Efficient Diethylzinc/Gallic Acid and Diethylzinc/Gallic Acid Ester Catalytic Systems for the Ring-Opening Polymerization of rac-Lactide
Abstract
:1. Introduction
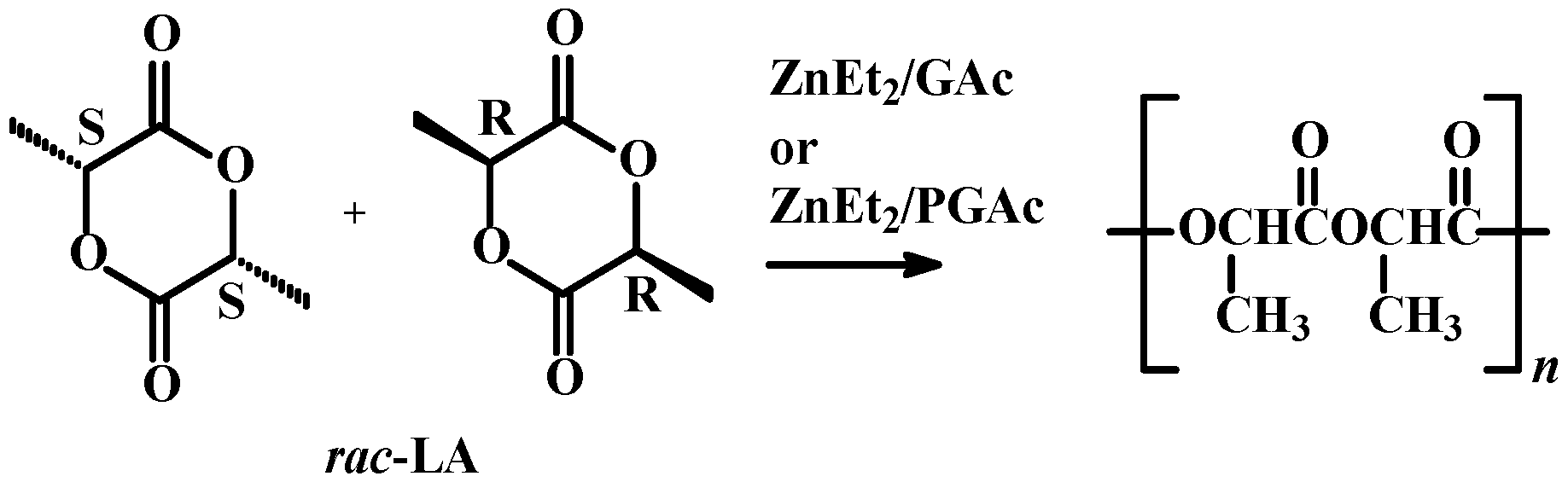
2. Results and Discussion
| Entry | Molar Ratio [Zn]/[rac-LA]0 | Temp. (°C) | Time (h) | Yield a (%) | Conv. b (%) | Mn c (Da) | PD c | MC d (%) | Mv e (Da) | Mn f (Da) | p2 | Li | T |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PLA 1 | 1/50 | 40 | 16 | 36 | 39 | 2500 | 1.26 | 3 | 2800 | 2700 | 0.70 | 2.86 | 0 |
| PLA 2 | 1/50 | 40 | 48 | 44 | 48 | 3200 | 1.48 | 9 | 3400 | 2900 | - | - | 0.22 |
| PLA 3 | 1/50 | 60 | 16 | 35 | 40 | 2000 | 1.18 | 3 | 2400 | 2100 | - | - | 0.19 |
| PLA 4 | 1/50 | 60 | 24 | 43 | 47 | 3000 | 1.54 | 10 | 3300 | 3200 | - | - | 0.22 |
| PLA 5 | 1/50 | 60 | 48 | 48 | 52 | 3300 | 2.71 | 22 | 3400 | 3100 | - | - | 0.57 |
| PLA 6 | 1/50 | 80 | 48 | 58 | 64 | 4000 | 3.39 | 33 | 4000 | 3700 | - | - | 0.85 |
| PLA 7 | 1/100 | 40 | 16 | 28 | 32 | 2100 | 1.49 | 6 | 2500 | 2300 | - | - | 0.08 |
| PLA 8 | 1/100 | 40 | 48 | 39 | 43 | 5400 | 1.56 | 7 | 5700 | 5200 | - | - | 0.14 |
| PLA 9 | 1/100 | 60 | 24 | 38 | 43 | 5600 | 1.63 | 9 | 5800 | 5300 | - | - | 0.17 |
| PLA 10 | 1/100 | 60 | 48 | 43 | 48 | 6000 | 2.49 | 18 | 6300 | 6100 | - | - | 0.47 |
| PLA 11 | 1/100 | 80 | 6 | 37 | 41 | 5200 | 2.32 | 6 | 5500 | 5000 | - | - | 0.36 |
| PLA 12 | 1/100 | 80 | 16 | 44 | 47 | 5900 | 2.48 | 17 | 6200 | 5800 | - | - | 0.41 |
| PLA 13 | 1/100 | 80 | 24 | 47 | 52 | 6600 | 2.67 | 30 | 6600 | 6200 | - | - | 0.49 |
| PLA 14 | 1/100 | 80 | 48 | 52 | 57 | 6800 | 3.21 | 39 | 7100 | 6400 | - | - | 0.76 |
| Entry | Molar Ratio [Zn]0/[rac-LA]0 | Medium | Temp. (°C) | Time (h) | Yield a (%) | Conv. b (%) | Mn c (Da) | PD c | MC d (%) | Mv e (Da) | Mn f (Da) | p2 | Li | T |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PLA 15 | 1/50 | THF | 40 | 16 | 23 | 26 | 1600 | 1.29 | 6 | 1800 | 1300 | 0.63 | 3.17 | 0 |
| PLA 16 | 1/50 | THF | 40 | 48 | 37 | 41 | 2500 | 2.25 | 13 | 2700 | 2100 | - | - | 0.33 |
| PLA 17 | 1/50 | THF | 60 | 48 | 43 | 47 | 2900 | 3.08 | 29 | 3200 | 2600 | - | - | 0.64 |
| PLA 18 | 1/100 | THF | 40 | 48 | 32 | 36 | 4500 | 2.37 | 11 | 4700 | 4300 | - | - | 0.26 |
| PLA 19 | 1/100 | THF | 60 | 48 | 35 | 38 | 4700 | 2.91 | 24 | 4900 | 3800 | - | - | 0.59 |
| PLA 20 | 1/50 | CH2Cl2 | 40 | 24 | traces | traces | - | - | - | - | - | - | - | - |
| PLA 21 | 1/50 | CH2Cl2 | 40 | 48 | 21 | 23 | 1300 | 2.86 | 17 | 1700 | 1200 | - | - | 0.42 |
| PLA 22 | 1/100 | CH2Cl2 | 40 | 48 | 16 | 17 | 2100 | 2.32 | 14 | 2400 | 2000 | - | - | 0.37 |
| Entry | Molar Ratio [Zn]0/[rac-LA]0 | Temp. (°C) | Time (h) | Yield a (%) | Conv. b (%) | Mn c (Da) | PD c | MC d (%) | Mv e [Da] | Mn f [Da] | p2 | Li | T |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PLA 23 | 1/50 | 40 | 16 | 39 | 43 | 2700 | 1.19 | 2 | 3100 | 2900 | 0.92 | 2.17 | 0 |
| PLA 24 | 1/50 | 40 | 48 | 61 | 69 | 4500 | 1.42 | 11 | 4800 | 4200 | - | - | 0.13 |
| PLA 25 | 1/50 | 60 | 16 | 53 | 58 | 3600 | 1.28 | 3 | 4100 | 3900 | 0.58 | 3.38 | 0 |
| PLA 26 | 1/50 | 60 | 24 | 59 | 65 | 4100 | 1.38 | 7 | 4400 | 4300 | - | - | 0.05 |
| PLA 27 | 1/50 | 60 | 48 | 68 | 74 | 4600 | 2.36 | 13 | 4900 | 4400 | - | - | 0.46 |
| PLA 28 | 1/50 | 80 | 48 | 83 | 91 | 5700 | 3.04 | 31 | 5900 | 5200 | - | - | 0.74 |
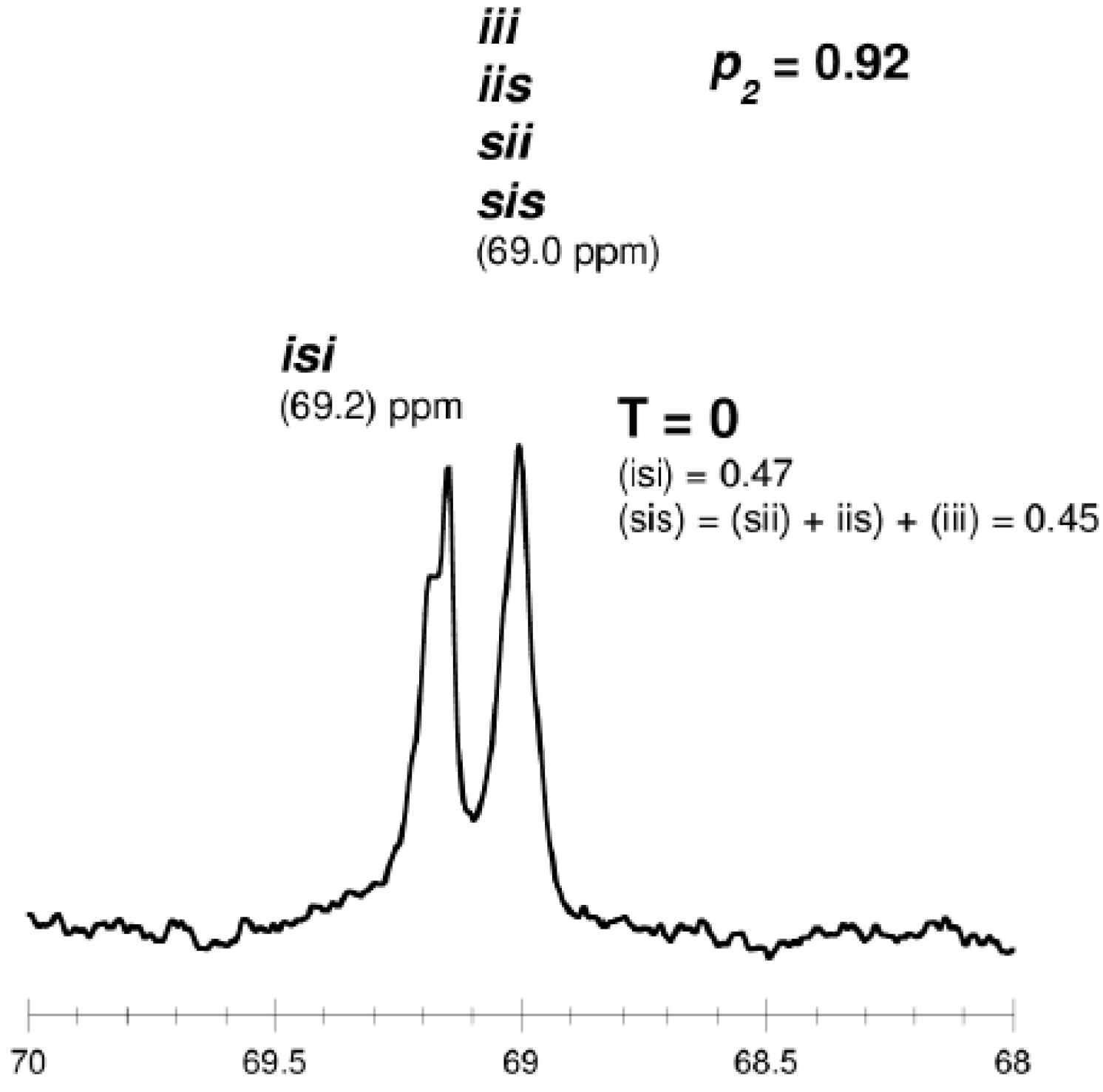
| PLA 29 | 1/100 | 40 | 16 | 35 | 39 | 4800 | 1.18 | 3 | 5300 | 5200 | 0.90 | 2.22 | 0 |
| PLA 30 | 1/100 | 40 | 48 | 54 | 61 | 7700 | 1.27 | 5 | 8100 | 7400 | 0.72 | 2.77 | 0 |
| PLA 31 | 1/100 | 60 | 16 | 42 | 46 | 5700 | 1.32 | 6 | 6200 | 5800 | 0.60 | 3.33 | 0 |
| PLA 32 | 1/100 | 60 | 24 | 56 | 62 | 7900 | 1.31 | 8 | 8300 | 7500 | 0.61 | 3.28 | 0 |
| PLA 33 | 1/100 | 60 | 48 | 63 | 68 | 8700 | 1.89 | 16 | 8900 | 8500 | - | - | 0.38 |
| PLA 34 | 1/100 | 80 | 6 | 54 | 59 | 7600 | 1.39 | 4 | 7800 | 7200 | - | - | 0.16 |
| PLA 35 | 1/100 | 80 | 16 | 62 | 68 | 8600 | 1.48 | 16 | 9000 | 8300 | - | - | 0.27 |
| PLA 36 | 1/100 | 80 | 24 | 67 | 73 | 9300 | 2.06 | 25 | 9500 | 9100 | - | - | 0.39 |
| PLA 37 | 1/100 | 80 | 48 | 75 | 82 | 9900 | 2.47 | 37 | 10,300 | 9400 | - | - | 0.59 |
| Entry | Molar ratio [Zn]0/[rac-LA]0 | Medium | Temp. (°C) | Time (h) | Yield a (%) | Conv. b (%) | Mn c (Da) | PD c | MC d (%) | Mv e [Da] | Mn f [Da] | p2 | Li | T |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PLA 38 | 1/50 | THF | 40 | 16 | 29 | 31 | 2000 | 1.35 | 7 | 2300 | 1800 | 0.71 | 2.82 | 0 |
| PLA 39 | 1/50 | THF | 40 | 48 | 40 | 46 | 2900 | 1.69 | 18 | 3300 | 2600 | - | - | 0.19 |
| PLA 40 | 1/50 | THF | 60 | 48 | 47 | 51 | 3300 | 2.61 | 22 | 3700 | 3100 | - | - | 0.49 |
| PLA 41 | 1/100 | THF | 40 | 48 | 38 | 41 | 5100 | 1.82 | 12 | 5700 | 4700 | - | - | 0.07 |
| PLA 42 | 1/100 | THF | 60 | 48 | 39 | 44 | 5500 | 2.39 | 26 | 5600 | 4900 | - | - | 0.44 |
| PLA 43 | 1/50 | CH2Cl2 | 40 | 16 | 15 | 17 | 1200 | 1.74 | 18 | 1500 | 1100 | - | - | 0.27 |
| PLA 44 | 1/50 | CH2Cl2 | 40 | 48 | 36 | 39 | 2400 | 2.89 | 29 | 2600 | 2100 | - | - | 0.53 |
| PLA 45 | 1/100 | CH2Cl2 | 40 | 48 | 32 | 34 | 4200 | 1.92 | 23 | 4700 | 3800 | - | - | 0.38 |
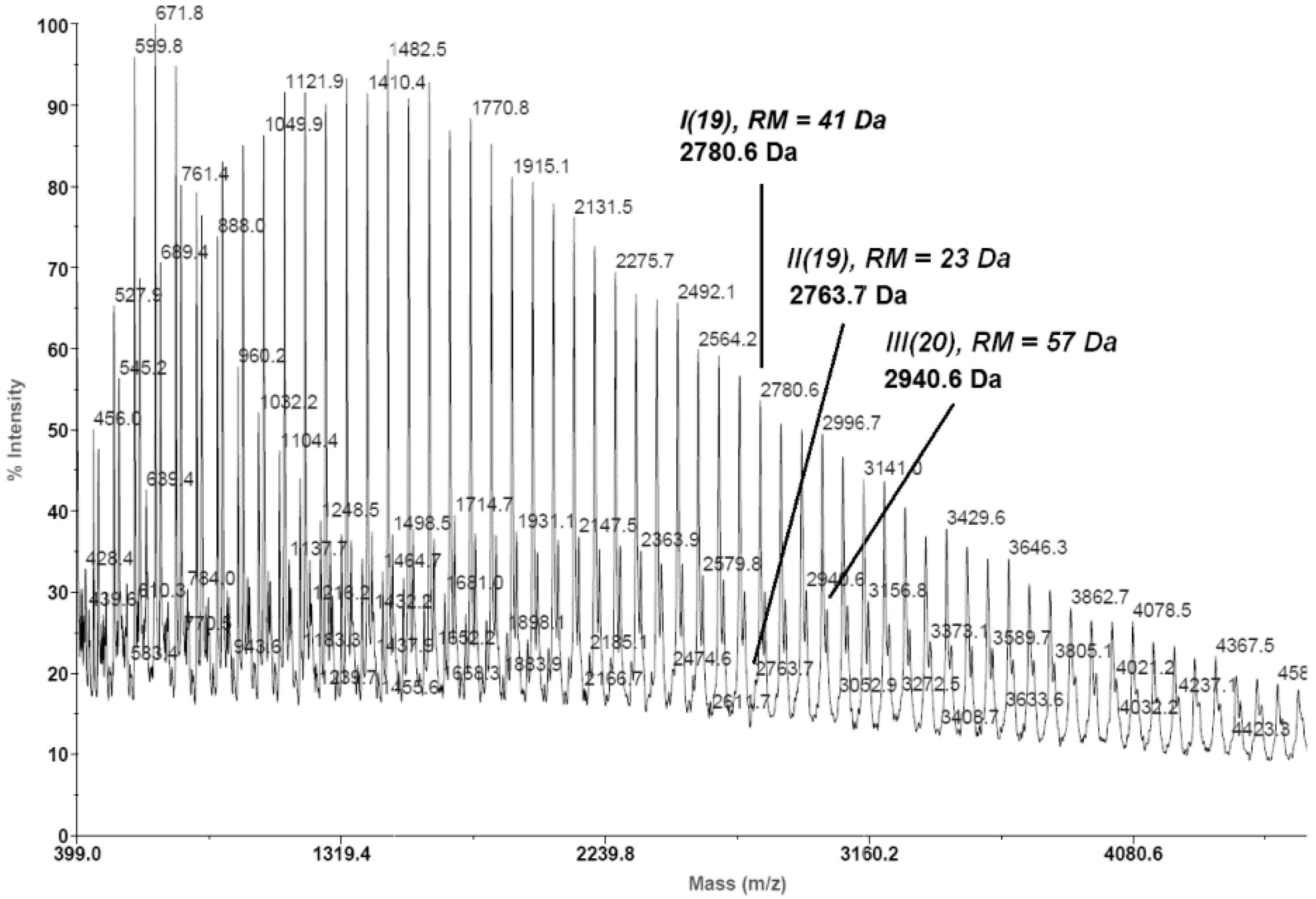
- -
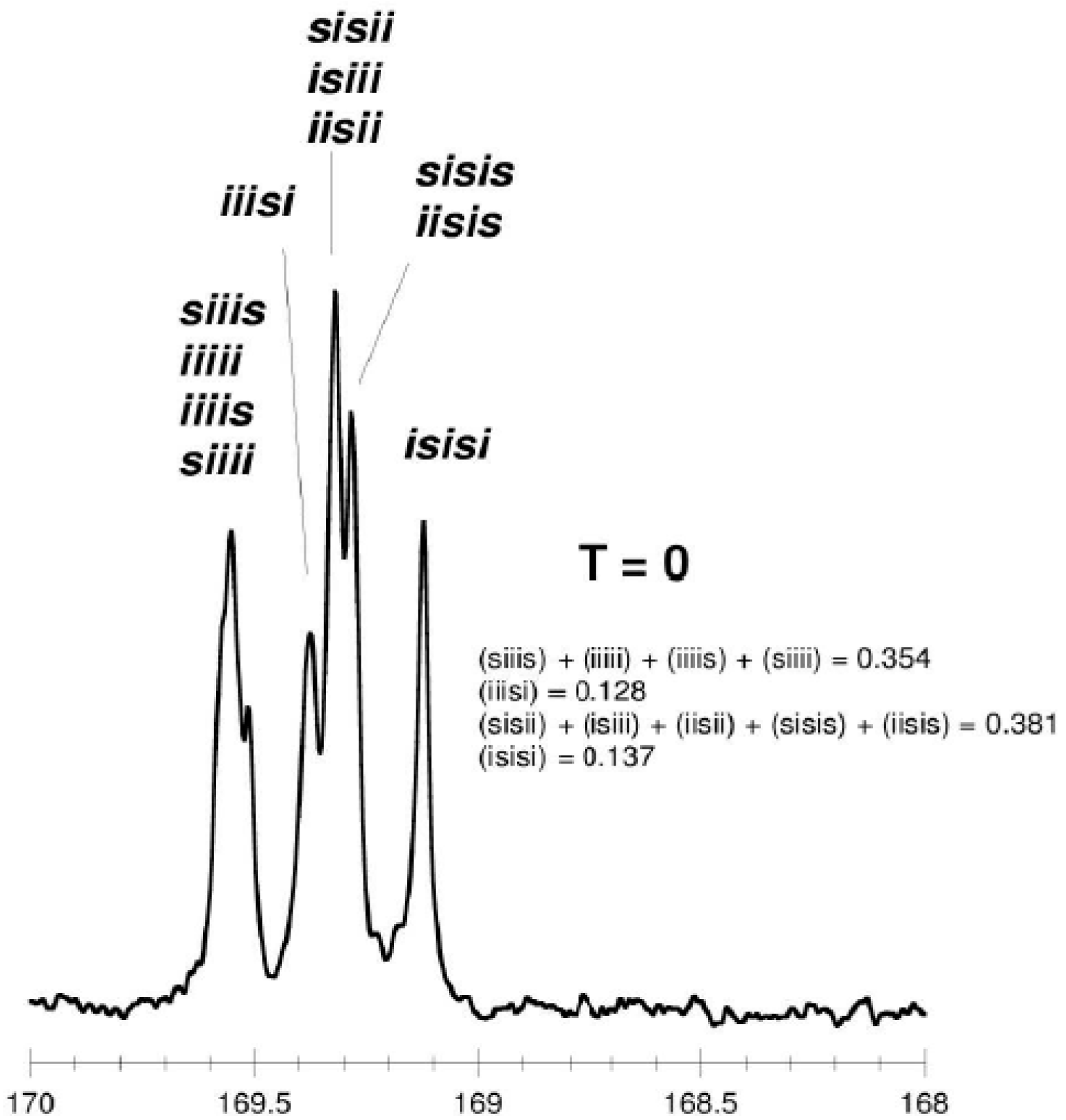
- for tetrads
- -
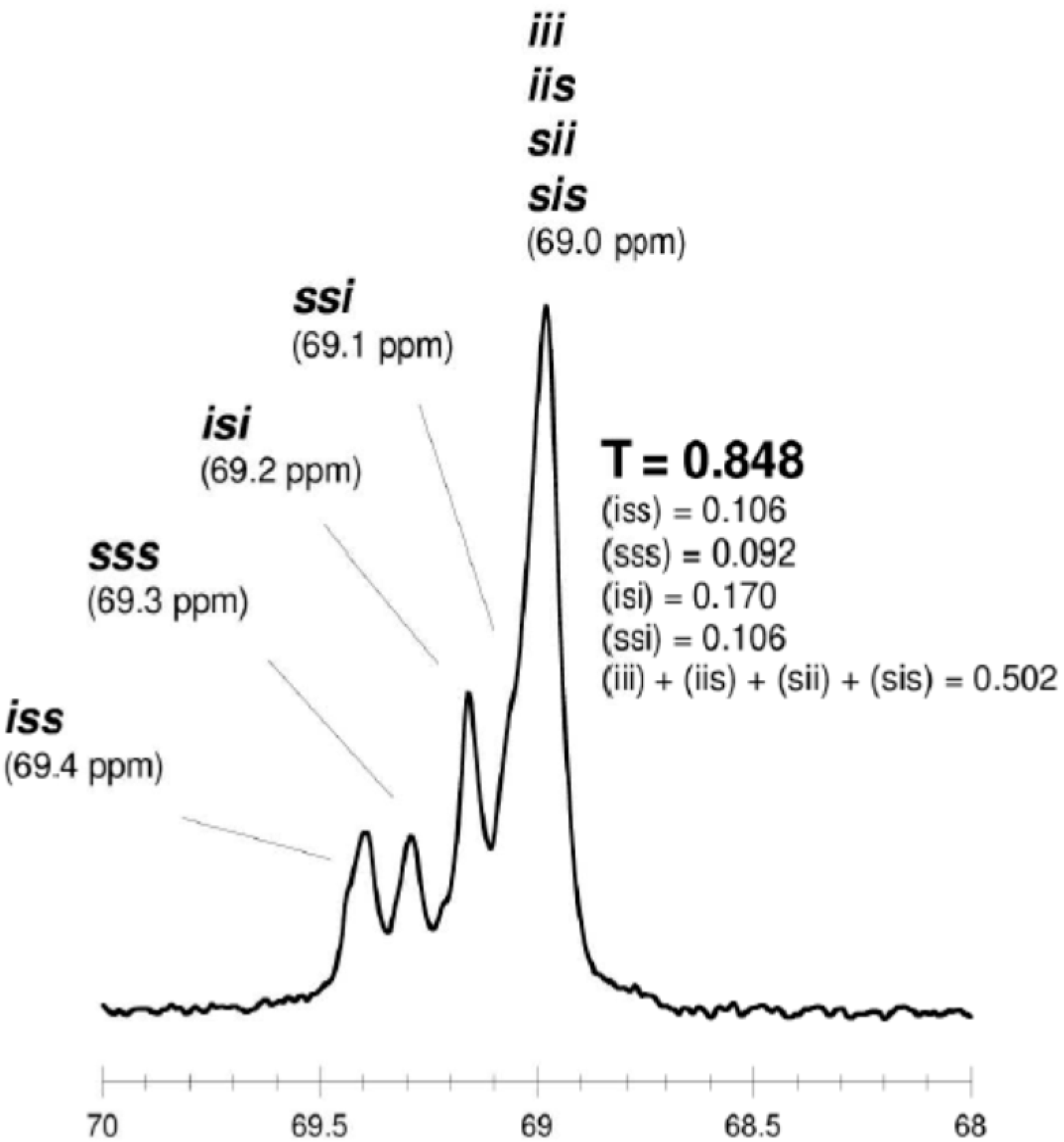
- for hexads
isotactic PLA (T = 0, p1 = 1, p2 = 0) ...SSSSSS... + ...RRRRRR...“predominantly isotactic” PLA (T = 0, p1 = 0.5, p2 = 0.5, Li = 4) ...SSSRRRSSSSRRR...“completely disyndiotactic” (heterotactic) PLA (T = 0, p1 = 0, p2 = 1, Li = 2) ...SSRRSSRRSSRR...


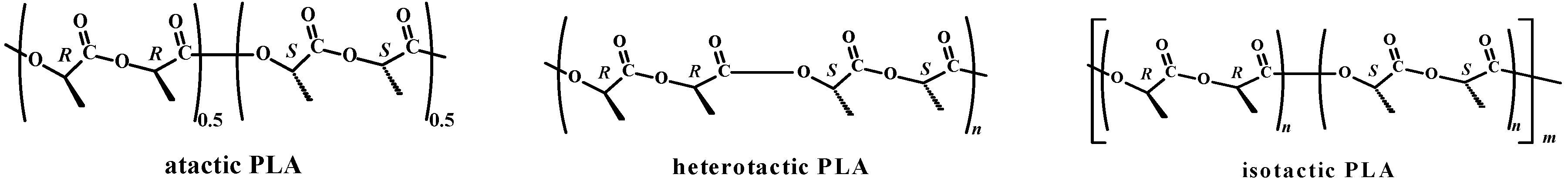
3. Experimental Section
3.1. Materials
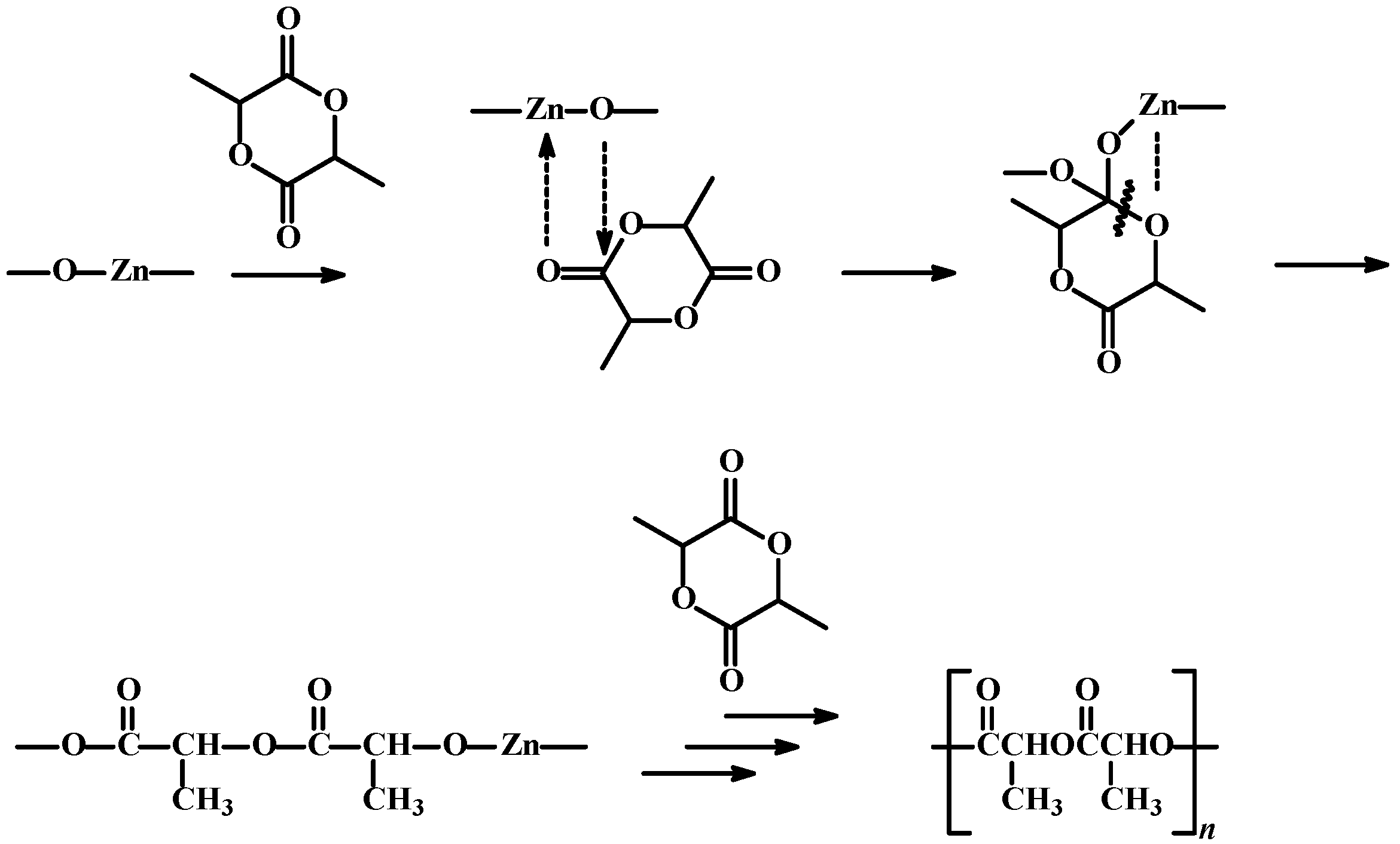
3.2. Synthesis of the Catalytic Systems
3.3. Synthesis of Polylactide
3.4. Spectroscopy Data
3.4.1. NMR Data
3.4.2. FT-IR Data
3.5. Measurements
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Platel, R.H.; Hodgson, L.M.; Williams, C.K. Biocompatible initiators for lactide polymerization. Polym. Rev. 2008, 48, 11–63. [Google Scholar] [CrossRef]
- Gupta, B.; Revagade, N.; Hilborn, J. Poly(lactic acid) fiber: An overview. Prog. Polym. Sci. 2007, 34, 455–482. [Google Scholar] [CrossRef]
- Xiao, L.; Wang, B.; Yang, G.; Gauthier, M. Poly(lactic acid)-based biomaterials: Synthesis, modification and applications. In Biomedical Science, Engineering and Technology; Ghista, D.N., Ed.; InTech Europe: Rijeka, Croatia, 2012; pp. 247–282. [Google Scholar]
- Lasprilla, A.J.R.; Martinez, G.A.R.; Lunelli, B.H.; Jardini, A.L.; Filho, R.M. Poly-lactic acid synthesis for application in biomedical devices—A review. Biotechnol. Adv. 2012, 30, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Olędzka, E.; Horeglad, P.; Gruszczyńska, Z.; Plichta, A.; Nałęcz-Jawecki, G.; Sobczak, M. Polylactide conjugates of camptothecin with different drug release abilities. Molecules 2014, 19, 19460–19470. [Google Scholar] [CrossRef] [PubMed]
- Sobczak, M.; Korzeniowska, A.; Goś, P.; Kołodziejski, W.L. Preparation and characterization of polyester- and poly(ester-carbonate)-paclitaxel conjugates. Eur. J. Med. Chem. 2011, 46, 3047–3051. [Google Scholar] [CrossRef] [PubMed]
- Sobczak, M.; Hajdaniak, M.; Goś, P.; Olędzka, E.; Kołodziejski, W.L. Use of aliphatic poly(amide urethane)s for the controlled release of 5-fluorouracil. Eur. J. Med. Chem. 2011, 46, 914–918. [Google Scholar] [CrossRef] [PubMed]
- Sobczak, M.; Nałęcz-Jawecki, G.; Kołodziejski, W.L.; Goś, P.; Żółtowska, K. Synthesis and study of controlled release of ofloxacin from polyester conjugates. Int. J. Pharm. 2010, 402, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Kricheldorf, H.R.; Berl, M.; Scharnagl, N. Poly(lactones). 9. Polymerization mechanism of metal alkoxide initiated polymerizations of lactide and various lactones. Macromolecules 1988, 21, 286–293. [Google Scholar] [CrossRef]
- Kowalski, A.; Duda, A.; Penczek, S. Kinetics and mechanism of cyclic esters polymerization initiated with Tin(II) octoate. 3. Polymerization of l,l-dilactide. Macromolecules 2000, 33, 7359–7370. [Google Scholar] [CrossRef]
- Marshall, E.L.; Gibson, V.C.; Rzepa, H.S. A computational analysis of the ring-opening polymerization of rac-lactide initiated by single-site β-diketiminate metal complexes: Defining the mechanistic pathway and the origin of stereocontrol. J. Am. Chem. Soc. 2005, 127, 6048–6051. [Google Scholar] [CrossRef] [PubMed]
- Ryner, M.; Stridsberg, K.; Albertsson, A.C.; von Schenck, H.; Svensson, M. Mechanism of ring-opening polymerization of 1,5-dioxepan-2-one and l-lactide with stannous 2-ethylhexanoate. A theoretical study. Macromolecules 2001, 34, 3877–3881. [Google Scholar] [CrossRef]
- Rasal, R.M.; Janorkar, A.V.; Hirt, D.E. Poly(lactic acid) modifications. Prog. Polym. Sci. 2010, 35, 338–356. [Google Scholar] [CrossRef]
- Kasperczyk, J. NMR investigation of biodegradable polyesters for medical application. Macromol. Symp. 2001, 175, 19–31. [Google Scholar] [CrossRef]
- Coudane, J.; Ustariz-Peyret, C.; Schwach, G.; Vert, M. More about the stereodependence of dd and ll pair linkages during the ring-opening polymerization of racemic lactide. J. Polym. Sci. A Polym. Chem. 1997, 35, 1651–1658. [Google Scholar] [CrossRef]
- Spassky, N.; Simic, V.; Montaudo, M.S.; Hubert-Pfalzgraf, L.G. Inter- and intramolecular ester exchange reactions in the ring-opening polymerization of (d,l)-lactide using lanthanide alkoxide initiators. Macromol. Chem. Phys. 2000, 201, 2432–2440. [Google Scholar] [CrossRef]
- Schwach, G.; Coudane, J.; Engel, R.; Vert, M. Zn lactate as initiator of d,l-lactide ring opening polymerization comparison with Sn octoate. Polym. Bull. 1996, 37, 771–776. [Google Scholar] [CrossRef]
- Stanford, M.J.; Dove, A.P. One-pot synthesis of α,ω-chain end functional, stereoregular, star-shaped poly(lactide). Macromolecules 2009, 42, 141–147. [Google Scholar] [CrossRef]
- Kreiser-Saunders, I.; Kricheldorf, H.R. Polylactones, 39. Zn lactate-catalyzed copolymerization of l-lactide with glycolide or ɛ-caprolactone. Macromol. Chem. Phys. 1998, 199, 1081–1087. [Google Scholar]
- Kricheldorf, H.R.; Damrau, D.-O. Polylactones, 43. Polymerization of l-lactide catalyzed by zinc amino acid salts. Macromol. Chem. Phys. 1998, 199, 1747–1752. [Google Scholar] [CrossRef]
- Kricheldorf, H.R.; Damrau, D.-O. Polylactones, 37. Polymerizations of l-lactide initiated with Zn(II) l-lactate and other resorbable Zn salts. Macromol. Chem. Phys. 1997, 198, 1753–1766. [Google Scholar] [CrossRef]
- Schwach, G.; Coudane, J.; Engel, R.; Vert, M. Ring opening polymerization of d,l-lactide in the presence of zinc metal and zinc lactate. Polym. Int. 1998, 46, 177–182. [Google Scholar] [CrossRef]
- Chisholm, M.H.; Huffman, J.C.; Phomphrai, K. Monomeric metal alkoxides and trialkyl siloxides: (BDI)Mg(OtBu)(THF) and (BDI)Zn(OSiPh3)(THF). Comments on single site catalysts for ring-opening polymerization of lactides. Dalton Trans. 2001, 3, 222–224. [Google Scholar] [CrossRef]
- Chisholm, M.H.; Gallucci, J.; Phomphrai, K. Coordination chemistry and reactivity of monomeric alkoxides and amides of magnesium and zinc supported by the diiminato ligand CH(CMeNC6H3–2,6-iPr2)2. A comparative study. Inorg. Chem. 2002, 41, 2785–2794. [Google Scholar] [CrossRef] [PubMed]
- Chisholm, M.H.; Phomphrai, K. Conformational effects in β-diiminate ligated magnesium and zinc amides. Solution dynamics and lactide polymerization. Inorg. Chim. Acta 2003, 350, 121–125. [Google Scholar] [CrossRef]
- Chisholm, M.H.; Gallucci, J.C.; Phomphrai, K. Lactide polymerization by well-defined calcium coordination complexes: Comparisons with related magnesium and zinc chemistry. Chem. Commun. 2003, 1, 48–49. [Google Scholar] [CrossRef]
- Hill, M.S.; Hitchcock, P.B. Synthesis of C2 and Cs symmetric zinc complexes supported by bis(phosphinimino)methyl ligands and their use in ring opening polymerisation catalysis. Dalton Trans. 2002, 24, 4694–4702. [Google Scholar] [CrossRef]
- Dove, A.P.; Gibson, V.C.; Marshall, E.L.; White, A.J.P.; Williams, D.J. Magnesium and zinc complexes of a potentially tridentate β-diketiminate ligand. Dalton Trans. 2004, 4, 570–578. [Google Scholar] [CrossRef] [PubMed]
- Chisholm, M.H.; Eilerts, N.W.; Huffman, J.C.; Iyer, S.S.; Pacold, M.; Phomphrai, K. Molecular design of single-site metal alkoxide catalyst precursors for ring-opening polymerization reactions leading to polyoxygenates. 1. Polylactide formation by achiral and chiral magnesium and zinc alkoxides, (η3-l)MOR, where l = trispyrazolyl- and trisindazolylborate ligands. J. Am. Chem. Soc. 2000, 122, 11845–11854. [Google Scholar]
- Williams, C.K.; Brooks, N.R.; Hillmyer, M.A.; Tolman, W.B. Metalloenzyme inspired dizinc catalyst for the polymerization of lactide. Chem. Commun. 2002, 18, 2132–2133. [Google Scholar] [CrossRef]
- Chisholm, M.H.; Gallucci, J.C.; Zhen, H.H.; Huffman, J.C. Three-coordinate zinc amide and phenoxide complexes supported by a bulky Schiff base ligand. Inorg. Chem. 2001, 40, 5051–5054. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.Y.; Tang, H.Y.; Lin, C.C. Ring-opening polymerization of lactides initiated by zinc alkoxides derived from NNO-tridentate ligands. Macromolecules 2006, 39, 3745–3752. [Google Scholar] [CrossRef]
- Breyfogle, L.E.; Williams, C.K.; Young, V.G.; Hillmyer, M.A.; Tolman, W.B. Comparison of structurally analogous Zn2, Co2, and Mg2 catalysts for the polymerization of cyclic esters. Dalton Trans. 2006, 7, 928–936. [Google Scholar] [CrossRef] [PubMed]
- Lian, B.; Thomas, C.M.; Casagrande, O.L., Jr.; Lehmann, C.W.; Roisnel, T.; Carpentier, J.F. Aluminum and zinc complexes based on amino-bis(pyrazolyl) ligand: Synthesis, structures and use in MMA and lactide polymerization. Inorg. Chem. 2007, 46, 328–340. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.H.; Lin, C.N.; Hsueh, M.L.; Athar, T.; Lin, C.C. Well-defined sterically hindered zinc aryloxides: Excellent catalysts for ring-opening polymerization of ε-caprolactone and L-lactide. Polym. J. 2006, 47. [Google Scholar] [CrossRef]
- Bukhaltsev, E.; Frish, L.; Cohen, Y.; Vigalok, A. Single-site catalysis by bimetallic zinc calixarene inclusion complexes. Org. Lett. 2005, 7, 5123–5126. [Google Scholar] [CrossRef] [PubMed]
- Coles, M.P.; Hitchcock, P.B. Zinc guanidinate complexes and their application in ringopening polymerisation catalysis. Eur. J. Inorg. Chem. 2004, 13, 2662–2672. [Google Scholar] [CrossRef]
- Dobrzynski, P. Initiation process of l-lactide polymerization carried out with zirconium (IV) acetylacetonate. J. Polym. Sci. A Polym. Chem. 2004, 42, 1886–1900. [Google Scholar] [CrossRef]
- Kricheldorf, H.R.; Serra, A. Polylactones: 6. Influence of various metal salts on the optical purity of Poly(l-lactide). Polym. Bull. 1985, 14, 497–502. [Google Scholar] [CrossRef]
- Żółtowska, K.; Sobczak, M.; Olędzka, E. Novel zinc-catalytic systems for ring-opening polymerization of ε-caprolactone. Molecules 2015, 20, 2816–2827. [Google Scholar] [CrossRef] [PubMed]
- Kowalski, A.; Duda, A.; Penczek, S. Polymerization of l,l-lactide initiated by aluminum isopropoxide trimer or tetramer. Macromolecules 1998, 31, 2114–2122. [Google Scholar] [CrossRef]
- Channuan, W.; Siripitayananon, J.; Molloy, R.; Sriyai, M.; Davis, F.J.; Mitchell, G.R. The structure of crystallisable copolymers of l-lactide, ε-caprolactone and glycolid. Polym. J. 2005, 46, 6411–6428. [Google Scholar] [CrossRef]
- Nijenhuis, A.J.; Grijpma, D.W.; Pennings, A.J. Lewis acid catalyzed polymerization of l-lactide. Kinetics and mechanism of the bulk polymerization. Macromolecules 1992, 25, 6419–6424. [Google Scholar] [CrossRef]
- Gupta, A.P.; Kumar, V. New emerging trends in synthetic biodegradable polymers—Polylactide: A critique. Eur. Polym. J. 2007, 43, 4053–4074. [Google Scholar] [CrossRef]
- Kasperczyk, J.E. Microstructure analysis of Poly(lactic acid) obtained by lithium tert-butoxide as initiator. Macromolecules 1995, 28, 3937–3939. [Google Scholar] [CrossRef]
- Kasperczyk, J.; Bero, M. Stereoselective polymerization of racemic d,l-lactide in the presence of butyllithium and butylmagnesium. Structural investigations of the polymers. Polymer 2000, 41, 391–395. [Google Scholar] [CrossRef]
- Stanford, M.J.; Dove, A.P. Stereocontrolled ring-opening polymerisation of lactide. Chem. Soc. Rev. 2010, 39, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.M. Stereocontrolled ring-opening polymerization of cyclic esters: Synthesis of new polyester microstructures. Chem. Soc. Rev. 2010, 39, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Dijkstra, P.J.; Du, H.; Feijen, J. Single site catalysts for stereoselective ring-opening polymerization of lactides. Polym. Chem. 2011, 2, 520–527. [Google Scholar] [CrossRef]
- Piedra-Arroni, E.; Ladaviere, C.; Amgoune, A.; Bourissou, D. Ring-opening polymerization with Zn(C6F5)2-based Lewis pairs: Original and efficient approach to cyclic polyesters. J. Am. Chem. Soc. 2013, 135, 13306–13309. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Ma, H. Highly diastereoselective synthesis of chiral aminophenolate zinc complexes and isoselective polymerization of rac-lactide. Chem. Commun. 2013, 49, 8686–8688. [Google Scholar] [CrossRef] [PubMed]
- Honrado, M.; Otero, A.; Fernandez-Baeza, J.; Sanchez-Barba, L.F.; Lara-Sanchez, A.; Tejeda, J.; Carrion, M.P.; Martinez-Ferrer, J.; Garces, A.; Rodriguez, A.M. Efficient synthesis of an unprecedented enantiopure hybrid scorpionate/cyclopentadienyl by diastereoselective nucleophilic addition to a fulvene. Organometallics 2013, 32, 3437–3440. [Google Scholar] [CrossRef]
- Zell, M.T.; Padden, B.E.; Paterick, A.J.; Thakur, K.A.M.; Kean, R.T.; Hillmyer, M.A.; Munson, E.J. Unambiguous determination of the 13C- and 1H-NMR stereosequence assignments of polylactide using high-resolution solution NMR spectroscopy. Macromolecules 2002, 35, 7700–7707. [Google Scholar] [CrossRef]
- Bero, M.; Kasperczyk, J.; Jedlinski, Z.J. Coordination polymerization of lactides, 1. Structure determination of obtained polymers. Macromol. Chem. Phys. 1990, 191, 2287–2296. [Google Scholar] [CrossRef]
- Bero, M.; Dobrzyński, P.; Kasperczyk, J. Synthesis of disyndiotactic polylactide. J. Polym. Sci. A. Polym. Chem. 1999, 37, 4038–4042. [Google Scholar] [CrossRef]
- Darensbourg, D.J.; Choi, W.; Karroonnirun, O.; Bhuvanesh, N. Ring-opening polymerization of cyclic monomers by complexes derived from biocompatible metals. Production of Poly(lactide), poly(trimethylene carbonate), and their copolymers. Macromolecules 2008, 41, 3493–3502. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the synthesized compounds are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Żółtowska, K.; Piotrowska, U.; Oledzka, E.; Sobczak, M. Efficient Diethylzinc/Gallic Acid and Diethylzinc/Gallic Acid Ester Catalytic Systems for the Ring-Opening Polymerization of rac-Lactide. Molecules 2015, 20, 21909-21923. https://doi.org/10.3390/molecules201219815
Żółtowska K, Piotrowska U, Oledzka E, Sobczak M. Efficient Diethylzinc/Gallic Acid and Diethylzinc/Gallic Acid Ester Catalytic Systems for the Ring-Opening Polymerization of rac-Lactide. Molecules. 2015; 20(12):21909-21923. https://doi.org/10.3390/molecules201219815
Chicago/Turabian StyleŻółtowska, Karolina, Urszula Piotrowska, Ewa Oledzka, and Marcin Sobczak. 2015. "Efficient Diethylzinc/Gallic Acid and Diethylzinc/Gallic Acid Ester Catalytic Systems for the Ring-Opening Polymerization of rac-Lactide" Molecules 20, no. 12: 21909-21923. https://doi.org/10.3390/molecules201219815
APA StyleŻółtowska, K., Piotrowska, U., Oledzka, E., & Sobczak, M. (2015). Efficient Diethylzinc/Gallic Acid and Diethylzinc/Gallic Acid Ester Catalytic Systems for the Ring-Opening Polymerization of rac-Lactide. Molecules, 20(12), 21909-21923. https://doi.org/10.3390/molecules201219815








