Roles of the Amino Group of Purine Bases in the Thermodynamic Stability of DNA Base Pairing
Abstract
:1. Introduction
2. Results and Discussion
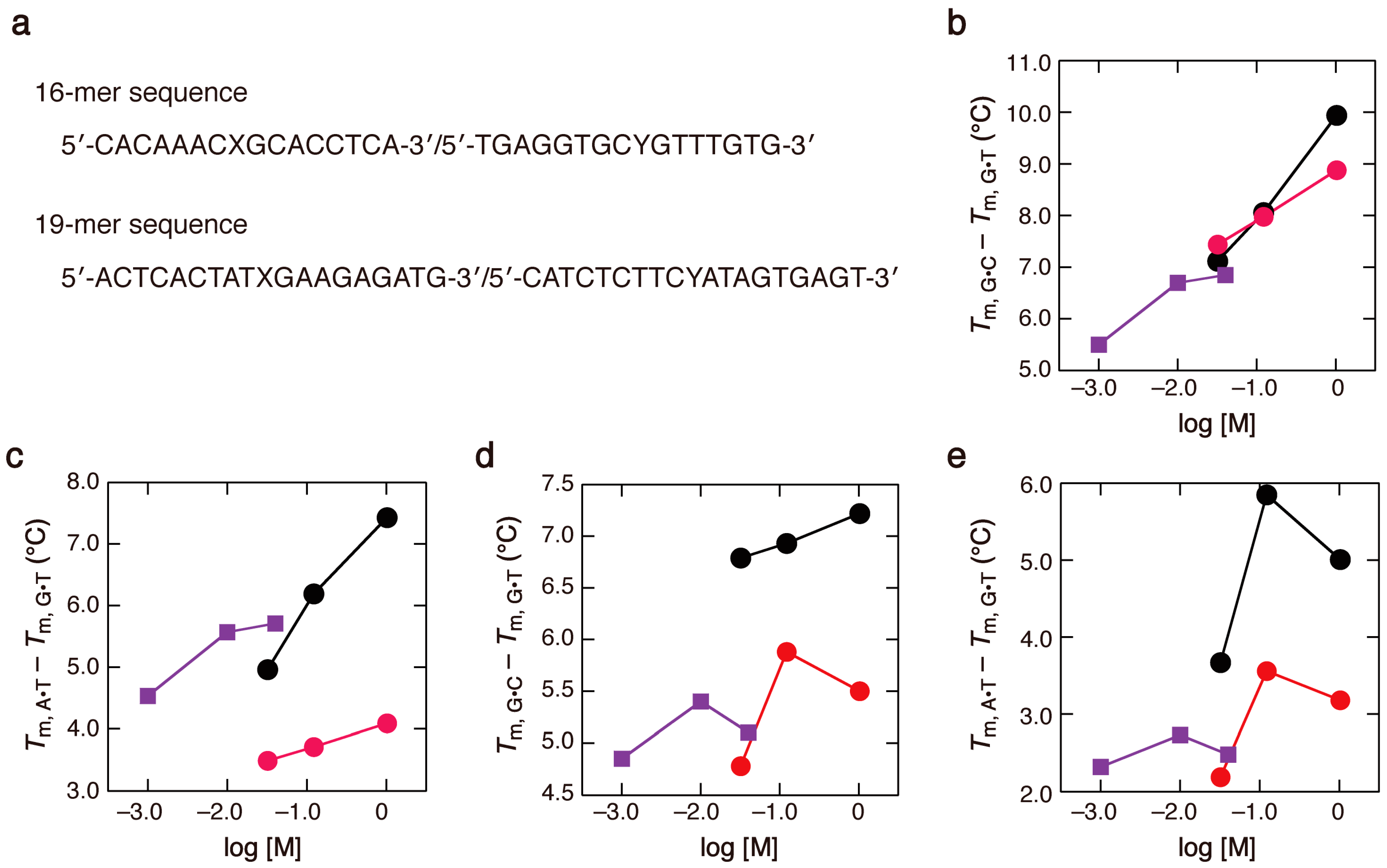
2.1. DNA Duplexes in Solutions Containing Na+ of Various Concentrations
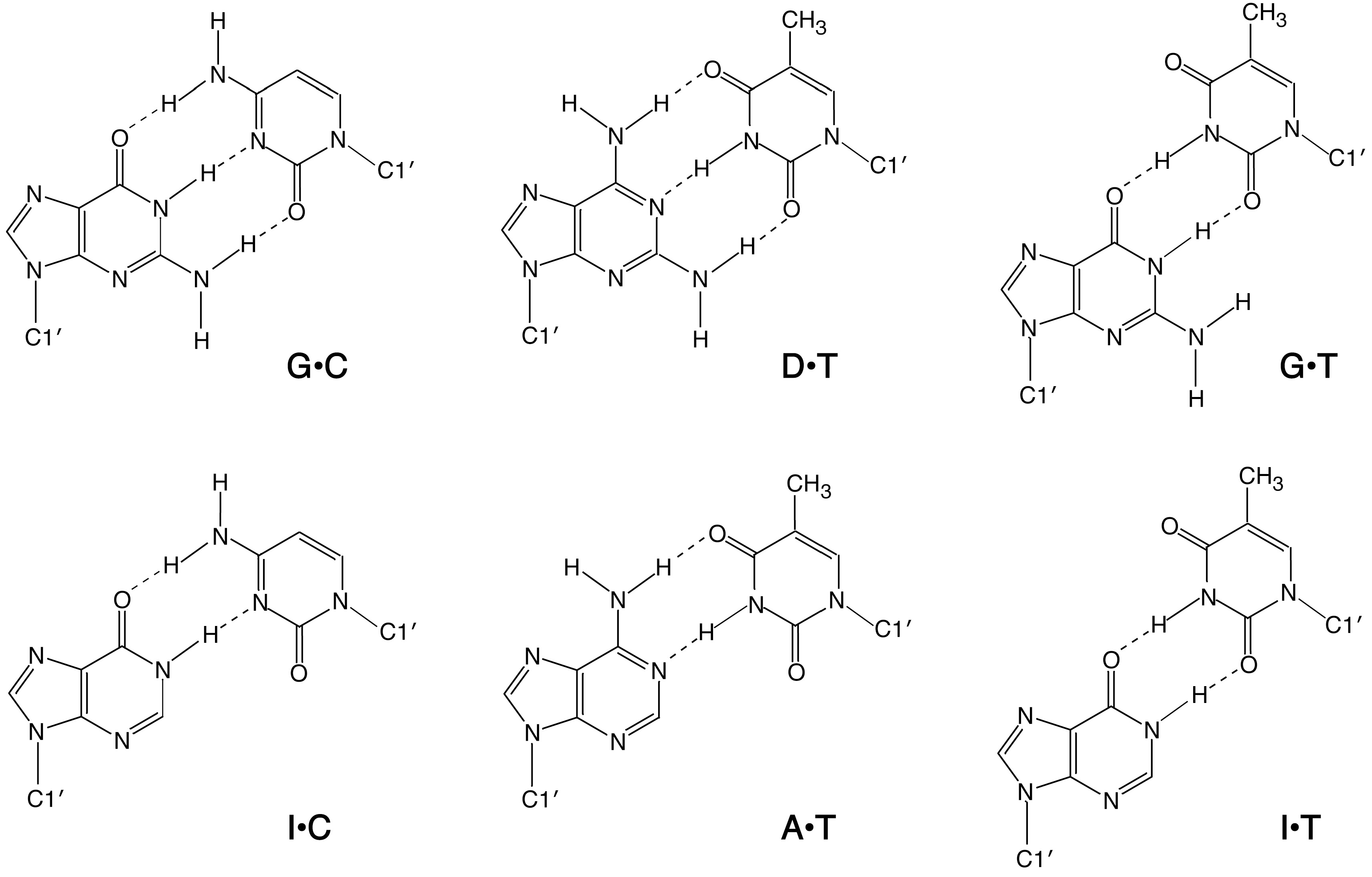
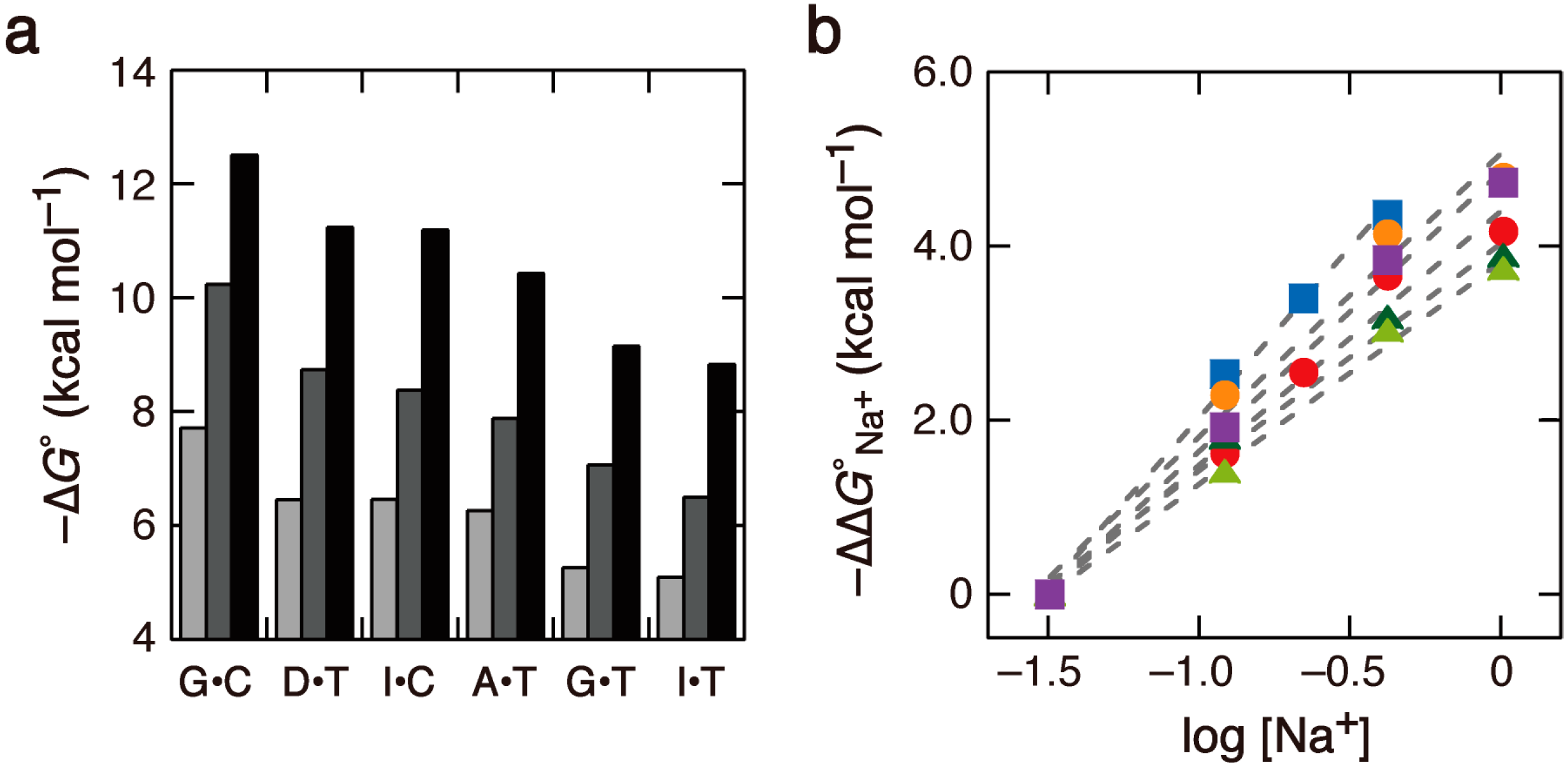
2.2. Duplex Stabilities in Water-Ethanol Mixed Solvents and Mg2+ Solutions
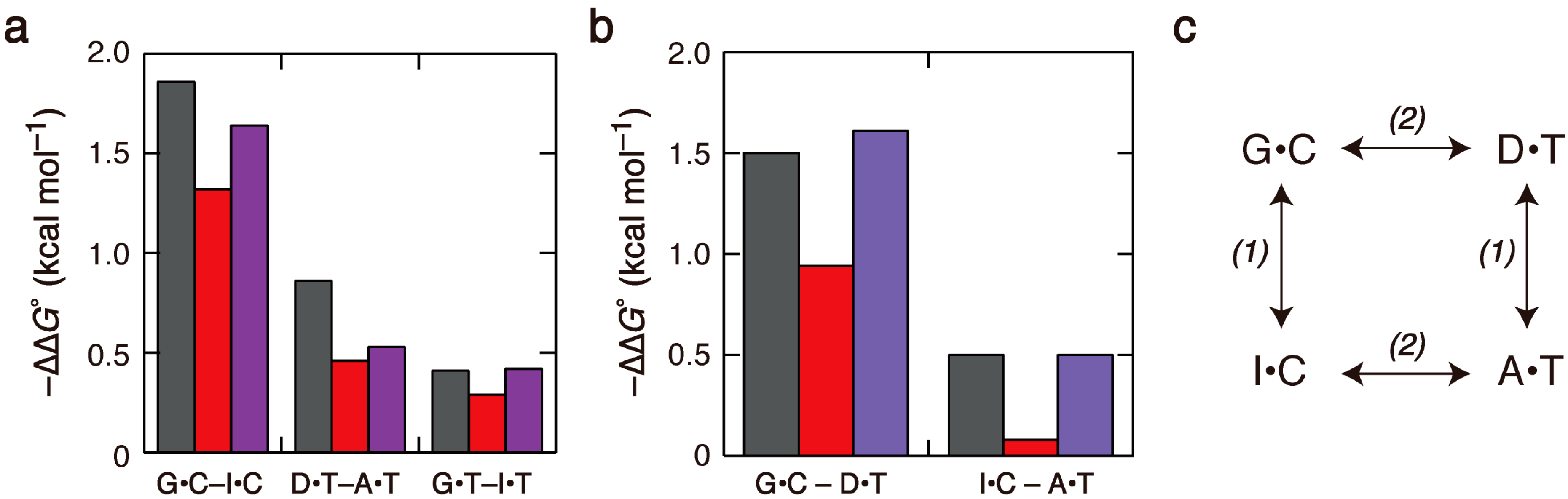
2.3. Comparison of the Salt Concentration Dependences
| X•Y pair | Na+ solution | Na+ with 3 M ethanol solution | Mg2+ solution |
|---|---|---|---|
| G•C | 3.91 ± 0.19 | 3.32 ± 0.09 | 2.63 ± 0.26 |
| D•T | 3.25 ± 0.32 | 3.17 ± 0.21 | 2.00 ± 0.22 |
| I•C | 3.21 ± 0.16 | 3.19 ± 0.11 | 2.15 ± 0.16 |
| A•T | 2.91 ± 0.23 | 2.74 ± 0.09 | 1.92 ± 0.19 |
| G•T | 2.61 ± 0.18 | 2.43 ± 0.17 | 1.59 ± 0.26 |
| I•T | 2.55 ± 0.13 | 2.41 ± 0.13 | 1.50 ± 0.26 |
2.4. Significance of Sequence-Dependent Salt Effects on Oligonucleotide Hybridization
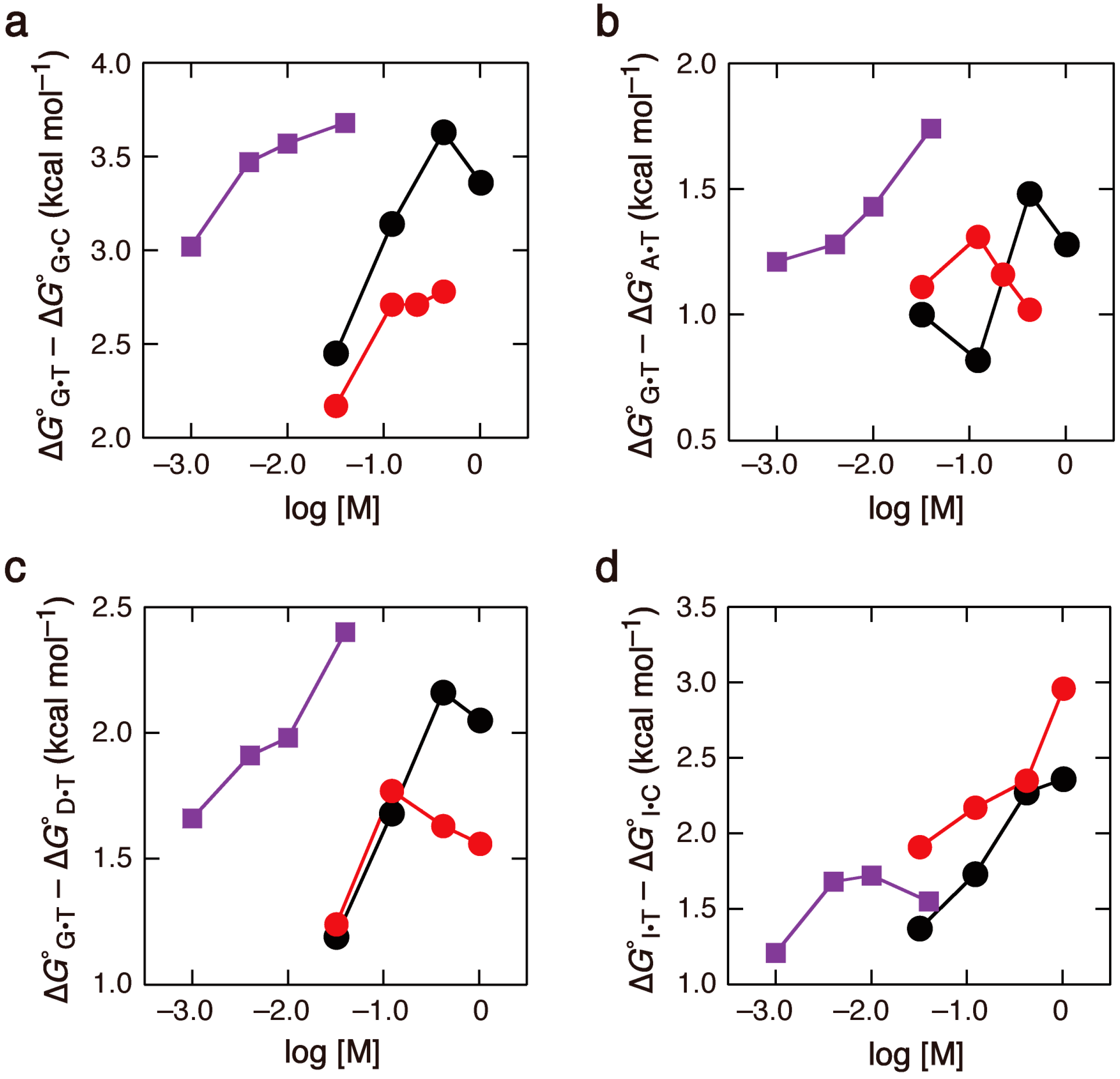
3. Experimental Section
3.1. Materials and Buffers
3.2. CD Spectra and UV Melting Curves
3.3. Analysis of the Number of Ions Bound during Duplex Formation
4. Conclusions
Supplementary Materials
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Turner, D.H. Conformational changes. In Nucleic Acids: Structures, Properties and Functions, 1st ed.; Bloomfield, V.A., Crothers, D.M., Tinoco, I., Jr., Eds.; University Science Books Press: California, CA, USA, 2000; pp. 259–334. [Google Scholar]
- Allawi, H.T.; SantaLucia, J. Thermodynamics and NMR of internal G•T mismatches in DNA. Biochemistry 1997, 36, 10581–10594. [Google Scholar] [CrossRef]
- Turner, D.H.; Sugimoto, N.; Kierzek, R.; Dreiker, S.D. Free energy increments for hydrogen bonds in nucleic acid base pairs. J. Am. Chem. Soc. 1987, 109, 3783–3785. [Google Scholar] [CrossRef]
- Watkins, N.E.; SantaLucia, J. Nearest-neighbor thermodynamics of deoxyinosine pairs in DNA duplexes. Nucl. Acids Res. 2005, 33, 6258–6267. [Google Scholar] [CrossRef]
- Siegfried, N.A.; Metzger, S.L.; Bevilacqua, P.C. Folding cooperativity in RNA and DNA is dependent on position in the helix. Biochemistry 2007, 46, 172–181. [Google Scholar] [CrossRef]
- Chollet, A.; Kawashima, E. DNA containing the base analogue 2-aminoadenine: Preparation, use as hybridization probes and cleavage by restriction endonucleases. Nucleic Acids Res. 1988, 16, 305–317. [Google Scholar] [CrossRef]
- Cheong, C.; Tinoco, I.; Chollet, A. Thermodynamic studies of base pairing involving 2,6-diaminopurine. Nucl. Acids Res. 1988, 16, 5115–5122. [Google Scholar] [CrossRef]
- Jorgensen, W.L.; Pranata, J. Importance of secondary interactions in triply hydrogen bonded complexes: guanine-cytosine vs. uracil-2,6-diaminopyridine. J. Am. Chem. Soc. 1990, 112, 2008–2010. [Google Scholar] [CrossRef]
- Leonard, G.A.; McAuley-Hecht, K.; Brown, T.; Hunter, W.N. Do C-H-O hydrogen bonds contribute to the stability of nucleic acid base pairs? Acta Cryst. 1995, D51, 136–139. [Google Scholar]
- Šponer, J.; Leszczynski, J.; Hobza, P. Electronic properties, hydrogen bonding, stacking, and cation binding of DNA and RNA bases. Biopolymers 2001, 61, 3–31. [Google Scholar] [CrossRef]
- Popelier, P.A.; Joubert, L. The elusive atomic rationale for DNA base pair stability. J. Am. Chem. Soc. 2002, 124, 8725–8729. [Google Scholar] [CrossRef]
- Parthasarathi, R.; Amutha, R.; Subramanian, V.; Nair, B.U.; Ramasami, T. Bader’s and reactivity descriptors’ analysis of DNA base pairs. J. Phys. Chem. A 2004, 108, 3817–3828. [Google Scholar]
- Sugimoto, N.; Nakano, M.; Nakano, S. Thermodynamics-structure relationship of single mismatches in RNA/DNA duplexes. Biochemistry 2000, 39, 11270–11281. [Google Scholar] [CrossRef]
- Nakano, S.; Fujimoto, M.; Hara, H.; Sugimoto, N. Nucleic acid duplex stability: Influence of base composition on cation effects. Nucleic Acids Res. 1999, 27, 2957–2965. [Google Scholar] [CrossRef]
- Albergo, D.D.; Turner, D.H. Solvent effects on thermodynamics of double-helix formation in (dG-dC)3. Biochemistry 1981, 20, 1413–1418. [Google Scholar] [CrossRef]
- Hickey, D.R.; Turner, D.H. Solvent effects on the stability of A7U7p. Biochemistry 1985, 24, 2086–2094. [Google Scholar] [CrossRef]
- Nakano, S.; Miyoshi, D.; Sugimoto, N. Effects of molecular crowding on the structures, interactions, and functions of nucleic acids. Chem. Rev. 2014, 114, 2733–2758. [Google Scholar] [CrossRef]
- Moody, E.M.; Bevilacqua, P.C. Folding of a stable DNA motif involves a highly cooperative network of interactions. J. Am. Chem. Soc. 2003, 125, 16285–16293. [Google Scholar] [CrossRef]
- Krepl, M.; Otyepka, M.; Banas, P.; Šponer, J. Effect of guanine to inosine substitution on stability of canonical DNA and RNA duplexes: Molecular dynamics thermodynamics integration study. J. Phys. Chem. B 2013, 117, 1872–1879. [Google Scholar]
- Murray, T.J.; Zimmerman, S.C. New triply hydrogen bonded complexes with highly variable stabilities. J. Am. Chem. Soc. 1992, 114, 4010–4011. [Google Scholar] [CrossRef]
- Chazin, W.J.; Rance, M.; Chollet, A.; Leupin, W. Comparative NMR analysis of the decadeoxynucleotide d-(GCATTAATGC)2 and an analogue containing 2-aminoadenine. Nucleic Acids Res. 1991, 19, 5507–5513. [Google Scholar] [CrossRef]
- Bailly, C.; Mollegaard, N.E.; Nielsen, P.E.; Waring, M.J. The influence of the 2-amino group of guanine on DNA conformation. Uranyl and DNase I probing of inosine/diaminopurine substituted DNA. EMBO J. 1995, 14, 2121–2131. [Google Scholar]
- Lankas, F.; Cheatham, T.E.; Špackova, N.; Hobza, P.; Langowski, J.; Šponer, J. Critical effect of the N2 amino group on structure, dynamics, and elasticity of DNA polypurine tracts. Biophys. J. 2002, 82, 2592–2609. [Google Scholar] [CrossRef]
- Anderson, C.F.; Record, M.T. Salt-nucleic acid interactions. Annu. Rev. Phys. Chem. 1995, 46, 657–700. [Google Scholar]
- Record, M.T.; Zhang, W.; Anderson, C.F. Analysis of effects of salts and uncharged solutes on protein and nucleic acid equilibria and processes: A practical guide to recognizing and interpreting polyelectrolyte effects, Hofmeister effects, and osmotic effects of salts. Adv. Protein Chem. 1998, 51, 281–353. [Google Scholar] [CrossRef]
- Leipply, D.; Lambert, D.; Draper, D.E. Ion-RNA interactions: Thermodynamic analysis of the effects of mono- and divalent ions on RNA conformational equilibria. Method. Enzymol. 2009, 469, 433–463. [Google Scholar] [CrossRef]
- Shkel, I.A.; Record, M.T. Effect of the number of nucleic acid oligomer charges on the salt dependence of stability (∆G°37) and melting temperature (Tm): NLPB analysis of experimental data. Biochemistry 2004, 43, 7090–7101. [Google Scholar] [CrossRef]
- Rozners, E. Determination of nucleic acid hydration using osmotic stress. Curr. Protoc. Nucleic Acid Chem. 2010, 43. [Google Scholar] [CrossRef]
- Williams, A.P.; Longfellow, C.E.; Freier, S.M.; Kierzek, R.; Turner, D.H. Laser temperature-jump, spectroscopic, and thermodynamic study of salt effects on duplex formation by dGCATGC. Biochemistry 1989, 28, 4283–4291. [Google Scholar] [CrossRef]
- Erie, D.; Sinha, N.; Olson, W.; Jones, R.; Breslauer, K. A dumbbell-shaped, double-hairpin structure of DNA: A thermodynamic investigation. Biochemistry 1987, 26, 7150–7159. [Google Scholar] [CrossRef]
- Braunlin, W.H.; Bloomfield, V.A. 1H-NMR study of the base-pairing reactions of d(GGAATTCC): Salt effects on the equilibria and kinetics of strand association. Biochemistry 1991, 30, 754–758. [Google Scholar] [CrossRef]
- Nakano, S.; Wu, L.; Oka, H.; Karimata, H.T.; Kirihata, T.; Sato, Y.; Fujii, S.; Sakai, H.; Kuwahara, M.; Sawai, H.; et al. Conformation and the sodium ion condensation on DNA and RNA structures in the presence of a neutral cosolute as a mimic of the intracellular media. Mol. BioSyst. 2008, 4, 579–588. [Google Scholar] [CrossRef]
- Bond, J.P.; Anderson, C.F.; Record, M.T. Conformational transitions of duplex and triplex nucleic acid helices: Thermodynamic analysis of effects of salt concentration on stability using preferential interaction coefficients. Biophys. J. 1994, 67, 825–836. [Google Scholar] [CrossRef]
- Tan, Z.J.; Chen, S.J. Ion-mediated nucleic acid helix-helix interactions. Biophys. J. 2006, 91, 518–536. [Google Scholar] [CrossRef]
- Nakano, S.; Kitagawa, Y.; Miyoshi, D.; Sugimoto, N. Hammerhead ribozyme activity and oligonucleotide duplex stability in mixed solutions of water and organic compounds. FEBS Open Bio 2014, 4, 643–650. [Google Scholar] [CrossRef]
- Chiu, T.K.; Kaczor-Grzeskowiak, M.; Dickerson, R.E. Absence of minor groove monovalent cations in the crosslinked dodecamer C-G-C-G-A-A-T-T-C-G-C-G. J. Mol. Biol. 1999, 292, 589–608. [Google Scholar] [CrossRef]
- Chiu, T.K.; Dickerson, R.E. 1 Å crystal structures of B-DNA reveal sequence-specific binding and groove-specific bending of DNA by magnesium and calcium. J. Mol. Biol. 2000, 301, 915–945. [Google Scholar] [CrossRef]
- Howerton, S.B.; Sines, C.C.; VanDerveer, D.; Williams, L.D. Locating monovalent cations in the grooves of B-DNA. Biochemistry 2001, 40, 10023–10031. [Google Scholar] [CrossRef]
- Hud, N.V.; Sklenar, V.; Feigon, J. Localization of ammonium ions in the minor groove of DNA duplexes in solution and the origin of DNA A-tract bending. J. Mol. Biol. 1999, 286, 651–660. [Google Scholar] [CrossRef]
- Cheatham, T.E.; Kollman, P.A. Molecular dynamics simulations highlight the structural differences among DNA:DNA, RNA:RNA, and DNA:RNA hybrid duplexes. J. Am. Chem. Soc. 1997, 119, 4805–4825. [Google Scholar] [CrossRef]
- Young, M.A.; Ravishanker, G.; Beveridge, D.L. A 5-nanosecond molecular dynamics trajectory for B-DNA: Analysis of structure, motions, and solvation. Biophys. J. 1997, 73, 2313–2336. [Google Scholar] [CrossRef]
- Barnett, R.N.; Cleveland, C.L.; Joy, A.; Landman, U.; Schuster, G.B. Charge migration in DNA: Ion-gated transport. Science 2001, 294, 567–571. [Google Scholar] [CrossRef]
- Hamelberg, D.; Williams, L.D.; Wilson, W.D. Influence of the dynamic positions of cations on the structure of the DNA minor groove: Sequence-dependent effects. J. Am. Chem. Soc. 2001, 123, 7745–7755. [Google Scholar] [CrossRef]
- Subitana, J.A.; Soler-Lopez, M. Cations as hydrogen bond donors: A view of electrostatic interactions in DNA. Annu. Rev. Biophys. Biomol. Struct. 2003, 32, 27–45. [Google Scholar] [CrossRef]
- Li, W.; Nordenskiold, L.; Mu, Y. Sequence-specific Mg2+-DNA interactions: A molecular dynamics simulation study. J. Phys. Chem. B 2011, 115, 14713–14720. [Google Scholar]
- Šponer, J.; Burda, J.V.; Sabat, M.; Leszczynski, J.; Hobza, P. Interaction between the guanine-cytosine Watson-Crick DNA base pair and hydrated group IIa (Mg2+, Ca2+, Sr2+, Ba2+) and group IIb (Zn2+, Cd2+, Hg2+) metal cations. J. Phys. Chem. A 1998, 102, 5951–5957. [Google Scholar]
- Shields, R.; Heinrichs, A. A Trends Guide to Genetic Variation and Genomic Medicine; Elsevier Science: Madison, WI, USA, 2002; pp. S25–S29. [Google Scholar]
- Yaku, H.; Yukimasa, T.; Nakano, S.; Sugimoto, N.; Oka, H. Design of allele-specific primers and detection of the human ABO genotyping to avoid the pseudopositive problem. Electrophoresis 2008, 29, 4130–4140. [Google Scholar] [CrossRef]
- Martin, F.H.; Castro, M.M.; Aboul-ela, F.; Tinoco, I. Base pairing involving deoxyinosine: Implications for probe design. Nucleic Acids Res. 1985, 13, 8927–8938. [Google Scholar] [CrossRef]
- Nakano, S.; Oka, H.; Yamaguchi, D.; Fujii, M.; Sugimoto, N. Base-pairing selectivity of a ureido-linked phenyl-2'-deoxycytidine derivative. Org. Biomol. Chem. 2012, 10, 9664–9670. [Google Scholar] [CrossRef]
- Richard, R.G. Handbook of Biochemistry and Molecular Biology: Nucleic Acids, 3rd ed.; CRC Press: Cleveland, OH, USA, 1975; p. 597. [Google Scholar]
- Mergny, J.L.; Lacroix, L. Analysis of thermal melting curves. Oligonucleotides 2003, 13, 515–537. [Google Scholar] [CrossRef]
- Puglisi, J.D.; Tinoco, I. Absorbance melting curves of RNA. Method. Enzymol. 1989, 180, 304–325. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nakano, S.-i.; Sugimoto, N. Roles of the Amino Group of Purine Bases in the Thermodynamic Stability of DNA Base Pairing. Molecules 2014, 19, 11613-11627. https://doi.org/10.3390/molecules190811613
Nakano S-i, Sugimoto N. Roles of the Amino Group of Purine Bases in the Thermodynamic Stability of DNA Base Pairing. Molecules. 2014; 19(8):11613-11627. https://doi.org/10.3390/molecules190811613
Chicago/Turabian StyleNakano, Shu-ichi, and Naoki Sugimoto. 2014. "Roles of the Amino Group of Purine Bases in the Thermodynamic Stability of DNA Base Pairing" Molecules 19, no. 8: 11613-11627. https://doi.org/10.3390/molecules190811613
APA StyleNakano, S.-i., & Sugimoto, N. (2014). Roles of the Amino Group of Purine Bases in the Thermodynamic Stability of DNA Base Pairing. Molecules, 19(8), 11613-11627. https://doi.org/10.3390/molecules190811613






