Characterization of a (2R,3R)-2,3-Butanediol Dehydrogenase from Rhodococcus erythropolis WZ010
Abstract
:1. Introduction
2. Results and Discussion
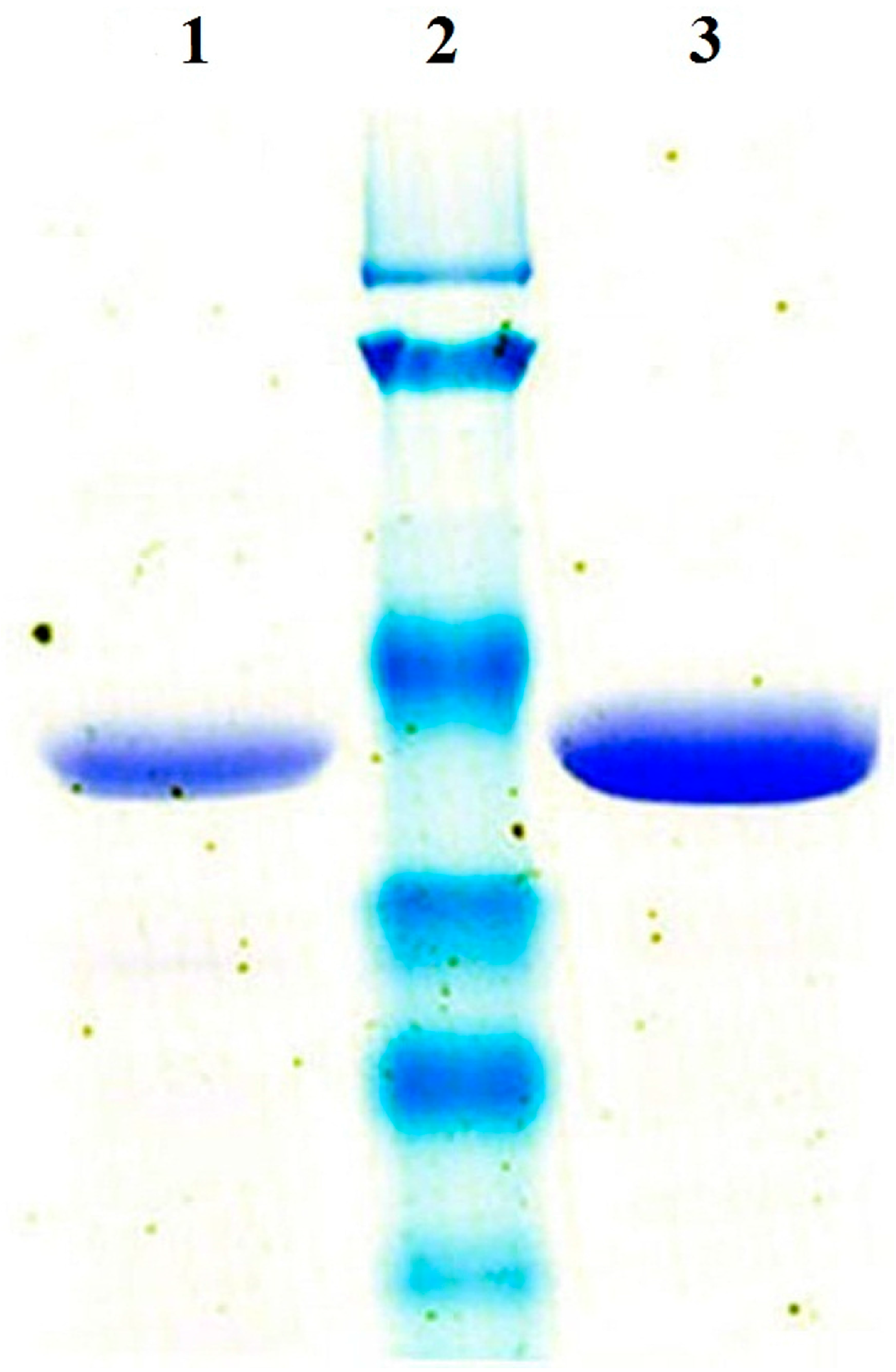
2.1. Over-Expression and Purification of ReBDH
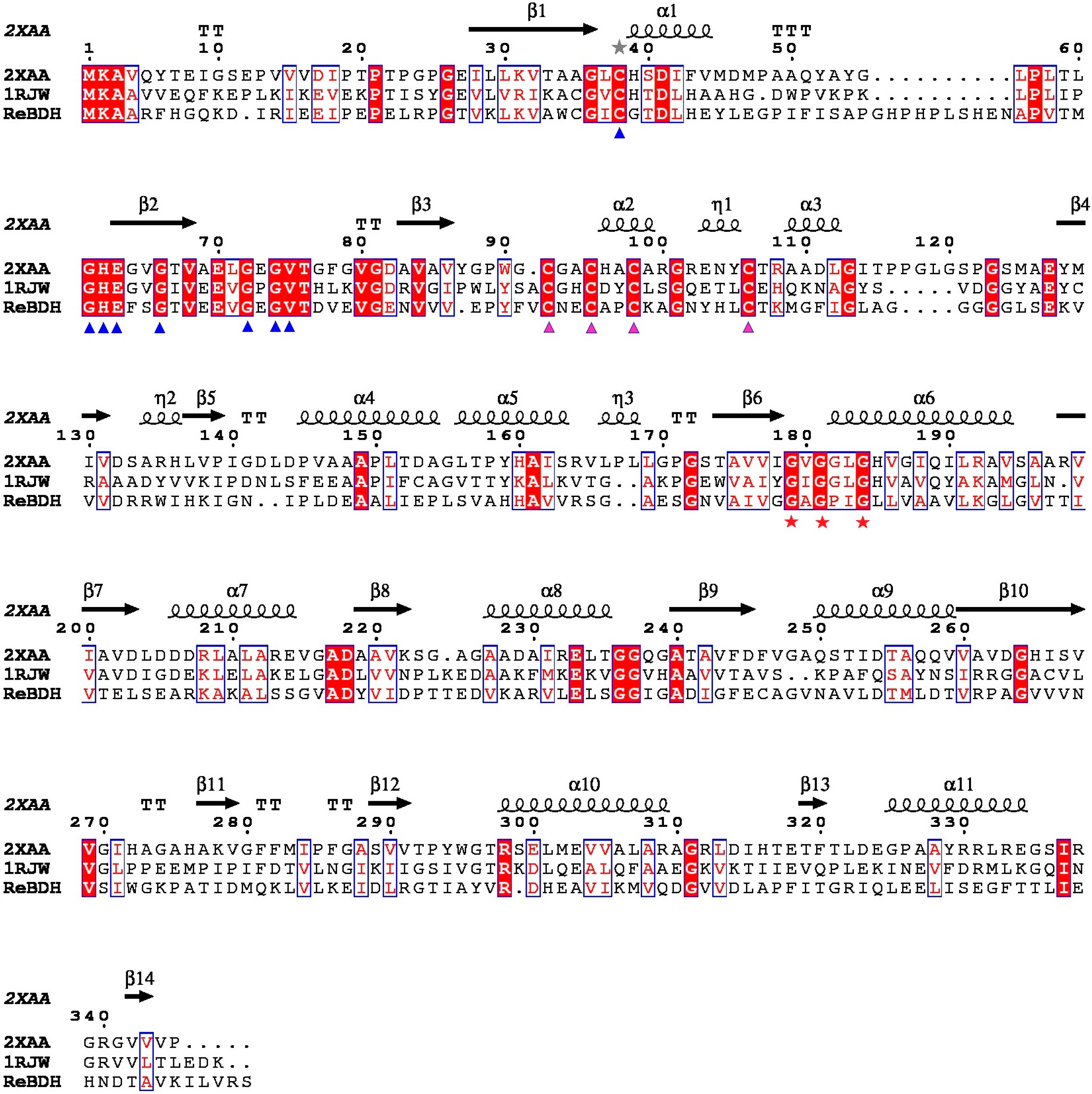
2.2. Sequence and Structure Analysis of ReBDH
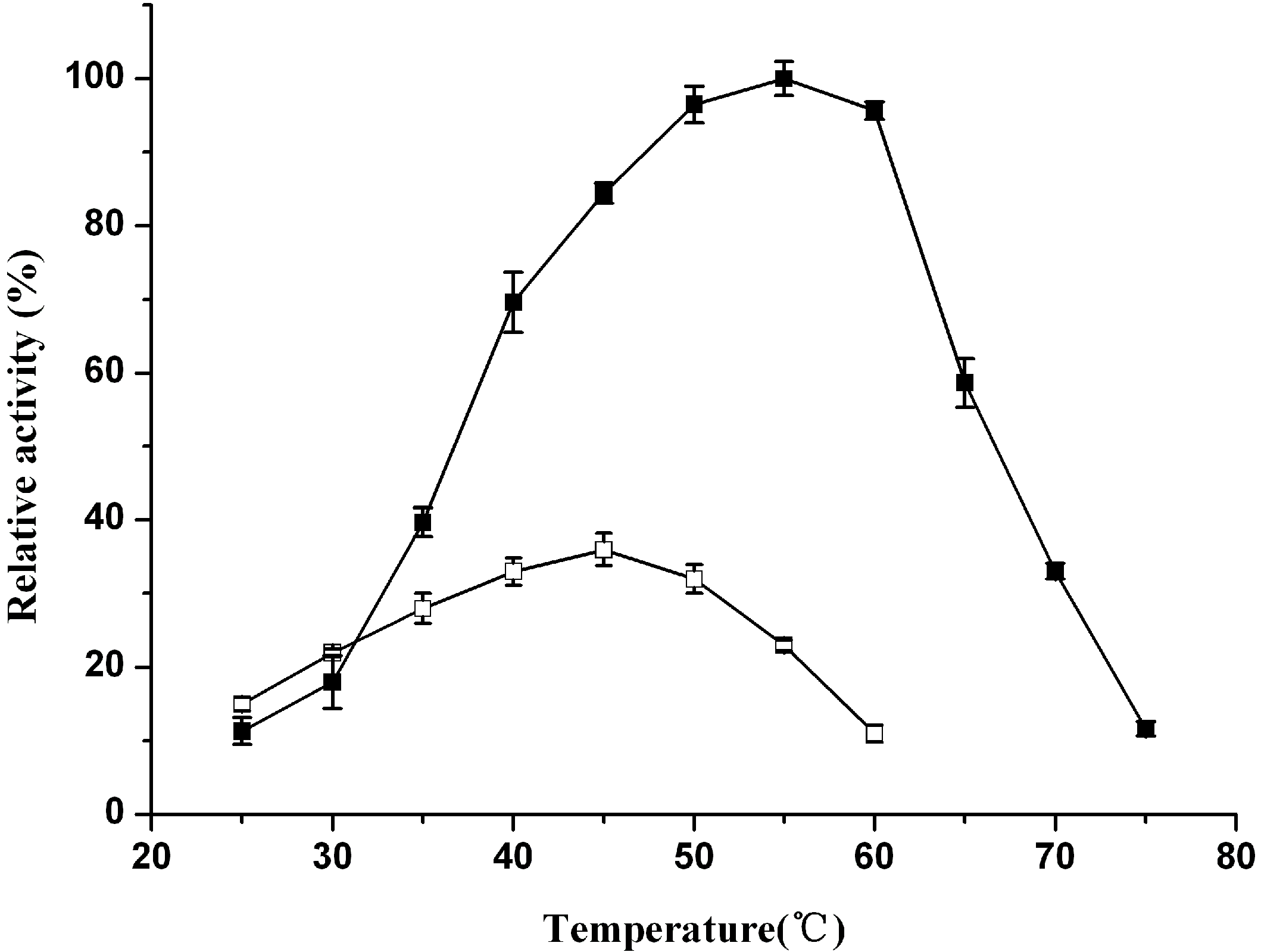
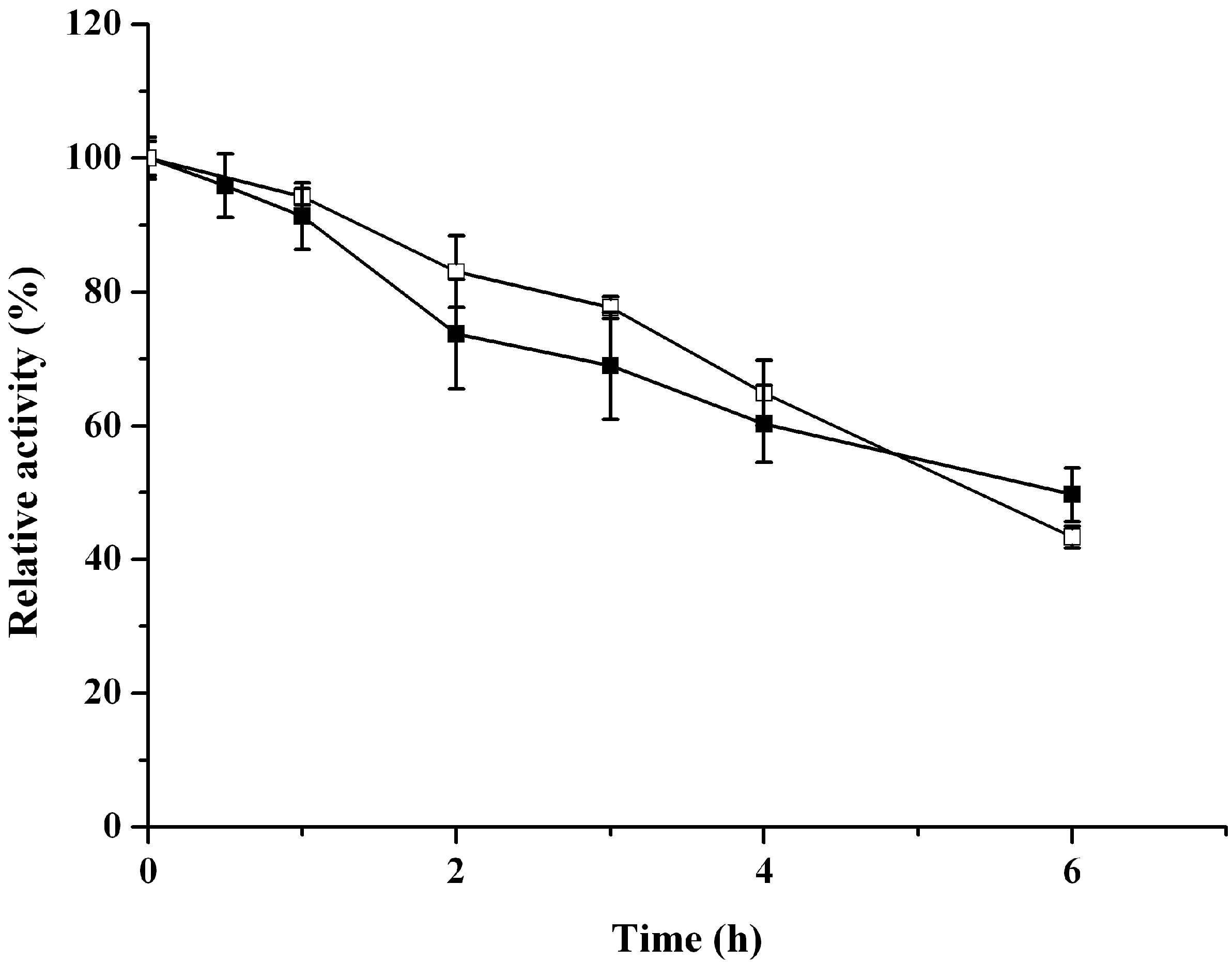
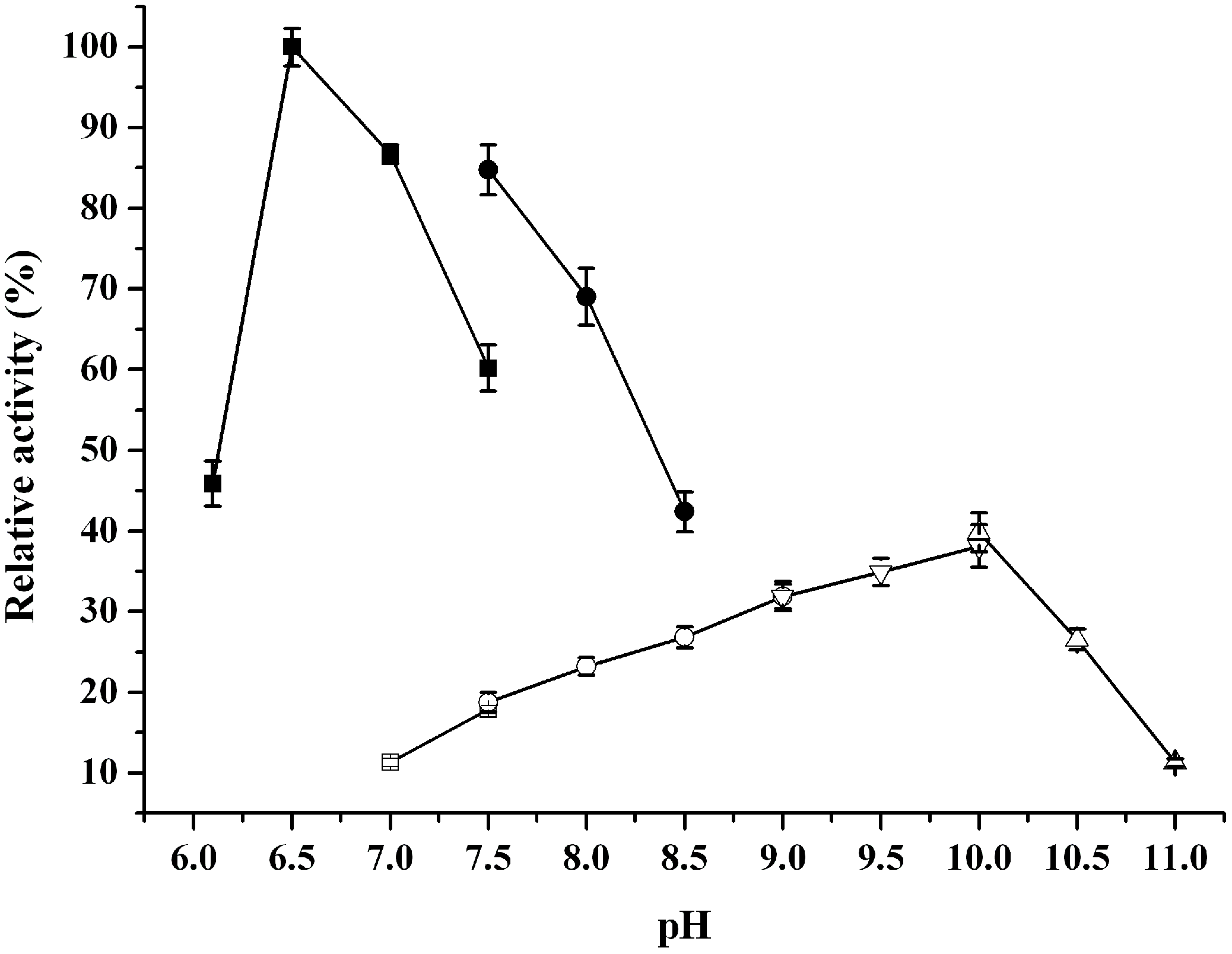
2.3. Catalytic Properties of ReBDH
| Alcohols | Relative Activity (100%) | Ketones | Relative Activity (100%) |
|---|---|---|---|
| 2,3-Butanediol a | 100 b ± 2.7 | Acetoin | 100 c ± 4.3 |
| Acetoin | 0 | Diacetyl | 69.3 ± 5.7 |
| (2R,3R)-2,3-Butanediol | 69.5 ± 5.9 | 2,2,2-Trifluoroacetophenone | 28.0 ± 1.8 |
| (2S,3S)-2,3-Butanediol | 0 | 3-Methyl-2-butenal | 16.7 ± 1.3 |
| (R)-1-Phenyl-1,2-ethanediol | 32.2 ± 2.6 | 2-Octanone | 9.3 ± 1.0 |
| (S)-1-Phenyl-1,2-ethanediol | 0 | 2-Hydroxyacetophenone | 8.7 ± 0.4 |
| Glycerol | 16.3 ± 0.5 | Acetophenone | 2.5 ± 0.2 |
| 1,3-Butanediol | 14.7 ± 1.0 | 4-Hydroxy-2-butanone | 0.9 ± 0.2 |
| Ethyl 4-chloro-3-hydroxybutyrate | 12.2 ± 0.6 | ||
| Sodium lactate | 10.2 ± 1.1 | ||
| 2-Butanol | 5.1 ± 0.3 | ||
| Ethyl lactate | 4.6 ± 0.3 | ||
| Isopropanol | 3.6 ± 0.4 | ||
| 1-Butanol | 3.1 ± 0.3 | ||
| Cyclohexanol | 1.5 ± 0.2 | ||
| 2-Pentanol | 1.5 ± 0.2 | ||
| 2-Octanol | 0.5 ± 0.1 |
| Compounds | Concentrations (mM) | Relative activity a (%) |
|---|---|---|
| Control | 0 | 100 b ± 4.3 |
| KCl | 10 | 138.9 ± 5.5 |
| NaCl | 10 | 102.2 ± 1.8 |
| MgCl2 | 10 | 97.0 ± 4.9 |
| CaCl2 | 1 | 89.3 ± 5.3 |
| BaCl2 | 1 | 85.8 ± 2.2 |
| MnCl2 | 1 | 81.1 ± 1.6 |
| CoCl2 | 1 | 79.5 ± 3.1 |
| AlCl3 | 1 | 21.6 ± 3.4 |
| EDTA | 1 | 9.9 ± 0.8 |
| ZnCl2 | 1 | 3.4 ± 0.3 |
| FeCl2 | 1 | 0 |
| CuSO4 | 1 | 0 |
| AgNO3 | 1 | 0 |
| Organic Solvents | Concentrations (%) | Relative Activity (100%) |
|---|---|---|
| Control | 0 | 100 a ± 4.3 |
| DMSO | 10 | 161.2 ± 2.8 |
| 20 | 122.5 ± 3.4 | |
| 30 | 83.8 ± 3.1 | |
| 40 | 8.4 ± 0.3 | |
| Acetone | 10 | 38.3 ± 1.1 |
| Methanol | 10 | 14.0 ± 0.3 |
| Ethanol | 10 | 3.7 ± 0.2 |
| Acetonitrile | 10 | 2.4 ± 0.2 |
2.4. Kinetic Parameters of ReBDH
| Substrate | Cosubstrate (mM) | Apparent Km (mM) | Vmax (U/mg) | Kcat (s−1) | Kcat/Km (s−1·M−1) |
|---|---|---|---|---|---|
| (2 R,3R)-2,3-Butanediol | NAD+ (0.64) | 0.58 ± 0.05 | 6.65 ± 0.18 | 4.28 ± 0.12 | 7379 ± 860 |
| NAD+ | (2 R,3R)-2,3-butanediol (50) | 1.04 ± 0.11 | 15.96 ± 1.07 | 10.27 ± 0.69 | 9875 ± 1777 |
| Diacetyl | NADH (0.4) | 0.1 ± 0.01 | 9.49 ± 0.22 | 6.11 ± 0.14 | 61100 ± 7650 |
| NADH | Diacetyl (50) | 0.08 ± 0.01 | 8.92 ± 0.12 | 5.73 ± 0.08 | 71625 ± 10078 |
2.5. Stereoselectivity of ReBDH
| Substrate (mM) | Conversion (%) | Products | Yield (%) | e.e. a (%) |
|---|---|---|---|---|
| Diacetyl (60) | 73.6 ± 4.0 | ( R)-acetoin | 35.7 ± 2.2 | 100 b |
| (2 R,3R)-2,3-butanediol | 34.2 ± 1.7 | 100 b | ||
| Racemic acetoin (60) | 42.6 ± 3.6 | (2 R,3R)-2,3-butanediol | 24.7 ± 2.4 | 100 c |
| meso-2,3-butanediol | 14 ± 1.1 | 100 c |
3. Experimental Section
3.1. Materials Used
3.2. Microorganisms and Growth Conditions
3.3. Construction of the Expression Plasmids pEASY-E1-rebdh and pEASY-E2-rebdh
3.4. Expression and Purification of ReBDH with or without N-terminal His-tag
3.5. Size Exclusion Chromatography and Structural Analysis
3.6. Determination of Zinc Content in ReBDH
3.7. Activity Assays of ReBDH
3.8. Catalytic Properties of ReBDH
3.9. Asymmetric Reduction of Diacetyl and Acetoin Catalyzed by ReBDH
3.10. Nucleotide Sequence Accession Number
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, Z.; Song, Q.; Yu, M.; Wang, Y.; Xiong, B.; Zhang, Y.; Zheng, J.; Ying, X. Characterization of a stereospecific acetoin(diacetyl) reductase from Rhodococcus erythropolis WZ010 and its application for the synthesis of (2S,3S)-2,3-butanediol. Appl. Microbiol. Biotechnol. 2014, 98, 641–650. [Google Scholar] [CrossRef] [PubMed]
- Zeng, A.; Sabra, W. Microbial production of diols as platform chemicals: recent progresses. Curr. Opin. Biotechnol. 2011, 22, 749–757. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wang, Y.; Zhang, L.; Ma, C.; Wang, A.; Tao, F.; Xu, P. Biocatalytic production of (2S,3S)-2,3-butanediol from diacetyl using whole cells of engineered Escherichia coli. Bioresour. Technol. 2012, 115, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Lee, C.; Liao, J.C. Enantioselective synthesis of pure (R,R)-2,3-butanediol in Escherichia coli with stereospecific secondary alcohol dehydrogenases. Org. Biomol. Chem. 2009, 7, 3914–3917. [Google Scholar] [CrossRef] [PubMed]
- Takeda, M.; Muranushi, T.; Inagaki, S.; Nakao, T.; Motomatsu, S.; Suzuki, I.; Koizumi, J. Identification and characterization of a mycobacterial (2R,3R)-2,3-butanediol dehydrogenase. Biosci. Biotechnol. Biochem. 2011, 75, 2384–2389. [Google Scholar] [CrossRef] [PubMed]
- González, E.; Fernández, M.R.; Larroy, C.; Solà, L.; Pericàs, M.A.; Parés, X.; Biosca, J.A. Characterization of a (2R,3R)-2,3-butanediol dehydrogenase as the Saccharomyces cerevisiae YAL060W gene product. J. Biol. Chem. 2000, 275, 35876–35885. [Google Scholar] [CrossRef] [PubMed]
- Raedts, J.; Siemerink, M.A.J.; Levisson, M.; van der Oost, J.; Kengen, S.W.M. Molecular characterization of an NADPH-dependent acetoin reductase/2,3-butanediol dehydrogenase from Clostridium beijerinckii NCIMB 8052. Appl. Environ. Microbiol. 2014, 80, 2011–2020. [Google Scholar] [CrossRef] [PubMed]
- Ying, X.; Ma, K. Characterization of a zinc-containing alcohol dehydrogenase with stereoselectivity from the hyperthermophilic archaeon Thermococcus guaymasensis. J. Bacteriol. 2011, 193, 3009–3019. [Google Scholar] [CrossRef] [PubMed]
- Giovannini, P.P.; Medici, A.; Bergamini, C.M.; Rippa, M. Properties of diacetyl(acetoin) reductases from Bacillus stearothermophilus. Bioorg. Med. Chem. 1996, 4, 1197–1201. [Google Scholar] [CrossRef] [PubMed]
- Park, J.M.; Hong, W.K.; Lee, S.M.; Heo, S.Y.; Jung, Y.R.; Kang, I.Y.; Oh, B.R.; Seo, J.W.; Kim, C.H. Identification and characterization of a short-chain acyl dehydrogenase from Klebsiella pneumoniae and its application for high-level production of l-2,3-butanediol. J. Ind. Microbiol. Biotechnol. 2014, 41, 1425–1433. [Google Scholar] [CrossRef] [PubMed]
- Ui, S.; Okajima, Y.; Minura, A.; Kanai, H.; Kobayashi, T.; Kudo, T. Sequence analysis of the gene for and characterization of d-acetoin forming meso-2,3-butanediol dehydrogenase of Klebsiella pneumoniae expressed in Escherichia coli. J. Ferment. Bioeng. 1997, 83, 32–37. [Google Scholar] [CrossRef]
- Ui, S.; Okajima, Y.; Mimura, A.; Dohmae, N.; Takio, K.; Ohkuma, M.; Kudo, T. Cloning, expression and nucleotide sequence of the L-2,3-butanediol dehydrogenase gene from Brevibacterium saccharolyticum C-1012. J. Ferment. Bioeng. 1998, 86, 290–295. [Google Scholar] [CrossRef]
- Shen, M.; Ji, X.; Nie, Z.; Xia, Z.; Yang, H.; Huang, H. Biotechnological production of 2,3-butanediol stereoisomers: synthetic mechanism and realized methods. Chin. J. Catal. 2013, 34, 351–360. [Google Scholar]
- Qi, G.; Kang, Y.; Li, L.; Xiao, A.; Zhang, S.; Wen, Z.; Xu, D.; Chen, S. Deletion of meso-2,3-butanediol dehydrogenase gene budC for enhanced d-2,3-butanediol production in Bacillus licheniformis. Biotechnol. Biofuels 2014, 7, 16. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, L.; Ma, C.; Gao, C.; Tao, F.; Xu, P. Engineering of cofactor regeneration enhances (2S,3S)-2,3-butanediol production from diacetyl. Sci. Rep. 2013, 3, 2643. [Google Scholar] [PubMed]
- Xu, Y.; Chu, H.; Gao, C.; Tao, F.; Zhou, Z.; Li, K.; Li, L.; Ma, C.; Xu, P. Systematic metabolic engineering of Escherichia coli for high-yield production of fuel bio-chemical 2,3-butanediol. Metab. Eng. 2014, 23, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, S.; Hattori, S.; Hata, H.; Yamada, H. One-step microbial conversion of a racemic mixture of pantoyl lactone to optically active d-(−)-pantoyl lactone. Appl. Environ. Microbiol. 1987, 53, 519–522. [Google Scholar] [PubMed]
- Wang, Y.; Li, J.; Wu, Q.; Zhu, D. Microbial stereospecific reduction of 3-quinuclidinone with newly isolated Nocardia sp. and Rhodococcus erythropolis. J. Mol. Catal. B Enzym. 2012, 88, 14–19. [Google Scholar] [CrossRef]
- Yang, C.; Ying, X.; Yu, M.; Zhang, Y.; Xiong, B.; Song, Q.; Wang, Z. Towards the discovery of alcohol dehydrogenases: NAD(P)H fluorescence-based screening and characterization of the newly isolated Rhodococcus erythropolis WZ010 in the preparation of chiral aryl secondary alcohols. J. Ind. Microbiol. Biotechnol. 2012, 39, 1431–1443. [Google Scholar] [CrossRef] [PubMed]
- Abokitse, K.; Hummel, W. Cloning, sequence analysis, and heterologous expression of the gene encoding a (S)-specific alcohol dehydrogenase from Rhodococcus erythropolis DSM 43297. Appl. Microbiol. Biotechnol. 2003, 62, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Hummel, W.; Abokitse, K.; Drauz, K.; Rollmann, C.; Gröger, H. Towards a large-scale asymmetric reduction process with isolated enzymes: expression of an (S)-alcohol dehydrogenase in E. coli and studies on the synthetic potential of this biocatalyst. Adv. Synth. Catal. 2003, 345, 153–159. [Google Scholar] [CrossRef]
- Kataoka, M.; Nakamura, Y.; Urano, N.; Ishige, T.; Shi, G.; Kita, S.; Sakamoto, K.; Shimizu, S. A novel NADP+-dependent L-1-amino-2-propanol dehydrogenase from Rhodococcus erythropolis MAK154: A promising enzyme for the production of double chiral aminoalcohols. Lett. Appl. Microbiol. 2006, 43, 430–435. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Chen, C.; Chen, H.; Hsu, W. Enantioselective synthesis of (S)-phenylephrine by whole cells of recombinant Escherichia coli expressing the amino alcohol dehydrogenase gene from Rhodococcus erythropolis BCRC 10909. Process Biochem. 2010, 45, 1529–1536. [Google Scholar] [CrossRef]
- Martinez-Rojas, E.; Kurt, T.; Schmidt, U.; Meyer, V.; Garbe, L. A bifunctional enzyme from Rhodococcus erythropolis exhibiting secondary alcohol dehydrogenase-catalase activities. Appl. Microbiol. Biotechnol. 2014, 98, 9249–9258. [Google Scholar] [CrossRef] [PubMed]
- Si, D.; Urano, N.; Nozaki, S.; Honda, K.; Shimizu, S.; Kataoka, M. L-Pantoyl lactone dehydrogenase from Rhodococcus erythropolis: genetic analyses and application to the stereospecific oxidation of l-pantoyl lactone. Appl. Microbiol. Biotechnol. 2012, 95, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Jia, H.; Li, Y.; Jia, L.; Ma, Y.; Wei, P. Cloning, expression and characterization of chiral alcohol dehydrogenase from Rhodococcus erythropolis ATCC 4277. Acta Microbiol. Sin. 2012, 52, 83–89. [Google Scholar]
- Sekine, M.; Tanikawa, S.; Omata, S.; Saito, M.; Fujisawa, T.; Tsukatani, N.; Tajima, T.; Sekigawa, T.; Kosugi, H.; Matsuo, Y.; et al. Sequence analysis of three plasmids harboured in Rhodococcus erythropolis strain PR4. Environ. Microbiol. 2006, 8, 334–346. [Google Scholar] [CrossRef] [PubMed]
- Karabec, M.; Łyskowski, A.; Tauber, K.C.; Steinkellner, G.; Kroutil, W.; Grogan, G.; Gruber, K. Structural insights into substrate specificity and solvent tolerance in alcohol dehydrogenase ADH-“A” from Rhodococcus ruber DSM 44541. Chem. Commun. 2010, 46, 6314–6316. [Google Scholar] [CrossRef]
- Ceccarelli, C.; Liang, Z.X.; Strickler, M.; Prehna, G.; Goldstein, B.M.; Klinman, J.P.; Bahnson, B.J. Crystal structure and amide H/D exchange of binary complexes of alcohol dehydrogenase from Bacillus stearothermophilus: Insight into thermostability and cofactor binding. Biochemistry 2004, 43, 5266–5277. [Google Scholar] [CrossRef] [PubMed]
- Korkhin, Y.; Kalb(Gilboa), A.J.; Peretz, M.; Bogin, O.; Burstein, Y.; Frolow, F. NADP-dependent bacterial alcohol dehydrogenases:crystal structure, cofactor-binding and cofactor specificity of the ADHs of Clostridium beijerinckii and Thermoanaerobacter brockii. J. Mol. Biol. 1998, 278, 967–981. [Google Scholar] [CrossRef] [PubMed]
- Ying, X.; Wang, Y.; Xiong, B.; Wu, T.; Xie, P.; Yu, M.; Wang, Z. Characterization of an allylic/benzyl alcohol dehydrogenase from Yokenella sp. strain WZY002, an organism potentially useful for the synthesis of α,β-unsaturated alcohols from allylic aldehydes and ketones. Appl. Environ. Microbiol. 2014, 80, 2399–2409. [Google Scholar] [CrossRef] [PubMed]
- Tani, A.; Sakai, Y.; Ishige, T.; Kato, N. Thermostable NADP+-dependent medium-chain alcohol dehydrogenase from Acinetobacter sp. strain M-1: Purification and characterization and gene expression in Escherichia coli. Appl. Environ. Microbiol. 2000, 66, 5231–5235. [Google Scholar] [CrossRef] [PubMed]
- Uthoff, S.; Steinbüchel, A. Purification and characterization of an NAD+-dependent XylB-like aryl alcohol dehydrogenase identified in Acinetobacter baylyi ADP1. Appl. Environ. Microbiol. 2012, 78, 8743–8752. [Google Scholar] [CrossRef] [PubMed]
- Lavandera, I.; Kern, A.; Schaffenberger, M.; Gross, J.; Glieder, A.; de Wildeman, S.; Kroutil, W. An exceptionally DMSO tolerant alcohol dehydrogenase for the stereoselective reduction of ketones. ChemSusChem 2008, 1, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Jung, M.; Ng, C.Y.; Song, H.; Lee, J.; Oh, M. Deletion of lactate dehydrogenase in Enterobacter aerogenes to enhance 2,3-butanediol production. Appl. Microbiol. Biotechnol. 2012, 95, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Madden, T.L.; Schäffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSI-BLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef] [PubMed]
- Gouet, P.; Courcelle, E.; Stuart, D.I.; Metoz, F. ESPript: Analysis of multiple sequence alignments in PostScript. Bioinformatics 1999, 15, 305–308. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J. CLUSTALW: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Slusarczyk, H.; Felber, S.; Kula, M.; Pohl, M. Stabilization of NAD-dependent formate dehydrogenase from Candida boidinii by site-directed mutagenesis of cysteine residues. Eur. J. Biochem. 2000, 267, 1280–1289. [Google Scholar] [CrossRef] [PubMed]
- Matsuyama, A.; Yamamoto, H.; Kawada, N.; Kobayashi, Y. Industrial production of (R)-1,3-butanediol by new biocatalysts. J. Mol. Catal. B Enzym. 2001, 11, 513–521. [Google Scholar] [CrossRef]
- Nie, Y.; Xu, Y.; Mu, X.Q.; Wang, H.Y.; Yang, M.; Xiao, R. Purification, characterization, gene cloning, and expression of a novel alcohol dehydrogenase with anti-Prelog stereospecificity from Candida parapsilosis. Appl. Environ. Microbiol. 2007, 73, 3759–3764. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds in miligram amounts are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, M.; Huang, M.; Song, Q.; Shao, J.; Ying, X. Characterization of a (2R,3R)-2,3-Butanediol Dehydrogenase from Rhodococcus erythropolis WZ010. Molecules 2015, 20, 7156-7173. https://doi.org/10.3390/molecules20047156
Yu M, Huang M, Song Q, Shao J, Ying X. Characterization of a (2R,3R)-2,3-Butanediol Dehydrogenase from Rhodococcus erythropolis WZ010. Molecules. 2015; 20(4):7156-7173. https://doi.org/10.3390/molecules20047156
Chicago/Turabian StyleYu, Meilan, Meijuan Huang, Qingqing Song, Jianzhong Shao, and Xiangxian Ying. 2015. "Characterization of a (2R,3R)-2,3-Butanediol Dehydrogenase from Rhodococcus erythropolis WZ010" Molecules 20, no. 4: 7156-7173. https://doi.org/10.3390/molecules20047156
APA StyleYu, M., Huang, M., Song, Q., Shao, J., & Ying, X. (2015). Characterization of a (2R,3R)-2,3-Butanediol Dehydrogenase from Rhodococcus erythropolis WZ010. Molecules, 20(4), 7156-7173. https://doi.org/10.3390/molecules20047156





