Limonin, a Component of Dictamni Radicis Cortex, Inhibits Eugenol-Induced Calcium and cAMP Levels and PKA/CREB Signaling Pathway in Non-Neuronal 3T3-L1 Cells
Abstract
:1. Introduction
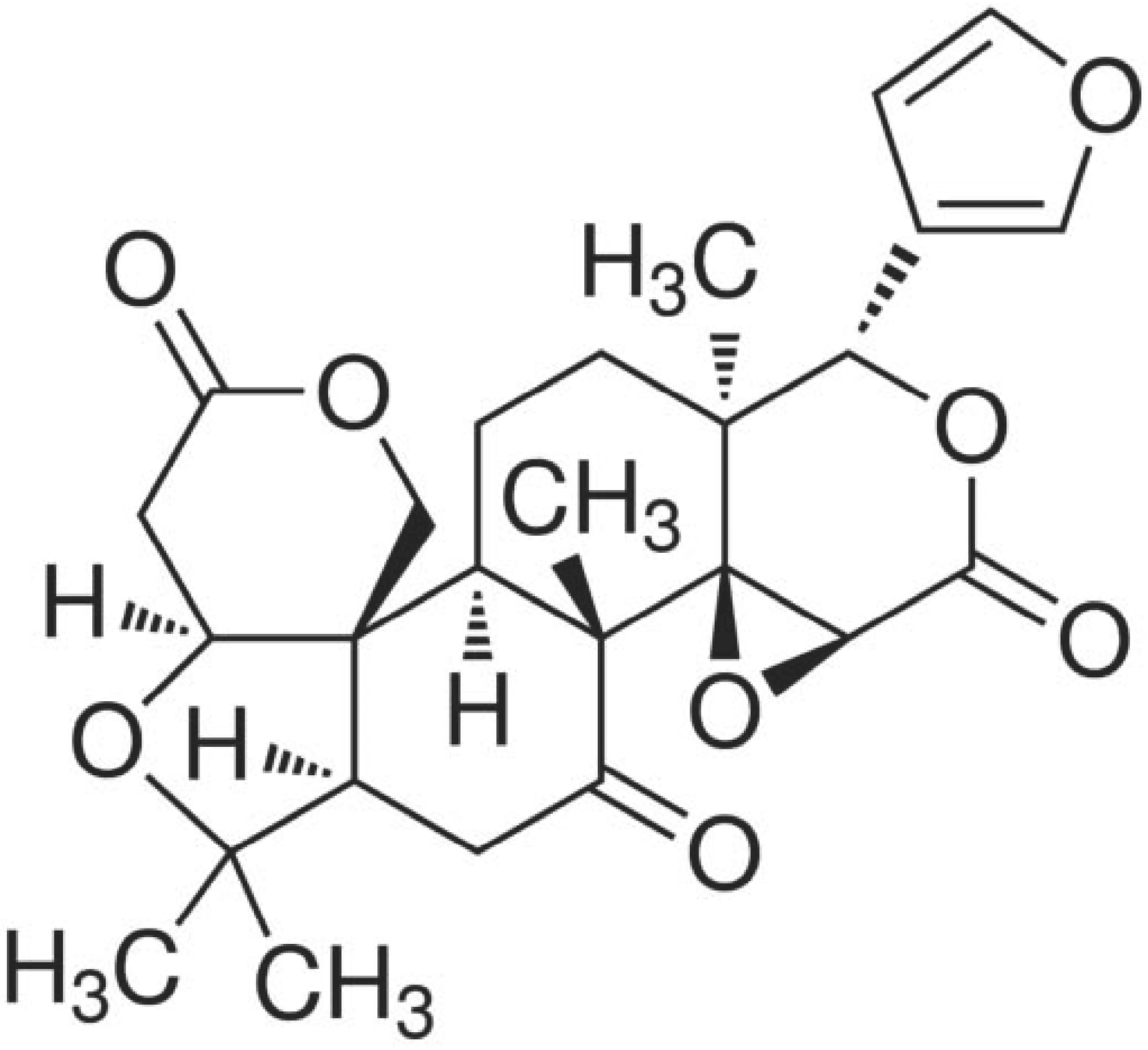
2. Results and Discussion
2.1. DRC Decreased Ca2+ and cAMP Levels in 3T3-L1 Cells
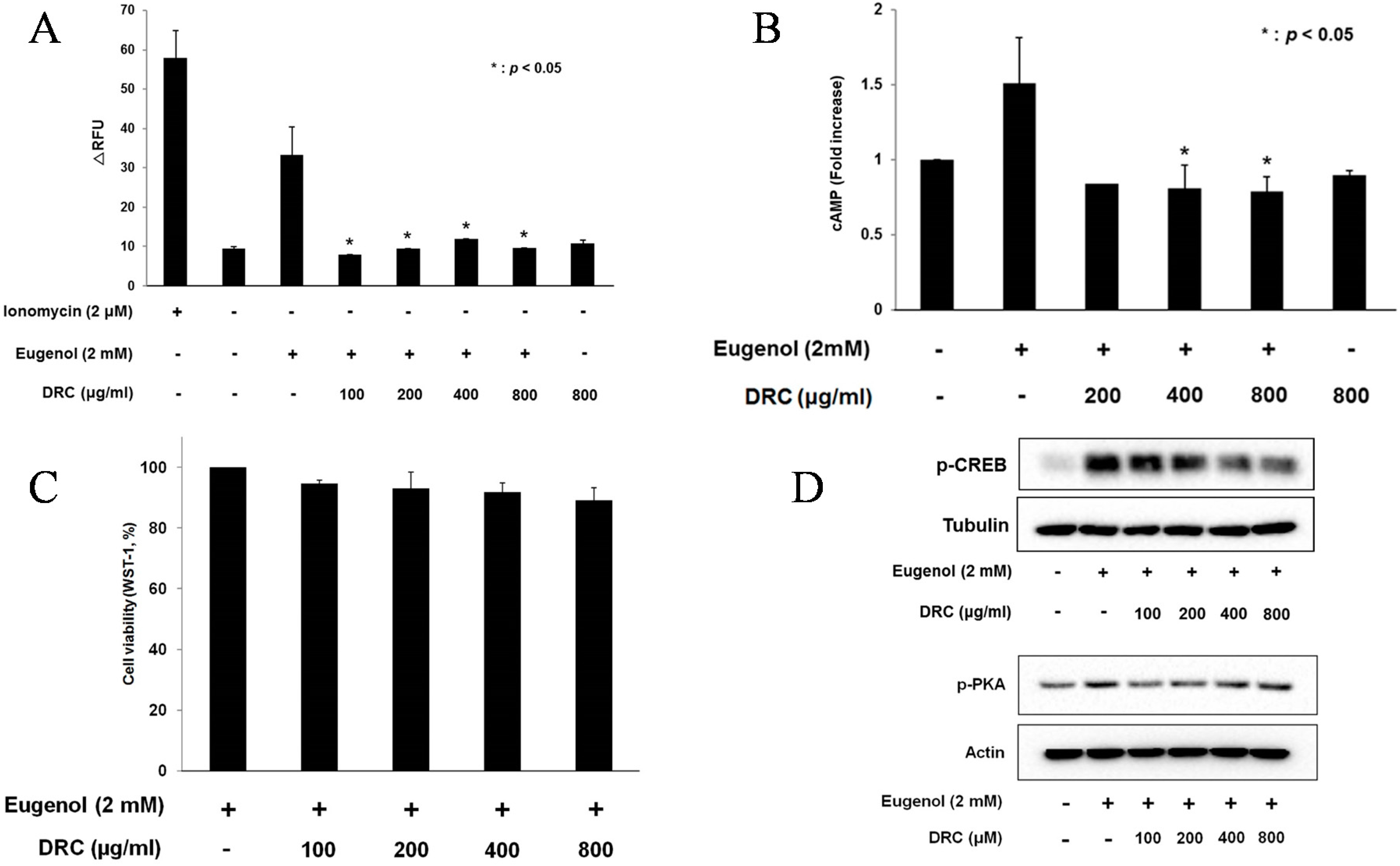
2.2. DRC Decreased the Phosphorylation of CREB in 3T3-L1 Cells
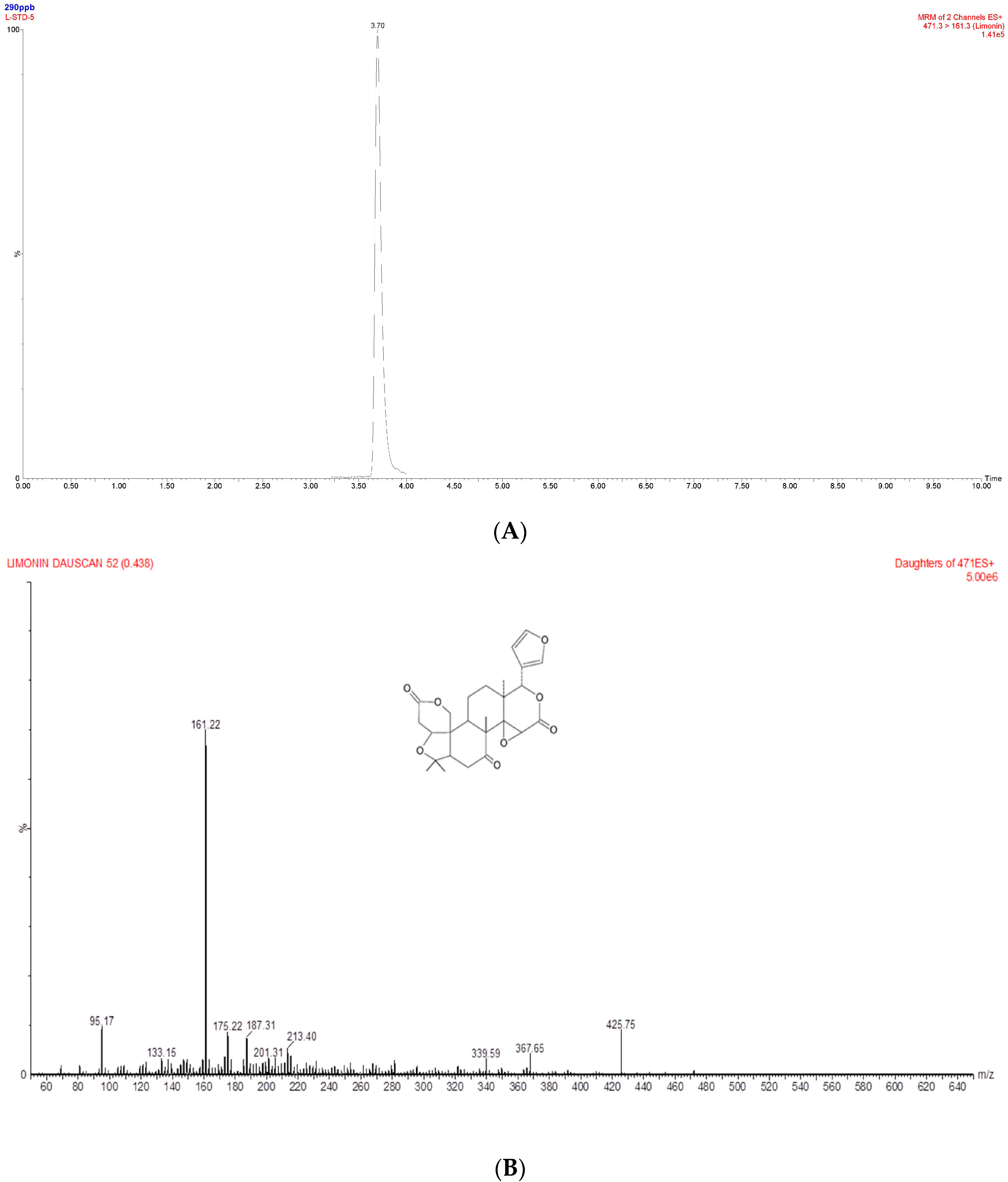
2.3. Determination of Limonin in the Ethanol Extract of DRC Using UPLC-MS/MS
2.4. Limonin Suppressed Eugenol-Induced Ca2+ and cAMP: Evels in 3T3-L1 Cells
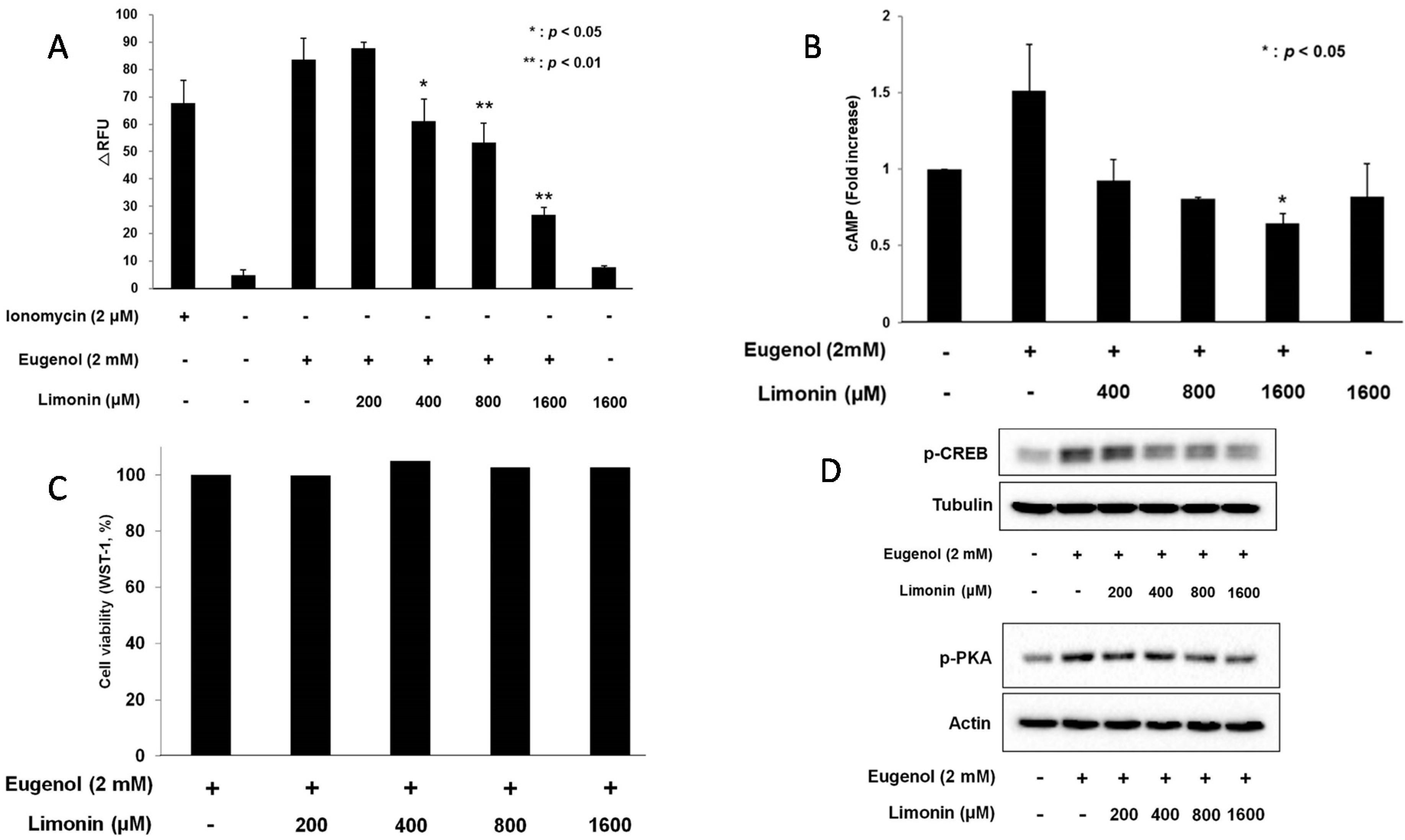
2.5. Limonin Decreased Eugenol-Induced Phosphorylation of PKA and CREB in 3T3-L1 Cells
3. Experimental Section
3.1. Plant Material
3.2. Reagents and Antibodies
3.3. Cell Culture
3.4. Ca2+ Assay
3.5. cAMP Assay
3.6. Cell Viability Analysis
3.7. Western Blot Analysis
3.8. Ultra-Performance Liquid Chromatography Tandem Mass Spectrometry (UPLC–MS/MS) Analysis
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jiang, S.; Nakano, Y.; Rahman, M.A.; Yatsuzuka, R.; Kamei, C. Effects of a Dictamnus dasycarpus T. Extract on Allergic Models in Mice. Biosci. Biotechnol. Biochem. 2008, 72, 660–665. [Google Scholar] [CrossRef] [PubMed]
- Lv, M.; Xu, P.; Tian, Y.; Liang, J.; Gao, Y.; Xu, F.; Zhang, Z.; Sung, J. Medicinal uses, phytochemistry and pharmacology of the genus Dictamnus (Rutaceae). J. Ethnopharmacol. 2015, 171, 247–263. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.B.; Qu, W.; Wang, P.; Wu, F.H.; Wang, L.Y.; Liang, J.Y. Degraded limonoids and quinoline alkaloids from Dictamnus angustifolius G. Don ex Sweet and their anti-platelet aggregation activity. Fitoterapia 2013, 90, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Geissman, T.A.; Tulagin, V. Some observations on the structure of limonin. J. Org. Chem. 1946, 11, 760–770. [Google Scholar]
- Du, C.F.; Yang, X.X.; Tu, P.F. Studies on chemical constitutents in bark of dictamnus dasycarpus. Zhongguo Zhong Yao Za Zhi 1992, 30, 1663–1666. [Google Scholar]
- Shimizu, S.; Miyamoto, S.; Fujii, G.; Nakanishi, R.; Onuma, W.; Ozaki, Y.; Fujimoto, K.; Yano, T.; Mutoh, M. Suppression of intestinal carcinogenesis in Apc-mutant mice by limonin. J. Clin. Biochem. Nutr. 2015, 57, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Fan, Y.Y.; Smith, R.; Patil, B.; Jayaprakasha, G.K.; McMurray, D.N.; Chapkin, R.S. Dietary curcumin and limonin suppress CD4+ T-cell proliferation and interleukin-2 production in mice. J. Nutr. 2009, 139, 1042–1048. [Google Scholar] [CrossRef] [PubMed]
- Ono, E.; Inoue, J.; Hashidume, T.; Shimizu, M.; Sato, R. Anti-obesity and anti-hyperglycemic effects of the dietary citrus limonoid nomilin in mice fed a high-fat diet. Biochem. Biophys. Res. Commun. 2011, 410, 677–681. [Google Scholar] [CrossRef] [PubMed]
- Halder, D.; Das, N.D.; Jung, K.H.; Choi, M.R.; Kim, M.S.; Lee, S.R.; Chai, Y.G. Cyclodextrin-clathrated limonin suppresses diet-induced obesity in mice. J. Food Biochem. 2014, 38, 216–226. [Google Scholar] [CrossRef]
- Nugent, C.; Prings, J.B.; Whitehead, J.P.; Savage, D.; Wentworth, J.M.; Chatterjee, V.K.; O’Rahily, S. Potentiation of glucose uptake in 3T3-L1 adipocytes by PPAR gamma agonists in maintained in cells expressing a PPAR gamma dominat-negative mutant: Evidence for selectivity in the downstream responses to PPAR gamma activation. Mol. Endocrinol 2001, 15, 1729–1738. [Google Scholar] [PubMed]
- Rayalam, S.; Yang, J.Y.; Ambati, S.; Della-Fera, M.A.; Baile, C.A. Resveratrol induces apoptosis and inhibits adipogenesis in 3T3-L1 adipocytes. Phytother. Res. 2008, 22, 1367–1371. [Google Scholar] [CrossRef] [PubMed]
- Reichert, M.; Eick, D. Analysis of cell cycle arrest in adipocyte differentiation. Oncogene 1999, 18, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Yoon, Y.C.; Kim, S.H.; Hwang, J.T.; Sung, M.J.; Kim, M.S.; Hur, H.J.; Rhyu, M.R.; Park, J.H. Ethanol extract of Polygonatum officinale rhizome inhibits odorant-induced cAMP and calcium levels in non-chemosensory 3T3-L1 cells. J. Food Nutr. Res. 2014, 2, 776–780. [Google Scholar] [CrossRef]
- Buck, L.; Axel, R. A novel multigene family may encode odorant receptors: a molecular basis for odor recognition. Cell 1991, 65, 175–187. [Google Scholar] [CrossRef]
- Restrepo, D.; Teeter, J.H.; Schild, D. Second messenger signaling in olfactory transduction. J. Neurobiol. 1996, 20, 37–48. [Google Scholar] [CrossRef]
- Sakano, H. Neural map formation in the mouse olfactory system. Neuron 2010, 67, 530–542. [Google Scholar] [CrossRef] [PubMed]
- Kang, N.; Koo, J. Olfactory receptors in non-chemosensory tissues. BMB Rep. 2012, 45, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Spehr, M.; Gisselmann, G.; Poplawski, A.; Riffell, J.A.; Wetzel, C.H.; Zimmer, R.K.; Haat, H. Identification of a testicular odorant receptor mediating human sperm chemotaxis. Science 2003, 299, 2054–2058. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, N.; Yomogida, K.; Okabe, M.; Touhara, K. Functional characterizatioin of a mouse testicular olfactory receptor and its role in chemosensing and in regulation of sperm motility. J. Cell Sci. 2004, 117, 5835–5845. [Google Scholar]
- Pluznick, J.L.; Zou, D.; Zhang, X.; Yan, Q.; Rodriguez-Gil, D.; Eisner, C.; Wells, E.; Greer, C.A.; Wang, T.; Firestein, S.; Schnermann, J.; Caplan, M.J. Functional expression of the olfactory signaling system in the kidney. Proc. Natl. Acad. Sci. USA 2009, 106, 2059–2064. [Google Scholar] [CrossRef] [PubMed]
- Griffin, C.A.; Kafadar, K.A.; Pavlath, G.K. MOR23 promotes muscle regeneration and regulates cell adhesion and migration. Dev. Cell 2009, 17, 649–661. [Google Scholar] [CrossRef] [PubMed]
- Busse, D.; Kudella, P.; Gruning, N.M.; Gisselmann, G.; Stander, S.; Luger, T.; Jacobsen, F.; Seinstraber, L.; Paus, R.; Gkogkolou, P.; et al. A synthetic sandalwood odorant induces would-healing process in human keratinocytes via the olfactory receptor OR2AT4. J. Investig. Dermatol. 2014, 134, 2823–2832. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Yoon, Y.C.; Lee, A.S.; Kang, N.; Koo, J.; Rhyu, M.R.; Park, J.H. Expression of human olfactory receptor 10J5 in heart aorta, coronary artery, and endothelial cells and its functional role in angiogenesis. Biochem. Biophys. Res. Commun. 2015, 460, 404–408. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.D.; Ford, D.A. Activation of myocardial cAMP-dependent protein kinase by lysoplasmenylcholine. FEBS Lett. 1997, 420, 33–38. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoon, Y.C.; Kim, S.-H.; Kim, M.J.; Yang, H.J.; Rhyu, M.-R.; Park, J.-H. Limonin, a Component of Dictamni Radicis Cortex, Inhibits Eugenol-Induced Calcium and cAMP Levels and PKA/CREB Signaling Pathway in Non-Neuronal 3T3-L1 Cells. Molecules 2015, 20, 22128-22136. https://doi.org/10.3390/molecules201219840
Yoon YC, Kim S-H, Kim MJ, Yang HJ, Rhyu M-R, Park J-H. Limonin, a Component of Dictamni Radicis Cortex, Inhibits Eugenol-Induced Calcium and cAMP Levels and PKA/CREB Signaling Pathway in Non-Neuronal 3T3-L1 Cells. Molecules. 2015; 20(12):22128-22136. https://doi.org/10.3390/molecules201219840
Chicago/Turabian StyleYoon, Yeo Cho, Sung-Hee Kim, Min Jung Kim, Hye Jeong Yang, Mee-Ra Rhyu, and Jae-Ho Park. 2015. "Limonin, a Component of Dictamni Radicis Cortex, Inhibits Eugenol-Induced Calcium and cAMP Levels and PKA/CREB Signaling Pathway in Non-Neuronal 3T3-L1 Cells" Molecules 20, no. 12: 22128-22136. https://doi.org/10.3390/molecules201219840
APA StyleYoon, Y. C., Kim, S.-H., Kim, M. J., Yang, H. J., Rhyu, M.-R., & Park, J.-H. (2015). Limonin, a Component of Dictamni Radicis Cortex, Inhibits Eugenol-Induced Calcium and cAMP Levels and PKA/CREB Signaling Pathway in Non-Neuronal 3T3-L1 Cells. Molecules, 20(12), 22128-22136. https://doi.org/10.3390/molecules201219840





