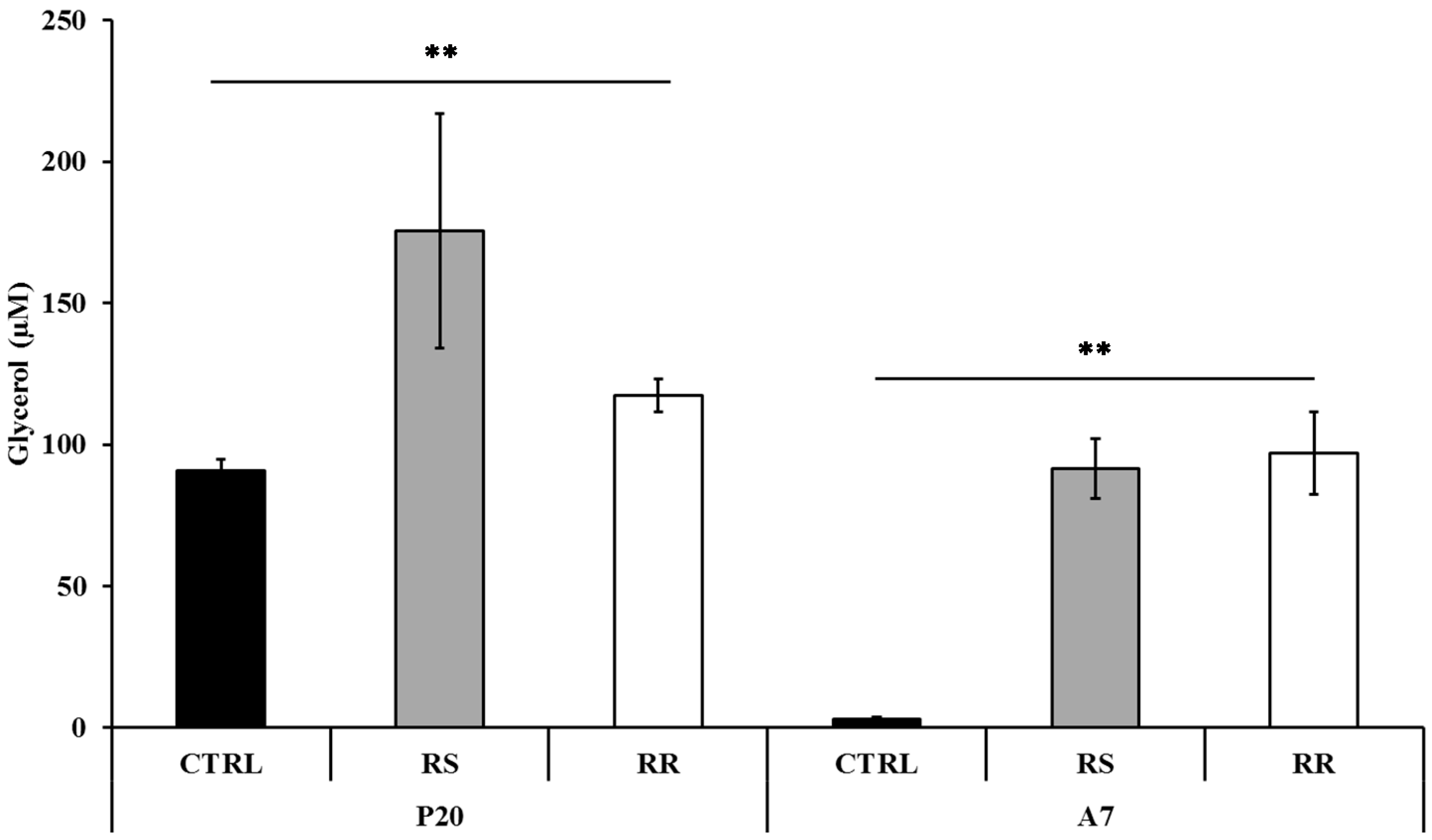
2.1.3. Effect of RR and RS on Apoptosis
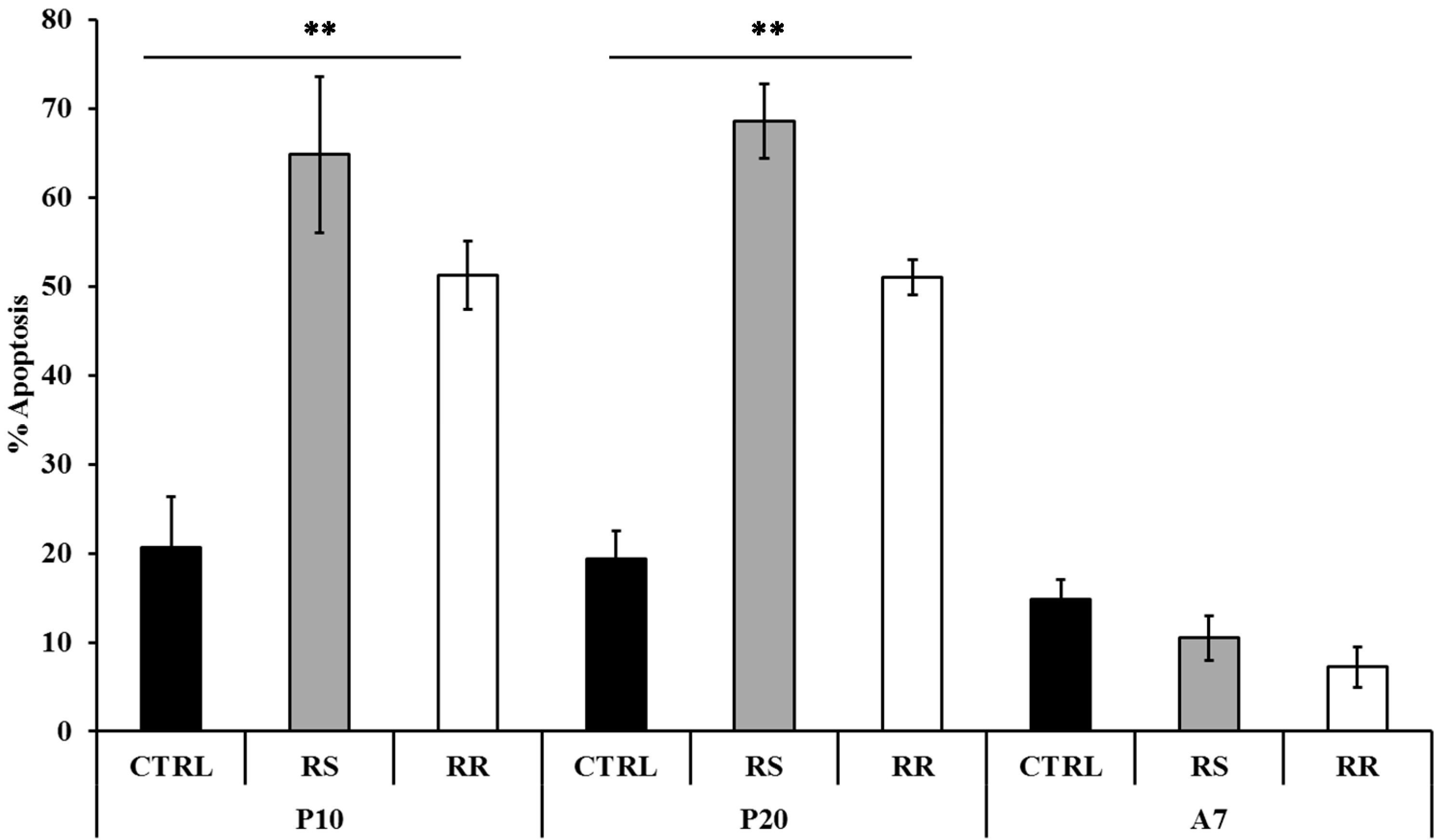
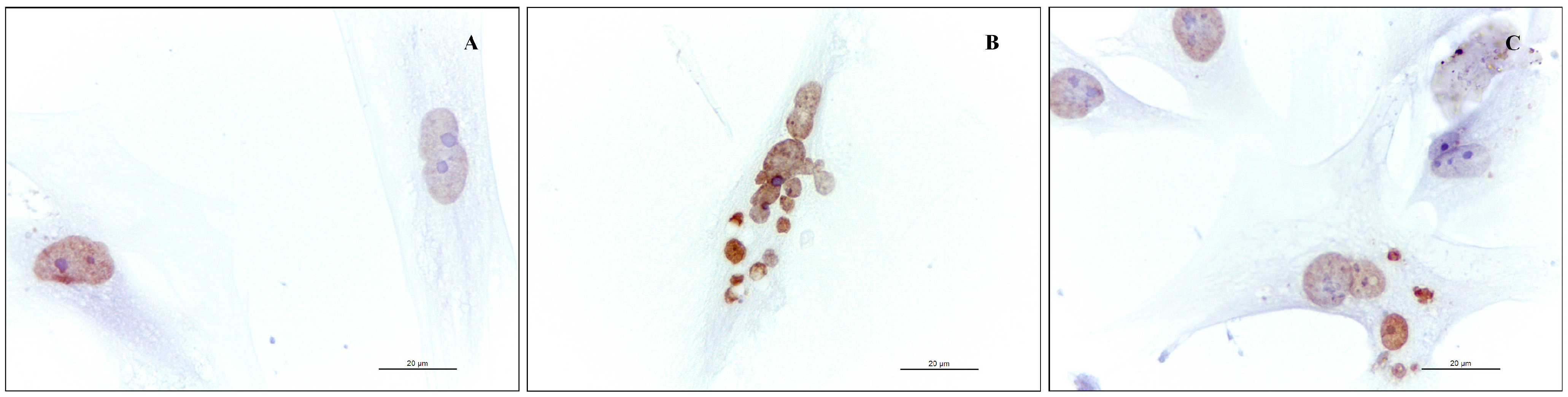
2.1.4. Effects of RR and RS Extracts on the Level of Expression of Adipogenesis-Associated Genes
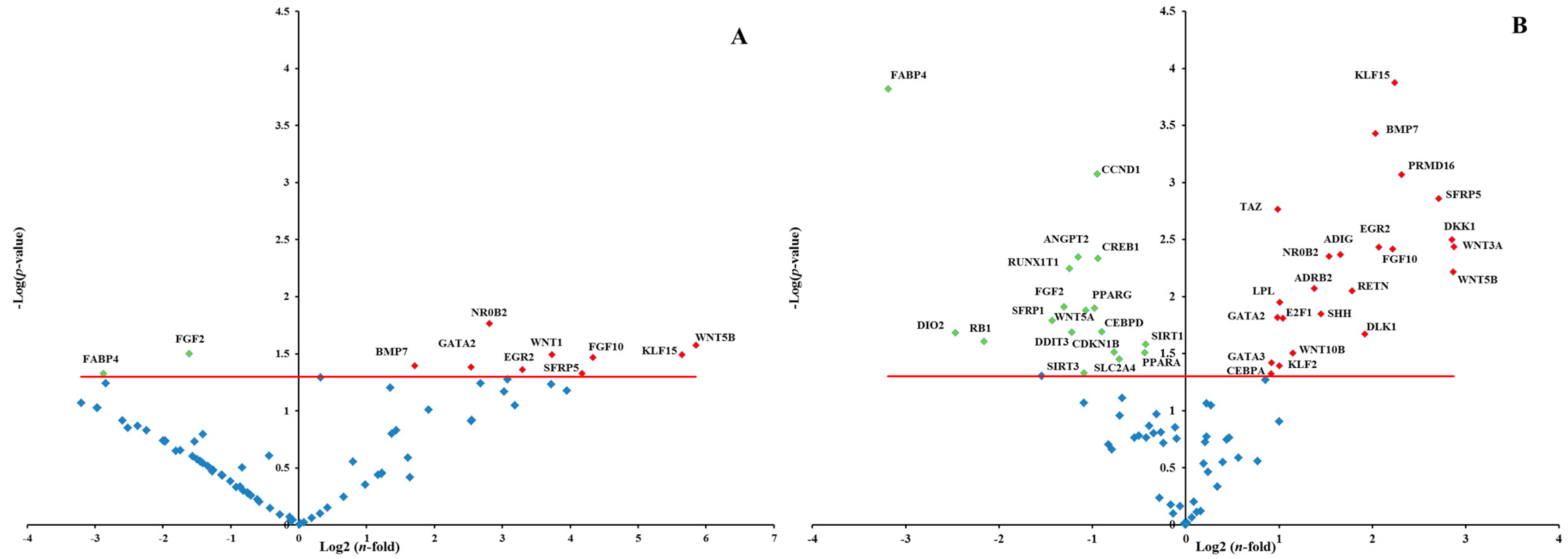
2.2. Discussion
3. Experimental Section
3.1. Materials
3.2. Cell Culture and Cell Treatment
3.2.1. Cell Viability
3.2.2. Oil Red O Staining and Measurement of Lipid Accumulation
3.2.3. Lipolysis Assay
3.2.4. Apoptosis Assay
3.2.5. TUNEL Assay
3.2.6. RNA Extraction and Adipogenesis PCR Array
3.3. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Abbreviations
| ACACB | acetyl-CoA carboxylase beta |
| ADIG | adipogenin |
| ADIPOQ | adiponectin |
| ADRB2 | adrenoceptor beta 2 |
| AGT | angiotensinogen |
| ANGPT2 | angiopoietin 2 |
| AXIN1 | axin 1 |
| BMP2 | bone morphogenetic protein 2 |
| BMP4 | bone morphogenetic protein 4 |
| BMP7 | bone morphogenetic protein 7 |
| CCND1 | cyclin D1 |
| CDK4 | cyclin-dependent kinase 4 |
| CDKN1A | cyclin-dependent kinase inhibitor 1A (p21, Cip1) |
| CDKN1B | cyclin-dependent kinase inhibitor 1B (p27, Kip1) |
| CEBPA | CCAAT/enhancer binding protein (C/EBP) alpha |
| CEBPB | CCAAT/enhancer binding protein (C/EBP) beta |
| CEBPD | CCAAT/enhancer binding protein (C/EBP) delta |
| CFD | complement factor D (adipsin) |
| CREB1 | cAMP responsive element binding protein 1 |
| DDIT3 | DNA-damage-inducible transcript 3 |
| DIO2 | deiodinase, iodothyronine, type II |
| DKK1 | dickkopf WNT signaling pathway inhibitor 1 |
| DLK1 | delta-like 1 homolog (Drosophila) |
| E2F1 | E2F transcription factor 1 |
| EGR2 | early growth response 2 |
| FABP4 | fatty acid binding protein 4, adipocyte |
| FASN | fatty acid synthase |
| FGF1 | fibroblast growth factor 1 (acidic) |
| FGF10 | fibroblast growth factor 10 |
| FGF2 | fibroblast growth factor 2 (basic) |
| FOXC2 | forkhead box C2 (MFH-1, mesenchyme forkhead 1) |
| FOXO1 | forkhead box O1 |
| GATA2 | GATA binding protein 2 |
| GATA3 | GATA binding protein 3 |
| HES1 | hes family bHLH transcription factor 1 |
| INSR | insulin receptor |
| IRS1 | insulin receptor substrate 1 |
| IRS2 | insulin receptor substrate 2 |
| JUN | jun proto-oncogene |
| KLF15 | Kruppel-like factor 15 |
| KLF2 | Kruppel-like factor 2 |
| KLF3 | Kruppel-like factor 3 |
| KLF4 | Kruppel-like factor 4 |
| LEP | leptin |
| LIPE | lipase, hormone-sensitive |
| LMNA | lamin A/C |
| LPL | lipoprotein lipase |
| LRP5 | low density lipoprotein receptor-related protein 5 |
| MAPK14 | mitogen-activated protein kinase 14 |
| NCOA2 | nuclear receptor coactivator 2 |
| NCOR2 | nuclear receptor corepressor 2 |
| NR0B2 | nuclear receptor subfamily 0, group B, member 2 |
| NR1H3 | nuclear receptor subfamily 1, group H, member 3 |
| NRF1 | nuclear respiratory factor 1 |
| PPARA | peroxisome proliferator-activated receptor alpha |
| PPARD | peroxisome proliferator-activated receptor delta |
| PPARG | peroxisome proliferator-activated receptor gamma |
| PPARGC1A | peroxisome proliferator-activated receptor gamma, coactivator 1 alpha |
| PPARGC1B | peroxisome proliferator-activated receptor gamma, coactivator 1 beta |
| PRDM16 | PR domain containing 16 |
| RB1 | retinoblastoma 1 |
| RETN | resistin |
| RUNX1T1 | runt-related transcription factor 1 |
| RXRA | retinoid X receptor, alpha |
| SFRP1 | secreted frizzled-related protein 1 |
| SFRP5 | secreted frizzled-related protein 5 |
| SHH | sonic hedgehog |
| SIRT1 | sirtuin 1 |
| SIRT2 | sirtuin 2 |
| SIRT3 | sirtuin 3 |
| SLC2A4 | solute carrier family 2 (facilitated glucose transporter), member 4 |
| SRC | v-src avian sarcoma (Schmidt-Ruppin A-2) viral oncogene homolog |
| SREBF1 | sterol regulatory element binding transcription factor 1 |
| TAZ | tafazzin |
| TCF7L2 | transcription factor 7-like 2 (T-cell specific, HMG-box) |
| TSC22D3 | TSC22 domain family, member 3 |
| TWIST1 | twist family bHLH transcription factor 1 |
| UCP1 | uncoupling protein 1 (mitochondrial proton carrier) |
| VDR | vitamin D (1,25- dihydroxyvitamin D3) receptor |
| WNT1 | wingless-type MMTV integration site family, member 1 |
| WNT10B | wingless-type MMTV integration site family, member 10B |
| WNT3A | wingless-type MMTV integration site family, member 3A |
| WNT5A | wingless-type MMTV integration site family, member 5A |
| WNT5B | wingless-type MMTV integration site family, member 5B |
Conflicts of Interest
References
- Functional Foods and Dietary Supplements: Processing Effects and Health Benefits; Noomhorm, A.; Ahmad, I.; Anal, A.K. (Eds.) Wiley-Blackwell: Oxford, UK, 2014.
- Colitti, M.; Gaspardo, B.; Della Pria, A.; Scaini, C.; Stefanon, B. Transcriptome modification of white blood cells after dietary administration of curcumin and non-steroidal anti-inflammatory drug in osteoarthritic affected dogs. Vet. Immunol. Immunopathol. 2012, 147, 136–146. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.W. Possible anti-obesity therapeutics from nature-a review. Phytochemistry 2010, 71, 1625–1641. [Google Scholar] [CrossRef] [PubMed]
- Colitti, M.; Grasso, S. Nutraceuticals and regulation of adipocyte life: Premises or promises. Biofactors 2014, 40, 398–418. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.T.; Park, I.J.; Shin, J.I.; Lee, Y.K.; Lee, S.K.; Baik, H.W.; Ha, J.; Park, O.J. Genistein, EGCG, and capsaicin inhibit adipocyte differentiation process via activating AMP-activated protein kinase. Biochem. Biophys. Res. Commun. 2005, 338, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Song, S.J.; Choi, S.; Park, T. Decaffeinated green coffee bean extract attenuates diet-induced obesity and insulin resistance in mice. Evid. Based Complement. Altern. Med. 2014, 2014, 718379. [Google Scholar]
- Takahashi, T.; Tabuchi, T.; Tamaki, Y.; Kosaka, K.; Takikawa, Y.; Satoh, T. Carnosic acid and carnosol inhibit adipocyte differentiation in mouse 3T3-L1 cells through induction of phase2 enzymes and activation of glutathione metabolism. Biochem. Biophys. Res. Commun. 2009, 382, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Stefanon, B.; Pomari, E.; Colitti, M. Effects of Rosmarinus officinalis extract on human primary omental preadipocytes and adipocytes. Exp. Biol. Med. (Maywood) 2015. [Google Scholar] [CrossRef]
- Shikov, A.N.; Pozharitskaya, O.N.; Makarov, V.G.; Wagner, H.; Verpoorte, R.; Heinrich, M. Medicinal plants of the Russian Pharmacopoeia; their history and applications. J. Ethnopharmacol. 2014, 154, 481–536. [Google Scholar] [CrossRef] [PubMed]
- Hung, S.K.; Perry, R.; Ernst, E. The effectiveness and efficacy of Rhodiola rosea L.: A systematic review of randomized clinical trials. Phytomedicine 2011, 18, 235–244. [Google Scholar]
- Brown, R.P.; Gerbarg, P.L.; Graham, B. The Rhodiola Revolution: Transform Your Health with the Herbal Breakthrough of the 21st Century; Rodale Press: Emmaus, PA, USA, 2005. [Google Scholar]
- Morgan, M.; Bone, K. Rhodiola rosea-Rhodiola. MediHerb. Newsl. 2005, 47, 1–4. [Google Scholar]
- Brown, R.P.; Gerbarg, P.L.; Ramazanov, Z. Rhodiola rosea: A phytomedicinal overview. Herbalgram 2002, 56, 40–52. [Google Scholar]
- Kelly, G.S. Rhodiola rosea: A possible plant adaptogen. Altern. Med. Rev. 2001, 6, 293–302. [Google Scholar] [PubMed]
- Kur’yanov, A.A.; Bondarenko, L.T.; Kurkin, V.A.; Zapesochnaya, G.G.; Dubichev, A.A.; Vorontsov, E.D. Determination of the biologically active components of the rhizomes of Rhodiola rosea. Chem. Nat. Comp. 1991, 27, 276–279. [Google Scholar] [CrossRef]
- Ming, D.S.; Hillhouse, B.J.; Guns, E.S.; Eberding, A.; Xie, S.; Vimalanathan, S.; Towers, G.H. Bioactive compounds from Rhodiola rosea (Crassulaceae). Phytother. Res. 2005, 19, 740–743. [Google Scholar] [CrossRef] [PubMed]
- Russian National Pharmacopoeia. PA 42–2126–83,Liquid Extract of Rhodiola rosea Root and Rhizome; The Russian Federation Ministry of Health and Medical Industry: Moscow, Russia, 1983.
- Chang, Y.W.; Yao, H.T.; Hsieh, S.H.; Lu, T.J.; Yeh, T.K. Quantitative determination of salidroside in rat plasma by on-line solid-phase extraction integrated with high-performance liquid chromatography/electrospray ionization tandem mass spectrometry. J. Chromatogr. B. Anal. Technol. Biomed. Life Sci. 2007, 857, 164–169. [Google Scholar] [CrossRef]
- Panossian, A.; Wikman, G.; Sarris, J. Rosenroot (Rhodiola rosea): Traditional use, chemical composition, pharmacology and clinical efficacy. Phytomedicine 2010, 17, 481–493. [Google Scholar] [CrossRef] [PubMed]
- Ishaque, S.; Shamseer, L.; Bukutu, C.; Vohra, S. Rhodiola rosea for physical and mental fatigue: A systematic review. BMC Complement. Altern. Med. 2012, 12, 70. [Google Scholar] [CrossRef] [PubMed]
- De Bock, K.; Eijnde, B.O.; Ramaekers, M.; Hespel, P. Acute Rhodiola rosea intake can improve endurance exercise performance. Int. J. Sport Nutr. Exerc. Metab. 2004, 14, 298–307. [Google Scholar] [PubMed]
- Chen, C.H.; Chan, H.C.; Chu, Y.T.; Ho, H.Y.; Chen, P.Y.; Lee, T.H.; Lee, C.K. Antioxidant activity of some plant extracts towards xanthine oxidase, lipoxygenase and tyrosinase. Molecules 2009, 14, 2947–2958. [Google Scholar] [CrossRef] [PubMed]
- Calcabrini, C.; de Bellis, R.; Mancini, U.; Cucchiarini, L.; Potenza, L.; de Sanctis, R.; Patrone, V.; Scesa, C.; Dachà, M. Rhodiola rosea ability to enrich cellular antioxidant defenses of cultured human keratinocytes. Arch. Dermatol. Res. 2010, 302, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Mao, G.X.; Wang, Y.; Qiu, Q.; Deng, H.B.; Yuan, L.G.; Li, R.G.; Song, D.Q.; Li, Y.Y.; Li, D.D.; Wang, Z. Salidroside protects human fibroblast cells from premature senescence induced by H(2)O(2) partly through modulating oxidative status. Mech. Ageing Dev. 2010, 131, 723–731. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Lin, S.; Yu, D.; Qiu, S.; Zhang, X.; Mei, R. A preliminary study: the anti-proliferation effect of salidroside on different human cancer cell lines. Cell Biol. Toxicol. 2010, 26, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Wang, Z.; Zheng, Q.; Zhang, H. Salidroside inhibits migration and invasion of human fibrosarcoma HT1080 cells. Phytomedicine 2012, 19, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Darbinyan, V.; Aslanyan, G.; Amroyan, E.; Gabrielyan, E.; Malmström, C.; Panossian, A. Clinical trial of Rhodiola rosea L. extract SHR-5 in the treatment of mild to moderate depression. Nord J. Psychiatry 2007, 61, 343–348. [Google Scholar]
- Ross, S.M. Rhodiola rosea (SHR-5), Part 2: A standardized extract of Rhodiola rosea is shown to be effective in the treatment of mild to moderate depression. Holist. Nurs. Pract. 2014, 28, 217–221. [Google Scholar] [CrossRef]
- Yu, S.; Shen, Y.; Liu, J.; Ding, F. Involvement of ERK1/2 pathway in neuroprotection by salidroside against hydrogen peroxide-induced apoptotic cell death. J. Mol. Neurosci. 2010, 40, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.; Lin, R.; Li, Z.; Mao, J.; Chen, L. Effects of Salidroside on cobalt chloride-induced hypoxia damage and mTOR signaling repression in PC12 cells. Biol. Pharm. Bull. 2014, 37, 1199–1206. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Zhou, H.; Jin, Z.; Bi, S.; Yang, X.; Yi, D.; Liu, W. Cardioprotection of salidroside from ischemia/reperfusion injury by increasing N-acetylglucosamine linkage to cellular proteins. Eur. J. Pharmacol. 2009, 613, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.Z.; Chen, L.J.; Lee, W.J.; Chen, M.F.; Jung, L.H.; Cheng, J.T. Increase of myocardial performance by Rhodiola-ethanol extract in diabetic rats. J. Ethnopharmacol. 2012, 144, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Song, E.K.; Kim, J.H.; Kim, J.S.; Cho, H.; Nan, J.X.; Sohn, D.H.; Ko, G.I.; Oh, H.; Kim, Y.C. Hepatoprotective phenolic constituents of Rhodiola sachalinensis on tacrine-induced cytotoxicity in Hep G2 cells. Phytother. Res. 2003, 17, 563–365. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.L.; Lian, L.H.; Jiang, Y.Z.; Nan, J.X. Hepatoprotective effects of salidroside on fulminant hepatic failure induced by d-galactosamine and lipopolysaccharide in mice. J. Pharm. Pharmacol. 2009, 61, 1375–1382. [Google Scholar] [CrossRef] [PubMed]
- Skopńska-Rózewska, E.; Wójcik, R.; Siwicki, A.K.; Sommer, E.; Wasiutyński, A.; Furmanowa, M.; Malinowski, M.; Mazurkiewicz, M. The effect of Rhodiola quadrifida extracts on cellular immunity in mice and rats. Pol. J. Vet. Sci. 2008, 11, 105–111. [Google Scholar] [PubMed]
- Siwicki, A.K.; Skopińska-Różewska, E.; Wasiutyński, A.; Wójcik, R.; Zdanowski, R.; Sommer, E.; Buchwald, W.; Furmanowa, M.; Bakuła, T.; Stankiewicz, W. The effect of Rhodiola kirilowii extracts on pigs’ blood leukocytes metabolic (RBA) and proliferative (LPS) activity, and on the bacterial infection and blood leukocytes number in mice. Cent. Eur. J. Immunol. 2012, 37, 145–150. [Google Scholar]
- Mishra, K.P.; Ganju, L.; Singh, S.B. Anti-cellular and immunomodulatory potential of aqueous extract of Rhodiola imbricata rhizome. Immunopharmacol. Immunotoxicol. 2012, 34, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Currier, A.; Ampong-Nyarko, K. Rhodiola Rosea; CRC Press: Boca Raton, FL, USA, 2015; p. 196. [Google Scholar]
- Lee, O.H.; Kwon, Y.I.; Apostolidis, E.; Shetty, K.; Kim, Y.C. Rhodiola-induced inhibition of adipogenesis involves antioxidant enzyme response associated with pentose phosphate pathway. Phytother. Res. 2011, 25, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Verpeut, J.L.; Walters, A.L.; Bello, N.T. Citrus aurantium and Rhodiola rosea in combination reduce visceral white adipose tissue and increase hypothalamic norepinephrine in a rat model of diet-induced obesity. Nutr. Res. 2013, 33, 503–512. [Google Scholar] [CrossRef] [PubMed]
- Peters, J.M.; Lee, S.S.; Li, W.; Ward, J.M.; Gavrilova, O.; Everett, C.; Reitman, M.L.; Hudson, L.D.; Gonzalez, F.J. Growth, adipose, brain, and skin alterations resulting from targeted disruption of the mouse peroxisome proliferator-activated receptor β(δ). Mol. Cell Biol. 2000, 20, 5119–5128. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.X.; Lee, C.H.; Tiep, S.; Yu, R.T.; Ham, J.; Kang, H.; Evans, R.M. Peroxisome-proliferator-activated receptor delta activates fat metabolism to prevent obesity. Cell 2003, 113, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Barish, G.D.; Narkar, V.A.; Evans, R.M. PPAR delta: A dagger in the heart of the metabolic syndrome. J. Clin. Investig. 2006, 116, 590–597. [Google Scholar] [CrossRef] [PubMed]
- El-Houri, R.B.; Kotowska, D.; Olsen, L.C.; Bhattacharya, S.; Christensen, L.P.; Grevsen, K.; Oksbjerg, N.; Færgeman, N.; Kristiansen, K.; Christensen, K.B. Screening for bioactive metabolites in plant extracts modulating glucose uptake and fat accumulation. Evid. Based Complement Altern. Med. 2014, 2014, 156398. [Google Scholar] [CrossRef]
- Abidoff, M.T.; Nelubov, M. 1997 Russian anti-stress herbal supplement promotes weight loss, reduces plasma perilipins and cortisol levels in obese patients: Double-blind placebo controlled clinical study. In Proceedings of Stress and Weight Management at Russian Perestroika/Healthy Diet, Republic of Dagestan, North Caucasian, Russia, 1–3 June 1997.
- Ramazanov, Z.; Mar Bernal Suarez, M.; Anderson, N. New Secret of Effective Natural Stress Weight Management Using Rhodiola Rosea and Rhododendron Cauca: Georgian Secres of Longevity, Stress and Natural; ATN/Safe Goods Publishing: East Canaan, CT, USA, 1999. [Google Scholar]
- Van Diermen, D.; Marston, A.; Bravo, J.; Reist, M.; Carrupt, P.A.; Hostettmann, K. Monoamine oxidase inhibition by Rhodiola rosea L. roots. J. Ethnopharmacol. 2009, 122, 397–401. [Google Scholar] [CrossRef]
- Lefterova, M.I.; Lazar, M.A. New developments in adipogenesis. Trends Endocrinol. Metab. 2009, 20, 107–114. [Google Scholar] [CrossRef]
- Panossian, A.; Hamm, R.; Wikman, G.; Efferth, T. Mechanism of action of Rhodiola, salidroside, tyrosol and triandrin in isolated neuroglial cells: An interactive pathway analysis of the downstream effects using RNA microarray data. Phytomedicine 2014, 21, 1325–1348. [Google Scholar] [CrossRef] [PubMed]
- Germano, C.; Ramanazov, Z. Artic Root (Rhodiola Rosea): The Powerful New Ginseng Alternative; Kensington Press: New York, NY, USA, 1999. [Google Scholar]
- Wang, J.; Rong, X.; Li, W.; Yang, Y.; Yamahara, J.; Li, Y. Rhodiola crenulata root ameliorates derangements of glucose and lipid metabolism in a rat model of the metabolic syndrome and type 2 diabetes. J. Ethnopharmacol. 2012, 142, 782–788. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.C.; Li, W.M.; Guo, R.; Xu, Y.W. Salidroside decreases atherosclerotic plaque formation in low density lipoprotein receptor-deficient mice. Evid. Based Complement. Altern. Med. 2012, 2012, 607508. [Google Scholar]
- Monforte, M.T.; Miceli, N.; Taviano, M.F.; Trovato, A. Hypolipidemic activity of Rhodiola rosea L. root extract. Pharmac. Mag. 2006, 2, 35–41. [Google Scholar]
- Wang, S.H.; Wang, W.J.; Wang, X.F.; Chen, W.H. Effects of salidroside on carbohydrate metabolism and differentiation of 3T3-L1 adipocytes. Zhong Xi Yi Jie He Xue Bao 2004, 2, 193–195. [Google Scholar] [CrossRef] [PubMed]
- Cifani, C.; Micioni Di B, M.V.; Vitale, G.; Ruggieri, V.; Ciccocioppo, R.; Massi, M. Effect of salidroside, active principle of Rhodiola rosea extract, on binge eating. Physiol. Behav. 2010, 101, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Tong, Q.; Tsai, J.; Hotamisligil, G.S. GATA transcription factors and fat cell formation. Drug News Perspect. 2003, 16, 585–588. [Google Scholar] [CrossRef] [PubMed]
- Tong, Q.; Tsai, J.; Tan, G.; Dalgin, G.; Hotamisligil, G.S. Interaction between GATA and the C/EBP family of transcription factors is critical in GATA-mediated suppression of adipocyte differentiation. Mol. Cell. Biol. 2005, 25, 706–715. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Fahmy, H.; Zjawiony, J.K.; Davies, G.E. Inhibitory effect and transcriptional impact of berberine and evodiamine on human white preadipocyte differentiation. Fitoterapia 2010, 81, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Srinivasan, S.V.; Neumann, J.C.; Lingrel, J.B. The KLF2 transcription factor does not affect the formation of preadipocytes but inhibits their differentiation into adipocytes. Biochemistry 2005, 44, 11098–11105. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.C.; Gotardo, E.M.; Brianti, M.T.; Piraee, M.; Gambero, A.; Ribeiro, M.L. Effects of yerba maté, a plant extract formulation (“YGD”) and resveratrol in 3T3-L1 adipogenesis. Molecules 2014, 19, 16909–16924. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.; Sakaue, H.; Iguchi, H.; Gomi, H.; Okada, Y.; Takashima, Y.; Nakamura, K.; Nakamura, T.; Yamauchi, T.; Kubota, N.; et al. Role of Kruppel-like factor 15 (KLF15) in transcriptional regulation of adipogenesis. J. Biol. Chem. 2005, 280, 12867–12875. [Google Scholar]
- Gray, S.; Feinberg, M.W.; Hull, S.; Kuo, C.T.; Watanabe, M.; Sen-Banerjee, S.; DePina, A.; Haspel, R.; Jain, M.K. The Kruppel-like factor KLF15 regulates the insulinsensitive glucose transporter GLUT4. J. Biol. Chem. 2002, 277, 34322–34328. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Kim, K.A.; Kim, J.H.; Sul, H.S. Pref-1, a preadipocyte secreted factor that inhibits adipogenesis. J. Nutr. 2006, 136, 2953–2956. [Google Scholar] [PubMed]
- Christodoulides, C.; Lagathu, C.; Sethi, J.K.; Vidal-Puig, A. Adipogenesis and WNT signalling. Trends Endocrinol. Metab. 2009, 20, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.E.; Erickson, R.L.; Gerin, I.; DeRose, P.M.; Bajnok, L.; Longo, K.A.; Misek, D.E.; Kuick, R.; Hanash, S.M.; Atkins, K.B.; et al. Microarray analyses during adipogenesis: Understanding the effects of Wnt signalling on adipogenesis and the roles of liver X receptor alpha in adipocyte metabolism. Mol. Cell. Biol. 2002, 22, 5989–5999. [Google Scholar]
- Nishizuka, M.; Koyanagi, A.; Osada, S.; Imagawa, M. Wnt4 and Wnt5a promote adipocyte differentiation. FEBS Lett. 2008, 582, 3201–3205. [Google Scholar] [CrossRef] [PubMed]
- Kawai, M.; Mushiakea, S.; Bessho, K.; Murakami, M.; Namba, N.; Kokubu, C.; Michigami, T.; Ozono, K. Wnt/Lrp/betacatenin signaling suppresses adipogenesis by inhibiting mutual activation of PPARgamma and C/EBPalpha. Biochem. Biophys. Res. Commun. 2007, 363, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Arçari, D.P.; Santos, J.C.; Gambero, A.; Ribeiro, M.L. The in vitro and in vivo effects of yerba maté (Ilex paraguariensis) extract on adipogenesis. Food Chem. 2013, 141, 809–815. [Google Scholar] [CrossRef] [PubMed]
- Christodoulides, C.; Scarda, A.; Granzotto, M.; Milan, G.; Dalla Nora, E.; Keogh, J.; de Pergola, G.; Stirling, H.; Pannacciulli, N.; Sethi, J.K.; et al. Wnt-10B mutations in human obesity. Diabetologia 2006, 49, 678–684. [Google Scholar]
- Bennett, C.N.; Ross, S.E.; Longo, K.A.; Bajnok, L.; Hemati, N.; Johnson, K.W.; Harrison, S.D.; MacDougald, O.A. Regulation of Wnt signaling during adipogenesis. J. Biol. Chem. 2002, 277, 30998–31004. [Google Scholar] [CrossRef] [PubMed]
- Murahovschi, V.; Pivovarova, O.; Ilkavets, I.; Dmitrieva, R.M.; Döcke, S.; Keyhani-Nejad, F.; Gögebakan, O.; Osterhoff, M.; Kemper, M.; Hornemann, S.; et al. WISP1 is a novel adipokine linked to inflammation in obesity. Diabetes 2015, 64, 856–866. [Google Scholar]
- Kanazawa, A.; Tsukada, S.; Kamiyama, M.; Yanagimoto, T.; Nakajima, M.; Maeda, S. Wnt5b partially inhibits canonical Wnt/β-catenin signaling pathway and promotes adipogenesis in 3T3-L1 preadipocytes. Biochem. Biophys. Res. Commun. 2005, 330, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Van Tienen, F.H.; Laeremans, H.; van der Kallen, C.J.; Smeets, H.J. Wnt5b stimulates adipogenesis by activating PPARgamma, and inhibiting the β-catenin dependent Wnt signaling pathway together with Wnt5a. Biochem. Biophys. Res. Commun. 2009, 387, 207–211. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, B.; Smith, U. Activation of canonical wingless-type MMTV integration site family (Wnt) signaling in mature adipocytes increases beta-catenin levels and leads to cell dedifferentiation and insulin resistance. J. Biol. Chem. 2010, 285, 14031–14041. [Google Scholar] [CrossRef] [PubMed]
- Ouchi, N.; Higuchi, A.; Ohashi, K.; Oshima, Y.; Gokce, N.; Shibata, R.; Akasaki, Y.; Shimono, A.; Walsh, K. Sfrp5 is an anti-inflammatory adipokine that modulates metabolic dysfunction in obesity. Science 2010, 329, 454–457. [Google Scholar] [CrossRef] [PubMed]
- Catalán, V.; Gómez-Ambrosi, J.; Rodríguez, A.; Pérez-Hernández, A.I.; Gurbindo, J.; Ramírez, B.; Méndez-Giménez, L.; Rotellar, F.; Valentí, V.; Moncada, R.; et al. Activation of noncanonical Wnt signaling through WNT5A in visceral adipose tissue of obese subjects is related to inflammation. J. Clin. Endocrinol. Metab. 2014, 99, E1407–E1417. [Google Scholar]
- Fajas, L.; Egler, V.; Reiter, R.; Hansen, J.; Kristiansen, K.; Debril, M.B.; Miard, S.; Auwerx, J. The retinoblastoma-histone deacetylase 3 complex inhibits PPARγ and adipocyte differentiation. Dev. Cell 2002, 3, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Fajas, L.; Landsberg, R.L.; Huss-Garcia, Y. E2Fs regulate adipocyte differentiation. Dev. Cell 2002, 3, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Chavey, C.; Lagarrigue, S.; Annicotte, J.-S.; Fajas, L. Emerging roles of cell cycle regulators in adipocyte metabolism in physiology and physiopathology of adipose tissue. In Physiology and Pathophysiology of Adipose Tissue; Bastard, J.-P., Fève, B., Eds.; Springer: Paris, France, 2013; Volume 1, pp. 17–25. [Google Scholar]
- Sarruf, D.A.; Iankova, I.; Abella, A.; Assou, S.; Miard, S.; Fajas, L. Cyclin D3 promotes adipogenesis through activation of peroxisome proliferator-activated receptor gamma. Mol. Cell. Biol. 2005, 25, 9985–9995. [Google Scholar] [CrossRef] [PubMed]
- Pomari, E.; Stefanon, B.; Colitti, M. Effect of Arctium lappa (burdock) extract on canine dermal fibroblasts. Vet. Immunol. Immunopathol. 2013, 156, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- SPSS®. Statistical Package for Social Science SPSS. In Advanced Statistics 7.5; SPSS Inc.: Chicago, IL, USA, 1997. [Google Scholar]
- Sample Availability: Not available.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pomari, E.; Stefanon, B.; Colitti, M. Effects of Two Different Rhodiola rosea Extracts on Primary Human Visceral Adipocytes. Molecules 2015, 20, 8409-8428. https://doi.org/10.3390/molecules20058409
Pomari E, Stefanon B, Colitti M. Effects of Two Different Rhodiola rosea Extracts on Primary Human Visceral Adipocytes. Molecules. 2015; 20(5):8409-8428. https://doi.org/10.3390/molecules20058409
Chicago/Turabian StylePomari, Elena, Bruno Stefanon, and Monica Colitti. 2015. "Effects of Two Different Rhodiola rosea Extracts on Primary Human Visceral Adipocytes" Molecules 20, no. 5: 8409-8428. https://doi.org/10.3390/molecules20058409
APA StylePomari, E., Stefanon, B., & Colitti, M. (2015). Effects of Two Different Rhodiola rosea Extracts on Primary Human Visceral Adipocytes. Molecules, 20(5), 8409-8428. https://doi.org/10.3390/molecules20058409






