Significance of β-Galactoside α2,6 Sialyltranferase 1 in Cancers
Abstract
:1. Introduction
| Sialyltransferases | Acceptor Sequence(s)/(Carrier Type) | Types of Cancers in Which the Altered Expression Observed | References ** |
|---|---|---|---|
| ST3Gal I | Galβ1-3GalNAc *, Galβ1-3GlcNAc/(O-glycans, glycolipids) | Breast, bladder, colon | [20,21,22] |
| ST3Gal II | Galβ1-3GalNAc/(O-glycans, glycolipids) | Prostate, colon | [22,23] |
| ST3Gal III | Galβ1-3*/4GlcNAc(N-glycans, O-glycans, glycolipids) | Stomach, pancreas, extrahepatic bile duct, cervix | [24,25,26,27] |
| ST3Gal IV | Galβ1-3GalNAc, Galβ1-3/4*GlcNAc/(N-glycans, O-glycans, glycolipids) | Renal cell, stomach | [28,29] |
| ST3Gal V | Galβ1-4Glc/(glycolipids) | Pediatric leukemia | [30] |
| ST3Gal VI | Galβ1-3/4*GlcNAc/(N-glycans, O-glycans, glycolipids) | ||
| ST6Gal I | Galβ1-4GlcNAc/(N-glycans) | Colon, breast, cervix, choriocarcinomas, acute myeloid leukemias, liver, brain | [18,31,32,33,34,35,36,37,38,39,40] |
| ST6Gal II | Galβ1-4GlcNAc/(N-glycans) | ||
| ST6GalNAc I | Galβ1-3GalNAc/(O-glycans) | Stomach, pancreas, colon, ovary, breast | [15,41,42,43,44] |
| ST6GalNAc II | Galβ1-3GalNAc/(O-glycans) | Colon | [42,45] |
| ST6GalNAc III | Galβ1-3GalNAc, GM1b/(O-glycans, glycolipids) | ||
| ST6GalNAc IV | Galβ1-3GalNAc, GM1b/(O-glycans, glycolipids) | ||
| ST6GalNAc V | GM1b/(glycolipids) | Colon, breast | [46,47] |
| ST6GalNAc VI | GM1b, GT1b/(glycolipids) | Colon | [48] |
| ST8Sia I | GM3/(glycolipids) | Breast cancer, pediatric acute leukemia | [30,49] |
| ST8Sia II | Sia2-3/6/8Galβ1-4GlcNAc/(N-glycans) | Liver | [37] |
| ST8Sia III | GT3, Siaα2-3Galβ1-4GlcNAc/(N-glycans, glycolipids) | Glioblastoma | [50] |
| ST8Sia IV | Sia2-3/6/8Galβ1-4GlcNAc/(N-glycans) | ||
| ST8Sia V | GD3, GM1b GD1a, GT1b, GQ1c/(glycolipids) |
2. The Structure of ST6Gal I Gene and Protein as well as the Lectins Specifically Recognizing α2,6 Sialylated Sugar Chains
3. Functions of ST6Gal I in Cancer Progression
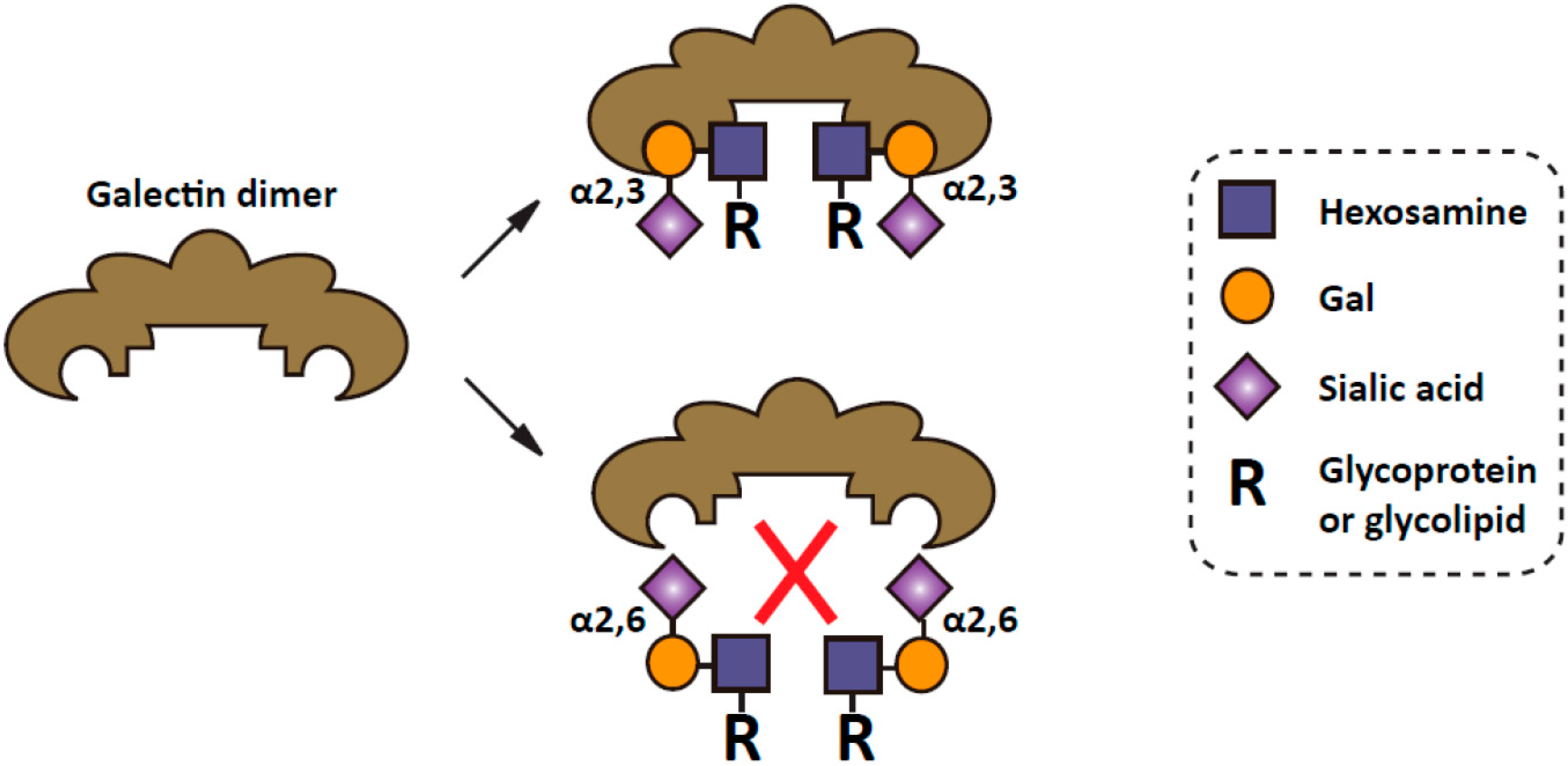
4. Mechanistic Roles of ST6Gal I in Cancer Progression
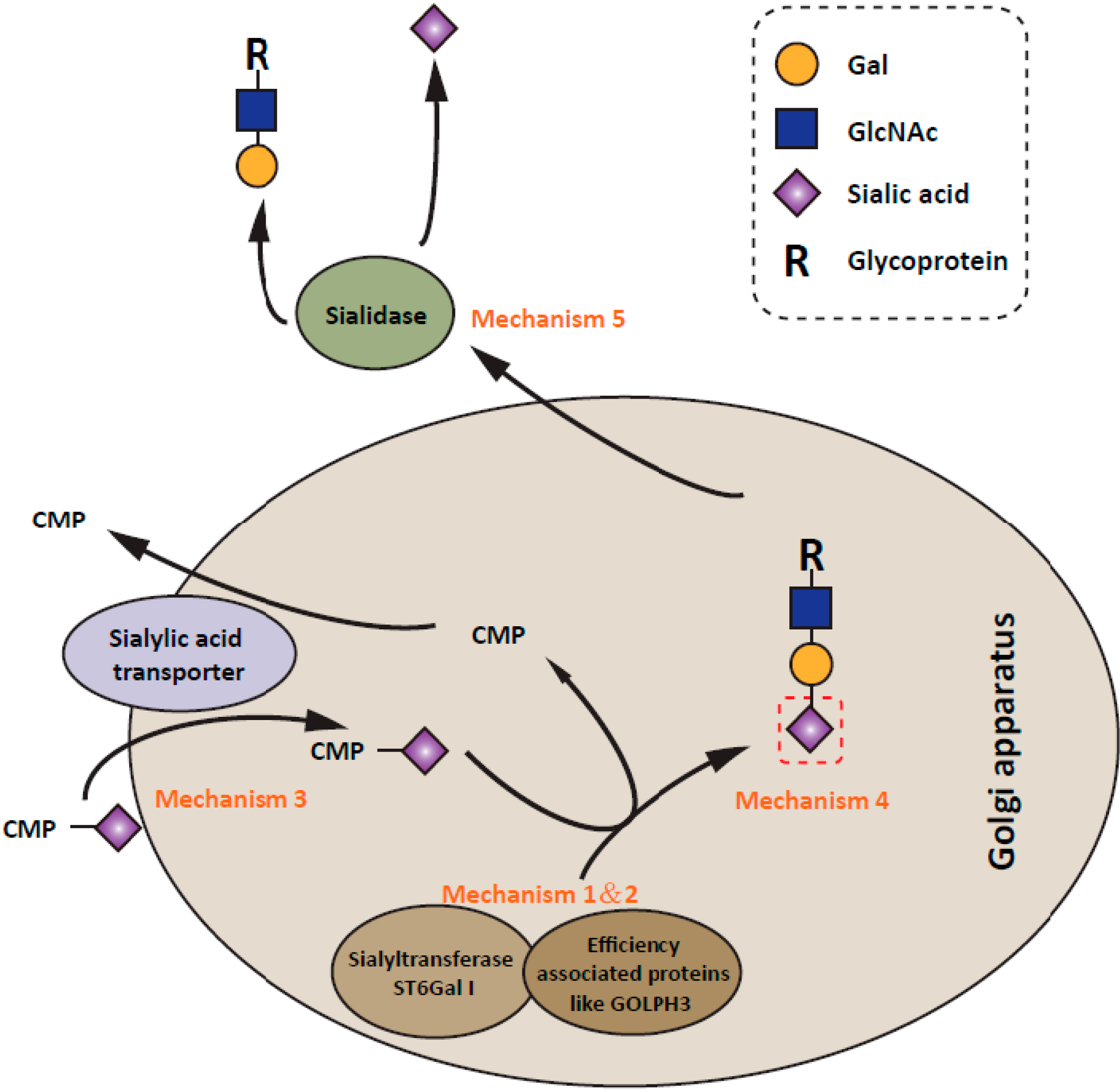
5. Regulatory Mechanisms of ST6Gal I Expression in Cancer Progression
6. Conclusions and Future Directions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kannagi, R.; Sakuma, K.; Cai, B.-H.; Yu, S.-Y. Tumor-Associated Glycans and Their Functional Roles in the Multistep Process of Human Cancer Progression. In Sugar Chains; Suzuki, T., Ohtsubo, K., Taniguchi, N., Eds.; Springer: Tokyo, Japan, 2015; pp. 139–158. [Google Scholar]
- Glavey, S.V.; Huynh, D.; Reagan, M.R.; Manier, S.; Moschetta, M.; Kawano, Y.; Roccaro, A.M.; Ghobrial, I.M.; Joshi, L.; O’Dwyer, M.E. The cancer glycome: Carbohydrates as mediators of metastasis. Blood Rev. 2015. [Google Scholar] [CrossRef]
- Drake, R.R.; Jones, E.E.; Powers, T.W.; Nyalwidhe, J.O. Altered Glycosylation in Prostate Cancer. In Advances in Cancer Research; Richard, R.D., Lauren, E.B., Eds.; Academic Press: New York, NY, USA, 2015; Volume 126, pp. 345–382. [Google Scholar]
- Lemjabbar-Alaoui, H.; McKinney, A.; Yang, Y.-W.; Tran, V.M.; Phillips, J.J. Glycosylation Alterations in Lung and Brain Cancer. In Advances in Cancer Research; Richard, R.D., Lauren, E.B., Eds.; Academic Press: New York, NY, USA, 2015; Volume 126, pp. 305–344. [Google Scholar]
- Varki, N.M.; Varki, A. Diversity in cell surface sialic acid presentations: Implications for biology and disease. Lab. Investig. 2007, 87, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Angata, T.; Varki, A. Chemical diversity in the sialic acids and related alpha-keto acids: An evolutionary perspective. Chem. Rev. 2002, 102, 439–469. [Google Scholar] [CrossRef] [PubMed]
- Varki, A.; Schauer, R. Sialic Acids. In Essentials of Glycobiology, 2nd ed.; Varki, A., Cummings, R.D., Esko, J.D., Freeze, H.H., Stanley, P., Bertozzi, C.R., Hart, G.W., Etzler, M.E., Eds.; Cold Spring Harbor: New York, NY, USA, 2009; pp. 199–217. [Google Scholar]
- Warren, L. The Distribution of Sialic Acids in Nature. Comp. Biochem. Physiol. 1963, 10, 153–171. [Google Scholar] [CrossRef] [PubMed]
- Varki, A. Diversity in the sialic acids. Glycobiology 1992, 2, 25–40. [Google Scholar] [CrossRef] [PubMed]
- Chou, H.-H.; Takematsu, H.; Diaz, S.; Iber, J.; Nickerson, E.; Wright, K.L.; Muchmore, E.A.; Nelson, D.L.; Warren, S.T.; Varki, A. A mutation in human CMP-sialic acid hydroxylase occurred after the Homo-Pan divergence. Proc. Natl. Acad. Sci. USA 1998, 95, 11751–11756. [Google Scholar] [CrossRef] [PubMed]
- Schultz, M.J.; Swindall, A.F.; Bellis, S.L. Regulation of the metastatic cell phenotype by sialylated glycans. Cancer Metastasis Rev. 2012, 31, 501–518. [Google Scholar] [CrossRef] [PubMed]
- Schauer, R. Sialic acids as regulators of molecular and cellular interactions. Curr. Opin. Struct. Biol. 2009, 19, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Bull, C.; den Brok, M.H.; Adema, G.J. Sweet escape: Sialic acids in tumor immune evasion. Biochim. Biophys. Acta 2014, 1846, 238–246. [Google Scholar] [PubMed]
- Varki, A.; Kannagi, R.; Toole, B.P. Glycosylation Changes in Cancer. In Essentials of Glycobiology, 2nd ed.; Varki, A., Cummings, R.D., Esko, J.D., Freeze, H.H., Stanley, P., Bertozzi, C.R., Hart, G.W., Etzler, M.E., Eds.; Cold Spring Harbor: New York, NY, USA, 2009; pp. 617–632. [Google Scholar]
- Brockhausen, I. Mucin-type O-glycans in human colon and breast cancer: Glycodynamics and functions. EMBO Rep. 2006, 7, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Dall’Olio, F.; Malagolini, N.; Trinchera, M.; Chiricolo, M. Sialosignaling: Sialyltransferases as engines of self-fueling loops in cancer progression. Biochim. Biophys. Acta 2014, 1840, 2752–2764. [Google Scholar] [CrossRef] [PubMed]
- Julien, S.; Delannoy, P. Sialic Acid and Cancer. In Glycoscience: Biology and Medicine; Endo, T., Seeberger, P.H., Hart, G.W., Wong, C.-H., Taniguchi, N., Eds.; Springer Japan: Tokyo, Japan, 2014; pp. 1–6. [Google Scholar]
- Dall’Olio, F.; Chiricolo, M. Sialyltransferases in cancer. Glycoconj. J. 2001, 18, 841–850. [Google Scholar] [CrossRef] [PubMed]
- Harduin-Lepers, A.; Krzewinski-Recchi, M.A.; Colomb, F.; Foulquier, F.; Groux-Degroote, S.; Delannoy, P. Sialyltransferases functions in cancers. Front. Biosci. 2012, 4, 499–515. [Google Scholar] [CrossRef]
- Burchell, J.; Poulsom, R.; Hanby, A.; Whitehouse, C.; Cooper, L.; Clausen, H.; Miles, D.; Taylor-Papadimitriou, J. An alpha2,3 sialyltransferase (ST3Gal I) is elevated in primary breast carcinomas. Glycobiology 1999, 9, 1307–1311. [Google Scholar] [CrossRef] [PubMed]
- Videira, P.A.; Correia, M.; Malagolini, N.; Crespo, H.J.; Ligeiro, D.; Calais, F.M.; Trindade, H.; Dall’Olio, F. ST3Gal.I sialyltransferase relevance in bladder cancer tissues and cell lines. BMC Cancer 2009, 9, 357. [Google Scholar] [CrossRef] [PubMed]
- Kudo, T.; Ikehara, Y.; Togayachi, A.; Morozumi, K.; Watanabe, M.; Nakamura, M.; Nishihara, S.; Narimatsu, H. Up-regulation of a set of glycosyltransferase genes in human colorectal cancer. Lab. Investig. 1998, 78, 797–811. [Google Scholar] [PubMed]
- Hatano, K.; Miyamoto, Y.; Mori, M.; Nimura, K.; Nakai, Y.; Nonomura, N.; Kaneda, Y. Androgen-regulated transcriptional control of sialyltransferases in prostate cancer cells. PLoS ONE 2012, 7, e31234. [Google Scholar] [CrossRef] [PubMed]
- Perez-Garay, M.; Arteta, B.; Pages, L.; de Llorens, R.; de Bolos, C.; Vidal-Vanaclocha, F.; Peracaula, R. alpha2,3-sialyltransferase ST3Gal III modulates pancreatic cancer cell motility and adhesion in vitro and enhances its metastatic potential in vivo. PLoS ONE 2010, 5, e12524. [Google Scholar] [CrossRef] [PubMed]
- Gretschel, S.; Haensch, W.; Schlag, P.M.; Kemmner, W. Clinical relevance of sialyltransferases ST6GAL-I and ST3GAL-III in gastric cancer. Oncology 2003, 65, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.L.; Zheng, S.S.; Wang, B.S.; Chen, H.L. Correlation of glycosyltransferases mRNA expression in extrahepatic bile duct carcinoma with clinical pathological characteristics. Hepatobiliary Pancreat. Dis. Int. 2004, 3, 292–295. [Google Scholar] [PubMed]
- Wang, P.H.; Li, Y.F.; Juang, C.M.; Lee, Y.R.; Chao, H.T.; Ng, H.T.; Tsai, Y.C.; Yuan, C.C. Expression of sialyltransferase family members in cervix squamous cell carcinoma correlates with lymph node metastasis. Gynecol. Oncol. 2002, 86, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Gomes, C.; Osorio, H.; Pinto, M.T.; Campos, D.; Oliveira, M.J.; Reis, C.A. Expression of ST3GAL4 leads to SLe(x) expression and induces c-Met activation and an invasive phenotype in gastric carcinoma cells. PLoS ONE 2013, 8, e66737. [Google Scholar] [CrossRef] [PubMed]
- Saito, S.; Yamashita, S.; Endoh, M.; Yamato, T.; Hoshi, S.; Ohyama, C.; Watanabe, R.; Ito, A.; Satoh, M.; Wada, T.; et al. Clinical significance of ST3Gal IV expression in human renal cell carcinoma. Oncol. Rep. 2002, 9, 1251–1255. [Google Scholar] [PubMed]
- Mondal, S.; Chandra, S.; Mandal, C. Elevated mRNA level of hST6Gal I and hST3Gal V positively correlates with the high risk of pediatric acute leukemia. Leuk. Res. 2010, 34, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Recchi, M.A.; Hebbar, M.; Hornez, L.; Harduin-Lepers, A.; Peyrat, J.P.; Delannoy, P. Multiplex reverse transcription polymerase chain reaction assessment of sialyltransferase expression in human breast cancer. Cancer Res. 1998, 58, 4066–4070. [Google Scholar] [PubMed]
- Dall’Olio, F.; Malagolini, N.; di Stefano, G.; Minni, F.; Marrano, D.; Serafini-Cessi, F. Increased CMP-NeuAc:Gal beta 1,4GlcNAc-R alpha 2,6 sialyltransferase activity in human colorectal cancer tissues. Int. J. Cancer 1989, 44, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Skacel, P.O.; Edwards, A.J.; Harrison, C.T.; Watkins, W.M. Enzymic control of the expression of the X determinant (CD15) in human myeloid cells during maturation: The regulatory role of 6-sialytransferase. Blood 1991, 78, 1452–1460. [Google Scholar] [PubMed]
- Fukushima, K.; Hara-Kuge, S.; Seko, A.; Ikehara, Y.; Yamashita, K. Elevation of alpha2-->6 sialyltransferase and alpha1-->2 fucosyltransferase activities in human choriocarcinoma. Cancer Res. 1998, 58, 4301–4306. [Google Scholar] [PubMed]
- Wang, P.H.; Li, Y.F.; Juang, C.M.; Lee, Y.R.; Chao, H.T.; Tsai, Y.C.; Yuan, C.C. Altered mRNA expression of sialyltransferase in squamous cell carcinomas of the cervix. Gynecol. Oncol. 2001, 83, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, Y.; Yamamoto, H.; Kersey, D.S.; Colley, K.J.; Leestma, J.E.; Moskal, J.R. The expression of Gal beta 1,4GlcNAc alpha 2,6 sialyltransferase and alpha 2,6-linked sialoglycoconjugates in human brain tumors. Acta Neuropathol. 1996, 91, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Li, Y.; Ma, H.; Dong, W.; Zhou, H.; Song, X.; Zhang, J.; Jia, L. Modification of sialylation mediates the invasive properties and chemosensitivity of human hepatocellular carcinoma. Mol. Cell. Proteomics 2014, 13, 520–536. [Google Scholar] [CrossRef] [PubMed]
- Dall’Olio, F.; Trere, D. Expression of alpha 2,6-sialylated sugar chains in normal and neoplastic colon tissues. Detection by digoxigenin-conjugated Sambucus nigra agglutinin. Eur. J. Histochem. 1993, 37, 257–265. [Google Scholar] [PubMed]
- Sata, T.; Roth, J.; Zuber, C.; Stamm, B.; Heitz, P.U. Expression of alpha 2,6-linked sialic acid residues in neoplastic but not in normal human colonic mucosa. A lectin-gold cytochemical study with Sambucus nigra and Maackia amurensis lectins. Am. J. Pathol. 1991, 139, 1435–1448. [Google Scholar] [PubMed]
- Dall’Olio, F.; Chiricolo, M.; D’Errico, A.; Gruppioni, E.; Altimari, A.; Fiorentino, M.; Grigioni, W.F. Expression of beta-galactoside alpha2,6 sialyltransferase and of alpha2,6-sialylated glycoconjugates in normal human liver, hepatocarcinoma, and cirrhosis. Glycobiology 2004, 14, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Julien, S.; Adriaenssens, E.; Ottenberg, K.; Furlan, A.; Courtand, G.; Vercoutter-Edouart, A.S.; Hanisch, F.G.; Delannoy, P.; Le Bourhis, X. ST6GalNAc I expression in MDA-MB-231 breast cancer cells greatly modifies their O-glycosylation pattern and enhances their tumourigenicity. Glycobiology 2006, 16, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Marcos, N.T.; Pinho, S.; Grandela, C.; Cruz, A.; Samyn-Petit, B.; Harduin-Lepers, A.; Almeida, R.; Silva, F.; Morais, V.; Costa, J.; et al. Role of the human ST6GalNAc-I and ST6GalNAc-II in the synthesis of the cancer-associated sialyl-Tn antigen. Cancer Res. 2004, 64, 7050–7057. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Martín, C.; Cuevas, E.; Gil-Martín, E.; Fernández-Briera, A. Correlation Analysis between Tumorous Associated Antigen Sialyl-Tn Expression and ST6GalNAc I Activity in Human Colon Adenocarcinoma. Oncology 2004, 67, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Pinho, S.; Marcos, N.T.; Ferreira, B.; Carvalho, A.S.; Oliveira, M.J.; Santos-Silva, F.; Harduin-Lepers, A.; Reis, C.A. Biological significance of cancer-associated sialyl-Tn antigen: Modulation of malignant phenotype in gastric carcinoma cells. Cancer Lett. 2007, 249, 157–170. [Google Scholar] [CrossRef] [PubMed]
- Schneider, F.; Kemmner, W.; Haensch, W.; Franke, G.; Gretschel, S.; Karsten, U.; Schlag, P.M. Overexpression of sialyltransferase CMP-sialic acid: Galbeta1,3GalNAc-R alpha6-Sialyltransferase is related to poor patient survival in human colorectal carcinomas. Cancer Res. 2001, 61, 4605–4611. [Google Scholar]
- Bos, P.D.; Zhang, X.H.F.; Nadal, C.; Shu, W.P.; Gomis, R.R.; Nguyen, D.X.; Minn, A.J.; van de Vijver, M.J.; Gerald, W.L.; Foekens, J.A.; et al. Genes that mediate breast cancer metastasis to the brain. Nature 2009, 459, 1005–1009. [Google Scholar] [CrossRef] [PubMed]
- Tsuchida, A.; Okajima, T.; Furukawa, K.; Ando, T.; Ishida, H.; Yoshida, A.; Nakamura, Y.; Kannagi, R.; Kiso, M.; Furukawa, K. Synthesis of disialyl Lewis a (Le(a)) structure in colon cancer cell lines by a sialyltransferase, ST6GalNAc VI, responsible for the synthesis of alpha-series gangliosides. J. Biol. Chem. 2003, 278, 22787–22794. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, K.; Ohmori, K.; Izawa, M.; Koike, T.; Kumamoto, K.; Furukawa, K.; Ando, T.; Kiso, M.; Yamaji, T.; Hashimoto, Y.; et al. Loss of disialyl Lewis(a), the ligand for lymphocyte inhibitory receptor sialic acid-binding immunoglobulin-like lectin-7 (Siglec-7) associated with increased sialyl Lewis(a) expression on human colon cancers. Cancer Res. 2004, 64, 4498–4505. [Google Scholar] [CrossRef] [PubMed]
- Steenackers, A.; Vanbeselaere, J.; Cazet, A.; Bobowski, M.; Rombouts, Y.; Colomb, F.; Le Bourhis, X.; Guerardel, Y.; Delannoy, P. Accumulation of unusual gangliosides G(Q3) and G(P3) in breast cancer cells expressing the G(D3) synthase. Molecules 2012, 17, 9559–9572. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Chung, T.W.; Jin, U.H.; Suh, S.J.; Lee, Y.C.; Kim, C.H. Molecular mechanisms involved in transcriptional activation of the human Sia-alpha2,3-Gal-beta1,4-GlcNAc-R: Alpha2,8-sialyltransferase (hST8Sia III) gene induced by KCl in human glioblastoma cells. Biochem. Biophys. Res. Commun. 2006, 344, 1057–1064. [Google Scholar] [CrossRef] [PubMed]
- Weinstein, J.; Lee, E.U.; McEntee, K.; Lai, P.H.; Paulson, J.C. Primary structure of beta-galactoside alpha 2,6-sialyltransferase. Conversion of membrane-bound enzyme to soluble forms by cleavage of the NH2-terminal signal anchor. J. Biol. Chem. 1987, 262, 17735–17743. [Google Scholar]
- Wen, D.X.; Svensson, E.C.; Paulson, J.C. Tissue-specific alternative splicing of the beta-galactoside alpha 2,6-sialyltransferase gene. J. Biol. Chem. 1992, 267, 2512–2518. [Google Scholar] [PubMed]
- Wang, X.; Vertino, A.; Eddy, R.L.; Byers, M.G.; Jani-Sait, S.N.; Shows, T.B.; Lau, J.T. Chromosome mapping and organization of the human beta-galactoside alpha 2,6-sialyltransferase gene. Differential and cell-type specific usage of upstream exon sequences in B-lymphoblastoid cells. J. Biol. Chem. 1993, 268, 4355–4361. [Google Scholar] [PubMed]
- Dall’Olio, F.; Chiricolo, M.; Lau, J.T.Y. Differential expression of the hepatic transcript of β-galactoside α2,6-sialyltransferase in human colon cancer cell lines. Int. J. Cancer 1999, 81, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Grundmann, U.; Nerlich, C.; Rein, T.; Zettlmeissl, G. Complete cDNA sequence encoding human beta-galactoside alpha-2,6-sialyltransferase. Nucleic Acids Res. 1990, 18, 667. [Google Scholar] [CrossRef] [PubMed]
- Keppler, O.T.; Moldenhauer, G.; Oppenlander, M.; Schwartz-Albiez, R.; Berger, E.G.; Funderud, S.; Pawlita, M. Human Golgi beta-galactoside alpha-2,6-sialyltransferase generates a group of sialylated B lymphocyte differentiation antigens. Eur. J. Immunol. 1992, 22, 2777–2781. [Google Scholar] [CrossRef] [PubMed]
- Aas-Eng, D.A.; Asheim, H.C.; Deggerdal, A.; Smeland, E.; Funderud, S. Characterization of a promoter region supporting transcription of a novel human beta-galactoside alpha-2,6-sialyltransferase transcript in HepG2 cells. Biochim. Biophys. Acta 1995, 1261, 166–169. [Google Scholar] [CrossRef] [PubMed]
- Dall’Olio, F.; Chiricolo, M.; Ceccarelli, C.; Minni, F.; Marrano, D.; Santini, D. Beta-galactoside alpha2,6 sialyltransferase in human colon cancer: Contribution of multiple transcripts to regulation of enzyme activity and reactivity with Sambucus nigra agglutinin. Int. J. Cancer 2000, 88, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Shibuya, N.; Goldstein, I.J.; Broekaert, W.F.; Nsimba-Lubaki, M.; Peeters, B.; Peumans, W.J. The elderberry (Sambucus nigra L.) bark lectin recognizes the Neu5Ac(alpha 2–6)Gal/GalNAc sequence. J. Biol. Chem. 1987, 262, 1596–1601. [Google Scholar] [PubMed]
- Yamashita, K.; Umetsu, K.; Suzuki, T.; Ohkura, T. Purification and characterization of a Neu5Ac alpha 2-->6Gal beta 1-->4GlcNAc and HSO3(-)-->6Gal beta 1-->GlcNAc specific lectin in tuberous roots of Trichosanthes japonica. Biochemistry 1992, 31, 11647–11650. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, K.; Fukushima, K.; Sakiyama, T.; Murata, F.; Kuroki, M.; Matsuoka, Y. Expression of Sia alpha 2-->6Gal beta 1-->4GlcNAc residues on sugar chains of glycoproteins including carcinoembryonic antigens in human colon adenocarcinoma: Applications of Trichosanthes japonica agglutinin I for early diagnosis. Cancer Res. 1995, 55, 1675–1679. [Google Scholar] [PubMed]
- Korekane, H.; Matsumoto, A.; Ota, F.; Hasegawa, T.; Misonou, Y.; Shida, K.; Miyamoto, Y.; Taniguchi, N. Involvement of ST6Gal I in the biosynthesis of a unique human colon cancer biomarker candidate, alpha2,6-sialylated blood group type 2H (ST2H) antigen. J. Biochem. 2010, 148, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Vierbuchen, M.J.; Fruechtnicht, W.; Brackrock, S.; Krause, K.T.; Zienkiewicz, T.J. Quantitative lectin-histochemical and immunohistochemical studies on the occurrence of alpha(2,3)- and alpha(2,6)-linked sialic acid residues in colorectal carcinomas. Relation to clinicopathologic features. Cancer 1995, 76, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Hedlund, M.; Ng, E.; Varki, A.; Varki, N.M. alpha 2–6-Linked sialic acids on N-glycans modulate carcinoma differentiation in vivo. Cancer Res. 2008, 68, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Chiricolo, M.; Malagolini, N.; Bonfiglioli, S.; Dall’Olio, F. Phenotypic changes induced by expression of beta-galactoside alpha2,6 sialyltransferase I in the human colon cancer cell line SW948. Glycobiology 2006, 16, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Park, J.J.; Yi, J.Y.; Jin, Y.B.; Lee, Y.J.; Lee, J.S.; Lee, Y.S.; Ko, Y.G.; Lee, M. Sialylation of epidermal growth factor receptor regulates receptor activity and chemosensitivity to gefitinib in colon cancer cells. Biochem. Pharmacol. 2012, 83, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, H.; Oviedo, A.; Sweeley, C.; Saito, T.; Moskal, J.R. Alpha2,6-sialylation of cell-surface N-glycans inhibits glioma formation in vivo. Cancer Res. 2001, 61, 6822–6829. [Google Scholar] [PubMed]
- Zhang, Z.; Sun, J.; Hao, L.; Liu, C.; Ma, H.; Jia, L. Modification of glycosylation mediates the invasive properties of murine hepatocarcinoma cell lines to lymph nodes. PLoS ONE 2013, 8, e65218. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Kemmner, W.; Grigull, S.; Schlag, P.M. Cell surface alpha 2,6 sialylation affects adhesion of breast carcinoma cells. Exp. Cell Res. 2002, 276, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Srivatana, U.; Ullah, A.; Gagneja, H.; Berenson, C.S.; Lance, P. Suppression of a sialyltransferase by antisense DNA reduces invasiveness of human colon cancer cells in vitro. Biochim. Biophys. Acta 2001, 1536, 148–160. [Google Scholar] [CrossRef] [PubMed]
- Christie, D.R.; Shaikh, F.M.; Lucas, J.A., 4th; Lucas, J.A., 3rd; Bellis, S.L. ST6Gal-I expression in ovarian cancer cells promotes an invasive phenotype by altering integrin glycosylation and function. J. Ovarian Res. 2008, 1. [Google Scholar] [CrossRef]
- Bresalier, R.S.; Rockwell, R.W.; Dahiya, R.; Duh, Q.Y.; Kim, Y.S. Cell surface sialoprotein alterations in metastatic murine colon cancer cell lines selected in an animal model for colon cancer metastasis. Cancer Res. 1990, 50, 1299–1307. [Google Scholar] [PubMed]
- Isaji, T.; Im, S.; Gu, W.; Wang, Y.; Hang, Q.; Lu, J.; Fukuda, T.; Hashii, N.; Takakura, D.; Kawasaki, N.; et al. An oncogenic protein Golgi phosphoprotein 3 up-regulates cell migration via sialylation. J. Biol. Chem. 2014, 289, 20694–20705. [Google Scholar] [CrossRef] [PubMed]
- Seales, E.C.; Jurado, G.A.; Brunson, B.A.; Wakefield, J.K.; Frost, A.R.; Bellis, S.L. Hypersialylation of beta1 integrins, observed in colon adenocarcinoma, may contribute to cancer progression by up-regulating cell motility. Cancer Res. 2005, 65, 4645–4652. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, F.M.; Seales, E.C.; Clem, W.C.; Hennessy, K.M.; Zhuo, Y.; Bellis, S.L. Tumor cell migration and invasion are regulated by expression of variant integrin glycoforms. Exp. Cell Res. 2008, 314, 2941–2950. [Google Scholar] [CrossRef] [PubMed]
- Danguy, A.; Camby, I.; Kiss, R. Galectins and cancer. Biochim. Biophys. Acta 2002, 1572, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Nakahara, S.; Raz, A. Biological modulation by lectins and their ligands in tumor progression and metastasis. Anticancer Agents Med. Chem. 2008, 8, 22–36. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.T.; Rabinovich, G.A. Galectins as modulators of tumour progression. Nat. Rev. Cancer 2005, 5, 29–41. [Google Scholar] [CrossRef] [PubMed]
- He, J.L.; Baum, L.G. Galectin interactions with extracellular matrix and effects on cellular function. Methods Enzymol. 2006, 417, 247–256. [Google Scholar] [PubMed]
- Ochieng, J.; Furtak, V.; Lukyanov, P. Extracellular functions of galectin-3. Glycoconj. J. 2002, 19, 527–535. [Google Scholar] [CrossRef]
- Elola, M.T.; Wolfenstein-Todel, C.; Troncoso, M.F.; Vasta, G.R.; Rabinovich, G.A. Galectins: Matricellular glycan-binding proteins linking cell adhesion, migration, and survival. Cell. Mol. Life Sci. 2007, 64, 1679–1700. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, Y.; Bellis, S.L. Emerging role of alpha2,6-sialic acid as a negative regulator of galectin binding and function. J. Biol. Chem. 2011, 286, 5935–5941. [Google Scholar] [CrossRef] [PubMed]
- Hirabayashi, J.; Hashidate, T.; Arata, Y.; Nishi, N.; Nakamura, T.; Hirashima, M.; Urashima, T.; Oka, T.; Futai, M.; Muller, W.E.; et al. Oligosaccharide specificity of galectins: A search by frontal affinity chromatography. Biochim. Biophys. Acta 2002, 1572, 232–254. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, Y.; Chammas, R.; Bellis, S.L. Sialylation of beta1 integrins blocks cell adhesion to galectin-3 and protects cells against galectin-3-induced apoptosis. J. Biol. Chem. 2008, 283, 22177–22185. [Google Scholar] [CrossRef] [PubMed]
- Swindall, A.F.; Bellis, S.L. Sialylation of the Fas death receptor by ST6Gal-I provides protection against Fas-mediated apoptosis in colon carcinoma cells. J. Biol. Chem. 2011, 286, 22982–22990. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Swindall, A.F.; Kesterson, R.A.; Schoeb, T.R.; Bullard, D.C.; Bellis, S.L. ST6Gal-I regulates macrophage apoptosis via alpha2–6 sialylation of the TNFR1 death receptor. J. Biol. Chem. 2011, 286, 39654–39662. [Google Scholar] [CrossRef]
- Moller, P.; Koretz, K.; Leithauser, F.; Bruderlein, S.; Henne, C.; Quentmeier, A.; Krammer, P.H. Expression of APO-1 (CD95), a member of the NGF/TNF receptor superfamily, in normal and neoplastic colon epithelium. Int. J. Cancer 1994, 57, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Lebel, M.; Bertrand, R.; Mes-Masson, A.M. Decreased Fas antigen receptor expression in testicular tumor cell lines derived from polyomavirus large T-antigen transgenic mice. Oncogene 1996, 12, 1127–1135. [Google Scholar] [PubMed]
- Strand, S.; Hofmann, W.J.; Hug, H.; Muller, M.; Otto, G.; Strand, D.; Mariani, S.M.; Stremmel, W.; Krammer, P.H.; Galle, P.R. Lymphocyte apoptosis induced by CD95 (APO-1/Fas) ligand-expressing tumor cells—A mechanism of immune evasion? Nat. Med. 1996, 2, 1361–1366. [Google Scholar] [CrossRef] [PubMed]
- Landowski, T.H.; GleasonGuzman, M.C.; Dalton, W.S. Selection for drug resistance results in resistance to Fas-mediated apoptosis. Blood 1997, 89, 1854–1861. [Google Scholar] [PubMed]
- Natoli, G.; Ianni, A.; Costanzo, A.; Depetrillo, G.; Ilari, I.; Chirillo, P.; Balsano, C.; Levrero, M. Resistance to Fas-Mediated Apoptosis in Human Hepatoma-Cells. Oncogene 1995, 11, 1157–1164. [Google Scholar] [PubMed]
- O’Connell, J.; Bennett, M.W.; O’Sullivan, G.C.; Roche, D.; Kelly, J.; Collins, J.K.; Shanahan, F. Fas ligand expression in primary colon adenocarcinomas: Evidence that the Fas counterattack is a prevalent mechanism of immune evasion in human colon cancer. J. Pathol. 1998, 186, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Keppler, O.T.; Peter, M.E.; Hinderlich, S.; Moldenhauer, G.; Stehling, P.; Schmitz, I.; Schwartz-Albiez, R.; Reutter, W.; Pawlita, M. Differential sialylation of cell surface glycoconjugates in a human B lymphoma cell line regulates susceptibility for CD95 (APO-1/Fas)-mediated apoptosis and for infection by a lymphotropic virus. Glycobiology 1999, 9, 557–569. [Google Scholar] [CrossRef] [PubMed]
- Peter, M.E.; Hellbardt, S.; Schwartz-Albiez, R.; Westendorp, M.O.; Walczak, H.; Moldenhauer, G.; Grell, M.; Krammer, P.H. Cell surface sialylation plays a role in modulating sensitivity towards APO-1-mediated apoptotic cell death. Cell Death Differ. 1995, 2, 163–171. [Google Scholar] [PubMed]
- Suzuki, O.; Nozawa, Y.; Abe, M. Sialic acids linked to glycoconjugates of Fas regulate the caspase-9-dependent and mitochondria-mediated pathway of Fas-induced apoptosis in Jurkat T cell lymphoma. Int. J. Oncol. 2003, 23, 769–774. [Google Scholar] [PubMed]
- Galea, G.L.; Price, J.S.; Lanyon, L.E. Emerging role of estrogen receptor-alpha in bone formation and bone sparing. Bonekey Rep. 2013, 2. [Google Scholar] [CrossRef]
- Ma, H.; Zhou, H.; Song, X.; Shi, S.; Zhang, J.; Jia, L. Modification of sialylation is associated with multidrug resistance in human acute myeloid leukemia. Oncogene 2015, 34, 726–740. [Google Scholar] [CrossRef] [PubMed]
- Swindall, A.F.; Londono-Joshi, A.I.; Schultz, M.J.; Fineberg, N.; Buchsbaum, D.J.; Bellis, S.L. ST6Gal-I protein expression is upregulated in human epithelial tumors and correlates with stem cell markers in normal tissues and colon cancer cell lines. Cancer Res. 2013, 73, 2368–2378. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Isaji, T.; Im, S.; Fukuda, T.; Hashii, N.; Takakura, D.; Kawasaki, N.; Gu, J. beta-Galactoside alpha2,6-sialyltranferase 1 promotes transforming growth factor-beta-mediated epithelial-mesenchymal transition. J. Biol. Chem. 2014, 289, 34627–34641. [Google Scholar] [CrossRef] [PubMed]
- Dawson, G.; Moskal, J.R.; Dawson, S.A. Transfection of 2,6 and 2,3-sialyltransferase genes and GlcNAc-transferase genes into human glioma cell line U-373 MG affects glycoconjugate expression and enhances cell death. J. Neurochem. 2004, 89, 1436–1444. [Google Scholar] [CrossRef] [PubMed]
- Antony, P.; Rose, M.; Heidenreich, A.; Knuchel, R.; Gaisa, N.T.; Dahl, E. Epigenetic inactivation of ST6GAL1 in human bladder cancer. BMC Cancer 2014, 14, 901. [Google Scholar] [CrossRef] [PubMed]
- Croci, D.O.; Cerliani, J.P.; Dalotto-Moreno, T.; Mendez-Huergo, S.P.; Mascanfroni, I.D.; Dergan-Dylon, S.; Toscano, M.A.; Caramelo, J.J.; Garcia-Vallejo, J.J.; Ouyang, J.; et al. Glycosylation-dependent lectin-receptor interactions preserve angiogenesis in anti-VEGF refractory tumors. Cell 2014, 156, 744–758. [Google Scholar] [CrossRef] [PubMed]
- Kimura, N.; Ohmori, K.; Miyazaki, K.; Izawa, M.; Matsuzaki, Y.; Yasuda, Y.; Takematsu, H.; Kozutsumi, Y.; Moriyama, A.; Kannagi, R. Human B-lymphocytes express alpha2–6-sialylated 6-sulfo-N-acetyllactosamine serving as a preferred ligand for CD22/Siglec-2. J. Biol. Chem. 2007, 282, 32200–32207. [Google Scholar] [CrossRef] [PubMed]
- Woodard-Grice, A.V.; McBrayer, A.C.; Wakefield, J.K.; Zhuo, Y.; Bellis, S.L. Proteolytic shedding of ST6Gal-I by BACE1 regulates the glycosylation and function of alpha4beta1 integrins. J. Biol. Chem. 2008, 283, 26364–26373. [Google Scholar] [CrossRef] [PubMed]
- Amano, M.; Galvan, M.; He, J.; Baum, L.G. The ST6Gal I sialyltransferase selectively modifies N-glycans on CD45 to negatively regulate galectin-1-induced CD45 clustering, phosphatase modulation, and T cell death. J. Biol. Chem. 2003, 278, 7469–7475. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.C.; Yen, H.Y.; Chen, C.Y.; Chen, C.H.; Cheng, P.F.; Juan, Y.H.; Chen, C.H.; Khoo, K.H.; Yu, C.J.; Yang, P.C.; et al. Sialylation and fucosylation of epidermal growth factor receptor suppress its dimerization and activation in lung cancer cells. Proc. Natl. Acad. Sci. USA 2011, 108, 11332–11337. [Google Scholar] [CrossRef] [PubMed]
- Kitazume, S.; Imamaki, R.; Ogawa, K.; Komi, Y.; Futakawa, S.; Kojima, S.; Hashimoto, Y.; Marth, J.D.; Paulson, J.C.; Taniguchi, N. Alpha2,6-sialic acid on platelet endothelial cell adhesion molecule (PECAM) regulates its homophilic interactions and downstream antiapoptotic signaling. J. Biol. Chem. 2010, 285, 6515–6521. [Google Scholar] [CrossRef] [PubMed]
- Cha, S.K.; Ortega, B.; Kurosu, H.; Rosenblatt, K.P.; Kuro, O.M.; Huang, C.L. Removal of sialic acid involving Klotho causes cell-surface retention of TRPV5 channel via binding to galectin-1. Proc. Natl. Acad. Sci. USA 2008, 105, 9805–9810. [Google Scholar] [CrossRef] [PubMed]
- Delannoy, P.; Pelczar, H.; Vandamme, V.; Verbert, A. Sialyltransferase activity in FR3T3 cells transformed withras oncogene: Decreased CMP-Neu5Ac:Galβ1–3GalNAc α-2,3-sialyltransferase. Glycoconj. J. 1993, 10, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Easton, E.W.; Bolscher, J.G.; van den Eijnden, D.H. Enzymatic amplification involving glycosyltransferases forms the basis for the increased size of asparagine-linked glycans at the surface of NIH 3T3 cells expressing the N-ras proto-oncogene. J. Biol. Chem. 1991, 266, 21674–21680. [Google Scholar] [PubMed]
- Le Marer, N.; Laudet, V.; Svensson, E.C.; Cazlaris, H.; van Hille, B.; Lagrou, C.; Stehelin, D.; Montreuil, J.; Verbert, A.; Delannoy, P. The c-Ha-ras oncogene induces increased expression of beta-galactoside alpha-2, 6-sialyltransferase in rat fibroblast (FR3T3) cells. Glycobiology 1992, 2, 49–56. [Google Scholar]
- Dalziel, M.; Dall’Olio, F.; Mungul, A.; Piller, V.; Piller, F. Ras oncogene induces β-galactoside α2,6-sialyltransferase (ST6Gal I) via a RalGEF-mediated signal to its housekeeping promoter. Eur. J. Biochem. 2004, 271, 3623–3634. [Google Scholar] [CrossRef] [PubMed]
- Sakakura, C.; Hasegawa, K.; Miyagawa, K.; Nakashima, S.; Yoshikawa, T.; Kin, S.; Nakase, Y.; Yazumi, S.; Yamagishi, H.; Okanoue, T.; et al. Possible involvement of RUNX3 silencing in the peritoneal metastases of gastric cancers. Clin. Cancer Res. 2005, 11, 6479–6488. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Fan, J.; Liu, L.; Zhang, L.; Wang, S.; Zhang, J. Caveolin-1 up-regulates integrin α2,6-sialylation to promote integrin α5β1-dependent hepatocarcinoma cell adhesion. FEBS Lett. 2013, 587, 782–787. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; O’Hanlon, T.P.; Young, R.F.; Lau, J.T. Rat beta-galactoside alpha 2,6-sialyltransferase genomic organization: Alternate promoters direct the synthesis of liver and kidney transcripts. Glycobiology 1990, 1, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Morales, D.; Velazquez-Marquez, N.; Valenzuela, O.; Santos-Lopez, G.; Reyes-Leyva, J.; Vallejo-Ruiz, V. Enhanced sialyltransferases transcription in cervical intraepithelial neoplasia. Investig. Clin. 2009, 50, 45–53. [Google Scholar]
- Milflores-Flores, L.; Millan-Perez, L.; Santos-Lopez, G.; Reyes-Leyva, J.; Vallejo-Ruiz, V. Characterization of P1 promoter activity of the beta-galactoside alpha2,6-sialyltransferase I gene (siat 1) in cervical and hepatic cancer cell lines. J. Biosci. 2012, 37, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Stanley, P. Altered Glycolipids of Cho Cells Resistant to Wheat-Germ Agglutinin. Abstr. Pap. Am. Chem. Soc. 1979, 70. [Google Scholar]
- Stanley, P. Membrane Mutants of Animal-Cells: Rapid Identification of Those with a Primary Defect in Glycosylation. Mol. Cell. Biol. 1985, 5, 923–929. [Google Scholar] [PubMed]
- Miyagi, T.; Sagawa, J.; Kuroki, T.; Matsuya, Y.; Tsuiki, S. Tumor-promoting phorbol ester induces alterations of sialidase and sialyltransferase activities of JB6 cells. Jpn. J. Cancer Res. 1990, 81, 1286–1292. [Google Scholar] [CrossRef] [PubMed]
- Sawada, M.; Moriya, S.; Saito, S.; Shineha, R.; Satomi, S.; Yamori, T.; Tsuruo, T.; Kannagi, R.; Miyagi, T. Reduced sialidase expression in highly metastatic variants of mouse colon adenocarcinoma 26 and retardation of their metastatic ability by sialidase overexpression. Int. J. Cancer 2002, 97, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Tokuyama, S.; Moriya, S.; Taniguchi, S.; Yasui, A.; Miyazaki, J.; Orikasa, S.; Miyagi, T. Suppression of pulmonary metastasis in murine B16 melanoma cells by transfection of a sialidase cDNA. Int. J. Cancer 1997, 73, 410–415. [Google Scholar] [CrossRef] [PubMed]
- Miyagi, T. Aberrant expression of sialidase and cancer progression. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2008, 84, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Uemura, T.; Shiozaki, K.; Yamaguchi, K.; Miyazaki, S.; Satomi, S.; Kato, K.; Sakuraba, H.; Miyagi, T. Contribution of sialidase NEU1 to suppression of metastasis of human colon cancer cells through desialylation of integrin beta4. Oncogene 2009, 28, 1218–1229. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.; Forouhar, F.; Gao, Z.; Ramiah, A.; Moniz, H.; Thieker, D.; Seetharaman, J.; Milaninia, S.; Su, M.; Veillon, L.; et al. Enzymatic basis for N-glycan sialylation: Structure of rat ST6GAL1 reveals conserved and unique features for glycan sialylation. Glycobiology 2013, 23, 1373–1374. [Google Scholar]
- Rao, F.V.; Rich, J.R.; Rakic, B.; Buddai, S.; Schwartz, M.F.; Johnson, K.; Bowe, C.; Wakarchuk, W.W.; DeFrees, S.; Withers, S.G.; et al. Structural insight into mammalian sialyltransferases. Nat. Struct. Mol. Biol. 2009, 16, 1186–1188. [Google Scholar] [CrossRef] [PubMed]
- Joziasse, D.H.; Schiphorst, W.E.C.M.; Vandeneijnden, D.H.; Vankuik, J.A.; Vanhalbeek, H.; Vliegenthart, J.F.G. Branch Specificity of Bovine Colostrum Cmp-Sialic Acid: N-Acetyllactosaminide Alpha-2,6-Sialyltransferase. Interaction with Biantennary Oligosaccharides and Glycopeptides of N-Glycosylproteins. J. Biol. Chem. 1985, 260, 714–719. [Google Scholar] [PubMed]
- Joziasse, D.H.; Schiphorst, W.E.C.M.; Vandeneijnden, D.H.; Vankuik, J.A.; Vanhalbeek, H.; Vliegenthart, J.F.G. Branch Specificity of Bovine Colostrum Cmp-Sialic Acid: Gal-Beta-1,4glcnac-R Alpha-2,6-Sialyltransferase. Sialylation of Biantennary, Triantennary, and Tetraantennary Oligosaccharides and Glycopeptides of the N-Acetyllactosamine Type. J. Biol. Chem. 1987, 262, 2025–2033. [Google Scholar] [PubMed]
- Guo, H.B.; Nairn, A.; Harris, K.; Randolph, M.; Alvarez-Manilla, G.; Moremen, K.; Pierce, M. Loss of expression of N-acetylglucosaminyltransferase Va results in altered gene expression of glycosyltransferases and galectins. FEBS Lett. 2008, 582, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.H.; Lee, W.L.; Lee, Y.R.; Juang, C.M.; Chen, Y.J.; Chao, H.T.; Tsai, Y.C.; Yuan, C.C. Enhanced expression of alpha 2,6-sialyltransferase ST6Gal I in cervical squamous cell carcinoma. Gynecol. Oncol. 2003, 89, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, I.; Futakawa, S.; Oka, R.; Ogawa, K.; Marth, J.D.; Miyoshi, E.; Taniguchi, N.; Hashimoto, Y.; Kitazume, S. Beta-galactoside alpha2,6-sialyltransferase I cleavage by BACE1 enhances the sialylation of soluble glycoproteins. A novel regulatory mechanism for alpha2,6-sialylation. J. Biol. Chem. 2007, 282, 34896–34903. [Google Scholar] [CrossRef] [PubMed]
- Bast, B.J.E.G.; Zhou, L.J.; Freeman, G.J.; Colley, K.J.; Ernst, T.J.; Munro, J.M.; Tedder, T.F. The Hb-6, Cdw75, and Cd76 Differentiation Antigens Are Unique Cell-Surface Carbohydrate Determinants Generated by the Beta-Galactoside Alpha-2,6-Sialyltransferase. J. Cell Biol. 1992, 116, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Baumann, H.; Gauldie, J. The Acute-Phase Response. Immunol. Today 1994, 15, 74–80. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, J.; Gu, J. Significance of β-Galactoside α2,6 Sialyltranferase 1 in Cancers. Molecules 2015, 20, 7509-7527. https://doi.org/10.3390/molecules20057509
Lu J, Gu J. Significance of β-Galactoside α2,6 Sialyltranferase 1 in Cancers. Molecules. 2015; 20(5):7509-7527. https://doi.org/10.3390/molecules20057509
Chicago/Turabian StyleLu, Jishun, and Jianguo Gu. 2015. "Significance of β-Galactoside α2,6 Sialyltranferase 1 in Cancers" Molecules 20, no. 5: 7509-7527. https://doi.org/10.3390/molecules20057509
APA StyleLu, J., & Gu, J. (2015). Significance of β-Galactoside α2,6 Sialyltranferase 1 in Cancers. Molecules, 20(5), 7509-7527. https://doi.org/10.3390/molecules20057509





