In Vitro and in Vivo Evaluation of Novel Cross-Linked Saccharide Based Polymers as Bile Acid Sequestrants
Abstract
:1. Introduction
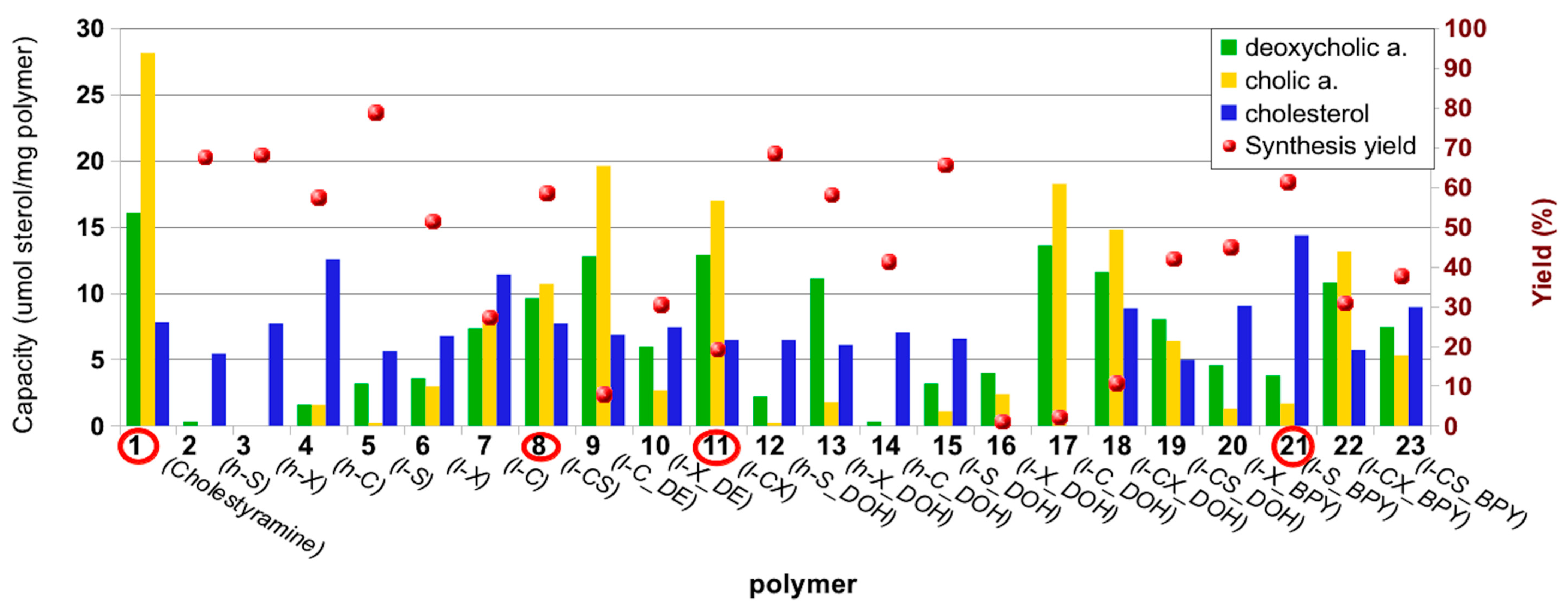
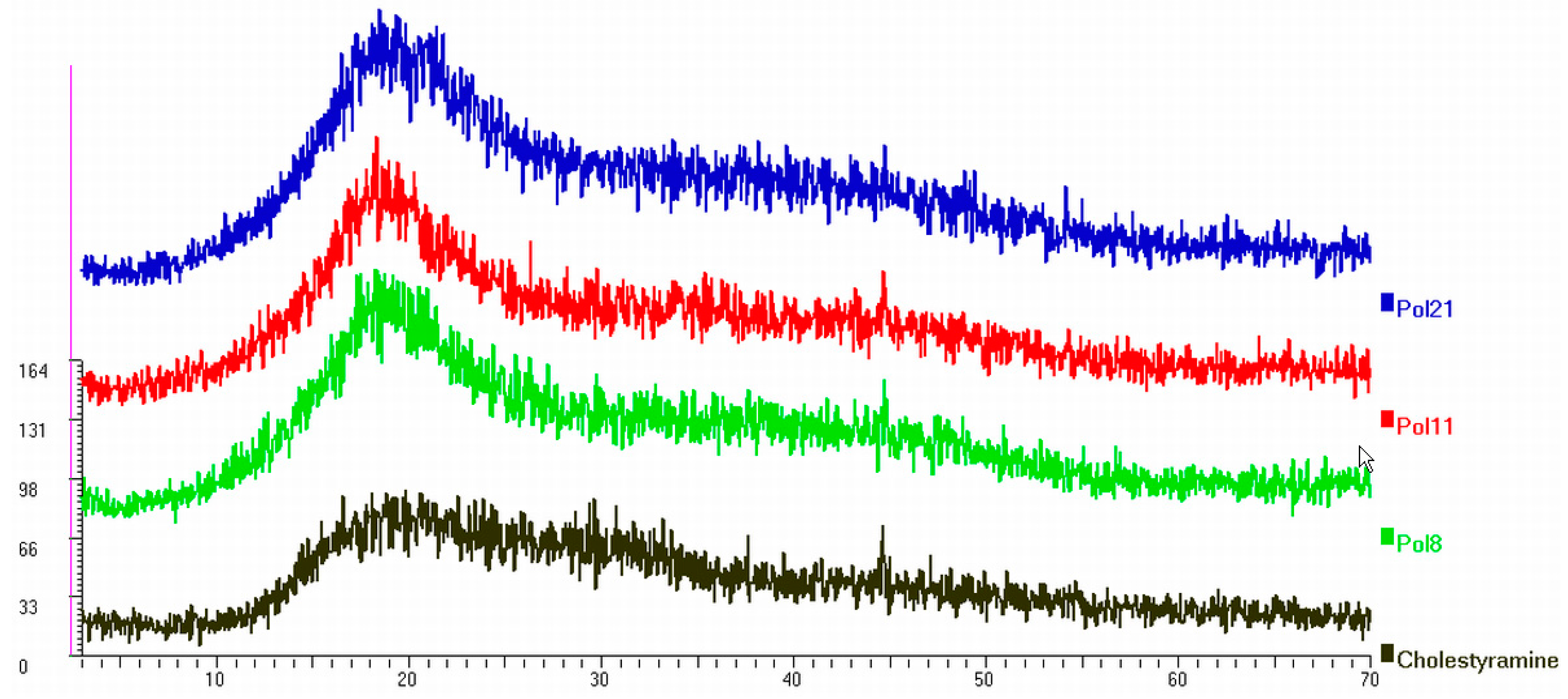
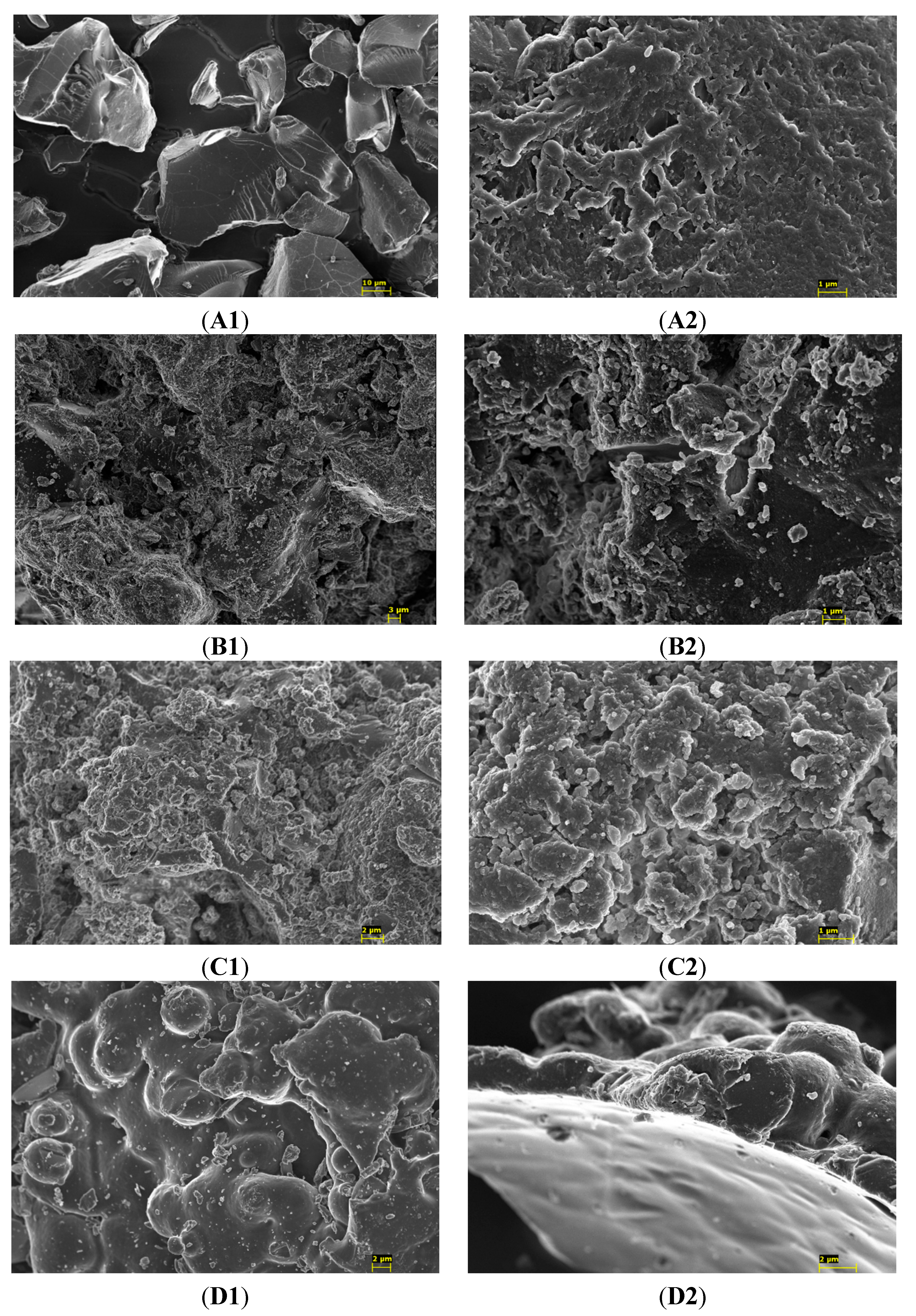
2. Results and Discussion
| Compound | % N | % C | % S |
|---|---|---|---|
| Cholestyramine | 5.6 ± 0.11 | 63.4 ± 0.15 | - |
| Polymer 8 | - | 39.9 ± 0.05 | 9.5 ± 0.11 |
| Polymer 11 | - | 40.3 ± 0.14 | 8.6 ± 0.03 |
| Polymer 21 | 0.4 ± 0.06 | 41.1 ± 0.00 | 8.5 ± 0.00 |
| CHOL (mmol/L) | TG (mmol/L) | Creatinine (µmol/L) | AST (U/L) | ALT (U/L) | GGT (U/L) | Glucose (mmol/L) | |
|---|---|---|---|---|---|---|---|
| Control | 1.93 ± 0.22 | 0.73 ± 0.15 | 51.27 ± 4.42 | 113.2 ± 26.8 | 52.4 ± 4.72 | 0.86 ± 1.23 | 12.17 ± 3.98 |
| Diet | 2.71 ± 0.54 (p < 0.02) | 1.25 ± 0.23 (p < 0.01) | 63.65 ± 7.96 (p < 0.05) | 156.0 ± 72.89 | 60.0 ± 9.06 | 0.99 ± 1.58 | 9.31 ± 3.34 |
| Cholestyramine | 1.8 ± 0.32 | 1.98 ± 0.83 (p < 0.02) | 64.53 ± 6.19 (p < 0.01) | 161.2 ± 8.59 | 74.0 ± 30.18 | 1.36 ± 1.58 | 11.01 ± 1.83 |
| Polymer 8 | 1.75 ± 0.2 | 0.89 ± 0.23 | 48.62 ± 6.19 | 118.4 ± 9.53 | 53.2 ± 5.02 | 4.06 ± 3.14 | 11.2 ± 1.64 |
| Polymer 11 | 1.61 ± 0.24 | 0.82 ± 0.22 | 58.22 ± 9.72 | 102.4 ± 13.20 | 41.0 ± 4.95 | 1.04 ± 0.97 | 7.64 ± 3.54 |
| Polymer 21 | 1.64 ± 0.41 | 0.74 ± 0.19 | 48.62 ± 4.42 | 118.2 ± 11.52 | 55.8 ± 10.38 | 1.47 ± 1.01 | 6.07 ± 0.34 (p < 0.01) |
3. Experimental Section
3.1. Materials
3.2. Synthesis of a Library of BAS Polymers
3.2.1. Neutral Homo-Polymers
3.2.2. Neutral Hetero-Polymers
3.2.3. Charged Homo- and Hetero-Polymers
3.3. Analysis of the Sequestrant Capability of the Library of BAS Polymers
3.4. Scale-Up Synthesis of BAS Polymers for Animal Experimentation
3.5. Characterization of the BAS Polymers Assayed in Animal Experimentation
3.6. Animal Experimentation
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Burden: Mortality, morbidity and risk factors. In Global Status Report on Noncommunicable Diseases 2010; Alwan, A. (Ed.) World Health Organization: Geneva, Switzerland, 2011; pp. 9–11.
- Rideout, T.C.; Hading, S.V.; Marinangeli, C.P.F.; Jones, P.J.H. Combination drug–diet therapies for dyslipidemia. Transl. Res. 2010, 155, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Rozman, D.; Monostory, K. Perspectives of the non-statin hypolipidemic agents. Pharmacol. Ther. 2010, 127, 19–40. [Google Scholar] [CrossRef] [PubMed]
- Brautbar, A.; Ballantyne, C.M. Pharmacological strategies for lowering LDL cholesterol: Statins and beyond. Nat. Rev. Cardiol. 2011, 8, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, V.; Khokhar, M. Mechanism of action of anti-hypercholesterolemia drugs and their resistance. Eur. J. Pharmacol. 2014, 741, 156–170. [Google Scholar] [CrossRef] [PubMed]
- Staels, B.; Fonseca, V.A. Bile acids and metabolic regulation. Diabetes Care 2009, 32, S237–S245. [Google Scholar] [CrossRef] [PubMed]
- Van der Wulp, M.Y.M.; Verkade, H.J.; Groen, A.K. Regulation of cholesterol homeostasis. Mol. Cell. Endocrinol. 2013, 368, 1–16. [Google Scholar]
- Mendoza, P.V.; Serra, A.C.; Silva, C.L.; Simoes, S.; Coelho, J.F.J. Polymeric bile acid sequestrants. Synthesis using conventional methods and new approaches based on “controlled”/living radical polymerization. Prog. Polym. Sci. 2013, 38, 445–461. [Google Scholar] [CrossRef]
- Nichifor, M.; Zhu, X.X.; Baille, W.; Cristea, D.; Carpov, A. Bile acid sequestrants based on cationic dextran hydrogel microspheres. 2. Influence of the length of alkyl substituents at the amino groups of the sorbents on the sorption of bile salts. J. Pharm. Sci. 2001, 90, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Nichifor, M.; Cristea, D.; Carpov, A. Sodium cholate sorption on cationic dextran hydrogel microspheres. 1. Influence of the chemical structure of functional groups. Int. J. Biol. Macromol. 2000, 28, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.K.; Kim, S.Y.; Kim, S.U.; Kim, J.H. Synthesis of cationic polysaccharide derivatives and their hypocholesterolaemic capacity. Biotechnol. Appl. Biochem. 2002, 35, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.K.; Kim, S.U.; Kim, J.H. Modification of chitosan to improve its hypocholesterolemic capacity. Biosci. Biotechnol. Biochem. 1999, 63, 833–839. [Google Scholar] [CrossRef] [PubMed]
- Murata, Y.; Kodoma, Y.; Hirai, D.; Kofugi, K.; Kawashima, S. Properties of an oral preparation containing a chitosan salt. Molecules 2009, 14, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Loftsson, T.; Brewster, E. Pharmaceutical applications of cyclodextrins: Effects on drug permeation through biological membranes. J. Pharm. Pharmacol. 2011, 63, 1119–1135. [Google Scholar] [CrossRef] [PubMed]
- Loftsson, T.; Brewster, E. Cyclodextrins as Functional Excipients: Methods to Enhance Complexation Efficiency. J. Pharm. Sci. 2012, 101, 3019–3032. [Google Scholar] [CrossRef] [PubMed]
- Favier, M.L.; Memesy, C.; Moundras, C.; Demigne, C. Effect of cyclodextrin on plasma lipids and cholesterol metabolism in the rat. Metabolism 1995, 44, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Trautwein, E.A.; Forgbert, K.; Rieckhoff, D.; Erbersdobler, H.F. Impact of beta-cyclodextrin and resistant starch on bile acid metabolism and fecal steroid excretion in regard to their hypolipidemic action in hamsters. Biochim. Biophys. Acta 1999, 1437, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Mediavilla, V.; Villares, C.; Culebras, J.M.; Bayón, J.E.; Gonzalez-Gallego, J. Effects of dietary β-cyclodextrin in hypercholesterolemic rats. Pharmacol. Toxicol. 2003, 92, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Park, B.S.; Jang, A. Effects of Dietary β-Cyclodextrin on Plasma Lipid and Tissue Cholesterol Content in Swine. Asian-Aust. J. Anim. Sci. 2007, 20, 100–105. [Google Scholar] [CrossRef]
- Kang, H.K.; Park, B.S. Cholesterol Contents of Pork Fed Dietary β-Cyclodextrin. J. Korean Soc. Food Sci. Nutr. 2007, 36, 180–185. [Google Scholar] [CrossRef]
- Wagner, E.M.; Jen, K.L.C.; Artiss, J.D.; Remaley, A.T. Dietary α-cyclodextrin lowers low-density lipoprotein cholesterol and alters plasma fatty acid profile in low-density lipoprotein receptor knockout mice on a high-fat diet. Metabolism 2008, 57, 1046–1051. [Google Scholar] [CrossRef] [PubMed]
- Abadie, C.; Hug, M.; Kübli, C.; Gains, N. Effect of cyclodextrins and undigested starch on the loss of chenodeoxycholate in the faeces. Biochem. J. 1994, 299, 725–730. [Google Scholar] [PubMed]
- Evaluation of Oral Alpha-Cyclodextrin for Decreasing Seum Cholesterol. Available online: http://clinicaltrials.gov/show/NCT01131299 (accessed on 4 November 2014).
- Van Der Manakker, F.; Vermonden, T.; van Nostrum, C.F.; Hennink, W.E. Cyclodextrin-based polymeric materials: Synthesis, properties, and pharmaceutical/biomedical applications. Biomacromolecules 2009, 10, 3157–3175. [Google Scholar] [CrossRef] [PubMed]
- Hoang Thi, T.H.; Chai, F.; Lepretre, S.; Blanchemain, N.; Martel, B.; Siepmann, F.; Hildebrand, H.F.; Siepmann, J.; Flament, M.P. Bone implants modified with cyclodextrin: Study of drug release in bulk fluid and into agarose gel. Int. J. Pharm. 2010, 400, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Blanchemain, N.; Karrout, Y.; Tabary, N.; Neut, C.; Bria, M.; Siepmann, J.; Hildebrand, H.F.; Martel, B. Methyl-beta-cyclodextrin modified vascular prosthesis: Influence of the modification level on the drug delivery properties in different media. Acta Biomater. 2011, 7, 304–314. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.G.; Zhang, J.F.; Hao, S.L.; Wu, D.-J.; Liu, L.; Xu, Y. Preparation, Characterization of Hydrophobic Drug in Combine Loaded Chitosan/Cyclodextrin/Trisodium Citrate Nanoparticles and in vitro Release Study. Colloids Surf. B 2011, 83, 103–107. [Google Scholar] [CrossRef]
- Anand, R.; Malanga, M.; Manet, I.; Manoli, F.; Tuza, K.; Aykac, A.; Ladaviere, C.; Fenyvesi, E.; Vargas-Berenguel, A.; Gref, R.; et al. Citric acid–γ-cyclodextrin crosslinked oligomers as carriers for doxorubicin delivery. Photochem. Photobiol. Sci. 2013, 12, 1841–1854. [Google Scholar] [CrossRef] [PubMed]
- Morales-Sanfrutos, J.; Lopez-Jaramillo, F.J.; Elremaily, M.A.A.; Hernandez-Mateo, F.; Santoyo-Gonzalez, F. Divinyl sulfone cross-linked cyclodextrin-based polymeric materials: Synthesis and applications as sorbents and encapsulating agents. Molecules 2015, 20, 3565–3581. [Google Scholar] [CrossRef] [PubMed]
- Baile, W.E.; Huang, W.Q.; Nichifor, M.; Zhu, X.X. Functionalized β-cyclodextrin polymers for the sorption of bile salts. J. Macromol. Sci. Part A: Pure Appl. Chem. 2000, 37, 677–690. [Google Scholar] [CrossRef]
- Gylling, H.M.; Mietinnen, T.A. The effect of cholesterol absorption inhibition on low density lipoprotein cholesterol level. Atherosclerosis 1995, 117, 305–308. [Google Scholar] [CrossRef] [PubMed]
- Staels, B.; Handelsman, Y.; Fonseca, V. Bile acid sequestrants for lipid and glucose control. Curr. Diab. Rep. 2010, 10, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Forristal, I. The chemistry of α,β-unsaturated sulfoxides and sulfones: An update. J. Sulfur Chem. 2005, 26, 163–195. [Google Scholar] [CrossRef]
- Aows, D.C.; Gervay-Hague, J. Vinyl sulfones: Synthetic preparations and medicinal chemistry applications. Med. Res. Rev. 2006, 26, 793–814. [Google Scholar] [CrossRef] [PubMed]
- Comini, S.; Olivier, P.; Riottot, M.; Duhamel, D. Interaction of β-cyclodextrin with bile acids and their competition with vitamins A and D3 as determined by 1H-NMR spectroscopy. Clin. Chim. Acta 1994, 228, 181–194. [Google Scholar] [CrossRef] [PubMed]
- Weingärtner, O.; Lütjohann, D.; Böhm, M.; Laufs, U. Relationship between cholesterol synthesis and intestinal absorption is associated with cardiovascular risk. Artherosclerosis 2010, 210, 362–365. [Google Scholar] [CrossRef]
- Han, E.M.; Kim, S.H.; Ahn, J.; Kwak, H.S. Comparison of cholesterol-reduced cream cheese manufactured using crosslinked β-cyclodextrin to regular cream cheese. Asian-Australas J. Anim. Sci. 2008, 21, 131–137. [Google Scholar] [CrossRef]
- Shonbeck, C.; Westh, P.; Madsen, J.C.; Larsen, K.L.; Stade, L.W.; Holm, R. Methylated β-Cyclodextrins: Influence of degree and pattern of substitution on the thermodynamics of complexation with tauro- and glyco-conjugated bile salts. Langmuir 2011, 27, 5832–5841. [Google Scholar] [CrossRef] [PubMed]
- Hinrichs, W.; Buttner, G.; Steifa, M.; Betzel, C.H.; Zabel, V.; Pfannemuller, B.; Saenger, W. An amylose antiparallel double helix at atomic resolution. Science 1987, 238, 205–208. [Google Scholar] [CrossRef] [PubMed]
- Boehm, O.; Zur, B.; Koch, A.; Tran, N.; Freyenhagen, R.; Harmann, M.; Zacharowski, K. Clinical chemistry reference database for Wistar rats anc C57/BL7 mice. Biol. Chem. 2007, 388, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Angelin, B.; Eriksson, M.; Rudling, M. Bile acids and lipoprotein metabolism: A renaissance for bile acids in the post-statin era? Curr. Opin. Lipidol. 1999, 10, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Hagen, E.; Istad, H.; Ose, L.; Bodd, E.; Eriksen, H.M.; Selvig, V.; Bard, J.M.; Fruchart, J.C.; Borge, M.; Wolf, M-C; et al. Fluvastatin efficacy and tolerability in comparison and in combination with cholestyramine. Eur. J. Clin. Pharmacol. 1994, 46, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Favier, M.-L.; Moundras, C.; Demigne, C.; Remesy, C. Fermentable carbohydrates exert a more potent cholesterol-lowering effect than cholestyramine. Biochim. Biophys. Acta 1995, 1258, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Leonard, T.B.; Naptun, D.A.; Popp, J.A. Serum gamma glutamyl transferase as a specific indicator of bile duct lesions in the rat liver. Am. J. Pathol. 1984, 116, 262–269. [Google Scholar] [PubMed]
- Scherer, M.; Gnewuch, C.; Schmitz, G.; Liebisch, G. Rapid quantification of bile acids and their conjugates in serum by liquid chromatography–tandem mass spectrometry. J. Chromatogr. B 2009, 877, 3920–3925. [Google Scholar] [CrossRef]
- Martin, J.D. Using XPowder: A software package for powder X-ray diffraction analysis; ISBN 84-609-1497-6; Spain. 2004. Available online: http://www.xpowder.com (accessed on 10 October 2014).
- Sample Availability: Not available.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lopez-Jaramillo, F.J.; Giron-Gonzalez, M.D.; Salto-Gonzalez, R.; Hernandez-Mateo, F.; Santoyo-Gonzalez, F. In Vitro and in Vivo Evaluation of Novel Cross-Linked Saccharide Based Polymers as Bile Acid Sequestrants. Molecules 2015, 20, 3716-3729. https://doi.org/10.3390/molecules20033716
Lopez-Jaramillo FJ, Giron-Gonzalez MD, Salto-Gonzalez R, Hernandez-Mateo F, Santoyo-Gonzalez F. In Vitro and in Vivo Evaluation of Novel Cross-Linked Saccharide Based Polymers as Bile Acid Sequestrants. Molecules. 2015; 20(3):3716-3729. https://doi.org/10.3390/molecules20033716
Chicago/Turabian StyleLopez-Jaramillo, Francisco Javier, Maria Dolores Giron-Gonzalez, Rafael Salto-Gonzalez, Fernando Hernandez-Mateo, and Francisco Santoyo-Gonzalez. 2015. "In Vitro and in Vivo Evaluation of Novel Cross-Linked Saccharide Based Polymers as Bile Acid Sequestrants" Molecules 20, no. 3: 3716-3729. https://doi.org/10.3390/molecules20033716
APA StyleLopez-Jaramillo, F. J., Giron-Gonzalez, M. D., Salto-Gonzalez, R., Hernandez-Mateo, F., & Santoyo-Gonzalez, F. (2015). In Vitro and in Vivo Evaluation of Novel Cross-Linked Saccharide Based Polymers as Bile Acid Sequestrants. Molecules, 20(3), 3716-3729. https://doi.org/10.3390/molecules20033716