A Galactose-Binding Lectin Isolated from Aplysia kurodai (Sea Hare) Eggs Inhibits Streptolysin-Induced Hemolysis
Abstract
:1. Introduction
2. Results and Discussion
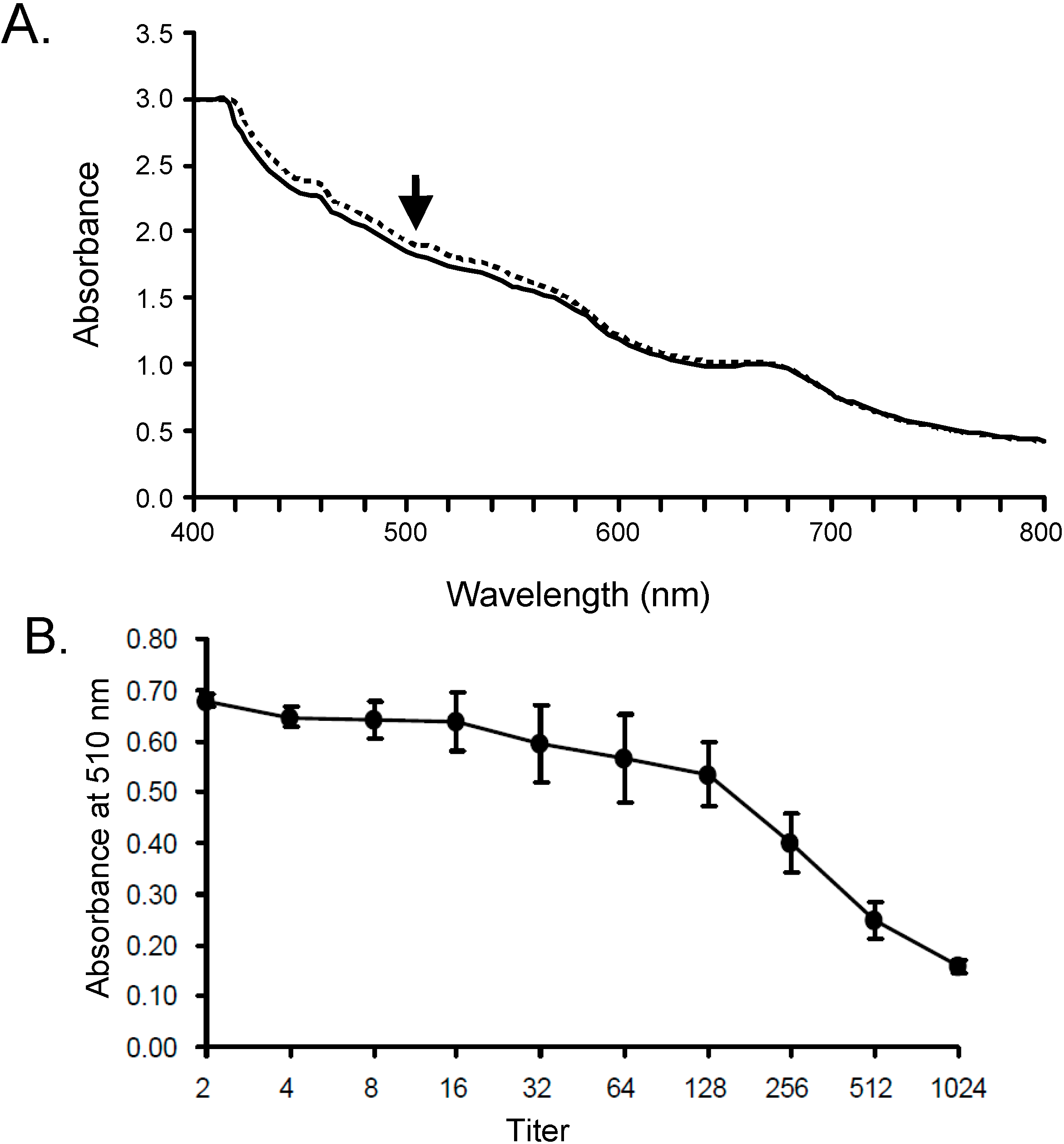
2.1. Optimization of Hemolysis Assay by SLO
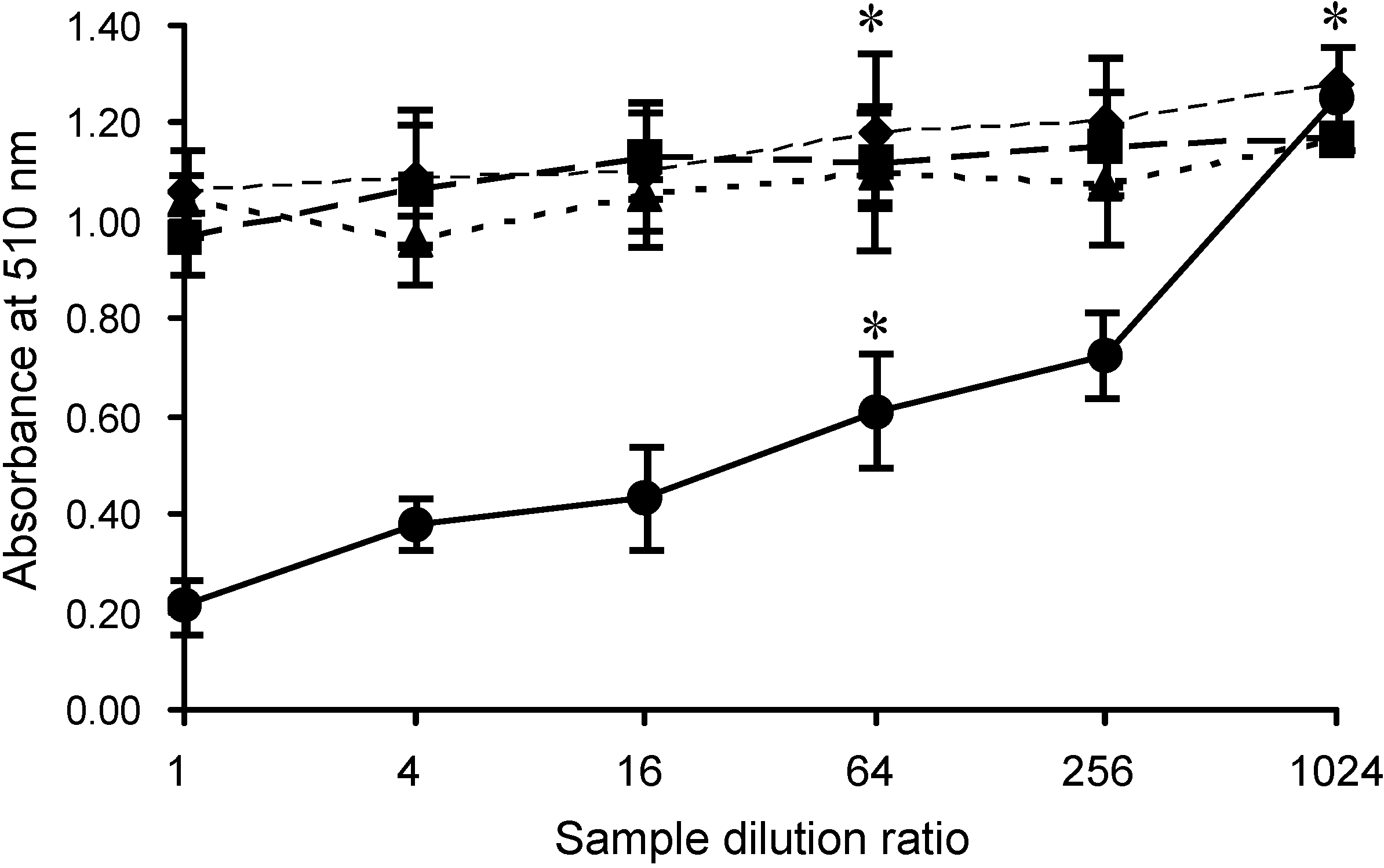
2.2. Specific Inhibition of SLO-Induced Hemolysis by AKL
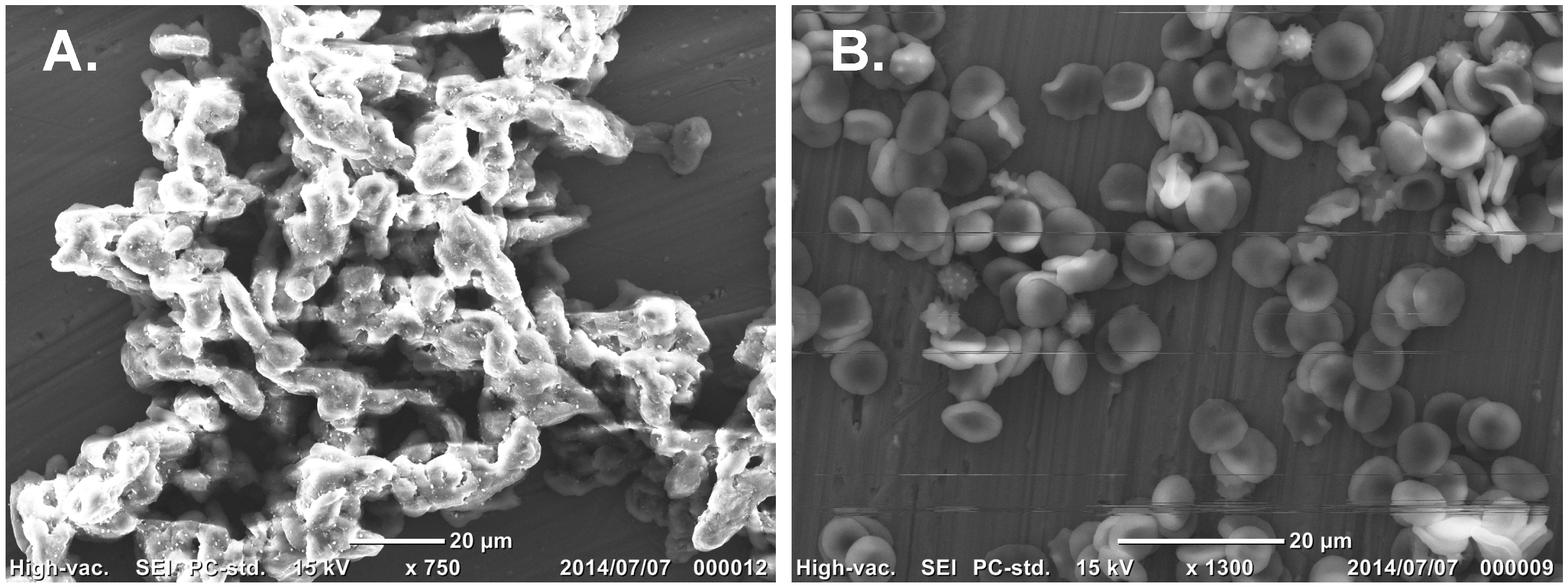
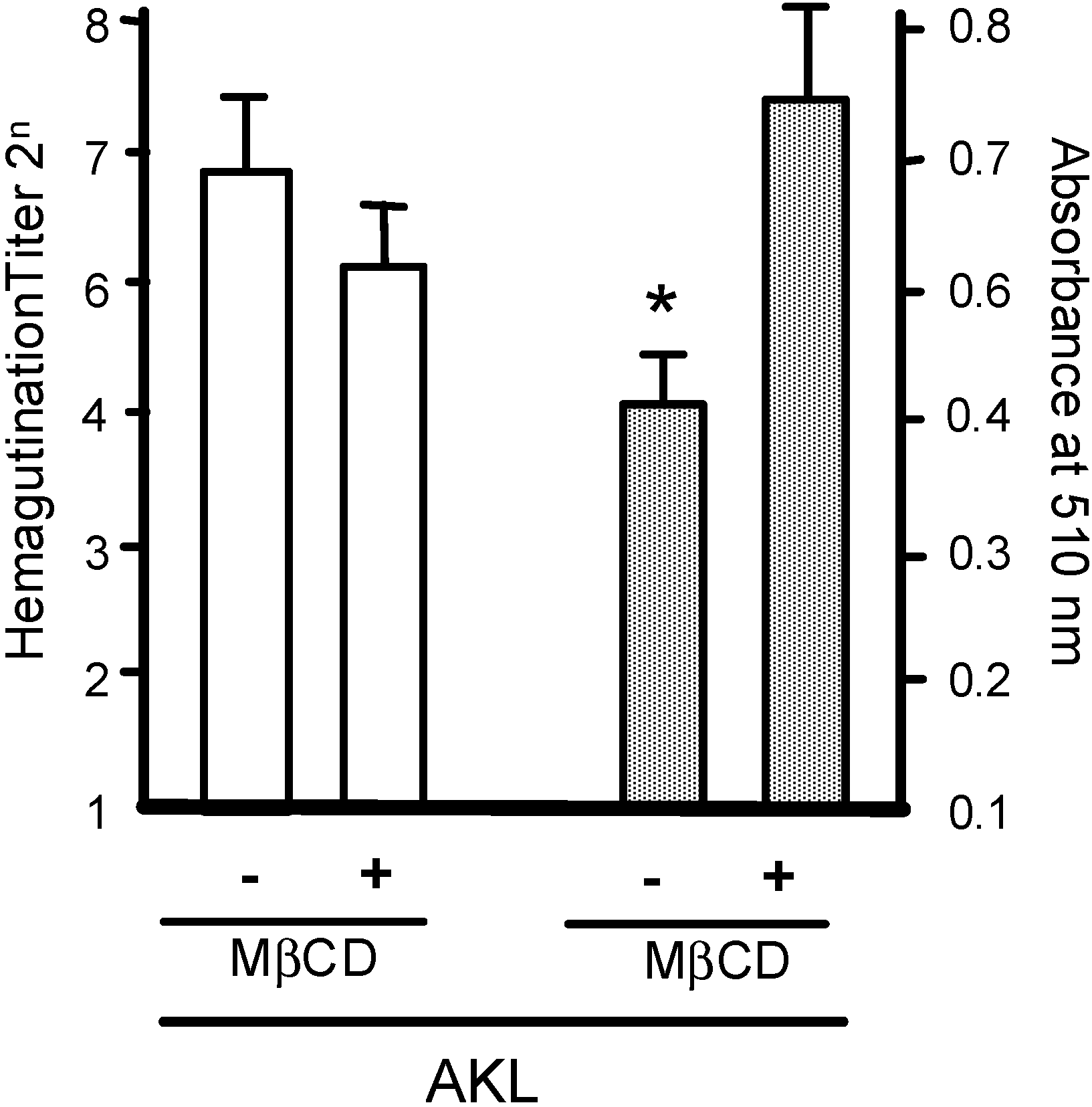
2.3. Treatment of RBCs with MβCD Blocks the Inhibitory Effect of AKL
2.4. Inhibition of S. pyogenes Growth by AKL
2.5. Stimulatory Effect of ECA on Growth of S. pyogenes
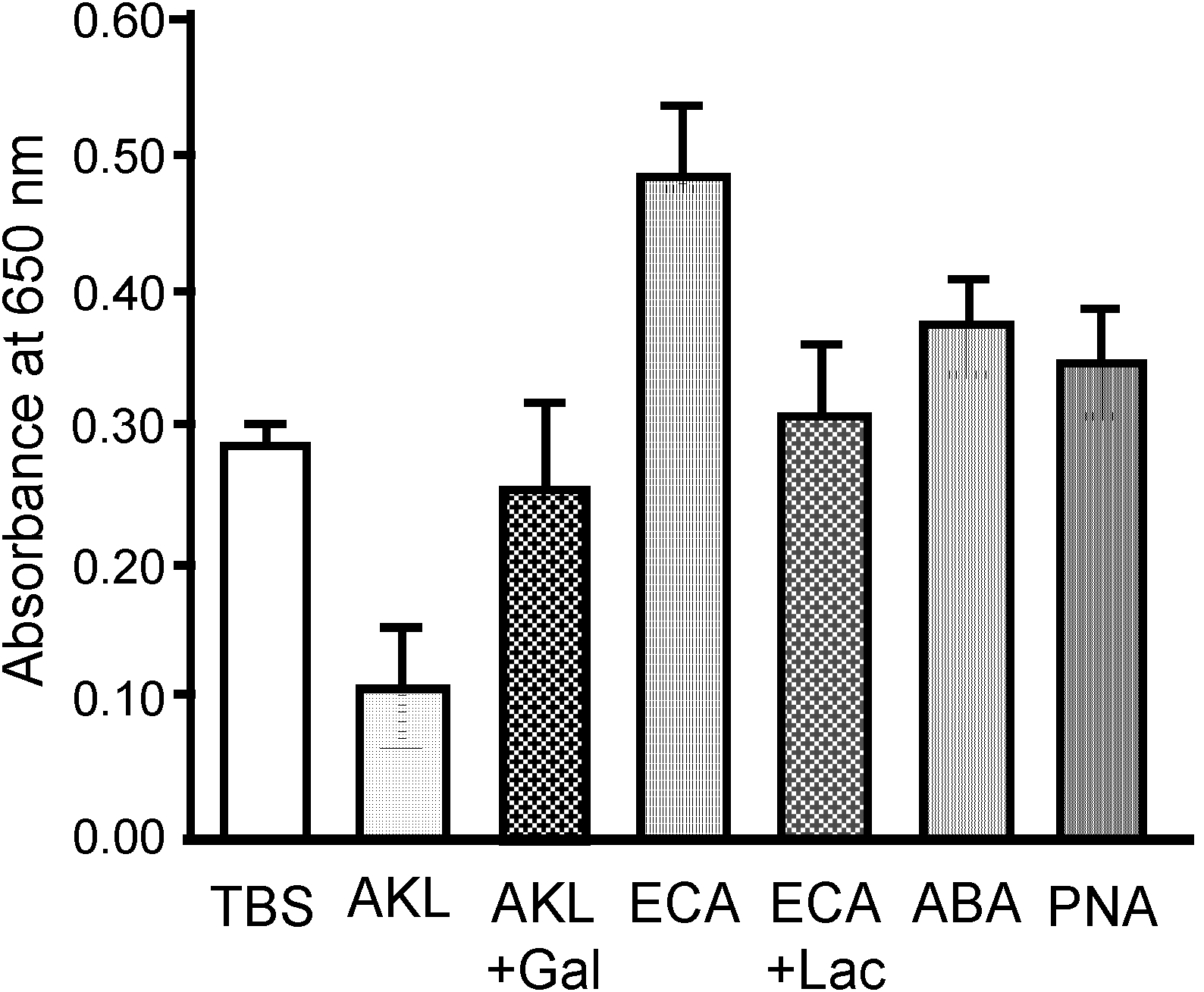
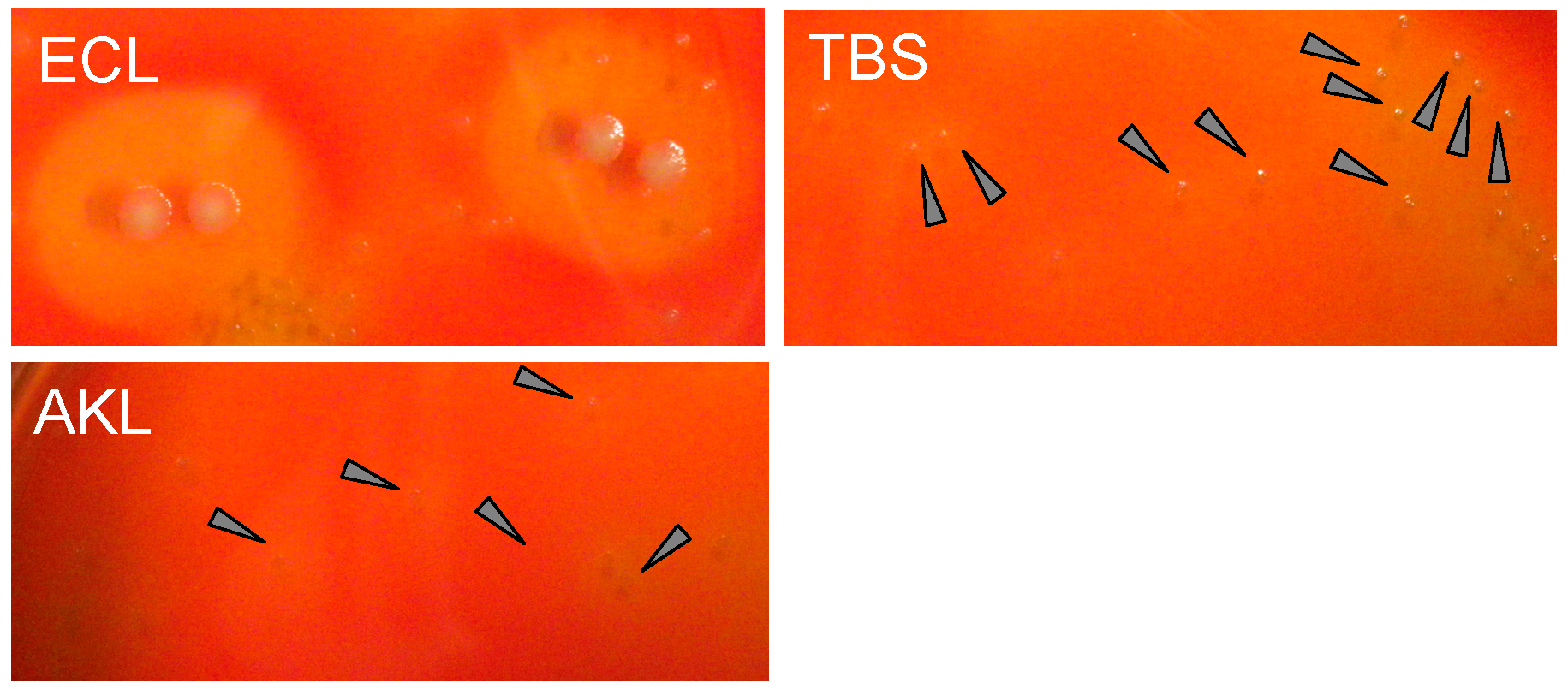
2.6. Bacteriostatic Activity of AKL
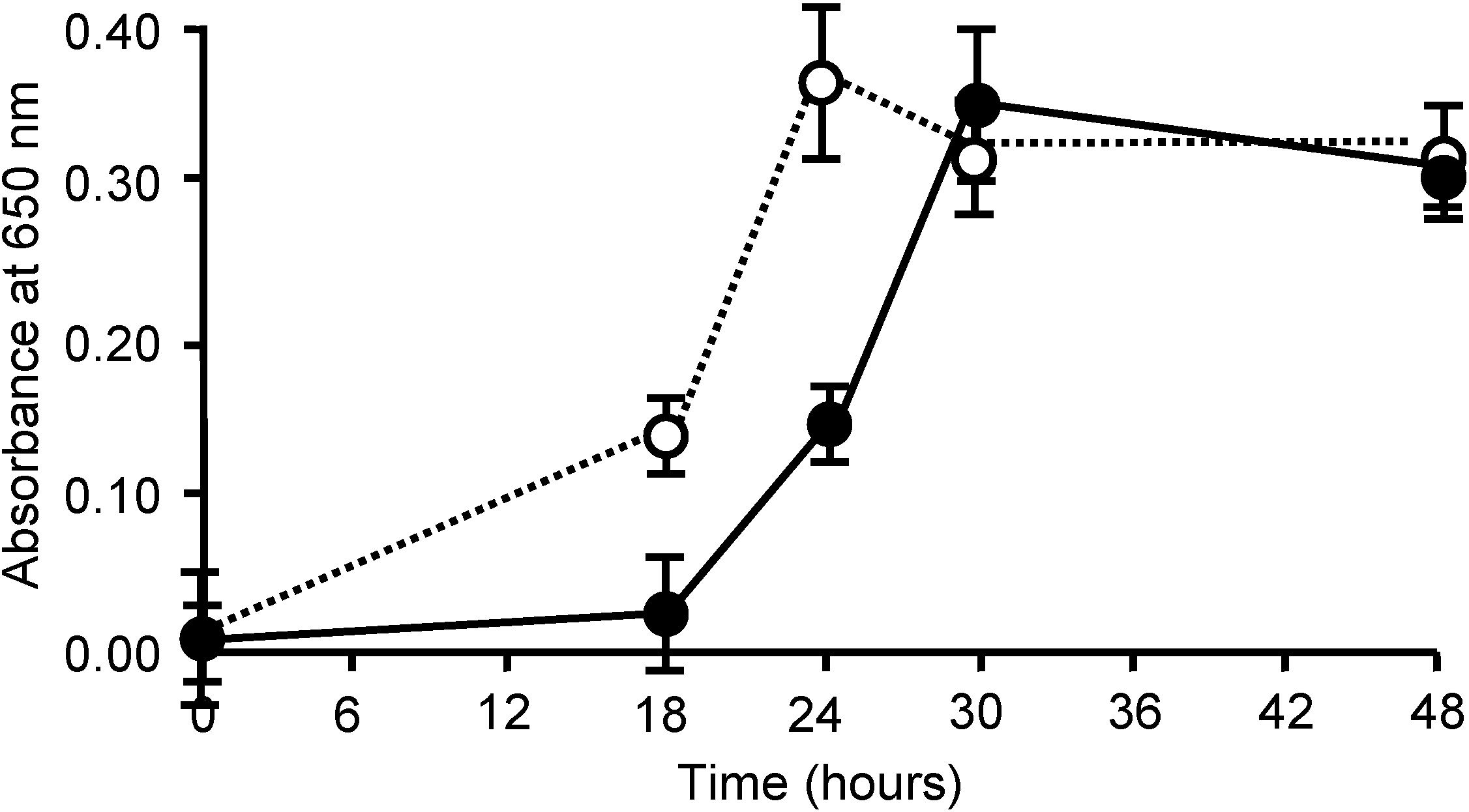
3. Experimental Section
3.1. Bacterial Strain, Culture, and Preparation of SLO
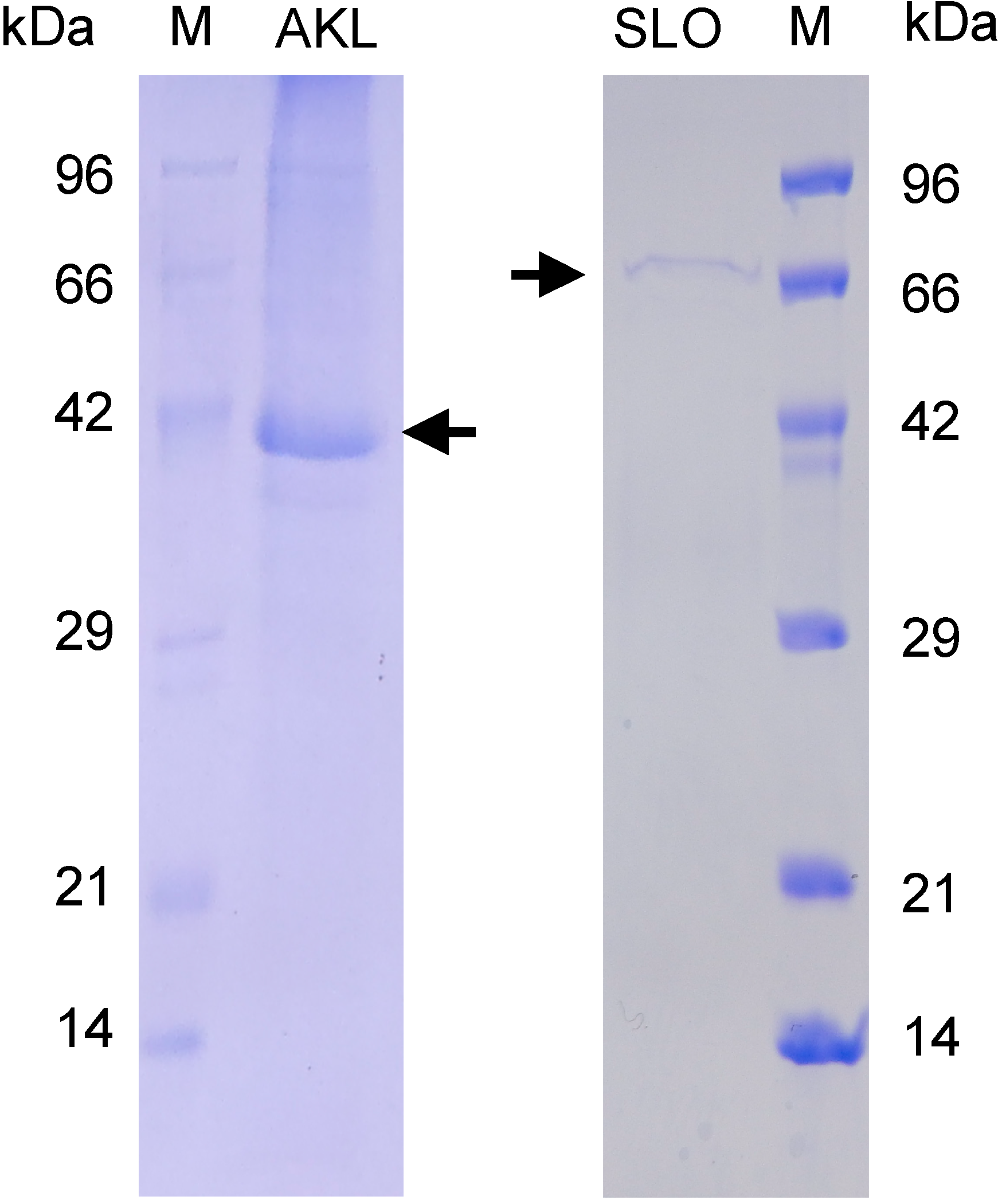
3.2. Lectin Preparation
3.3. RBC Preparation
3.4. Hemagglutination Assay and Sugar Binding Specificity
3.5. Hemolytic Activity of SLO
3.6. SEM Observationof AKL-Induced Hemagglutination in the Presence of SLO
3.7. Evaluation of AKL-Dependent Inhibition of Hemolysis Using Rbcs Treated with Methyl-β-d-Cyclodextrin
3.8. Effects of Lectins on SLO-Induced Hemolysis
3.9. Effects of Lectins on Growth of S. pyogenes
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Weis, W.I.; Drickamer, K. Structural basis of lectin-carbohydrate recognition. Annu. Rev. Biochem. 1996, 65, 441–473. [Google Scholar] [CrossRef] [PubMed]
- Rowe, P.C. Escherichia coli O157: H7, other verotoxin-producing E. coli and the hemolytic uremic syndrome in childhood. Can. J. Infect. Dis. 1995, 6, 105–110. [Google Scholar]
- Suzuki, J.; Kobayashi, S.; Kagaya, K.; Fukuzawa, Y. Heterogeneity of hemolytic efficiency and isoelectric point of streptolysin O. Infect. Immun. 1988, 56, 2474–2478. [Google Scholar] [PubMed]
- Shiseki, M.; Miwa, K.; Nemoto, Y.; Kato, H.; Suzuki, J.; Sekiya, K.; Murai, T.; Kikuchi, T.; Yamashita, N.; Totsuka, K.; et al. Comparison of pathogenic factors expressed by group A Streptococci isolated from patients with streptococcal toxic shock syndrome and scarlet fever. Microb. Pathog. 1999, 27, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Kanno, T.; Sakaguchi, K.; Fukuyama, M.; Suzuki, J. Properties of metabolic substances produced by group A streptococcus from a food-borne epidemic. J. Infect. Chemother. 2011, 17, 462–467. [Google Scholar] [CrossRef] [PubMed]
- Kanno, T.; Sakaguchi, K.; Suzuki, J. Time course of virulence factors produced by group A streptococcus during a food-borne epidemic. J. Infect. Chemother. 2012, 18, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Murphy, P.V.; André, S.; Gabius, H.J. The third dimension of reading the sugar code by lectins: Design of glycoclusters with cyclic scaffolds as tools with the aim to define correlations between spatial presentation and activity. Molecules 2013, 18, 4026–4053. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, T.; Watanabe, M.; Naganuma, T.; Muramoto, K. Diversified carbohydrate-binding lectins from marine resources. J. Amino Acids 2011. [Google Scholar] [CrossRef]
- Kawsar, S.M.A.; Matsumoto, R.; Fujii, Y.; Yasumitsu, H.; Dogasaki, C.; Hosono, M.; Nitta, K.; Hamako, J.; Matsui, T.; Kojima, N.; et al. Purification and biochemical characterization of a D-galactose binding lectin from Japanese sea hare (Aplysia kurodai) eggs. Biochemistry (Mosc.) 2009, 74, 709–716. [Google Scholar] [CrossRef]
- Kawsar, S.M.; Matsumoto, R.; Fujii, Y.; Matsuoka, H.; Masuda, N.; Chihiro, I.; Yasumitsu, H.; Kanaly, R.A.; Sugawara, S.; Hosono, M.; et al. Cytotoxicity and glycan-binding profile of a d-galactose-binding lectin from the eggs of a Japanese sea hare (Aplysia kurodai). Protein J. 2011, 30, 509–519. [Google Scholar]
- Kuo, C.C.; Takahashi, N.; Swanson, A.F.; Ozeki, Y.; Hakomori, S. An N-linked high-mannose type oligosaccharide, expressed at the major outer membrane protein of Chlamydia trachomatis, mediates attachment and infectivity of the microorganism to HeLa cells. J. Clin. Investig. 1996, 98, 2813–2818. [Google Scholar] [CrossRef] [PubMed]
- Glover, K.J.; Weerapana, E.; Imperiali, B. In vitro assembly of the undecaprenylpyrophosphate-linked heptasaccharide for prokaryotic N-linked glycosylation. Proc. Natl. Acad. Sci. USA 2006, 102, 14255–142559. [Google Scholar] [CrossRef]
- Hartley, M.D.; Morrison, M.J.; Aas, F.E.; Børud, B.; Koomey, M.; Imperiali, B. Biochemical characterization of the O-linked glycosylation pathway in Neisseria gonorrhoeae responsible for biosynthesis of protein glycans containing N,N'-diacetylbacillosamine. Biochemistry 2011, 50, 4936–4948. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.M.; Wu, J.H.; Tsai, M.-S.; Yang, Z.; Sharon, N.; Herp, A. Differential affinities of Erythrina cristagalli lectin (ECL) toward monosaccharides and polyvalent mammalian structural units. Glycoconj. J. 2007, 24, 591–604. [Google Scholar] [CrossRef] [PubMed]
- Arasu, A.; Kumaresan, V.; Sathyamoorthi, A.; Chaurasia, M.K.; Bhatt, P.; Gnanam, A.J.; Palanisamy, R.; Marimuthu, K.; Pasupuleti, M.; Arockiaraj, J. Molecular characterization of a novel proto-type antimicrobial protein galectin-1 from striped murrel. Microbiol. Res. 2014. [Google Scholar] [CrossRef]
- Mukherjee, S.; Zheng, H.; Derebe, M.G.; Callenberg, K.M.; Partch, C.L.; Rollins, D.; Propheter, D.C.; Rizo, J.; Grabe, M.; Jiang, Q.X.; et al. Antibacterial membrane attack by a pore-forming intestinal C-type lectin. Nature 2014, 505, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Hasan, I.; Ozeki, Y.; Kabir, S.R. Purifiction of a novel chitin-binding lectin with antimicrobial and antibiofilm activities from a Bangladesh cultivar of potato (Solanum tuberosum). Ind. J. Biochem. Biophys. 2014, 51, 142–148. [Google Scholar]
- Stephenson, A.E.; Wu, H.; Novak, J.; Tomana, M.; Mintz, K.; Fives-Taylor, P. The Fap1 fimbrial adhesin is a glycoprotein: Antibodies specific for the glycan moiety block the adhesion of Streptococcus parasanguis in an in vitro tooth model. Mol. Microbiol. 2002, 43, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Chaze, T.; Guillot, A.; Valot, B.; Langella, O.; Chamot-Rooke, J.; di Guilmi, A.M.; Trieu-Cuot, P.; Dramsi, S.; Mistou, M.Y. O-glycosylation of the N-terminal region of the serine-rich adhesin Srr1 of Streptococcus agalactiae explored by mass spectrometry. Mol. Cell Proteomics 2014, in press. [Google Scholar]
- Chiarot, E.; Faralla, C.; Chiappini, N.; Tuscano, G.; Falugi, F.; Gambellini, G.; Taddei, A.; Capo, S.; Cartocci, E.; Veggi, D.; et al. Targeted amino acid substitutions impair streptolysin O toxicity and group A Streptococcus virulence. MBio 2013, 4. [Google Scholar] [CrossRef]
- Feil, S.C.; Ascher, D.B.; Kuiper, M.J.; Tweten, R.K.; Parker, M.W. Structural studies of Streptococcus pyogenes streptolysin O provide insights into the early steps of membrane penetration. J. Mol. Biol. 2014, 426, 785–792. [Google Scholar] [CrossRef] [PubMed]
- Ilangumaran, S.; Hoessli, D.C. Effects of cholesterol depletion by cyclodextrin on the sphingolipid microdomains of the plasma membrane. Biochem. J. 1998, 335, 433–440. [Google Scholar] [PubMed]
- Ogawa, Y.; Sugawara, S.; Tatsuta, T.; Hosono, M.; Nitta, K.; Fujii, Y.; Kobayashi, H.; Fujimura, T.; Taka, H.; Koide, Y.; et al. Sialyl-glycoconjugates in cholesterol-rich microdomains of P388 cells are the triggers for apoptosis induced by Rana catesbeiana oocyte ribonuclease. Glycoconj. J. 2014, 31, 171–184. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.Y.; Lee, S.H.; Park, J.H.; Han, H. Interaction of galectin-1 with caveolae induces mouse embryonic stem cell proliferation through the Src, ERas, Akt and mTOR signaling pathways. Cell. Mol. Life Sci. 2009, 66, 1467–1478. [Google Scholar] [CrossRef] [PubMed]
- Ikuta, A.; Mizuta, N.; Kitahara, S.; Murata, T.; Usui, T.; Koizumi, K.; Tanimoto, T. Preparation and characterization of novel branched β-cyclodextrins having β-d-galactose residues on the non-reducing terminal of the side chains and their specific interactions with peanut (Arachis hypogaea) agglutinin. Chem. Pham. Bull. 2004, 52, 51–56. [Google Scholar] [CrossRef]
- Stellner, K.; Saito, H.; Hakomori, S.-I. Determination of aminosugar linkages in glycolipids by methylation. Aminosugar linkages of ceramide pentasaccharides of rabbit erythrocytes and of Forssman antigen. Arch. Biochem. Biophys. 1973, 155, 464–472. [Google Scholar]
- Cisar, J.O.; Sandberg, A.L.; Abeygunawardana, C.; Reddy, G.P.; Bush, C.A. Lectin recognition of host-like saccharide motifs in streptococcal cell wall polysaccharides. Glycobiology 1995, 5, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, H.; Morizane, S.; Yamasaki, O.; Oono, T.; Iwatsuki, K. Assessment of Streptococcus pyogenes microcolony formation in infected skin by confocal laser scanning microscopy. J. Dermatol. Sci. 2003, 32, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Islam, B.; Khan, S.N.; Naeem, A.; Sharma, V.; Khan, A.U. Novel effect of plant lectins on the inhibition of Streptococcus mutans biofilm formation on saliva-coated surface. J. Appl. Microbiol. 2009, 106, 1682–1689. [Google Scholar] [CrossRef] [PubMed]
- Cavalcante, T.T.; Anderson Matias da Rocha, B.; Alves Carneiro, V.; Vassiliepe Sousa Arruda, F.; do Nascimento, A.S.F.; Cardoso Sá, N.; do Nascimento, K.S.; Cavada, B.S.; Teixeira, E.H. Effect of lectins from Diocleinae subtribe against oral Streptococci. Molecules 2011, 16, 3530–3543. [Google Scholar] [CrossRef] [PubMed]
- Grushoff, P.S.; Shany, S.; Bernheimer, W. Purification and properties of streptococcal nicotinamide adenine dinucleotide glycohydrolase. J. Bacteriol. 1975, 122, 599–605. [Google Scholar] [PubMed]
- Smith, P.K.; Krohn, R.I.; Hermanson, G.T.; Mallia, A.K.; Gartner, F.H.; Provenzano, M.D.; Fujimoto, E.K.; Goeke, N.M.; Olson, B.J.; Klenk, D.C. Measurement of protein using bicinchoninic acid. Anal. Biochem. 1985, 150, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Wiechelman, K.J.; Braun, R.D.; Fitzpatrick, J.D. Investigation of the bicinchoninic acid protein assay: Identification of the groups responsible for color formation. Anal. Biochem. 1988, 175, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Gourdine, J.P.; Cioci, G.; Miguet, L.; Unverzagt, C.; Silva, D.V.; Varrot, A.; Gautier, C.; Smith-Ravin, E.J.; Imberty, A. High affinity interaction between a bivalve C-type lectin and a biantennary complex-type N-glycan revealed by crystallography and microcalorimetry. J. Biol. Chem. 2008, 283, 30112–30120. [Google Scholar] [CrossRef] [PubMed]
- Kusama, H.; Ohashi, M.; Shimazaki, H.; Fukumi, H. Studies on streptolysin O and antistreptolysin O. III. Spectrophotometric determination of the hemolyic activity of streptolysin O by the fifty per cent end-point titration. Jpn. J. Med. Sci. Biol. 1958, 11, 347–357. [Google Scholar]
- Gniadecki, R.; Christoffersen, N.; Wulf, H.C. Cholesterol-rich plasma membrane domains (lipid rafts) in keratinocytes: Importance in the baseline and UVA-induced generation of reactive oxygen species. J. Investig. Dermatol. 2001, 118, 582–588. [Google Scholar] [CrossRef]
- Sample Availability: AKL sample is available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hasan, I.; Watanabe, M.; Ishizaki, N.; Sugita-Konishi, Y.; Kawakami, Y.; Suzuki, J.; Dogasaki, C.; Rajia, S.; Kawsar, S.M.A.; Koide, Y.; et al. A Galactose-Binding Lectin Isolated from Aplysia kurodai (Sea Hare) Eggs Inhibits Streptolysin-Induced Hemolysis. Molecules 2014, 19, 13990-14003. https://doi.org/10.3390/molecules190913990
Hasan I, Watanabe M, Ishizaki N, Sugita-Konishi Y, Kawakami Y, Suzuki J, Dogasaki C, Rajia S, Kawsar SMA, Koide Y, et al. A Galactose-Binding Lectin Isolated from Aplysia kurodai (Sea Hare) Eggs Inhibits Streptolysin-Induced Hemolysis. Molecules. 2014; 19(9):13990-14003. https://doi.org/10.3390/molecules190913990
Chicago/Turabian StyleHasan, Imtiaj, Miharu Watanabe, Naoto Ishizaki, Yoshiko Sugita-Konishi, Yasushi Kawakami, Jun Suzuki, Chikaku Dogasaki, Sultana Rajia, Sarkar M. A. Kawsar, Yasuhiro Koide, and et al. 2014. "A Galactose-Binding Lectin Isolated from Aplysia kurodai (Sea Hare) Eggs Inhibits Streptolysin-Induced Hemolysis" Molecules 19, no. 9: 13990-14003. https://doi.org/10.3390/molecules190913990
APA StyleHasan, I., Watanabe, M., Ishizaki, N., Sugita-Konishi, Y., Kawakami, Y., Suzuki, J., Dogasaki, C., Rajia, S., Kawsar, S. M. A., Koide, Y., Kanaly, R. A., Sugawara, S., Hosono, M., Ogawa, Y., Fujii, Y., Iriko, H., Hamako, J., Matsui, T., & Ozeki, Y. (2014). A Galactose-Binding Lectin Isolated from Aplysia kurodai (Sea Hare) Eggs Inhibits Streptolysin-Induced Hemolysis. Molecules, 19(9), 13990-14003. https://doi.org/10.3390/molecules190913990








