Clones of FeSOD, MDHAR, DHAR Genes from White Clover and Gene Expression Analysis of ROS-Scavenging Enzymes during Abiotic Stress and Hormone Treatments
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
2.1.1. Cloning and Sequence Analysis of FeSOD, DHAR and MDHAR Genes in White Clover
| Gene | Species | Identity(%) | Accession No. |
|---|---|---|---|
| FeSOD | Medicago sativa | 94 | AF377344.1 |
| Pisum sativum | 94 | AJ496175.1 | |
| Lotus japonicus | 89 | AY525601.1 | |
| Vigna unguiculata | 85 | AF077224.2 | |
| Glycine max | 84 | NM 001250972.1 | |
| Fagus sylvatica | 81 | DQ787261.1 | |
| DHAR | Medicago truncatula | 92 | DQ006811.1 |
| Cicer arietinum | 91 | KF276974.1 | |
| Lotus corniculatus | 89 | DQ013362.1 | |
| Glycine max | 86 | NM 001250000.1 | |
| MDHAR | Medicago sativa | 94 | JN979555.1 |
| Cicer arietinum | 91 | KF276975.1 | |
| Pisum sativum | 90 | AY730589.1 | |
| Glycine max | 88 | NM 001289382.1 |
2.1.2. Phylogenetic Analysis
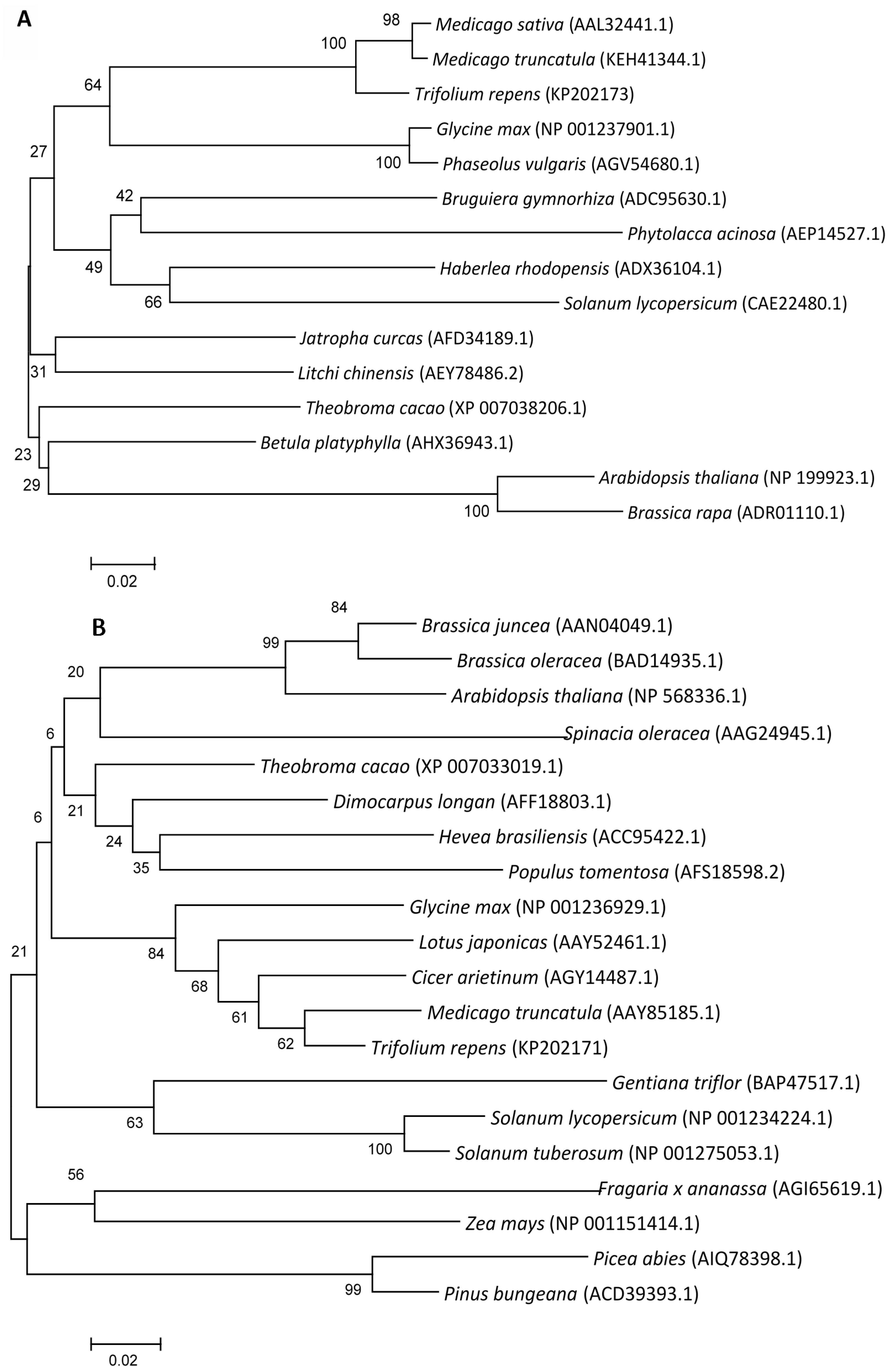
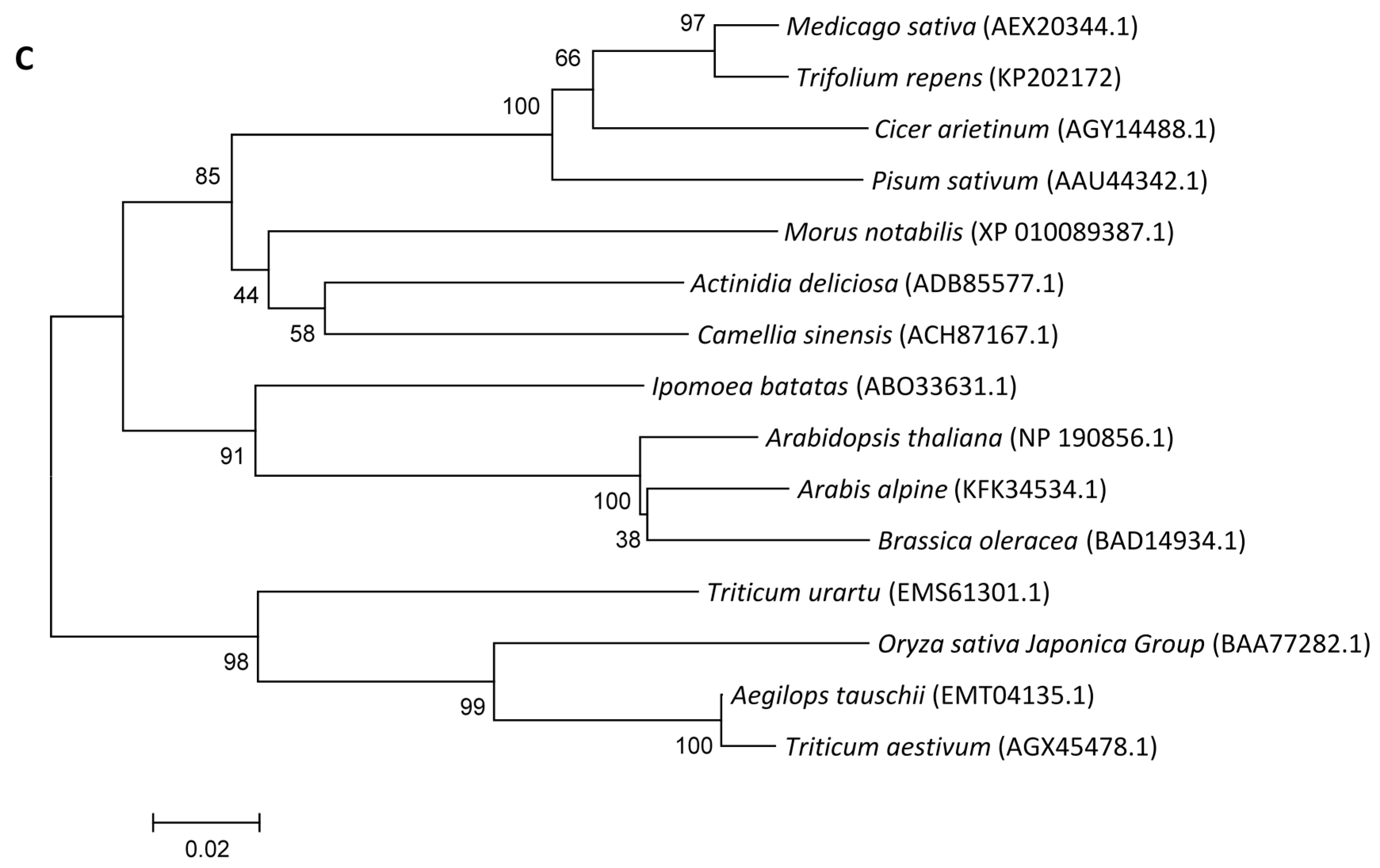
2.1.3. Expression Patterns of Genes Encoding ROS-Scavenging Enzymes in Response to Abiotic Stress
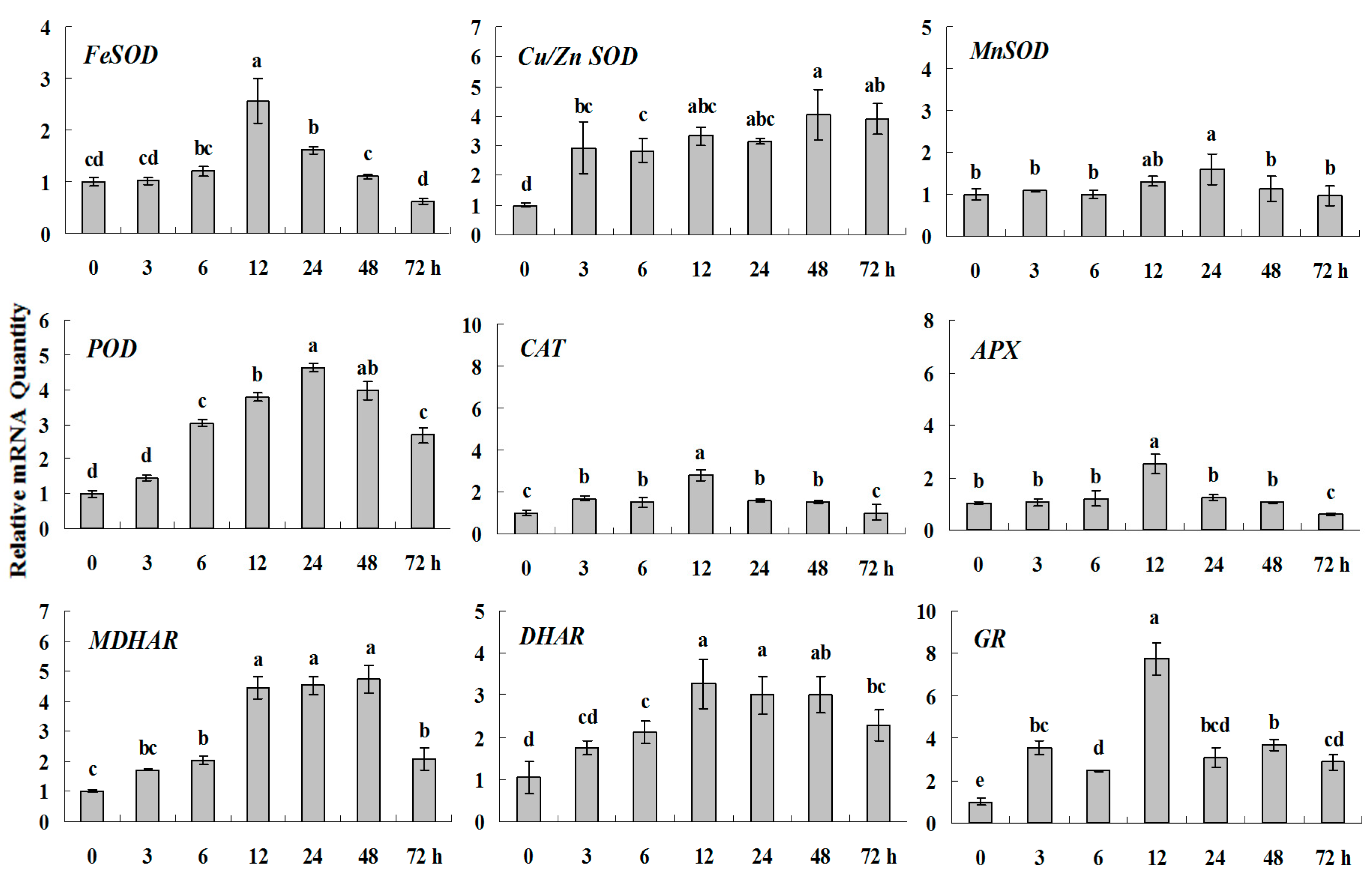
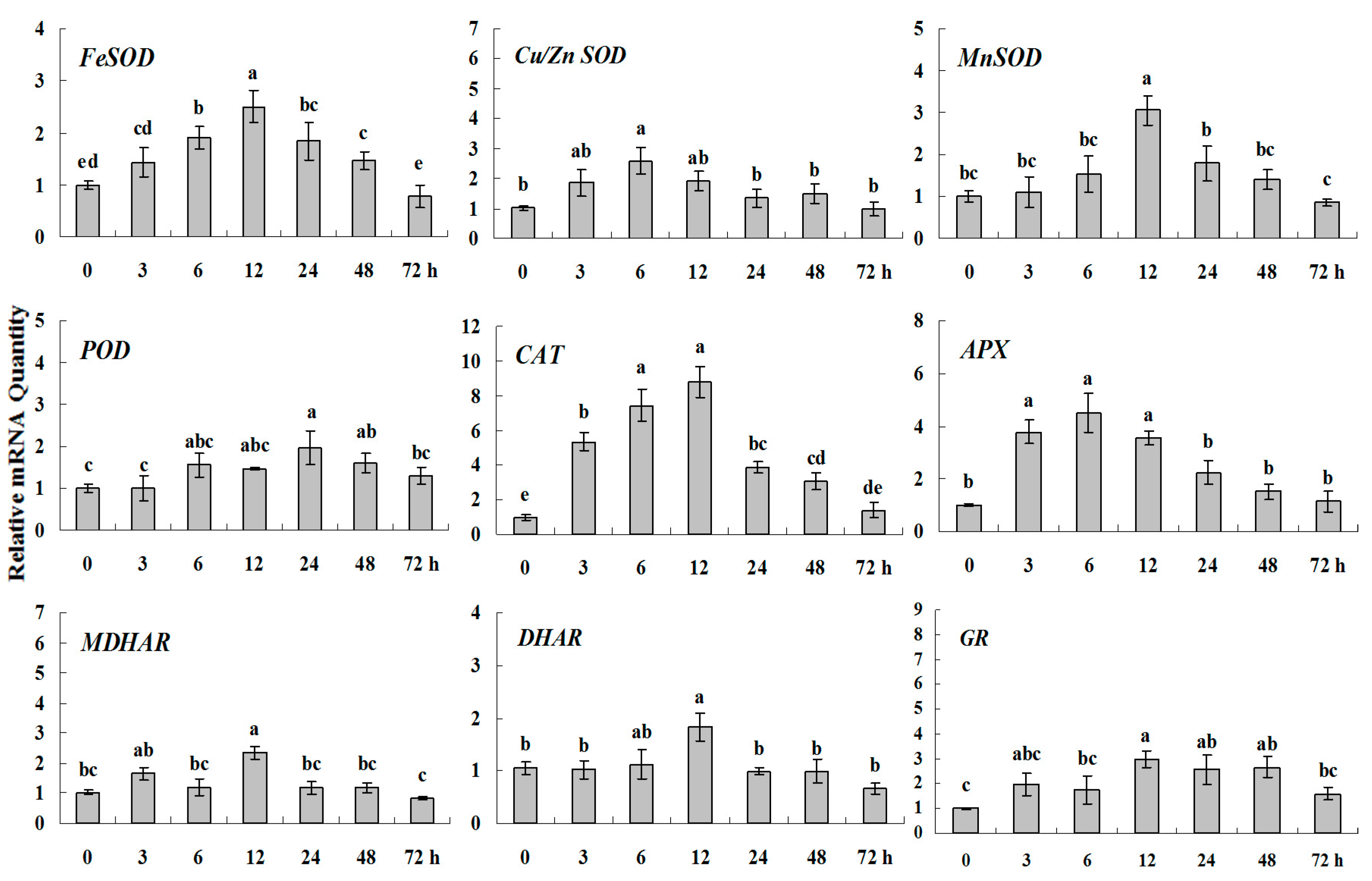
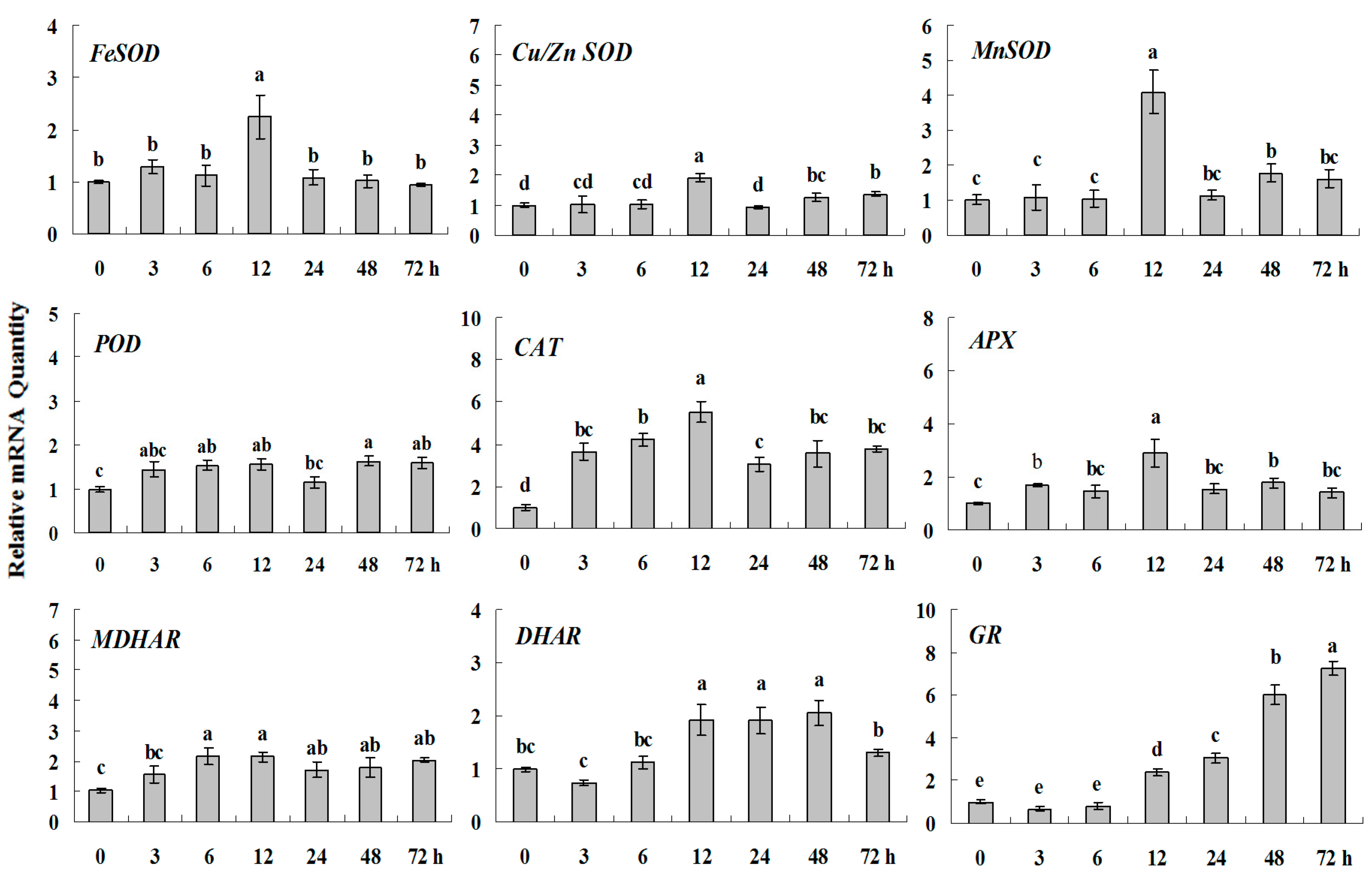
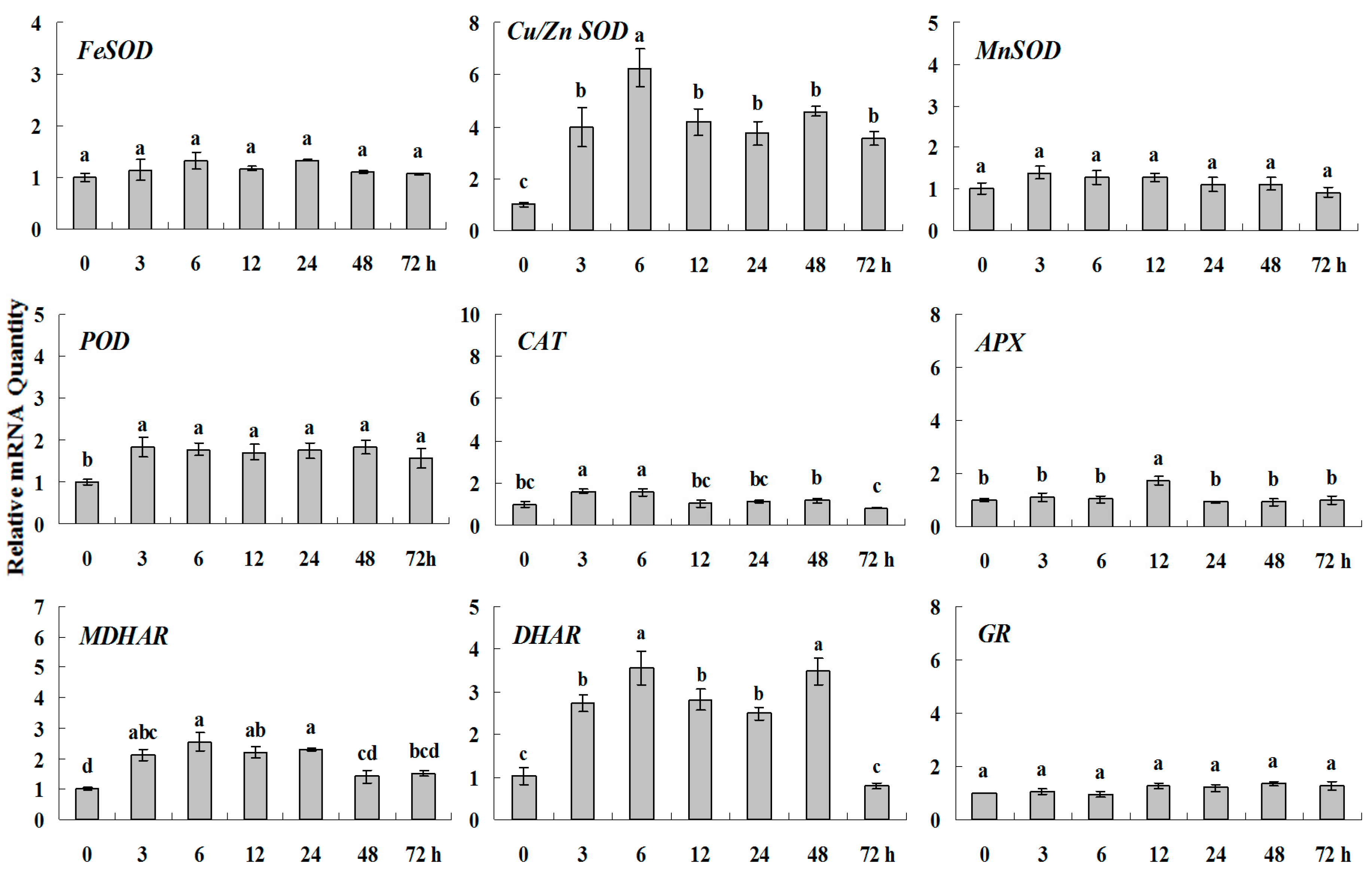
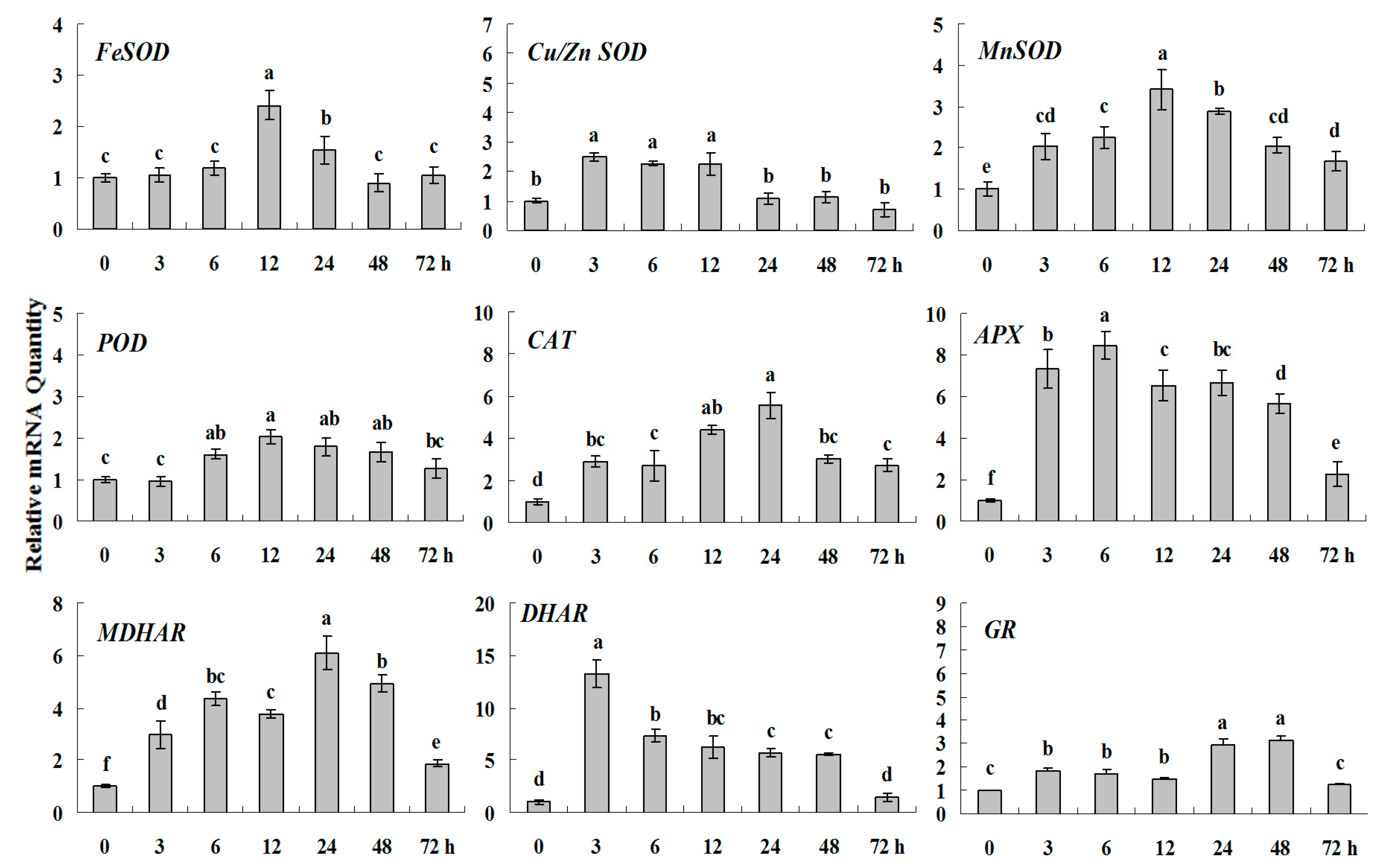
2.1.4. Expression Patterns of Genes Encoding ROS-Scavenging Enzymes in Response to PGRs
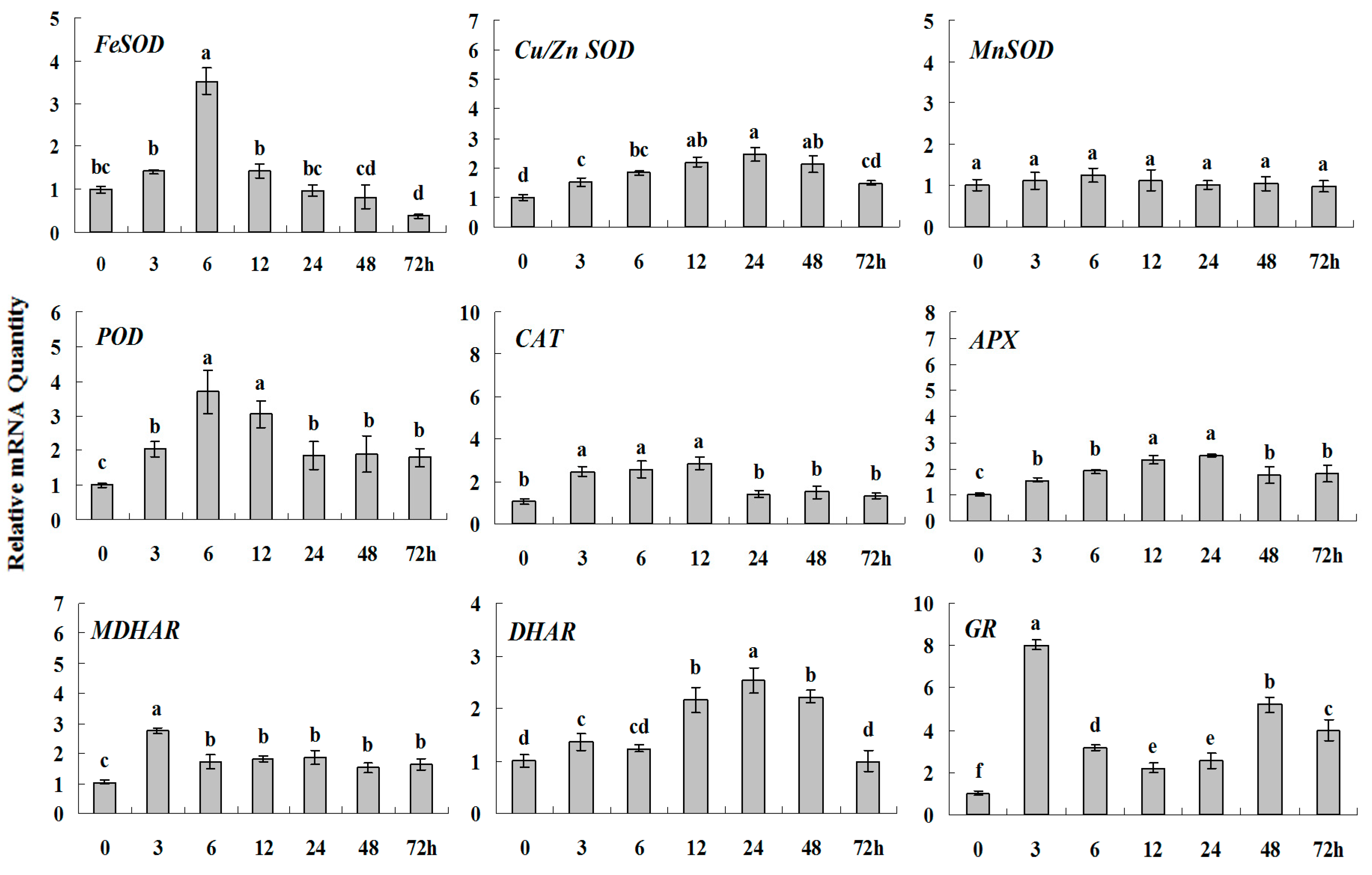
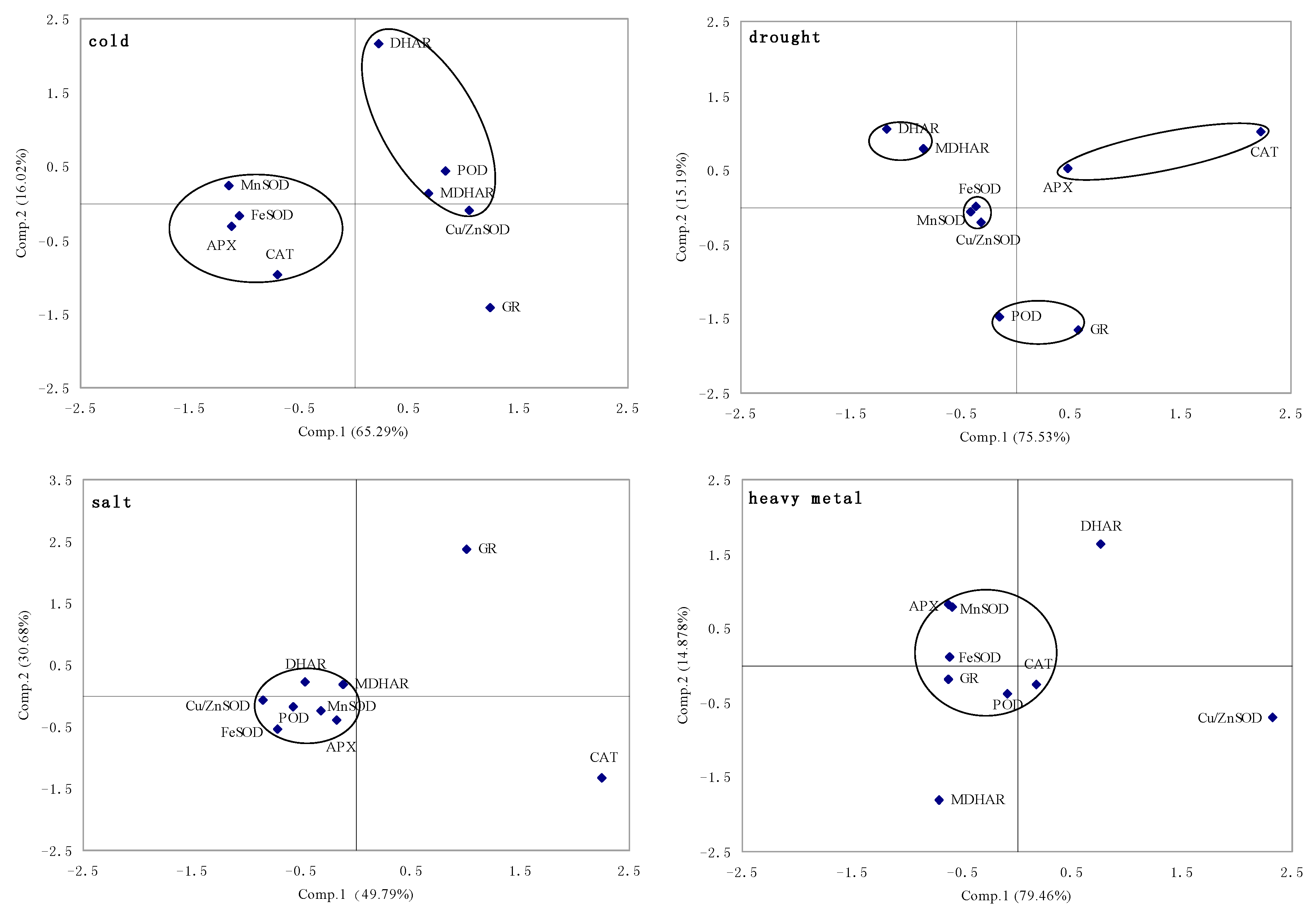
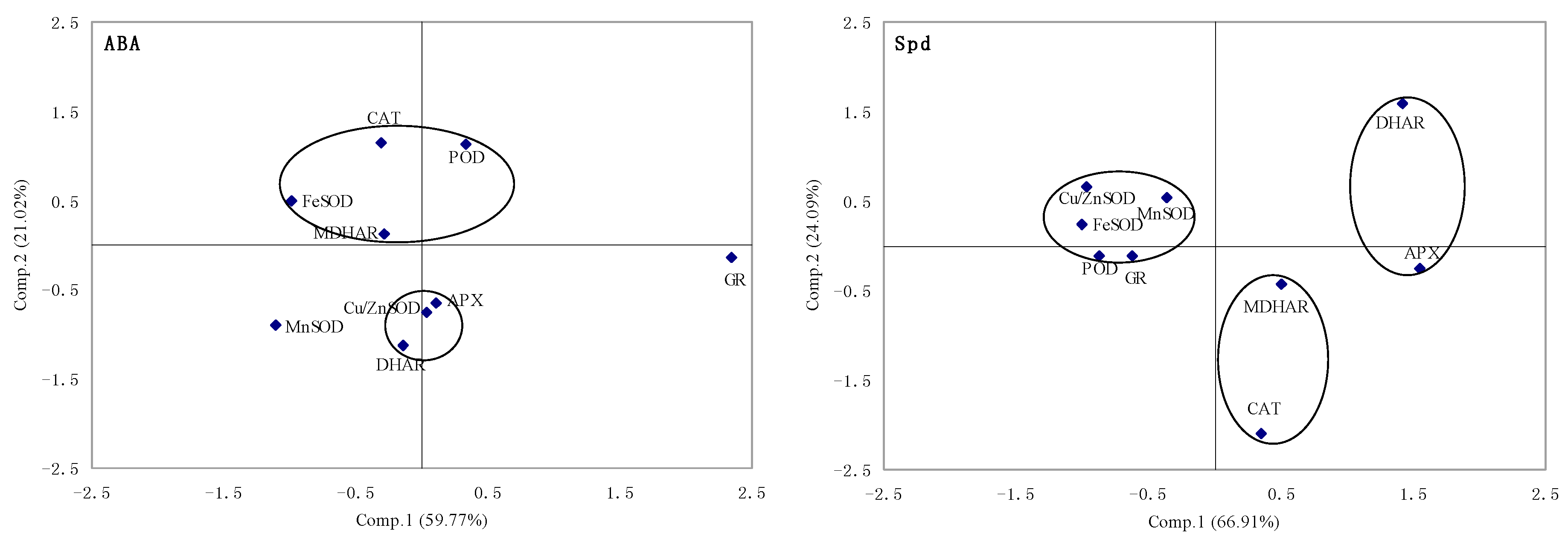
2.1.5. Principal Component Analysis of Genes Expression
2.2. Discussion
3. Experimental Section
3.1. Plant Materials and Growth Conditions
3.2. RNA Isolation and cDNA Synthesis
3.3. cDNA Sequence Isolation
| Gene | Forward Primer (5′-3′) | Reverse Primer (5′-3′) |
|---|---|---|
| FeSOD | TCACTGCAAAGTTTGAGCTG | ACTGCTTCCCAGGAAACAAG |
| DHAR | CTAAATGGTATAGCTTTGGTCC | GAAATCGCTGTTAAAGCTTC |
| MDHAR | TTATGCAGCAAGGGAGTTTGTG | ACTTCTTTTACCTCTCCATCGG |
3.4. Sequence Alignment, Identity Analysis and Principal Components Analysis
3.5. Quantitative Real-Time RT-PCR
| Gene | Accession No. | Forward Primer (5′-3′) | Reverse Primer (5′-3′) |
|---|---|---|---|
| β-Actin | JF968419 | TTACAATGAATTGCGTGTTG | AGAGGACAGCCTGAATGG |
| FeSOD | KP202173 | ACACGATTTCTCAGGGTTACGAC | GCGGCCAAGACTATCAGTTCCAT |
| CuZnSOD | JQ321597.1 | AACTGTGTACCACGAGGACTTC | AGACTAACAGGTGCTAACAACG |
| MnSOD | JQ321598.1 | TAAGGGAACCTACCCGATAACT | CCAGGACCAAACGTCACCAAAG |
| CAT | JQ321596.1 | AACAGGACGGGAATAGCACG | ACCAGGTTCAGACACGGAGACA |
| POD | AJ011939.1 | TCTAGGGCAACGGTTAATTCATTC | GGTACGGATTTTCCCATTTCTTG |
| APX | JQ321599.1 | TAAAGATAGTCAACCCACCTCAACA | ACCAGTCTTGGGAAACAACGTA |
| DHAR | KP202171 | TGGTTACCTCCCGACCCTAT | TCTTACCAAGGAACTTTAGTCAGG |
| MDHAR | KP202172 | CCAACTGCCTAAAGCCACATCT | GAAGAAAGGAAACTAACGGAGCA |
| GR | JQ321602.1 | TAAACTTCCACTCCCTTTCTATCG | CTACAATATGGGTTGAGGACAGGT |
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
References
- Hendry, G.A. Oxygen, free radical processes and seed longevity. Seed Sci. Res. 1993, 3, 141–153. [Google Scholar] [CrossRef]
- Sharma, P.; Jha, A.; Dubey, R.; Pessarakli, M. Oxidative stress and antioxidative defense system in plants growing under abiotic stresses. In Handbook Plant Crop Stress; CRC Press: Boca Raton, FL, USA, 2010; pp. 89–138. [Google Scholar]
- Zelko, I.N.; Mariani, T.J.; Folz, R.J. Superoxide dismutase multigene family: A comparison of the CuZn-SOD (SOD1), Mn-SOD (SOD2), and Fe-SOD (SOD3) gene structures, evolution, and expression. Free Radic. Biol. Med. 2002, 33, 337–349. [Google Scholar] [CrossRef]
- Zhang, J.; Kirkham, M.B. Enzymatic responses of the ascorbate-glutathione cycle to drought in sorghum and sunflower plants. Plant Sci. 1996, 113, 139–147. [Google Scholar] [CrossRef]
- Asada, K. Production and scavenging of reactive oxygen species in chloroplasts and their functions. Plant Physiol. 2006, 141, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, J.; Jimenez, A.; Mullineaux, P.; Sevilia, F. Tolerance of pea (Pisum sativum L.) to long-term salt stress is associated with induction of antioxidant defences. Plant Cell Environ. 2000, 23, 853–862. [Google Scholar] [CrossRef]
- Vaidyanathan, H.; Sivakumar, P.; Chakrabarty, R.; Thomas, G. Scavenging of reactive oxygen species in nacl-stressed rice (Oryza sativa L.)-differential response in salt-tolerant and sensitive varieties. Plant Sci. 2003, 165, 1411–1418. [Google Scholar] [CrossRef]
- Khan, F.; Siddiqi, T.O.; Mahmooduzzafar; Ahmad, A. Morphological changes and antioxidant defence systems in soybean genotypes as affected by salt stress. J. Plant Interact. 2009, 4, 295–306. [Google Scholar] [CrossRef] [Green Version]
- Sairam, R.; Srivastava, G.; Agarwal, S.; Meena, R. Differences in antioxidant activity in response to salinity stress in tolerant and susceptible wheat genotypes. Biol. Plant. 2005, 49, 85–91. [Google Scholar] [CrossRef]
- De Souza, T.C.; Magalhães, P.C.; de Castro, E.M.; Carneiro, N.P.; Padilha, F.A.; Júnior, C.C.G. Aba application to maize hybrids contrasting for drought tolerance: Changes in water parameters and in antioxidant enzyme activity. Plant Growth Regul. 2014, 73, 205–217. [Google Scholar] [CrossRef]
- Wang, Y.; Wisniewski, M.; Meilan, R.; Cui, M.; Webb, R.; Fuchigami, L. Overexpression of cytosolic ascorbate peroxidase in tomato confers tolerance to chilling and salt stress. J. Am. Soc. Hortic. Sci. 2005, 130, 167–173. [Google Scholar]
- Li, Z.; Peng, Y.; Zhang, X.-Q.; Pan, M.-H.; Ma, X.; Huang, L.-K.; Yan, Y.-H. Exogenous spermidine improves water stress tolerance of white clover (Trifolium repens L.) involved in antioxidant defence, gene expression and proline metabolism. Plant Omics 2014, 7, 517. [Google Scholar]
- Kaminaka, H.; Morita, S.; Nakajima, M.; Masumura, T.; Tanaka, K. Gene cloning and expression of cytosolic glutathione reductase in rice (Oryza sativa L.). Plant Cell Physiol. 1998, 39, 1269–1280. [Google Scholar] [CrossRef] [PubMed]
- Baek, K.-H.; Skinner, D.Z. Alteration of antioxidant enzyme gene expression during cold acclimation of near-isogenic wheat lines. Plant Sci. 2003, 165, 1221–1227. [Google Scholar] [CrossRef]
- Agarwal, S.; Sairam, R.; Srivastava, G.; Tyagi, A.; Meena, R. Role of aba, salicylic acid, calcium and hydrogen peroxide on antioxidant enzymes induction in wheat seedlings. Plant Sci. 2005, 169, 559–570. [Google Scholar] [CrossRef]
- Lu, Z.; Liu, D.; Liu, S. Two rice cytosolic ascorbate peroxidases differentially improve salt tolerance in transgenic arabidopsis. Plant Cell Rep. 2007, 26, 1909–1917. [Google Scholar] [CrossRef] [PubMed]
- Eltayeb, A.E.; Kawano, N.; Badawi, G.H.; Kaminaka, H.; Sanekata, T.; Morishima, I.; Shibahara, T.; Inanaga, S.; Tanaka, K. Enhanced tolerance to ozone and drought stresses in transgenic tobacco overexpressing dehydroascorbate reductase in cytosol. Physiol. Plant. 2006, 127, 57–65. [Google Scholar] [CrossRef]
- Lee, Y.-P.; Kim, S.-H.; Bang, J.-W.; Lee, H.-S.; Kwak, S.-S.; Kwon, S.-Y. Enhanced tolerance to oxidative stress in transgenic tobacco plants expressing three antioxidant enzymes in chloroplasts. Plant Cell Rep. 2007, 26, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Jiang, J.; Chang, Q.; Gu, C.; Song, A.; Chen, S.; Dong, B.; Chen, F. Cold acclimation induces freezing tolerance via antioxidative enzymes, proline metabolism and gene expression changes in two chrysanthemum species. Mol. Biol. Rep. 2014, 41, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Li, L.; Jung, W.; Jin, Y.; Avice, J.; Ourry, A.; Kim, T. Water deficit-induced oxidative stress and the activation of antioxidant enzymes in white clover leaves. Biol. Plant. 2009, 53, 505–510. [Google Scholar] [CrossRef]
- Vaseva, I.; Akiscan, Y.; Simova-Stoilova, L.; Kostadinova, A.; Nenkova, R.; Anders, I.; Feller, U.; Demirevska, K. Antioxidant response to drought in red and white clover. Acta Physiol. Plant. 2012, 34, 1689–1699. [Google Scholar] [CrossRef]
- Mhadhbi, H.; Fotopoulos, V.; Mylona, P.V.; Jebara, M.; Elarbi Aouani, M.; Polidoros, A.N. Antioxidant gene-enzyme responses in medicago truncatula genotypes with different degree of sensitivity to salinity. Physiol. Plant. 2011, 141, 201–214. [Google Scholar] [CrossRef] [PubMed]
- Sung, M.S.; Chow, T.J.; Lee, T.M. Polyamine acclimation alleviates hypersalinity-induced oxidative stress in a marine green macroalga, Ulva fasciata, by modulation of antioxidative enzyme gene expression1. J. Phycol. 2011, 47, 538–547. [Google Scholar] [CrossRef]
- Parvin, S.; Lee, O.R.; Sathiyaraj, G.; Khorolragchaa, A.; Kim, Y.-J.; Yang, D.-C. Spermidine alleviates the growth of saline-stressed ginseng seedlings through antioxidative defense system. Gene 2014, 537, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.C.; Qu, G.Z.; Li, H.Y.; Wu, Y.J.; Wang, C.; Liu, G.F.; Yang, C.P. Enhanced salt tolerance of transgenic poplar plants expressing a manganese superoxide dismutase from Tamarix androssowii. Mol. Biol. Rep. 2010, 37, 1119–1124. [Google Scholar] [CrossRef] [PubMed]
- Eltayeb, A.E.; Kawano, N.; Badawi, G.H.; Kaminaka, H.; Sanekata, T.; Shibahara, T.; Inanaga, S.; Tanaka, K. Overexpression of monodehydroascorbate reductase in transgenic tobacco confers enhanced tolerance to ozone, salt and polyethylene glycol stresses. Planta 2007, 225, 1255–1264. [Google Scholar] [CrossRef] [PubMed]
- Ushimaru, T.; Nakagawa, T.; Fujioka, Y.; Daicho, K.; Naito, M.; Yamauchi, Y.; Nonaka, H.; Amako, K.; Yamawaki, K.; Murata, N. Transgenic arabidopsis plants expressing the rice dehydroascorbate reductase gene are resistant to salt stress. J. Plant Physiol. 2006, 163, 1179–1184. [Google Scholar] [CrossRef] [PubMed]
- Verma, K.; Mehta, S.; Shekhawat, G. Nitric oxide (no) counteracts cadmium induced cytotoxic processes mediated by reactive oxygen species (ros) in brassica juncea: Cross-talk between ros, no and antioxidant responses. BioMetals 2013, 26, 255–269. [Google Scholar] [CrossRef] [PubMed]
- Manier, N.; Brulle, F.; le Curieux, F.; Vandenbulcke, F.; Deram, A. Biomarker measurements in trifolium repens and eisenia fetida to assess the toxicity of soil contaminated with landfill leachate: A microcosm study. Ecotoxicol. Environ. Saf. 2012, 80, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Romero-Puertas, M.C.; Corpas, F.J.; Rodríguez-Serrano, M.; Gómez, M.; Luis, A.; Sandalio, L.M. Differential expression and regulation of antioxidative enzymes by cadmium in pea plants. J. Plant Physiol. 2007, 164, 1346–1357. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.-B.; Kim, Y.-H.; Lee, H.-S.; Kim, K.-Y.; Deng, X.-P.; Kwak, S.-S. Analysis of antioxidant enzyme activity during germination of alfalfa under salt and drought stresses. Plant Physiol. Biochem. 2009, 47, 570–577. [Google Scholar] [CrossRef] [PubMed]
- Štork, F.; Bačkor, M.; Klejdus, B.; Hedbavny, J.; Kováčik, J. Changes of metal-induced toxicity by H2O2/NO modulators in Scenedesmus quadricauda (chlorophyceae). Environ. Sci. Pollut. Res. 2013, 20, 5502–5511. [Google Scholar] [CrossRef] [PubMed]
- Kováčik, J.; Babula, P.; Hedbavny, J.; Švec, P. Manganese-induced oxidative stress in two ontogenetic stages of chamomile and amelioration by nitric oxide. Plant Sci. 2014, 215, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Shi, G.; Xu, Q.; Hu, J. Exogenous polyamines enhance copper tolerance of nymphoides peltatum. J. Plant Physiol. 2007, 164, 1062–1070. [Google Scholar] [CrossRef] [PubMed]
- Mullineaux, P.M.; Creissen, G.P. Glutathione reductase: Regulation and role in oxidative stress. Cold Spring Harb. Monogr. Arch. 1997, 34, 667–713. [Google Scholar]
- Guo, W.; Chen, R.; Gong, Z.; Yin, Y.; Ahmedand, S.; He, Y. Exogenous abscisic acid increases antioxidant enzymes and related gene expression in pepper (Capsicum annuum) leaves subjected to chilling stress. Genet. Mol. Res. 2012, 11, 4063–4080. [Google Scholar] [CrossRef] [PubMed]
- Ghassemian, M.; Lutes, J.; Chang, H.-S.; Lange, I.; Chen, W.; Zhu, T.; Wang, X.; Lange, B.M. Abscisic acid-induced modulation of metabolic and redox control pathways in Arabidopsis thaliana. Phytochemistry 2008, 69, 2899–2911. [Google Scholar] [CrossRef] [PubMed]
- Aronova, E.; Shevyakova, N.; Stetsenko, L.; Kuznetsov, V.V. In Cadaverine-induced induction of superoxide dismutase gene expression in Mesembryanthemum crystallinum L. Dokl. Biol. Sci. 2005, 403, 257–259. [Google Scholar] [CrossRef] [PubMed]
- Hiraga, S.; Ito, H.; Yamakawa, H.; Ohtsubo, N.; Seo, S.; Mitsuhara, I.; Matsui, H.; Honma, M.; Ohashi, Y. An hr-induced tobacco peroxidase gene is responsive to spermine, but not to salicylate, methyl jasmonate, and ethephon. Mol. Plant-Microbe Interact. 2000, 13, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Hoagland, D.R.; Arnon, D.I. The Water-Culture Method for Growing Plants without Soil; Circular California Agricultural Experiment Station 347: Berkeley, CA, USA, 1950. [Google Scholar]
- NCBI. Available online: http://www.ncbi.nlm.gov/blast/ (accessed on 15 October 2015).
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are not available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Li, Z.; Peng, Y.; Wang, X.; Peng, D.; Li, Y.; He, X.; Zhang, X.; Ma, X.; Huang, L.; et al. Clones of FeSOD, MDHAR, DHAR Genes from White Clover and Gene Expression Analysis of ROS-Scavenging Enzymes during Abiotic Stress and Hormone Treatments. Molecules 2015, 20, 20939-20954. https://doi.org/10.3390/molecules201119741
Zhang Y, Li Z, Peng Y, Wang X, Peng D, Li Y, He X, Zhang X, Ma X, Huang L, et al. Clones of FeSOD, MDHAR, DHAR Genes from White Clover and Gene Expression Analysis of ROS-Scavenging Enzymes during Abiotic Stress and Hormone Treatments. Molecules. 2015; 20(11):20939-20954. https://doi.org/10.3390/molecules201119741
Chicago/Turabian StyleZhang, Yan, Zhou Li, Yan Peng, Xiaojuan Wang, Dandan Peng, Yaping Li, Xiaoshuang He, Xinquan Zhang, Xiao Ma, Linkai Huang, and et al. 2015. "Clones of FeSOD, MDHAR, DHAR Genes from White Clover and Gene Expression Analysis of ROS-Scavenging Enzymes during Abiotic Stress and Hormone Treatments" Molecules 20, no. 11: 20939-20954. https://doi.org/10.3390/molecules201119741
APA StyleZhang, Y., Li, Z., Peng, Y., Wang, X., Peng, D., Li, Y., He, X., Zhang, X., Ma, X., Huang, L., & Yan, Y. (2015). Clones of FeSOD, MDHAR, DHAR Genes from White Clover and Gene Expression Analysis of ROS-Scavenging Enzymes during Abiotic Stress and Hormone Treatments. Molecules, 20(11), 20939-20954. https://doi.org/10.3390/molecules201119741








