The Phe-Phe Motif for Peptide Self-Assembly in Nanomedicine
Abstract
:1. Introduction
2. Preparation of Compounds Bearing the Phe-Phe Motif
3. Characterization of Compounds Bearing the Phe-Phe Motif
4. Nanomorphologies Formed by the Phe-Phe Motif
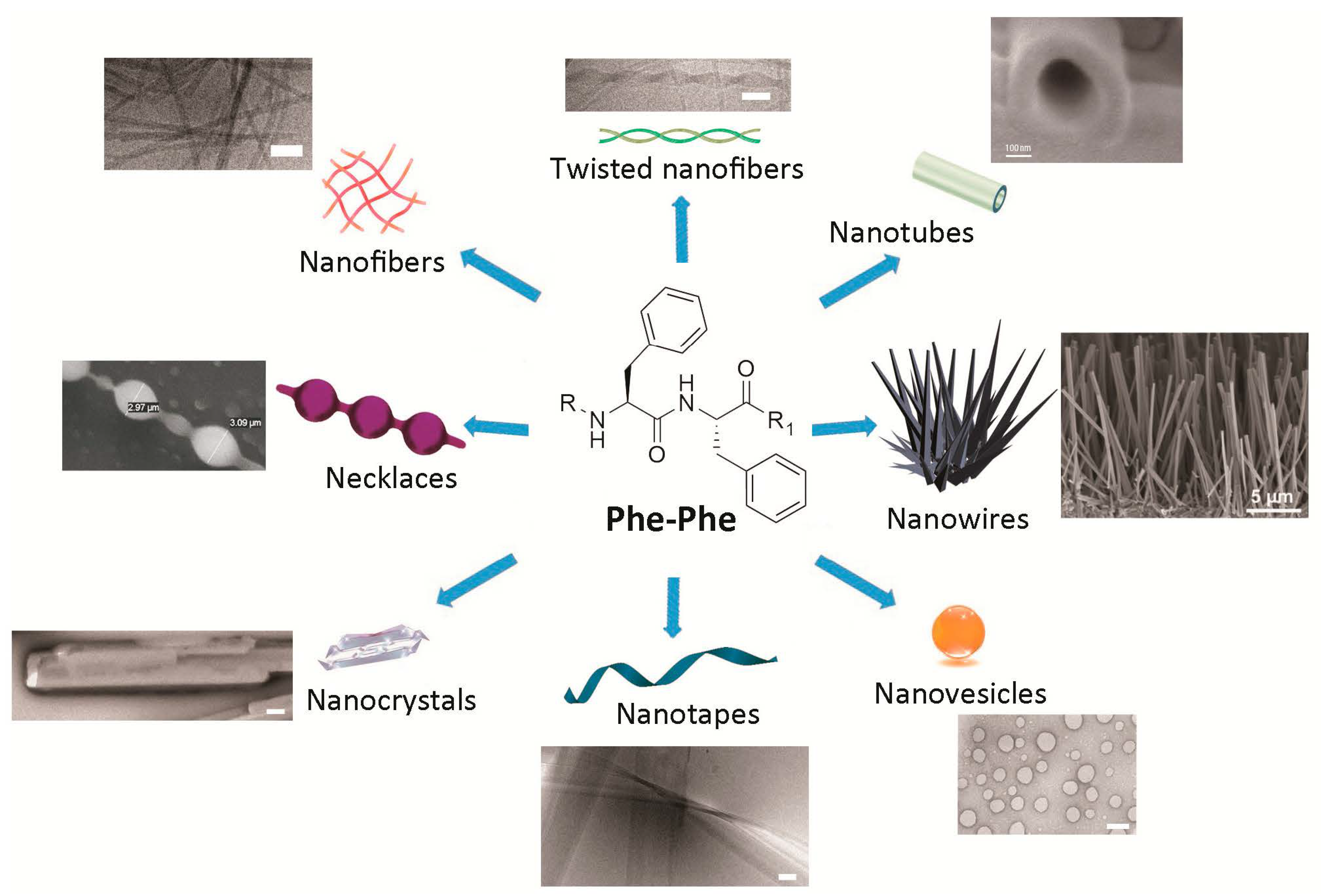
5. Use for Drug-Delivery
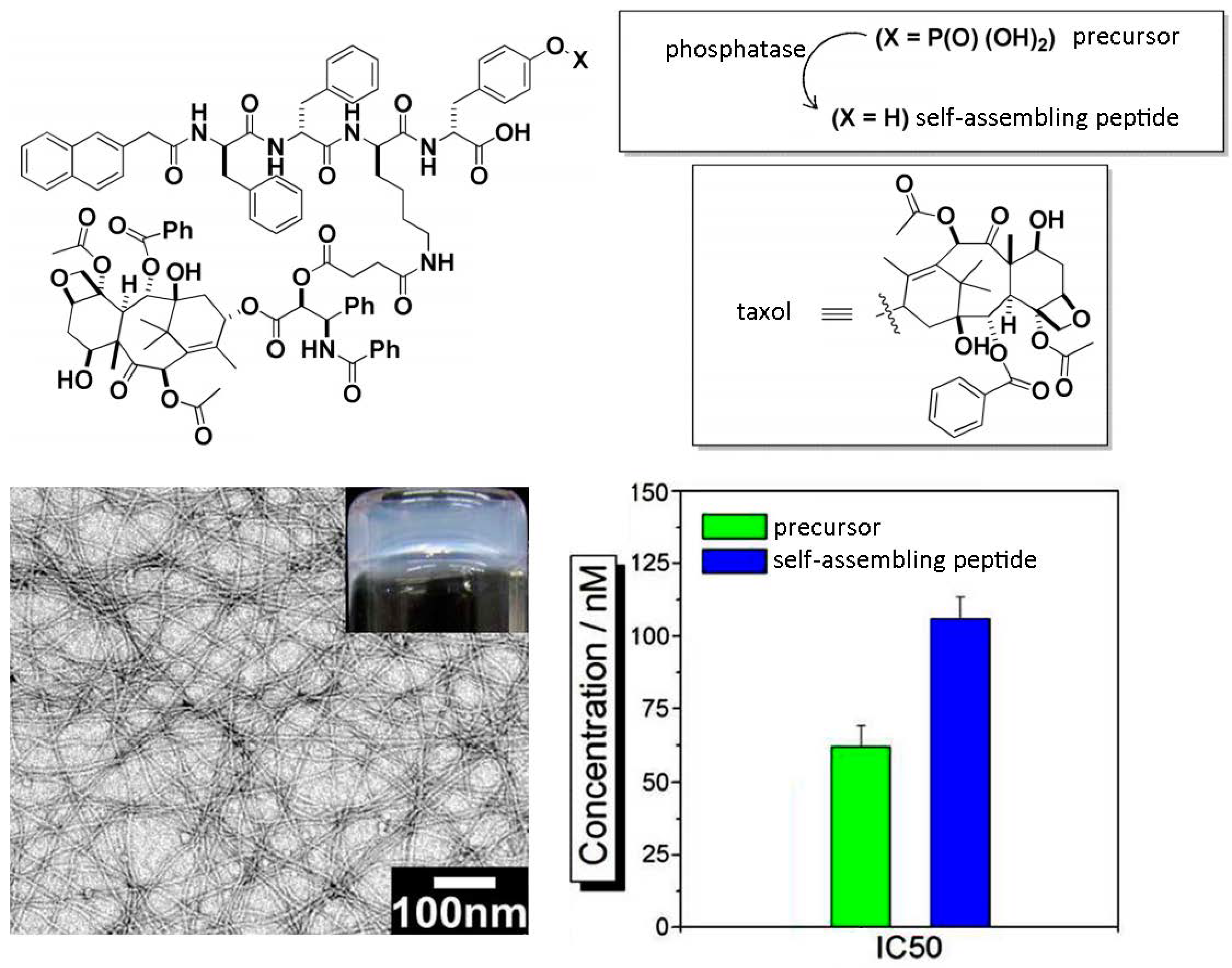

6. Biomaterials Formed by Self-Assembly of Phe-Phe Derivatives
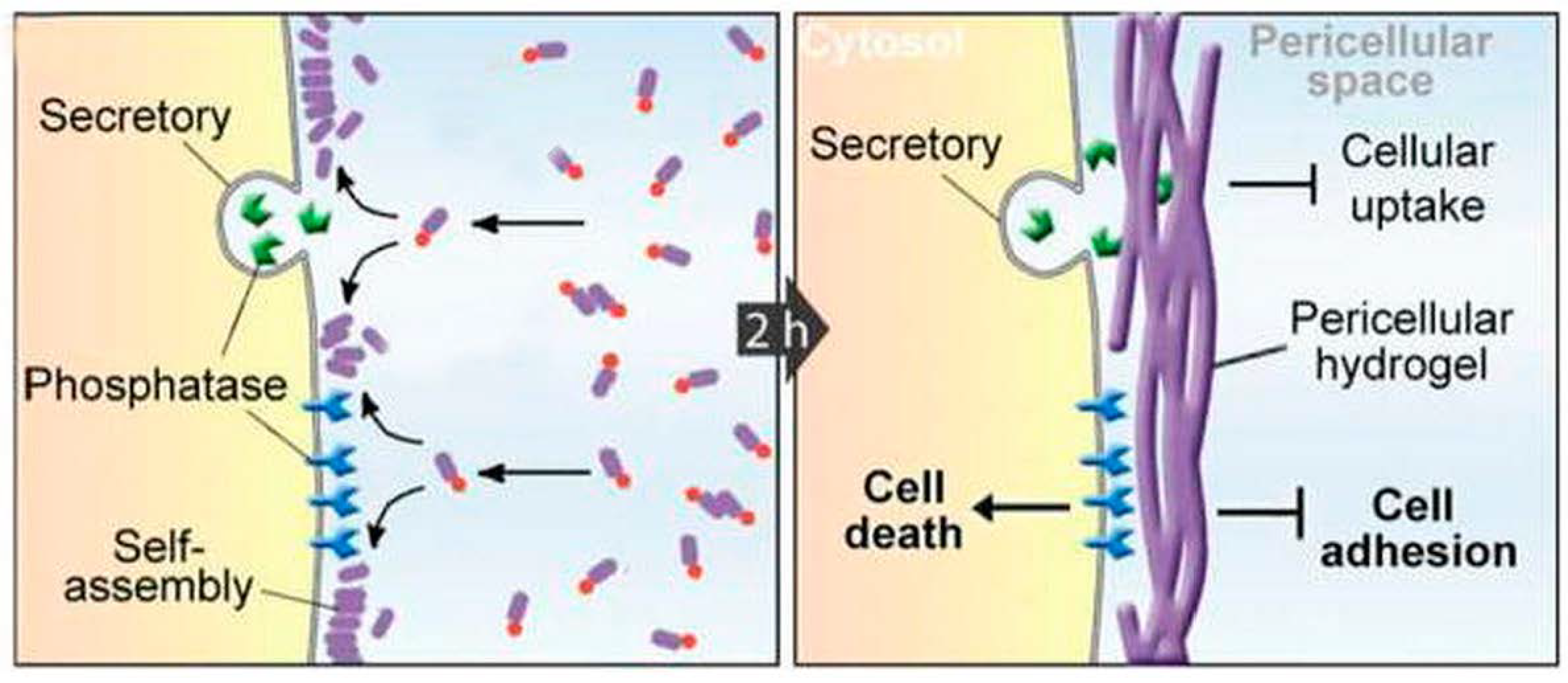
7. Other Applications
8. Conclusions
Acknowledgments
Conflicts of Interest
References
- Min, Y.; Caster, J.M.; Eblan, M.J.; Wang, A.Z. Clinical Translation of Nanomedicine. Chem. Rev. 2015, 115, 11147–11190. [Google Scholar] [CrossRef]
- Marchesan, S.; Prato, M. Nanomaterials for (Nano)medicine. ACS Med. Chem. Lett. 2013, 4, 147–149. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.Y.; Huang, W.; Li, Z.P.; Lei, X.Y.; He, D.X.; Sun, L. Progress in Self-assembling Peptide-based Nanomaterials for Biomedical Applications. Curr. Top. Med. Chem. 2016, 16, 281–290. [Google Scholar] [CrossRef]
- Collier, J.H.; Segura, T. Evolving the use of peptides as components of biomaterials. Biomaterials 2011, 32, 4198–4204. [Google Scholar] [CrossRef] [PubMed]
- Adams, D.J. Dipeptide and tripeptide conjugates as low-molecular-weight hydrogelators. Macromol. Biosci. 2011, 11, 160–173. [Google Scholar] [CrossRef] [PubMed]
- Fleming, S.; Ulijn, R.V. Design of nanostructures based on aromatic peptide amphiphiles. Chem. Soc. Rev. 2014, 43, 8150–8177. [Google Scholar] [CrossRef] [PubMed]
- Mba, M.; Mazzier, D.; Silvestrini, S.; Toniolo, C.; Fatás, P.; Jiménez, A.I.; Cativiela, C.; Moretto, A. Photocontrolled Self-Assembly of a Bis-Azobenzene-Containing α-Amino Acid. Chem. Eur. J. 2013, 19, 15841–15846. [Google Scholar] [CrossRef] [PubMed]
- Reches, M.; Gazit, E. Casting metal nanowires within discrete self-assembled peptide nanotubes. Science 2003, 300, 625–627. [Google Scholar] [CrossRef] [PubMed]
- Adler-Abramovich, L.; Vaks, L.; Carny, O.; Trudler, D.; Magno, A.; Caflisch, A.; Frenkel, D.; Gazit, E. Phenylalanine assembly into toxic fibrils suggests amyloid etiology in phenylketonuria. Nat. Chem. Biol. 2012, 8, 701–706. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Zhu, P.; Li, J. Self-assembly and application of diphenylalanine-based nanostructures. Chem. Soc. Rev. 2010, 39, 1877–1890. [Google Scholar] [CrossRef] [PubMed]
- Dudukovic, N.A.; Zukoski, C.F. Gelation of Fmoc-diphenylalanine is a first order phase transition. Soft Matter 2015, 11, 7663–7673. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; He, Q.; Wang, K.; Duan, L.; Cui, Y.; Li, J. Transition of Cationic Dipeptide Nanotubes into Vesicles and Oligonucleotide Delivery. Angew. Chem. Int. Ed. 2007, 46, 2431–2434. [Google Scholar] [CrossRef] [PubMed]
- Reches, M.; Gazit, E. Formation of Closed-Cage Nanostructures by Self-Assembly of Aromatic Dipeptides. Nano Lett. 2004, 4, 581–585. [Google Scholar] [CrossRef]
- Adler-Abramovich, L.; Gazit, E. Controlled patterning of peptide nanotubes and nanospheres using inkjet printing technology. J. Pept. Sci. 2008, 14, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Marchesan, S.; Easton, C.D.; Kushkaki, F.; Waddington, L.; Hartley, P.G. Tripeptide self-assembled hydrogels: Unexpected twists of chirality. Chem. Commun. 2012, 48, 2195–2197. [Google Scholar] [CrossRef] [PubMed]
- Maity, S.; Nir, S.; Zada, T.; Reches, M. Self-assembly of a tripeptide into a functional coating that resists fouling. Chem. Commun. 2014, 50, 11154–11157. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Li, B.; Huang, Z.; Li, Y.; Yang, Y. Terminal Is Important for the Helicity of the Self-Assemblies of Dipeptides Derived from Alanine. Langmuir 2013, 29, 6013–6017. [Google Scholar] [CrossRef] [PubMed]
- Das, A.K.; Collins, R.; Ulijn, R.V. Exploiting enzymatic (reversed) hydrolysis in directed self-assembly of peptide nanostructures. Small 2008, 4, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Chronopoulou, L.; Sennato, S.; Bordi, F.; Giannella, D.; di Nitto, A.; Barbetta, A.; Dentini, M.; Togna, A.R.; Togna, G.I.; Moschini, S.; et al. Designing unconventional Fmoc-peptide-based biomaterials: Structure and related properties. Soft Matter 2014, 10, 1944–1952. [Google Scholar] [CrossRef] [PubMed]
- Ostrov, N.; Gazit, E. Genetic engineering of biomolecular scaffolds for the fabrication of organic and metallic nanowires. Angew. Chem. Int. Ed. 2010, 49, 3018–3021. [Google Scholar] [CrossRef] [PubMed]
- Conlon, J.M. Purification of naturally occurring peptides by reversed-phase HPLC. Nat. Protoc. 2007, 2, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Mant, C.T.; Chen, Y.; Yan, Z.; Popa, T.V.; Kovacs, J.M.; Mills, J.B.; Tripet, B.P.; Hodges, R.S. HPLC analysis and purification of peptides. Methods Mol. Biol. 2007, 386, 3–55. [Google Scholar] [PubMed]
- Arai, T.; Kino, K. New l-amino acid ligases catalyzing oligopeptide synthesis from various microorganisms. Biosci. Biotechnol. Biochem. 2010, 74, 1572–1577. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Toledano, S.; Williams, R.J.; Jayawarna, V.; Ulijn, R.V. Enzyme-Triggered Self-Assembly of Peptide Hydrogels via Reversed Hydrolysis. J. Am. Chem. Soc. 2006, 128, 1070–1071. [Google Scholar] [CrossRef] [PubMed]
- Taylor Paul, P.; Grinter Nigel, J.; McCarthy Shelly, L.; Pantaleone David, P.; Ton Jennifer, L.; Yoshida Roberta, K.; Fotheringham Ian, G. d-Phenylalanine Biosynthesis Using Escherichia coli: Creation of a New Metabolic Pathway. In Applied Biocatalysis in Specialty Chemicals and Pharmaceuticals; American Chemical Society: Washington, DC, USA, 2001; Volume 776, pp. 65–75. [Google Scholar]
- Ollivaux, C.; Soyez, D.; Toullec, J.-Y. Biogenesis of d-amino acid containing peptides/proteins: Where, when and how? J. Pept. Sci. 2014, 20, 595–612. [Google Scholar] [CrossRef] [PubMed]
- Fusetani, N. Antifungal peptides in marine invertebrates. ISJ 2010, 7, 53–66. [Google Scholar]
- Yuran, S.; Reches, M. Formation of ordered biomolecular structures by the self-assembly of short peptides. J. Vis. Exp. 2013, 21, e50946. [Google Scholar] [CrossRef] [PubMed]
- Adamcik, J.; Mezzenga, R. Study of amyloid fibrils via atomic force microscopy. Curr. Opin. Coll. Interface Sci. 2012, 17, 369–376. [Google Scholar] [CrossRef]
- Gras, S.L.; Waddington, L.J.; Goldie, K.N. Transmission electron microscopy of amyloid fibrils. Methods Mol. Biol. 2011, 752, 197–214. [Google Scholar] [PubMed]
- Wang, Z.; Zhou, C.; Wang, C.; Wan, L.; Fang, X.; Bai, C. AFM and STM study of β-amyloid aggregation on graphite. Ultramicroscopy 2003, 97, 73–79. [Google Scholar] [CrossRef]
- Takai, E.; Ohashi, G.; Ueki, R.; Yamada, Y.; Fujita, J.-I.; Shiraki, K. Scanning Electron Microscope Imaging of Amyloid Fibrils. Am. J. Biochem. Biotechnol. 2014, 10, 31–39. [Google Scholar] [CrossRef]
- Schermelleh, L.; Heintzmann, R.; Leonhardt, H. A guide to super-resolution fluorescence microscopy. J. Cell Biol. 2010, 190, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Biancalana, M.; Koide, S. Molecular Mechanism of Thioflavin-T Binding to Amyloid Fibrils. Biochim. Biophys. Acta 2010, 1804, 1405–1412. [Google Scholar] [CrossRef] [PubMed]
- Howie, A.J.; Brewer, D.B. Optical properties of amyloid stained by Congo red: History and mechanisms. Micron 2009, 40, 285–301. [Google Scholar] [CrossRef] [PubMed]
- Marchesan, S.; Styan, K.; Easton, C.D.; Waddington, L.; Vargiu, A.V. Higher and lower supramolecular order for the design of self-assembled heterochiral tripeptide hydrogel biomaterials. J. Mater. Chem. B 2015, 3, 8123–8132. [Google Scholar] [CrossRef]
- Ryu, J.; Kim, S.-W.; Kang, K.; Park, C.B. Mineralization of Self-assembled Peptide Nanofibers for Rechargeable Lithium Ion Batteries. Adv. Mater. 2010, 22, 5537–5541. [Google Scholar] [CrossRef] [PubMed]
- Van den Hout, K.P.; Martin-Rapun, R.; Vekemans, J.A.; Meijer, E.W. Tuning the stacking properties of C3-symmetrical molecules by modifying a dipeptide motif. Chem. Eur. J. 2007, 13, 8111–8123. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.; Park, C.B. High-Temperature Self-Assembly of Peptides into Vertically Well-Aligned Nanowires by Aniline Vapor. Adv. Mater. 2008, 20, 3754–3758. [Google Scholar] [CrossRef]
- Görbitz, C.H. Microporous Organic Materials from Hydrophobic Dipeptides. Chem. Eur. J. 2007, 13, 1022–1031. [Google Scholar] [CrossRef] [PubMed]
- Krysmann, M.J.; Castelletto, V.; Hamley, I.W. Fibrillisation of hydrophobically modified amyloid peptide fragments in an organic solvent. Soft Matter 2007, 3, 1401–1406. [Google Scholar] [CrossRef]
- Pudakalakatti, S.M.; Chandra, K.; Thirupathi, R.; Atreya, H.S. Rapid Characterization of Molecular Diffusion by NMR Spectroscopy. Chem. Eur. J. 2014, 20, 15719–15722. [Google Scholar] [PubMed]
- Tamamis, P.; Adler-Abramovich, L.; Reches, M.; Marshall, K.; Sikorski, P.; Serpell, L.; Gazit, E.; Archontis, G. Self-assembly of phenylalanine oligopeptides: Insights from experiments and simulations. Biophys. J. 2009, 96, 5020–5029. [Google Scholar] [CrossRef] [PubMed]
- Tuttle, T. Computational Approaches to Understanding the Self-assembly of Peptide-based Nanostructures. Isr. J. Chem. 2015, 55, 724–734. [Google Scholar] [CrossRef]
- Colombo, G.; Soto, P.; Gazit, E. Peptide self-assembly at the nanoscale: A challenging target for computational and experimental biotechnology. Trends Biotechnol. 2007, 25, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Tamamis, P.; Kasotakis, E.; Mitraki, A.; Archontis, G. Amyloid-Like Self-Assembly of Peptide Sequences from the Adenovirus Fiber Shaft: Insights from Molecular Dynamics Simulations. J. Phys. Chem. B 2009, 113, 15639–15647. [Google Scholar] [CrossRef] [PubMed]
- Azuri, I.; Adler-Abramovich, L.; Gazit, E.; Hod, O.; Kronik, L. Why Are Diphenylalanine-Based Peptide Nanostructures so Rigid? Insights from First Principles Calculations. J. Am. Chem. Soc. 2014, 136, 963–969. [Google Scholar] [PubMed]
- Guo, C.; Luo, Y.; Zhou, R.; Wei, G. Probing the Self-Assembly Mechanism of Diphenylalanine-Based Peptide Nanovesicles and Nanotubes. ACS Nano 2012, 6, 3907–3918. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Luo, Y.; Zhou, R.; Wei, G. Triphenylalanine peptides self-assemble into nanospheres and nanorods that are different from the nanovesicles and nanotubes formed by diphenylalanine peptides. Nanoscale 2014, 6, 2800–2811. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.M.; Northey, T.; Ryan, K.; Brooks, B.R.; Kholkin, A.L.; Rodriguez, B.J.; Buchete, N.V. Conformational dynamics and aggregation behavior of piezoelectric diphenylalanine peptides in an external electric field. Biophys. Chem. 2015, 196, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Jeon, J.; Mills, C.E.; Shell, M.S. Molecular Insights into Diphenylalanine Nanotube Assembly: All-Atom Simulations of Oligomerization. J. Phys. Chem. B 2013, 117, 3935–3943. [Google Scholar] [CrossRef] [PubMed]
- Frederix, P.W.J.M.; Ulijn, R.V.; Hunt, N.T.; Tuttle, T. Virtual Screening for Dipeptide Aggregation: Toward Predictive Tools for Peptide Self-Assembly. J. Phys. Chem. Lett. 2011, 2, 2380–2384. [Google Scholar] [CrossRef] [PubMed]
- Frederix, P.W.J.M.; Scott, G.G.; Abul-Haija, Y.M.; Kalafatovic, D.; Pappas, C.G.; Javid, N.; Hunt, N.T.; Ulijn, R.V.; Tuttle, T. Exploring the sequence space for (tri-)peptide self-assembly to design and discover new hydrogels. Nat. Chem. 2015, 7, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Marchesan, S.; Waddington, L.; Easton, C.D.; Winkler, D.A.; Goodall, L.; Forsythe, J.; Hartley, P.G. Unzipping the role of chirality in nanoscale self-assembly of tripeptide hydrogels. Nanoscale 2012, 4, 6752–6760. [Google Scholar] [CrossRef] [PubMed]
- Yuran, S.; Razvag, Y.; Reches, M. Coassembly of aromatic dipeptides into biomolecular necklaces. ACS Nano 2012, 6, 9559–9566. [Google Scholar] [CrossRef] [PubMed]
- Marchesan, S.; Easton, C.D.; Styan, K.E.; Waddington, L.J.; Kushkaki, F.; Goodall, L.; McLean, K.M.; Forsythe, J.S.; Hartley, P.G. Chirality effects at each amino acid position on tripeptide self-assembly into hydrogel biomaterials. Nanoscale 2014, 6, 5172–5180. [Google Scholar] [CrossRef] [PubMed]
- Reches, M.; Gazit, E. Controlled patterning of aligned self-assembled peptide nanotubes. Nat. Nanotechnol. 2006, 1, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Challa, S.R.; Medforth, C.J.; Qiu, Y.; Watt, R.K.; Pena, D.; Miller, J.E.; Swol, F.V.; Shelnutt, J.A. Synthesis of peptide-nanotube platinum-nanoparticle composites. Chem. Commun. 2004, 1044–1045. [Google Scholar] [CrossRef] [PubMed]
- Yuran, S.; Razvag, Y.; Das, P.; Reches, M. Self-assembly of azide containing dipeptides. J. Pept. Sci. 2014, 20, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Pappas, C.G.; Frederix, P.W.J.M.; Mutasa, T.; Fleming, S.; Abul-Haija, Y.M.; Kelly, S.M.; Gachagan, A.; Kalafatovic, D.; Trevino, J.; Ulijn, R.V.; et al. Alignment of nanostructured tripeptide gels by directional ultrasonication. Chem. Commun. 2015, 51, 8465–8468. [Google Scholar] [CrossRef] [PubMed]
- Bonetti, A.; Pellegrino, S.; Das, P.; Yuran, S.; Bucci, R.; Ferri, N.; Meneghetti, F.; Castellano, C.; Reches, M.; Gelmi, M.L. Dipeptide Nanotubes Containing Unnatural Fluorine-Substituted β-Diarylamino Acid and l-Alanine as Candidates for Biomedical Applications. Org. Lett. 2015, 17, 4468–4471. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, J.; Chu, L.; Zhang, Y.; Xu, H.; Kong, D.; Yang, Z.; Yang, C.; Ding, D. Self-Assembling Peptide of d-Amino Acids Boosts Selectivity and Antitumor Efficacy of 10-Hydroxycamptothecin. ACS Appl. Mater. Interfaces 2014, 6, 5558–5565. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Gao, Y.; Kuang, Y.; Shi, J.; Du, X.; Zhou, J.; Wang, H.; Yang, Z.; Xu, B. Dephosphorylation of d-Peptide Derivatives to Form Biofunctional, Supramolecular Nanofibers/Hydrogels and Their Potential Applications for Intracellular Imaging and Intratumoral Chemotherapy. J. Am. Chem. Soc. 2013, 135, 9907–9914. [Google Scholar] [CrossRef]
- Mao, L.N.; Wang, H.M.; Tan, M.; Ou, L.L.; Kong, D.L.; Yang, Z.M. Conjugation of two complementary anti-cancer drugs confers molecular hydrogels as a co-delivery system. Chem. Commun. 2012, 48, 395–397. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Kuang, Y.; Gao, Y.; Du, X.; Shi, J.; Xu, B. d-amino acids boost the selectivity and confer supramolecular hydrogels of a nonsteroidal anti-inflammatory drug (NSAID). J. Am. Chem. Soc. 2013, 135, 542–545. [Google Scholar] [CrossRef] [PubMed]
- Liang, G.; Yang, Z.; Zhang, R.; Li, L.; Fan, Y.; Kuang, Y.; Gao, Y.; Wang, T.; Lu, W.W.; Xu, B. Supramolecular Hydrogel of a d-Amino Acid Dipeptide for Controlled Drug Release in Vivo. Langmuir 2009, 25, 8419–8422. [Google Scholar] [CrossRef] [PubMed]
- Marchesan, S.; Qu, Y.; Waddington, L.J.; Easton, C.D.; Glattauer, V.; Lithgow, T.J.; McLean, K.M.; Forsythe, J.S.; Hartley, P.G. Self-assembly of ciprofloxacin and a tripeptide into an antimicrobial nanostructured hydrogel. Biomaterials 2013, 34, 3678–3687. [Google Scholar] [CrossRef] [PubMed]
- Marchesan, S.; Waddington, L.; Easton, C.D.; Kushkaki, F.; McLean, K.M.; Forsythe, J.S.; Hartley, P.G. Tripeptide Self-Assembled Hydrogels: Soft Nanomaterials for Biological Applications. BioNanoScience 2013, 3, 21–29. [Google Scholar] [CrossRef]
- Johnson, E.K.; Adams, D.J.; Cameron, P.J. Peptide based low molecular weight gelators. J. Mater. Chem. 2011, 21, 2024–2027. [Google Scholar] [CrossRef]
- Dasgupta, A.; Mondal, J.H.; Das, D. Peptide hydrogels. RSC Adv. 2013, 3, 9117–9149. [Google Scholar] [CrossRef]
- Martin, A.D.; Robinson, A.B.; Mason, A.F.; Wojciechowski, J.P.; Thordarson, P. Exceptionally strong hydrogels through self-assembly of an indole-capped dipeptide. Chem. Commun. 2014, 50, 15541–15544. [Google Scholar] [CrossRef] [PubMed]
- Raeburn, J.; Zamith Cardoso, A.; Adams, D.J. The importance of the self-assembly process to control mechanical properties of low molecular weight hydrogels. Chem. Soc. Rev. 2013, 42, 5143–5156. [Google Scholar] [CrossRef] [PubMed]
- Jayawarna, V.; Ali, M.; Jowitt, T.A.; Miller, A.F.; Saiani, A.; Gough, J.E.; Ulijn, R.V. Nanostructured Hydrogels for Three-Dimensional Cell Culture Through Self-Assembly of Fluorenylmethoxycarbonyl-Dipeptides. Adv. Mater. 2006, 18, 611–614. [Google Scholar] [CrossRef]
- Zhou, M.; Smith, A.M.; Das, A.K.; Hodson, N.W.; Collins, R.F.; Ulijn, R.V.; Gough, J.E. Self-assembled peptide-based hydrogels as scaffolds for anchorage-dependent cells. Biomaterials 2009, 30, 2523–2530. [Google Scholar] [CrossRef] [PubMed]
- Kuang, Y.; Shi, J.; Li, J.; Yuan, D.; Alberti, K.A.; Xu, Q.; Xu, B. Pericellular hydrogel/nanonets inhibit cancer cells. Angew. Chem. Int. Ed. 2014, 53, 8104–8107. [Google Scholar] [CrossRef] [PubMed]
- Das, P.; Yuran, S.; Yan, J.; Lee, P.S.; Reches, M. Sticky tubes and magnetic hydrogels co-assembled by a short peptide and melanin-like nanoparticles. Chem. Commun. 2015, 51, 5432–5435. [Google Scholar] [CrossRef] [PubMed]
- Adler-Abramovich, L.; Reches, M.; Sedman, V.L.; Allen, S.; Tendler, S.J.; Gazit, E. Thermal and chemical stability of diphenylalanine peptide nanotubes: Implications for nanotechnological applications. Langmuir 2006, 22, 1313–1320. [Google Scholar] [CrossRef] [PubMed]
- Yemini, M.; Reches, M.; Gazit, E.; Rishpon, J. Peptide nanotube-modified electrodes for enzyme-biosensor applications. Anal. Chem. 2005, 77, 5155–5159. [Google Scholar] [CrossRef] [PubMed]
- Ikezoe, Y.; Washino, G.; Uemura, T.; Kitagawa, S.; Matsui, H. New Autonomous Motors of Metal-Organic Framework (MOF) Powered by Reorganization of Self-Assembled Peptides at interfaces. Nat. Mater. 2012, 11, 1081–1085. [Google Scholar] [PubMed]
- Gan, Z.; Wu, X.; Zhu, X.; Shen, J. Light-Induced Ferroelectricity in Bioinspired Self-Assembled Diphenylalanine Nanotubes/Microtubes. Angew. Chem. Int. Ed. 2013, 52, 2055–2059. [Google Scholar] [CrossRef] [PubMed]
- Ryan, K.; Beirne, J.; Redmond, G.; Kilpatrick, J.I.; Guyonnet, J.; Buchete, N.V.; Kholkin, A.L.; Rodriguez, B.J. Nanoscale Piezoelectric Properties of Self-Assembled Fmoc-FF Peptide Fibrous Networks. ACS Appl. Mater. Interfaces 2015, 7, 12702–12707. [Google Scholar] [CrossRef] [PubMed]
- Handelman, A.; Lavrov, S.; Kudryavtsev, A.; Khatchatouriants, A.; Rosenberg, Y.; Mishina, E.; Rosenman, G. Nonlinear Optical Bioinspired Peptide Nanostructures. Adv. Opt. Mater. 2013, 1, 875–884. [Google Scholar] [CrossRef]
- Ryu, J.; Lim, S.Y.; Park, C.B. Photoluminescent Peptide Nanotubes. Adv. Mater. 2009, 21, 1577–1581. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds dLeu-Phe-Phe, dVal-Phe-Phe, dPhe-Phe-Val, Val-dPhe-dPhe, Phe-dPhe-dVal and other isomers are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marchesan, S.; Vargiu, A.V.; Styan, K.E. The Phe-Phe Motif for Peptide Self-Assembly in Nanomedicine. Molecules 2015, 20, 19775-19788. https://doi.org/10.3390/molecules201119658
Marchesan S, Vargiu AV, Styan KE. The Phe-Phe Motif for Peptide Self-Assembly in Nanomedicine. Molecules. 2015; 20(11):19775-19788. https://doi.org/10.3390/molecules201119658
Chicago/Turabian StyleMarchesan, Silvia, Attilio V. Vargiu, and Katie E. Styan. 2015. "The Phe-Phe Motif for Peptide Self-Assembly in Nanomedicine" Molecules 20, no. 11: 19775-19788. https://doi.org/10.3390/molecules201119658
APA StyleMarchesan, S., Vargiu, A. V., & Styan, K. E. (2015). The Phe-Phe Motif for Peptide Self-Assembly in Nanomedicine. Molecules, 20(11), 19775-19788. https://doi.org/10.3390/molecules201119658








