Covalent Modification of Human Serum Albumin by the Natural Sesquiterpene Lactone Parthenolide
Abstract
:1. Introduction
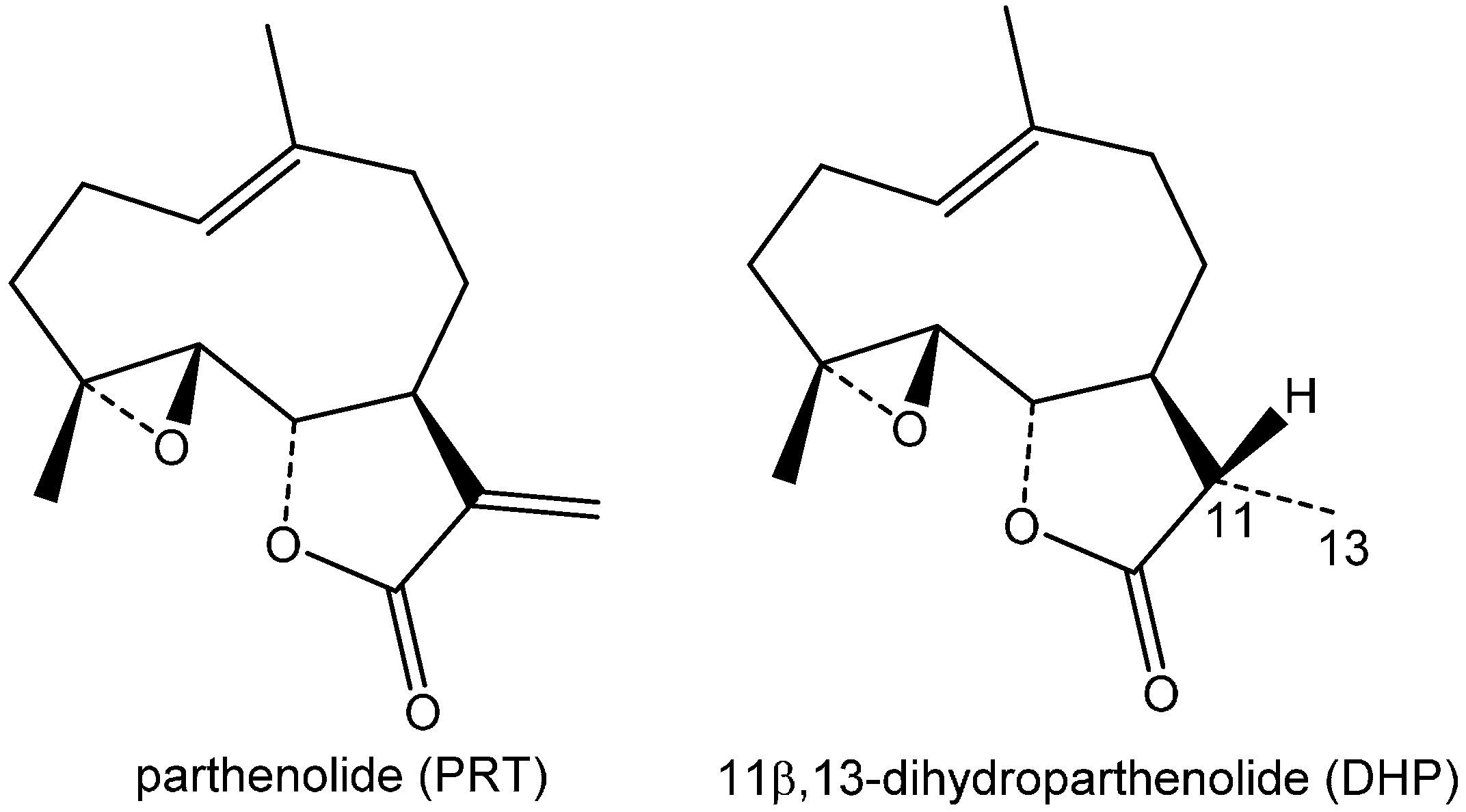
2. Results and Discussion
2.1. LC/MS-Analysis of Untreated HSA Digest
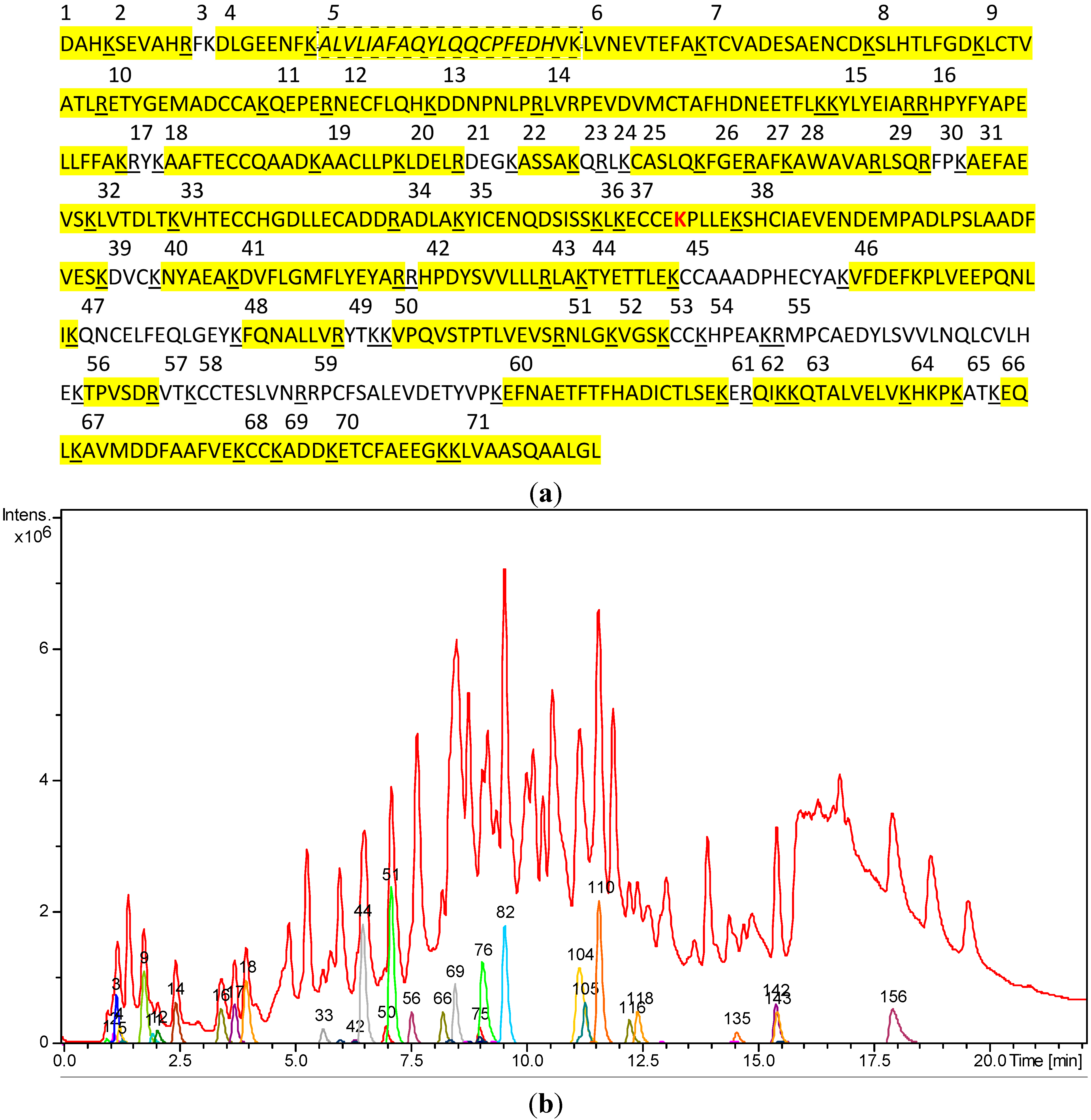
| Tryptic Fragment No. | Theoretical Molecular Mass of [M+H]+ | Molecular Mass of [M+H]+ after Deconvolution | Deviation (mDa/ppm) | Charge States (n) of Detected Ions [M+nH]n+ | Retention Time (min) | Compound No. (Figure 2b) |
|---|---|---|---|---|---|---|
| 1 | 470.2358 | 470.2340 | 1.8/3.8 | 1 | 0.99 | 1 |
| 2 | 698.3580 | 698.3579 | 0.1/1.0 | 1, 2 | 1.13 | 2 |
| 3 | 294.1812 | n.d. | ||||
| 4 | 951.4418 | 951.4377 | 4.1/4.3 | 1, 2 | 3.72 | 17 |
| 5 | 2433.2635 | n.d. | ||||
| 5-SS-5 | 4863.5037 | 4863.5344 | 30.7/6.3 | 4, 5, 6, 7 | 17.87 | 156 |
| 5-SS-Cys | 2552.2676 | 2552.2778 | 10.2/4.0 | 3, 4, 5 | 15.38 | 143 |
| 5-SS-hCys | 2566.2832 | 2566.3146 | 31/12.1 | 3, 4, 5 | 9.00 | 75 |
| 6 | 1149.6150 | 1149.6221 | 7.1/6.2 | 1,2 | 7.10 | 51 |
| 7 | 1382.5195 | 1382.5358 | 16.3/11.8 | 1, 2 | 1.19 | 3 |
| 8 | 1017.5364 | 1017.5353 | 1.1/1.1 | 1, 2, 3 | 6.98 | 50 |
| 9 + 10 + 11 + 12 | 3848.7117 | 3848.7399 | 28.2/7.3 | 5, 6 | 6.32 | 42 |
| 13 | 940.4483 | 940.4493 | 1.0/1.1 | 1,2 | 1.78 | 9 |
| 14 + 18 + 19 | 4560.1368 | 4560.1741 | 37.3/8.2 | 4, 5, 6, 7, 8 | 9.53 | 82 |
| 15 | 927.4934 | 927.4921 | 1.3/1.4 | 1, 2 | 6.49 | 44 |
| 16 | 1742.8941 | 1742.8983 | 4.2/2.4 | 2, 3 | 3.43 | 135 |
| 17 | 310.1761 | n.d. | ||||
| 20 | 645.3566 | 645.3621 | 5.5/8.5 | 1, 2 | 1.96 | 11 |
| 21 | 448.2038 | n.d. | ||||
| 22 | 463.2511 | 463.2496 | 1.5/3.2 | 1 | 0.99 | 1 |
| 23 | 303.1775 | n.d. | ||||
| 24 | 260.1969 | n.d. | ||||
| 25 + 33 | 2560.0676 | 2560.0647 | 2.9/1,1 | 4, 5 | 5.64 | 33 |
| 26 | 508.2514 | 508.2525 | 1.1/2.2 | 1 | 1.25 | 4 |
| 27 | 365.2183 | 365.2185 | 0.2/0.5 | 1 | 1.19 | 3 |
| 28 | 673.3780 | 673.3788 | 0.8/1.2 | 1, 2 | 3.43 | 16 |
| 29 | 503.2936 | 503.2932 | 0.4/0.8 | 1 | 1.13 | 2 |
| 30 | 391.2340 | 391.2326 | 1.4/3.6 | 1 | 1.32 | 5 |
| 31 | 880.4411 | 880.4411 | 0/0 | 1, 2 | 2.46 | 14 |
| 32 | 789.4716 | 789.4691 | 2.5/3.2 | 1, 2 | 3.98 | 18 |
| 34 | 517.2980 | 517.2995 | 1.5/2.9 | 1 | 1.19 | 3 |
| 35 + 36 + 37 + 38 | 5730.6464 | 5,730.6533 | 6.9/1.2 | 4, 5, 6, 7, 8, 9 | 11.14 | 104 |
| 39 | 464.2173 | n.d. | ||||
| 40 | 695.3359 | 695.3439 | 8.0/11.5 | 1 | 1.19 | 3 |
| 41 | 1623.7876 | 1623.7938 | 6.2/3.8 | 2, 3 | 15.36 | 142 |
| 42 | 1311.7419 | 1311.7362 | 5.7/4.3 | 1, 2, 3 | 12.39 | 118 |
| 43 | 331.2340 | 331.2354 | 1.4/4.2 | 1 | 1.19 | 3 |
| 44 | 984.4884 | 984.4889 | 0.5/0.5 | 1, 2 | 2.07 | 12 |
| 45 | 1381.5334 | n.d. | ||||
| 46 | 2045.0954 | 2,045.1124 | 17.0/8.3 | 2, 3, 4 | 11.56 | 110 |
| 47 | 1600.7312 | n.d. | ||||
| 48 | 960.5630 | 960.5584 | 4.6/4.8 | 1, 2 | 7.54 | 56 |
| 49 | 411.2238 | n.d. | ||||
| 50 | 1511.8428 | 1511.8521 | 9.3/6.2 | 2, 3 | 8.21 | 66 |
| 51 | 431.2613 | 431.2608 | 0.5/1.2 | 1 | 1.19 | 3 |
| 52 | 390.2350 | 390.2300 | 5.0/17.2 | 1 | 0.99 | 1 |
| 53 | 353.1312 | n.d. | ||||
| 54 | 581.3052 | n.d. | ||||
| 55 | 2404.1709 | n.d. | ||||
| 56 | 674.3468 | 674.3529 | 6.1/9.0 | 1 | 1.19 | 3 |
| 57 | 347.2289 | n.d. | ||||
| 58 | 1024.4550 | n.d. | ||||
| 59 | 1853.9102 | n.d. | ||||
| 60 + 68 + 69 + 70 + 71 | 3563.5102 | 3,563.6537 | 143/40 | 4, 5 | 12.21 | 116 |
| 61 | 304.1615 | n.d. | ||||
| 62 | 388.2554 | 388.2550 | 0.4/1.0 | 1 | 1.19 | 3 |
| 63 | 1000.6037 | 1000.6029 | 0.8/0.8 | 1, 2 | 8.47 | 69 |
| 64 | 509.3194 | 509.3184 | 1.0/2.0 | 1 | 0.99 | 1 |
| 65 | 319.1976 | n.d. | ||||
| 66 | 517.2980 | 517.2995 | 1.5/2.9 | 1 | 1.19 | 3 |
| 67 | 1342.6348 | 1342.6417 | 6.9/5.1 | 2, 3 | 11.26 | 105 |
| 68 | 1013.5990 | 1013.6084 | 9.4/9.3 | 1, 2 | 9.06 | 76 |
2.2. Covalent Modification of C34 by Parthenolide

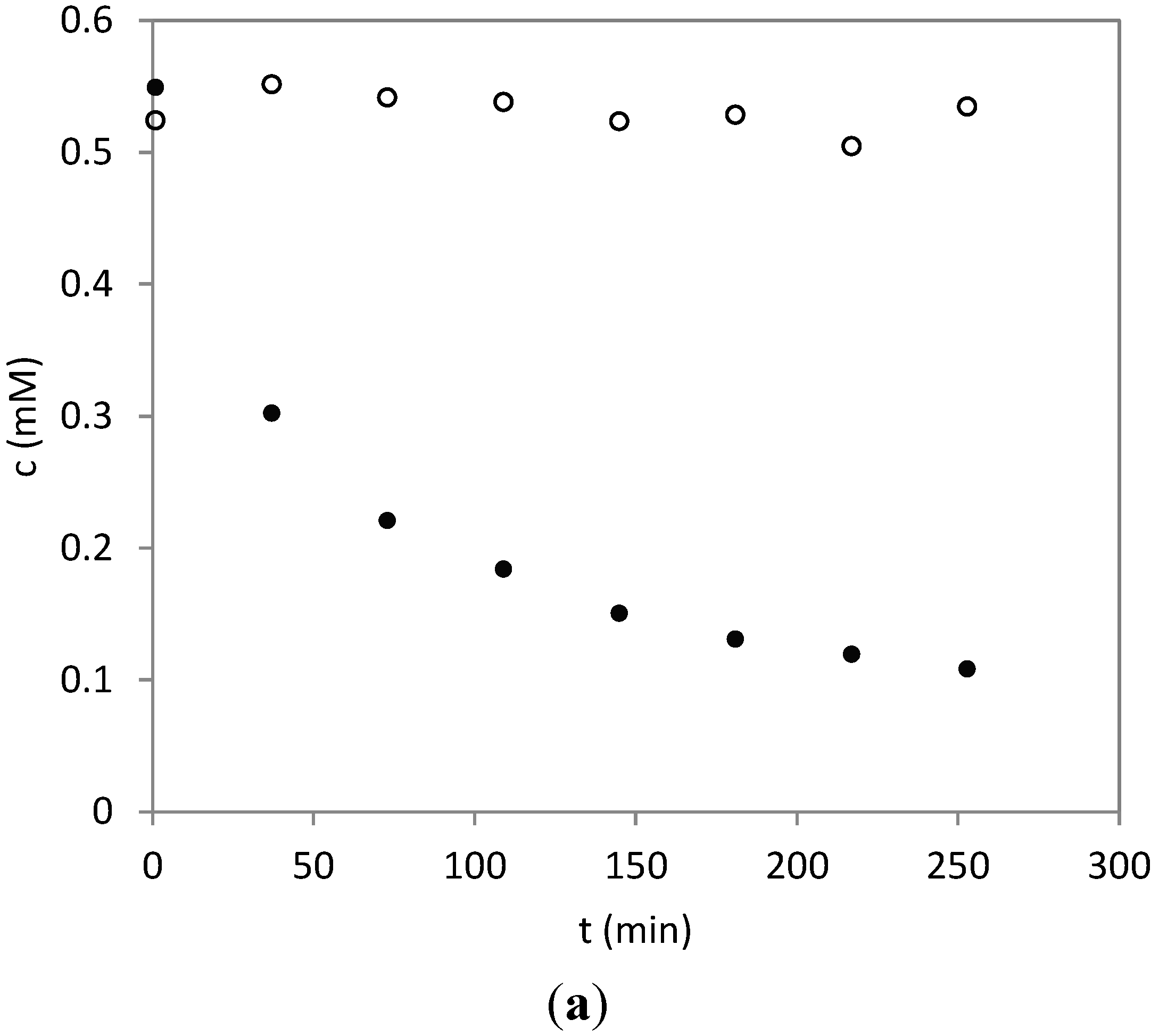
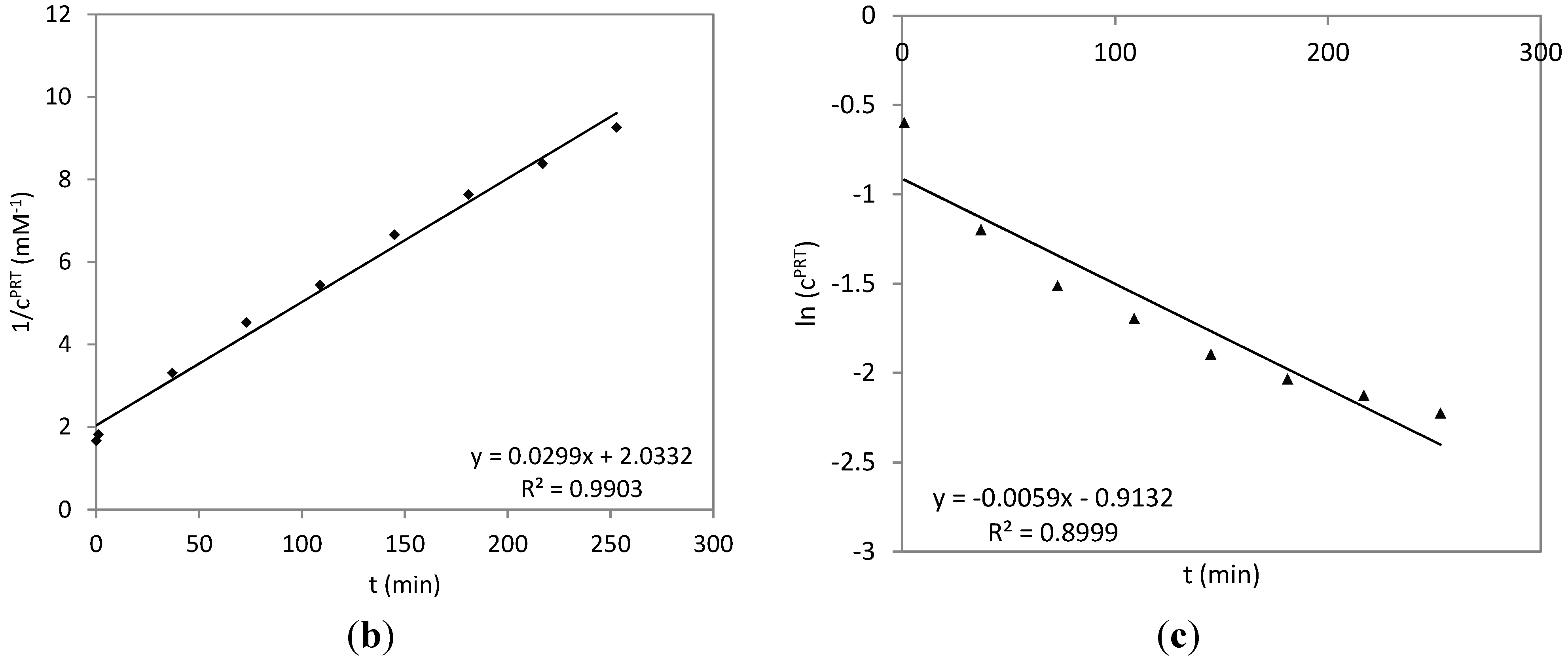
2.3. Time Course of the Reaction between Parthenolide and HSA
3. Experimental Section
3.1. Investigated Compounds
3.2. Sample Preparation
3.2.1. Preparation of Parthenolide-HSA Conjugate
3.2.2. Tryptic Digestion
3.3. High-Performance Liquid Chromatography-Electrospray Ionization Mass Spectrometry
3.4. Data Analysis
3.5. Time Course of the Reaction of HSA with PRT and DHP
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Schmidt, T.J. Structure-activity relationships of sesquiterpene lactones. In Studies in Natural Products Chemistry; Atta-ur-Rahman, Ed.; Elsevier: Amsterdam, The Netherlands, 2006; Volume 33, pp. 309–392. [Google Scholar]
- Lepoittevin, J.-P.; Berl, V.; Gimenez-Arnau, E. α-Methylene-γ-butyrolactones: Versatile skin bioactive natural products. Chem. Rec. 2009, 9, 258–270. [Google Scholar] [CrossRef] [PubMed]
- Jacob, M.; Brinkmann, J.; Schmidt, T.J. Sesquiterpene Lactone Mix as a diagnostic tool for Asteraceae allergic contact dermatitis: Chemical explanation for its poor performance and “SL-Mix 2” as a proposed improvement. Contact Dermat. 2012, 66, 233–240. [Google Scholar] [CrossRef]
- Schmidt, T.J. Helenanolide type sesquiterpene lactones. III. rates and stereochemistry in the reaction of helenalin and related helenanolides with sulfhydryl containing biomolecules. Bioorg. Med. Chem. 1997, 5, 645–653. [Google Scholar]
- Schmidt, T.J.; Lyß, G.; Pahl, H.L.; Merfort, I. Helenanolide type sesquiterpene lactones-V. The role of glutathione addition under physiological conditions. Bioorg. Med. Chem. 1999, 7, 2849–2855. [Google Scholar]
- Kupchan, S.M.; Fessler, D.C.; Eakin, M.A.; Giacobbe, T.J. Reactions of alpha methylene lactone tumor inhibitors with model biological nucelophiles. Science 1970, 168, 376–377. [Google Scholar] [PubMed]
- Kupchan, S.M.; Eakin, M.A.; Thomas, A.M. Tumor inhibitors. 69. Structure-cytotoxicity relationships among the sesquiterpene lactones. J. Med. Chem. 1971, 14, 1147–1152. [Google Scholar]
- Wagner, S.; Kratz, F.; Merfort, I. In vitro behaviour of sesquiterpene lactones and sesquiterpene lactone-containing plant preparations in human blood, plasma and human serum albumin solutions. Planta Med. 2004, 70, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Carter, D.C.; Ho, J.X. Structure of serum albumin. Adv. Protein Chem. 1994, 45, 153–203. [Google Scholar] [PubMed]
- Fogh-Andersen, N.; Altura, B.M.; Altura, B.T.; Sigaard-Andersen, O. Composition of interstitial fluid. Clin. Chem. 1995, 41, 1522–1525. [Google Scholar] [PubMed]
- Liu, Q.; Simpson, D.C.; Gronert, S. The reactivity of human serum albumin toward trans-4-hydroxy-2-nonenal. J. Mass. Spectrom. 2012, 47, 411–424. [Google Scholar] [CrossRef] [PubMed]
- Graeser, R.; Esser, N.; Unger, H.; Fichtner, I.; Zhu, A.; Unger, C.; Kratz, F. INNO-206, the (6-maleimidocaproyl hydrazone derivative of doxorubicin), shows superior antitumor efficacy compared to doxorubicin in different tumor xenograft models and in an orthotopic pancreas carcinoma model. Investig. New Drugs 2010, 28, 14–19. [Google Scholar] [CrossRef]
- Abu Ajaj, K.; Graeser, R.; Kratz, F. Zosuquidar and an albumin-binding prodrug of zosuquidar reverse multidrug resistance in breast cancer cells of doxorubicin and an albumin-binding prodrug of doxorubicin. Breast Cancer Res. Treat. 2012, 134, 117–129. [Google Scholar] [CrossRef] [PubMed]
- Elsadek, B.; Kratz, F. Impact of albumin on drug delivery—New applications on the horizon. J. Control. Release 2012, 157, 4–28. [Google Scholar] [CrossRef] [PubMed]
- Pleitez, M.; von Lilienfeld-Toal, H.; Mäntele, W. Infrared spectroscopic analysis of human interstitial fluid in vitro and in vivo using FT-IR spectroscopy and pulsed quantum cascade lasers (QCL): Establishing a new approach to non invasive glucose measurement. Spectrochim. Acta A 2012, 85, 61–65. [Google Scholar] [CrossRef]
- Pajak, B.; Orzechowski, A.; Gajkowska, B. Molecular basis of parthenolide-dependent proapoptotic activity in cancer cells. Folia Histochem. Cytobiol. 2008, 46, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Mathema, V.B.; Koh, Y.S.; Thakuri, B.C.; Sillanpää, M. Parthenolide, a sesquiterpene lactone, expresses multiple anti-cancer and anti-inflammatory activities. Inflammation 2012, 35, 560–565. [Google Scholar] [CrossRef] [PubMed]
- Pareek, A.; Suthar, M.; Rathore, G.S.; Bansal, V. Feverfew (Tanacetum parthenium L.): A systematic review. Pharmacogn. Rev. 2011, 5, 103–110. [Google Scholar] [CrossRef]
- Amorim, M.H.; Gil da Costa, R.M.; Lopes, C.; Bastos, M.M. Sesquiterpene lactones: Adverse health effects and toxicity mechanisms. Crit. Rev. Toxicol. 2013, 43, 559–579. [Google Scholar] [CrossRef] [PubMed]
- Pfaffenrath, V.; Diener, H.C.; Fischer, M.; Friede, M.; Henneicke-von Zepelin, H.H. The efficacy and safety of Tanacetum parthenium (feverfew) in migraine prophylaxis—A double-blind, multicentre, randomized placebo-controlled dose-response study. Cephalalgia 2002, 22, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Tassorelli, C.; Greco, R.; Morazzoni, P.; Riva, A.; Sandrini, G.; Nappi, G. Parthenolide is the component of Tanacetum parthenium that inhibits nitroglycerin-induced Fos activation: Studies in an animal model of migraine. Cephalalgia 2005, 25, 612–621. [Google Scholar] [CrossRef] [PubMed]
- Warshaw, E.M.; Zug, K.A. Sesquiterpene lactone allergy. Am. J. Contact Dermat. 1996, 7, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Le Coz, C.L.; Ducombs, G. Plants and plant products. In Contact Dermatitis, 4th ed.; Frosch, P.J., Menne, T., Lepottevin, J.-P., Eds.; Springer-Verlag: Berlin/Heidelberg, Germany, 2006; pp. 751–800. [Google Scholar]
- Mahajan, V.K.; Sharma, V.; Gupta, M.; Chauhan, P.S.; Mehta, K.S.; Garg, S. Parthenium dermatitis: Is parthenolide an effective choice for patch testing? Contact Dermat. 2014, 70, 340–343. [Google Scholar] [CrossRef]
- Kratz, F.; Warnecke, A.; Scheuermann, K.; Stockmar, C.; Schwab, J.; Lazar, P.; Drückes, P.; Esser, N.; Drevs, J.; Rognan, D.; et al. Probing the cysteine-34 position of endogenous serum albumin with thiol-binding doxorubicin derivatives. improved efficacy of an acid-sensitive doxorubicin derivative with specific albumin-binding properties compared to that of the parent compound. J. Med. Chem. 2002, 45, 5523–5533. [Google Scholar]
- Sogami, M.; Era, S.; Nagaoka, S.; Kuwata, K.; Kida, K.; Shigemi, J.; Miura, K.; Suzuki, E.; Muto, Y.; Tomita, E.; et al. High performance liquid chromatographic studies on non-mercapt—Mercapt conversion of human serum albumin. II. J. Chromatogr. A 1985, 332, 19–27. [Google Scholar]
- Etoh, T.; Miyazaki, M.; Harada, K.; Nakayama, M.; Sugii, A. Rapid analysis of human serum albumin by high-performance liquid chromatography. J. Chromatogr. B 1992, 578, 292–296. [Google Scholar] [CrossRef]
- Taguchi, K.; Chuang, V.T.G.; Maruyama, T.; Otagiri, M. Pharmaceutical aspects of the recombinant human serum albumin dimer: Structural characteristics, biological properties, and medical applications. J. Pharm. Sci. 2012, 101, 3033–3046. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, A.A.; Curry, S.; Franks, N.P. Binding of the general anesthetics propofol and halothane to human serum albumin. High resolution crystal structures. J. Biol. Chem. 2000, 275, 38731–38738. [Google Scholar]
- ProteinProspector. UCSF Mass Spectrometry Facility: San Francisco, CA, USA. Available online: http://prospector.ucsf.edu/prospector/mshome.htm (accessed on 8 April 2015).
- Sample Availability: Not applicable.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Plöger, M.; Sendker, J.; Langer, K.; Schmidt, T.J. Covalent Modification of Human Serum Albumin by the Natural Sesquiterpene Lactone Parthenolide. Molecules 2015, 20, 6211-6223. https://doi.org/10.3390/molecules20046211
Plöger M, Sendker J, Langer K, Schmidt TJ. Covalent Modification of Human Serum Albumin by the Natural Sesquiterpene Lactone Parthenolide. Molecules. 2015; 20(4):6211-6223. https://doi.org/10.3390/molecules20046211
Chicago/Turabian StylePlöger, Michael, Jandirk Sendker, Klaus Langer, and Thomas J. Schmidt. 2015. "Covalent Modification of Human Serum Albumin by the Natural Sesquiterpene Lactone Parthenolide" Molecules 20, no. 4: 6211-6223. https://doi.org/10.3390/molecules20046211
APA StylePlöger, M., Sendker, J., Langer, K., & Schmidt, T. J. (2015). Covalent Modification of Human Serum Albumin by the Natural Sesquiterpene Lactone Parthenolide. Molecules, 20(4), 6211-6223. https://doi.org/10.3390/molecules20046211







