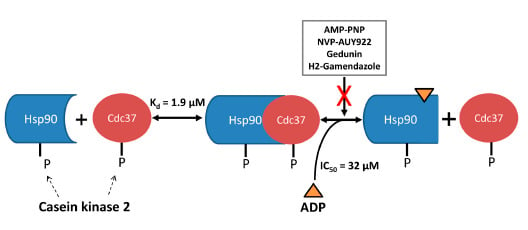
Stability of the Human Hsp90-p50Cdc37 Chaperone Complex against Nucleotides and Hsp90 Inhibitors, and the Influence of Phosphorylation by Casein Kinase 2
Abstract
:1. Introduction
2. Results and Discussion
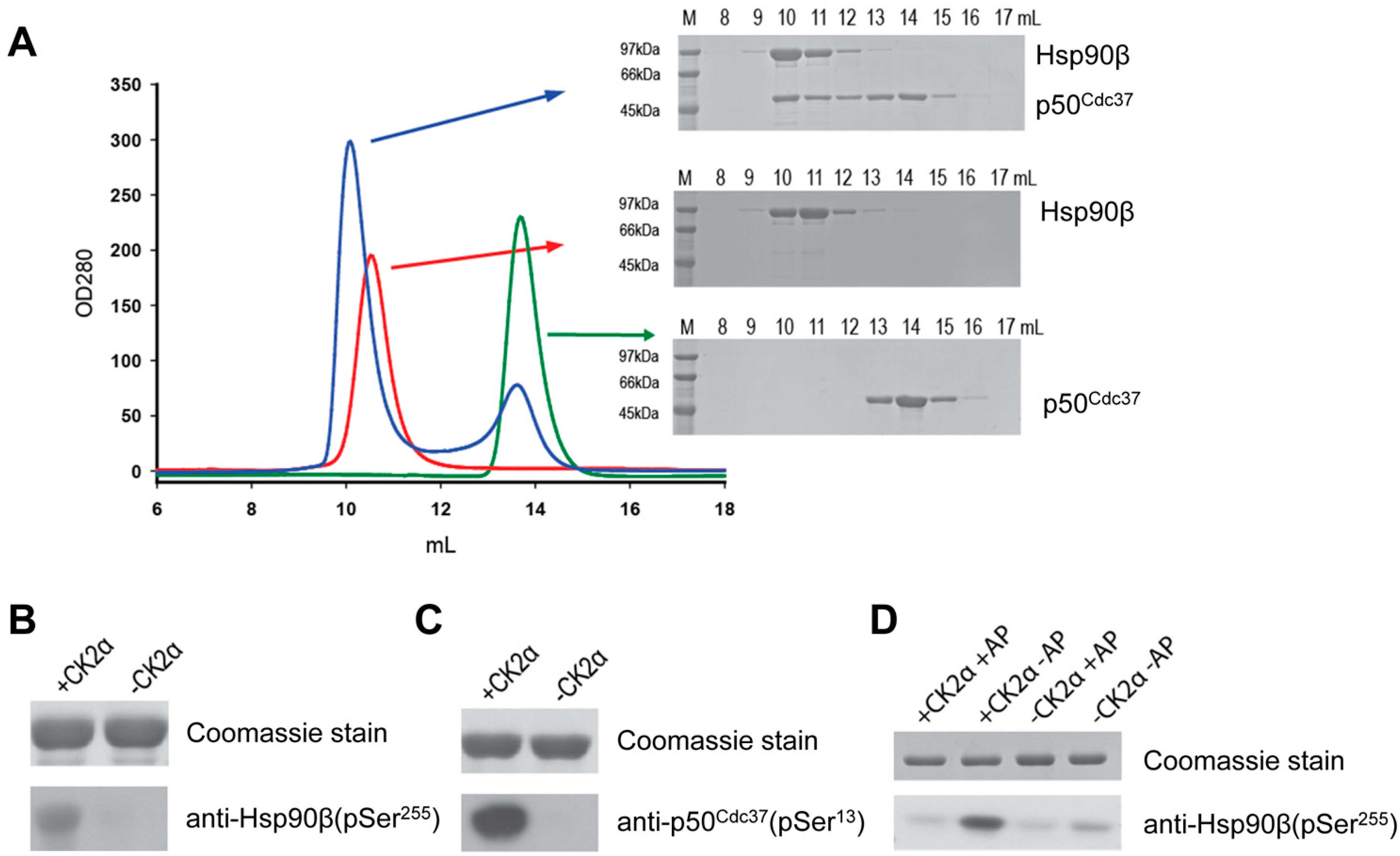
2.1. In Vitro Reconstitution of the Human Hsp90β-p50Cdc37 Complex and Phosphorylation by CK2
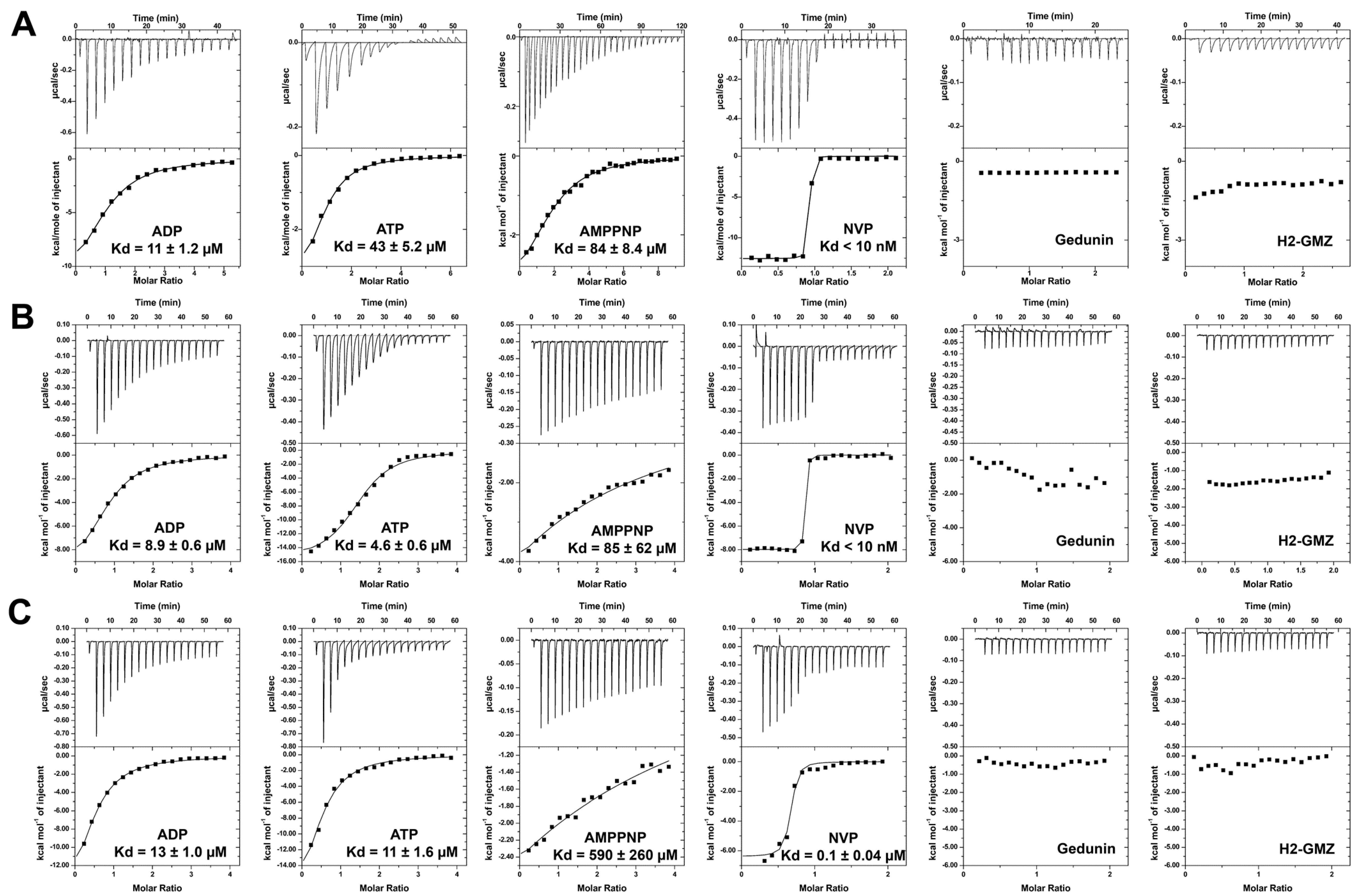
2.2. Effect of Phosphorylation on the Interaction of Hsp90β with Nucleotides and Inhibitors
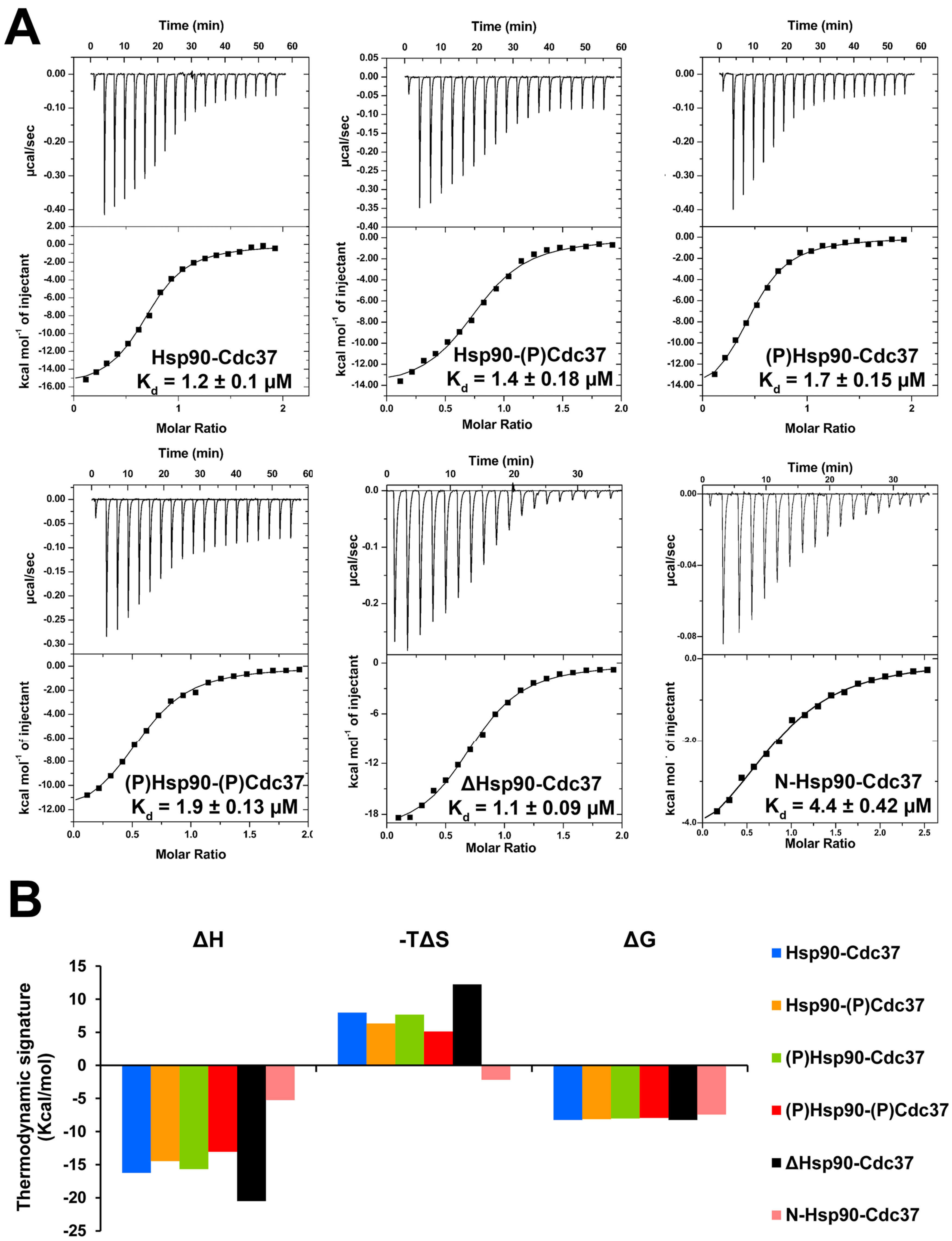
| Binding Partner/Ligand | Hsp90 | Observed Kd (µM) | Reported Kd (µM) | Reference |
|---|---|---|---|---|
| p50Cdc37 | yHsp82-FL | n/d | 1.46 | [30] |
| hΔHsp90β | 1.1 | n/a | ||
| hHsp90β-N | 4.4 | n/a | ||
| hHsp90β-FL | 1.2 | n/a | ||
| h(p)Hsp90β-FL | 1.7 | n/a | ||
| (p)p50Cdc37 | hHsp90β-FL | 5.1 | n/a | |
| h(p)Hsp90β-FL | 8.6 | n/a | ||
| ADP | hΔHsp90β | 11 | n/a | |
| hHsp90β-FL | 8.9 | 7.2 | [31] | |
| h(p)Hsp90β-FL | 13 | n/a | ||
| ATP | hΔHsp90β | 43 | n/a | |
| hHsp90β-FL | 4.6 | 240 | [31] | |
| h(p)Hsp90β-FL | 11 | n/a | ||
| AMP-PNP | hΔHsp90β | 84 | n/a | |
| hHsp90β-FL | 85 | 148 | [31] | |
| h(p)Hsp90β-FL | 590 | n/a | ||
| NVP-AUY922 | hΔHsp90β | <0.01 | n/a | |
| hHsp90β-FL | <0.01 | 0.0017 | [32] | |
| h(p)Hsp90β-FL | 0.1 | n/a | ||
| 17-DMAG | hHsp90β-FL | n/d | 0.35 | [33] |
| hΔHsp90β | 0.79 | n/a | ||
| Radicicol | yHsp82-FL | n/d | 0.019 | [34] |
| hΔHsp90β | <0.01 | n/a |
2.3. Evaluation of Small Molecules as PPI Inhibitors of the Hsp90β-p50Cdc37 Complex
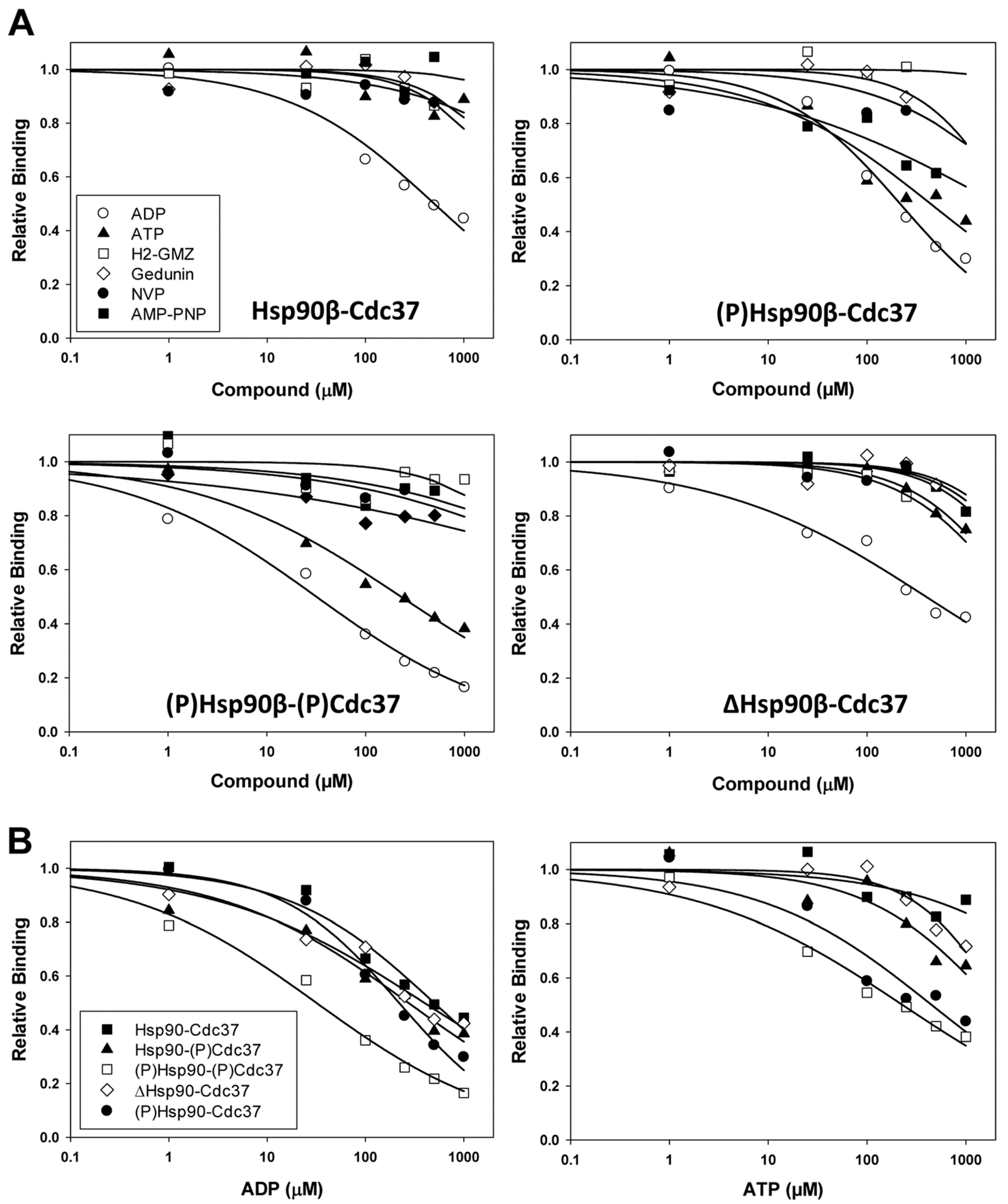
| Complex | IC50 (µM) | ||||||
|---|---|---|---|---|---|---|---|
| ADP | ATP | AMP-PNP | H2-GMZ | H2-GMZ | Gedunin | NVP | |
| Hsp90-p50Cdc37 | 500 ± 94 | >1000 | >1000 | >1000 | >1000 | >1000 | >1000 |
| (p)Hsp90-p50Cdc37 | 220 ± 24 | 450 ± 150 | >1000 | >1000 | >1000 | >1000 | >1000 |
| Hsp90-(p)p50Cdc37 | 270 ± 35 | >1000 | n/d | n/d | n/d | n/d | n/d |
| (p)Hsp90-(p)p50Cdc37 | 32 ± 5.6 | 230 ± 40 | >1000 | >1000 | >1000 | >1000 | >1000 |
| ΔHsp90-p50Cdc37 | 400 ± 81 | >1000 | >1000 | >1000 | >1000 | >1000 | >1000 |
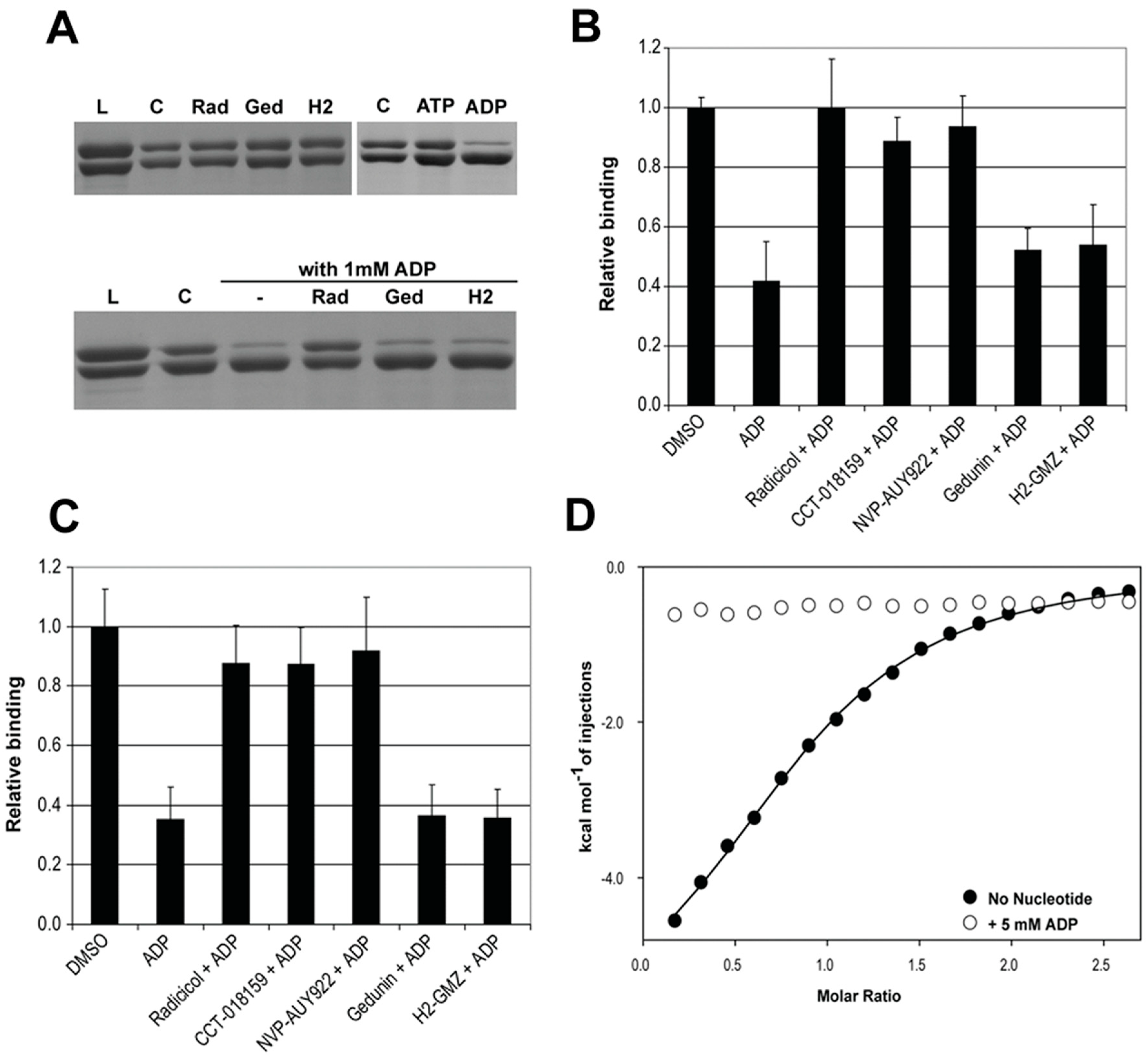
2.4. ADP Exerts Inhibitory Activity through Binding to the ATP Site in the N-Terminal Hsp90 Domain
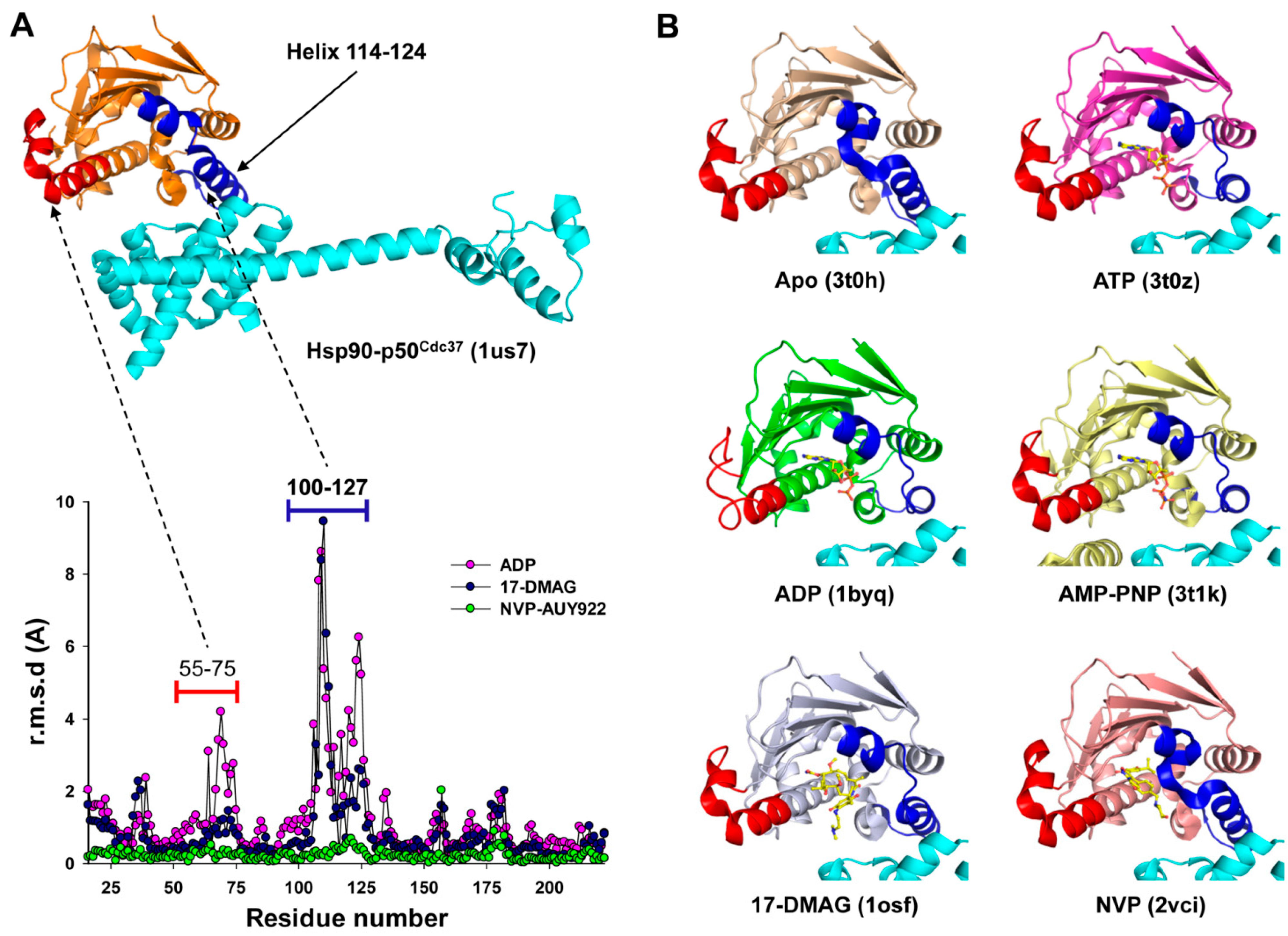
2.5. Possible Structural Basis for the PPI Inhibitory Activity of ADP
3. Experimental Section
3.1. Materials
3.2. Cloning and Expression
3.3. Protein Purification
3.4. GST Pull-Down
3.5. In Vitro Phosphorylation
3.6. SDS-PAGE and Western Blot Analysis
3.7. ELISA Microtiter Plate Assay
3.8. Isothermal Titration Calorimetry (ITC)
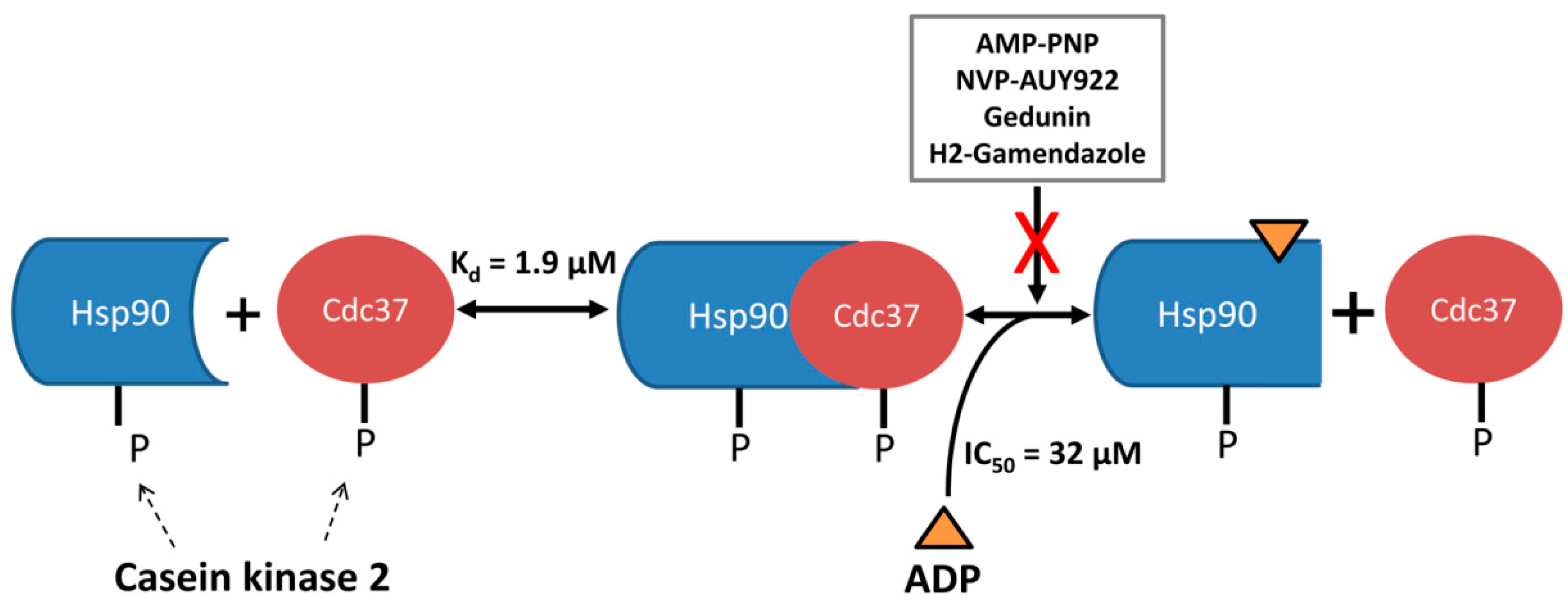
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Da Silva, V.C.; Ramos, C.H. The network interaction of the human cytosolic 90 kDa heat shock protein Hsp90: A target for cancer therapeutics. J. Proteomics 2012, 75, 2790–2802. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.L. Evolution and function of diverse Hsp90 homologs and cochaperone proteins. Biochim. Biophys. Acta Mol. Cell Res. 2012, 1823, 607–613. [Google Scholar] [CrossRef]
- Neckers, L. Heat shock protein 90: The cancer chaperone. J. Biosci. 2007, 32, 517–530. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.R.; Workman, P. Targeting CDC37: An alternative, kinase-directed strategy for disruption of oncogenic chaperoning. Cell Cycle 2009, 8, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Carbonero, R.; Carnero, A.; Paz-Ares, L. Inhibition of HSP90 molecular chaperones: Moving into the clinic. Lancet Oncol. 2013, 14, e358–369. [Google Scholar] [CrossRef] [PubMed]
- Dickey, C.A.; Koren, J.; Zhang, Y.J.; Xu, Y.F.; Jinwal, U.K.; Birnbaum, M.J.; Monks, B.; Sun, M.; Cheng, J.Q.; Patterson, C.; et al. Akt and CHIP coregulate tau degradation through coordinated interactions. Proc. Natl. Acad. Sci. USA 2008, 105, 3622–3627. [Google Scholar] [CrossRef] [PubMed]
- Geller, R.; Vignuzzi, M.; Andino, R.; Frydman, J. Evolutionary constraints on chaperone-mediated folding provide an antiviral approach refractory to development of drug resistance. Genes Dev. 2007, 21, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Shah, V.; Wiest, R.; Garcia-Cardena, G.; Cadelina, G.; Groszmann, R.J.; Sessa, W.C. Hsp90 regulation of endothelial nitric oxide synthase contributes to vascular control in portal hypertension. Am. J. Physiol. 1999, 277, G463–G468. [Google Scholar] [PubMed]
- Li, J.; Soroka, J.; Buchner, J. The Hsp90 chaperone machinery: Conformational dynamics and regulation by co-chaperones. Biochim. Biophys. Acta Mol. Cell Res. 2012, 1823, 624–635. [Google Scholar] [CrossRef]
- Smith, J.R.; Clarke, P.A.; de Billy, E.; Workman, P. Silencing the cochaperone CDC37 destabilizes kinase clients and sensitizes cancer cells to HSP90 inhibitors. Oncogene 2009, 28, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Legagneux, V.; Morange, M.; Bensaude, O. Heat shock increases turnover of 90 kDa heat shock protein phosphate groups in HeLa cells. FEBS Lett. 1991, 291, 359–362. [Google Scholar] [CrossRef] [PubMed]
- Lees-Miller, S.P.; Anderson, C.W. Two human 90-kDa heat shock proteins are phosphorylated in vivo at conserved serines that are phosphorylated in vitro by casein kinase II. J. Biol. Chem. 1989, 264, 2431–2437. [Google Scholar] [PubMed]
- Mollapour, M.; Tsutsumi, S.; Donnelly, A.C.; Beebe, K.; Tokita, M.J.; Lee, M.J.; Lee, S.; Morra, G.; Bourboulia, D.; Scroggins, B.T.; et al. Swe1Wee1-dependent tyrosine phosphorylation of Hsp90 regulates distinct facets of chaperone function. Mol. Cell 2010, 37, 333–343. [Google Scholar] [CrossRef] [PubMed]
- Mollapour, M.; Tsutsumi, S.; Neckers, L. Hsp90 phosphorylation, Wee1, and the cell cycle. Cell Cycle 2010, 9, 2310–2316. [Google Scholar] [CrossRef] [PubMed]
- Mollapour, M.; Neckers, L. Post-translational modifications of Hsp90 and their contributions to chaperone regulation. Biochim. Biophys. Acta Mol. Cell Res. 2012, 1823, 648–655. [Google Scholar] [CrossRef]
- Mollapour, M.; Tsutsumi, S.; Truman, A.W.; Xu, W.; Vaughan, C.K.; Beebe, K.; Konstantinova, A.; Vourganti, S.; Panaretou, B.; Piper, P.W.; et al. Threonine 22 phosphorylation attenuates Hsp90 interaction with cochaperones and affects its chaperone activity. Mol. Cell 2011, 41, 672–681. [Google Scholar] [CrossRef] [PubMed]
- Ogiso, H.; Kagi, N.; Matsumoto, E.; Nishimoto, M.; Arai, R.; Shirouzu, M.; Mimura, J.; Fujii-Kuriyama, Y.; Yokoyama, S. Phosphorylation analysis of 90 kDa heat shock protein within the cytosolic arylhydrocarbon receptor complex. Biochemistry (Mosc.) 2004, 43, 15510–15519. [Google Scholar] [CrossRef]
- Kurokawa, M.; Zhao, C.; Reya, T.; Kornbluth, S. Inhibition of apoptosome formation by suppression of Hsp90beta phosphorylation in tyrosine kinase-induced leukemias. Mol. Cell. Biol. 2008, 28, 5494–5506. [Google Scholar] [CrossRef] [PubMed]
- Wayne, N.; Bolon, D.N. Charge-rich regions modulate the anti-aggregation activity of Hsp90. J. Mol. Biol. 2010, 401, 931–939. [Google Scholar] [CrossRef] [PubMed]
- Hainzl, O.; Lapina, M.C.; Buchner, J.; Richter, K. The charged linker region is an important regulator of Hsp90 function. J. Biol. Chem. 2009, 284, 22559–22567. [Google Scholar] [CrossRef] [PubMed]
- Miyata, Y.; Nishida, E. CK2 controls multiple protein kinases by phosphorylating a kinase-targeting molecular chaperone, Cdc37. Mol. Cell. Biol. 2004, 24, 4065–4074. [Google Scholar] [CrossRef] [PubMed]
- Shao, J.; Prince, T.; Hartson, S.D.; Matts, R.L. Phosphorylation of serine 13 is required for the proper function of the Hsp90 co-chaperone, Cdc37. J. Biol. Chem. 2003, 278, 38117–38120. [Google Scholar] [CrossRef] [PubMed]
- Beebe, K.; Mollapour, M.; Scroggins, B.; Prodromou, C.; Xu, W.; Tokita, M.; Taldone, T.; Pullen, L.; Zierer, B.K.; Lee, M.J.; et al. Posttranslational modification and conformational state of Heat Shock Protein 90 differentially affect binding of chemically diverse small molecule inhibitors. Oncotarget 2013, 4, 1065–1074. [Google Scholar] [PubMed]
- Jhaveri, K.; Taldone, T.; Modi, S.; Chiosis, G. Advances in the clinical development of heat shock protein 90 (Hsp90) inhibitors in cancers. Biochim. Biophys. Acta Mol. Cell Res. 2012, 1823, 742–755. [Google Scholar] [CrossRef]
- Patki, J.M.; Pawar, S.S. HSP90: Chaperone-me-not. Pathol. Oncol. Res. POR 2013, 19, 631–640. [Google Scholar] [CrossRef]
- Zhang, T.; Li, Y.; Yu, Y.; Zou, P.; Jiang, Y.; Sun, D. Characterization of celastrol to inhibit hsp90 and cdc37 interaction. J. Biol. Chem. 2009, 284, 35381–35389. [Google Scholar] [CrossRef] [PubMed]
- Tash, J.S.; Attardi, B.; Hild, S.A.; Chakrasali, R.; Jakkaraj, S.R.; Georg, G.I. A novel potent indazole carboxylic acid derivative blocks spermatogenesis and is contraceptive in rats after a single oral dose. Biol. Reprod. 2008, 78, 1127–1138. [Google Scholar] [CrossRef] [PubMed]
- Tash, J.S.; Chakrasali, R.; Jakkaraj, S.R.; Hughes, J.; Smith, S.K.; Hornbaker, K.; Heckert, L.L.; Ozturk, S.B.; Hadden, M.K.; Kinzy, T.G.; et al. Gamendazole, an orally active indazole carboxylic acid male contraceptive agent, targets HSP90AB1 (HSP90BETA) and EEF1A1 (eEF1A), and stimulates Il1a transcription in rat Sertoli cells. Biol. Reprod. 2008, 78, 1139–1152. [Google Scholar] [CrossRef] [PubMed]
- Matts, R.L.; Brandt, G.E.; Lu, Y.; Dixit, A.; Mollapour, M.; Wang, S.; Donnelly, A.C.; Neckers, L.; Verkhivker, G.; Blagg, B.S. A systematic protocol for the characterization of Hsp90 modulators. Bioorg. Med. Chem. 2011, 19, 684–692. [Google Scholar] [CrossRef] [PubMed]
- Roe, S.M.; Ali, M.M.; Meyer, P.; Vaughan, C.K.; Panaretou, B.; Piper, P.W.; Prodromou, C.; Pearl, L.H. The Mechanism of Hsp90 regulation by the protein kinase-specific cochaperone p50(cdc37). Cell 2004, 116, 87–98. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, S.H.; Ventouras, L.A.; Lobbezoo, B.; Jackson, S.E. Independent ATPase activity of Hsp90 subunits creates a flexible assembly platform. J. Mol. Biol. 2004, 344, 813–826. [Google Scholar] [CrossRef] [PubMed]
- Eccles, S.A.; Massey, A.; Raynaud, F.I.; Sharp, S.Y.; Box, G.; Valenti, M.; Patterson, L.; de Haven Brandon, A.; Gowan, S.; Boxall, F.; et al. NVP-AUY922: A novel heat shock protein 90 inhibitor active against xenograft tumor growth, angiogenesis, and metastasis. Cancer Res. 2008, 68, 2850–2860. [Google Scholar] [CrossRef] [PubMed]
- Onuoha, S.C.; Mukund, S.R.; Coulstock, E.T.; Sengerova, B.; Shaw, J.; McLaughlin, S.H.; Jackson, S.E. Mechanistic studies on Hsp90 inhibition by ansamycin derivatives. J. Mol. Biol. 2007, 372, 287–297. [Google Scholar] [CrossRef] [PubMed]
- Roe, S.M.; Prodromou, C.; O’Brien, R.; Ladbury, J.E.; Piper, P.W.; Pearl, L.H. Structural basis for inhibition of the Hsp90 molecular chaperone by the antitumor antibiotics radicicol and geldanamycin. J. Med. Chem. 1999, 42, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Sun, L.; Xu, C.; Yu, F.; Zhou, H.; Zhao, Y.; Zhang, J.; Cai, J.; Mao, C.; Tang, L.; et al. Structure insights into mechanisms of ATP hydrolysis and the activation of human heat-shock protein 90. Acta Biochim. Biophys. Sin. 2012, 44, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Obermann, W.M.; Sondermann, H.; Russo, A.A.; Pavletich, N.P.; Hartl, F.U. In vivo function of Hsp90 is dependent on ATP binding and ATP hydrolysis. J. Cell Biol. 1998, 143, 901–910. [Google Scholar] [CrossRef] [PubMed]
- Jez, J.M.; Chen, J.C.; Rastelli, G.; Stroud, R.M.; Santi, D.V. Crystal structure and molecular modeling of 17-DMAG in complex with human Hsp90. Chem. Biol. 2003, 10, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Brough, P.A.; Aherne, W.; Barril, X.; Borgognoni, J.; Boxall, K.; Cansfield, J.E.; Cheung, K.M.; Collins, I.; Davies, N.G.; Drysdale, M.J.; et al. 4,5-diarylisoxazole Hsp90 chaperone inhibitors: Potential therapeutic agents for the treatment of cancer. J. Med. Chem. 2008, 51, 196–218. [Google Scholar] [CrossRef] [PubMed]
- Siligardi, G.; Panaretou, B.; Meyer, P.; Singh, S.; Woolfson, D.N.; Piper, P.W.; Pearl, L.H.; Prodromou, C. Regulation of Hsp90 ATPase activity by the co-chaperone Cdc37p/p50cdc37. J. Biol. Chem. 2002, 277, 20151–20159. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Guo, X.; Borkan, S.C.; Bharti, A.; Kuramochi, Y.; Calderwood, S.; Sawyer, D.B. Heat shock protein 90 stabilization of ErbB2 expression is disrupted by ATP depletion in myocytes. J. Biol. Chem. 2005, 280, 13148–13152. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are not available.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olesen, S.H.; Ingles, D.J.; Zhu, J.-Y.; Martin, M.P.; Betzi, S.; Georg, G.I.; Tash, J.S.; Schönbrunn, E. Stability of the Human Hsp90-p50Cdc37 Chaperone Complex against Nucleotides and Hsp90 Inhibitors, and the Influence of Phosphorylation by Casein Kinase 2. Molecules 2015, 20, 1643-1660. https://doi.org/10.3390/molecules20011643
Olesen SH, Ingles DJ, Zhu J-Y, Martin MP, Betzi S, Georg GI, Tash JS, Schönbrunn E. Stability of the Human Hsp90-p50Cdc37 Chaperone Complex against Nucleotides and Hsp90 Inhibitors, and the Influence of Phosphorylation by Casein Kinase 2. Molecules. 2015; 20(1):1643-1660. https://doi.org/10.3390/molecules20011643
Chicago/Turabian StyleOlesen, Sanne H., Donna J. Ingles, Jin-Yi Zhu, Mathew P. Martin, Stephane Betzi, Gunda I. Georg, Joseph S. Tash, and Ernst Schönbrunn. 2015. "Stability of the Human Hsp90-p50Cdc37 Chaperone Complex against Nucleotides and Hsp90 Inhibitors, and the Influence of Phosphorylation by Casein Kinase 2" Molecules 20, no. 1: 1643-1660. https://doi.org/10.3390/molecules20011643
APA StyleOlesen, S. H., Ingles, D. J., Zhu, J.-Y., Martin, M. P., Betzi, S., Georg, G. I., Tash, J. S., & Schönbrunn, E. (2015). Stability of the Human Hsp90-p50Cdc37 Chaperone Complex against Nucleotides and Hsp90 Inhibitors, and the Influence of Phosphorylation by Casein Kinase 2. Molecules, 20(1), 1643-1660. https://doi.org/10.3390/molecules20011643