Phenolic Content and Antioxidant Capacity in Algal Food Products
Abstract
:1. Introduction
2. Results and Discussion
2.1. Total Phenolic Content
| Algae | (1) | (2) | (3) | (4) | (5) |
|---|---|---|---|---|---|
| Eisenia bicyclis | 192.6 ± 3.3 a | 192.8 ± 0.8 a | 143.2 ± 9.5 b | 84.1 ± 0.3 c | 9.5 ± 0.5 d |
| Hizikia fusiformis | 34.5 ± 5.8 a | 26.9 ± 0.1 b | 9.5 ± 0.1 c | 13.1 ± 0.1 c | 6.0 ± 0.1 c |
| Laminaria japonica | 8.7 ± 0.3 a | 8.5 ± 0.3 a | 14.9 ± 0.1 b | 8.8 ± 0.1 a | 0.7 ± 0.1 c |
| Undaria pinnatifida (W) | 8.6 ± 0.4 a | 3.7 ± 0.1 b | 5.9 ± 0.1 c | 5.7 ± 0.1 c | 1.3 ± 0.1 d |
| Undaria pinnatifida (Wi) | 8.0 ± 0.5 a | 5.0 ± 0.1 b | 6.5 ± 0.1 c | 4.6 ± 0.1 b | 8.4 ± 0.2 a |
| Palmaria palmata | 31.8 ± 1.0 a | 22.1 ± 0.7 b | 26.5 ± 0.4 c | 25.0 ± 0.1 d | 10.7 ± 0.3 e |
| Porphyra tenera | 18.2 ± 0.6 a | 16.2 ± 0.4 b | 15.1 ± 0.1 b | 11.1 ± 0.1 c | 4.7 ± 0.6 d |
| Chlorella pyrenoidosa | 18.0 ± 0.2 a | 13.2 ± 0.2 b | 16.8 ± 0.1 a,c | 15.5 ± 0.1 c | 25.8 ± 1.7 d |
| Spirulina platensis | 43.2 ± 1.0 a | 17.0 ± 0.5 b | 23.9 ± 0.1 c | 18.4 ± 0.1 d | 24.4 ± 0.2 c |
2.2. Selected Phenolic Compounds Content
| GA | HBA | C | EC | CG | ECG | EGC | EGCG | PC | |
|---|---|---|---|---|---|---|---|---|---|
| A | 2.8 ± 0.1 | - | - | 3.2 ± 0.3 | 2.9 ± 1.0 | - | - | + | - |
| H | 14.1 ± 0.5 | - | - | 8.2 ± 0.1 | - | - | - | + | - |
| K | - | - | - | 3.1 ± 0.1 | - | - | 4.0 ± 0.2 | + | - |
| W | - | 1.9 ± 0.1 | - | - | - | - | 4.8 ± 0.1 | - | - |
| Wi | - | 8.1 ± 0.1 | - | 6.3 ± 0.5 | 2.0 ± 0.2 | - | 21.4 ± 0.1 | 7.5 ± 0.1 | - |
| D | - | 5.8 ± 0.1 | - | + | - | - | - | - | - |
| N | 3.5 ± 0.1 | 1.6 ± 0.1 | 128.8 ± 2.9 | 16.4 ± 0.7 | - | + | 16.0 ± 0.5 | 4.0 ± 0.1 | - |
| C | 5.0 ± 0.2 | 20.5 ± 0.1 | - | - | - | - | 20.2 ± 0.7 | - | - |
| S | - | - | 22.7 ± 2.3 | 27.5 ± 1.3 | - | - | - | - | 28.9 ± 0.6 |
2.3. Antioxidant Capacity of Water Soluble Compounds (ACW)
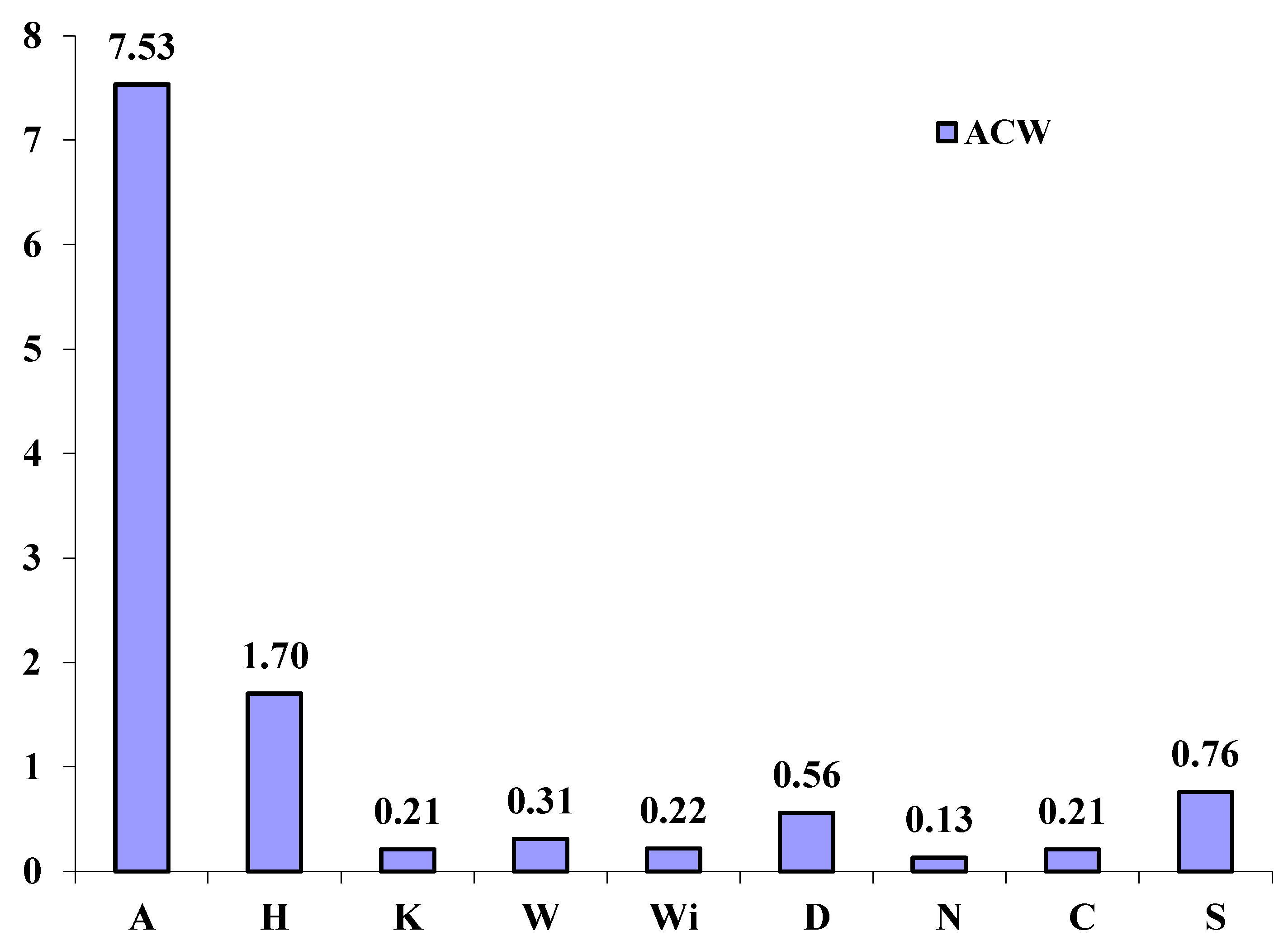
2.4. Statistical Correlations
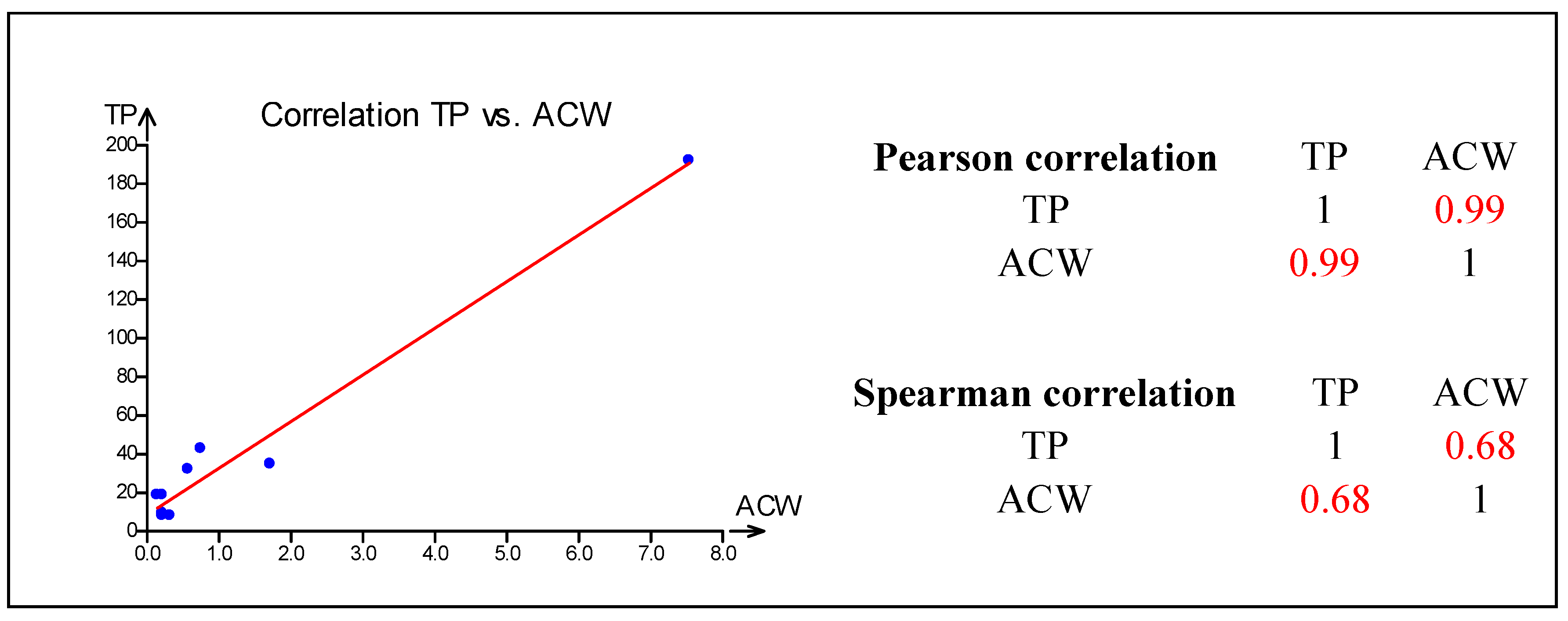
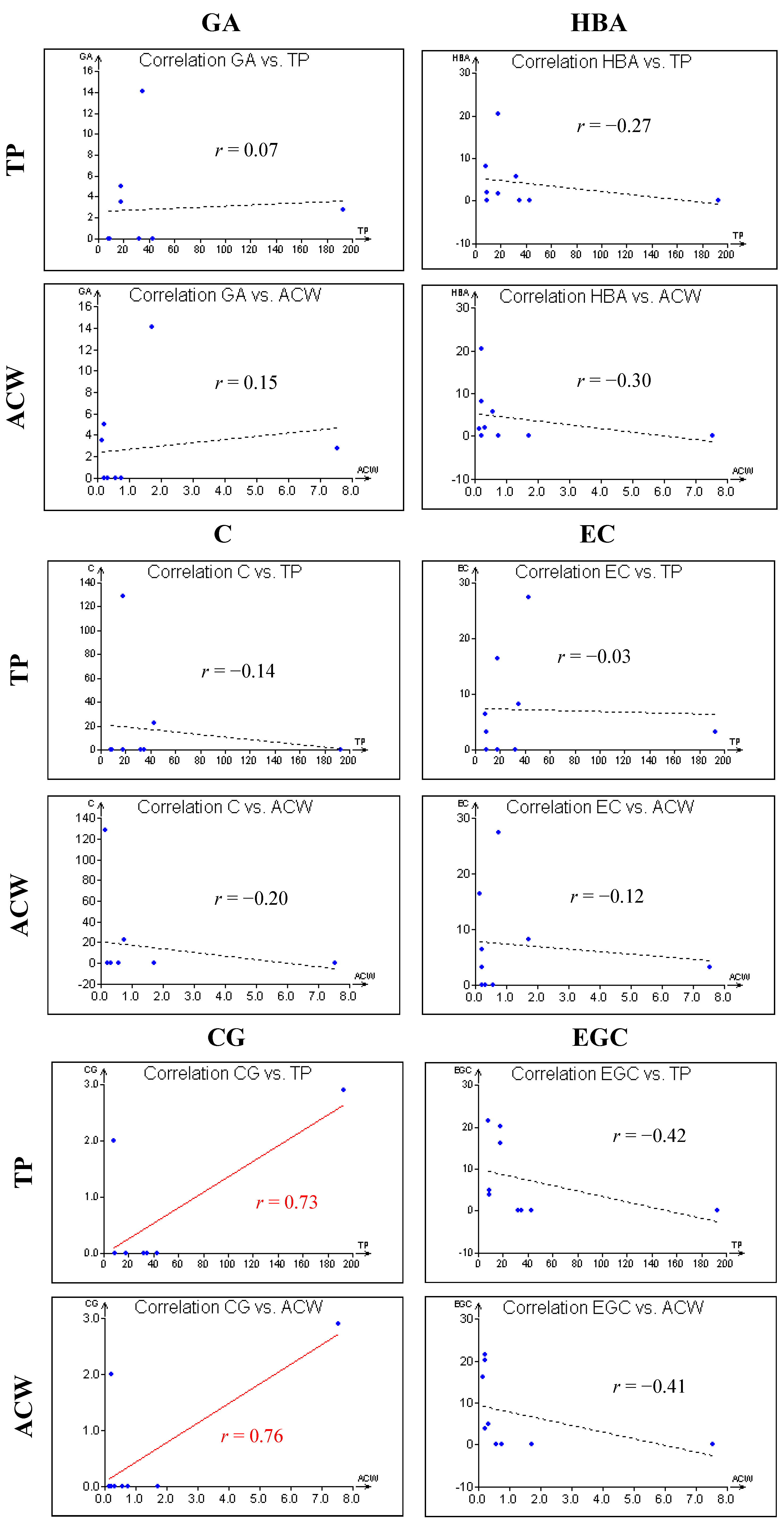
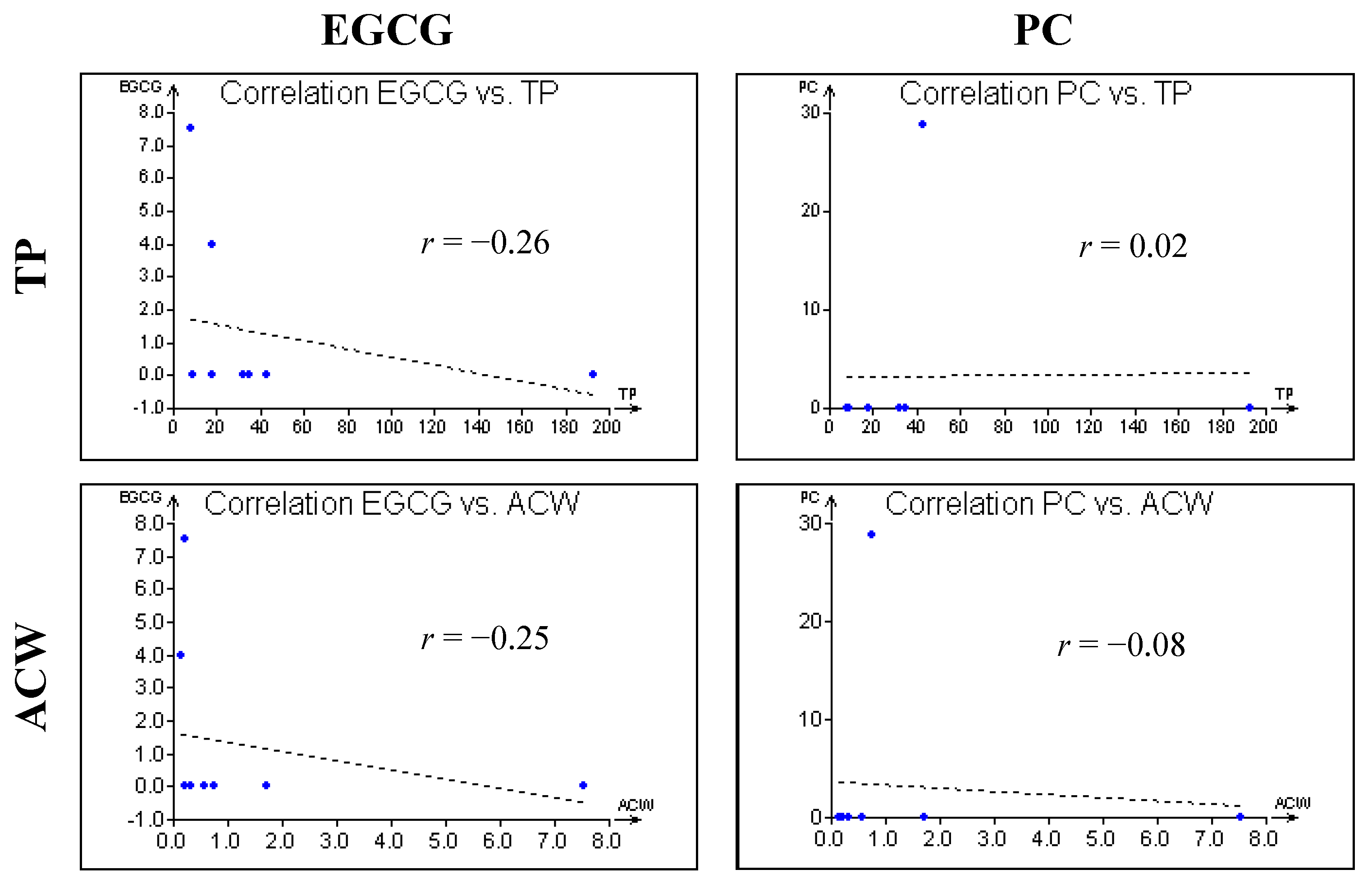
3. Experimental Section
3.1. Algal Samples
| Species | Algal Product | Type | Country of Origin |
|---|---|---|---|
| Eisenia bicyclis | Arame | Brown | Japan |
| Hizikia fusiformis | Hijiki | Brown | Japan |
| Laminaria japonica | Kombu | Brown | Japan |
| Undaria pinnatifida (W) | Wakame | Brown | Japan |
| Undaria pinnatifida (Wi) | Wakame-instant | Brown | Japan |
| Palmaria palmata | Dulse flakes Bio | Red | USA |
| Porphyra tenera | Nori flakes | Red | Japan |
| Chlorella pyrenoidosa | Chlorella Tabs | Green | Taiwan |
| Spirulina platensis | Spirulina Bio | Cyanobacteria | India |
3.2. Chemicals
3.3. Phenolic Compounds Extraction
- /1/
- distilled water (80 °C for 10 min in water bath with constant shaking);
- /2/
- methanol–water–acetic acid (30:69:1, v/v/v) (70 °C for 50 min in water bath with constant shaking);
- /3/
- 80% methanol (70 °C for 1 h in water bath with constant shaking);
- /4/
- 70% acetone (30 °C for 30 min in water bath with constant shaking);
- /5/
- 100% methanol (23 °C for 24 h, constant shaking).
3.4. Total Phenolic Content
3.5. HPLC Analysis of Selected Phenolic Compounds
3.6. Antioxidant Capacity of the Water Soluble Compounds (ACW)
3.7. Statistical and Data Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mišurcová, L. Chemical composition of seaweeds. In Handbook of Marine Macroalgae: Biotechnology and Applied Phycology; Kim, S.-K., Ed.; John Wiley & Sons: Chichester, UK, 2011; pp. 173–192. [Google Scholar]
- Ambrozova, J.V.; Misurcova, L.; Vicha, R.; Machu, L.; Samek, D.; Baron, M.; Mlcek, J.; Sochor, J.; Jurikova, T. Influence of extractive solvents on lipid and fatty acids content of edible freshwater algal and seaweed products, the green microalga Chlorella kessleri and the cyanobacterium Spirulina platensis. Molecules 2014, 19, 2344–2360. [Google Scholar] [CrossRef] [PubMed]
- Rop, O.; Mlcek, J.; Jurikova, T.; Neugebauerova, J.; Vabkova, J. Edible flowers—A new promising source of mineral elements in human nutrition. Molecules 2012, 17, 6672–6683. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [PubMed]
- Zern, T.L.; Fernandez, M.L. Cardioprotective effects of dietary polyphenols. J. Nutr. 2005, 135, 2291–2294. [Google Scholar] [PubMed]
- Li, Y.X.; Wijesekara, I.; Li, Y.; Kim, S.K. Phlorotannins as bioactive agents from brown algae. Process Biochem. 2011, 46, 2219–2224. [Google Scholar] [CrossRef]
- Thomas, N.V.; Kim, S.K. Potential pharmacological applications of polyphenolic derivates from marine brown algae. Environ. Toxicol. Pharmacol. 2011, 32, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Popov, I.; Lewin, G. Antioxidative homeostasis: Characterization by means of chemiluminescent technique. Methods Enzymol. 1999, 300, 437–456. [Google Scholar] [PubMed]
- Craft, B.D.; Kerrihard, A.L.; Amarowicz, R.; Pegg, R.B. Phenol-based antioxiadants and the in vitro methods used for their assessment. Compr. Rev. Food Sci. Food Saf. 2012, 11, 148–173. [Google Scholar] [CrossRef]
- Yuan, Y.V.; Walsh, N.A. Antioxidant and antiproliferative activities of extracts from a variety of edible seaweeds. Food Chem. Toxicol. 2006, 44, 1144–1150. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Escrig, A.; Jiménez-Jiménez, I.; Pulido, R.; Saura-Calixto, F. Antioxidant activity of fresh and processed edible seaweeds. J. Sci. Food Agric. 2001, 81, 530–534. [Google Scholar] [CrossRef]
- Marinho-Soriano, E.; Fonseca, P.C.; Carneiro, M.A.A.; Moreira, W.S.C. Seasonal variation in the chemical composition of two tropical seaweeds. Bioresour. Technol. 2006, 97, 2402–2406. [Google Scholar] [CrossRef] [PubMed]
- López, A.; Rico, M.; Rivero, A.; Suárez de Tangil, M. The effects of solvents on the phenolic contents and antioxidant activity of Stypocaulon scoparium algae extracts. Food Chem. 2011, 125, 1104–1109. [Google Scholar] [CrossRef]
- Li, S.; Li, S.K.; Gan, R.Y.; Song, F.L.; Kuang, L.; Li, H.B. Antioxidant capacities and total phenolic contents of infusions from 223 medicinal plants. Ind. Crops Prod. 2013, 51, 289–298. [Google Scholar] [CrossRef]
- Fu, L.; Xu, B.T.; Xu, X.R.; Gan, R.Y.; Zhang, Y.; Xia, E.Q.; Li, H.B. Antioxidant capacities and total phenolic contents of 62 fruits. Food Chem. 2011, 129, 345–350. [Google Scholar] [CrossRef]
- Deng, G.F.; Lin, X.; Xu, X.R.; Gao, L.L.; Xie, J.F.; Li, H.B. Antioxidant capacities and total phenolic contents of 56 vegetables. J. Funct. Foods 2013, 5, 260–266. [Google Scholar] [CrossRef]
- Dudonné, S.; Vitrac, X.; Coutière, P.; Woillez, M.; Mérillon, J.M. Comparative study of antioxidant properties and total phenolic content of 30 plant extracts of industrial interest using DPPH, ABTS, FRAP, SOD, and ORAC assays. J. Agric. Food Chem. 2009, 57, 1768–1774. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.W.; Kim, Y.J.; Lee, H.J.; Lee, C.Y. Cocoa has more phenolic phytochemicals and a higher antioxidant capacity than teas and red wine. J. Agric. Food Chem. 2003, 51, 7292–7295. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.M.; Kang, K.; Jeon, J.S.; Jho, E.H.; Kim, C.Y.; Nho, C.W.; Um, B.H. Isolation of phlorotannins from Eisenia bicyclis and their hepatoprotective effects against oxidative stress induced by tert-butyl hyperoxide. Appl. Biochem. Biotechnol. 2011, 165, 1296–1307. [Google Scholar] [CrossRef] [PubMed]
- Heo, S.J.; Cha, S.H.; Lee, K.W.; Cho, S.K.; Jeon, Y.J. Antioxidant activities of chlorophyta and phaeophyta from Jeju Island. Algae 2005, 20, 251–260. [Google Scholar] [CrossRef]
- Zhang, W.W.; Duan, X.J.; Huang, H.L.; Zhang, Y.; Wang, B.G. Evaluation of 28 marine algae from the Qingdao coast for antioxidative capacity and determination of antioxidant efficiency and total phenolic content of fractions and subfractions derived from Symphyocladia latiuscula (Rhodomelaceae). J. Appl. Phycol. 2007, 19, 97–108. [Google Scholar] [CrossRef]
- Yuan, Y.V.; Bone, D.E.; Carrington, M.F. Antioxidant activity of dulse (Palmaria palmata) extract evaluated in vitro. Food Chem. 2005, 91, 485–494. [Google Scholar] [CrossRef]
- Heo, S.J.; Cha, S.H.; Lee, K.W.; Jeon, Y.J. Antioxidant activities of red algae from Jeju Island. Algae 2006, 21, 149–156. [Google Scholar] [CrossRef]
- Li, H.B.; Cheng, K.W.; Wong, C.C.; Fan, K.W.; Chen, F.; Jiang, Y. Evaluation of antioxidant capacity and total phenolic content of different fractions of selected microalgae. Food Chem. 2007, 102, 771–776. [Google Scholar] [CrossRef]
- Liu, J.G.; Hou, C.W.; Lee, S.Y.; Chuang, Y.; Lin, C.C. Antioxidant effects and UVB protective activity of Spirulina (Arthrospira platensis) products fermented with lactic acid bacteria. Process Biochem. 2011, 46, 1405–1410. [Google Scholar] [CrossRef]
- Tomás-Barberán, F.A.; Clifford, M.N. Flavonones, chalcones and dihydrochalcones—Nature, occurrence and dietary burden. J. Sci. Food Agric. 2000, 80, 1073–1080. [Google Scholar] [CrossRef]
- Yoshie, Y.; Wang, W.; Petillo, D.; Suzuki, T. Distribution of catechins in Japanese seaweeds. Fish. Sci. 2000, 66, 998–1000. [Google Scholar] [CrossRef]
- Rodríguez-Bernaldo de Quirós, A.; Lage-Yusty, M.A.; López-Hernández, J. Determination of phenolic compounds in macroalgae for human consumption. Food Chem. 2010, 121, 634–638. [Google Scholar]
- Onofrejová, L.; Vašíčková, J.; Klejdus, B.; Stratil, P.; Mišurcová, L.; Kráčmar, S.; Kopecký, J.; Vacek, J. Bioactive phenols in algae: The application of pressurized-liquid and solid-phase extraction techniques. J. Pharm. Biomed. Anal. 2010, 51, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Kranl, K.; Schlesier, K.; Bitsch, R.; Hermann, H.; Rohe, M.; Böhm, V. Comparing antioxidative food additives and secondary plant products—Use of different assays. Food Chem. 2005, 93, 171–175. [Google Scholar] [CrossRef]
- Besco, E.; Braccioli, E.; Vertuani, S.; Ziosi, P.; Brazzo, F.; Bruni, R.; Sacchetti, G.; Manfredini, S. The use of photochemiluminescence for the measurement of the integral antioxidant capacity of baobab products. Food Chem. 2007, 102, 1352–1356. [Google Scholar] [CrossRef]
- Chua, M.T.; Tung, Y.T.; Chang, S.T. Antioxidant activities of ethanolic extracts from the twigs of Cinnamomum osmophloeum. Bioresour. Technol. 2008, 99, 1918–1925. [Google Scholar] [CrossRef] [PubMed]
- Punithavathi, V.R.; Stanely Mainzen Prince, P.; Kumar, M.R.; Selvakumari, C.J. Protective effects of gallic acid on hepatic lipid peroxide metabolism, glycoprotein components and lipids in streptozotocin-induced type II diabetic Wistar rats. J. Biochem. Mol. Toxicol. 2011, 25, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Peungvicha, P.; Temsiririrkkul, R.; Prasain, J.K.; Tezuka, Y.; Kadota, S.; Thirawarapan, S.S.; Watanabe, H. 4-Hydroxybeozoic acid: A hypoglycemic constituent of aqueous extract of Pandanus odorus root. J. Ethnopharmacol. 1998, 62, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Al-Hazzani, A.A.; Alshatwi, A.A. Catechin hydrate inhibits proliferation and mediates apoptosis of SiHa human cervical cancer cells. Food Chem. Toxicol. 2011, 49, 3281–3286. [Google Scholar] [CrossRef] [PubMed]
- Babich, H.; Zuckerbraun, H.L.; Weinerman, S.M. In vitro cytotoxicity of (–)-catechin gallate, a minor polyphenol in green tea. Toxicol. Lett. 2007, 171, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Terao, J.; Piskula, M.; Yao, Q. Protective Effect of Epicatechin, Epicatechin Gallate, and Quercetin on Lipid Peroxidation in Phospholipid Bilayers. Arch. Biochem. Biophys. 1994, 308, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Suganuma, M.; Okabe, S.; Oniyama, M.; Tada, Y.; Ito, H.; Fujiki, H. Wide distribution of [3H](–)-epigallocatechin gallate, a cancer preventive tea polyphenol, in mouse tissue. Carcinogenesis 1998, 19, 1771–1776. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.N.; Shankar, S.; Srivastava, R. Green tea catechin, epigallocatechin-3-gallate (EGCG): Mechanism, perspectives and clinical applications. Biochem. Pharmacol. 2011, 82, 1807–1821. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Bernaldo de Quirós, A.; Frecha-Ferreiro, S.; Vidal-Pérez, A.M.; López-Hernández, J. Antioxidant compounds in edible brown seaweeds. Eur. Food Res. Technol. 2010, 231, 495–498. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are not available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Machu, L.; Misurcova, L.; Vavra Ambrozova, J.; Orsavova, J.; Mlcek, J.; Sochor, J.; Jurikova, T. Phenolic Content and Antioxidant Capacity in Algal Food Products. Molecules 2015, 20, 1118-1133. https://doi.org/10.3390/molecules20011118
Machu L, Misurcova L, Vavra Ambrozova J, Orsavova J, Mlcek J, Sochor J, Jurikova T. Phenolic Content and Antioxidant Capacity in Algal Food Products. Molecules. 2015; 20(1):1118-1133. https://doi.org/10.3390/molecules20011118
Chicago/Turabian StyleMachu, Ludmila, Ladislava Misurcova, Jarmila Vavra Ambrozova, Jana Orsavova, Jiri Mlcek, Jiri Sochor, and Tunde Jurikova. 2015. "Phenolic Content and Antioxidant Capacity in Algal Food Products" Molecules 20, no. 1: 1118-1133. https://doi.org/10.3390/molecules20011118
APA StyleMachu, L., Misurcova, L., Vavra Ambrozova, J., Orsavova, J., Mlcek, J., Sochor, J., & Jurikova, T. (2015). Phenolic Content and Antioxidant Capacity in Algal Food Products. Molecules, 20(1), 1118-1133. https://doi.org/10.3390/molecules20011118






