New Synthesis Method for Sultone Derivatives: Synthesis, Crystal Structure and Biological Evaluation of S-CA
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
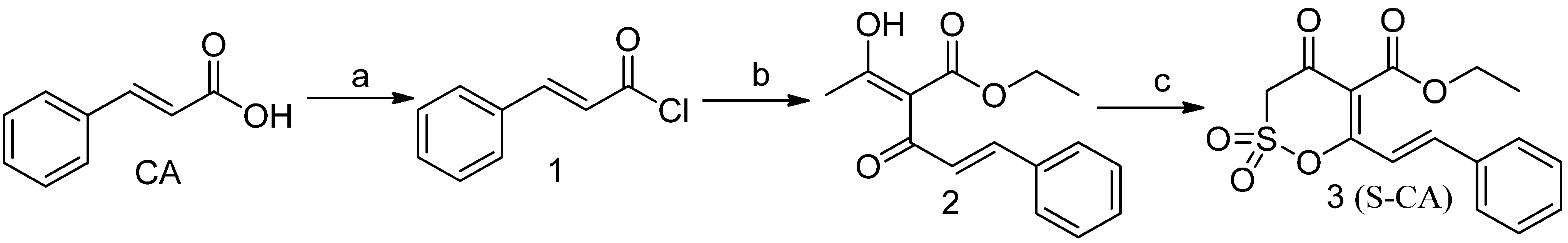
2.1.1. Synthesis of S-CA
2.1.2. Crystal Structure of S-CA
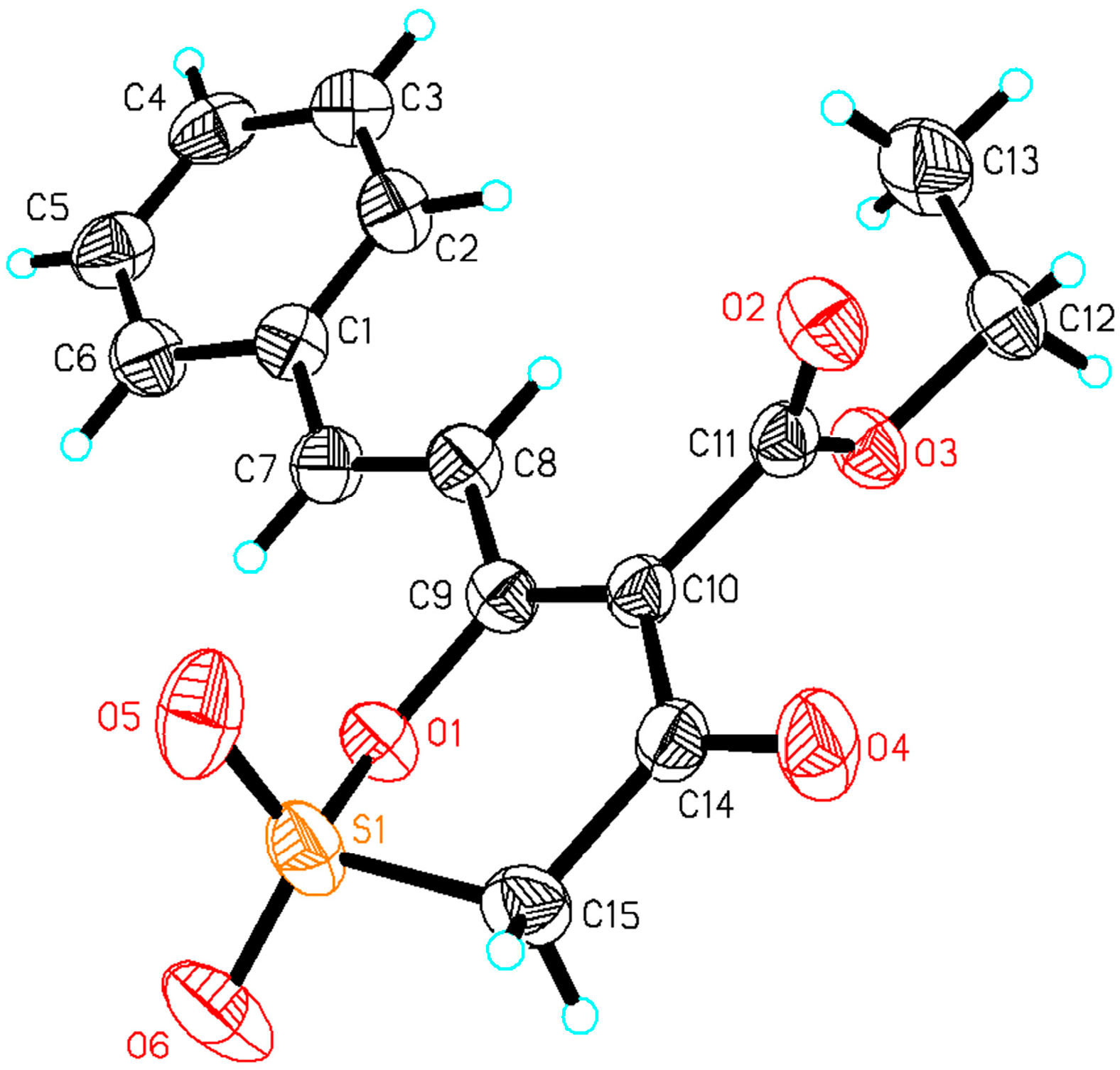
| Phase | Compound |
|---|---|
| Molecular formula | C15 H14 O6 S |
| Formula weight | 322.32 |
| T/K | 296(2) K |
| Wavelength/nm | 0.71073 |
| Crystal system | Triclinic |
| Space group | P-1 |
| a/Å | 8.5023(2) |
| b/Å | 9.1314(2) |
| c/Å | 10.6477(2) |
| α (°) | 71.512(2) |
| β (°) | 83.356(2) |
| γ (°) | 72.179(2) |
| V (Å3) | 746.22(3) |
| Z | 2 |
| F(000) | 336 |
| Dcalc (mg/m3) | 1.434 |
| Absorption coefficient (mm−1) | 0.243 |
| θ range/(°) | 2.02–27.59 |
| Limiting indices | −10 ≤ h ≤ 11, −11 ≤ k ≤ 11, −13 ≤ l ≤ 13 |
| Reflections collected/unique | 7340/3419 [R(int) = 0.0197] |
| Completeness to theta | 99.0% |
| Data/restraints/parameters | 3419/0/199 |
| Refinement method | Full-matrix least-squares on F2 |
| Final R indices[I > 2σ(I)] | R1 = 0.0513, wR2 = 0.1377 |
| R indices (all data) | R1 = 0.0822, wR2 = 0.1586 |
| Goodness-of-fit on F2 | 1.055 |
| Largest diff. peak and hole/(e·Å ×10−3) | 0.656 and −0.215 |
| CCDC | 1052338 |
2.2. Biological Evaluation
2.2.1. Angiogenesis Activity
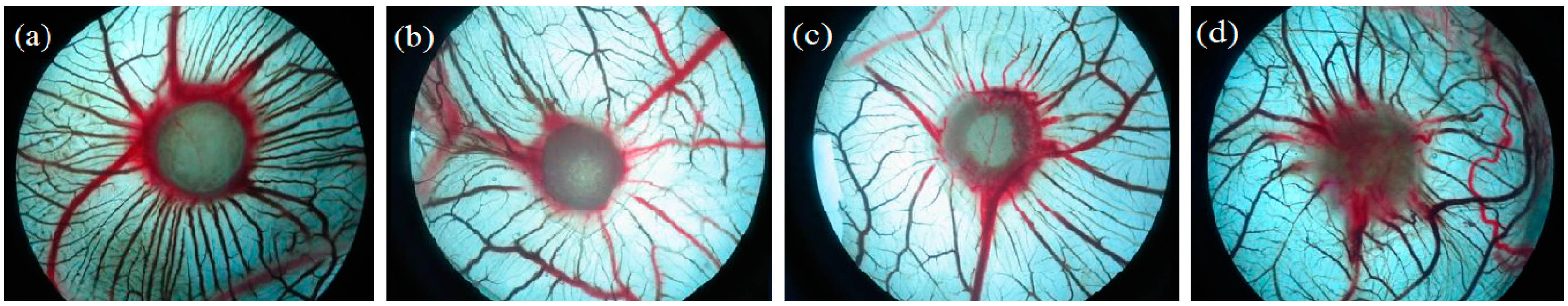
| Group | n | Dose (μg/egg) | Large Vessels | Medium Vessels | Small Vessels |
|---|---|---|---|---|---|
| Control | 13 | - | 8.92 ± 4.80 | 11.84 ± 3.33 | 11.6 ± 2.07 |
| Thalidomide | 13 | 20 | 12.4 ± 4.77 | 12.1 ± 3.92 | 4.16 ± 3.04 * |
| S-CA | 12 | 10 | 12.5 ± 6.94 | 10.33 ± 5.25 | 5.1 ± 1.22 * |
| S-CA | 12 | 40 | 12.6 ± 7.68 | 9.00 ± 4.44 | 3.5 ± 2.73 * |
2.2.2. Acute Toxicity
| Group | Mice Number Start/End | Dose (mg/kg) | Death Rate (%) | LD50 (mg/kg) | 95% CIs |
|---|---|---|---|---|---|
| 1 | 10/10 | 10 | 10 | 25.624 | 13.04–50.34 |
| 2 | 10/8 | 20 | 20 | ||
| 3 | 10/4 | 30 | 60 | ||
| 4 | 10/0 | 50 | 100 |
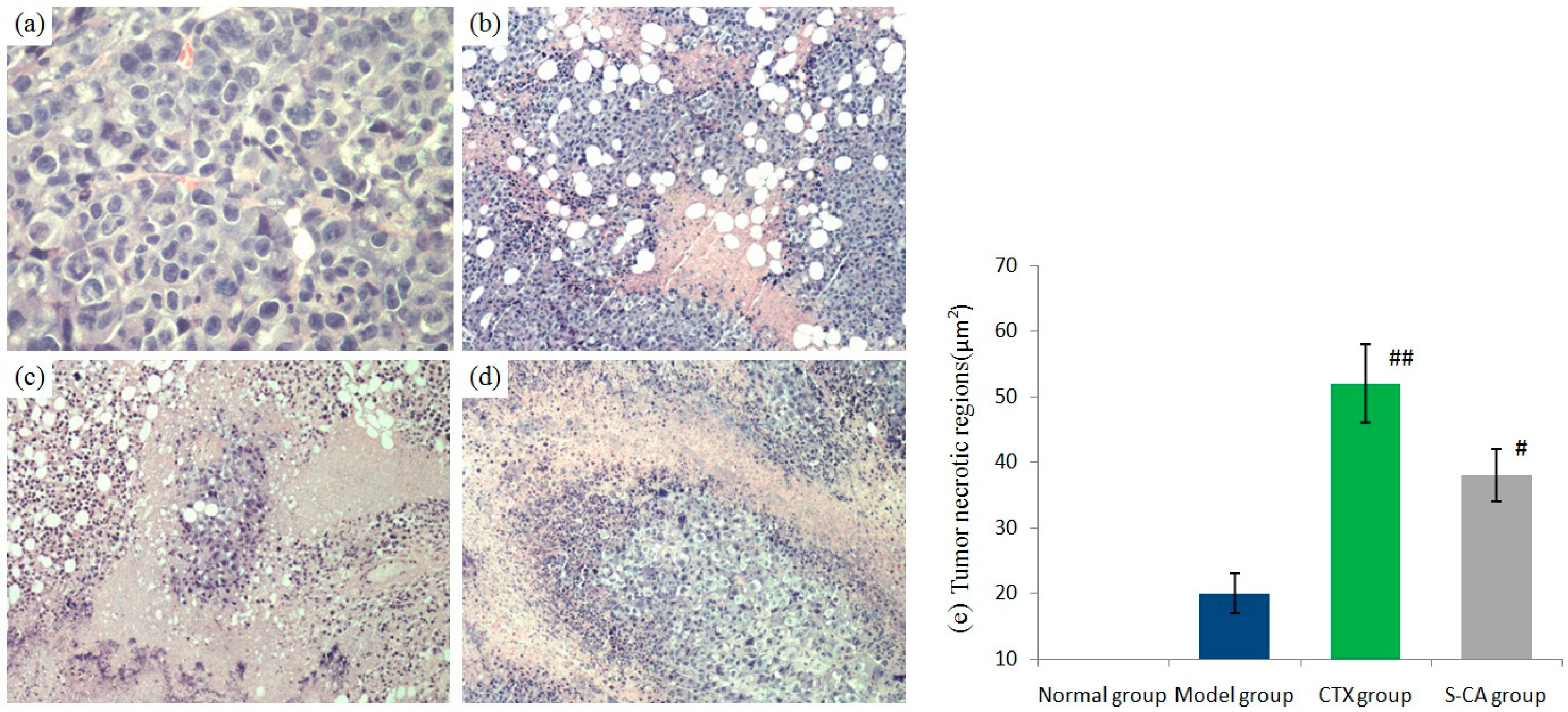
2.2.3. S-CA’s Antitumor Activity in Vivo
| Group | Dose (mg/kg) | Mice Number Start/End | Tumor Weight (g) | Inhibitory Rate (%) | Liver Index (100× g/g) | Spleen Index (mg/g) |
|---|---|---|---|---|---|---|
| Normal | - | 10/10 | - | - | 4.24 ± 0.54 | 4.27 ± 0.75 |
| Model | - | 10/10 | 0.684 ± 0.416 | - | 4.51 ± 0.53 | 5.25 ± 0.94 |
| CTX | 20 | 10/10 | 0.249 ± 0.142 # | 63.52% | 4.98 ± 0.26 | 4.50 ± 1.39 |
| S-CA | 10 | 10/10 | 0.342 ± 0.291 # | 44.71% | 4.81 ± 0.32 | 6.33 ± 1.51 * |
3. Experimental Section
3.1. General Information
3.2. Chemistry
3.2.1. Synthesis of S-CA
3.2.2. Crystal Structure
3.3. Bio-Evaluation Methods
3.3.1. Angiogenesis Assay
3.3.2. Acute Toxicity Test
3.3.3. Antitumor Activity in Vivo
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhang, C.P.; Chen, Q.Y.; Guo, Y.; Xiao, J.C.; Gu, Y.C. Difluoromethylation and trifluoromethylation reagents derived from tetrafluoroethane β-sultone: Synthesis, reactivity and applications. Coord. Chem. Rev. 2014, 261, 28–72. [Google Scholar] [CrossRef]
- Morimoto, Y.; Kurihara, H.; Kinoshita, T. Can α-sultone exist as a chemical species? First experimental implication for intermediacy of α-sultone. Chem. Commun. 2000, 3, 189–190. [Google Scholar] [CrossRef]
- Bhowmik, B.B.; Ganguly, P. Photophysics of xanthene dyes in surfactant solution. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2005, 61, 1997–2003. [Google Scholar] [CrossRef] [PubMed]
- Ghandi, M.; Taghi Nazeri, M.; Kubicki, M. An efficient one-pot, regio-and stereoselective synthesis of novel pentacyclic-fused pyrano[3,2,c]chromenone or quinolinone benzosultone derivatives in water. Tetrahedron 2013, 69, 4979–4989. [Google Scholar] [CrossRef]
- Karsch, S.; Freitag, D.; Schwab, P. Ring closing metathesis in the synthesis of sultones and sultams. Synthesis 2004, 10, 1696–1712. [Google Scholar]
- Natrajan, A.; Wen, D. A green synthesis of chemiluminescent N-sulfopropyl acridinium esters in ionic liquids without using the carcinogen 1, 3-propane sultone. Green Chem. Lett. Rev. 2013, 6, 237–248. [Google Scholar] [CrossRef]
- Camarasa, M.J. Synthesis of 3'-Spiro-substituted Nucleosides: Chemistry of TSAO Nucleoside Derivatives. In Chemical Synthesis of Nucleoside Analogues; Merino, P., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2013; Chapter 10; pp. 427–472. [Google Scholar]
- Mondal, S. Recent Developments in the Synthesis and Application of Sultones. Chem. Rev. 2012, 112, 5339–5355. [Google Scholar] [CrossRef] [PubMed]
- Mondal, S.; Debnath, S.; Das, B. Synthesis of seven-membered fused sultones by reductive Heck cyclization: An investigation for stereochemistry through DFT study. Tetrahedron 2014, 71, 476–486. [Google Scholar] [CrossRef]
- Qi, Z.; Wang, M.; Li, X. Rh (III)-Catalyzed synthesis of sultones through C–H activation directed by a sulfonic acid group. Chem. Commun. 2014, 50, 9776–9778. [Google Scholar] [CrossRef]
- Lewis, F.W.; Grayson, D.H. An improved synthesis of 10-isobornylsultone. Tetrahedron: Asymmetry 2014, 25, 1150–1152. [Google Scholar] [CrossRef]
- De Castro, S.; García-Aparicio, C.; Andrei, G.; Snoeck, R.; Blazarini, J.; Carmarasa, M.J.; Velazquez, S. 4-Benzyloxy-γ-Sultone Derivatives: Discovery of a Novel Family of Non-Nucleoside Inhibitors of Human Cytomegalovirus and Varicella Zoster Virus. J. Med. Chem. 2009, 52, 1582–1591. [Google Scholar] [CrossRef] [PubMed]
- De Castro, S.; Familiar, O.; Andrei, G.; Snoeck, R.; Balzarini, J.; Camarasa, M.J.; Zquez, S.V. From β-Amino-g-sultone to Unusual Bicyclic Pyridine and Pyrazine Heterocyclic Systems: Synthesis and Cytostatic and Antiviral Activities. ChemMedChem 2011, 6, 686–697. [Google Scholar] [CrossRef] [PubMed]
- De Castro, S.; Peromingo, M.T.; Naesens, L.; Andrei, G.; Snoeck, R.; Balzarini, J.; Velazquez, S.; Camarasa, M.J. 4''-Benzoylureido-TSAO Derivatives as Potent and Selective Non-Nucleoside HCMV Inhibitors. Structure-Activity Relationship and Mechanism of Antiviral Action. J. Med. Chem. 2008, 51, 5823–5832. [Google Scholar] [CrossRef] [PubMed]
- Hensel, A. γ-Propoxy-sulfo-lichenin, an antitumor polysaccharide derived from lichenin. Pharm. Acta Helv. 1995, 70, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Walles, S.A. Determination of reaction rate constants for alkylation of 4-(p-Nitrobenzyl) pyridine by different alkylating agents. Toxicol. Lett. 1980, 5, 161–167. [Google Scholar] [CrossRef] [PubMed]
- García-Fernández, L.; Halstenberg, S.; Unger, R.E. Anti-angiogenic activity of heparin-like polysulfonated polymeric drugs in 3D human cell culture. Biomaterials 2010, 31, 7863–7872. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.L.; She, G.M.; Yang, Y.N.; Li, Q.; Zhang, H.G.; Liu, J.; Cao, Y.Q.; Xu, X.; Lei, H.M. Synthesis and Biological Evaluation of New Ligustrazine Derivatives as Anti-Tumor Agents. Molecules 2012, 17, 4972–4985. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Jin, Z.X.; Liu, X.J.; Fan, D.; Sun, Y.; Zhao, H.Y.; Zhu, Z.H.; Liu, Z.F.; Jia, B.; Wang, F. PET Imaging of Neovascularization with 68Ga-3PRGD2 for Assessing Tumor Early Response to Endostar Antiangiogenic Therapy. Mol. Pharm. 2014, 11, 3915–3922. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.M.; Shu, Z.; He, W.J.; Chen, L.Y.; Yang, S.L.; Yang, G.; Liu, Y.L.; Li, X.R. Antitumor activity of Pulsatilla chinensis (Bunge) Regel saponins in human liver tumor 7402 cells in vitro and in vivo. Phytomedicine 2012, 19, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.L.; Zhang, Y.Z.; Xu, K.; Li, Q.; Zhang, H.G.; Guo, J.; Pang, D.D.; Cheng, Y.T.; Lei, H.M. A new ligustrazine derivative--pharmacokinetic evaluation and antitumor activity by suppression of NF-kappaB/p65 and COX-2 expression in S180 mice. Pharmazie 2013, 68, 782–789. [Google Scholar] [PubMed]
- Liao, N.; Ao, M.; Zhang, P.; Yu, L. Extracts of Lycoris aurea induce apoptosis in murine sarcoma S180 cells. Molecules 2012, 17, 3723–3735. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Sample of the compound S-CA is available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, B.; Yan, W.; Zhang, C.; Zhang, Y.; Liang, M.; Chu, F.; Gong, Y.; Xu, B.; Wang, P.; Lei, H. New Synthesis Method for Sultone Derivatives: Synthesis, Crystal Structure and Biological Evaluation of S-CA. Molecules 2015, 20, 4307-4318. https://doi.org/10.3390/molecules20034307
Li B, Yan W, Zhang C, Zhang Y, Liang M, Chu F, Gong Y, Xu B, Wang P, Lei H. New Synthesis Method for Sultone Derivatives: Synthesis, Crystal Structure and Biological Evaluation of S-CA. Molecules. 2015; 20(3):4307-4318. https://doi.org/10.3390/molecules20034307
Chicago/Turabian StyleLi, Bi, Wenqiang Yan, Chenze Zhang, Yuzhong Zhang, Miao Liang, Fuhao Chu, Yan Gong, Bing Xu, Penglong Wang, and Haimin Lei. 2015. "New Synthesis Method for Sultone Derivatives: Synthesis, Crystal Structure and Biological Evaluation of S-CA" Molecules 20, no. 3: 4307-4318. https://doi.org/10.3390/molecules20034307
APA StyleLi, B., Yan, W., Zhang, C., Zhang, Y., Liang, M., Chu, F., Gong, Y., Xu, B., Wang, P., & Lei, H. (2015). New Synthesis Method for Sultone Derivatives: Synthesis, Crystal Structure and Biological Evaluation of S-CA. Molecules, 20(3), 4307-4318. https://doi.org/10.3390/molecules20034307






