Chemically-Modified Cellulose Paper as a Microstructured Catalytic Reactor
Abstract
:1. Introduction

2. In Situ Modification of Cellulose Paper with Functional Groups Using the Silane-Coupling Technique
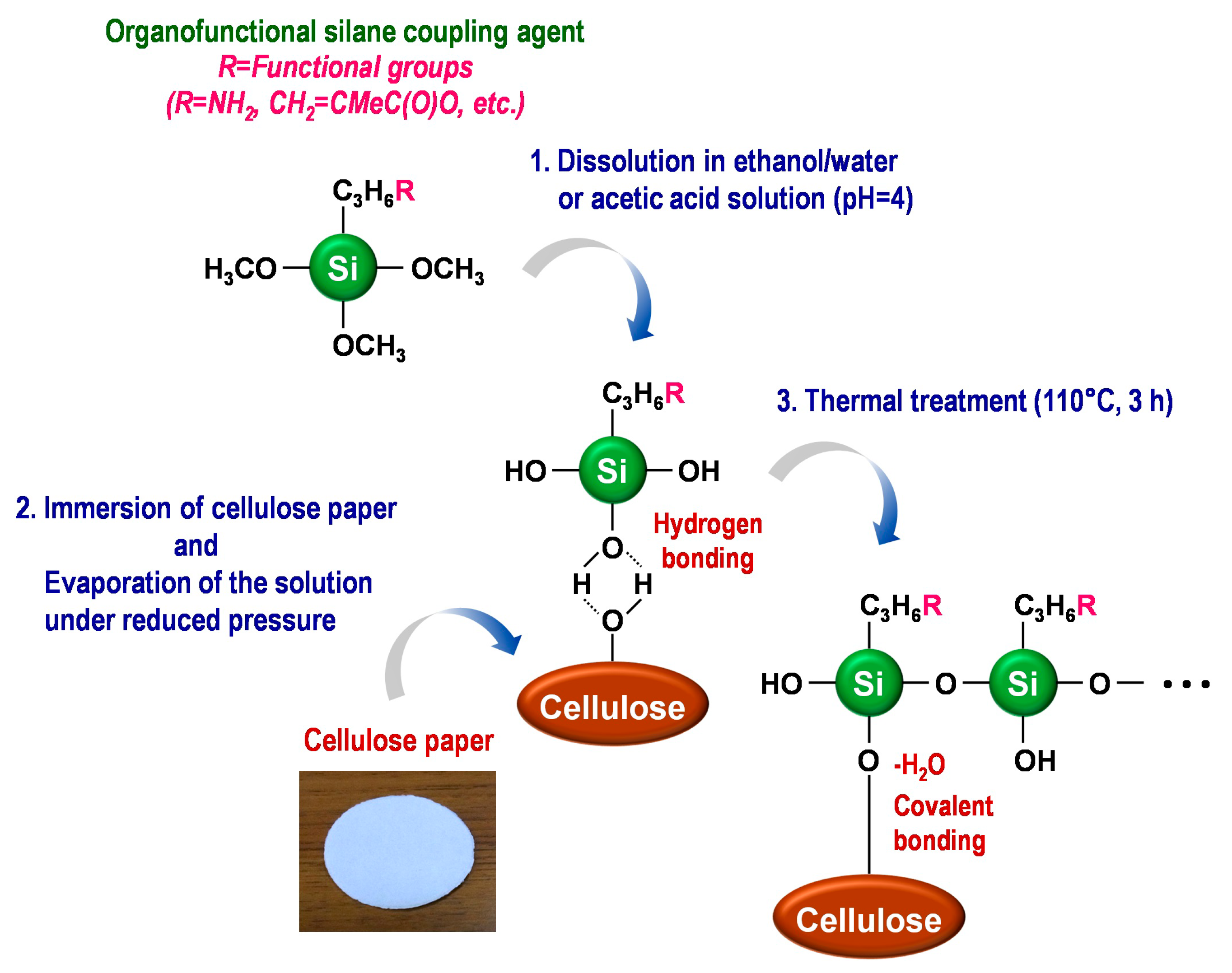
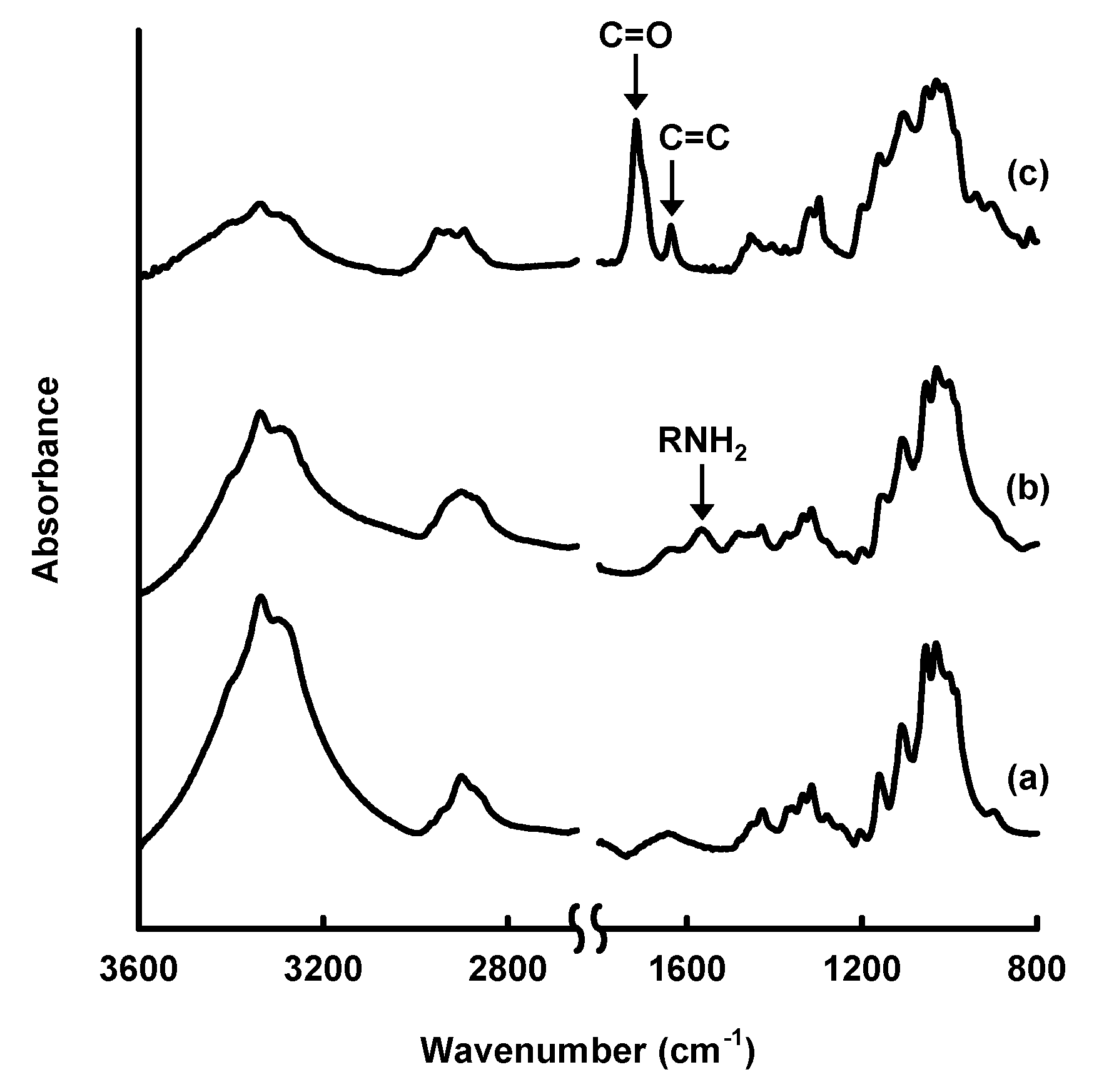

3. Amine-Modified Paper for Knoevenagel Condensation
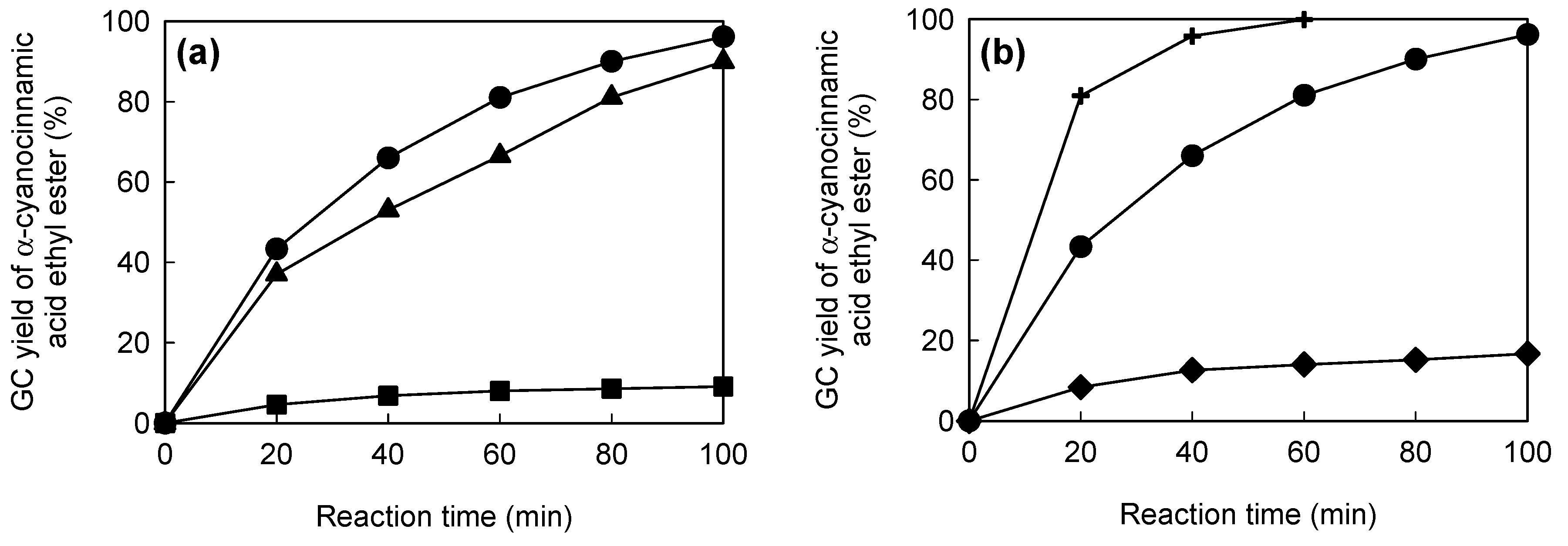
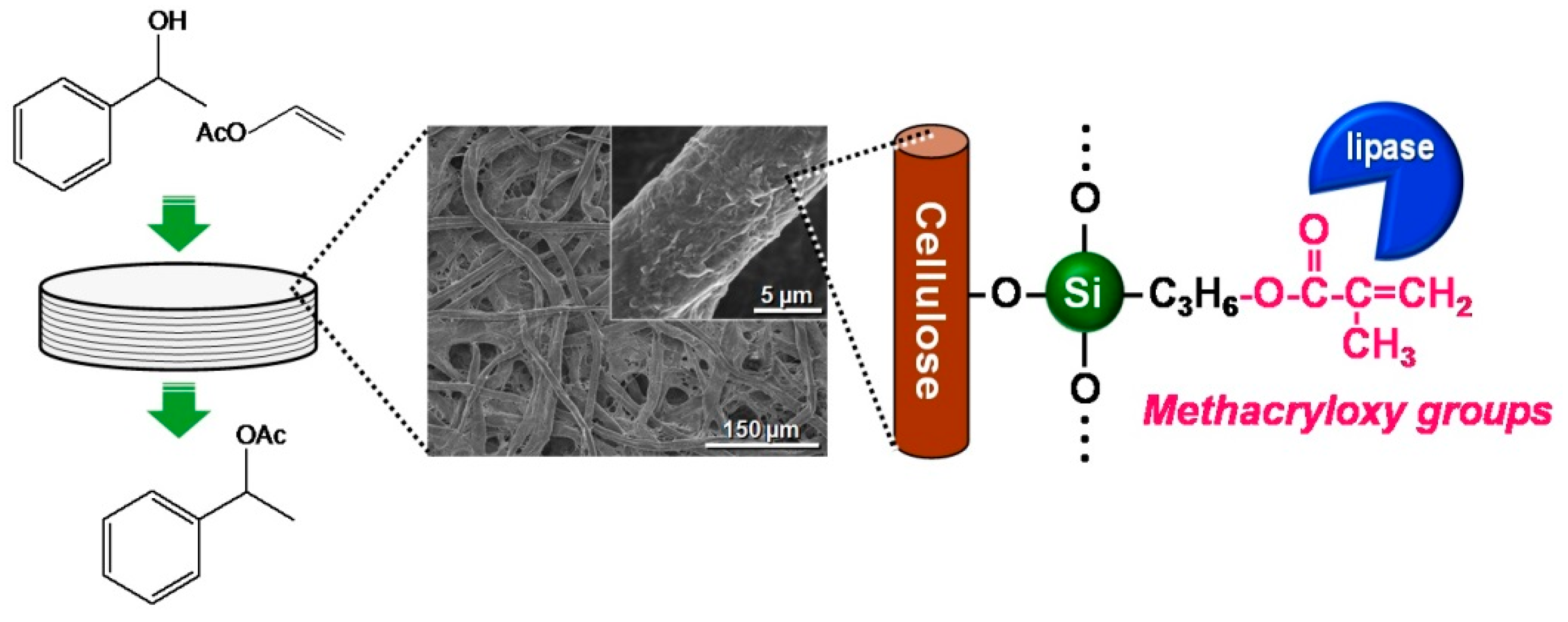
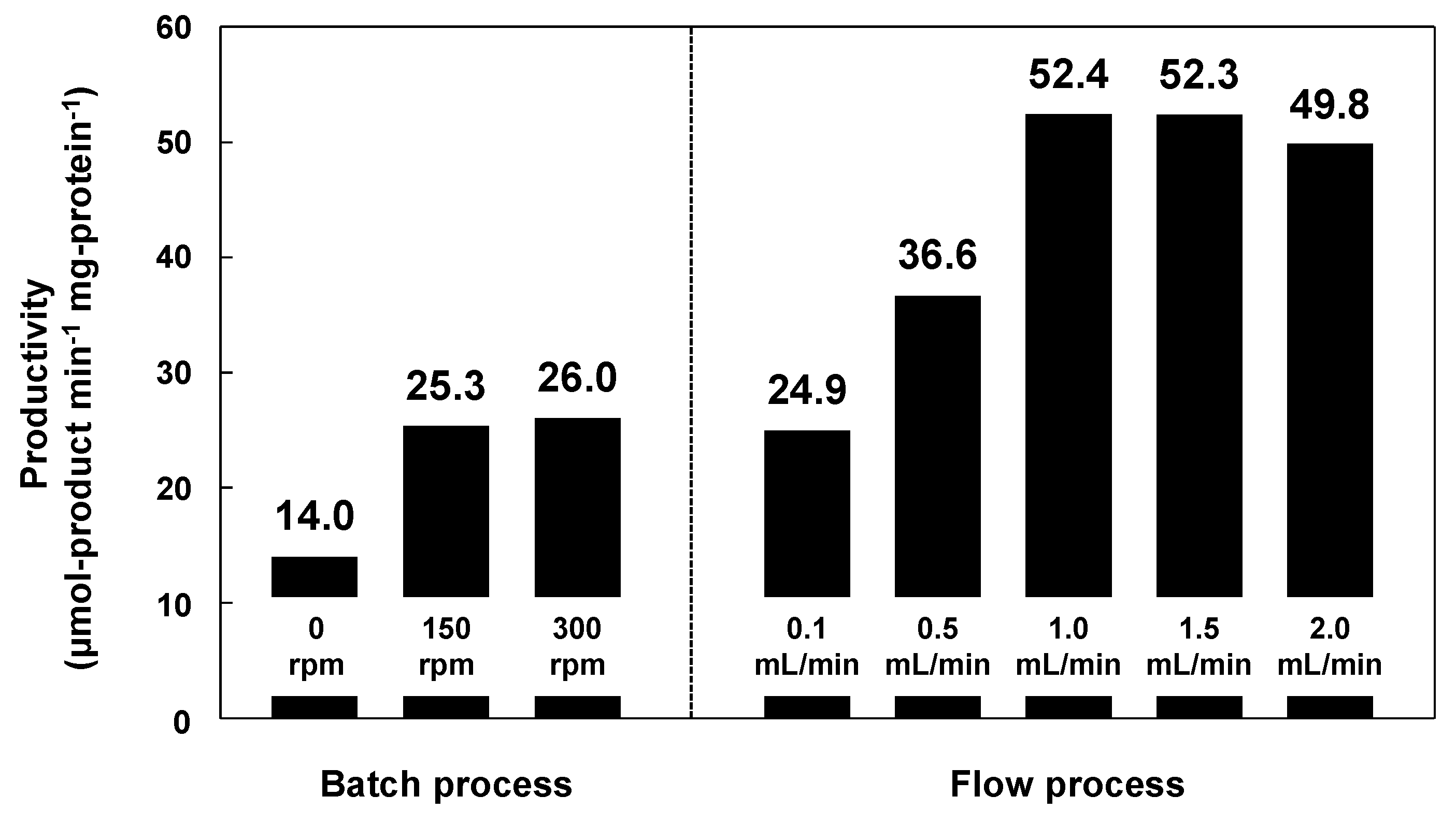
4. Immobilized Lipase on Methacrylate-Modified Paper for Nonaqueous Transesterification
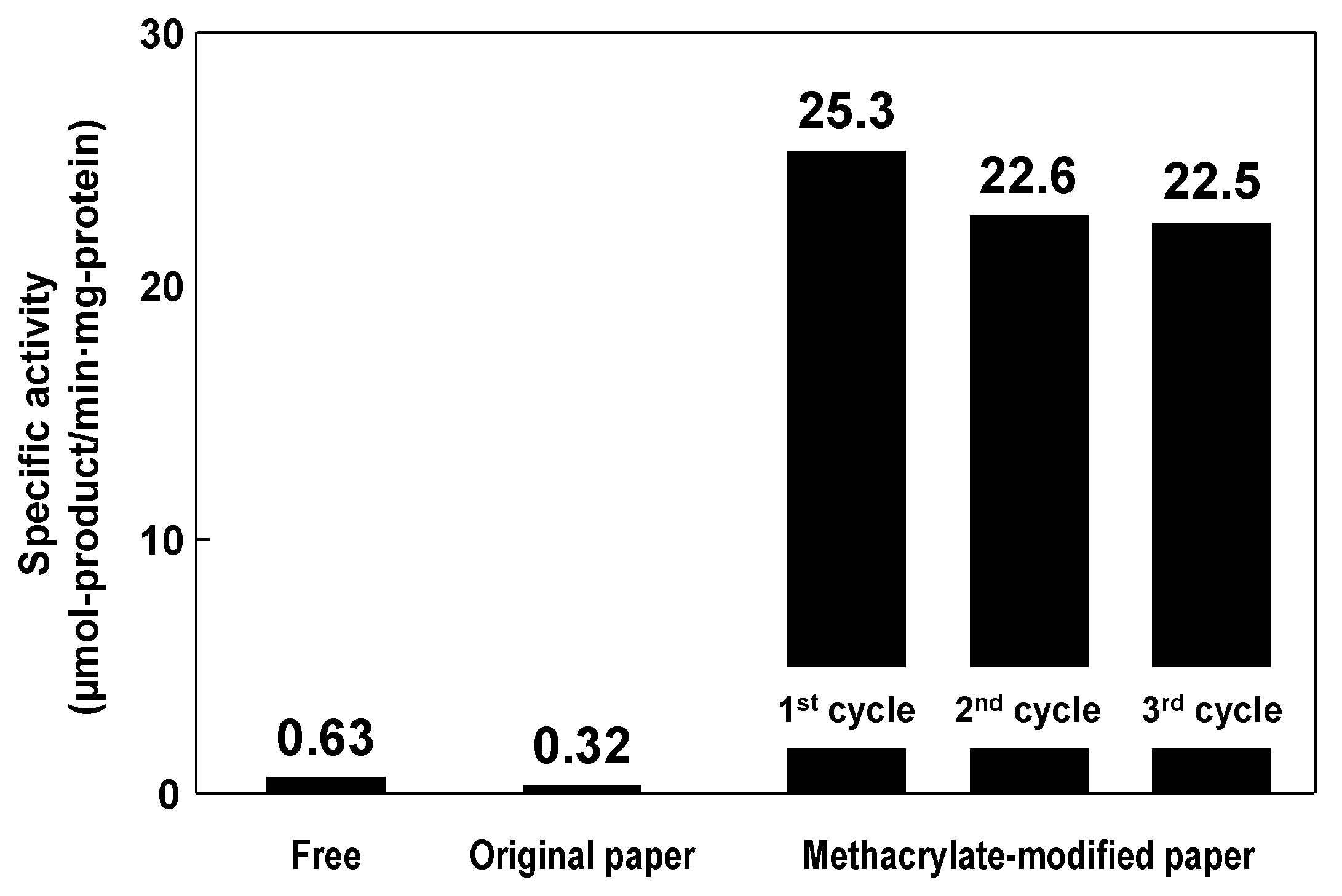
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ishida, T.; Haruta, M. Gold catalysts: Towards sustainable chemistry. Angew. Chem. Int. Ed. 2007, 46, 7154–7156. [Google Scholar]
- Campelo, J.M.; Luna, D.; Luque, R.; Marinas, J.M.; Romero, A.A. Sustainable preparation of supported metal nanoparticles and their applications in catalysis. ChemSusChem 2009, 2, 18–45. [Google Scholar] [CrossRef] [PubMed]
- D’yakonov, V.A.; Trapeznikova, O.A.; de Meijere, A.; Dzhemilev, U.M. Metal complex catalysis in the synthesis of spirocarbocycles. Chem. Rev. 2014, 114, 5775–5814. [Google Scholar] [CrossRef] [PubMed]
- List, B.; Lerner, R.A.; Barbas, C.F., III. Proline-catalyzed direct asymmetric aldol reactions. J. Am. Chem. Soc. 2000, 122, 2395–2396. [Google Scholar] [CrossRef]
- Northrup, A.B.; MacMillan, D.W.C. Two step synthesis of carbohydrates by selective aldol reactions. Science 2004, 305, 1752–1755. [Google Scholar] [CrossRef] [PubMed]
- Koeller, K.M. Enzymes for chemical synthesis. Nature 2001, 409, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Schmid, A.; Dordick, J.S.; Hauer, B.; Kiener, A.; Wubbolts, M.; Witholt, B. Industrial biocatalysis today and tomorrow. Nature 2001, 409, 258–268. [Google Scholar] [CrossRef] [PubMed]
- Fujisawa, S.; Ikeuchi, T.; Takeuchi, M.; Saito, T.; Isogai, A. Superior reinforcement effect of TEMPO-oxidized cellulose nanofibrils in polystyrene matrix: Optical, thermal and mechanical studies. Biomacromolecules 2012, 13, 2188–2194. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Saito, T.; Fujisawa, S.; Fukuzumi, H.; Isogai, A. Ultrastrong and high gas-barrier nanocellulose/clay layered composites. Biomacromolecules 2012, 13, 1927–1932. [Google Scholar] [CrossRef] [PubMed]
- Fukuzumi, H.; Saito, T.; Kumamoto, Y.; Iwata, T.; Isogai, A. Transparent and high gas barrier films of cellulose nanofibers prepared by TEMPO-mediated oxidation. Biomacromolecules 2009, 10, 162–165. [Google Scholar] [CrossRef] [PubMed]
- Nogi, M.; Yano, H. Optically transparent nanofiber paper. Adv. Mater. 2009, 21, 1595–1598. [Google Scholar] [CrossRef]
- Nogi, M.; Kim, C.; Sugahara, T.; Inui, T.; Takahashi, T.; Suganuma, K. High thermal stability of optical transparency in cellulose nanofiber paper. Appl. Phys. Lett. 2013, 102, 181911. [Google Scholar] [CrossRef]
- Koga, H.; Saito, T.; Kitaoka, T.; Nogi, M.; Suganuma, K.; Isogai, A. Transparent, conductive and printable composites consisting of TEMPO-oxidized nanocellulose and carbon nanotube. Biomacromolecules 2013, 14, 1160–1165. [Google Scholar] [CrossRef] [PubMed]
- Fujisaki, Y.; Koga, H.; Nakajima, Y.; Nakata, M.; Tsuji, H.; Yamamoto, T.; Kurita, T.; Nogi, M.; Shimidzu, N. Transparent nanopaper-based flexible organic thin-film transistor array. Adv. Funct. Mater. 2014, 24, 1657–1663. [Google Scholar] [CrossRef]
- Koga, H.; Nogi, M.; Komoda, N.; Nge, T.T.; Sugahara, T.; Suganuma, K. Uniformly connected conductive networks on cellulose nanofiber paper for transparent paper electronics. NPG Asia Mater. 2014, 6, e93. [Google Scholar] [CrossRef]
- Nagashima, K.; Koga, H.; Celano, U.; Zhuge, F.; Kanai, M.; Rahong, S.; Meng, G.; He, Y.; Boeck, J.D.; Jurczak, M.; et al. Cellulose nanofiber paper as an ultra flexible nonvolatile memory. Sci. Rep. 2014, 4, 5532. [Google Scholar] [CrossRef] [PubMed]
- Inui, T.; Koga, H.; Nogi, M.; Komoda, N.; Suganuma, K. Miniaturized flexible antenna printed on high dielectric constant nanopaper composite. Adv. Mater. 2015, in press. [Google Scholar]
- Koga, H.; Tokunaga, E.; Hidaka, M.; Umemura, Y.; Saito, T.; Isogai, A.; Kitaoka, T. Topochemical synthesis and catalysis of metal nanoparticles exposed on crystalline cellulose nanofibers. Chem. Commun. 2010, 46, 8567–8569. [Google Scholar] [CrossRef]
- Azetsu, A.; Koga, H.; Isogai, A.; Kitaoka, T. Synthesis and catalytic features of hybrid metal nanoparticles supported on cellulose nanofibers. Catalysts 2011, 1, 83–96. [Google Scholar] [CrossRef]
- Koga, H.; Azetsu, A.; Tokunaga, E.; Saito, T.; Isogai, A.; Kitaoka, T. Topological loading of Cu(I) catalysts onto crystalline cellulose nanofibrils for the Huisgen click reaction. J. Mater. Chem. 2012, 22, 5538–5542. [Google Scholar] [CrossRef]
- Tsutsumi, Y.; Koga, H.; Qi, Z.-D.; Saito, T.; Isogai, A. Nanofibrillar chitin aerogels as renewable base catalysts. Biomacromolecules 2014, 15, 4314–4319. [Google Scholar] [CrossRef] [PubMed]
- Kiwi-Minsker, L.; Renken, A. Microstructured reactors for catalytic reactions. Catal. Today 2005, 110, 2–14. [Google Scholar] [CrossRef]
- Costantini, F.; Benetti, E.M.; Reinhoudt, D.N.; Huskens, J.; Vancso, G.J.; Verboom, W. Enzyme-functionalized polymer brush films on the inner wall of silicon-glass microreactors with tunable biocatalytic activity. Lab Chip 2010, 10, 3407–3412. [Google Scholar] [CrossRef] [PubMed]
- Lindström, B.; Agrell, J.; Pettersson, L.J. Combined methanol reforming for hydrogen generation over monolithic catalysts. Chem. Eng. J. 2003, 93, 91–101. [Google Scholar] [CrossRef]
- Patcas, F.C.; Garrido, G.I.; Kraushaar-Czarnetzki, B. CO oxidation over structured carriers: A comparison of ceramic foams, honeycombs and beads. Chem. Eng. Sci. 2007, 62, 3984–3990. [Google Scholar] [CrossRef]
- Lin, Y.-G.; Hsu, Y.-K.; Chen, S.-Y.; Lin, Y.-K.; Chen, L.-C.; Chen, K.-H. Nanostructured zinc oxide nanorods with copper nanoparticles as a microreformation catalyst. Angew. Chem. Int. Ed. 2009, 48, 7586–7590. [Google Scholar] [CrossRef]
- Nishihara, H.; Mukai, S.R.; Yamashita, D.; Tamon, H. Ordered macroporous silica by ice templating. Chem. Mater. 2005, 17, 683–689. [Google Scholar] [CrossRef]
- Hyun, S.H.; Jo, S.Y.; Kang, B.S. Surface modification of γ-alumina membranes by silane coupling for CO2 separation. J. Membr. Sci. 1996, 120, 197–206. [Google Scholar] [CrossRef]
- Yamaguchi, K.; Kitabatake, T.; Izawa, M.; Fujiwara, T.; Nishimura, H.; Futami, T. Novel silane coupling agents containing a photolabile 2-nitrobenzyl ester for introduction of a carboxy group on the surface of silica gel. Chem. Lett. 2000, 29, 228–229. [Google Scholar] [CrossRef]
- Abdelmouleh, M.; Boufi, S.; ben Salah, A.; Belgacem, M.N.; Gandini, A. Interaction of silane coupling agents with cellulose. Langmuir 2002, 18, 3203–3208. [Google Scholar] [CrossRef]
- Abdelmouleh, M.; Boufi, S.; Belgacem, M.N.; Duarte, A.P.; ben Salah, A.; Gandini, A. Modification of cellulosic fibres with functionalised silanes: Development of surface properties. Int. J. Adhes. Adhes. 2004, 24, 43–54. [Google Scholar] [CrossRef]
- Gazit, O.M.; Charmot, A.; Katz, A. Grafted cellulose strands on the surface of silica: Effect of environment on reactivity. Chem. Commun. 2011, 47, 376–378. [Google Scholar] [CrossRef]
- Koga, H.; Kitaoka, T.; Isogai, A. In situ modification of cellulose paper with amino groups for catalytic applications. J. Mater. Chem. 2011, 21, 9356–9361. [Google Scholar] [CrossRef]
- Koga, H.; Kitaoka, T.; Isogai, A. Paper-immobilized enzyme as a green microstructured catalyst. J. Mater. Chem. 2012, 22, 11591–11597. [Google Scholar] [CrossRef]
- Krishnan, G.R.; Sreekumar, K. First example of organocatalysis by polystyrene-supported PAMAM dendrimers: Highly efficient and reusable catalyst for Knoevenagel condensations. Eur. J. Org. Chem. 2008, 28, 4763–4768. [Google Scholar] [CrossRef]
- Chtchigrovsky, M.; Primo, A.; Gonzalez, P.; Molvinger, K.; Robitzer, M.; Quignard, F.; Taran, F. Functionalized chitosan as a green, recyclable, biopolymer-supported catalyst for the [3+2] Huisgen cycloaddition. Angew. Chem. Int. Ed. 2009, 48, 5916–5920. [Google Scholar] [CrossRef]
- Hicks, J.C.; Drese, J.H.; Fauth, D.J.; Gray, M.L.; Qi, G.; Jones, C.W. Designing adsorbents for CO2 capture from flue gas-hyperbranched aminosilicas capable of capturing CO2 reversibly. J. Am. Chem. Soc. 2008, 130, 2902–2903. [Google Scholar] [CrossRef] [PubMed]
- Knoevenagel, E. Condensation von malonsäure mit aromatischen aldehyden durch ammoniak und amine. Ber. Dtsch. Chem. Ges. 1898, 31, 2596–2619. [Google Scholar] [CrossRef]
- Fondjo, E.S.; Döpp, D. Condensation of some ketones with methylene active nitriles in varied stoichiometries. J. Heterocycl. Chem. 2006, 43, 1037–1044. [Google Scholar] [CrossRef]
- McCluskey, A.; Robinson, P.J.; Hill, T.; Scott, J.L.; Edwards, J.K. Green chemistry approaches to the Knoevenagel condensation: Comparison of ethanol, water and solvent free (dry grind) approaches. Tetrahedron Lett. 2002, 43, 3117–3120. [Google Scholar] [CrossRef]
- Reddy, K.R.; Rajgopal, K.; Maheswari, C.U.; Kantam, M.L. Chitosan hydrogel: A green and recyclable biopolymer catalyst for aldol and Knoevenagel reactions. New J. Chem. 2006, 30, 1549–1552. [Google Scholar] [CrossRef]
- Sudheesh, N.; Sharma, S.K.; Shukla, R.S. Chitosan as an eco-friendly solid base catalyst for the solvent-free synthesis of jasminaldehyde. J. Mol. Catal. A Chem. 2010, 321, 77–82. [Google Scholar] [CrossRef]
- Ricci, A.; Bernardi, L.; Gioia, C.; Vierucci, S.; Robitzer, M.; Quignard, F. Chitosan aerogel: A recyclable, heterogeneous organocatalyst for the asymmetric direct aldol reaction in water. Chem. Commun. 2010, 46, 6288–6290. [Google Scholar] [CrossRef]
- Zaks, A.; Klibanov, A.M. Enzyme-catalyzed processes in organic solvents. Proc. Natl. Acad. Sci. USA 1985, 82, 3192–3196. [Google Scholar] [CrossRef] [PubMed]
- Foresti, M.L.; Ferreira, M.L. Solvent-free ethyl oleate synthesis mediated by lipase from Candida antarctica B adsorbed on polypropylene powder. Catal. Today 2005, 107, 23–30. [Google Scholar] [CrossRef]
- Yadav, G.D.; Lathi, P.S. Synthesis of citronellol laurate in organic media catalyzed by immobilized lipases: Kinetic studies. J. Mol. Catal. B Enzym. 2004, 27, 113–119. [Google Scholar] [CrossRef]
- Yagiz, F.; Kazan, D.; Akin, A.N. Biodiesel production from waste oils by using lipase immobilized on hydrotalcite and zeolites. Chem. Eng. J. 2007, 134, 262–267. [Google Scholar] [CrossRef]
- Klibanov, A.M. Why are enzymes less active in organic solvents than in water? Trends Biotechnol. 1997, 15, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Blanco, R.M.; Terreros, P.; Fernández-Pérez, M.; Otero, C.; Diaz-González, G. Functionalization of mesoporous silica for lipase immobilization: Characterization of the support and the catalysts. J. Mol. Catal. B Enzym. 2004, 30, 83–93. [Google Scholar] [CrossRef]
- Bai, Y.-X.; Li, Y.-F.; Yang, Y.; Yi, L.-X. Covalent immobilization of triacylglycerol lipase onto functionalized novel mesoporous silica supports. J. Biotechnol. 2006, 125, 574–582. [Google Scholar] [CrossRef] [PubMed]
- Kamori, M.; Hori, T.; Yamashita, Y.; Hirose, Y.; Naoshima, Y. Immobilization of lipase on a new inorganic ceramics support, toyonite, and the reactivity and enantioselectivity of the immobilized lipase. J. Mol. Catal. B Enzym. 2000, 9, 269–274. [Google Scholar] [CrossRef]
- Huang, L.; Cheng, Z.-M. Immobilization of lipase on chemically modified bimodal ceramic foams for olive oil hydrolysis. Chem. Eng. J. 2008, 144, 103–109. [Google Scholar] [CrossRef]
- Shah, S.; Solanki, K.; Gupta, M.N. Enhancement of lipase activity in non-aqueous media upon immobilization on multi-walled carbon nanotubes. Chem. Cent. J. 2007, 1, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Sakai, S.; Liu, Y.; Yamaguchi, T.; Watanabe, R.; Kawabe, M.; Kawakami, K. Immobilization of Pseudomonas cepacia lipase onto electrospun polyacrylonitrile fibers through physical adsorption and application to transesterification in nonaqueous solvent. Biotechnol. Lett. 2010, 32, 1059–1062. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, N.N.; Vijayalakshmi, V.; Sawant, S.B.; Joshi, J.B. Immobilization of Mucor miehei lipase on ion exchange resins. Chem. Eng. J. Biochem. Eng. J. 1996, 61, 149–156. [Google Scholar] [CrossRef]
- Mateo, C.; Palomo, J.M.; Fernandez-Lorente, G.; Guisan, J.M.; Fernandez-Lafuente, R. Improvement of enzyme activity, stability and selectivity via immobilization techniques. Enzym. Microb. Technol. 2007, 40, 1451–1463. [Google Scholar] [CrossRef]
- Tischer, W.; Wedekind, F. Immobilized enzymes: Methods and applications. Top. Curr. Chem. 1999, 200, 95–126. [Google Scholar]
- Bastida, A.; Sabuquillo, P.; Armisen, P.; Fernandez-Lafuente, R.; Huguet, J.; Guisan, J.M. A single step purification, immobilization, and hyperactivation of lipases via interfacial adsorption on strongly hydrophobic supports. Biotechnol. Bioeng. 1998, 58, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Lafuente, R.; Armisen, P.; Sabuquillo, P.; Fernandez-Lorente, G.; Guisan, J.M. Immobilization of lipases by selective adsorption on hydrophobic supports. Chem. Phys. Lipids 1998, 93, 185–197. [Google Scholar] [CrossRef] [PubMed]
- Mason, B.P.; Price, K.E.; Steinbacher, J.L.; Bogdan, A.R.; McQuade, D.T. Greener approaches to organic synthesis using microreactor technology. Chem. Rev. 2007, 107, 2300–2318. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, J.; Nagaki, A.; Yamada, T. Flash chemistry: Fast chemical synthesis by using microreactors. Chem. Eur. J. 2008, 14, 7450–7459. [Google Scholar] [CrossRef] [PubMed]
- Bolivar, J.M.; Wiesbauer, J.; Nidetzky, B. Biotransformations in microstructured reactors: More than flowing with the stream? Trends Biotechnol. 2009, 29, 333–342. [Google Scholar] [CrossRef]
- Debeche, T.; Marmet, C.; Kiwi-Minsker, L.; Renken, A.; Juillerat, M.-A. Structured fiber supports for gas phase biocatalysis. Enzym. Microb. Technol. 2005, 36, 911–916. [Google Scholar] [CrossRef]
- O’Connell, P.J.; Varley, J. Immobilization of Candida rugosa lipase on colloidal gas aphrons (CGAs). Biotechnol. Bioeng. 2001, 74, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Fukahori, S.; Ichiura, H.; Kitaoka, T.; Tanaka, H. Capturing of bisphenol A photodecomposition intermediates by composite TiO2-zeolite sheets. Appl. Catal. B 2003, 46, 453–462. [Google Scholar] [CrossRef]
- Fukahori, S.; Iguchi, Y.; Ichiura, H.; Kitaoka, T.; Tanaka, H.; Wariishi, H. Effect of void structure of photocatalyst paper on VOC decomposition. Chemosphere 2007, 66, 2136–2141. [Google Scholar] [CrossRef] [PubMed]
- Koga, H.; Kitaoka, T.; Nakamura, M.; Wariishi, H. Influence of a fiber-network microstructure of paper-structured catalyst on methanol reforming behavior. J. Mater. Sci. 2009, 44, 5836–5841. [Google Scholar] [CrossRef]
- Koga, H.; Umemura, Y.; Tomoda, A.; Suzuki, R.; Kitaoka, T. In situ synthesis of platinum nanocatalysts on a microstructured paperlike matrix for the catalytic purification of exhaust gases. ChemSusChem 2010, 3, 604–608. [Google Scholar] [CrossRef] [PubMed]
- Banús, E.D.; Ulla, M.A.; Galván, M.V.; Zanuttini, M.A.; Milt, V.G.; Miró, E.E. Catalytic ceramic paper for the combustion of diesel soot. Catal. Commun. 2010, 12, 46–49. [Google Scholar] [CrossRef]
- Homma, T.; Kitaoka, T. Multiphase catalytic oxidation of alcohols over paper-structured catalysts with micrometer-size pores. Appl. Catal. A 2014, 486, 201–209. [Google Scholar] [CrossRef]
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koga, H.; Kitaoka, T.; Isogai, A. Chemically-Modified Cellulose Paper as a Microstructured Catalytic Reactor. Molecules 2015, 20, 1495-1508. https://doi.org/10.3390/molecules20011495
Koga H, Kitaoka T, Isogai A. Chemically-Modified Cellulose Paper as a Microstructured Catalytic Reactor. Molecules. 2015; 20(1):1495-1508. https://doi.org/10.3390/molecules20011495
Chicago/Turabian StyleKoga, Hirotaka, Takuya Kitaoka, and Akira Isogai. 2015. "Chemically-Modified Cellulose Paper as a Microstructured Catalytic Reactor" Molecules 20, no. 1: 1495-1508. https://doi.org/10.3390/molecules20011495
APA StyleKoga, H., Kitaoka, T., & Isogai, A. (2015). Chemically-Modified Cellulose Paper as a Microstructured Catalytic Reactor. Molecules, 20(1), 1495-1508. https://doi.org/10.3390/molecules20011495








