Antimicrobial Activity of Rhoeo discolor Phenolic Rich Extracts Determined by Flow Cytometry
Abstract
:1. Introduction
2. Results and Discussion
2.1. Agar Diffusion Assays

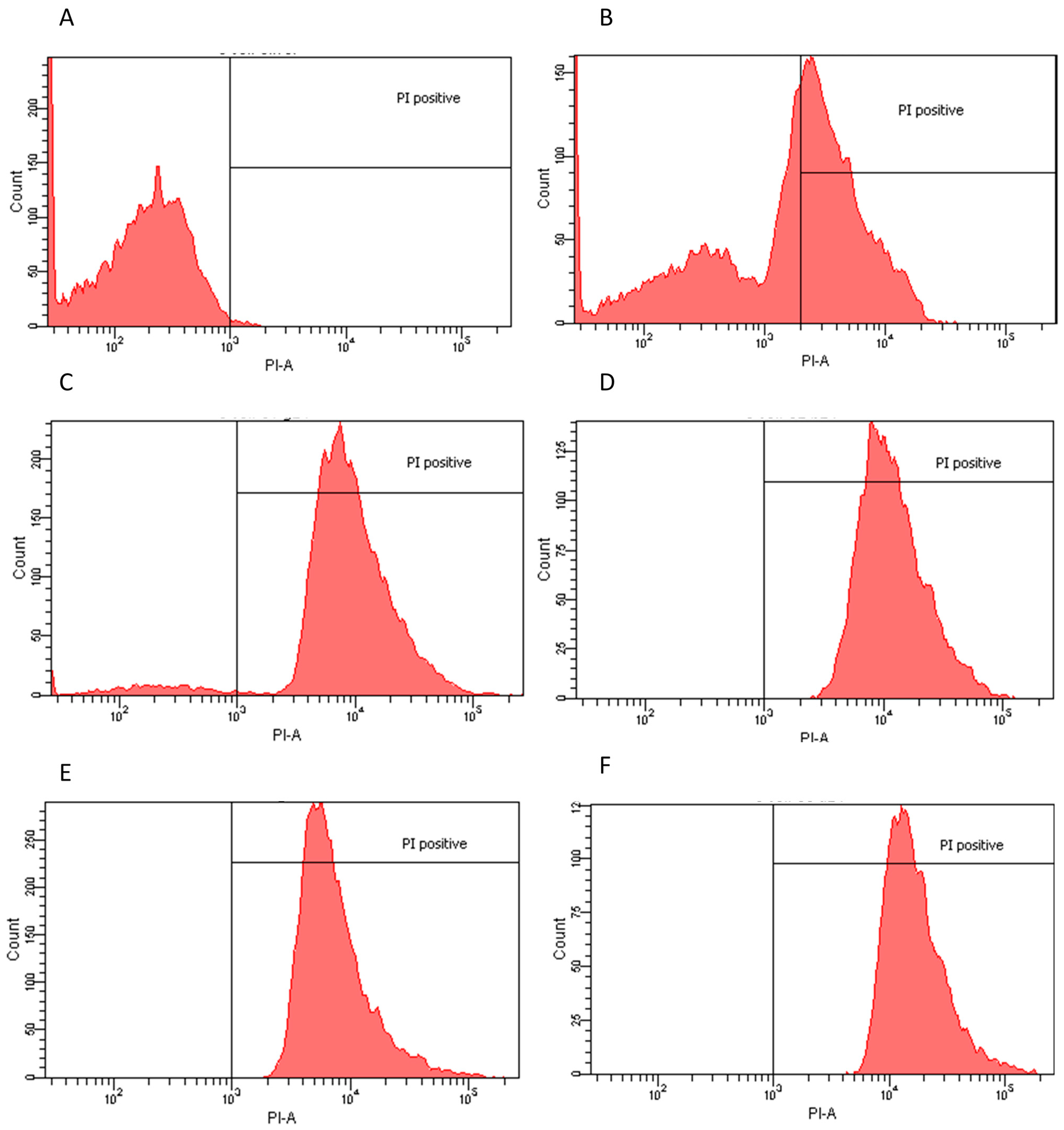
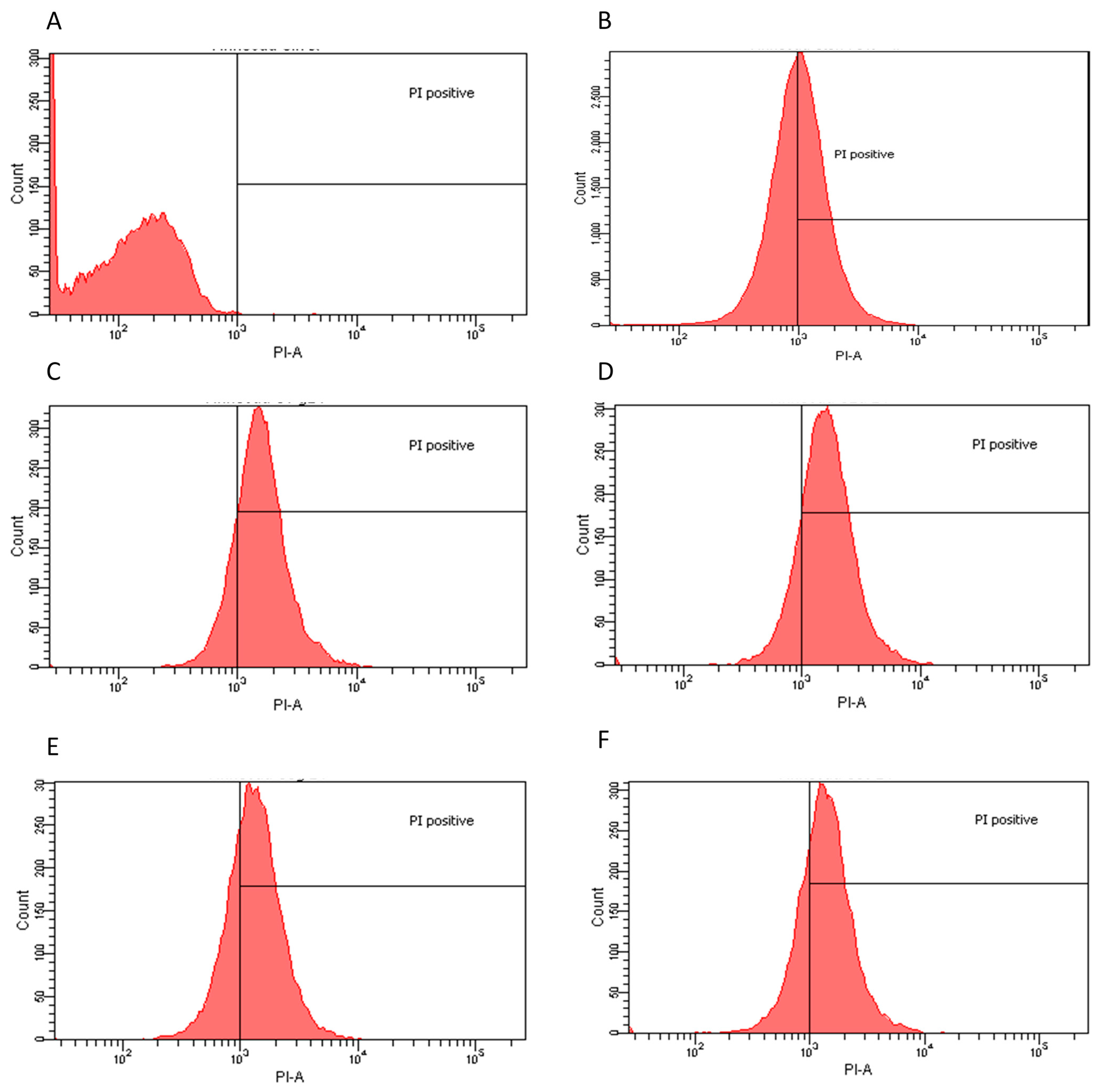

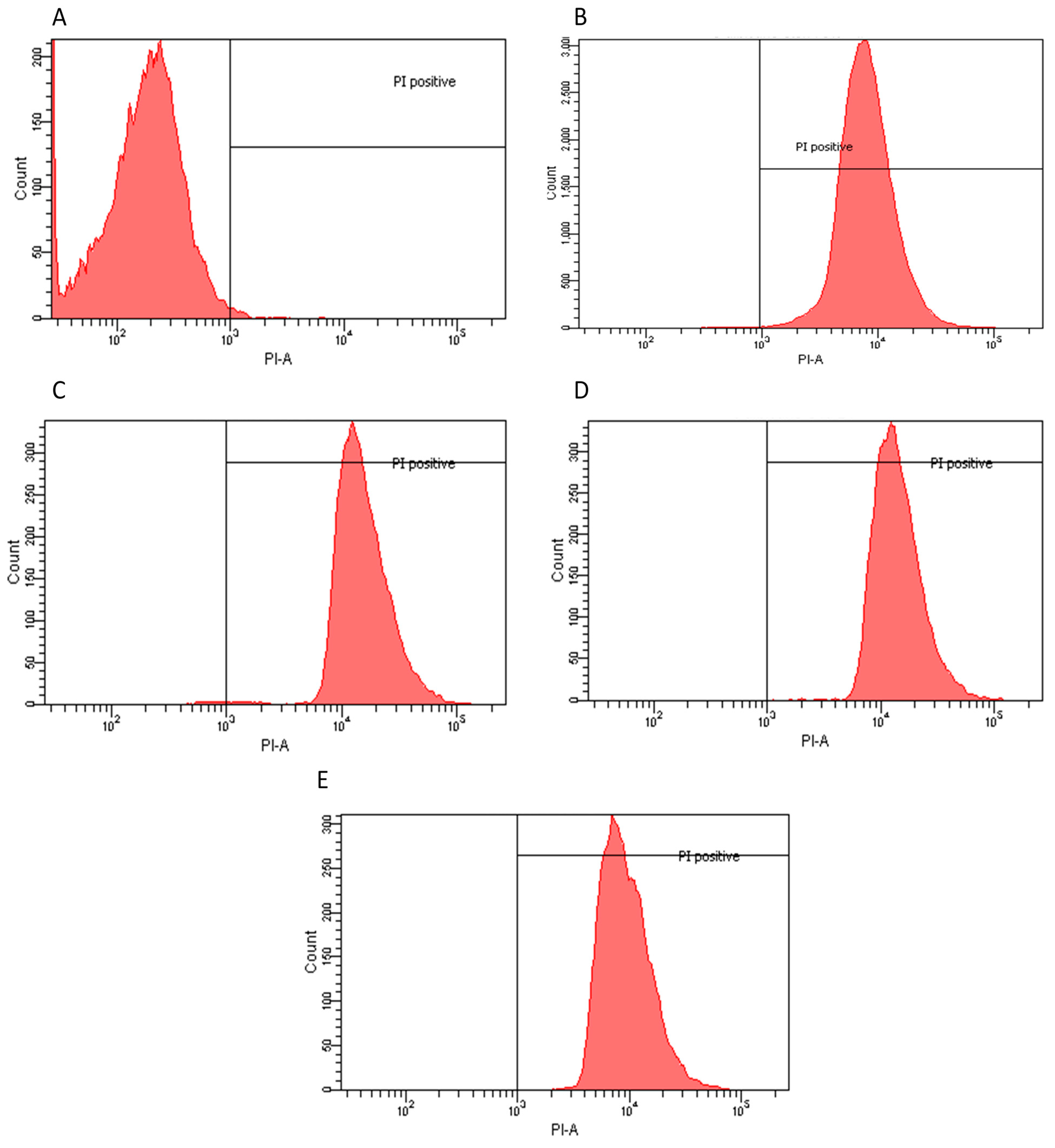
2.2. Flow Cytometry Assay
| Extract | Dose | Effect | PI Positive |
|---|---|---|---|
| E. coli | |||
| Ethanol | 4 μg/mL | Bactericidal | 99.90% |
| Water non-dried leaf non-boiled | 1.5 μg/mL | Bactericidal | 99.90% |
| Ethyl Acetate | 1 μg/mL | Bactericidal | 99.70% |
| Water dry leaf non-boiled | 4 μg/mL | Bactericidal | 94.30% |
| L. innocua | |||
| Water dry leaf non-boiled | 4 μg/mL | Bacteriostatic | 82.50% |
| Water dry leaf boiled | 2 μg/mL | Bacteriostatic | 74.40% |
| Water non-dried leaf non-boiled | 2.5 μg/mL | Bacteriostatic | 83.30% |
| Ethanol | 4 μg/mL | Bacteriostatic | 71.50% |
| S. mutans | |||
| Petroleum Ether | 1 μg/mL | Bacteriostatic | 58.40% |
| Chloroform | 3.5 μg/mL | Bactericidal | 99.70% |
| Acetone | 4 μg/mL | Bactericidal | 99.50% |
| Water dry leaf non-boiled | 1 μg/mL | Bactericidal | 98.50% |
| C. albicans | |||
| Water non-dry leaf non-boiled | 1.5 μg/mL | Fungicide | 99.80% |
| Methanol | 1 μg/mL | Fungicide | 99.90% |
| Water dry leaf non-boiled | 1 μg/mL | Fungicide | 99.40% |
2.3. Total Phenolic Count
| Extract | µg GAE/mg |
|---|---|
| Extract #1: H2O dry leaf, non-boiled | 8.5 ± 3.7 |
| Extract #2: H2O non-dried leaf, non-boiled | 7.5 ± 1.5 |
| Extract #3: H2O dry leaf, boiled | 16.9 ± 3.7 |
| Extract #4: Methanol (MeOH) | 1.5 ± 0.7 |
| Extract #5: Ethanol (EtOH) | 1.6 ± 0.2 |
| Extract #6: Ethyl Acetate (EtAc) | 9.4 ± 8.8 |
| Extract #7: Acetone (Ac) | 5.5 ± 1.1 |
| Extract #8: Petroleum Ether (PtEt) | 1.4 ± 2.4 |
| Extract #9: Chloroform (CHCl3) | 1.7 ± 2.5 |
| Extract #10: Hexane (Hx) | 0.7 ± 0.0 |
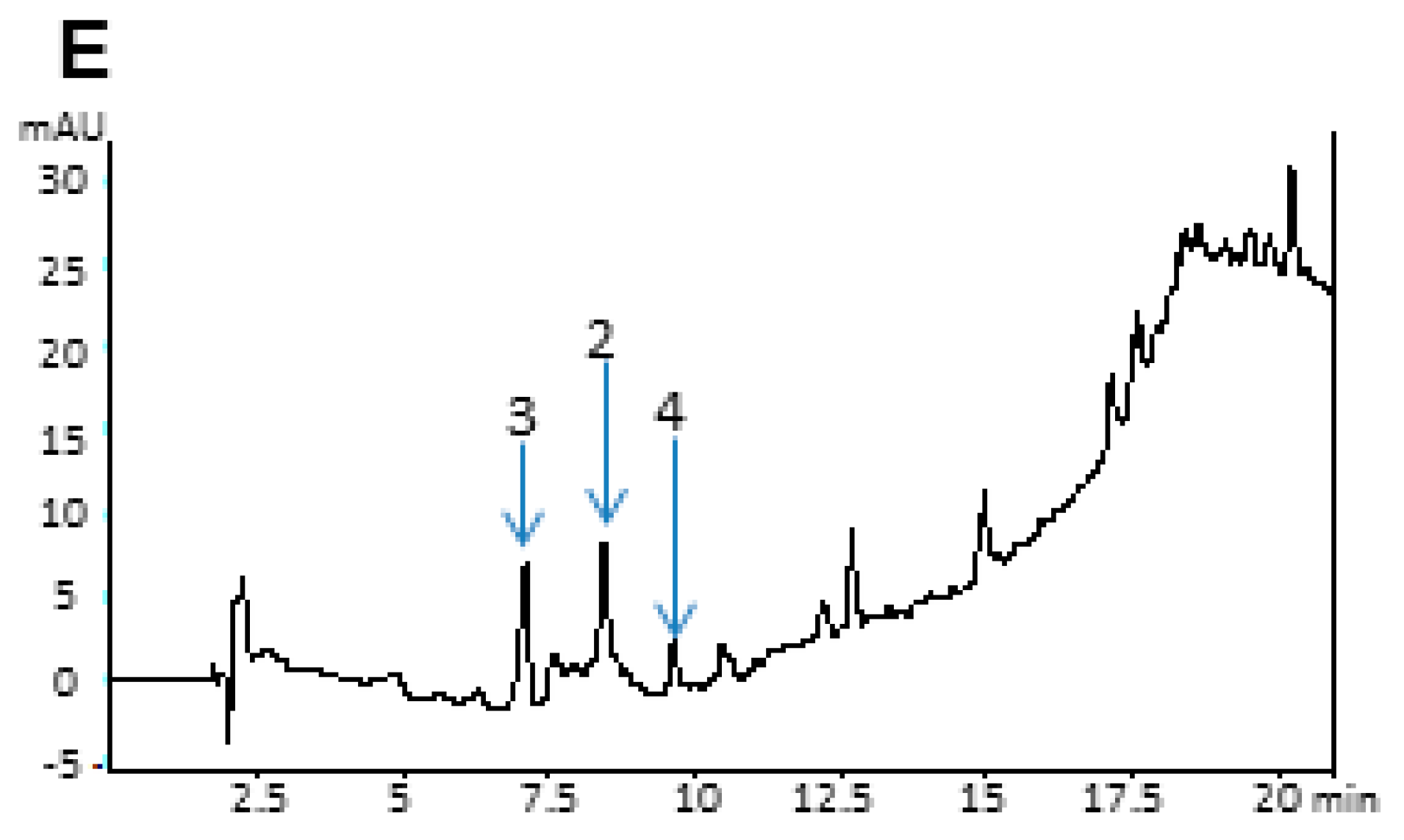
2.4. Analytical Data
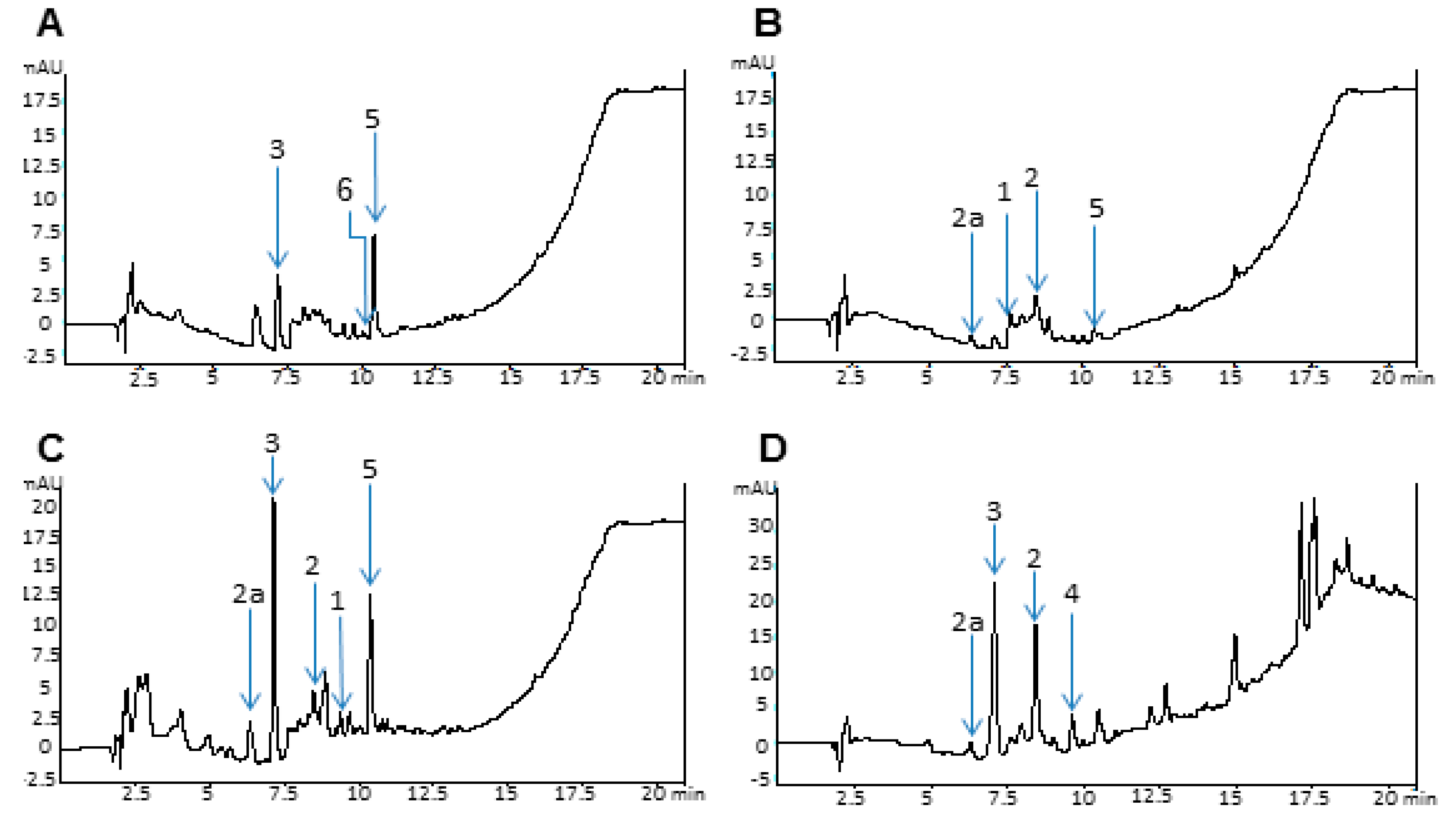
3. Experimental Section
3.1. Plant Material
3.2. Aqueous Extract Preparations
3.3. Organic Solvent Extract Preparation
3.4. Dose Preparation
3.5. Organisms and Growth Conditions
3.6. Inoculum Preparation
3.7. Agar-Disc Diffusion Assay
3.8. Total Phenolic Count
3.9. High-Performance Liquid Chromatography (HPLC) Analysis
3.10. High-Performance Liquid Chromatograpjy Coupled with Mass Spectrometry-Time of flight (HPLC-MS-TOF) Analysis
3.11. Flow Cytometry Analysis
3.12. Statistical Analyses
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rosales-Reyes, T.; de la Garza, M.; Arias-Castro, C.; Rodriguez-Mendiola, M.; Fattel-Fazenda, S.; Arce-Popoca, E.; Hernandez-Garcia, S.; Villa-Trevino, S. Aqueous crude extract of Rhoeo discolor, a mexican medicinal plant, decreases the formation of liver preneoplastic foci in rats. J. Ethnopharmacol. 2008, 115, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Avila, M.; Arriaga-Alba, M.; de la Garza, M.; del Carmen, H.M.; Dominguez-Ortiz, M.A.; Fattel-Fazenda, S.; Villa-Trevino, S. Antigenotoxic, antimutagenic and ros scavenging activities of a Rhoeo discolor ethanolic crude extract. Toxicol. In Vitro 2003, 17, 77–83. [Google Scholar] [CrossRef]
- Giovannini, P.; Reyes-Garcia, V.; Waldstein, A.; Heinrich, M. Do pharmaceuticals displace local knowledge and use of medicinal plants? Estimates from a cross-sectional study in a rural indigenous community, mexico. Soc. Sci. Med. 2011, 72, 928–936. [Google Scholar] [CrossRef] [PubMed]
- Mosa, A.I.; Emara, A.A.; Yousef, J.M.; Saddiq, A.A. Novel transition metal complexes of 4-hydroxy-coumarin-3-thiocarbohydrazone: Pharmacodynamic of Co(Iii) on rats and antimicrobial activity. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2011, 81, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Wattenberg, L.W. Inhibition of carcinogenesis by minor dietary constituents. Cancer Res. 1992, 52, 2085s–2091s. [Google Scholar] [PubMed]
- Martin, C.; Zhang, Y.; Tonelli, C.; Petroni, K. Plants, diet, and health. Annu. Rev. Plant Biol. 2013, 64, 19–46. [Google Scholar] [CrossRef] [PubMed]
- Idaka, E.; Ogawa, T.; Kondo, T.; Goto, T. Isolation of highly acylated anthocyanins from commelinaceae plants, zebrina pendula, rhoeo spathacea and setcreasea purpurea. Agric. Biol. Chem. 1987, 51, 2215–2220. [Google Scholar] [CrossRef]
- Arriaga-Alba, M.; Blasco, J.L.; Ruiz-Perez, N.J.; Sanchez-Navarrete, J.; Rivera-Sanchez, R.; Gonzalez-Avila, M. Antimutagenicity mechanisms of the Rhoeo discolor ethanolic extract. Exp. Toxicol. Pathol. 2011, 63, 243–248. [Google Scholar] [PubMed]
- Arenas-Hernandez, M.M.; Martinez-Laguna, Y.; Torres, A.G. Clinical implications of enteroadherent Escherichia coli. Curr. Gastroenterol. Rep. 2012, 14, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Hoiby, N.; Frederiksen, B.; Pressler, T. Eradication of early Pseudomonas aeruginosa infection. J. Cyst. Fibros. 2005, 4, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Siegman-Igra, Y.; Levin, R.; Weinberger, M.; Golan, Y.; Schwartz, D.; Samra, Z.; Konigsberger, H.; Yinnon, A.; Rahav, G.; Keller, N.; et al. Listeria monocytogenes infection in israel and review of cases worldwide. Emerg. Infect. Dis. 2002, 8, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, P.J. Ignorance about listeria. BMJ 1989, 299, 276–277. [Google Scholar] [CrossRef]
- Forssten, S.D.; Bjorklund, M.; Ouwehand, A.C. Streptococcus mutans, caries and simulation models. Nutrients 2010, 2, 290–298. [Google Scholar] [CrossRef]
- Moyes, D.L.; Naglik, J.R. Mucosal immunity and Candida albicans infection. Clin. Dev. Immunol. 2011, 2011. [Google Scholar] [CrossRef]
- Tan, J.B.L.; Yap, W.J.; Tan, S.Y.; Lim, Y.Y.; Lee, S.M. Antioxidant content, antioxidant activity, and antibacterial activity of five plants from the commelinaceae family. Antioxidants 2014, 3, 758–769. [Google Scholar] [CrossRef]
- Tan, J.B.L.; Lim, Y.Y.; Lee, S.M. Antioxidant and antibacterial activity of rhoeo spathacea (swartz) stearn leaves. J. Food Sci. Technol. 2013, 52, 2394–2400. [Google Scholar] [CrossRef] [PubMed]
- Praveen, N.C.; Rajesh, A.; Madan, M.; Chaurasia, V.R.; Hiremath, N.V.; Sharma, A.M. In vitro evaluation of antibacterial efficacy of pineapple extract (bromelain) on periodontal pathogens. J. Int. Oral Health 2014, 6, 96–98. [Google Scholar] [PubMed]
- Garcia-Garcia, R.; Lopez-Malo, A.; Palou, E. Bactericidal action of binary and ternary mixtures of carvacrol, thymol, and eugenol against Listeria innocua. J. Food Sci. 2011, 76. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Garcia, R.; Escobedo-Avellaneda, Z.; Tejada-Ortigoza, V.; Martin-Belloso, O.; Valdez-Fragoso, A.; Welti-Chanes, J. Hurdle technology applied to prickly pear beverages for inhibiting saccharomyces cerevisiae and Escherichia coli. Lett. Appl. Microbiol. 2015, 60, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Pedras, M.S.; Yaya, E.E. Plant chemical defenses: Are all constitutive antimicrobial metabolites phytoanticipins? Nat. Prod. Commun. 2015, 10, 209–218. [Google Scholar] [PubMed]
- Cushnie, T.P.; Lamb, A.J. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agents 2005, 26, 343–356. [Google Scholar] [CrossRef] [PubMed]
- Mujeeb, F.; Bajpai, P.; Pathak, N. Phytochemical evaluation, antimicrobial activity, and determination of bioactive components from leaves of aegle marmelos. Biomed. Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Tatsuzawa, F.; Saito, N.; Maeyama, K.; Yokoi, M.; Shigihara, A.; Honda, T. Triacylated anthocyanidin 3-arabinosylglucoside-7,3ʹ-diglucosides isolated from the bluish flowers of tradescantia virginiana cultivars and their distribution in the tradescantieae. 2010, 81. [Google Scholar] [CrossRef]
- Cisowska, A.; Wojnicz, D.; Hendrich, A.B. Anthocyanins as antimicrobial agents of natural plant origin. Nat. Prod. Commun. 2011, 6, 149–156. [Google Scholar] [PubMed]
- Mira, N.V.M.; Barros, R.M.C.; Schiocchet, M.A.; Noldin, J.A.; Lanfer-Marquez, U.M. Extraction, analysis and distribution of phenolic acids in pigmented and non-pigmented genotypes of rice (Oryza sativa L.). Ciênc. Tecnol. Aliment. 2008, 28, 994–1002. [Google Scholar] [CrossRef]
- Gardjeva, P.A.; Dimitrova, S.Z.; Kostadinov, I.D.; Murdjeva, M.A.; Peyche, L.P.; Lukanov, L.K.; Stanimirova, I.V.; Alexandrov, A.S. A study of chemical composition and antimicrobial activity of bulgarian propolis. Folia Med.(Plovdiv.) 2007, 49, 63–69. [Google Scholar] [PubMed]
- Mueller, U.; Sauer, T.; Weigel, I.; Pichner, R.; Pischetsrieder, M. Identification of H2O2 as a major antimicrobial component in coffee. Food Funct. 2011, 2, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Kozyra, M.; Biernasiuk, A.; Malm, A.; Chowaniec, M. Chemical compositions and antibacterial activity of extracts obtained from the inflorescences of Cirsium canum (L.) all. Nat. Prod. Res. 2015, 29, 2059–2063. [Google Scholar] [CrossRef] [PubMed]
- Popova, M.P.; Chinou, I.B.; Marekov, I.N.; Bankova, V.S. Terpenes with antimicrobial activity from cretan propolis. Phytochemistry 2009, 70, 1262–1271. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Cao, F.; Su, E.; Wu, C.; Zhao, L.; Ying, R. Improving flavonoid extraction from ginkgo biloba leaves by prefermentation processing. J. Agric. Food Chem. 2013, 61, 5783–5791. [Google Scholar] [CrossRef] [PubMed]
- Harborne, J.B.; Williams, C.A. Advances in flavonoid research since 1992. Phytochemistry 2000, 55, 481–504. [Google Scholar] [CrossRef]
- Tekeli, Y.; Karpuz, E.; Danahaliloglu, H.; Bucak, S.; Guzel, Y.; Erdmann, H. Phenolic composition, antioxidant capacity of salvia verticcilata and effect on multidrug resistant bacteria by flow-cytometry. Afr. J. Tradit. Complement Altern. Med. 2014, 11, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Patra, J.K.; Kim, E.S.; Oh, K.; Kim, H.J.; Kim, Y.; Baek, K.H. Antibacterial effect of crude extract and metabolites of phytolacca americana on pathogens responsible for periodontal inflammatory diseases and dental caries. BMC. Complement Altern. Med. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.B.; Lim, Y.Y. Critical analysis of current methods for assessing the in vitro antioxidant and antibacterial activity of plant extracts. Food Chem. 2015, 172, 814–822. [Google Scholar] [CrossRef] [PubMed]
- De Araujo, A.A.; Soares, L.A.; Assuncao Ferreira, M.R.; de Souza Neto, M.A.; da Silva, G.R.; de Araujo, R.F.J.; Guerra, G.C.; de Melo, M.C. Quantification of polyphenols and evaluation of antimicrobial, analgesic and anti-inflammatory activities of aqueous and acetone-water extracts of libidibia ferrea, parapiptadenia rigida and psidium guajava. J. Ethnopharmacol. 2014, 156, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Tamburini, S.; Ballarini, A.; Ferrentino, G.; Moro, A.; Foladori, P.; Spilimbergo, S.; Jousson, O. Comparison of quantitative pcr and flow cytometry as cellular viability methods to study bacterial membrane permeabilization following supercritical CO2 treatment. Microbiology 2013, 159, 1056–1066. [Google Scholar] [CrossRef] [PubMed]
- Herman, A.; Herman, A.P.; Domagalska, B.W.; Mlynarczyk, A. Essential oils and herbal extracts as antimicrobial agents in cosmetic emulsion. Indian J. Microbiol. 2013, 53, 232–237. [Google Scholar] [PubMed]
- Hofling, J.F.; Anibal, P.C.; Obando-Pereda, G.A.; Peixoto, I.A.; Furletti, V.F.; Foglio, M.A.; Goncalves, R.B. Antimicrobial potential of some plant extracts against candida species. Braz. J. Biol. 2010, 70, 1065–1068. [Google Scholar] [CrossRef] [PubMed]
- Efstratiou, E.; Hussain, A.I.; Nigam, P.S.; Moore, J.E.; Ayub, M.A.; Rao, J.R. Antimicrobial activity of Calendula officinalis petal extracts against fungi, as well as gram-negative and gram-positive clinical pathogens. Complement Ther. Clin. Pract. 2012, 18, 173–176. [Google Scholar] [CrossRef] [PubMed]
- Agusti, G.; Fittipaldi, M.; Morato, J.; Codony, F. Viable quantitative pcr for assessing the response of Candida albicans to antifungal treatment. Appl. Microbiol. Biotechnol. 2013, 97, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Assaf, A.M.; Haddadin, R.N.; Aldouri, N.A.; Alabbassi, R.; Mashallah, S.; Mohammad, M.; Bustanji, Y. Anti-cancer, anti-inflammatory and anti-microbial activities of plant extracts used against hematological tumors in traditional medicine of jordan. J. Ethnopharmacol. 2013, 145, 728–736. [Google Scholar] [CrossRef] [PubMed]
- Urias-Lugo, D.A.; Heredia, J.B.; Muy-Rangel, M.D.; Valdez-Torres, J.B.; Serna-SaldÃvar, S.O.; Gutiérrez-Uribe, J.A. Anthocyanins and phenolic acids of hybrid and native blue maize (Zea mays L.) extracts and their antiproliferative activity in mammary (MCF7), liver (HepG2), colon (Caco2 and HT29) and prostate (PC3) cancer cells. Plant Foods Hum. Nutr. 2015, 70, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Zare, K.; Movafeghi, A.; Mohammadi, S.A.; Asnaashari, S.; Nazemiyeh, H. New phenolics from Linum mucronatum subsp. Orientale. Bioimpacts. 2014, 4, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Berney, M.; Hammes, F.; Bosshard, F.; Weilenmann, H.U.; Egli, T. Assessment and interpretation of bacterial viability by using the live/dead baclight kit in combination with flow cytometry. Appl. Environ. Microbiol. 2007, 73, 3283–3290. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the extracts are not currently available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Varela, R.; García-García, R.M.; Barba-Dávila, B.A.; Fajardo-Ramírez, O.R.; Serna-Saldívar, S.O.; Cardineau, G.A. Antimicrobial Activity of Rhoeo discolor Phenolic Rich Extracts Determined by Flow Cytometry. Molecules 2015, 20, 18685-18703. https://doi.org/10.3390/molecules201018685
García-Varela R, García-García RM, Barba-Dávila BA, Fajardo-Ramírez OR, Serna-Saldívar SO, Cardineau GA. Antimicrobial Activity of Rhoeo discolor Phenolic Rich Extracts Determined by Flow Cytometry. Molecules. 2015; 20(10):18685-18703. https://doi.org/10.3390/molecules201018685
Chicago/Turabian StyleGarcía-Varela, Rebeca, Rebeca M. García-García, Bertha A. Barba-Dávila, Oscar R. Fajardo-Ramírez, Sergio O. Serna-Saldívar, and Guy A. Cardineau. 2015. "Antimicrobial Activity of Rhoeo discolor Phenolic Rich Extracts Determined by Flow Cytometry" Molecules 20, no. 10: 18685-18703. https://doi.org/10.3390/molecules201018685
APA StyleGarcía-Varela, R., García-García, R. M., Barba-Dávila, B. A., Fajardo-Ramírez, O. R., Serna-Saldívar, S. O., & Cardineau, G. A. (2015). Antimicrobial Activity of Rhoeo discolor Phenolic Rich Extracts Determined by Flow Cytometry. Molecules, 20(10), 18685-18703. https://doi.org/10.3390/molecules201018685






