Rumphellaones B and C, New 4,5-Seco-Caryophyllane Sesquiterpenoids from Rumphella
Abstract
:1. Introduction
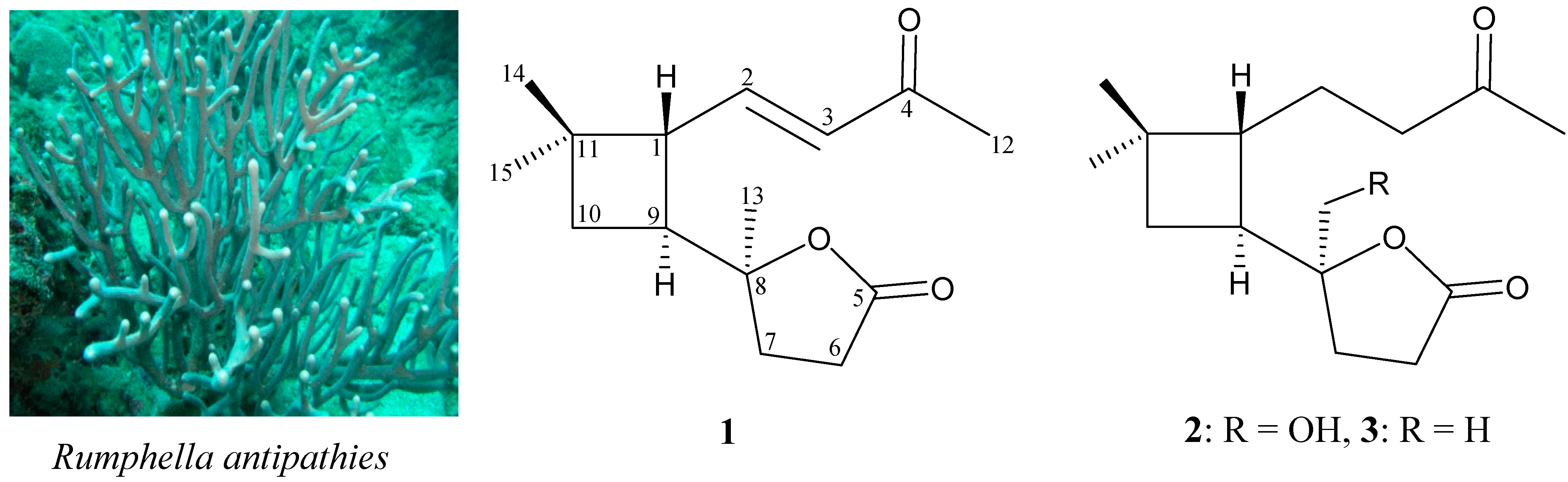
2. Results and Discussion
| Position | δH (J in Hz) | δC, Multiple | 1H–1H COSY | HMBC |
|---|---|---|---|---|
| 1 | 2.78 dd (9.2, 8.4) | 48.0, CH | H-2, H-9 | C-2, -3, -8, -9, -11, -14, -15 |
| 2 | 6.77 dd (16.0, 8.4) | 147.1, CH | H-1, H-3 | C-4, -9 |
| 3 | 6.09 d (16.0) | 131.4, CH | H-2 | C-1, -4 |
| 4 | 198.1, C | |||
| 5 | 176.7, C | |||
| 6 | 2.51–2.68 m | 29.1, CH2 | H2-7 | C-5 |
| 7 | 1.85–2.02 m | 30.9, CH2 | H2-6 | C-5, -6 |
| 8 | 86.6, C | |||
| 9 | 2.39 m | 43.0, CH | H-1, H2-10 | C-1, -2, -10 |
| 10 | 1.51–1.70 m | 33.1, CH2 | H-9 | C-1, -8, -9, -11, -14, -15 |
| 11 | 36.0, C | |||
| 12 | 2.25 s | 27.4, CH3 | C-3, -4 | |
| 13 | 1.25 s | 24.6, CH3 | C-7, -8, -9 | |
| 14 | 1.04 s | 23.8, CH3 | C-1, -10, -11, -15 | |
| 15 | 1.10 s | 29.6, CH3 | C-1, -10, -11, -14 |
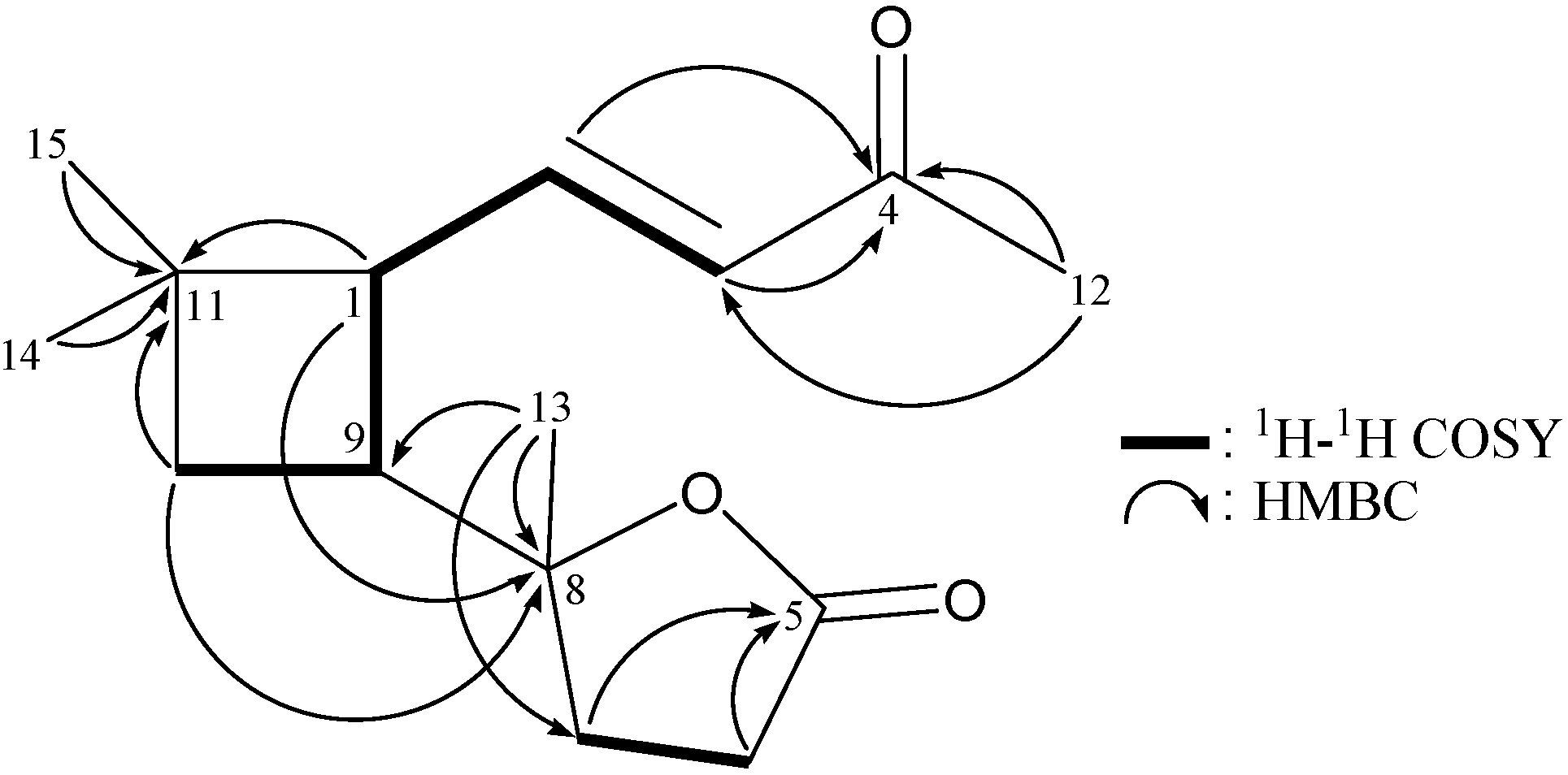
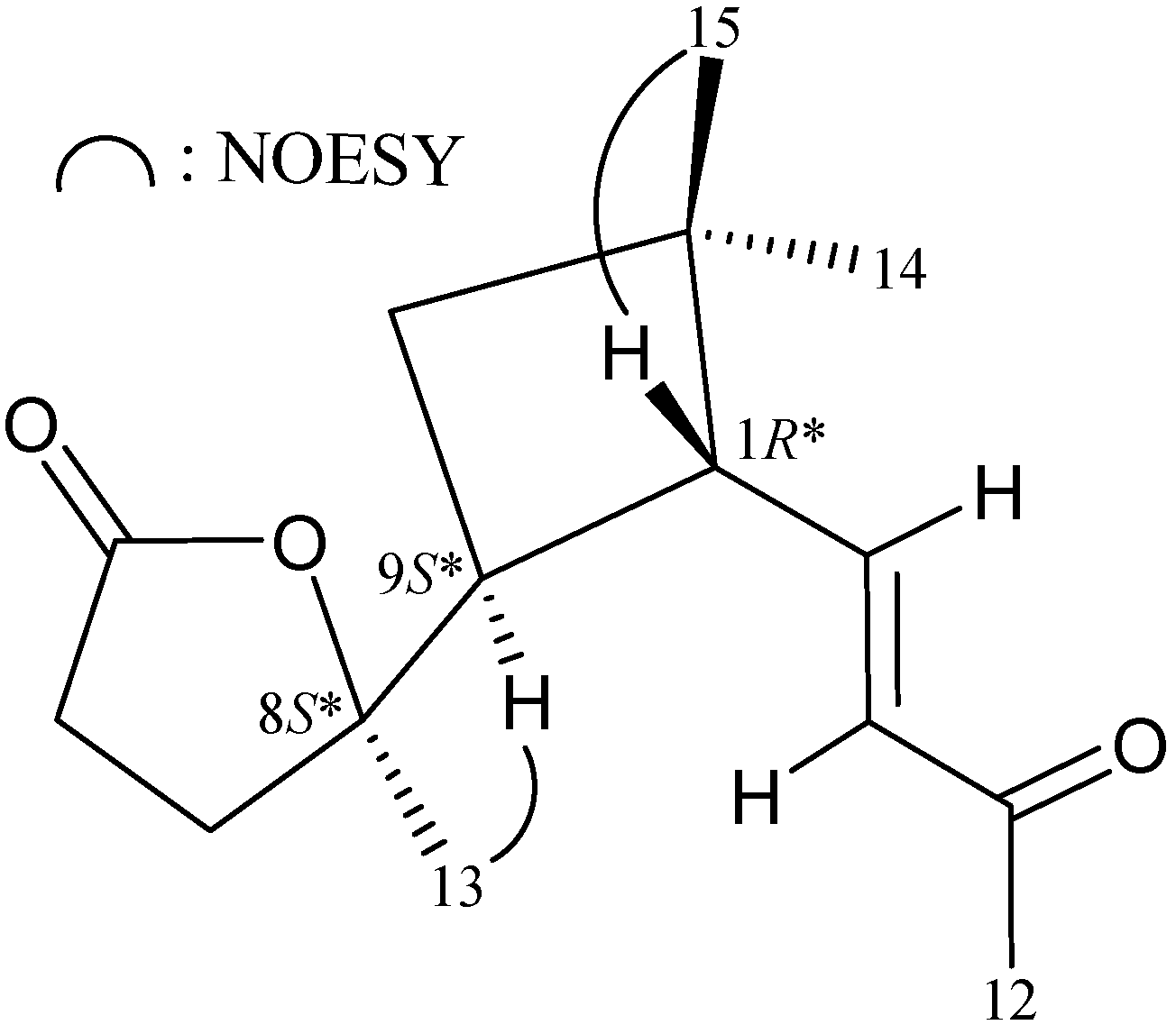
| Position | δH (J in Hz) | δC, Multiple | 1H–1H COSY | HMBC |
|---|---|---|---|---|
| 1 | 1.90 m | 44.0, CH | H2-2, H-9 | C-10, -11, -15 |
| 2 | 1.62 m | 24.9, CH2 | H-1, H2-3 | C-1, -3, -4, -9, -11 |
| 3 | 2.36 t (7.2) | 41.8, CH2 | H2-2 | C-1, -2, -4 |
| 4 | 208.6, C | |||
| 5 | 177.6, C | |||
| 6 | 2.54 ddd (18.0, 10.8, 5.2) | 29.7, CH2 | H2-7 | C-5, -7 |
| 2.71 ddd (18.0, 10.8, 7.6) | ||||
| 7 | 1.94 ddd (14.2, 10.8, 7.6) | 25.6, CH2 | H2-6 | C-5, -8, -9, -13 |
| 2.20 ddd (14.2, 10.8, 5.2) | ||||
| 8 | 89.5, C | |||
| 9 | 2.12 ddd (10.0, 10.0, 9.6) | 40.2, CH | H-1, H2-10 | n. o. a |
| 10 | 1.43 dd (10.4, 10.0) | 33.0, CH2 | H-9 | C-1, -8, -9, -11, -14, -15 |
| 1.57 dd (10.4, 9.6) | ||||
| 11 | 33.5, C | |||
| 12 | 2.12 s | 30.0, CH3 | C-3, -4 | |
| 13 | 3.43 d (11.6) | 66.6, CH2 | C-7, -8 | |
| 3.73 d (11.6) | ||||
| 14 | 1.03 s | 22.5, CH3 | C-1, -10, -11, -15 | |
| 15 | 1.07 s | 30.8, CH3 | C-1, -10, -11, -14 |
3. Experimental Section
3.1. General Experimental Procedures
3.2. Animal Material
3.3. Extraction and Isolation
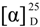 +18 (c 0.25, CHCl3); IR (neat) νmax 1,767, 1,712 cm−1; 1H-NMR (CDCl3, 400 MHz) and 13C-NMR (CDCl3, 100 MHz) data, see Table 1; ESIMS m/z 249 [M − H]+; HRESIMS m/z 249.1493 (calcd. for C15H22O3–H, 249.1485).
+18 (c 0.25, CHCl3); IR (neat) νmax 1,767, 1,712 cm−1; 1H-NMR (CDCl3, 400 MHz) and 13C-NMR (CDCl3, 100 MHz) data, see Table 1; ESIMS m/z 249 [M − H]+; HRESIMS m/z 249.1493 (calcd. for C15H22O3–H, 249.1485).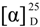 −8 (c 0.18, CHCl3); IR (neat) νmax 3,435, 1,763, 1,714 cm−1; 1H-NMR(CDCl3, 400 MHz) and 13C-NMR (CDCl3, 100 MHz) data: see Table 2; ESIMS m/z 291 [M + Na]+; HRESIMS m/z 291.1570 (calcd. for C15H24O4+Na, 291.1572).
−8 (c 0.18, CHCl3); IR (neat) νmax 3,435, 1,763, 1,714 cm−1; 1H-NMR(CDCl3, 400 MHz) and 13C-NMR (CDCl3, 100 MHz) data: see Table 2; ESIMS m/z 291 [M + Na]+; HRESIMS m/z 291.1570 (calcd. for C15H24O4+Na, 291.1572).3.4. Generation of Superoxide Anions and Release of Elastase by Human Neutrophils
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Anjaneyulu, V.; Rao, K.N.; Kobayashi, M. (24S)-24-Methylcholes-4-ene-3β,6β-diol from a gorgonian (Rumphella. aggregata) of the Andaman and the Nicobar Islands. Indian J. Chem 1995, 34B, 78–80. [Google Scholar]
- Puglisi, M.P.; Paul, V.J.; Biggs, J.; Slattery, M. Co-occurrence of chemical and structural defenses in the gorgonian corals of Guam. Mar. Ecol. Prog. Ser 2002, 239, 105–114. [Google Scholar]
- Nourry, M.; Urvois, P.-A.; Tomasoni, C.; Biard, J.F.; Verbist, J.F.; Roussakis, C. Antiproliferative effects of a product isolated from the gorgonian Rumphella. aggregata. Anticancer Res 1999, 19, 1881–1885. [Google Scholar]
- Chuang, L.-F.; Fan, T.-Y.; Li, J.-J.; Sung, P.-J. Kobusone: occurrence of a norsesquiterpenoid in the gorgonian coral Rumphella. antipathies (Gorgoniidae). Biochem. Syst. Ecol 2007, 35, 470–471. [Google Scholar] [CrossRef]
- Chuang, L.-F.; Fan, T.-Y.; Li, J.-J.; Kuo, J.; Fang, L.-S.; Wang, W.-H.; Sung, P.-J. Isokobusone, a caryophyllane-type norsesquiterpenoid from the gorgonian coral Rumphella antipathies (Gorgoniidae). Platax 2007, 2007, 61–67. [Google Scholar]
- Sung, P.-J.; Chuang, L.-F.; Kuo, J.; Chen, J.-J.; Fan, T.-Y.; Li, J.-J.; Fang, L.-S.; Wang, W.-H. Rumphellolides A–F, six new caryophyllane-related derivatives fromthe Formosan gorgonian coral Rumphella antipathies. Chem. Pharm. Bull 2007, 55, 1296–1301. [Google Scholar] [CrossRef]
- Sung, P.-J.; Chuang, L.-F.; Fan, T.-Y.; Chou, H.-N.; Kuo, J.; Fang, L.-S.; Wang, W.-H. Rumphellolide G, a new caryophyllane-type tetrahydropyran norsesquiterpenoid from the gorgonian coral Rumphella antipathies (Gorgoniidae). Chem. Lett. 2007, 36, 1322–1323. [Google Scholar] [CrossRef]
- Hwang, T.-L.; Su, Y.-D.; Hu, W.-P.; Chuang, L.-F.; Sung, P.-J. Rumphellolide H, anewnaturalcaryophyllane from the gorgonian Rumphella antipathies. Heterocycles 2009, 78, 1563–1567. [Google Scholar] [CrossRef]
- Sung, P.-J.; Su, Y.-D.; Hwang, T.-L.; Chuang, L.-F.; Chung, H.-M.; Chen, J.-J.; Li, J.-J.; Fang, L.-S.; Wang, W.-H. Rumphellolide I, a novel caryophyllane-related tetrahydropyran norsesquiterpenoid from gorgonian coral Rumphella antipathies. Chem. Lett 2009, 38, 282–283. [Google Scholar] [CrossRef]
- Sung, P.-J.; Chuang, L.-F.; Kuo, J.; Fan, T.-Y.; Hu, W.-P. Rumphellatin A, the first chloride-containing caryophyllane-typenorsesquiterpenoid from Rumphella antipathies. Tetrahedron Lett 2007, 48, 3987–3989. [Google Scholar] [CrossRef]
- Sung, P.-J.; Chuang, L.-F.; Hu, W.-P. Rumphellatins B and C, two new caryophyllane-type hemiketal norsesquiterpenoids from the Formosangorgonian coral Rumphella antipathies. Bull. Chem. Soc. Jpn. 2007, 80, 2395–2399. [Google Scholar] [CrossRef]
- Sung, P.-J.; Su, Y.-D.; Hwang, T.-L.; Chuang, L.-F.; Chen, J.-J.; Li, J.-J.; Fang, L.-S.; Wang, W.-H. Rumphellatin D, a novel chlorinated caryophyllane from gorgonian coral Rumphella antipathies. Chem. Lett 2008, 37, 1244–1245. [Google Scholar] [CrossRef]
- Chung, H.-M.; Chen, Y.-H.; Lin, M.-R.; Su, J.-H.; Wang, W.-H.; Sung, P.-J. Rumphellaone A, a novel caryophyllane-related derivative from the gorgoniancoral Rumphella antipathies. Tetrahedron Lett 2010, 51, 6025–6027. [Google Scholar]
- Yang, S.-C.; Chung, P.-J.; Ho, C.-M.; Kuo, C.-Y.; Hung, M.-F.; Huang, Y.-T.; Chang, W.-Y.; Chang, Y.-W.; Chan, K.-H.; Hwang, T.-L. Propofol inhibits superoxide production, elastase release, and chemotaxis in formyl peptide-activated human neutrophils by blocking formyl peptide receptor 1. J. Immunol. 2013, 190, 6511–6519. [Google Scholar]
- Yu, H.-P.; Hsieh, P.-W.; Chang, Y.-J.; Chung, P.-J.; Kuo, L.-M.; Hwang, T.-L. 2-(2-Fluorobenzamido)benzoate ethyl ester (EFB-1) inhibits superoxide production by human neutrophils and attenuates hemorrhagic shock-induced organ dysfunction in rats. Free Radic. Biol. Med. 2011, 50, 1737–1748. [Google Scholar]
- Samples Availability: Samples of the compounds are not available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chung, H.-M.; Wang, W.-H.; Hwang, T.-L.; Li, J.-J.; Fang, L.-S.; Wu, Y.-C.; Sung, P.-J. Rumphellaones B and C, New 4,5-Seco-Caryophyllane Sesquiterpenoids from Rumphella . Molecules 2014, 19, 12320-12327. https://doi.org/10.3390/molecules190812320
Chung H-M, Wang W-H, Hwang T-L, Li J-J, Fang L-S, Wu Y-C, Sung P-J. Rumphellaones B and C, New 4,5-Seco-Caryophyllane Sesquiterpenoids from Rumphella . Molecules. 2014; 19(8):12320-12327. https://doi.org/10.3390/molecules190812320
Chicago/Turabian StyleChung, Hsu-Ming, Wei-Hsien Wang, Tsong-Long Hwang, Jan-Jung Li, Lee-Shing Fang, Yang-Chang Wu, and Ping-Jyun Sung. 2014. "Rumphellaones B and C, New 4,5-Seco-Caryophyllane Sesquiterpenoids from Rumphella " Molecules 19, no. 8: 12320-12327. https://doi.org/10.3390/molecules190812320
APA StyleChung, H.-M., Wang, W.-H., Hwang, T.-L., Li, J.-J., Fang, L.-S., Wu, Y.-C., & Sung, P.-J. (2014). Rumphellaones B and C, New 4,5-Seco-Caryophyllane Sesquiterpenoids from Rumphella . Molecules, 19(8), 12320-12327. https://doi.org/10.3390/molecules190812320







