Evaluation of the Hypoglycemic Effects of Flavonoids and Extracts from Jatropha gossypifolia L.
Abstract
:1. Introduction
2. Results and Discussion
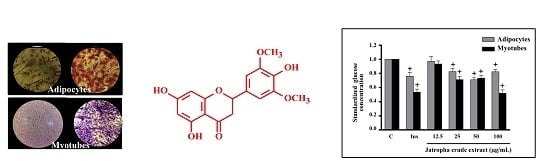
2.1. Glucose Uptake in Myotubes and Adipocytes Treated with Extracts of Jatropha gossypifolia L.
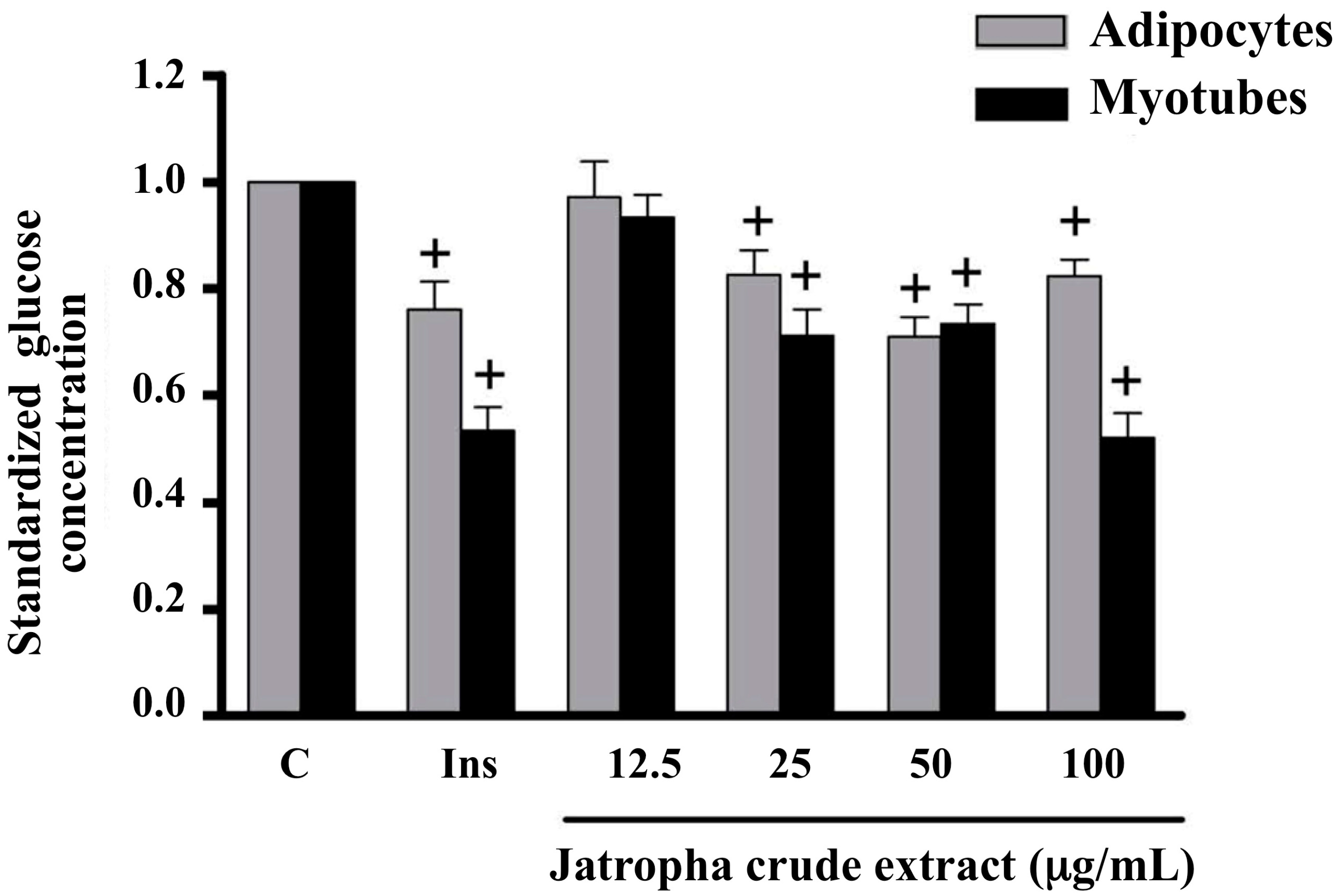
2.2. Glucose Uptake in Insulin-Resistant Myotubes Treated with Extracts of Jatropha gossypifolia L.
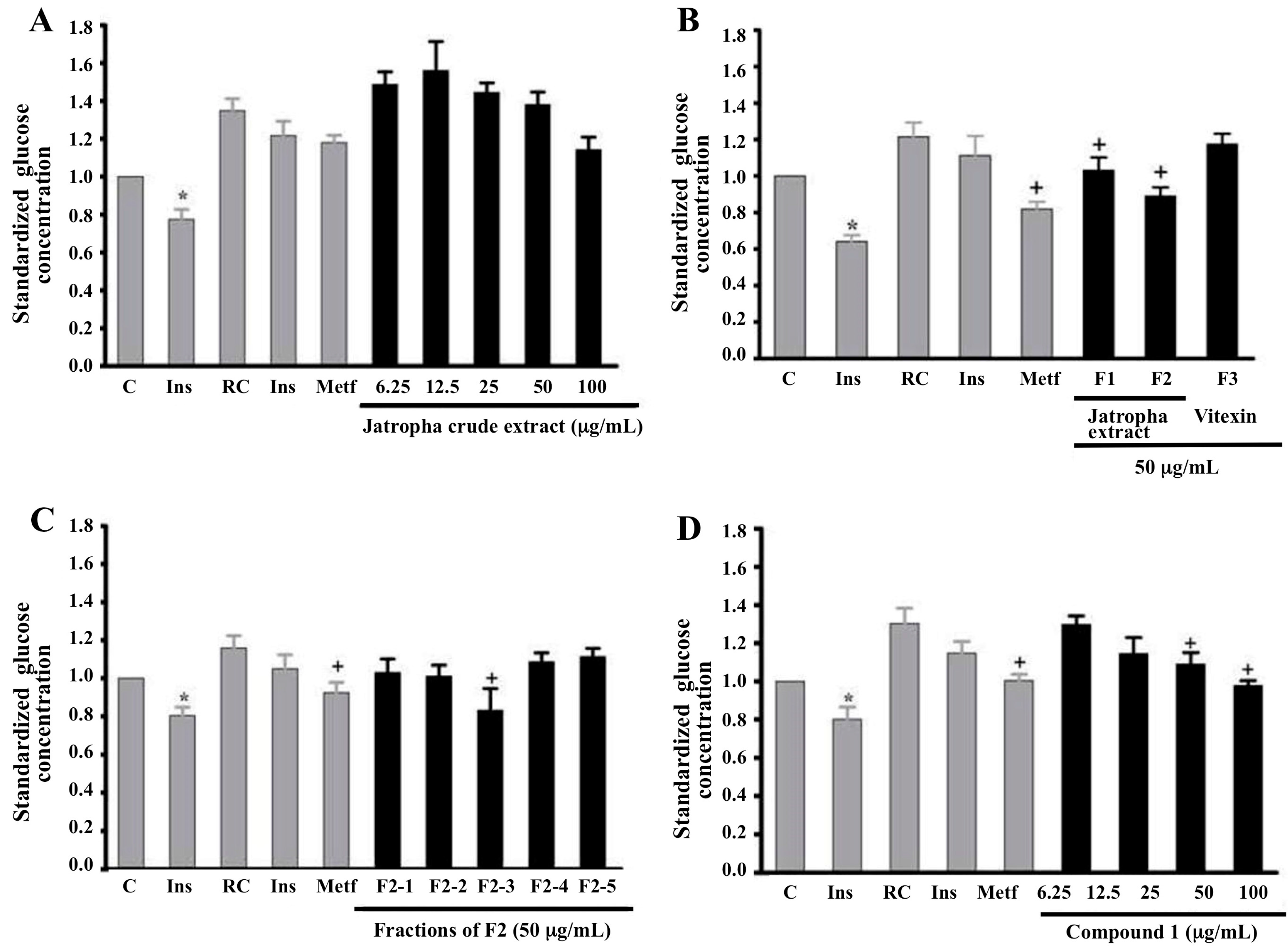
2.3. Effect of Chromatographic Fractions and Pure Compounds on Glucose Uptake in Insulin-Resistant Myotubes
| Plant Material | IC50 (µg/mL) C2C12 | SD | IC50 (µg/mL) HepG2 | SD |
|---|---|---|---|---|
| Crude Jatropha extract | 135 | 4.6 | 108 | 14 |
| Diethyl ether (F1) | 57 | 12.7 | 70 | 9.7 |
| Ethyl acetate (F2) | >200 | 6.9 | >200 | 5.6 |
| Vitexin (F3) | >200 | 4.3 | >200 | 8.3 |
2.4. Effect of Compound 1 on Glucose Uptake
2.5. Structure of Compound 1 with Hypoglycemic Effects
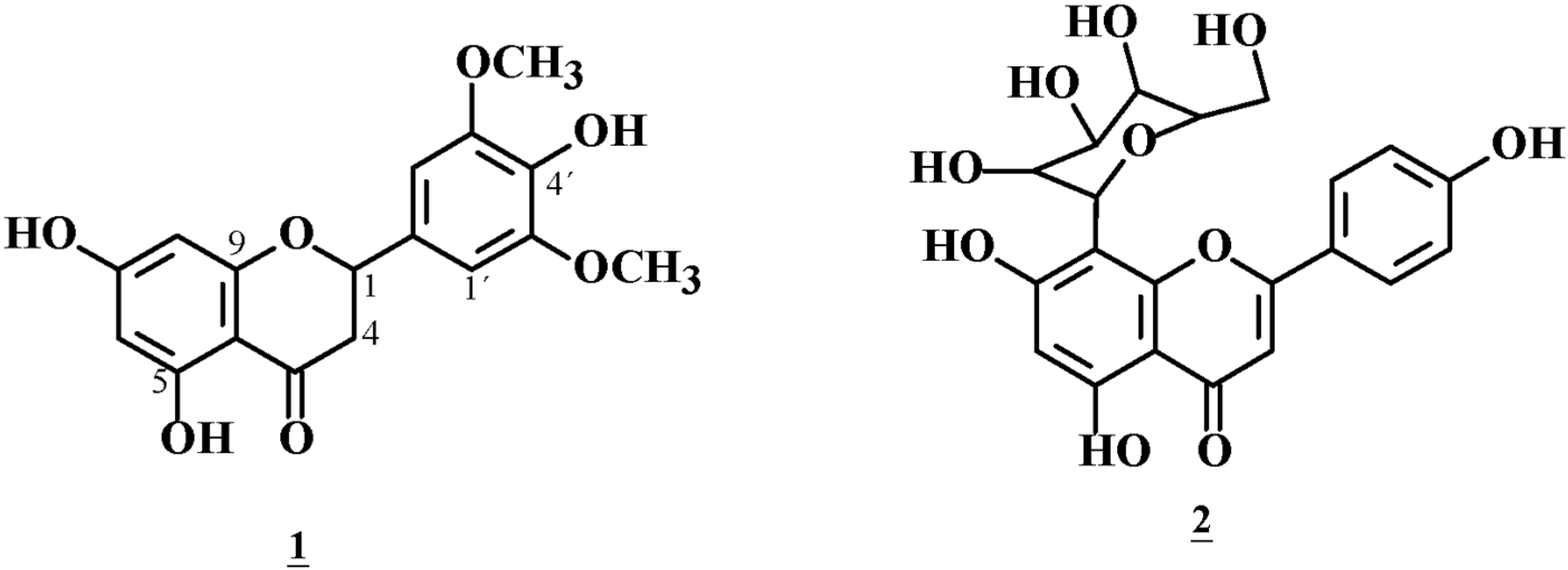
2.6. Structure of the Glucose Uptake Suppressor
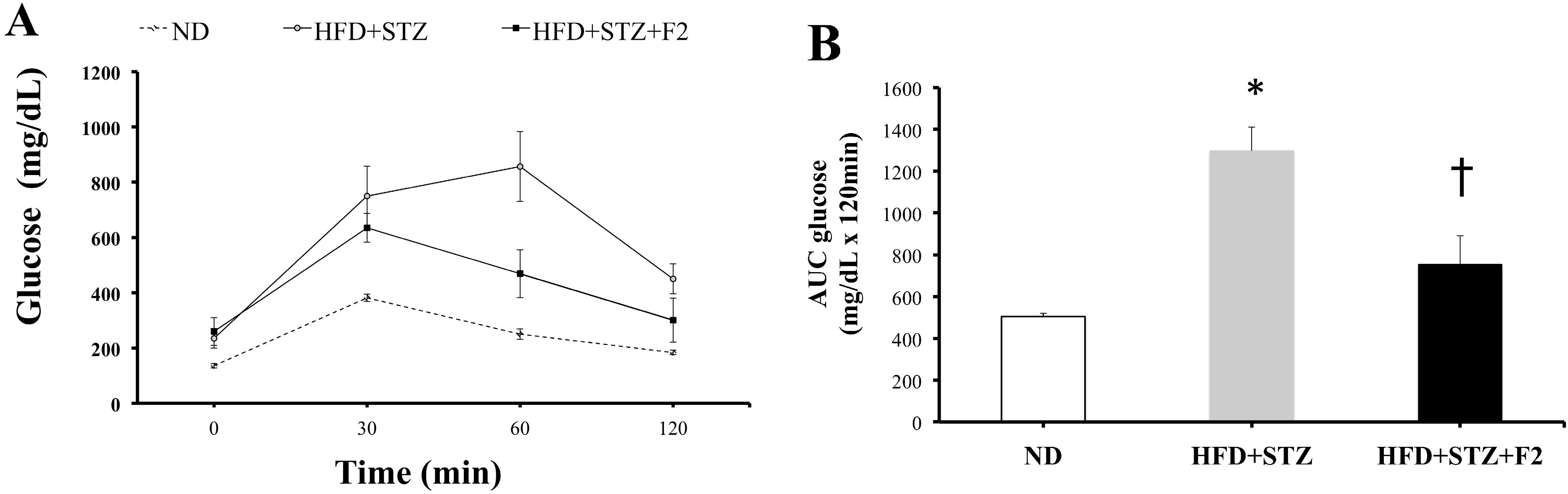
2.7. Intraperitoneal Glucose Tolerance Test (IPGTT)
3. Experimental Section
3.1. General Procedures
3.2. Preparation of the Extract and Chromatographic Fractionation
3.3. Cell Cultures
3.4. Glucose Tolerance Test in the Mouse Model of T2DM
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ross, S.A.; Gulve, E.A.; Wang, M. Chemistry and Biochemistry of Type 2 Diabetes. Chem. Rev. 2004, 104, 1255–1282. [Google Scholar] [CrossRef] [PubMed]
- Goh, S.Y.; Cooper, M.E. Clinical review: The role of advanced glycation end products in progression and complications of diabetes. J. Clin. Endocrinol. Metab. 2008, 93, 1143–1152. [Google Scholar] [CrossRef] [PubMed]
- Scheen, A.J.; Paquot, N. Metformin revisited: A critical review of the benefit–risk balance in at-risk patients with type 2 diabetes. Diabetes Metab. 2013, 39, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Hung, H.Y.; Qian, K.; Morris-Natschke, S.L.; Hsu, C.S.; Lee, K.H. Recent discovery of plant-derived anti-diabetic natural products. Nat. Prod. Rep. 2012, 29, 580–606. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, CA.; Simó, L.Y.; Ortiz, M.; Mañón, D. Estudio de seis plantas medicinales dominicanas. Acta Med. Dominic. 1999, 21, 86–93. [Google Scholar]
- Singh, H.; Sharma, S.K. Antidiabetic activity of Jatropha gossypifolia Linn root extracts in alloxan induced diabetic mice. Int. Res. J. Pharm. 2013, 4, 210–212. [Google Scholar] [CrossRef]
- Rahuja, N.; Mishra, A.; Maurya, R.; Srivastava, M.N.; Tamrakar, A.K.; Jain, S.K.; Srivastava, A.K. Antidiabetic and hypolipidemic activity in stem of Jatropha gossypifolia L. Int. Res. J. Pharm. 2013, 5, 706–715. [Google Scholar]
- Yang, W.Q.; Wang, H.C.; Wang, W.J.; Wang, Y.; Zhang, X.Q.; Ye, W.C. Chemical constituents from the fruits of Areca catechu. Zhong Yao Cai 2012, 35, 400–403. [Google Scholar] [PubMed]
- Na, M.; Jang, J.; Njamen, D.; Mbafor, J.T.; Fomum, Z.T.; Kim, B.Y.; Oh, W.K.; Ahn, J.S. Protein tyrosine phosphatase-1B inhibitory activity of isoprenylated flavonoids isolated from Erythrina mildbraedii. J. Nat. Prod. 2006, 69, 1572–1576. [Google Scholar] [CrossRef] [PubMed]
- Talhi, O.; Silva, A.M. Advances in C-glycosylflavonoid research. Curr. Org. Chem. 2012, 16, 859–896. [Google Scholar] [CrossRef]
- Kumar, N.; Dey, C.S. Metformin enhances insulin signalling in insulin-dependent and-independent pathways in insulin resistant muscle cells. Br. J. Pharmacol. 2002, 137, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Pérez, A.; Ojeda, P.; Ojeda, L.; Salas, M.; Rivas, C.I.; Vera, J.C.; Reyes, A.M. Hexose transporter GLUT1 harbors several distinct regulatory binding sites for flavones and tyrphostins. Biochemistry 2011, 50, 8834–8845. [Google Scholar] [CrossRef] [PubMed]
- Kodama, S.; Horikawa, C.; Fujihara, K.; Yoshizawa, S.; Yachi, Y.; Tanaka, S.; Ohara, N.; Matsunaga, S.; Yamada, T.; Hanyu, O.; et al. Quantitative relationship between body weight gain in adulthood and incident type 2 diabetes: A meta-analysis. Obes. Rev. 2014, 15, 202–214. [Google Scholar]
- Tortorella, L.L.; Pilch, P.F. C2C12 myocytes lack an insulin-responsive vesicular compartment despite dexamethasone-induced GLUT4 expression. Am. J. Physiol. Endocrinol. Metab. 2002, 283, E514–E524. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, H.; Tajiri, Y.; Sako, Y.; Hashimoto, T.; Umeda, F.; Nawata, H. Effects of free fatty acids on beta-cell functions: A possible involvement of peroxisome proliferator-activated receptors alpha or pancreatic/duodenal homeobox. Metabolism 2004, 50, 613–618. [Google Scholar] [CrossRef]
- Strobel, P.; Allard, C.; Perez-Acle, T.; Calderon, R.; Aldunate, R.; Leighton, F. Myricetin, quercetin and catechin-gallate inhibit glucose uptake in isolated rat adipocytes. Biochem. J. 2005, 386, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Harmon, A.W.; Patel, Y.M. Naringenin inhibits glucose uptake in MCF-7 breast cancer cells: A mechanism for impaired cellular proliferation. Breast Cancer Res. Treat. 2004, 85, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Spencer, J.P.; Abd-el-Mohsen, M.M.; Rice-Evans, C. Cellular uptake and metabolism of flavonoids and their metabolites: Implications for their bioactivity. Arch. Biochem. Biophys. 2004, 423, 148–161. [Google Scholar] [CrossRef] [PubMed]
- Leiter, E.H. Selecting the “right” mouse model for metabolic syndrome and type 2 diabetes research. Methods Mol. Biol. 2009, 560, 1–17. [Google Scholar] [PubMed]
- Sabandar, C.W.; Ahmat, N.; Jaafar, F.M.; Sahidin, I. Medicinal property, phytochemistry and pharmacology of several Jatropha species (Euphorbiaceae): A review. Phytochemistry 2013, 85, 7–29. [Google Scholar] [CrossRef] [PubMed]
- Lam, K.S. New aspects of natural products in drug discovery. Trends Microbiol. 2007, 15, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Schmitz-Peiffer, C.; Craig, D.L.; Biden, T.J. Ceramide generation is sufficient to account for the inhibition of the insulin-stimulated PKB pathway in C2C12 skeletal muscle cells pretreated with palmitate. J. Biol. Chem. 1999, 274, 24202–24210. [Google Scholar] [CrossRef] [PubMed]
- Zebisch, K.; Voigt, V.; Wabitsch, M.; Brandsch, M. Protocol for effective differentiation of 3T3-L1 cells to adipocytes. Anal. Biochem. 2012, 425, 88–90. [Google Scholar] [CrossRef] [PubMed]
- Chavez, J.A.; Summers, S.A. Characterizing the effects of saturated fatty acids on insulin signaling and ceramide and diacylglycerol accumulation in 3T3-L1 adipocytes and C2C12 myotubes. Arch. Biochem. Biophys. 2003, 419, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Hansen, M.B.; Nielsen, S.E.; Berg, K. Re-examination and further development of a precise and rapid dye method for measuring cell growth/cell kill. J. Immunol. Methods 1989, 119, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds 1 and 2 are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Granados, S.; Balcázar, N.; Guillén, A.; Echeverri, F. Evaluation of the Hypoglycemic Effects of Flavonoids and Extracts from Jatropha gossypifolia L. Molecules 2015, 20, 6181-6193. https://doi.org/10.3390/molecules20046181
Granados S, Balcázar N, Guillén A, Echeverri F. Evaluation of the Hypoglycemic Effects of Flavonoids and Extracts from Jatropha gossypifolia L. Molecules. 2015; 20(4):6181-6193. https://doi.org/10.3390/molecules20046181
Chicago/Turabian StyleGranados, Sergio, Norman Balcázar, Alis Guillén, and Fernando Echeverri. 2015. "Evaluation of the Hypoglycemic Effects of Flavonoids and Extracts from Jatropha gossypifolia L." Molecules 20, no. 4: 6181-6193. https://doi.org/10.3390/molecules20046181
APA StyleGranados, S., Balcázar, N., Guillén, A., & Echeverri, F. (2015). Evaluation of the Hypoglycemic Effects of Flavonoids and Extracts from Jatropha gossypifolia L. Molecules, 20(4), 6181-6193. https://doi.org/10.3390/molecules20046181