Insecticidal Activity of Plant Lectins and Potential Application in Crop Protection
Abstract
:1. Introduction
2. Structural Characteristics and Classification of Plant Lectins
3. Constitutively Expressed and Inducible Lectins
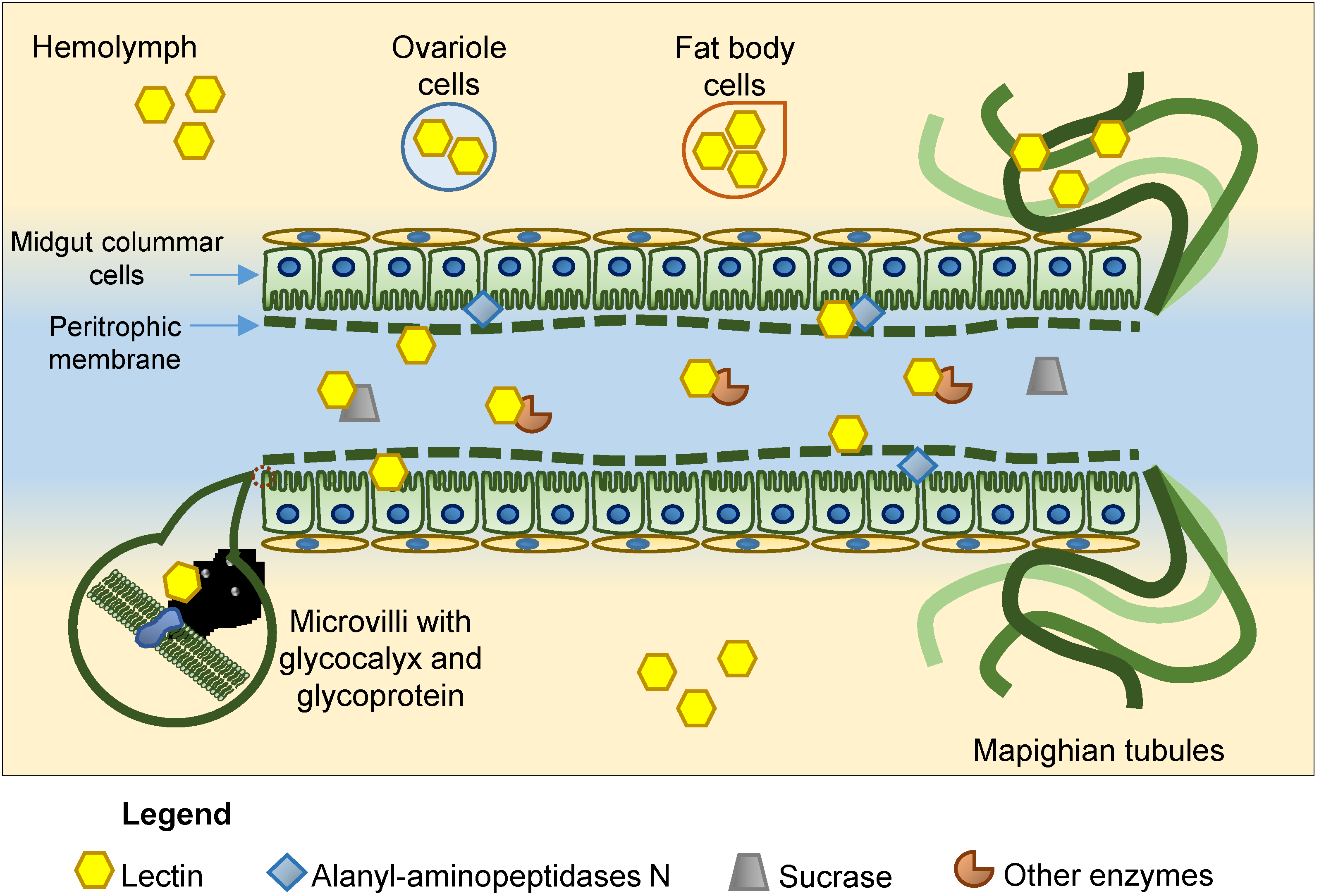
4. Anti-Insect Activity and Mode of Action of Plant Lectins
5. Biotechnological Applications
6. Tri-Trophic Interaction and Safety Assessment of Lectin-Expressing Plants
7. Conclusions and Future Directions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Stillmark, H. Uber Ricin, Eines Giftiges Ferment aus den Samen von Ricinus communis L. und Anderson Euphorbiacen. Inaugural Disseration, University of Dorpat, Dorpat, Estonia, 1888. [Google Scholar]
- Sumner, J.B. The globulins of the jack bean, Canavalia ensiformis. J. Biol. Chem. 1919, 37, 137–142. [Google Scholar]
- Sumner, J.B.; Howell, S.F. Identification of hemagglutinin of jack bean with concanavalin A. J. Bacteriol. 1936, 32, 227. [Google Scholar] [PubMed]
- Peumans, W.J.; van Damme, E. Lectins as plant defense proteins. Plant Physiol. 1995, 109, 347. [Google Scholar] [CrossRef] [PubMed]
- Van Damme, E.J.; Lannoo, N.; Fouquaert, E.; Peumans, W.J. The identification of inducible cytoplasmic/nuclear carbohydrate-binding proteins urges to develop novel concepts about the role of plant lectins. Glycoconj. J. 2003, 20, 449–460. [Google Scholar] [CrossRef]
- Van Damme, E.J.M.; Peumans, W.J.; Barre, A.; Rouge, P. Plant lectins: A composite of several distinct families of structurally and evolutionary related proteins with diverse biological roles. Crit. Rev. Plant Sci. 1998, 17, 575–692. [Google Scholar] [CrossRef]
- Van Damme, E.J.M.; Lannoo, N.; Peumans, W.J. Chapter 3 Plant Lectins. In Advances in Botanical Research; Jean-Claude, K., Michel, D., Eds.; Academic Press: Waltham, MA, USA, 2008; Volume 48, pp. 107–209. [Google Scholar]
- Gaidamashvili, M.; Ohizumi, Y.; Iijima, S.; Takayama, T.; Ogawa, T.; Muramoto, K. Characterization of the yam tuber storage proteins from Dioscorea batatas exhibiting unique lectin activities. J. Biol. Chem. 2004, 279, 26028–26035. [Google Scholar] [CrossRef] [PubMed]
- Sharon, N.; Lis, H. Legume lectins—A large family of homologous proteins. FASEB J. 1990, 4, 3198–3208. [Google Scholar] [PubMed]
- Young, N.M.; Oomen, R.P. Analysis of sequence variation among legume lectins: A ring of hypervariable residues forms the perimeter of the carbohydrate-binding site. J. Mol. Biol. 1992, 228, 924–934. [Google Scholar] [CrossRef] [PubMed]
- Rüdiger, H.; Gabius, H.-J. Plant lectins: Occurrence, biochemistry, functions and applications. Glycoconj. J. 2001, 18, 589–613. [Google Scholar] [CrossRef] [PubMed]
- Al Atalah, B.; Smagghe, G.; van Damme, E.J.M. Orysata, a jacalin-related lectin from rice, could protect plants against biting-chewing and piercing-sucking insects. Plant Sci. 2014, 221–222, 21–28. [Google Scholar]
- Roy, A.; Gupta, S.; Hess, D.; Das, K.P.; Das, S. Binding of insecticidal lectin Colocasia esculenta tuber agglutinin (CEA) to midgut receptors of Bemisia tabaci and Lipaphis erysimi provides clues to its insecticidal potential. Proteomics 2014, 14, 1646–1659. [Google Scholar] [CrossRef] [PubMed]
- Guo, P.; Wang, Y.; Zhou, X.; Xie, Y.; Wu, H.; Gao, X. Expression of soybean lectin in transgenic tobacco results in enhanced resistance to pathogens and pests. Plant Sci. 2013, 211, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Peumans, W.J.; Barre, A.; Hao, Q.; Rougé, P.; van Damme, E.J.M. Higher plants developed structurally different motifs to recognize foreign glycans. Trends Glycosci. Glycotechnol. 2000, 12, 83–101. [Google Scholar] [CrossRef]
- Bohlool, B.; Schmidt, E. Lectins: A possible basis for specificity in the Rhizobium—Legume root nodule symbiosis. Science 1974, 185, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Michiels, K.; van Damme, E.; Smagghe, G. Plant-insect interactions: What can we learn from plant lectins? Arch. Insect Biochem. Physiol. 2010, 73, 193–212. [Google Scholar] [CrossRef] [PubMed]
- Lannoo, N.; van Damme, E.J.M. Nucleocytoplasmic plant lectins. Biochim. Biophys. Acta Gen. Subj. 2010, 1800, 190–201. [Google Scholar] [CrossRef]
- Zhang, W.; Peumans, W.J.; Barre, A.; Houles Astoul, C.; Rovira, P.; Rougé, P.; Proost, P.; Truffa-Bachi, P.; Jalali, A.A.H.; van Damme, E.J.M. Isolation and characterization of a jacalin-related mannose-binding lectin from salt-stressed rice (Oryza sativa) plants. Planta 2000, 210, 970–978. [Google Scholar] [CrossRef] [PubMed]
- Claes, B.; Dekeyser, R.; Villarroel, R.; van den Bulcke, M.; Bauw, G.; van Montagu, M.; Caplan, A. Characterization of a rice gene showing organ-specific expression in response to salt stress and drought. Plant Cell 1990, 2, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Moons, A.; Prinsen, E.; Bauw, G.; van Montagu, M. Antagonistic effects of abscisic acid and jasmonates on salt-stress-inducible transcripts in rice shoots. Plant Cell 1997, 9, 2243–2259. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.; Collier, C.; Nemacheck, J.; Liang, C.; Cambron, S. A lectin-like wheat gene responds systemically to attempted feeding by avirulent first-instar hessian fly larvae. J. Chem. Ecol. 2002, 28, 1411–1428. [Google Scholar] [CrossRef] [PubMed]
- Puthoff, D.P.; Sardesai, N.; Subramanyam, S.; Nemacheck, J.A.; Williams, C.E. Hfr-2, a wheat cytolytic toxin-like gene, is up-regulated by virulent hessian fly larval feeding. Mol. Plant Pathol. 2005, 6, 411–423. [Google Scholar] [CrossRef] [PubMed]
- Subramanyam, S.; Smith, D.F.; Clemens, J.C.; Webb, M.A.; Sardesai, N.; Williams, C.E. Functional characterization of HFR1, a high-mannose N-glycan-specific wheat lectin induced by hessian fly larvae. Plant Physiol. 2008, 147, 1412–1426. [Google Scholar] [CrossRef] [PubMed]
- Giovanini, M.P.; Saltzmann, K.D.; Puthoff, D.P.; Gonzalo, M.; Ohm, H.W.; Williams, C.E. A novel wheat gene encoding a putative chitin-binding lectin is associated with resistance against hessian fly. Mol. Plant Pathol. 2007, 8, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Pyati, P.; Chellamuthu, A.; Gatehouse, A.M.R.; Fitches, E.; Gatehouse, J.A. Insecticidal activity of wheat hessian fly responsive proteins HFR-1 and HFR-3 towards a non-target wheat pest, cereal aphid (Sitobion avenae F.). J. Insect Physiol. 2012, 58, 991–999. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Peumans, W.J.; Hause, B.; Bras, J.; Kumar, M.; Proost, P.; Barre, A.; Rougé, P.; van Damme, E.J.M. Jasmonate methyl ester induces the synthesis of a cytoplasmic/nuclear chitooligosaccharide-binding lectin in tobacco leaves. FASEB J. 2002, 16, 905–907. [Google Scholar] [PubMed]
- Vandenborre, G.; van Damme, E.M.; Smagghe, G. Nicotiana tabacum agglutinin expression in response to different biotic challengers. Arthropod Plant Interact. 2009, 3, 193–202. [Google Scholar] [CrossRef]
- Walling, L.L. Avoiding effective defenses: Strategies employed by phloem-feeding insects. Plant Physiol. 2008, 146, 859–866. [Google Scholar] [CrossRef] [PubMed]
- Lam, S.; Ng, T. Lectins: Production and practical applications. Appl. Microbiol. Biotechnol. 2011, 89, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Vandenborre, G.; Smagghe, G.; van Damme, E.J.M. Plant lectins as defense proteins against phytophagous insects. Phytochemistry 2011, 72, 1538–1550. [Google Scholar] [CrossRef] [PubMed]
- Mohan Babu, R.; Sajeena, A.; Seetharaman, K.; Reddy, M.S. Advances in genetically engineered (transgenic) plants in pest management—An over view. Crop Prot. 2003, 22, 1071–1086. [Google Scholar]
- Carlini, C.R.; Grossi-de-Sá, M.F. Plant toxic proteins with insecticidal properties. A review on their potentialities as bioinsecticides. Toxicon 2002, 40, 1515–1539. [Google Scholar] [CrossRef] [PubMed]
- Vandenborre, G.; Smagghe, G.; Ghesquière, B.; Menschaert, G.; Rao, R.N.; Gevaert, K.; van Damme, E.J.M. Diversity in protein glycosylation among insect species. PLoS One 2011, 6, e16682. [Google Scholar] [CrossRef] [PubMed]
- Vandenborre, G.; van Damme, E.J.M.; Ghesquière, B.; Menschaert, G.; Hamshou, M.; Rao, R.N.; Gevaert, K.; Smagghe, G. Glycosylation signatures in Drosophila: Fishing with lectins. J. Proteome Res. 2010, 9, 3235–3242. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.F.R.; Luz, L.A.; Paiva, P.M.G.; Coelho, L.C.B.B.; Marangoni, S.; Macedo, M.L.R. Evaluation of seed coagulant Moringa oleifera lectin (cMoL) as a bioinsecticidal tool with potential for the control of insects. Process Biochem. 2011, 46, 498–504. [Google Scholar] [CrossRef]
- Felton, G.W. Indigestion is a plant’s best defense. Proc. Natl. Acad. Sci. USA 2005, 102, 18771–18772. [Google Scholar] [CrossRef] [PubMed]
- Lagarda-Diaz, I.; Guzman-Partida, A.M.; Urbano-Hernandez, G.; Ortega-Nieblas, M.M.; Robles-Burgueño, M.R.; Winzerling, J.; Vazquez-Moreno, L. Insecticidal action of PF2 lectin from Olneya tesota (Palo Fierro) against Zabrotes subfasciatus larvae and midgut glycoconjugate binding. J. Agric. Food Chem. 2008, 57, 689–694. [Google Scholar] [CrossRef]
- Vandenborre, G.; Groten, K.; Smagghe, G.; Lannoo, N.; Baldwin, I.T.; van Damme, E.J.M. Nicotiana tabacum agglutinin is active against Lepidopteran pest insects. J. Exp. Bot. 2010, 61, 1003–1014. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, V.; Uniyal, P.L.; Bhattacharya, R. Aphid resistance in Brassica crops: Challenges, biotechnological progress and emerging possibilities. Biotechnol. Adv. 2011, 29, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Goggin, F.L. Plant–aphid interactions: Molecular and ecological perspectives. Curr. Opin. Plant Biol. 2007, 10, 399–408. [Google Scholar] [CrossRef] [PubMed]
- Powell, K.S.; Spence, J.; Bharathi, M.; Gatehouse, J.A.; Gatehouse, A.M.R. Immunohistochemical and developmental studies to elucidate the mechanism of action of the snowdrop lectin on the rice brown planthopper, Nilaparvata lugens (Stal). J. Insect Physiol. 1998, 44, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Rao, K.V.; Rathore, K.S.; Hodges, T.K.; Fu, X.; Stoger, E.; Sudhakar, D.; Williams, S.; Christou, P.; Bharathi, M.; Bown, D.P.; et al. Expression of snowdrop lectin (GNA) in transgenic rice plants confers resistance to rice brown planthopper. Plant J. 1998, 15, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Bandyopadhyay, S.; Roy, A.; Das, S. Binding of garlic (Allium sativum) leaf lectin to the gut receptors of homopteran pests is correlated to its insecticidal activity. Plant Sci. 2001, 161, 1025–1033. [Google Scholar] [CrossRef]
- Dutta, I.; Saha, P.; Majumder, P.; Sarkar, A.; Chakraborti, D.; Banerjee, S.; Das, S. The efficacy of a novel insecticidal protein, Allium sativum leaf lectin (ASAL), against homopteran insects monitored in transgenic tobacco. Plant Biotechnol. J. 2005, 3, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Dutta, I.; Majumder, P.; Saha, P.; Ray, K.; Das, S. Constitutive and phloem specific expression of Allium sativum leaf agglutinin (ASAL) to engineer aphid (Lipaphis erysimi) resistance in transgenic indian mustard (Brassica juncea). Plant Sci. 2005, 169, 996–1007. [Google Scholar] [CrossRef]
- Fitches, E.; Wiles, D.; Douglas, A.E.; Hinchliffe, G.; Audsley, N.; Gatehouse, J.A. The insecticidal activity of recombinant garlic lectins towards aphids. Insect Biochem. Mol. Biol. 2008, 38, 905–915. [Google Scholar] [CrossRef] [PubMed]
- Cristofoletti, P.T.; Mendonça de Sousa, F.A.; Rahbé, Y.; Terra, W.R. Characterization of a membrane-bound aminopeptidase purified from Acyrthosiphon pisum midgut cells. FEBS J. 2006, 273, 5574–5588. [Google Scholar] [CrossRef] [PubMed]
- Napoleão, T.H.; Pontual, E.V.; Lima, T.A.; Santos, N.D.L.; Sá, R.A.; Coelho, L.C.B.B.; Navarro, D.M.A.F.; Paiva, P.M.G. Effect of Myracrodruon urundeuva leaf lectin on survival and digestive enzymes of Aedes aegypti larvae. Parasitol. Res. 2012, 110, 609–616. [Google Scholar] [CrossRef] [PubMed]
- Agra-Neto, A.C.; Napoleão, T.H.; Pontual, E.V.; Santos, N.D.; Luz Lde, A.; de Oliveira, C.M.; de Melo-Santos, M.A.; Coelho, L.C.; Navarro, D.M.; Paiva, P.M. Effect of Moringa oleifera lectins on survival and enzyme activities of Aedes aegypti larvae susceptible and resistant to organophosphate. Parasitol. Res. 2014, 113, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Macedo, M.; das Graças Machado Freire, M.; da Silva, M.; Coelho, L. Insecticidal action of Bauhinia monandra leaf lectin (BmoLL) against Anagasta kuehniella (Lepidoptera: Pyralidae), Zabrotes subfasciatus and Callosobruchus maculatus (Coleoptera: Bruchidae). Comp. Biochem. Physiol. A Comp. Physiol. 2007, 146, 486–498. [Google Scholar] [CrossRef]
- Sprawka, I.; Goławska, S.; Goławski, A.; Chrzanowski, G.; Czerniewicz, P.; Sytykiewicz, H. Entomotoxic action of jackbean lectin (Con A) in bird cherry-oat aphid through the effect on insect enzymes. J. Plant Interact. 2013, 9, 425–433. [Google Scholar] [CrossRef]
- Coelho, M.B.; Marangoni, S.; Macedo, M.L.R. Insecticidal action of Annona coriacea lectin against the flour moth Anagasta kuehniella and the rice moth Corcyra cephalonica (Lepidoptera: Pyralidae). Comp. Biochem. Physiol. Part C Toxicol. Pharmcol. 2007, 146, 406–414. [Google Scholar] [CrossRef]
- Du, J.; Foissac, X.; Carss, A.; Gatehouse, A.M.R.; Gatehouse, J.A. Ferritin acts as the most abundant binding protein for snowdrop lectin in the midgut of rice brown planthoppers (Nilaparvata lugens). Insect Biochem. Mol. Biol. 2000, 30, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Bala, A.; Roy, A.; Behura, N.; Hess, D.; Das, S. Insight to the mode of action of Allium sativum leaf agglutinin (ASAL) expressing in T3 rice lines on brown planthopper. Am. J. Plant Sci. 2013, 4, 400–407. [Google Scholar] [CrossRef]
- Bharathi, Y.; Vijaya Kumar, S.; Pasalu, I.C.; Balachandran, S.M.; Reddy, V.D.; Rao, K.V. Pyramided rice lines harbouring Allium sativum (ASAL) and Galanthus nivalis (GNA) lectin genes impart enhanced resistance against major sap-sucking pests. J. Biotechnol. 2011, 152, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Machuka, J.; van Damme, E.J.M.; Peumans, W.J.; Jackai, L.E.N. Effect of plant lectins on larval development of the legume pod borer, Maruca vitrata. Entomol. Exp. Appl. 1999, 93, 179–187. [Google Scholar] [CrossRef]
- Gatehouse, J.A.; Gatehouse, A.M.R.; Fitches, E. Effects of snowdrop lectin (GNA) delivered via artificial diet and transgenic plants on the development of tomato moth (Lacanobia oleracea) larvae in laboratory and glasshouse trials. J. Insect Physiol. 1997, 43, 727–739. [Google Scholar] [CrossRef] [PubMed]
- Sétamou, M.; Bernal, J.S.; Legaspi, J.C.; Mirkov, T.E.; Legaspi, B.C., Jr. Evaluation of lectin-expressing transgenic sugarcane against stalkborers (Lepidoptera: Pyralidae): Effects on life history parameters. J. Econ. Entomol. 2002, 95, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi, A.; Smagghe, G.; Proost, P.; van Damme, E.J.M. Ferritin acts as a target site for the snowdrop lectin (GNA) in the midgut of the cotton leafworm Spodoptera littoralis. Insect Sci. 2008, 15, 513–519. [Google Scholar] [CrossRef]
- Caccia, S.; van Damme, E.J.M.; de Vos, W.H.; Smagghe, G. Mechanism of entomotoxicity of the plant lectin from Hippeastrum hybrid (Amaryllis) in Spodoptera littoralis larvae. J. Insect Physiol. 2012, 58, 1177–1183. [Google Scholar] [CrossRef] [PubMed]
- Li, H.M.; Sun, L.; Mittapalli, O.; Muir, W.M.; Xie, J.; Wu, J.; Schemerhorn, B.J.; Sun, W.; Pittendrigh, B.R.; Murdock, L.L. Transcriptional signatures in response to wheat germ agglutinin and starvation in Drosophila melanogaster larval midgut. Insect Mol. Biol. 2009, 18, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Hegedus, D.; Erlandson, M.; Gillott, C.; Toprak, U. New insights into peritrophic matrix synthesis, architecture, and function. Annu. Rev. Entomol. 2009, 54, 285–302. [Google Scholar] [CrossRef] [PubMed]
- Harper, M.S.; Hopkins, T.L.; Czapla, T.H. Effect of wheat germ agglutinin on formation and structure of the peritrophic membrane in european corn borer (Ostrinia nubilalis) larvae. Tissue Cell 1998, 30, 166–176. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, T.L.; Harper, M.S. Lepidopteran peritrophic membranes and effects of dietary wheat germ agglutinin on their formation and structure. Arch. Insect Biochem. Physiol. 2001, 47, 100–109. [Google Scholar] [CrossRef] [PubMed]
- Ohizumi, Y.; Gaidamashvili, M.; Ohwada, S.; Matsuda, K.; Kominami, J.; Nakamura-Tsuruta, S.; Hirabayashi, J.; Naganuma, T.; Ogawa, T.; Muramoto, K. Mannose-binding lectin from yam (Dioscorea batatas) tubers with insecticidal properties against Helicoverpa armigera (Lepidoptera: Noctuidae). J. Agric. Food Chem. 2009, 57, 2896–2902. [Google Scholar] [CrossRef] [PubMed]
- Walski, T.; van Damme, E.J.M.; Smagghe, G. Penetration through the peritrophic matrix is a key to lectin toxicity against Tribolium castaneum. J. Insect Physiol. 2014, 70, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Shahidi-Noghabi, S.; Damme, E.J.M.V.; Iga, M.; Smagghe, G. Exposure of insect midgut cells to Sambucus nigra L. agglutinins I and II causes cell death via caspase-dependent apoptosis. J. Insect Physiol. 2010, 56, 1101–1107. [Google Scholar] [CrossRef] [PubMed]
- Shahidi-Noghabi, S.; van Damme, E.J.M.; Mahdian, K.; Smagghe, G. Entomotoxic action of Sambucus nigra agglutinin I in Acyrthosiphon pisum aphids and Spodoptera exigua caterpillars through caspase-3-like-dependent apoptosis. Arch. Insect Biochem. Physiol. 2010, 75, 207–220. [Google Scholar] [CrossRef] [PubMed]
- Tabashnik, B.E.; Carrière, Y.; Dennehy, T.J.; Morin, S.; Sisterson, M.S.; Roush, R.T.; Shelton, A.M.; Zhao, J.-Z. Insect resistance to transgenic Bt crops: Lessons from the laboratory and field. J. Econ. Entomol. 2003, 96, 1031–1038. [Google Scholar] [CrossRef] [PubMed]
- Van Damme, E.J.; Allen, A.K.; Peumans, W.J. Isolation and characterization of a lectin with exclusive specificity towards mannose from snowdrop (Galanthus nivalis) bulbs. FEBS Lett. 1987, 215, 140–144. [Google Scholar]
- Down, R.E.; Gatehouse, A.M.R.; Hamilton, W.D.O.; Gatehouse, J.A. Snowdrop lectin inhibits development and decreases fecundity of the glasshouse potato aphid (Aulacorthum solani) when administered in vitro and via transgenic plants both in laboratory and glasshouse trials. J. Insect Physiol. 1996, 42, 1035–1045. [Google Scholar] [CrossRef]
- Nagadhara, D.; Ramesh, S.; Pasalu, I.; Rao, Y.K.; Sarma, N.; Reddy, V.; Rao, K. Transgenic rice plants expressing the snowdrop lectin gene (GNA) exhibit high-level resistance to the whitebacked planthopper (Sogatella furcifera). Theor. Appl. Genet. 2004, 109, 1399–1405. [Google Scholar] [CrossRef] [PubMed]
- Boulter, D.; Edwards, G.A.; Gatehouse, A.M.R.; Gatehouse, J.A.; Hilder, V.A. Additive protective effects of different plant-derived insect resistance genes in transgenic tobacco plants. Crop Prot. 1990, 9, 351–354. [Google Scholar] [CrossRef]
- Zhu-Salzman, K.; Ahn, J.E.; Salzman, R.A.; Koiwa, H.; Shade, R.E.; Balfe, S. Fusion of a soybean cysteine protease inhibitor and a legume lectin enhances anti-insect activity synergistically. Agric. For. Entomol. 2003, 5, 317–323. [Google Scholar] [CrossRef]
- Zhu-Salzman, K.; Koiwa, H.; Salzman, R.; Shade, R.; Ahn, J.E. Cowpea bruchid Callosobruchus maculatus uses a three-component strategy to overcome a plant defensive cysteine protease inhibitor. Insect Mol. Biol. 2003, 12, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Lange, A.B.; Bendena, W.G.; Tobe, S.S. The effect of the thirteen Dip-allatostatins on myogenic and induced contractions of the cockroach (Diploptera punctata) hindgut. J. Insect Physiol. 1995, 41, 581–588. [Google Scholar] [CrossRef]
- Weaver, R.J.; Edwards, J.P.; Bendena, W.G.; Tobe, S.S. Structures, Functions and Occurrences of Insect Allatostatic Peptides; Seminar Series-Society for Experimental Biology; Cambridge University Press: New York, NY, USA, 1998; pp. 3–32. [Google Scholar]
- Audsley, N.; Weaver, R.J.; Edwards, J.P. In vivo effects of Manduca sexta allatostatin and allatotropin on larvae of the tomato moth, Lacanobia oleracea. Physiol. Entomol. 2001, 26, 181–188. [Google Scholar] [CrossRef]
- Fitches, E.; Audsley, N.; Gatehouse, J.A.; Edwards, J.P. Fusion proteins containing neuropeptides as novel insect contol agents: Snowdrop lectin delivers fused allatostatin to insect haemolymph following oral ingestion. Insect Biochem. Mol. Biol. 2002, 32, 1653–1661. [Google Scholar] [CrossRef] [PubMed]
- Fitches, E.C.; Bell, H.A.; Powell, M.E.; Back, E.; Sargiotti, C.; Weaver, R.J.; Gatehouse, J.A. Insecticidal activity of scorpion toxin (ButaIT) and snowdrop lectin (GNA) containing fusion proteins towards pest species of different orders. Pest Manag. Sci. 2010, 66, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Fitches, E.C.; Pyati, P.; King, G.F.; Gatehouse, J.A. Fusion to snowdrop lectin magnifies the oral activity of insecticidal ω-hexatoxin-Hv1a peptide by enabling its delivery to the central nervous system. PLoS One 2012, 7, e39389. [Google Scholar] [CrossRef] [PubMed]
- Nakasu, E.Y.T.; Williamson, S.M.; Edwards, M.G.; Fitches, E.C.; Gatehouse, J.A.; Wright, G.A.; Gatehouse, A.M.R. Novel biopesticide based on a spider venom peptide shows no adverse effects on honeybees. Proc. R. Soc. B 2014, 281. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Pyati, P.; Fitches, E.; Gatehouse, J.A. A recombinant fusion protein containing a spider toxin specific for the insect voltage-gated sodium ion channel shows oral toxicity towards insects of different orders. Insect Biochem. Mol. Biol. 2014, 47, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Tajne, S.; Boddupally, D.; Sadumpati, V.; Vudem, D.R.; Khareedu, V.R. Synthetic fusion-protein containing domains of Bt Cry1Ac and Allium sativum lectin (ASAL) conferred enhanced insecticidal activity against major lepidopteran pests. J. Biotechnol. 2014, 171, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Birch, A.N.E.; Geoghegan, I.E.; Majerus, M.E.; McNicol, J.W.; Hackett, C.A.; Gatehouse, A.M.; Gatehouse, J.A. Tri-trophic interactions involving pest aphids, predatory 2-spot ladybirds and transgenic potatoes expressing snowdrop lectin for aphid resistance. Mol. Breed. 1999, 5, 75–83. [Google Scholar] [CrossRef]
- Lawo, N.C.; Romeis, J. Assessing the utilization of a carbohydrate food source and the impact of insecticidal proteins on larvae of the green lacewing, Chrysoperla carnea. Biol. Control 2008, 44, 389–398. [Google Scholar] [CrossRef]
- Li, Y.; Romeis, J. Impact of snowdrop lectin (Galanthus nivalis agglutinin; GNA) on adults of the green lacewing, Chrysoperla carnea. J. Insect Physiol. 2009, 55, 136–143. [Google Scholar] [CrossRef]
- Thaler, J.S. Jasmonate-inducible plant defences cause increased parasitism of herbivores. Nature 1999, 399, 686–688. [Google Scholar] [CrossRef]
- Broadway, R.M.; Duffey, S.S.; Pearce, G.; Ryan, C.A. Plant proteinase inhibitors: A defense against herbivorous insects? Entomol. Exp. Appl. 1986, 41, 33–38. [Google Scholar] [CrossRef]
- Lövei, G.; Arpaia, S. The impact of transgenic plants on natural enemies: A critical review of laboratory studies. Entomol. Exp. Appl. 2005, 114, 1–14. [Google Scholar] [CrossRef]
- Poulsen, M.; Pedersen, J.W. Assessing biosafety of GM plants containing lectins. Plant Sci. Rev. 2011, 2010, 157. [Google Scholar]
- Poulsen, M.; Kroghsbo, S.; Schroder, M.; Wilcks, A.; Jacobsen, H.; Miller, A.; Frenzel, T.; Danier, J.; Rychlik, M.; Shu, Q.; et al. A 90-day safety study in wistar rats fed genetically modified rice expressing snowdrop lectin Galanthus nivalis (GNA). Food Chem. Toxicol. 2007, 45, 350–363. [Google Scholar] [CrossRef] [PubMed]
- Kroghsbo, S.; Madsen, C.; Poulsen, M.; Schrøder, M.; Kvist, P.H.; Taylor, M.; Gatehouse, A.; Shu, Q.; Knudsen, I. Immunotoxicological studies of genetically modified rice expressing PHA-E lectin or Bt toxin in wistar rats. Toxicology 2008, 245, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Gould, F.; Kennedy, G.G.; Johnson, M.T. Effects of natural enemies on the rate of herbivore adaptation to resistant host plants. Entomol. Exp. Appl. 1991, 58, 1–14. [Google Scholar] [CrossRef]
- Van Emden, H. Host plant–aphidophaga interactions. Agric. Ecosyst. Environ. 1995, 52, 3–11. [Google Scholar]
- Poppy, G.M.; Sutherland, J.P. Can biological control benefit from genetically-modified crops? Tritrophic interactions on insect-resistant transgenic plants. Physiol. Entomol. 2004, 29, 257–268. [Google Scholar] [CrossRef]
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Macedo, M.L.R.; Oliveira, C.F.R.; Oliveira, C.T. Insecticidal Activity of Plant Lectins and Potential Application in Crop Protection. Molecules 2015, 20, 2014-2033. https://doi.org/10.3390/molecules20022014
Macedo MLR, Oliveira CFR, Oliveira CT. Insecticidal Activity of Plant Lectins and Potential Application in Crop Protection. Molecules. 2015; 20(2):2014-2033. https://doi.org/10.3390/molecules20022014
Chicago/Turabian StyleMacedo, Maria Lígia R., Caio F. R. Oliveira, and Carolina T. Oliveira. 2015. "Insecticidal Activity of Plant Lectins and Potential Application in Crop Protection" Molecules 20, no. 2: 2014-2033. https://doi.org/10.3390/molecules20022014
APA StyleMacedo, M. L. R., Oliveira, C. F. R., & Oliveira, C. T. (2015). Insecticidal Activity of Plant Lectins and Potential Application in Crop Protection. Molecules, 20(2), 2014-2033. https://doi.org/10.3390/molecules20022014




