Surface Properties and Photocatalytic Activity of KTaO3, CdS, MoS2 Semiconductors and Their Binary and Ternary Semiconductor Composites
Abstract
:1. Introduction
2. Results and Discussion
2.1. BET Surface Area
| Sample Label | KTaO3:CdS:MoS2/Molar Ratio | Preparation Method | BET Surface Area/[m2/g] | Pore Volume/[cm3/g] |
|---|---|---|---|---|
| KTaO3 | 1:0:0 | hydrothermal | 0.1 | 0.0001 |
| CdS | 0:1:0 | solvothermal | 1.2 | 0.0006 |
| MoS2 | 0:0:1 | hydrothermal | 1.8 | 0.0009 |
| CdS-MoS2 5-1 | 0:5:1 | solvothermal mixed solutions | 5.0 | 0.0026 |
| CdS-MoS2 4-1 | 0:4:1 | solvothermal mixed solutions | 11.1 | 0.0056 |
| CdS-MoS2 1-1 | 0:1:1 | solvothermal mixed solutions | 10.4 | 0.0052 |
| CdS-MoS2 1-5 | 0:1:5 | solvothermal mixed solutions | 1.0 | 0.0006 |
| KTaO3-CdS 10-1_MS | 10:1:0 | solvothermal mixed solutions | 17.5 | 0.0088 |
| KTaO3-CdS 10-1_C | 10:1:0 | hydro/solvothermal and calcination | 0.4 | 0.0002 |
| KTaO3-MoS2 10-1_MS | 10:0:1 | hydrothermal mixed solutions | 2.8 | 0.0014 |
| KTaO3-MoS2 10-1_C | 10:0:1 | hydrothermal and calcination | <0.1 | ~0 |
| KTaO3-CdS-MoS2 10-1-1_MS | 10:1:1 | solvothermal mixed solutions | 4.0 | 0.0019 |
| KTaO3-CdS-MoS2 10-1-1_C | 10:1:1 | hydro/solvothermal and calcination | <0.1 | ~0 |
| KTaO3-CdS-MoS2 10-5-1_MS | 10:5:1 | solvothermal mixed solutions | 10.3 | 0.0051 |
| KTaO3-CdS-MoS2 10-5-1_C | 10:5:1 | hydro/solvothermal and calcination | 0.5 | 0.0003 |
2.2. XRD Analysis
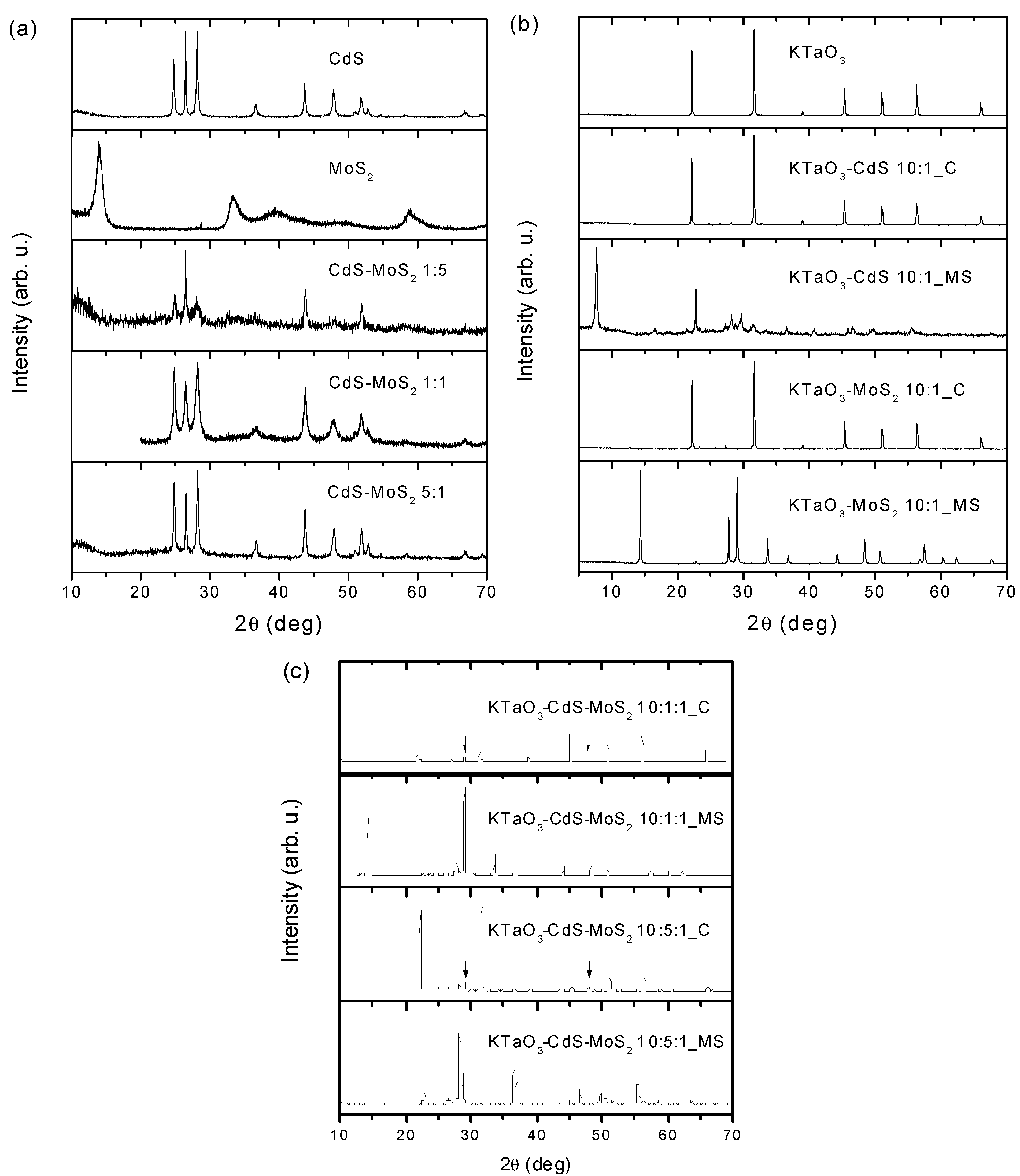
2.3. Morphology



2.4. UV-Vis Properties

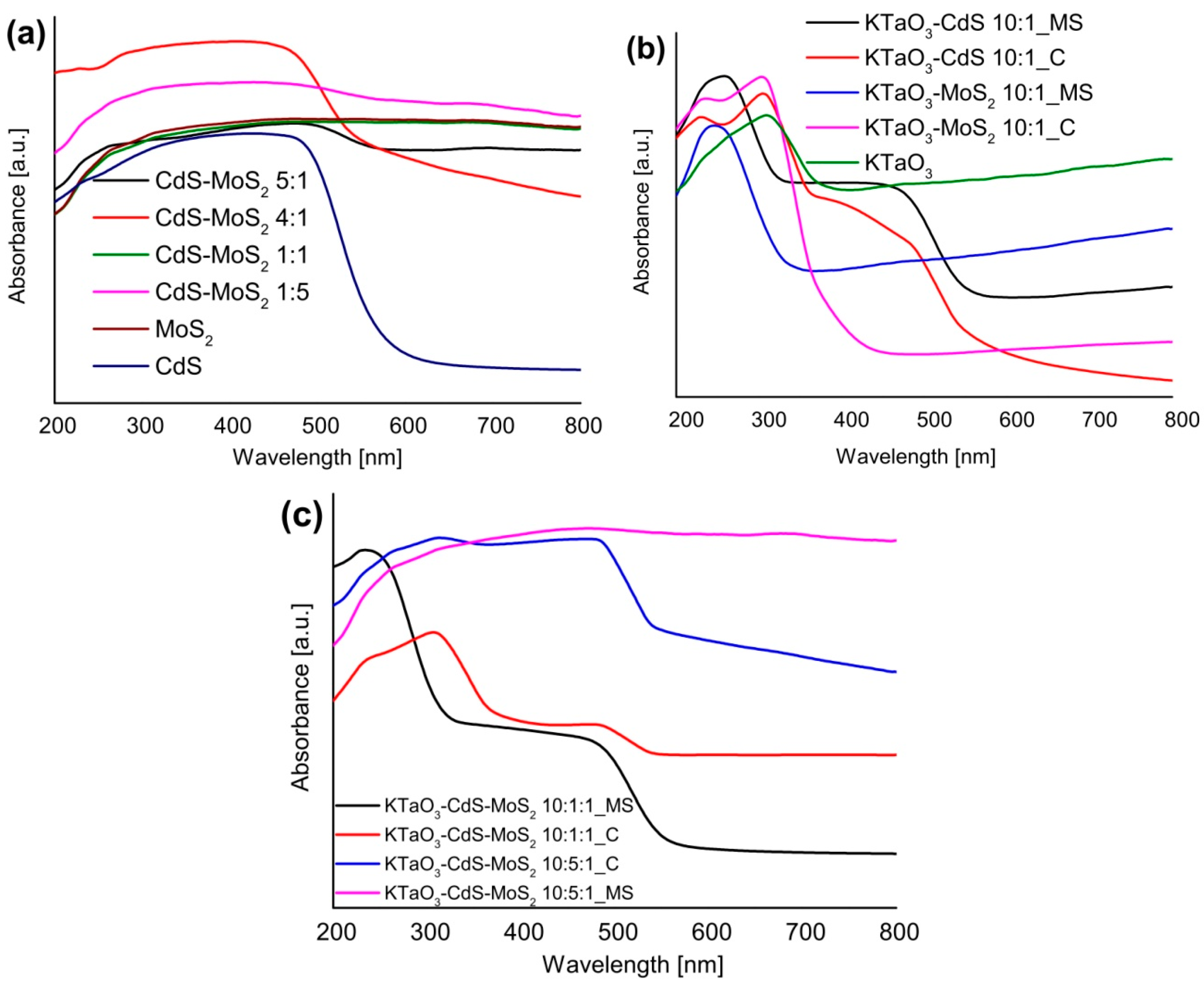
2.5. The Photocatalytic Degradation of Phenol in the Aqueous Phase
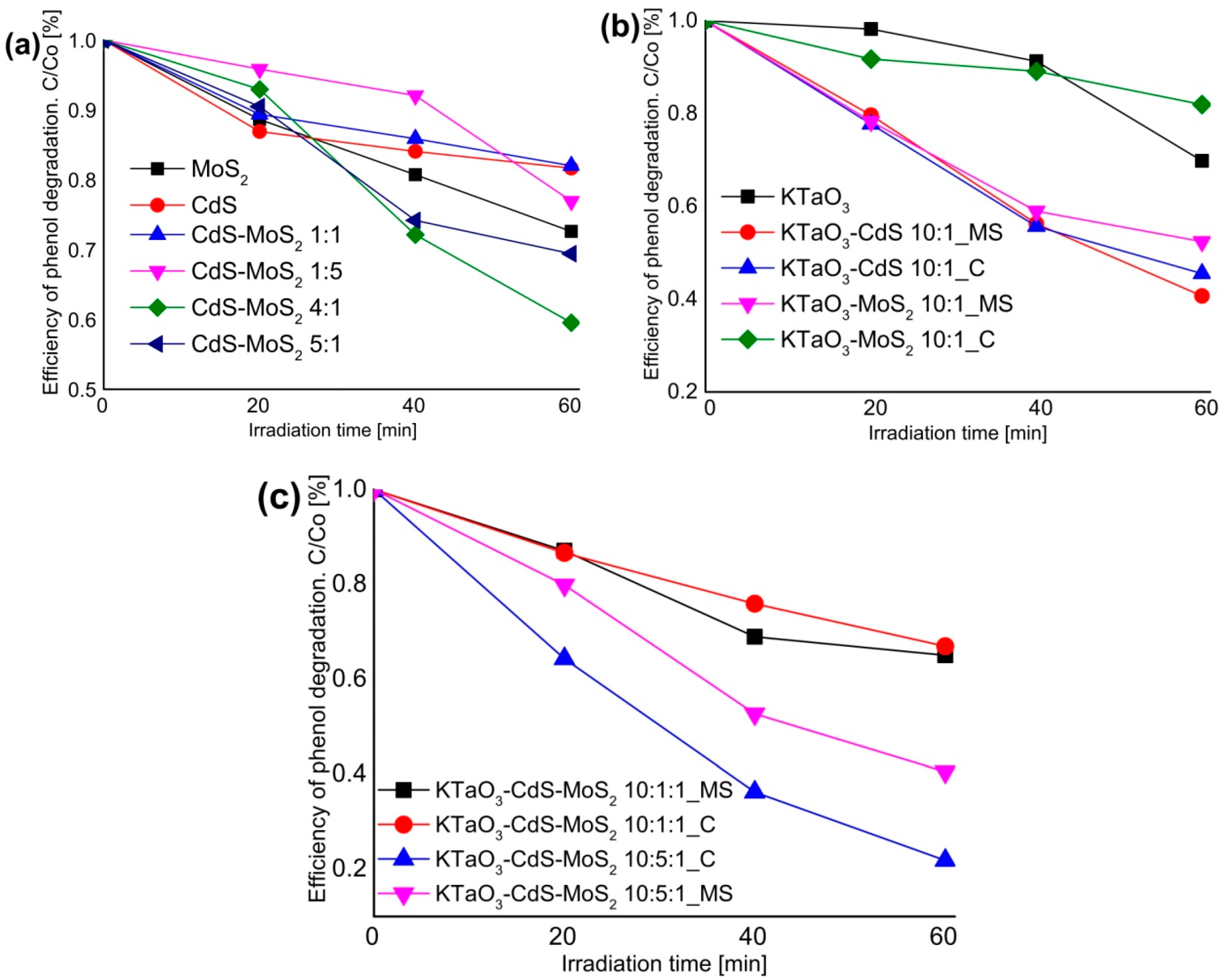
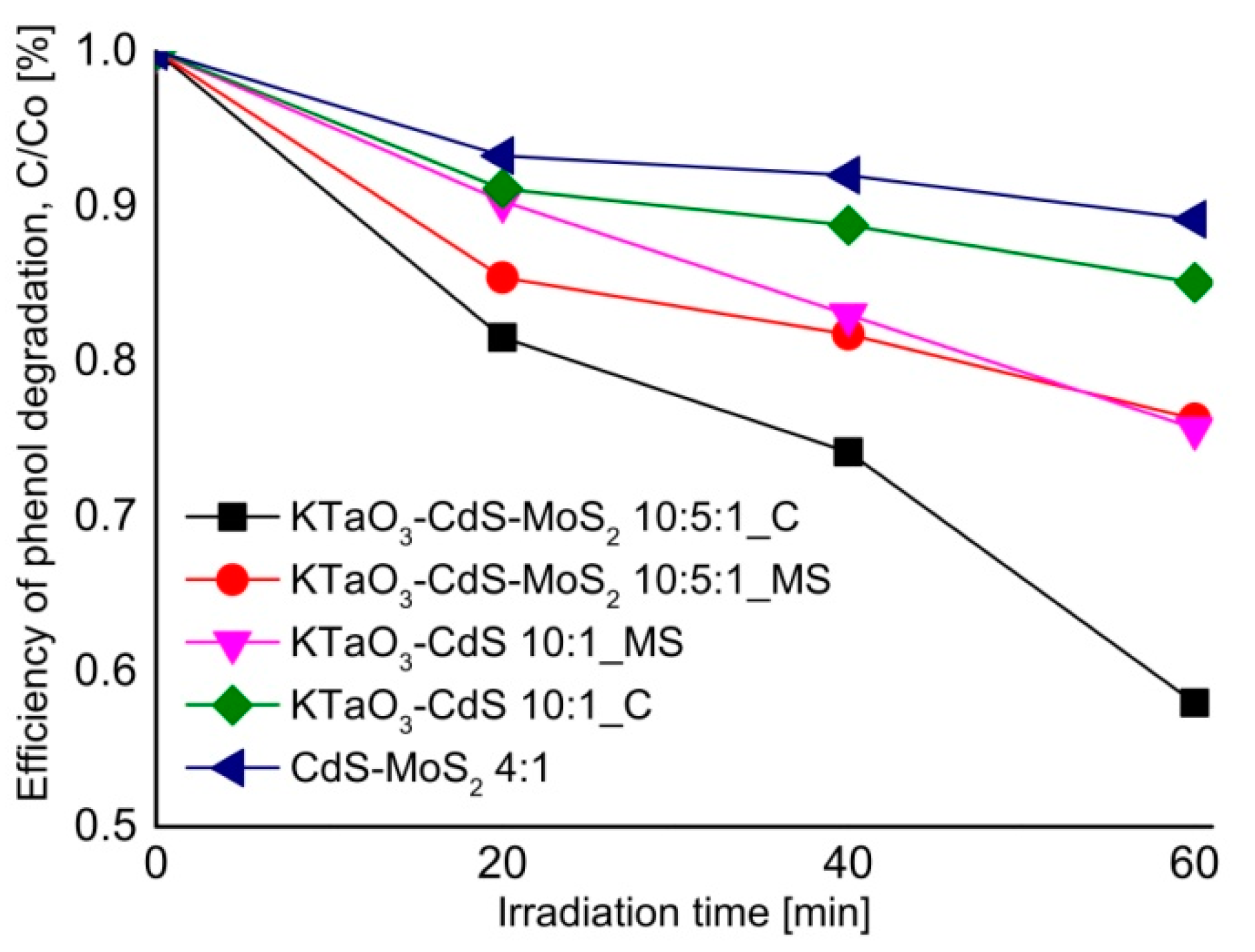
2.6. The Photocatalytic Degradation of Toluene in the Gas Phase
| Sample Label | Phenol Degradation Rate under UV-Vis/(µmol·dm−3·min−1) | Toluene Degradation after 1 h Irradiation (LEDs, λmax = 375 nm) [%] | |||
|---|---|---|---|---|---|
| 1st Cycle | 2nd Cycle | 3rd Cycle | 4th Cycle | ||
| KTaO3 | 0.79 | 64 | 63 | 42 | 37 |
| CdS | 0.61 | 57 | 57 | 57 | 52 |
| MoS2 | 0.90 | 46 | 23 | 22 | 22 |
| CdS-MoS2 1-5 | 0.77 | 57 | 53 | 44 | 27 |
| CdS-MoS2 1-1 | 0.62 | 61 | 53 | 62 | 52 |
| CdS-MoS2 5-1 | 0.81 | 70 | 60 | 49 | 48 |
| CdS-MoS2 4-1 | 1.41 | 53 | 56 | 60 | 62 |
| KTaO3-CdS 10-1_MS | 2.08 | 47 | 45 | 41 | 40 |
| KTaO3-CdS 10-1_C | 1.75 | 53 | 48 | 52 | 50 |
| KTaO3-MoS2 10-1_MS | 1.69 | 55 | 51 | 49 | 51 |
| KTaO3-MoS2 10-1_C | 0.55 | 46 | 34 | 37 | 35 |
| KTaO3-CdS-MoS2 10-1-1_MS | 1.15 | 50 | 52 | 48 | 39 |
| KTaO3-CdS-MoS2 10-1-1_C | 1.11 | 53 | 54 | 49 | 40 |
| KTaO3-CdS-MoS2 10-5-1_MS | 1.99 | 50 | 41 | 43 | 41 |
| KTaO3-CdS-MoS2 10-5-1_C | 2.81 | 48 | 48 | 50 | 46 |
| TiO2 (P25) | 2.87 | 98 | 96 | 95 | 95 |
2.7. The Origin of Photocatalytic Activity
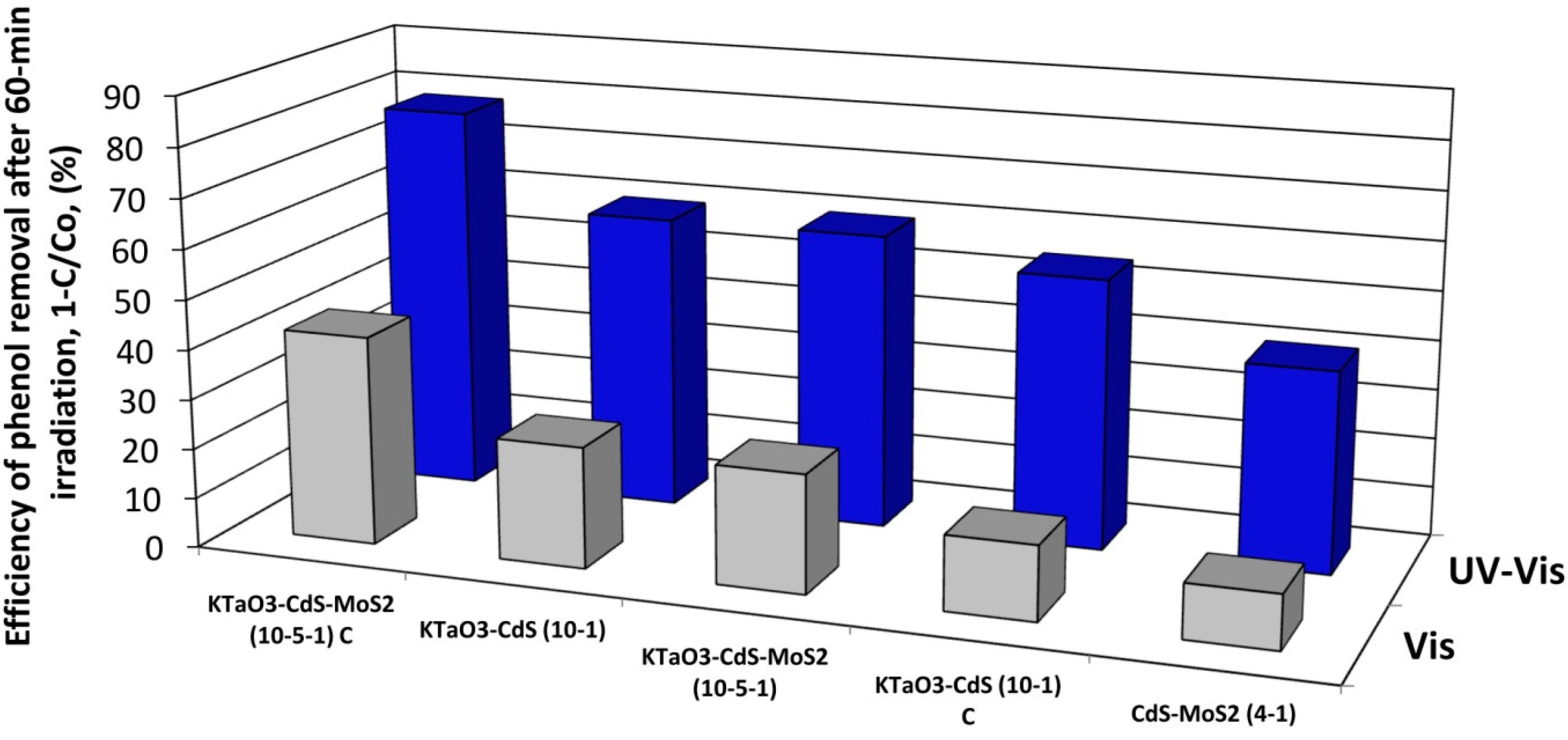
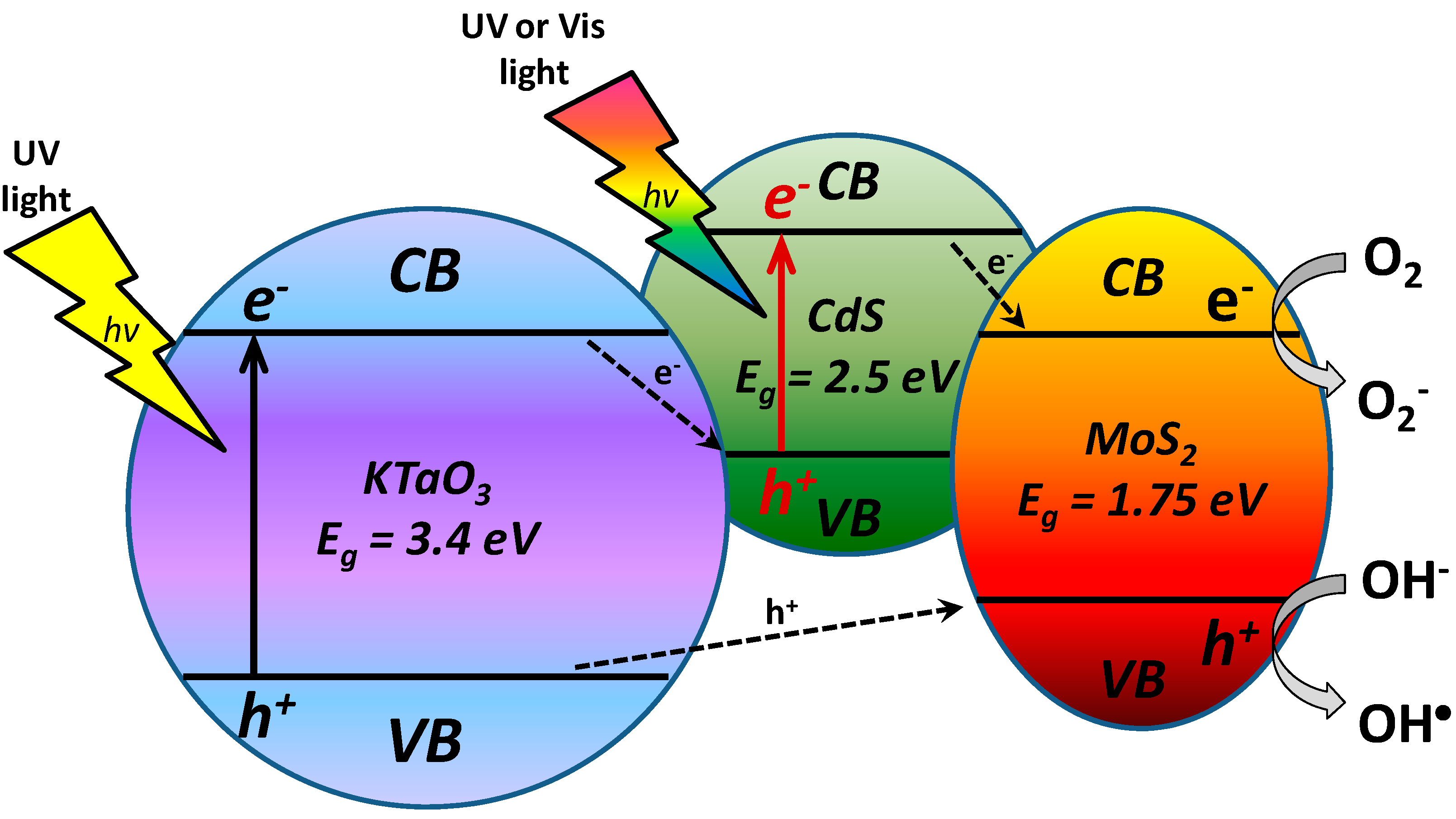
3. Experimental
3.1. Materials and Instruments
3.2. Synthesis of Single KTaO3, CdS and MoS2 Semiconductors
3.3. Synthesis of Binary and Ternary Semiconductor Composites
3.4. Measurement of Photocatalytic Activity in the Aqueous Phase
3.5. Measurement of Photocatalytic Activity in the Gas Phase
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fujishima, A.; Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 1972, 238, 37–38. [Google Scholar]
- Chen, X.B.; Mao, S.S. Titanium dioxide nanomaterials: Synthesis, properties, modifications, and applications. Chem. Rev. 2007, 107, 2891–2959. [Google Scholar]
- Tong, H.; Ouyang, S.X.; Bi, Y.P.; Umezawa, N.; Oshikiri, M.; Ye, J.H. Nanophotocatalytic materials: Possibilities and challenges. Adv. Mater. 2012, 24, 229–251. [Google Scholar]
- Bahnemann, D. Photocatalytic water treatment: Solar energy applications. Sol. Energy 2004, 77, 445–459. [Google Scholar]
- Ahmad, M.; Ahmed, E.; Zhang, Y.W.; Khalid, N.R.; Xu, J.F.; Ullah, M.; Hong, Z.L. Preparation of highly efficient Al-doped ZnO photocatalyst by combustion synthesis. Curr. Appl. Phys. 2013, 4, 697–704. [Google Scholar]
- Liao, C.H.; Huang, C.W.; Wu, J.C.S. Hydrogen production from semiconductor-based photocatalysis via water splitting. Catalysts 2012, 2, 490–516. [Google Scholar]
- Zhang, N.; Ciriminna, R.; Pagliaro, M.; Xu, Y.-J. Nanochemistry-derived Bi2WO6 nanostructures: Towards production of sustainable chemicals and fuels induced by visible light. Chem. Soc. Rev. 2014, 43, 5276–5287. [Google Scholar]
- Zhang, N.; Zhang, Y.; Xu, Y.-J. Recent progress on graphene-based photocatalysts: Current status and future perspectives. Nanoscale 2012, 4, 5792–5813. [Google Scholar]
- Zhang, N.; Liu, S.; Xu, Y.-J. Recent progress on metal core@semiconductor shell nanocomposites as a promising type of photocatalyst. Nanoscale 2012, 4, 2227–2238. [Google Scholar]
- Pan, X.; Yang, M.-Q.; Fu, X.; Zhang, N.; Xu, Y.-J. Defective TiO2 with oxygen vacancies: Synthesis, properties and photocatalytic applications. Nanoscale 2013, 5, 3601–3614. [Google Scholar]
- Yang, M.-Q.; Xu, Y.-J. Selective photoredox using graphene-based composite photocatalysts. Phys. Chem. Chem. Phys. 2013, 15, 19102–19118. [Google Scholar]
- Fujishima, A.; Zhang, X.; Tryk, D.A. TiO2 photocatalysis and related surface phenomena. Surf. Sci. Rep. 2008, 63, 515–582. [Google Scholar]
- Marschall, R. Semiconductor composites: Strategies for enhancing charge carrier separation to improve photocatalytic activity. Adv. Funct. Mater. 2014, 17, 2420–2440. [Google Scholar]
- Yang, G.; Yan, Z.; Xiao, T. Preparation and characterization of SnO/ZnO/TiO2 composite semiconductor with enhanced photocatalytic activity. Appl. Surf. Sci. 2012, 258, 8704–8712. [Google Scholar]
- Emeline, A.V.; Kuznetsov, V.N.; Ryabchuk, V.K.; Serpone, N. On the way to the creation of next generation photoactive materials. Environ. Sci. Pollut. Res. 2012, 19, 3666–3675. [Google Scholar]
- Serpone, N.; Emeline, A.V. Semiconductor photocatalysis—Past, present, and future outlook. J. Phys. Chem. Lett. 2012, 3, 673–677. [Google Scholar]
- Zhang, Y.; Tang, Z.-R.; Fu, H.; Xu, Y.-J. TiO2-graphene nanocomposites for gas-phase photocatalytic degradation of volatile aromatic pollutant: Is TiO2-graphene truly different from other TiO2-carbon composite materials? ACS Nano 2010, 4, 7303–7314. [Google Scholar]
- Kato, H.; Kudo, A. Photocatalytic water splitting into H2 and O2 over various tantalate photocatalysts. Catal. Today 2003, 78, 561–569. [Google Scholar]
- Liu, J.W.; Chen, G.; Li, Z.H.; Zhang, Z.G. Hydrothermal synthesis and photocatalytic properties of ATaO3 and AnbO3 (A = Na and K). Int. J. Hydrogen Energy 2007, 32, 2269–2272. [Google Scholar]
- Kudo, A. Development of photocatalyst materials for water splitting. Int. J. Hydrog. Energy 2006, 31, 197–202. [Google Scholar]
- Lin, W.; Chen, C.; Hu, C.; Teng, H. NaTaO3 photocatalysts of different crystalline structures for water splitting into H2 and O2. Appl. Phys. Lett. 2006, 89, 211904. [Google Scholar]
- Torres-Martínez, L.M.; Cruz-López, A.; Juárez-Ramírez, I.; Meza-de la Rosa, M.E. Methylene blue degradation by NaTaO3 sol-gel doped with Sm and La. J. Hazard. Mater. 2009, 165, 774–779. [Google Scholar]
- Zhu, H.; Jiang, R.; Xiao, L.; Chang, Y.; Guan, Y.; Li, X.; Zeng, G. Photocatalytic decolorization and degradation of Congo Red on innovative crosslinked chitosan/nano-CdS composite catalyst under visible light irradiation. J. Hazard. Mater. 2009, 169, 933–940. [Google Scholar]
- Zhai, T.Y.; Fang, X.S.; Bando, Y.S.; Liao, Q.; Xu, X.J.; Zeng, H.B.; Ma, Y.; Yao, J.N.; Golberg, D. Morphology-dependent stimulated emission and field emission of ordered CdS nanostructure arrays. ACS Nano 2009, 3, 949–959. [Google Scholar]
- Dongre, J.K.; Nogriya, V.; Ramrakhiani, M. Structural, optical and photoelectrochemical characterization of CdS nanowire synthesized by chemical bath deposition and wet chemical etching. Appl. Surf. Sci. 2009, 255, 6115–6120. [Google Scholar]
- Zhang, H.; Zhu, Y. Significant visible photoactivity and antiphotocorrosion performance of CdS photocatalysts after monolayer polyaniline hybridization. J. Phys. Chem. C 2010, 114, 5822–5826. [Google Scholar]
- Zhong, M.; Shi, J.; Xiong, F.; Zhang, W.; Li, C. Enhancement of photoelectrochemical activity of nanocrystalline CdS photoanode by surface modification with TiO2 for hydrogen production and electricity generation. Sol. Energy 2012, 86, 756–763. [Google Scholar]
- Li, X.; Xia, T.; Xu, C.; Murowchick, J.; Chen, X. Synthesis and photoactivity of nanostructured CdS–TiO2 composite catalysts. Catal. Today 2014, 225, 64–73. [Google Scholar]
- He, D.; Chen, M.; Teng, F.; Li, G.; Shi, H.; Wang, J.; Xu, M.; Lu, T.; Ji, X.; Lv, Y. Enhanced cyclability of CdS/TiO2 photocatalyst by stable interface structure. Superlattices Microstruct. 2012, 51, 799–808. [Google Scholar]
- Panigrahi, S.; Basak, D. Morphology driven ultraviolet photosensitivity in ZnO–CdS composite. J. Colloid Interface Sci. 2011, 364, 10–17. [Google Scholar]
- Jana, T.K.; Pal, A.; Chatterjee, K. Self assembled flower like CdS–ZnO nanocomposite and its photo catalytic activity. J. Alloys Compd. 2014, 583, 510–515. [Google Scholar]
- Liu, S.; Li, H.; Yan, L. Synthesis and photocatalytic activity of three-dimensional ZnS/CdS composites. Mater. Res. Bull. 2013, 48, 3328–3334. [Google Scholar]
- Liu, X.; Yan, Y.; Da, Z.; Shi, W.; Ma, C.; Lv, P.; Tang, Y.; Yao, G.; Wu, Y.; Huo, P.; et al. Significantly enhanced photocatalytic performance of CdS coupled WO3 nanosheets and the mechanism study. Chem. Eng. J. 2014, 241, 243–250. [Google Scholar]
- Zhao, Y.; Zhang, Y.; Yang, Z.; Yan, Y.; Sun, K. Synthesis of MoS2 and MoO2 for their applications in H2 generation and lithium ion batteries: A review. Sci. Technol. Adv. Mater. 2013, 14, 043501. [Google Scholar]
- Zong, X.; Wu, G.; Yan, H.; Ma, G.; Shi, J.; Wen, F.; Wang, L.; Li, C. Photocatalytic H2 Evolution on MoS2/CdS Catalysts under Visible Light Irradiation. J. Phys. Chem. C 2010, 114, 1963–1968. [Google Scholar]
- Chen, G.; Li, D.; Li, F.; Fan, Y.; Zhao, H.; Luo, Y.; Yu, R.; Meng, O. Ball-milling combined calcination synthesis of MoS2/CdS photocatalysts for high photocatalytic H2 evolution activity under visible light irradiation. Appl. Cat. A 2012. [Google Scholar]
- Liu, Y.; Yu, Y.X.; Zhang, W.D. MoS2/CdS Heterojunction with high photoelectrochemical activity for H2 evolution under visible light: The Role of MoS2. J. Phys. Chem. C 2013, 117, 12949–12957. [Google Scholar]
- Liu, Y.; Yu, H.; Quan, X.; Chen, S. Green synthesis of feather-shaped MoS2/CdS photocatalyst for effective hydrogen production. Int. J. Photoenergy 2013, 2013, 247516–247520. [Google Scholar]
- Zong, X.; Yan, H.; Wu, G.; Ma, G.; Wen, F.; Wang, L.; Li, C. Enhancement of photocatalytic H2 evolution on CdS by loading MoS2 as cocatalyst under visible light irradiation. J. Am. Chem. Soc. 2008, 130, 7176–7177. [Google Scholar]
- Wiedemeier, H.; Khan, A.A. Phase studies in the system managanese sulfioe–cadmium sulfide. Trans. Metall. Soc. AIME 1968, 242, 1969–1972. [Google Scholar]
- Hu, C.-C.; Yeh, T.-F.; Teng, H. Pyrochlore-like K2Ta2O8 synthesized from different methods as efficient photocatalysts for water splitting. Catal. Sci. Technol. 2013, 3, 1798–1804. [Google Scholar]
- Zhong, S.; Zhang, L.; Huang, Z.; Wang, S. Mixed-solvothermal synthesis of CdS micro/nanostructures and their optical properties. Appl. Surf. Sci. 2011, 257, 2599–2603. [Google Scholar]
- Zou, Y.; Hu, Y.; Gu, H.; Wang, Y. Optical properties of octahedral KTaO3 nanocrystalline. Mater. Chem. Phys. 2009, 115, 151–153. [Google Scholar]
- Adhikari, R.; Malla, S.; Gyawali, G.; Sekino, T.; Lee, S.W. Synthesis, characterization and evaluation of the photocatalytic performance of Ag-CdMoO4 solar light driven plasmonic photocatalysts. Mat. Res. Bull. 2013, 48, 3367–3373. [Google Scholar]
- Wang, W.-S.; Zhen, L.; Xu, C.-Y.; Shao, W.-Z.; Chen, Z.-L. Formation of CdMO4 porous hollow nanospheres via self-assembly accompanied with Ostwald ripening process and their photocatalytic performance. CrystEngComm 2013, 15, 8014–8021. [Google Scholar]
- Wang, S.; Li, G.; Du, G.; Jiang, X.; Feng, C.; Guo, Z.; Kim, S. Hydrothermal synthesis of molybdenum disulfide for lithium ion battery applications. Chin. J. Chem. Eng. 2010, 18, 910–913. [Google Scholar]
- Nischk, M.; Mazierski, P.; Gazda, M.; Zaleska, A. Ordered TiO2 nanotubes: The effect of preparation parameters on the photocatalytic activity in air purification process. Appl. Catal. B 2014, 144, 674–685. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bajorowicz, B.; Cybula, A.; Winiarski, M.J.; Klimczuk, T.; Zaleska, A. Surface Properties and Photocatalytic Activity of KTaO3, CdS, MoS2 Semiconductors and Their Binary and Ternary Semiconductor Composites. Molecules 2014, 19, 15339-15360. https://doi.org/10.3390/molecules190915339
Bajorowicz B, Cybula A, Winiarski MJ, Klimczuk T, Zaleska A. Surface Properties and Photocatalytic Activity of KTaO3, CdS, MoS2 Semiconductors and Their Binary and Ternary Semiconductor Composites. Molecules. 2014; 19(9):15339-15360. https://doi.org/10.3390/molecules190915339
Chicago/Turabian StyleBajorowicz, Beata, Anna Cybula, Michał J. Winiarski, Tomasz Klimczuk, and Adriana Zaleska. 2014. "Surface Properties and Photocatalytic Activity of KTaO3, CdS, MoS2 Semiconductors and Their Binary and Ternary Semiconductor Composites" Molecules 19, no. 9: 15339-15360. https://doi.org/10.3390/molecules190915339
APA StyleBajorowicz, B., Cybula, A., Winiarski, M. J., Klimczuk, T., & Zaleska, A. (2014). Surface Properties and Photocatalytic Activity of KTaO3, CdS, MoS2 Semiconductors and Their Binary and Ternary Semiconductor Composites. Molecules, 19(9), 15339-15360. https://doi.org/10.3390/molecules190915339





