New Sulphated Flavonoids from Wissadula periplocifolia (L.) C. Presl (Malvaceae)
Abstract
:1. Introduction
2. Results and Discussion
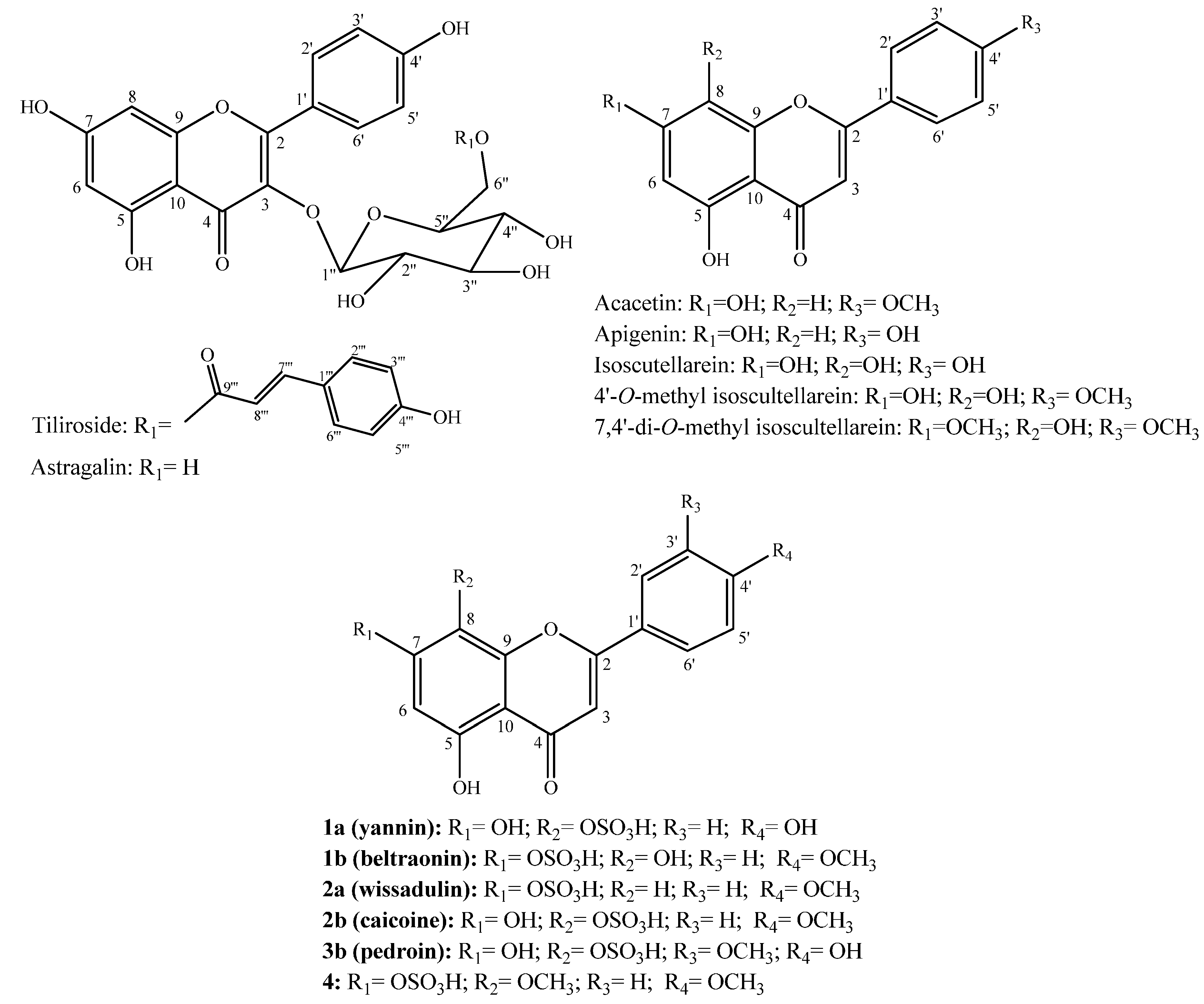
2.1. Structure Elucidation of Compounds
| Position | 1a | 1b | 2a | 2b | 3b | 4 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| δH (J in Hz) | δC | δH (J in Hz) | δC | δH (J in Hz) | δC | δH (J in Hz) | δC | δH (J in Hz) | δC | δH (J in Hz) | δC | |
| 2 | - | 164.5, C | - | 164.3, C | - | 164.3, C | - | 163.9, C | - | 164.4, C | - | 164.4, C |
| 3 | 6.81, s | 103.1, CH | 6.93, s | 103.9, CH | 6.93, s | 104.2, CH | 6.89, s | 103.7, CH | 6.87, s | 103.3, CH | 6.87, s | 103.3, CH |
| 4 | - | 182.5, C | - | 183.1, C | - | 182.6, C | - | 182.4, C | - | 182.5, C | - | 182.8, C |
| 5 | - | 157.5, C | - | 152.0, C | - | 161.0, C | - | 157.4, C | - | 157.5, C | - | 157.6, C |
| 6 | 6.29, s | 100.0, CH | 6.90, s | 104.1, CH | 6.60, d (1.8) | 102.1, CH | 6.30, s | 100.0, CH | 6.29, s | 99.9, CH | 6.53, s | 96.6, CH |
| 7 | - | 157.5, C | - | 148.3, C | - | 160.1, C | - | 157.5, C | - | 157.5, C | - | 159.7, C |
| 8 | - | 121.9, C | - | 129.5, C | 7.05, d (1.8) | 98.0, CH | - | 121.9, C | - | 121.1, C | - | 123.3, C |
| 9 | - | 150.2, C | - | 145.7, C | - | 156.9, C | - | 150.1, C | - | 151.9, C | - | 149.9, C |
| 10 | - | 104.4, C | - | 106.8, C | - | 106.2, C | - | 104.4, C | - | 104.4, C | - | 104.3, C |
| 1′ | - | 121.9, C | - | 123.5, C | - | 123.1, C | - | 123.4, C | - | 122.3, C | - | 123.7, C |
| 2′ | 8.03, d (8.7) | 129.0, CH | 8.11, d (8.9) | 129.3, CH | 8.07, d (8.7) | 129.0, CH | 8.14, d (8.9) | 129.1, CH | 7.64, dd (1.8, 8.5) | 121.1, CH | 8.28, d (8.7) | 129.7, CH |
| 3′ | 6.93, d (8.7) | 116.4, CH | 7.14, d (8.9) | 115.2, CH | 7.12, d (8.7) | 115.1, CH | 7.13, d (8.9) | 115.0, CH | 7.14, dd (0.7, 8.5) | 115.2, CH | 7.08, d (8.7) | 114.9, CH |
| 4′ | - | 161.8, C | - | 163.0, C | - | 162.9, C | - | 162.9, C | - | 151.2, C | - | 162.8, C |
| 5′ | 6.93, d (8.7) | 116.4, CH | 7.14, d (8.9) | 115.2, CH | 7.12, d (8.7) | 115.1, CH | 7.13, d (8.9) | 115.0, CH | - | 148.5, C | 7.08, d (8.7) | 114.9, CH |
| 6′ | 8.03, d (8.7) | 129.0, CH | 8.11, d (8.9) | 129.3, CH | 8.07, d (8.7) | 129.0, CH | 8.14, d (8.9) | 129.1, CH | 7.82, d (1.8) | 111.2, CH | 8.28, d (8.7) | 129.7, CH |
| OCH3-4′ | - | - | 3.86, s | 56.1, CH3 | 3.86, s | 56.0, CH3 | 3.87, s | 56.0, CH3 | - | - | 3.86, s | 56.1, CH3 |
| OCH3-7 | - | - | - | - | - | - | - | - | - | - | 3.85, s | 57.0, CH3 |
| OCH3-5′ | - | - | - | - | - | - | - | - | 3.88, s | 56.4, CH3 | - | - |
| OH-5 | 12.75, s | - | 12.16, s | - | 12.82, s | - | 12.70, s | - | 12.71, s | - | 12.87, s | - |
| OH-8 | - | - | 8.98, s | - | - | - | - | - | - | - | - | - |
| OH-7 | 9.96, s | - | - | - | - | - | - | - | - | - | - | - |
| OH-4′ | 10.42, s | - | - | - | - | - | - | - | - | - | - | - |
2.2. Evaluation of Cytotoxicity
| Treatment | IC50 (µg/mL) | |||
|---|---|---|---|---|
| Tumour Cell Lines | Normal Cell Lines | |||
| UVW | PC-3M | PNT2A | Hs27 | |
| acacetin | 27.37 ± 1.09 ab | 21.13 ± 1.16 ab | 51.33 ± 1.09 | >100 |
| 7,4′-di-O-methylisoscutellarein | 98.55 ± 1.13 ab | >100 | 88.73 ± 1.13 | NA |
| tiliroside | >100 | 60.55 ± 1.12 ab | >100 | NA |
| acacetin + apigenin (1.2:1) | >100 | 48.13 ± 1.09 ab | >100 | >100 |
| 2a + 2b (2:1) | >100 | 92.14 ± 1.09 ab | >100 | NA |
3. Experimental Section
3.1. General Procedures
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Compound Identification
3.5. Liquid Chromatography-Mass Spectrometry (LC-MS)
| Time (min) | A% | B% |
|---|---|---|
| 0 | 75 | 25 |
| 15 | 25 | 75 |
3.6. Cell lines and Cell Culture
3.7. Evaluation of Cytotoxicity
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rocha, J.F.; Neves, L.J. Anatomia foliar de Hibiscus tiliaceus L. e Hibiscus pernambucensis Arruda (Malvaceae). Rodriguesia 2000, 51, 113–132. [Google Scholar]
- Silva, D.A.; Falcão-Silva, V.S.; Gomes, A.Y.S.; Costa, D.A.; Lemos, V.S.; Agra, M.F.; Braz-Filho, R.; Siqueira-Junior, J.P.; Souza, M.F.V. Triterpenes and phenolic compounds isolated from the aerial parts of Herissantia tiubae and evaluation of 5,4′,-dihydroxy-3,6,7,8,3′-pentamethoxyflavone as modulator of bacterial drug resistance. Pharm. Biol. 2009, 47, 279–284. [Google Scholar] [CrossRef]
- Gomes, R.A.; Maciel, J.K.S.; Agra, M.F.; Souza, M.F.V.; Falcão-Silva, V.S.; Siqueira-Junior, J.P. Phenolic compounds from Sidastrum micranthum (A. St.-Hil.) Fryxell and evaluation of acacetin and 7,4′-Di-O-methylisoscutellarein as motulator of bacterial drug resistence. Quim. Nova 2011, 34, 1385–1388. [Google Scholar] [CrossRef]
- Oliveira, A.M.F.; Pinheiro, L.S.; Pereira, C.K.S.; Matias, W.N.; Gomes, R.A.; Chaves, O.S.; Souza, M.F.V.; Almeida, R.N.; Assis, T.S. Total phenolic content and antioxidant activity of some Malvaceae family species. Antioxidants 2012, 1, 33. [Google Scholar] [CrossRef]
- Billeter, M.; Meier, B.; Sticher, O. 8-Hydroxyflavonoid glucuronides from Malva sylvestris. Phytochemistry. 1991, 30, 987–990. [Google Scholar] [CrossRef]
- Nawwar, M.; Buddrus, J. A gossypetin glucuronide sulphate from the leaves of Malva sylvestris. Phytochemistry 1981, 20, 2446–2448. [Google Scholar] [CrossRef]
- Buchholz, H.; Wirth, C.; Carola, C.; Alves Fontes, R. Flavonoid derivative. U.S. Patent 20,070,134,172 A1, 14 July 2007. [Google Scholar]
- Guglielmone, H.A.; Agnese, A.M.; Montoya, S.C.N.; Cabrera, J.L. Inhibitory effects of sulphated flavonoids isolated from Flaveria bidentis on platelet aggregation. Thromb. Res. 2005, 115, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Gurni, A.A.; König, W.A.; Kubitzki, K. Flavonoid glycosides and sulphates from the Dilleniaceae. Phytochemistry 1981, 20, 1057–1059. [Google Scholar] [CrossRef]
- Harborne, J. B. Flavonoid sulphates: A new class of sulphur compounds in higher plants. Phytochemistry 1975, 14, 1147–l155. [Google Scholar] [CrossRef]
- Tomás-Barberán, F.; Harborne, J.B.; Self, R. Twelve 6-Oxygenated-Flavone Sulphates from Lippia nodiflora and L. canescens. Phytochemistry 1987, 26, 2281–2284. [Google Scholar] [CrossRef]
- Bylka, W.; Stobiecki, M.; Frahski, R. Sulphated flavonoid glycosides from leaves of Atriplex hortensis. Acta Physiol. Plant. 2001, 23, 285–290. [Google Scholar] [CrossRef]
- Correia-da-Silva, M.; Sousa, E.; Pinto, M.M. Emerging sulfated flavonoids and other polyphenols as drugs: Nature as an inspiration. Med. Res. Rev. 2014, 34, 223–279. [Google Scholar] [CrossRef] [PubMed]
- Varin, L.; Marsolais, F.; Richard, M.; Rouleau, M. Sulfation and sulfotransferases 6: Biochemistry and molecular biology of plant sulfotransferases. FASEB J. 1997, 11, 517–525. [Google Scholar] [PubMed]
- Varin, L.; Marsolais, F.; Brisson, N. Chimeric flavonol sulfotransferases define a domain responsible for substrate and position specificities. J. Biol. Chem. 1995, 270, 12498–12502. [Google Scholar] [PubMed]
- Kopriva, S.; Mugford, S.G.; Baraniecka, P.; Lee, B.; Matthewman, C.A.; Koprivova, A. Control of sulfur partitioning between primary and secondary metabolism in Arabidopsis. Front. Plant Sci. 2012, 3, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Correia-da-Silva, M.; Sousa, E.; Duarte, B.; Marques, F.; Carvalho, F.; Cunha-Ribeiro, L.M.; Pinto, M.M. Flavonoids with an oligopolysulfated moiety: a new class of anticoagulant agents. J. Med. Chem. 2011, 54, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Gunnarsson, G.T.; Desai, U.R. Interaction of designed sulfated flavanoids with antithrombin: Lessons on the design of organic activators. J. Med. Chem. 2002, 45, 4460–4470. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Liang, N.-C. Inhibitory effect of disodium quercetin-7,4′-disulfate on aggregation of pig platelets induced by thrombin and its mechanism. Acta Pharmacol. Sin. 2000, 21, 737–741. [Google Scholar] [PubMed]
- Teles, Y.C.F.; Gomes, R.A.; Oliveira, M.S.; Lucena, K.L.; Nascimento, J.S.; Agra, M.F.; Igoli, J.O.; Gray, A.I.; Souza, M.F.V. Phytochemical investigation of Wissadula periplocifolia (L.) C. Presl and evaluation of its antibacterial activity. Quim. Nova 2014, 37, 1491–1495. [Google Scholar]
- Yoon, K.D.; Jeong, D.G.; Hwang, Y.H.; Ryu, J.M.; Kim, J. Inhibitors of osteoclast differentiation from Cephalotaxus koreana. J. Nat. Prod. 2007, 70, 2029–2032. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Qiu, F.; Zhu, S.; Zhang, T.; Qu, G.; Yao, X. Isolation and identification of ten metabolites of breviscapine in rat urine. Biol. Pharm. Bull. 2007, 30, 1308–1316. [Google Scholar] [CrossRef] [PubMed]
- Meselhy, M.R. Constituents from Moghat, the roots of Glossostemon bruguieri (Desf.). Molecules 2003, 8, 614–621. [Google Scholar] [CrossRef]
- Lin, Y.; Kong, L. Studies on the chemical constituents of Desmodium styracifolium (Osbeck) Merr. Asian J. Tradit. Med. 2006, 1, 34–36. [Google Scholar]
- Hsu, Y.L.; Kuo, P.L.; Lin, C.C. Acacetin inhibits the proliferation of Hep G2 by blocking cell cycle progression and inducing apoptosis. Biochem. Pharmacol. 2004, 67, 823–829. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.H.; Lai, C.S.; Hsu, P.C.; Wang, Y.J. Acacetin induces apoptosis in human gastric carcinoma cells accompanied by activation of caspase cascades and production of reactive oxygen species. J. Agric. Food Chem. 2005, 53, 620–630. [Google Scholar] [CrossRef] [PubMed]
- Shim, H.Y.; Park, J.H.; Paik, H.D.; Nah, S.Y.; Kim, D.S.; Han, Y.S. Acacetin-induced apoptosis of human breast cancer MCF-7 cells involves caspase cascade, mitochondria-mediated death signaling and SAPK/JNK1/2-c-Jun activation. Mol. Cells 2007, 24, 95–104. [Google Scholar] [PubMed]
- Freitas, S.; Costa, S.; Azevedo, C.; Carvalho, G.; Freire, S.; Barbosa, P.; Velozo, E.; Schaer, R.; Tardy, M.; Meyer, R.; Nascimento, I. Flavonoids inhibit angiogenic cytokine production by human glioma cells. Phytother. Res. 2011, 25, 916–921. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.R.; Park, C.G.; Jung, J.Y. Acacetin (5,7-dihydroxy-4′-methoxyflavone) exhibits in vitro and in vivo anticancer activity through the suppression of NF-κB/Akt signaling in prostate cancer cells. Int. J. Mol. Med. 2014, 33, 317–324. [Google Scholar] [PubMed]
- Kim, C.; Kim, M.C.; Kim, S.M.; Nam, D.; Choi, S.H.; Kim, S.H.; Ahn, K.S.; Lee, E.H.; Jung, S.H.; Ahn, K.S. Chrysanthemum indicum L. extract induces apoptosis through suppression of constitutive STAT3 activation in human prostate cancer DU145 Cells. Phytother. Res. 2013, 27, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Shen, K.H.; Hung, S.H.; Yin, L.T.; Huang, C.S.; Chao, C.H.; Liu, C.L.; Shih, Y.W. Acacetin, a flavonoid, inhibits the invasion and migration of human prostate cancer DU145 cells via inactivation of the p38 MAPK signaling pathway. Mol. Cell. Biochem. 2010, 333, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; Gupta, S. Apigenin: A promising molecule for cancer prevention. Pharm Res. 2010, 27, 962–978. [Google Scholar] [CrossRef] [PubMed]
- Knowles, L.M.; Zigrossi, D.A.; Tauber, R.A.; Hightower, C.; Milner, J.A. Flavonoids suppress androgen-independent human prostate tumor proliferation. Nutr. Cancer 2000, 38, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Morrissey, C.; O’Neill, A.; Spengler, B.; Christoffel, V.; Fitzpatrick, J.M.; Watson, R.W. Apigenin drives the production of reactive oxygen species and initiates a mitochondrial mediated cell death pathway in prostate epithelial cells. Prostate 2005, 63, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; Gupta, S. Apigenin-induced prostate cancer cell death is initiated by reactive oxygen species and p53 activation. Free Radic. Biol. Med. 2008, 44, 1833–1845. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Banik, N.L.; Ray, S.K. Flavonoids activated caspases for apoptosis in human glioblastoma t98g and u87mg cells but not in human normal astrocytes. Cancer 2010, 116, 164–176. [Google Scholar] [CrossRef] [PubMed]
- Seibert, H.; Maser, E.; Schweda, K.; Seibert, S.; Gülden, M. Cytoprotective activity against peroxide-induced oxidative damage and cytotoxicity of flavonoids in C6 rat glioma cells. Food Chem. Toxicol. 2011, 49, 2398–2407. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.R.; Kuo, Y.H.; Ho, Y.L.; Wang, C.Y.; Yang, C.S.; Lin, C.W.; Chang, Y.S. Studies on cytotoxic constituents from the leaves of Elaeagnus oldhamii Maxim. In non-small cell lung cancer a549 cells. Molecules 2014, 19, 9515–9534. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, P.R.C.; Aguiar, J.S.; Matias, W.N.; Gomes, R.A.; Teles, Y.C.F.; Souza, M.F.V.; Medeiros, P.L.; Silva, E.C.; Gonçalves-Silva, T.; Nascimento, S.C. In vitro and in vivo antitumor effects of the flavonol glycosides isolated of Herissantia crispa (L.) Brizicky. Lat. Am. J. Pharm. 2011, 30, 253–257. [Google Scholar]
- Tsimplouli, C.; Demetzos, C.; Hadzopoulou-Cladaras, M.; Pantazis, P.; Dimas, K. In vitro activity of dietary flavonol congeners against human cancer cell lines. Eur. J. Nutr. 2012, 51, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Tomczyk, M.; Tumanov, A.; Zaniewska, A.; Surazynski, A. The potential mechanism of tiliroside-dependent inhibition of t-butylhydroperoxide-induced oxidative stress in endometrial carcinoma cells. Planta Med. 2010, 76, 963–968. [Google Scholar] [CrossRef] [PubMed]
- Dimas, K.; Demetzos, C.; Vaos, B.; Marselos, M.; Kokkinopoulos, D. Cytotoxic and antiproliferative effects of heptaacetyl tilirosid in human leulemic cell lines. Leuk. Res. 1999, 23, 1021–1033. [Google Scholar] [CrossRef]
- Teles, Y.C.F.; Ribeiro-Filho, J.; Bozza, P.T.; Agra, M.F.; Siheri, W.; Igoli, J.O.; Gray, A.I.; Souza, M.F.V. Phenolic constituents from Wissadula periplocifolia (L.) C. Presl. and anti-inflammatory activity of 7,4′-di-O -methylisoscutellarein. Nat. Prod. Res. 2015, 1, 1–5. [Google Scholar] [CrossRef]
- Beninger, C.W.; Hosfield, G.L.; Bassett, M.J. Flavonoid Composition of Three Genotypes of Dry Bean (Phaseolus. vulgaris) differing in Seedcoat Color. J. Am. Soc. Hort. Sci. 1999, 124, 514–518. [Google Scholar]
- Boyd, M.; Mairs, R.J.; Keith, W.N.; Ross, S.C.; Welsh, P.; Akabani, G.; Owens, J.; Vaidyanathan, G.; Carruthers, R.; Dorrens, J.; et al. An efficient targeted radiotherapy/gene therapy strategy utilising human telomerase promoters and radioastatine and harnessing radiation-mediated bystander effects. J. Gene Med. 2004, 6, 937–947. [Google Scholar] [PubMed]
- Ahmed, S.A.; Gogal, R.M., Jr.; Walsh, J.E. A new rapid and simple non-radioactive assay to monitor and determine the proliferation of lymphocytes: An alternative to [3H]thymidine incorporation assay. J. Immunol. Methods 1994, 170, 211–224. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1 to 4 are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teles, Y.C.F.; Horta, C.C.R.; Agra, M.D.F.; Siheri, W.; Boyd, M.; Igoli, J.O.; Gray, A.I.; De Souza, M.D.F.V. New Sulphated Flavonoids from Wissadula periplocifolia (L.) C. Presl (Malvaceae). Molecules 2015, 20, 20161-20172. https://doi.org/10.3390/molecules201119685
Teles YCF, Horta CCR, Agra MDF, Siheri W, Boyd M, Igoli JO, Gray AI, De Souza MDFV. New Sulphated Flavonoids from Wissadula periplocifolia (L.) C. Presl (Malvaceae). Molecules. 2015; 20(11):20161-20172. https://doi.org/10.3390/molecules201119685
Chicago/Turabian StyleTeles, Yanna C. F., Carolina Campolina Rebello Horta, Maria De Fátima Agra, Weam Siheri, Marie Boyd, John O. Igoli, Alexander I. Gray, and Maria De Fátima Vanderlei De Souza. 2015. "New Sulphated Flavonoids from Wissadula periplocifolia (L.) C. Presl (Malvaceae)" Molecules 20, no. 11: 20161-20172. https://doi.org/10.3390/molecules201119685
APA StyleTeles, Y. C. F., Horta, C. C. R., Agra, M. D. F., Siheri, W., Boyd, M., Igoli, J. O., Gray, A. I., & De Souza, M. D. F. V. (2015). New Sulphated Flavonoids from Wissadula periplocifolia (L.) C. Presl (Malvaceae). Molecules, 20(11), 20161-20172. https://doi.org/10.3390/molecules201119685