A Review of Heterogeneous Photocatalysis for Water and Surface Disinfection
Abstract
:1. Heterogeneous Photocatalysis
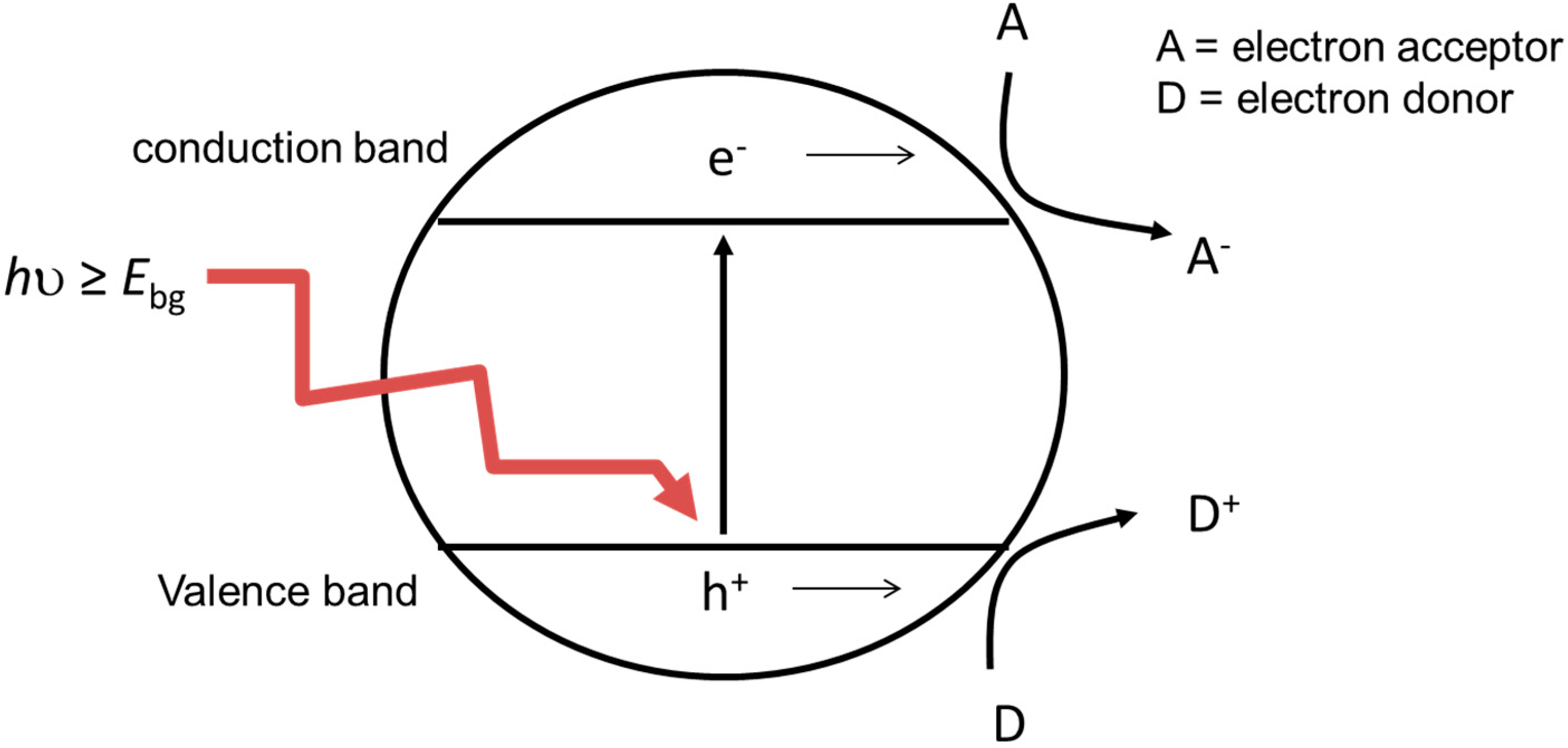
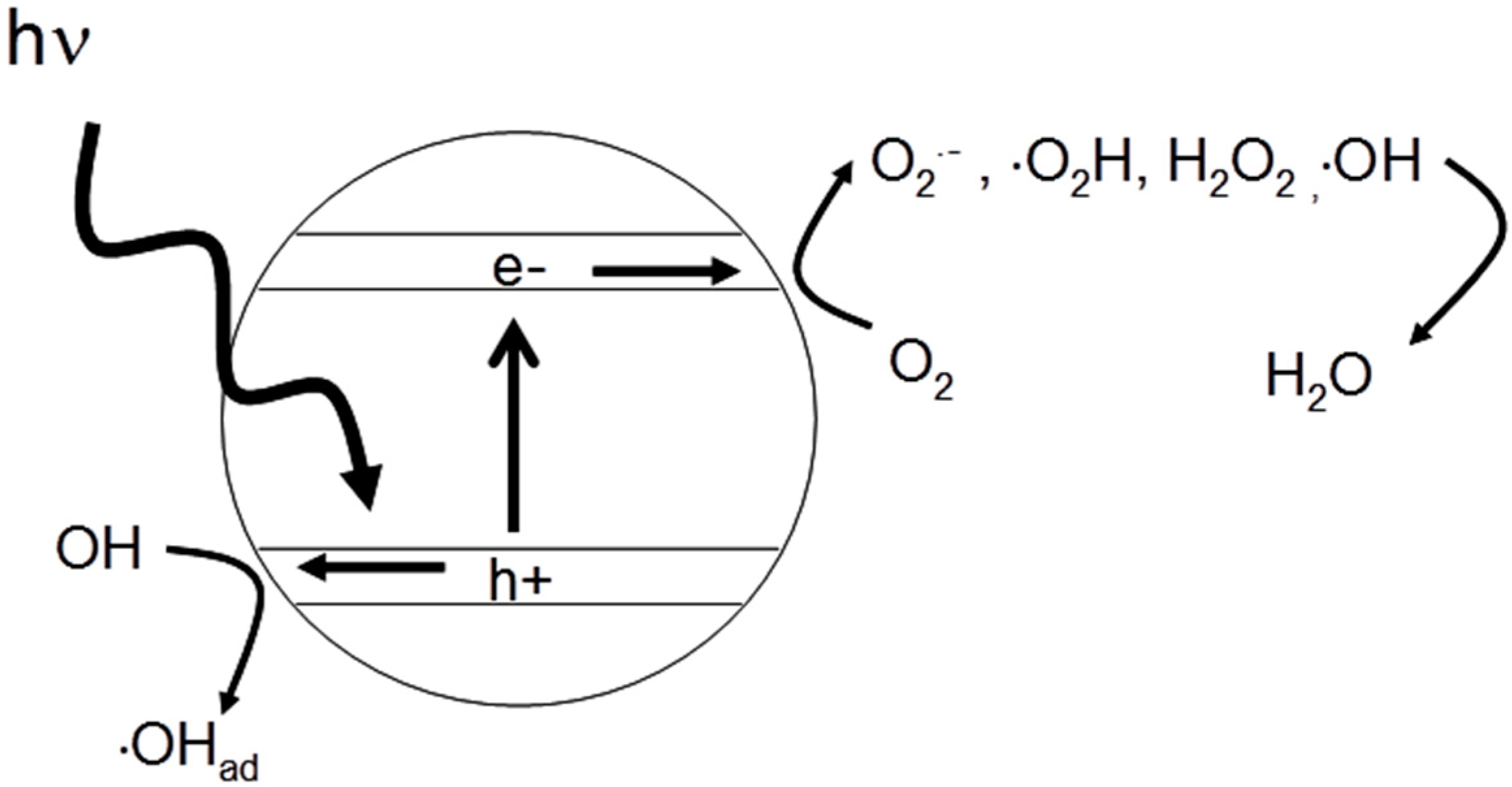
2. Mechanism of Photocatalytic Inactivation of Microorganisms
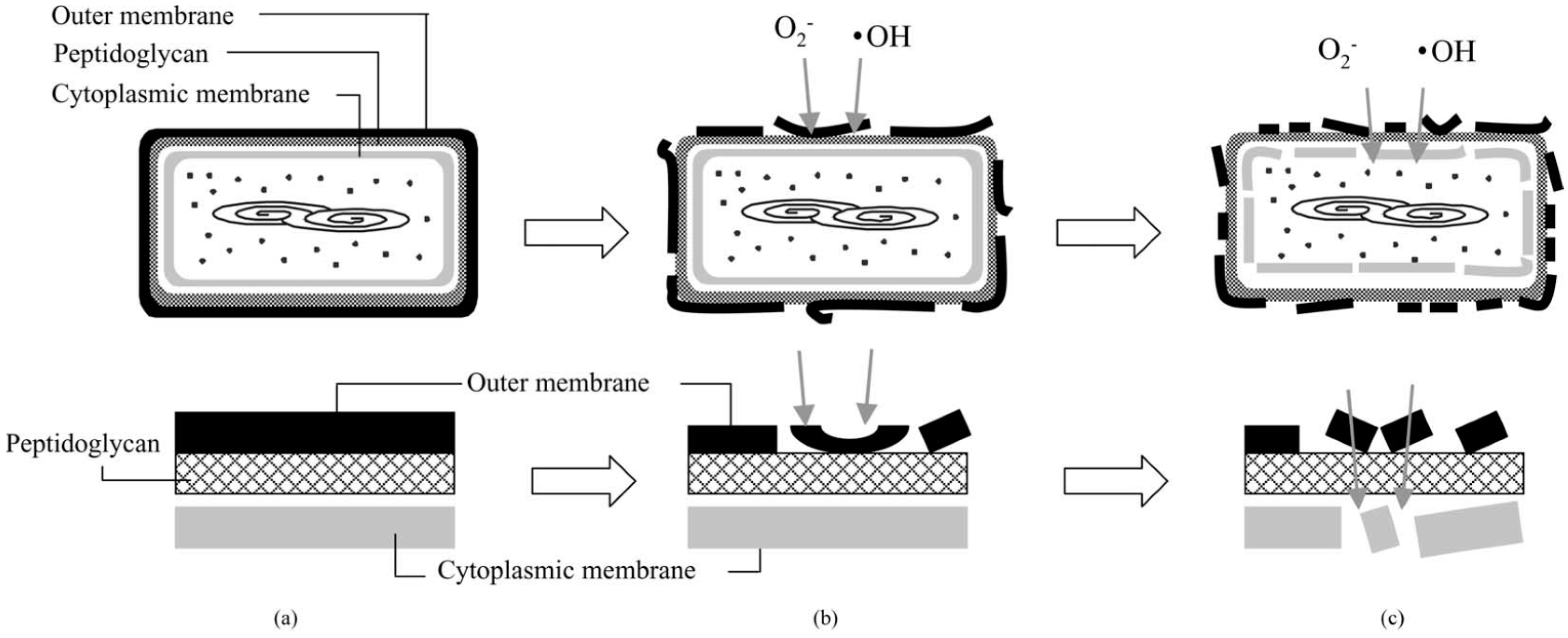
3. Solar Photocatalytic Disinfection of Water
3.1. Photocatalytic Materials Tested under Solar or Solar Simulated Conditions
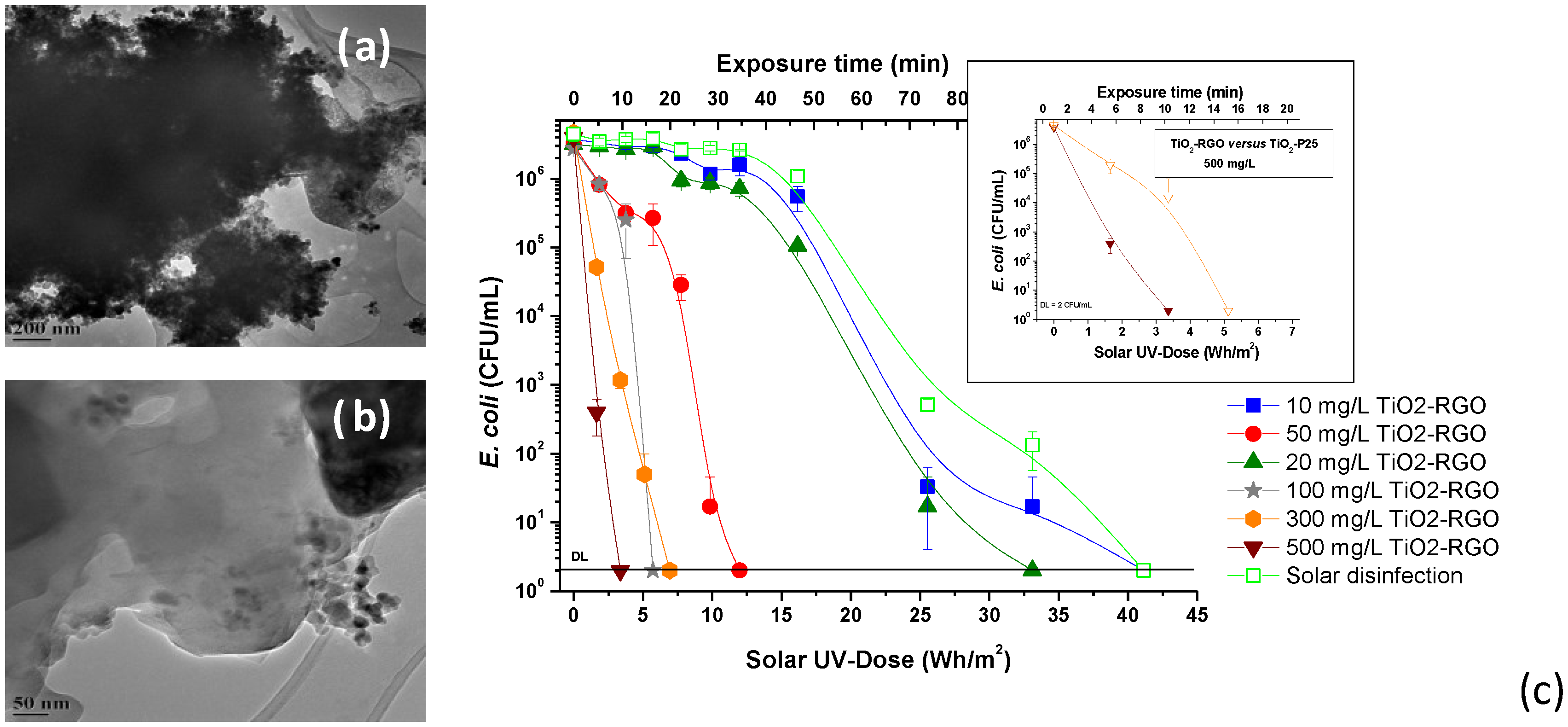
3.2. Solar Reactor Design and Applications
- (i)
- Increasing the appropriate solar photons entering the photo-reactor system.
- (ii)
- Improving the efficacy of the treatment against certain resistant water pathogens.
- (iii)
- Increasing the total volume of treated water for a certain treatment time (solar exposure).
- (iv)
- Reducing complexity of the water treatment system and decreasing the user dependency of the process.
- (v)
- Using low-cost systems based on simple designs and local and cheap materials to construct the water disinfection systems for application in developing countries.
- (vi)
- Design optimized photo-reactor systems that avoid post-treatment recovery or re-growth of microorganisms and prevent recontamination of treated water due to inappropriate handling or storage.

- (i)
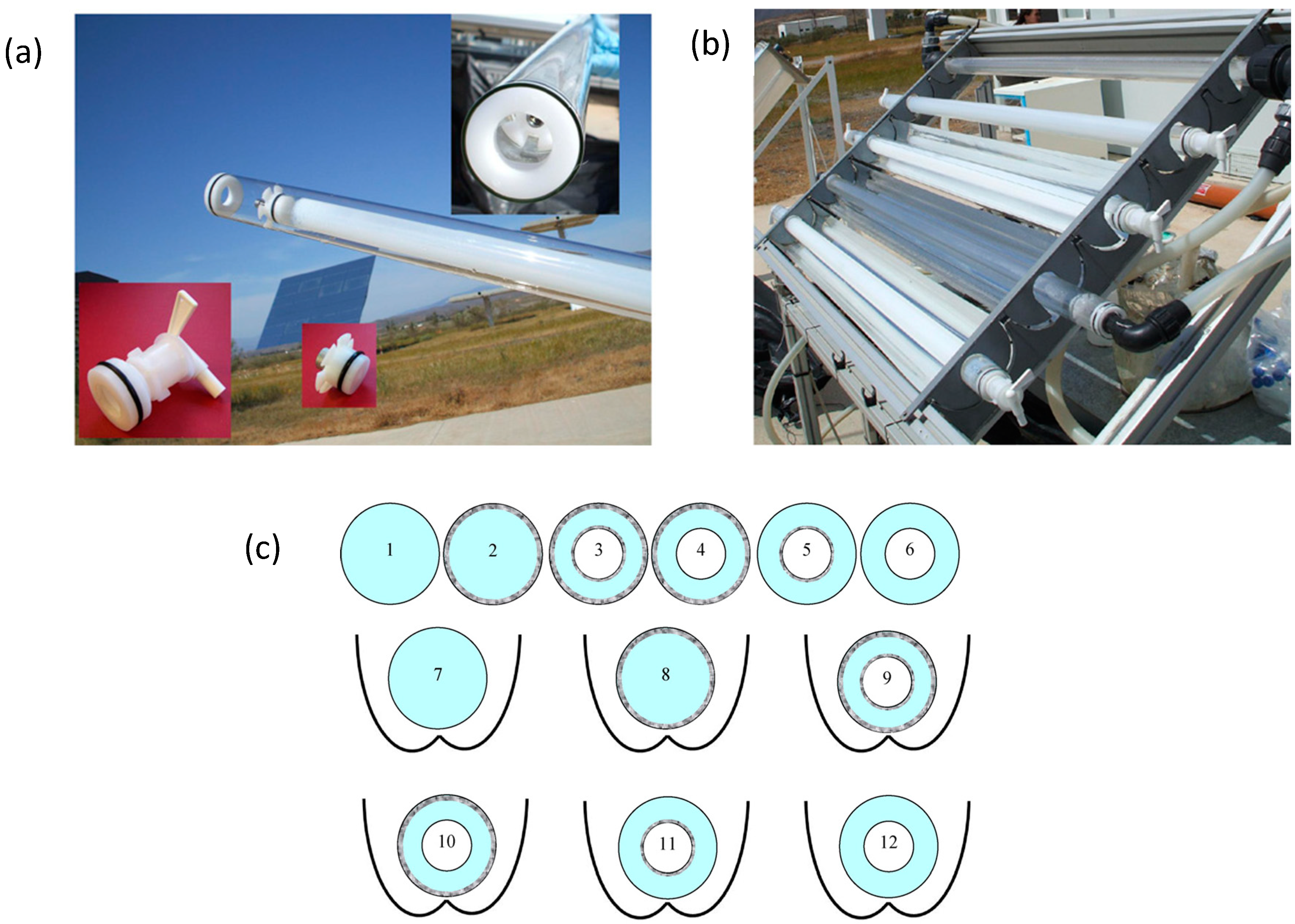
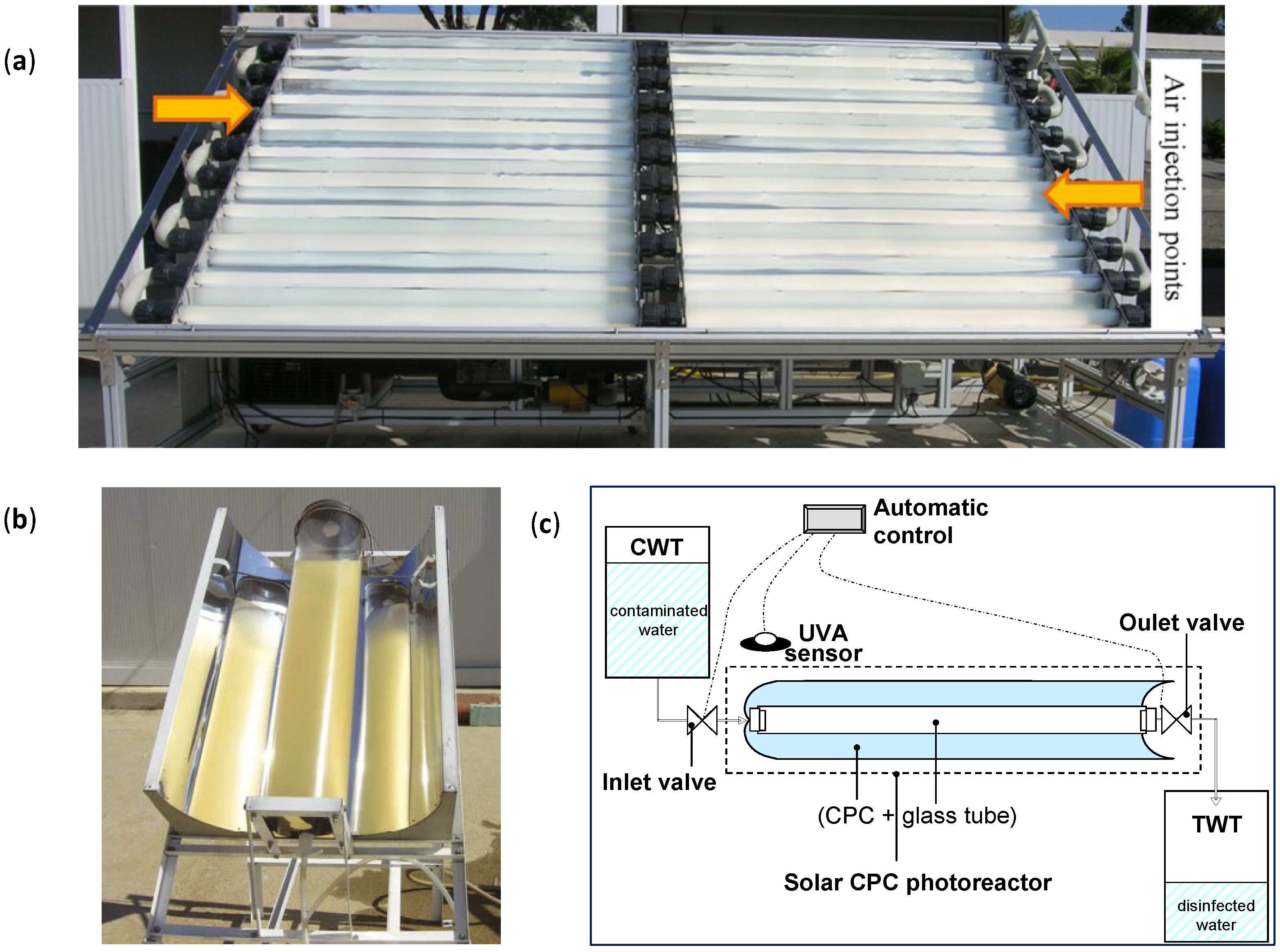
- The incoming photon-flux inside solar photo-reactor must be as high as possible (using either CPC solar mirrors or other low-cost reflectors, which increase the solar light collection). It should be efficiently and homogeneously distributed in the photo-reactor water, even for large volumes of water. Therefore light scattering and absorption of water samples is a key factor, thus there must be a compromise between the optical properties of the water and the physical path length of the photo-reactor (diameter if it is a tube, or depth if it is a pond or raceway). If the UV-Vis transmission of the water is good enough (turbidity < 10 NTU), the optical reactor path length (i.e., diameter of the tubular photo-reactor) can be increased until 10–20 cm (Figure 7b) [62]. The use of solar mirrors accelerates the disinfection rate, the increase in solar irradiance decreases the treatment time and accelerates the disinfection rate, but not necessarily in a linear proportionality manner [63].
- (ii)
- When the water does not have good UV-Vis transmission (i.e., when suspended TiO2 is used or turbid waters are treated), then the reactor tube must be reduced to few centimeters and the large volume requirement may be accomplished by connection of several photo-reactor modules [64].
- (iii)
- The use of immobilized photocatalyst is desirable, mainly for drinking water applications. In this case, the water flow rate must be low enough to increase the residence time required to achieve the desired disinfection in a single pass (the contact time will depend on the nature of the pathogen and the immobilized photocatalyst used) so that the recovery mechanisms of some microorganisms cannot effectively work after the treatment within the residence time. This important limitation has been widely investigated [53,65,66] with the conclusion that the flow rate must be low enough (only a few L/min) to ensure that a minimum lethal dose of energy is delivered before the water circulates to dark regions of the reactor. In the dark regions of the reactor, bacteria may recover and re-grow when the damage produced by the disinfecting agent (either by direct action of solar photons or by hydroxyl radicals) is not strong enough to achieve total inactivation (i.e., complete disinfection) and desired disinfection efficiencies may not be achieved [63,65,66]. Therefore, the design of the reactor must be done with prior knowledge of the lethal solar energy dose required so that complete disinfection is achieved in one run. Obviously, this energy requirement for complete killing depends strongly on several parameters like water composition, turbidity and nature of the biological contamination. A number of examples of the influence of the presence of ions, organic matter and natural occurring bacteria present in wastewater are available in the literature [65,67,68].
- (iv)
- There are limitations of solar disinfection (without any photocatalyst) when it is scaled-up through the use of large batch volumes or continuous flow recirculation reactors [63]. Increasing the flow rate has a negative effect on inactivation of bacteria, as at a given time point there needs to be maximum exposure of bacteria to UV to ensure inactivation as compared to having bacteria exposed to sub-lethal doses over a long period of time. When the water is kept static under solar light it is constantly illuminated and hence the required uninterrupted UV dose can be easily achieved. With continuous flow systems, the lethal dose can be delivered but in an intermittent manner and complete inactivation is not observed. This statement has important implications for those attempting to scale-up solar systems through the use of pumped, re-circulatory, continuous flow reactors. If the operational parameters are set such that the microbial pathogens are repeatedly exposed to sub-lethal doses of solar radiation followed by a period within which the cells have an opportunity to recover or repair, complete inactivation may not be achieved.
- (v)
- In photocatalytic disinfection, the electron acceptor is normally dissolved oxygen, which is easily available from the air. In the case of static batch systems, the concentration of dissolved oxygen will be rapidly depleted and must be replenished to maintain photocatalytic activity. Furthermore, the solubility of oxygen in water is reduced by temperature. This must be taken into consideration as the temperature within solar irradiated reactors can reach 55 °C. New designs of reactor must address the need for replenishment of dissolved oxygen in photocatalytic disinfection systems. This was investigated in the recent contribution of Garcia-Fernandez et al. [67]. These authors investigated the influence of temperature and dissolved oxygen using a solar 60 L-CPC reactor with suspended TiO2 (0.1 g/L) (Figure 7a). They injected air in the reactor pipeline (160 L/h) and evaluated several controlled (fixed) temperatures (15, 25, 35 and 45 °C) for the photocatalytic inactivation of two models of water pathogen, E. coli (vegetative cell model of fecal contamination) and Fusarium solani (a spore model of resistant fungi). In this work, they also assessed the effect of the chemical composition of the water and compared real urban wastewater with synthetic ones. They observed that increasing the water temperature, from 15 to 45 °C, had clear benefits on the disinfection rate for both pathogens. They also observed that air injection led to an important enhancement on the inactivation efficiency, which was even stronger for F. solani spores, the most resistant pathogen evaluated. The composition of the water matrix significantly affected negatively the efficiency of the photocatalytic treatment, showing a better inactivation rate in simulated urban wastewater effluent than for real urban wastewater effluent.
- (vi)
- An alternative to enhance photocatalytic performance may be to introduce other oxidants e.g., H2O2, persulfate radical, etc. however, this would give rise to a dependence on consumable chemicals which may be affordable or undesirable depending on the final application of the system.
- (vii)
- Other aspects like reducing the user dependence of the process or making the system as cheap and robust as possible are worthy to consider so that reactors can be used worldwide for solving a number of issues related with water safety, mainly for human consumption purposes in developing countries. The automated SODIS reactor developed by Polo-Lopez et al. [66] addressed some practical problems associated with increasing the treated water output using a continuous flow concept. This novel sequential batch photo-reactor was designed and constructed with the aim of decreasing the treatment time, increasing the total volume of water treated per day and reducing user-dependency. The photoreactor incorporated a CPC concentration factor of 1.89 and the treatment time was automatically controlled by an electronic UVA sensor (Figure 7c). The feedback sensor system controlled the gravity-filling of the reactor from an untreated water reservoir, and controlled the discharge of the treated water into a clean reservoir tank following receipt of the pre-defined UVA dose. The reactor was tested using E. coli in well water under real sun conditions in Southern Spain. They found that this system would permit processing of six sequential batches of 2.5 L each day (i.e., 15 L of solar purified water each per day). The system is modular, therefore it may be scaled up to allow several CPC photoreactors to be used under the control of a single UVA sensor. For example, six systems like this could produce around 90 L of potable water per day (for several households), and it could produce approximately 31,500 L during a typical year. Of course static systems do not present a good option when using immobilized photocatalyst, as mass transfer limitations will predominate unless a mechanism for forced convection and mixing is introduced.
4. Photocatalytic Coatings for Healthcare Applications
4.1. Self-Disinfecting Coatings for Environmental Surfaces
4.2. Photocatalytic Coatings for the Disinfection and Decontamination of Medical Devices
5. Novel Photocatalytic Materials
| Proposed Application | Photocatalyst | Microorganisms Tested | Reference |
|---|---|---|---|
| Environmental surfaces and medical devices | Evonik Aeroxide P25 immobilized on glass slides | extended-spectrum beta-lactamase(ESBL) Escherichia coli, methicillin resistant Staphylococcus aureus (MRSA), Pseudomonas aeruginosa and Clostridium difficile spores | [11] |
| General environmental cleaning | TiO2 (P25) on glass | E. coli, P. aeruginosa, S. aureus, E. faecium, C.albicans | [87] |
| Coating for hard surfaces in hospital environment | TiO2 and Ag-TiO2 on glass slides via sol-gel dip coating method | E. coli, S. aureus, B. cereus | [88] |
| Self-disinfecting catheters | TiO2 dip coated onto silicone catheters | E. coli | [79] |
| Lancet | TiO2 layer created by sputter depositions | E. coli | [84] |
| Percutaneous implant | Direct oxidation of pure Titanium plates | MRSA | [86] |
| Metal pin for external/percutaneous fixation | TiO2 coated on stainless steel via sol-gel dip coating | E. coli | [93] |
| Metal implant | Stainless steel and titanium coated with TiO2 via PSII | S. aureus | [94] |
| Intraocular lenses | PMMA plasma pre-treated and dip-coated with TiO2 | E. coli S. aureus | [97] |
| Dental implant | Titanium disc coated via PSII | A. actinomyce-temcomitans F. nucleatum | [98] |
| Dental adhesives | TiO2 nanoparticles mixed with commercially available dental adhesives | S. epidermidis | [85] |
| Antibacterial surface | TiO2 films sputter deposited onto silicon wafers | Diplococcus pneumoniae, S. aureus, E. coli | [89] |
| VLA coatings for environmental surfaces | N-doped TiO2 created by ion-assisted electron beam evaporation | S. flexneri, L. monocytogenes, V. para-haemolyticus, S. aureus, S. pyogenes, A. baumannii, E. coli | [90] |
| Self-disinfecting hospital surfaces | C-TiO2 and TiO2 modified with platinum (IV) chloride | E. coli, S. aureus, E. faecalis, C. albicans, A. niger | [91] |
| Antimicrobial healthcare surfaces | Sulfur and nitrogen doped titanium dioxide composites create via APCVD | E. coli | [92] |
| Antimicrobial healthcare surfaces | Ag loaded TiO2 films created via sol-gel | E. coli, EMRSA | [93] |
5.1. Narrow Band Gap Materials for Visible Light Activity
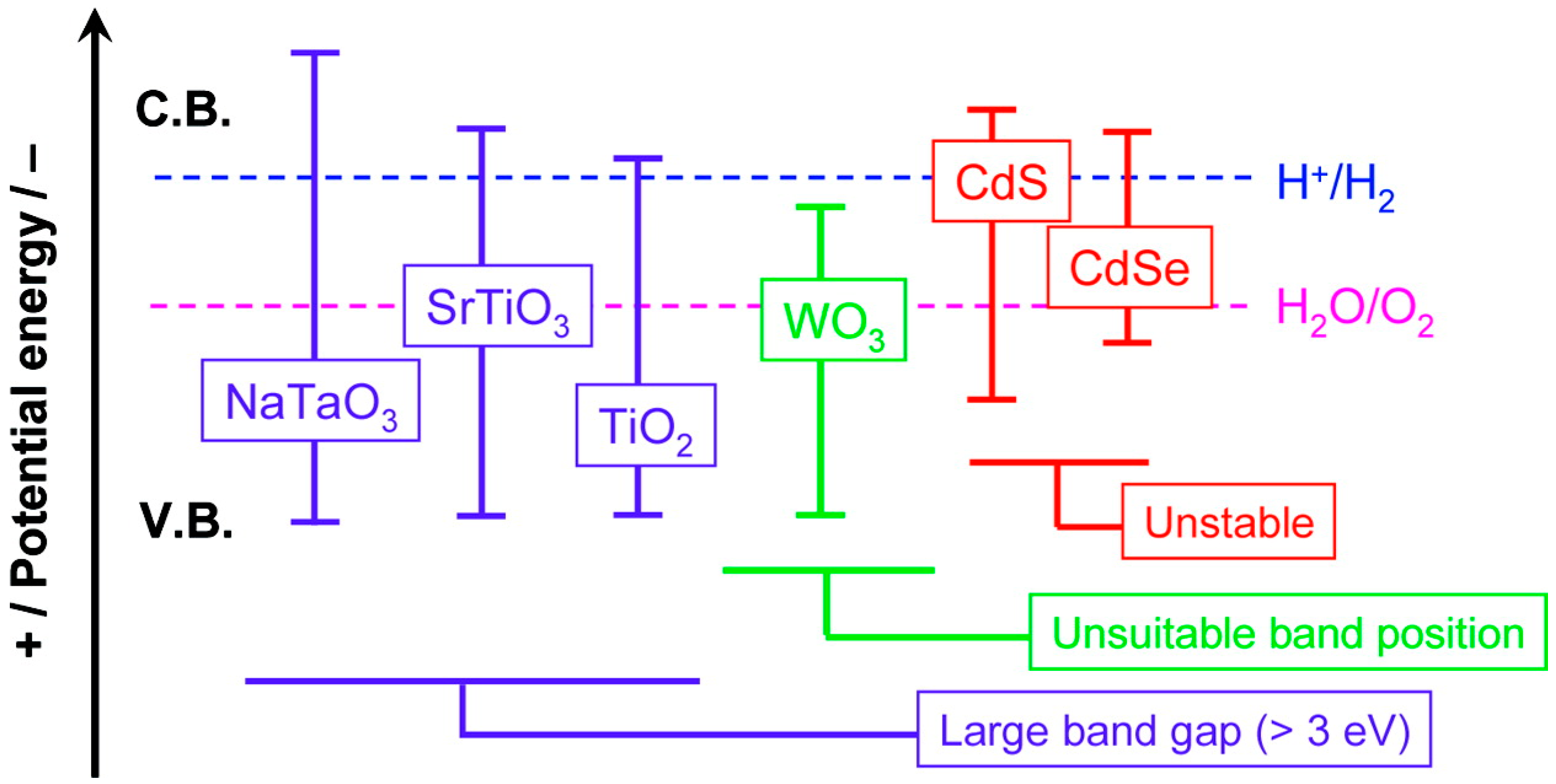
5.2. Modification and Sensitization of Existing Materials
5.3. Modification by Doping (Including Surface Loading)
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- IUPAC. Compendium of Chemical Terminology. In The Gold Book, 2nd ed.; McNaught, A.D., Wilkinson, A., Eds.; Blackwell Scientific Publications: Oxford, UK, 1997. [Google Scholar]
- Mills, A.; Le Hunte, S. An overview of semiconductor photocatalysis. J. Photochem. Photobiol. A 1997, 108, 1–35. [Google Scholar] [CrossRef]
- Tryk, D.A.; Fujishima, A.; Honda, K. Recent topics in photoelectrochemistry: Achievements and future prospects. Electrochim. Acta 2000, 45, 2363–2376. [Google Scholar] [CrossRef]
- Bahnemann, D. Photocatalytic water treatment: Solar energy applications. Sol. Energy 2004, 77, 445–459. [Google Scholar] [CrossRef]
- Agrios, A.G.; Pichat, P. State of the art and perspectives on materials and applications of photocatalysis over TiO2. J. Appl. Electrochem. 2005, 35, 655–663. [Google Scholar] [CrossRef]
- Augugliaro, V.; Litter, M.; Palmisano, L.; Soria, J. The combination of heterogeneous photocatalysis with chemical and physical operations: A tool for improving the photoprocess performance. J. Photochem. Photobiol. C 2006, 7, 127–144. [Google Scholar] [CrossRef]
- Fujishima, A.; Zhang, X.; Tryk, D.A. TiO2 photocatalysis and related surface phenomena. Surf. Sci. Rep. 2008, 63, 515–582. [Google Scholar] [CrossRef]
- Malato, S.; Fernandez-Ibanez, P.; Maldonado, M.I.; Blanco, J.; Gernjak, W. Decontamination and disinfection of water by solar photocatalysis: Recent overview and trends. Catal. Today 2009, 147, 1–59. [Google Scholar] [CrossRef]
- Matsunaga, T.; Tomoda, R.; Nakajima, T.; Wake, H. Photoelectrochemical sterilization of microbial cells by semiconductor powders. FEMS Microbiol. Lett. 1985, 29, 211–214. [Google Scholar] [CrossRef]
- Dunlop, P.S.M.; Byrne, J.A.; Manga, N.; Eggins, B.R. The photocatalytic removal of bacterial pollutants from drinking water. J. Photochem. Photobiol. A 2002, 148, 355–363. [Google Scholar] [CrossRef]
- Dunlop, P.S.M.; Sheeran, C.P.; Byrne, J.A.; McMahon, M.A.S.; Boyle, M.A.; McGuigan, K.G. Inactivation of clinically relevant pathogens by photocatalytic coatings. Photochem. Photobiol. A 2010, 216, 303–310. [Google Scholar] [CrossRef]
- Dunlop, P.S.M.; McMurray, T.A.; Hamilton, J.W.J.; Byrne, J.A. Photocatalytic inactivation of Clostridium perfringens spores on TiO2 electrodes. J. Photochem. Photobiol. A 2008, 196, 113–119. [Google Scholar] [CrossRef]
- Watts, R.J.; Kong, S.; Orr, M.P.; Miller, G.C.; Henry, B.E. Photocatalytic inactivation of coliform bacteria and viruses in secondary wastewater effluent. Water Res. 1995, 29, 95–100. [Google Scholar] [CrossRef]
- Sunnotel, O.; Verdoold, R.; Dunlop, P.S.M.; Snelling, W.J.; Lowery, C.J.; Dooley, J.S.G.; Moore, J.E.; Byrne, J.A. Photocatalytic inactivation of Cryptosporidium parvum on nanostructured titanium dioxide films. J. Water Health 2010, 8, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Sichel, C.; de Cara, M.; Tello, J.; Blanco, J.; Fernández-Ibánez, P. Solar photocatalytic disinfection of agricultural pathogenic fungi: Fusarium species. Appl. Catal. B-Environ. 2007, 74, 152–160. [Google Scholar] [CrossRef]
- Linkous, C.A.; Carter, G.J.; Locuson, D.B.; Ouellette, A.J.; Slattery, D.K.; Smith, L.A. Photocatalytic Inhibition of Algae Growth Using TiO2, WO3, and Cocatalyst Modifications. Environ. Sci. Technol. 2000, 34, 4754–4758. [Google Scholar] [CrossRef]
- McCullagh, C.; Robertson, J.M.C.; Bahnemann, D.W.; Robertson, P.J.K. The application of TiO2 photocatalysis for disinfection of water contaminated with pathogenic micro-organisms: A review. Res. Chem. Intermed. 2007, 33, 359–375. [Google Scholar] [CrossRef]
- Robertson, P.K.J.; Robertson, J.M.C.; Bahnemann, D.W. Removal of microorganisms and their chemical metabolites from water using semiconductor photocatalysis. J. Hazard. Mater. 2012, 211, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Dalrymple, O.K.; Stefanakos, E.; Trotz, M.A.; Goswami, D.Y. A review of the mechanisms and modeling of photocatalytic disinfection. Appl. Catal. B-Environ. 2010, 98, 27–38. [Google Scholar] [CrossRef]
- Fernandez-Ibanez, P.; Polo-Lopez, M.I.; Malato, S.; Wadhwa, S.; Hamilton, J.W.J.; Dunlop, P.S.M.; D’Sa, R.; Magee, E.; O’Shea, K.; Dionysiou, D.D.; et al. Solar photocatalytic disinfection of water using titanium dioxide graphene composites. Chem. Eng. J. 2015, 261, 36–44. [Google Scholar] [CrossRef]
- Cho, M.; Chung, H.; Choi, W.; Yoon, J. Linear correlation between inactivation of E. coli and OH radical concentration in TiO2 photocatalytic disinfection. Water Res. 2004, 38, 1069–1077. [Google Scholar] [CrossRef] [PubMed]
- Goulhen-Chollet, F.; Josset, S.; Keller, N.; Keller, V.; Lett, M.C. Monitoring the bactericidal effect of UV-A photocatalysis: A first approach through 1D and 2D protein electrophoresis. Catal. Today 2009, 147, 169–172. [Google Scholar] [CrossRef]
- Wu, P.; Xie, R.; Imlay, J.A.; Shang, J.K. Visible-light-induced photocatalytic inactivation of bacteria by composite photocatalysts of palladium oxide and nitrogen-doped titanium oxide. Appl. Catal. B-Environ. 2009, 88, 576–581. [Google Scholar] [CrossRef]
- Kiwi, J.; Nadtochenko, V. Evidence for the mechanism of photocatalytic degradation of the bacterial wall membrane at the TiO2 interface by ATR-FTIR and laser kinetic spectroscopy. Langmuir 2005, 21, 4631–41. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Maness, P.; Blake, D.M.; Wolfrum, E.J.; Smolinski, S.L.; Jacoby, W.A. Bactericidal mode of titanium dioxide photocatalysis. J. Photochem. Photobiol. A 2000, 130, 163–170. [Google Scholar] [CrossRef]
- Wainwright, M. Methylene blue derivatives—Suitable photoantimicrobials for blood product disinfection? Int. J. Antimicrob. Agents 2000, 16, 381–394. [Google Scholar] [CrossRef] [PubMed]
- Sunada, K.; Watanabe, T.; Hashimoto, K. Studies on photokilling of bacteria on TiO2 thin film. J. Photochem. Photobiol. A 2003, 156, 227–233. [Google Scholar] [CrossRef]
- Carré, G.; Hamon, E.; Ennahar, S.; Estner, M.; Lett, M.C.; Horvatovich, P.; Gies, J.P.; Keller, V.; Keller, N.; Andre, P. TiO2 Photocatalysis Damages Lipids and Proteins in Escherichia coli. Appl. Environ. Microbiol. 2014, 80, 2573–2581. [Google Scholar] [CrossRef] [PubMed]
- Rincon, A.G.; Pulgarin, C. Field solar E. coli inactivation in the absence and presence of TiO2: Is UV solar dose an appropriate parameter for standardization of water solar disinfection? Sol. Energy 2004, 77, 635–648. [Google Scholar] [CrossRef]
- Pigeot-Rémy, S.; Simonet, F.; Errazuriz-Cerda, E.; Lazzaroni, J.C.; Atlan, D.; Guillard, C. Photocatalysis and disinfection of water: Identification of potential bacterial targets. Appl. Catal. B-Environ. 2011, 104, 390–398. [Google Scholar] [CrossRef]
- Kubacka, A.; Diez, M.S.; Rojo, D.; Bargiela, R.; Ciordia, S.; Zapico, I.; Albar, J.P.; Barbas, C.; Martins dos Santos, V.A.P.; Fernandez-Garcia, M.; et al. Understanding the antimicrobial mechanism of TiO2-based nanocomposite films in a pathogenic bacterium. Sci. Rep. 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Gogniat, G.; Dukan, S. TiO2 Photocatalysis Causes DNA Damage via Fenton Reaction-Generated Hydroxyl Radicals during the Recovery Period. Appl. Environ. Microbiol. 2007, 73, 7740–7743. [Google Scholar] [CrossRef] [PubMed]
- Rincon, A.-G.; Pulgarin, C. Bactericidal action of illuminated TiO2 on pure Escherichia coli and natural bacterial consortia: Post-irradiation events in the dark and assessment of the effective disinfection time. Appl. Catal. B-Environ. 2004, 49, 99–112. [Google Scholar] [CrossRef]
- Dunlop, P.S.M.; Ciavola, M.; Rizzo, L.; McDowell, D.A.; Byrne, J.A. Effect of photocatalysis on the transfer of antibiotic resistance genes in urban wastewater. Catal. Today 2014, in press. [Google Scholar]
- WHO; UNICEF. Progress on Drinking Water and Sanitation: 2014 Update; World Health Organization; UNICEF: Geneva, Switzerland, 2014. [Google Scholar]
- Clasen, T.; Edmondson, P. Sodium dichloroisocyanurate (NaDCC) tablets as an alternative to sodium hypochlorite for the routine treatment of drinking water at a household level. Int. J. Hyg. Environ. Health 2006, 209, 173–181. [Google Scholar] [CrossRef] [PubMed]
- WHO. Economic and Health Effects of Increasing Coverage of Low Cost Household Drinking Water Supply and Sanitation Interventions to Countries off-Track To Meet MDG Target 10; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- Burch, J.D.; Thomas, K.E. Water disinfection for developing countries and potential for solar thermal pasteurization. Sol. Energy 1998, 64, 87–97. [Google Scholar] [CrossRef]
- WHO. Cause-Specific Mortality: Regional Estimates for 2008; World Health Organization: Geneva, Switzerland, 2011. Available online: http://www.who.int/healthinfo/global_burden_disease/estimates_regional/en/index.html (accessed on 20 March 2015).
- Liu, L.; Johnson, H.L.; Cousens, S.; Perin, J.; Scott, S.; Lawn, J.E.; Rudan, I.; Campbell, H.; Cibulskis, R.; Li, M.; et al. Child Health Epidemiology, Global, regional, and national causes of child mortality: An updated systematic analysis for 2010 with time trends since 2000. Lancet 2012, 379, 2151–2161. [Google Scholar] [CrossRef] [PubMed]
- WHO. Cholera. Weekly Epidemiological Report. 2011. Available online: http://www.who.int/wer/2012/wer873132/en/index.html (accessed on 20 March 2015).
- Clasen, T.F.; Haller, L. Water Quality Interventions to Prevent Diarrhoea: Cost and Cost-Effectiveness; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Michael, I.; Rizzo, L.; McArdell, C.S.; Manaia, C.M.; Merlin, C.; Schwartz, T.; Dagot, C.; Fatta-Kassinos, D. Urban wastewater treatment plants as hotspots for the release of antibiotics in the environment: A review. Water Res. 2013, 47, 957–995. [Google Scholar] [CrossRef] [PubMed]
- Bouki, C.; Venieri, D.; Diamadopoulos, E. Detection and fate of antibiotic resistant bacteria in wastewater treatment plants: A review. Ecotox. Environ. Safe 2013, 91, 1–9. [Google Scholar] [CrossRef]
- Huang, J.J.; Hu, H.Y.; Tang, F.; Li, Y.; Lu, S.Q.; Lu, Y. Inactivation and reactivation of antibiotic-resistant bacteria by chlorination in secondary effluents of a municipal wastewater treatment plant. Water Res. 2011, 45, 2775–2781. [Google Scholar] [CrossRef] [PubMed]
- Hedin, G.; Rynbäckm, J.; Loré, B. Reduction of bacterial surface contamination in the hospital environment by application of a new product with persistent effect. J. Hosp. Infect. 2010, 75, 112–115. [Google Scholar] [CrossRef] [PubMed]
- Markowska-Szczupak, A.; Ulfig, K.; Morawski, A.W. The application of titanium dioxide for deactivation of bioparticulates: An overview. Catal. Today 2011, 169, 249–257. [Google Scholar] [CrossRef]
- Alrousan, D.M.A.; Dunlop, P.S.M.; McMurray, T.A.; Byrne, J.A. Photocatalytic inactivation of E. coli in surface water using immobilised nanoparticle TiO2 films. Water Res. 2009, 43, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Amezaga-Madrid, P.; Silveyra-Morales, R.; Cordoba-Fierro, L.; Nevarez-Moorillon, G.V.; Miki-Yoshida, M.; Orrantia-Borunda, E.; Solı́, F.J. TEM evidence of ultrastructural alteration on Pseudomonas aeruginosa by photocatalytic TiO2 thin films. J. Photochem. Photobiol. B 2003, 70, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Hermida, F.; Ares-Mazás, E.; McGuigan, K.G.; Boyle, M.; Sichel, C.; Fernández-Ibáñez, P. Disinfection of drinking water contaminated with Cryptosporidium parvum oocysts under natural sunlight and using the photocatalyst TiO2. J. Photochem. Photobiol. B 2007, 88, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Weir, A.; Westerhoff, P.; Fabricius, L.; von Goetz, N. Titanium Dioxide Nanoparticles in Food and Personal Care Products. Environ. Sci. Technol. 2012, 46, 2242–2250. [Google Scholar] [CrossRef] [PubMed]
- Byrne, J.A.; Fernandez-Ibanez, P.A.; Dunlop, P.S.M.; Alrousan, D.M.A.; Hamilton, J.W.J. Photocatalytic Enhancement for Solar Disinfection of Water: A Review. Int. J. Photoenergy 2011, 12. [Google Scholar] [CrossRef]
- Sordo, C.; van Grieken, R.; Marugán, J.; Fernández-Ibáñez, P. Solar photocatalytic disinfection with immobilised TiO2 at pilot-plant scale. Water Sci. Technol. 2010, 61, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Alrousan, D.M.A.; Polo-López, M.I.; Dunlop, P.S.M.; Fernández-Ibáñez, P.; Byrne, J.A. Solar photocatalytic disinfection of water with immobilised titanium dioxide in re-circulating flow CPC reactors. Appl. Catal. B-Environ. 2012, 128, 126–134. [Google Scholar] [CrossRef]
- Rengifo-Herrera, J.A.; Pulgarin, C. Photocatalytic activity of N, S co-doped and N-doped commercial anatase TiO2 powders towards phenol oxidation and E. coli inactivation under simulated solar light irradiation. Sol. Energy 2010, 84, 37–43. [Google Scholar] [CrossRef]
- Helali, S.; Polo-López, M.I.; Fernández-Ibáñez, P.; Ohtani, B.; Amano, F.; Malato, S.; Guillard, C. Solar photocatalysis: A green technology for E. coli contaminated water disinfection. Effect of concentration and different types of suspended catalyst. J. Photochem. Photobiol. A 2013, 276, 31–40. [Google Scholar] [CrossRef]
- Turki, A.; Kochkara, H.; García-Fernández, I.; Polo-López, M.I.; Ghorbel, A.; Guillard, C.; Berhault, G.; Fernández-Ibáñez, P. Solar photocatalytic inactivation of Fusarium solani over TiO2 nanomaterials with controlled morphology—Formic acid effect. Catal. Today 2013, 209, 147–152. [Google Scholar] [CrossRef]
- Booshehri, A.Y.; Goh, S.C.K.; Hong, J.; Jiang, R.; Xu, R. Effect of depositing silver nanoparticles on BiVO4 in enhancing visible light photocatalytic inactivation of bacteria in water. J. Mater. Chem. A 2014, 2, 6209–6217. [Google Scholar] [CrossRef]
- Vidal, A.; Díaz, A.I.; el Hraiki, A.; Romero, M.; Muguruza, I.; Senhaji, F.; González, J. Solar photocatalysis for detoxification and disinfection of contaminated water: Pilot plant studies. Catal. Today 1999, 54, 283–290. [Google Scholar] [CrossRef]
- McLoughlin, O.A.; Fernández Ibáñez, P.; Gernjak, W.; Malato Rodríguez, S.; Gill, L.W. Photocatalytic disinfection of water using low cost compound parabolic collectors. Sol. Energy 2004, 77, 625–633. [Google Scholar] [CrossRef]
- Puralytics. Available online: http://www.puralytics.com/html/home.php (accessed on 22 December 2014).
- Ubomba-Jaswa, E.; Fernández-Ibáñez, P.; Navntoft, C.; Polo-López, M.I.; McGuigan, K.G. Investigating the microbial inactivation efficiency of a 25 L batch solar disinfection (SODIS) reactor enhanced with a compound parabolic collector (CPC) for household use. J. Chem. Technol. Biotechnol. 2010, 85, 1028–1037. [Google Scholar] [CrossRef]
- Ubomba-Jaswa, E.; Navntoft, C.; Polo-López, M.I.; Fernandez-Ibáñez, P.; McGuigan, K.G. Solar disinfection of drinking water (SODIS): An investigation of the effect of UV-A dose on inactivation efficiency. Photochem. Photobiol. Sci. 2009, 8, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Polo-López, M.I.; Fernández-Ibáñez, P.; García-Fernández, I.; Oller, I.; Salgado-Tránsito, I.; Sichel, C. Resistance of Fusarium sp spores to solar TiO2 photocatalysis: Influence of spore type and water (scaling-up results). J. Chem. Technol. Biotechnol. 2010, 85, 1038–1048. [Google Scholar] [CrossRef]
- Sichel, C.; Tello, J.C.; de Cara, M.; Fernández-Ibáñez, P. Effect of UV solar intensity and dose on the photocatalytic disinfection of bacteria and fungi. Catal. Today 2007, 129, 152–160. [Google Scholar] [CrossRef]
- Polo-López, M.I.; Fernández-Ibáñez, P.; Ubomba-Jaswa, E.; Navntoft, C.; Garcia-Fernandez, I.; Dunlop, P.S.M.; Schmidt, M.; Byrne, J.A.; McGuigan, K.G. Elimination of water pathogens with solar radiation using and automated sequential batch CPC Reactor. J. Hazard. Mater. 2011, 196, 16–21. [Google Scholar] [CrossRef] [PubMed]
- García-Fernández, I.; Fernández-Calderero, I.; Polo-López, M.I.; Fernández-Ibáñez, P. Disinfection of urban effluents using solar TiO2 photocatalysis: A study of significance of dissolved oxygen, temperature, type of microorganism and water matrix. Catal. Today 2014, 240, 30–38. [Google Scholar] [CrossRef]
- Polo-López, M.I.; Oller, I.; Castro-Alferez, M.; Fernández-Ibáñez, P. Assessment of solar photo-Fenton, photocatalysis, and H2O2 for removal of phytopathogenic fungi spores in synthetic and real effluents of urban wastewater. Chem. Eng. J. 2014, 257, 122–130. [Google Scholar] [CrossRef]
- Ducel, G.; Fabry, J.; Nicolle, L. Prevention of Hospital Acquired Infections: A Practical Guide, 2nd ed.; World Health Organisation: Geneva, Switzerland, 2002. [Google Scholar]
- Anon. Improving Patient Care by Reducing the Risk of Hospital Acquired Infection: A Progress Report. 2004. Available online: https://www.nao.org.uk/report/improving-patient-care-by-reducing-the-risk-of-hospital-acquired-infection-a-progress-report/ (accessed on 30 April 2014).
- Anon. MRSA Rates Slashed, but other Bugs a Threat. NHS Choices 2012. Available online: http://www.nhs.uk/news/2012/05may/Pages/mrsa-hospital-acquired-infection-rates.aspx (accessed on 25 April 2014).
- Boyce, J.M. Environmental contamination makes an important contribution to hospital infection. J. Hosp. Infect. 2007, 65, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Dancer, S.J. The role of environmental cleaning in the control of hospital-acquired infection. J. Hosp. Infect. 2009, 73, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Kramer, A.; Schwebke, I.; Kampf, G. How long do nosocomial pathogens persist on inanimate surfaces? A systematic review. BMC Infect. Dis. 2006, 6. [Google Scholar] [CrossRef]
- Pratt, R.J.; Pellowe, C.M.; Wilson, J.A.; Loveday, H.P.; Harper, P.J.; Jones, S.R.; McDougall, C.; Wilcox, M.H. National Evidence-Based Guidelines for Preventing Healthcare-Associated Infections in NHS Hospitals in England. J. Hosp. Infect. 2007, 65, S1–S64. [Google Scholar] [CrossRef] [PubMed]
- Weber, D.J.; Rutala, W.A.; Miller, M.B.; Huslage, K.; Sickbert-Bennett, E. Role of hospital surfaces in the transmission of emerging health care-associated pathogens: Norovirus, Clostridium difficile, and Acinetobacter species. Am. J. Infect. Control 2010, 38, S25–S33. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.C. Environmental Engineering Dictionary, 4th ed.; Government Institutes: Lanham, MD, USA, 2005. [Google Scholar]
- Schabrun, S.; Chipchase, L. Healthcare equipment as a source of nosocomial infection: A systematic review. J. Hosp. Infect. 2006, 63, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Ohko, Y.; Utsumi, Y.; Niwa, C.; Tatsuma, T.; Kobayakawa, K.; Satoh, Y.; Kubota, Y.; Fujishima, A. Self-sterilizing and self-cleaning of silicone catheters coated with TiO2 photocatalyst thin films: A preclinical work. J. Biomed. Mater. Res. 2001, 58, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Brun-Buisson, C. New Technologies and Infection Control Practices to Prevent Intravascular Catheter-related Infections. Am. J. Respir. Crit. Care Med. 2001, 164, 1557–1558. [Google Scholar] [CrossRef] [PubMed]
- Pichat, P. Self-cleaning materials based on solar photocatalysis in “New and future developments in catalysis”. In Solar Catalysis; Suib, S.L., Ed.; Elsevier: Amsterdam, The Netherlands, 2013; Volume 7, pp. 167–190. [Google Scholar]
- Pilkington, Self-cleaning glass. Available online: https://www.pilkington.com/en-gb/uk/householders/types-of-glass/self-cleaning-glass (accessed on 27 March 2015).
- TOTO. Amazing New Tiles that Keep Their Beauty and Cleanliness by the Power of Light. 2011. Available online: http://www.toto.com.hk/tech/hydrotect.html (accessed on 17 April 2014).
- Nakamura, H.; Tanaka, M.; Shinohara, S.; Gotoh, M.; Karube, I. Development of a self-sterilizing lancet coated with a titanium dioxide photocatalytic nano-layer for self-monitoring of blood glucose. Biosens. Bioelectron. 2007, 22, 1920–1925. [Google Scholar] [CrossRef] [PubMed]
- Welch, K.; Cai, Y.; Engqvist, H.; Strømme, M. Dental adhesives with bioactive and on-demand bactericidal properties. Dent. Mater. 2010, 26, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Oka, Y.; Kim, W.-C.; Yoshida, T.; Hirashima, T.; Mouri, H.; Urade, H.; Itoh, Y.; Kubo, T. Efficacy of titanium dioxide photocatalyst for inhibition of bacterial colonization on percutaneous implants. J. Biomed. Mater. Res. B Appl. Biomater. 2008, 86, 530–540. [Google Scholar] [CrossRef] [PubMed]
- Kühn, K.P.; Chaberny, I.F.; Massholder, K.; Stickler, M.; Benz, V.W.; Sonntag, H.-G.; Erdinger, L. Disinfection of surfaces by photocatalytic oxidation with titanium dioxide and UVA light. Chemosphere 2003, 53, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Page, K.; Palgrave, R.G.; Parkin, I.P.; Wilson, M.; Savin, S.L.P.; Chadwick, A.V. Titania and silver-titania composite films on glass-potent antimicrobial coatings. J. Mater. Chem. 2007, 17, 95–104. [Google Scholar] [CrossRef]
- Miron, C.; Roca, A.; Hoisie, S.; Cozorici, P.; Sirghi, L. Photoinduced bactericidal activity of TiO2 thin films obtained by radiofrequency magnetron sputtering deposition. J. Optoelectron. Adv. Mater. 2004, 7, 915–919. [Google Scholar]
- Wong, M.-S.; Chu, W.-C.; Sun, D.-S.; Huang, H.-S.; Chen, J.-H.; Tsai, P.-J.; Lin, N.-T.; Yu, M.-S.; Hsu, S.-F.; Wang, S.-L.; et al. Visible-Light-Induced Bactericidal Activity of a Nitrogen-Doped Titanium Photocatalyst against Human Pathogens. Appl. Environ. Microbiol. 2006, 72, 6111–6116. [Google Scholar] [CrossRef] [PubMed]
- Mitoraj, D.; Janczyk, A.; Strus, M.; Kisch, H.; Stochel, G.; Heczko, P.B.; Macyk, W. Visible light inactivation of bacteria and fungi by modified titanium dioxide. Photochem. Photobiol. Sci. 2007, 6, 642–648. [Google Scholar] [CrossRef] [PubMed]
- Dunnill, C.W.; Aiken, Z.A.; Pratten, J.; Wilson, M.; Parkin, I.P. Sulfur- and Nitrogen-Doped Titania Biomaterials via APCVD. Chem. Vapor Depos. 2010, 16, 50–54. [Google Scholar] [CrossRef]
- Dunnill, C.W.; Page, K.; Aiken, Z.A.; Noimark, S.; Hyett, G.; Kafizas, A.; Pratten, J.; Wilson, M.; Parkin, I.P. Nanoparticulate silver coated-titania thin films—Photo-oxidative destruction of stearic acid under different light sources and antimicrobial effects under hospital lighting conditions. J. Photochem. Photobiol. A 2011, 220, 113–123. [Google Scholar] [CrossRef]
- Tsuang, Y.-H.; Sun, J.-S.; Huang, Y.-C.; Lu, C.-H.; Chang, W.H.-S.; Wang, C.-C. Studies of Photokilling of Bacteria Using Titanium Dioxide Nanoparticles. Artif. Organs 2008, 32, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Shiraishi, K.; Koseki, H.; Tsurumoto, T.; Baba, K.; Naito, M.; Nakayama, K.; Shindo, H. Antibacterial metal implant with a TiO2-conferred photocatalytic bactericidal effect against Staphylococcus aureus. Surf. Interface Anal. 2009, 41, 17–22. [Google Scholar] [CrossRef]
- Bombac, D.; Brojan, M.; Fajfar, P.; Kosel, F.; Turk, R. Review of materials in medical applications. RMZ-Mater. Geoenviron. 2007, 54, 471. [Google Scholar]
- Amin, S.Z.; Smith, L.; Luthert, P.J.; Cheetham, M.E.; Buckley, R.J. Minimising the risk of prion transmission by contact tonometry. Br. J. Ophthalmol. 2003, 87, 1360–1362. [Google Scholar] [CrossRef] [PubMed]
- Su, W.; Wang, S.; Wang, X.; Fu, X.; Weng, J. Plasma pre-treatment and TiO2 coating of PMMA for the improvement of antibacterial properties. Surf. Coat. Technol. 2010, 205, 465–469. [Google Scholar] [CrossRef]
- Suketa, N.; Sawase, T.; Kitaura, H.; Naito, M.; Baba, K.; Nakayama, K.; Wennerberg, A.; Atsuta, M. An Antibacterial Surface on Dental Implants, Based on the Photocatalytic Bactericidal Effect. Clin. Implant Dent. Relat. Res. 2005, 7, 105–111. [Google Scholar] [CrossRef] [PubMed]
- National Health Service UK. Creutzfeldt-Jakob Disease. Available online: http://www.nhs.uk/conditions/Creutzfeldt-Jakob-disease/Pages/Introduction.aspx (accessed on 28 December 2014).
- National CJD Research and Surveillance Unit. CJD in the UK by calendar year. 2014. Available online: http://www.cjd.ed.ac.uk (accessed on 28 December 2014).
- Paspaltsis, I.; Kotta, K.; Lagoudaki, R.; Grigoriadis, N.; Poulios, I.; Sklaviadis, T. Titanium dioxide photocatalytic inactivation of prions. J. Gen. Virol. 2006, 87, 3125–3130. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.H.; Keyes, T.E.; Byrne, J.A. The photocatalytic inactivation effect of Ag–TiO2 on β-amyloid peptide (1–42). J. Photochem. Photobiol. A 2013, 254, 1–11. [Google Scholar] [CrossRef]
- Zhang, D.; Li, G.; Yu, J.C. Inorganic materials for photocatalytic water disinfection. J. Mater. Chem. 2010, 20. [Google Scholar] [CrossRef]
- Gondal, M.A.; Dastageer, M.A.; Khalil, A.; Hayat, K.; Yamani, Z.H. Nanostructured ZnO synthesis and its application for effective disinfection of Escherichia coli microorganism in water. J. Nanopart. Res. 2011, 13, 3423–3430. [Google Scholar] [CrossRef]
- Rodríguez, J.; Paraguay-Delgado, F.; López, A.; Alarcón, J.; Estrada, W. Synthesis and characterization of ZnO nanorod films for photocatalytic disinfection of contaminated water. Thin Solid Films 2010, 519, 729–735. [Google Scholar] [CrossRef]
- Hill, J.C.; Choi, K.-S. Effect of Electrolytes on the Selectivity and Stability of n-type WO3 Photoelectrodes for Use in Solar Water Oxidation. J. Phys. Chem. C 2012, 116, 7612–7620. [Google Scholar] [CrossRef]
- Wang, P.; Huang, B.; Qin, X.; Zhang, X.; Dai, Y.; Whangbo, M-H. Ag/AgBr/WO3·H2O: Visible-Light Photocatalyst for Bacteria Destruction. Inorg. Chem. 2009, 48, 10697–10702. [Google Scholar] [CrossRef] [PubMed]
- Basnet, P.; Larsen, G.K.; Jadeja, R.P.; Hung, Y-C.; Zhao, Y. α-Fe2O3 Nanocolumns and Nanorods Fabricated by Electron Beam Evaporation for Visible Light Photocatalytic and Antimicrobial Applications. Appl. Mater. Interfaces 2013, 5, 2085–2095. [Google Scholar] [CrossRef]
- Meissner, D.; Memming, R.; Kastening, B. Photoelectrochemistry of Cadmium Sulfide 1. Reanalysis of Photocorrosion and Flat-Band Potential. J. Phys. Chem. 1988, 92, 3476–3483. [Google Scholar] [CrossRef]
- Huang, L.; Peng, F.; Yu, H.; Wang, H. Preparation of cuprous oxides with different sizes and their behaviors of adsorption, visible-light driven photocatalysis and photocorrosion. Solid State Sci. 2009, 11, 129–138. [Google Scholar] [CrossRef]
- Hamilton, J.W.J.; Byrne, J.A.; Dunlop, P.S.M.; Brown, N.M.D. Photo-Oxidation of Water Using Nanocrystalline Tungsten Oxide under Visible Light. Int. J. Photoenergy 2008. [Google Scholar] [CrossRef]
- Maeda, K.; Domen, K. New Non-Oxide Photocatalysts Designed for Overall Water Splitting under Visible Light. J. Phys. Chem. C 2007, 111, 7851–7861. [Google Scholar] [CrossRef]
- Yu, H.; Quan, X.; Zhang, Y.; Ma, N.; Chen, S.; Zhao, H. Electrochemically Assisted Photocatalytic Inactivation of Escherichia coli under Visible Light Using a ZnIn2S4 Film Electrode. Langmuir 2008, 24, 7599–7604. [Google Scholar] [CrossRef] [PubMed]
- Woodhouse, M.; Herman, G.S.; Parkinson, B.A. Combinatorial Approach to Identification of Catalysts for the Photoelectrolysis of Water. Chem. Mater. 2005, 17, 4318–4324. [Google Scholar] [CrossRef]
- Takata, T.; Tanaka, A.; Hara, M.; Kondo, J.N.; Domen, K. Recent progress of photocatalysts for overall water splitting. Catal. Today 1998, 44, 17–26. [Google Scholar] [CrossRef]
- Yan, J.; Yang, H.; Tang, Y.; Lu, Z.; Zheng, S.; Yao, M.; Han, Y. Synthesis and photocatalytic activity of CuYyFe2-yO4–CuCo2O4 nanocomposites for H2 evolution under visible light irradiation. Renew. Energy 2009, 34, 2399–2403. [Google Scholar] [CrossRef]
- Yang, J.; Zhong, H.; Li, M.; Zhang, L.; Zhang, Y. Markedly enhancing the visible-light photocatalytic activity of LaFeO3 by post-treatment in molten salt React. Kinet. Catal. Lett. 2009, 97, 269–274. [Google Scholar] [CrossRef]
- Zhang, L.; Tan, P.Y.; Chow, C.L.; Lim, C.K.; Tan, O.K.; Tse, M.S.; Sze, C.C. Antibacterial activities of mechanochemically synthesized perovskite strontium titanate ferrite metal oxide. Colloids Surf. A 2014, 456, 169–175. [Google Scholar] [CrossRef]
- Bessekhouad, Y.; Trari, M. Photocatalytic hydrogen production from suspension of spinel powders AMn2O4 (A = Cu and Zn). Int. J. Hydrog. Energy 2002, 27. [Google Scholar] [CrossRef]
- Wang, D.; Zou, Z.; Ye, J. A new spinel-type photocatalyst BaCr2O4 for H2 evolution under UV and visible light irradiation. Chem. Phys. Lett. 2003, 373, 191–196. [Google Scholar] [CrossRef]
- Yin, J.; Zou, Z.; Ye, J. Photophysical and photocatalytic properties of new photocatalysts MCrO4 (M=Sr, Ba). Chem. Phys. Lett. 2003, 378, 24–28. [Google Scholar] [CrossRef]
- Cui, B.; Lin, H.; Li, Y.Z.; Li, J.B.; Sun, P.; Zhao, X.C.; Liu, C.J. Photophysical and Photocatalytic Properties of Core-Ring Structured NiCo2O4 Nanoplatelets. J. Phys. Chem. C 2009, 113, 14083–14087. [Google Scholar] [CrossRef]
- Wu, W.; Zhang, H.; Chang, S.; Gao, J.; Jia, L.J. Study on the antibacterial performance of perovskite LaCoO3 under visible light illumination. Chem. Ind. Times 2009, 7, 25–28. [Google Scholar]
- Wang, Y.; Zhang, Z.; Zhu, Y.; Li, Z.; Vajtai, R.; Ci, L.; Ajayan, P.M. Nanostructured VO2 Photocatalysts for Hydrogen Production. ACS Nano 2008, 2, 1492–1496. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Zou, Z.; Oshikiri, M.; Matsushita, A.; Shimoda, M.; Imai, M.; Shishido, T. A novel hydrogen-evolving photocatalyst InVO4 active under visible light irradiation. Chem. Phys. Lett. 2002, 356, 221–226. [Google Scholar] [CrossRef]
- Kudo, A.; Ueda, K.; Kato, H. Photocatalytic O2 evolution under visible light irradiation on BiVO4 in aqueous AgNO3 solution. Catal. Lett. 1998, 53, 229–230. [Google Scholar] [CrossRef]
- Kohtani, S.; Makino, S.; Kudo, A.; Tokumura, K.; Ishigaki, Y.; Matsunaga, T.; Nikaido, O.; Hayakawa, K.; Nakagaki, R. Photocatalytic degradation of 4-n-nonylphenol under irradiation from solar simulator: Comparison between BiVO4 and TiO2 photocatalysts. Chem. Lett. 2002, 7, 660–661. [Google Scholar] [CrossRef]
- Tokunaga, S.; Kato, H.; Kudo, A. Selective Preparation of Monoclinic and Tetragonal BiVO4 with Scheelite Structure and Their Photocatalytic Properties. Chem. Mater. 2001, 13, 4624–4628. [Google Scholar] [CrossRef]
- Xie, B. Bactericidal Activity of monoclinic BiVO4 Under Visible Light Irradiation. Chin. J. Disinfect. 2010, 1, 14–16. [Google Scholar]
- Wang, W.; Yu, Y.; An, T.; Li, G.; Yip, H.Y.; Yu, J.C.; Wong, P.K. Visible-Light-Driven Photocatalytic Inactivation of E. coli K-12 by Bismuth Vanadate Nanotubes: Bactericidal Performance and Mechanism. Environ. Sci. Technol. 2012, 46, 4599–4606. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Li, Y.W.; Wu, P.; Xie, R.; Shang, J.K. Palladium Oxide Nanoparticles on Nitrogen-Doped Titanium Oxide: Accelerated Photocatalytic Disinfection and Post-Illumination Catalytic “Memory”. Adv. Mater. 2008, 20, 3717–3723. [Google Scholar] [CrossRef]
- Pena, M.A.; Fie, J.L.G. Chemical Structures and Performance of Perovskite Oxides. Chem. Rev. 2001, 101, 1981–2017. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, H.; Misono, M. Advances in designing perovskite catalysts. Curr. Opin. Solid State Mater. Sci. 2001, 5, 381–387. [Google Scholar] [CrossRef]
- Eng, H.W.; Barnes, P.W.; Auer, B.M.; Woodward, P.M. Investigations of the electronic structure of d0 transition metal oxides belonging to the perovskite family. J. Solid State Chem. 2003, 175, 94–109. [Google Scholar] [CrossRef]
- Fei, D.Q.; Hudaya, T.; Adesina, A.A. Visible-light activated titania perovskite photocatalysts: Characterisation and initial activity studies. Catal. Commun. 2005, 6, 253–258. [Google Scholar] [CrossRef]
- Kato, H.; Kobayashi, H.; Kudo, A. Role of Ag+ in the Band Structures and Photocatalytic Properties of AgMO3 (M: Ta and Nb) with the Perovskite Structure. J. Phys. Chem. B 2002, 106, 12441–12447. [Google Scholar] [CrossRef]
- Yea, J.; Zou, Z.; Matsushita, A. A novel series of water splitting photocatalysts NiM2O6 (M = Nb; Ta) active under visible light. Int. J. Hydrog. Energy 2003, 28, 651–655. [Google Scholar] [CrossRef]
- Gan, H.; Zhang, G.; Huang, H. Enhanced visible-light-driven photocatalytic inactivation of Escherichia coli by Bi2O2CO3/Bi3NbO7 composites. J. Hazard. Mater. 2013, 250, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Lin, H. Visible-light photocatalytic inactivation of Escherichia coli by K4Nb6O17 and Ag/Cu modified K4Nb6O17. J. Hazard. Mater. 2012, 217, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Liu, J.; Sun, D.D.; Ng, W. Graphene oxide–CdS composite with high photocatalytic degradation and disinfection activities under visible light irradiation. J. Hazard. Mater. 2013, 250–251, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yu, J.C.; Xia, D.; Wong, P.K.; Li, Y. Graphene and g-C3N4 Nanosheets Cowrapped Elemental α-Sulfur As a Novel Metal-Free Heterojunction Photocatalyst for Bacterial Inactivation under Visible-Light. Environ. Sci. Technol. 2013, 47, 8724–8732. [Google Scholar] [PubMed]
- Krishna, V.; Pumprueg, S.; Lee, S.-H.; Zhao, J.; Sigmund, W.; Koopman, B.; Moudgi, B.M. Photocatalytic Disinfection with Titanium Dioxide Coated Multi-Wall Carbon Nanotubes. Proc. Saf. Environ. Prot. 2005, 83, 393–397. [Google Scholar] [CrossRef]
- Yao, K.S.; Wang, D.Y.; Chang, C.Y.; Weng, K.W.; Yang, L.Y.; Lee, S.J.; Cheng, T.C.; Hwang, C.C. Photocatalytic disinfection of phytopathogenic bacteria by dye-sensitized TiO2 thin film activated by visible light. Surf. Coat. Technol. 2007, 202, 1329–1332. [Google Scholar] [CrossRef]
- Hou, Y.; Li, X.; Zhao, Q.; Chen, G.; Raston, C.L. Role of Hydroxyl Radicals and Mechanism of Escherichia coli Inactivation on Ag/AgBr/TiO2 Nanotube Array Electrode under Visible Light Irradiation. Environ. Sci. Technol. 2012, 46, 4042–4050. [Google Scholar] [CrossRef] [PubMed]
- Scanlon, D.O.; Dunnill, C.W.; Buckeridge, J.; Shevlin, S.A.; Logsdail, A.J.; Woodley, S.M.; Catlow, C.R.A.; Powell, M.J.; Palgrave, R.G.; Parkin, I.P.; et al. Band alignment of rutile and anatase TiO2. Nat. Mater. 2013, 12, 798–801. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.M.; Chen, C.K.; Chen, C.-J.; Cheng, L.-C.; Wu, P.C.; Cheng, B.H.; Ho, Y.Z.; Tseng, M.L.; Hsu, Y.-Y.; Chan, T.-S.; et al. Plasmon Inducing Effects for Enhanced Photoelectrochemical Water Splitting: X-ray Absorption Approach to Electronic Structures. ACS Nano 2012, 6, 7362–7372. [Google Scholar] [CrossRef] [PubMed]
- Tributsch, H. Multi-electron transfer catalysis for energy conversion based on abundant transition metals. Electrochim. Acta 2007, 52, 2302–2316. [Google Scholar] [CrossRef]
- Hayden, S.C.; Allam, N.K.; el-Sayed, M.A. TiO2 Nanotube/CdS Hybrid Electrodes: Extraordinary Enhancement in the Inactivation of Escherichia coli. J. Am. Chem. Soc. 2010, 132, 14406–14408. [Google Scholar] [CrossRef] [PubMed]
- Vogel, R.; Hoyer, P.; Weller, H. Quantum-Sized PbS, CdS, Ag2S, Sb2S3, and Bi2S3 Particles as Sensitizers for Various Nanoporous Wide-Bandgap Semiconductors. J. Phys. Chem. 1994, 98, 3183–3188. [Google Scholar] [CrossRef]
- Tada, H.; Jin, Q.; Iwaszuk, A.; Nolan, M. Molecular-Scale Transition Metal Oxide Nanocluster Surface-Modified Titanium Dioxide as Solar-Activated Environmental Catalysts. J. Phys. Chem. C 2014, 118, 12077–12086. [Google Scholar] [CrossRef]
- Nolan, M. Surface modification of TiO2 with metal oxide nanoclusters: A route to composite photocatalytic materials. Chem. Commun. 2011, 47, 8617–8619. [Google Scholar] [CrossRef]
- Iwaszuka, A.; Nolan, N. SnO-nanocluster modified anatase TiO2 photocatalyst: Exploiting the Sn(II) lone pair for a new photocatalyst material with visible light absorption and charge carrier separation. J. Mater. Chem. A 2013, 1, 6670–6677. [Google Scholar] [CrossRef]
- Khalil, A.; Gondal, M.A.; Dastageer, M.A. Augmented photocatalytic activity of palladium incorporated ZnO nanoparticles in the disinfection of Escherichia coli microorganism from water. Appl. Catal. A 2011, 402, 162–167. [Google Scholar] [CrossRef]
- Karunakaran, C.; Gomathisankar, P.; Manikandan, G. Preparation and characterization of antimicrobial Ce-doped ZnO nanoparticles for photocatalytic detoxification of cyanide. Mater. Chem. Phys. 2010, 123, 585–594. [Google Scholar]
- Xu, C.; Cao, L.; Su, G.; Liu, W.; Qu, X.; Yu, Y. Preparation, characterization and photocatalytic activity of Co-doped ZnO powders. J. Alloys Compd. 2010, 497, 373–376. [Google Scholar] [CrossRef]
- Karunakaran, C.; Rajeswari, V.; Gomathisankar, P. Enhanced photocatalytic and antibacterial activities of sol–gel synthesized ZnO and Ag-ZnO. Mater. Sci. Semicond. Proc. 2011, 14, 133–138. [Google Scholar] [CrossRef]
- Karunakaran, C.; Rajeswari, V.; Gomathisankar, P. Combustion synthesis of ZnO and Ag-doped ZnO and their bactericidal and photocatalytic activities. Superlattice Microst. 2011, 50, 234–241. [Google Scholar] [CrossRef]
- Karunakaran, C.; Rajeswari, V.; Gomathisankar, P. Optical, electrical, photocatalytic, and bactericidal properties of microwave synthesized nanocrystalline Ag-ZnO and ZnO. Solid State Sci. 2011, 13, 923–928. [Google Scholar] [CrossRef]
- Hamilton, J.W.J.; Byrne, J.A.; McCullagh, C.; Dunlop, P.S.M. Electrochemical Investigation of Doped Titanium Dioxide. Int. J. Photoenergy 2008, 2008. [Google Scholar] [CrossRef]
- Vohra, A.; Goswami, D.Y.; Deshpande, D.A.; Block, S.S. Enhanced photocatalytic disinfection of indoor air. Appl. Catal. B-Environ. 2006, 65, 57–65. [Google Scholar] [CrossRef]
- Sökmena, M.; Candana, F.; Sümer, Z. Disinfection of E. coli by the Ag-TiO2/UV system: Lipid peroxidation. J. Photochem. Photobiol. A 2001, 143, 241–244. [Google Scholar] [CrossRef]
- Karunakarana, C.; Abiramasundaria, G.; Gomathisankara, P.; Manikandana, G.; Anandib, V. Cu-doped TiO2 nanoparticles for photocatalytic disinfection of bacteria under visible light. J. Colloid Interface Sci. 2010, 352, 68–74. [Google Scholar] [CrossRef]
- Yu, J.C.; Ho, W.; Yu, J.; Hoyin Yip, H.; Wong, P.K.; Zhao, J. Efficient Visible-Light-Induced Photocatalytic Disinfection on Sulfur-Doped Nanocrystalline Titania. Environ. Sci. Technol. 2005, 39, 1175–1179. [Google Scholar] [CrossRef] [PubMed]
- Rehman, S.; Ullah, R.; Butt, A.M.; Gohar, N.D. Strategies of making TiO2 and ZnO visible light active. J. Hazard. Mater. 2009, 170, 560–569. [Google Scholar] [CrossRef] [PubMed]
- Im, J.S.; Yun, S.; Lee, Y. Investigation of multielemental catalysts based on decreasing the band gap of titania for enhanced visible light photocatalysis. J. Colloid Interface Sci. 2009, 336, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Xie, R.; Li, Y.W.; Mintz, E.A.; Shang, J.K. Enhanced Visible-Light-Induced Photocatalytic disinfection of E. coli by Carbon-Sensitized Nitrogen-Doped Titanium Oxide. Environ. Sci. Technol. 2007, 41, 5050–5056. [Google Scholar] [CrossRef] [PubMed]
- Hameed, A.; Gondal, M.A.; Yamani, Z.H. Effect of transition metal doping on photocatalytic activity of WO3 for water splitting under laser illumination: role of 3d-orbitals. Cat. Commun. 2004, 5, 715–719. [Google Scholar] [CrossRef]
- Bagabas, A.; Gondal, M.; Khalil, A.; Dastageer, A.; Yamani, Z.; Ashameria, M. Laser-induced photocatalytic inactivation of coliform bacteria from water using Pd-loaded nano-WO3. Stud. Surf. Sci. Catal. 2010, 175, 279–282. [Google Scholar]
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Byrne, J.A.; Dunlop, P.S.M.; Hamilton, J.W.J.; Fernández-Ibáñez, P.; Polo-López, I.; Sharma, P.K.; Vennard, A.S.M. A Review of Heterogeneous Photocatalysis for Water and Surface Disinfection. Molecules 2015, 20, 5574-5615. https://doi.org/10.3390/molecules20045574
Byrne JA, Dunlop PSM, Hamilton JWJ, Fernández-Ibáñez P, Polo-López I, Sharma PK, Vennard ASM. A Review of Heterogeneous Photocatalysis for Water and Surface Disinfection. Molecules. 2015; 20(4):5574-5615. https://doi.org/10.3390/molecules20045574
Chicago/Turabian StyleByrne, John Anthony, Patrick Stuart Morris Dunlop, Jeremy William John Hamilton, Pilar Fernández-Ibáñez, Inmaculada Polo-López, Preetam Kumar Sharma, and Ashlene Sarah Margaret Vennard. 2015. "A Review of Heterogeneous Photocatalysis for Water and Surface Disinfection" Molecules 20, no. 4: 5574-5615. https://doi.org/10.3390/molecules20045574
APA StyleByrne, J. A., Dunlop, P. S. M., Hamilton, J. W. J., Fernández-Ibáñez, P., Polo-López, I., Sharma, P. K., & Vennard, A. S. M. (2015). A Review of Heterogeneous Photocatalysis for Water and Surface Disinfection. Molecules, 20(4), 5574-5615. https://doi.org/10.3390/molecules20045574







