Leishmanicidal Evaluation of Tetrahydroprotoberberine and Spirocyclic Erythrina-Alkaloids
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation and Structural Determination of Alkaloids
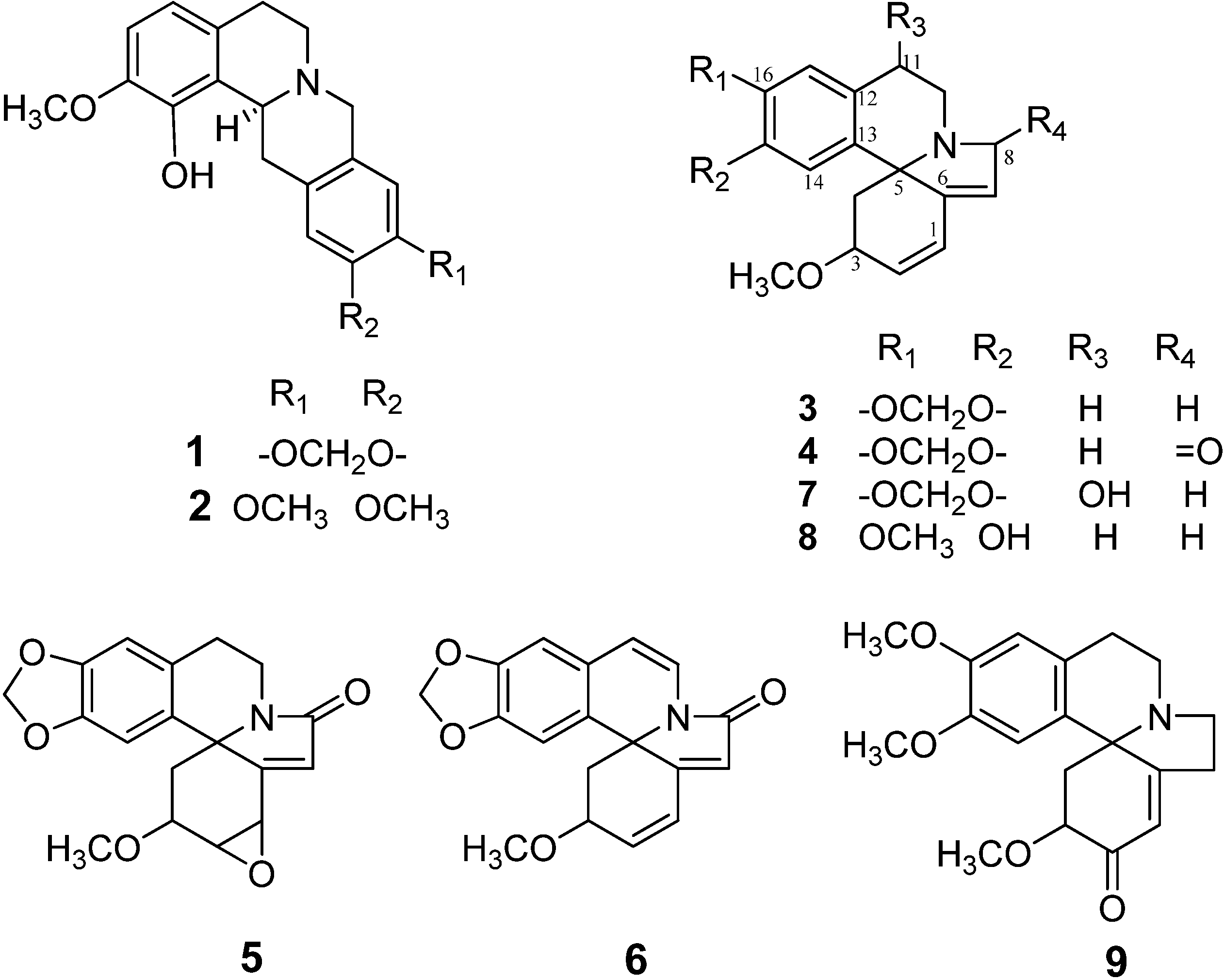
2.2. Evaluation of Leishmanicidal Activity
| Compound | L. amazonensis | Macrophage cells (J774) | SI |
|---|---|---|---|
| IC50 (µg/mL) | CC50 (µg/mL) | ||
| 1 | 0.18 | 51.22 | 284.55 |
| 2 | >1000 | 96.56 | ND |
| 3 | 65.27 | 69.33 | 1.06 |
| 4 | >1000 | >1000 | ND |
| 5 | 48.17 | 47.66 | 0.98 |
| 6 | 71.53 | 58.97 | 0.82 |
| 7 | 39.53 | 59.97 | 1.52 |
| 8 | >1000 | >1000 | ND |
| 9 | >1000 | >1000 | ND |
| Amphotericin B | 0.20 | NT | NT |
3. Experimental Section
3.1. General
3.2. Isolation of Alkaloids
3.2.1. Plant Materials
3.2.2. Extraction and Isolation
3.2.3. Spectral Data of Alkaloids
3.3. Leishmanicidal Assay
3.4. Macrophages in Vitro Cytotoxic Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wouters, O.J.; Downs, P.W.; Zoerhoff, K.L.; Crowley, K.R.; Frawley, H.; Einberg, J.; Chu, B.K.; Brady, M.A.; Oscar, R.; Jeudi, M.; et al. Resource planning for Neglected Tropical Disease (NTD) control nrograms: Feasibility study of the tool for integrated planning and costing (TIPAC). PLoS Negl. Trop. Dis. 2014, 8, 1–8. [Google Scholar]
- Sachs-Barrable, K.; Conway, J.; Gershkovich, P.; Ibrahim, F.; Wasan, K.M. The use of the United States FDA programs as a strategy to advance the development of drug products for neglected tropical diseases. Drug Dev. Ind. Pharm. 2014, 1–6. [Google Scholar]
- Johnston, K.L.; Ford, L.; Taylor, M.J. Overcoming the challenges of drug discovery for neglected tropical diseases: The A·WOL Experience. J. Biomol. Screen. 2014, 19, 335–343. [Google Scholar] [CrossRef]
- David, C.V.; Craft, N. Cutaneous and mucocutaneous leishmaniasis. Dermatol. Ther. 2009, 22, 491–502. [Google Scholar] [CrossRef]
- Control of the Leishmaniasis; NO. 949; World Health Organization: Geneva, Switzerland, 2010; pp. 1–202.
- Bhandari, V.; Sundar, S.; Dujardin, J.C.; Salotra, P. Elucidation of cellular mechanisms involved in experimental paromomycin resistance in Leishmania donovani. Antimicrob. Agents Chemother. 2014, 58, 2580–2585. [Google Scholar] [CrossRef]
- Stockdale, L.; Newton, R. A review of preventative methods against human leishmaniasis infection. PLoS Negl. Trop. Dis. 2013, 20, 1–15. [Google Scholar]
- Rath, S.; Trivelin, L.A.; Imbrunito, T.R.; Tomazela, D.M.; Jesús, M.N.; Marzal, P.C.; Andrade Junior, H.F.; Tempone, A.G. Antimoniais empregados no tratamento da leishmaniose: Estado da arte. Quím. Nova 2003, 26, 550–550. [Google Scholar]
- Schmidt, T.J.; Khalid, S.A.; Romanha, A.J.; Alves, T.M.; Biavatti, M.W.; Brun, R.; da costa, f.b.; de Castro, S.L.; Ferreira, V.F.; de Lacerda, M.V.; et al. The potential of secondary metabolites from plants as drugs or leads against protozoan neglected diseases—Part I. Curr. Med. Chem. 2012, 19, 2128–2175. [Google Scholar]
- Schmidt, T.J.; Khalid, S.A.; Romanha, A.J.; Alves, T.M.; Biavatti, M.W.; Brun, R.; da Costa, F.B.; de Castro, S.L.; Ferreira, V.F.; de Lacerda, M.V.; et al. The potential of secondary metabolites from plants as drugs or leads against protozoan neglected diseases—Part II. Curr. Med. Chem. 2012, 19, 2176–2228. [Google Scholar]
- Singh, N.; Mishra, B.B.; Bajpai, S.; Singh, R.K.; Tiwari, V.K. Natural product based leads to fight against leishmaniasis. Bioorg. Med. Chem. 2014, 22, 18–45. [Google Scholar] [CrossRef]
- Luize, P.S.; Tiuman, T.S.; Morello, L.G.; Maza, P.K.; Ueda-Nakamura, T.; Filho, B.P.D.; Cortez, D.A.G.; Mello, J.C.P.; Nakamura, C.V. Effects of medicinal plant extracts on growth of Leishmania (L.) amazonensis and Trypanosoma cruzi. Braz. J. Pharm. Sci. 2005, 41, 85–94. [Google Scholar]
- Miguel-Chavez, R.S.; Soto-Hernandez, M.; Ramos-Valdivia, A.C.; Kite, G. Alkaloid production in elicited cell suspension cultures of Erythrina americana Miller. Phytochem. Rev. 2007, 6, 167–173. [Google Scholar] [CrossRef]
- Tanaka, H.; Etoh, H.; Shimizu, H.; Oh-Uchi, T.; Terada, Y.; Tateishi, Y. Erythrinan alkaloids and isoflavonoids from Eryhtina poepiigiana. Planta Med. 2001, 67, 871–873. [Google Scholar] [CrossRef]
- Wanjala, C.C.W.; Juma, B.F.; Bojase, G.; Gache, B.A.; Majinda, R.R.T. Erythrinaline alkaloids and antimicrobial flavonoids from Eryrhina latissima. Planta Med. 2002, 68, 640–642. [Google Scholar] [CrossRef]
- Juma, B.F.; Majinda, R.R.T. Erythrinaline alkaloids from the flowers and pods of Erythrina lysistemon and their DPPH radical scavenging proprieties. Phytochemistry 2004, 65, 1397–1404. [Google Scholar] [CrossRef]
- Tanaka, H.; Hattori, H.; Tanaka, T.; Sakai, E.; Tanaka, N.; Kulkarni, A.; Etoh, H. A new Erythrina alkaloid from Erythrina herbacea. J. Nat. Med. 2008, 62, 228–231. [Google Scholar] [CrossRef]
- Cui, L.; Thoung, P.T.; Fomum, T.; Oh, W.K. A new erythrinan alkaloid from the seed of Erythrina addsoniae. Arch. Pharm. Res. 2009, 32, 325–328. [Google Scholar]
- Silva, D.B.; Guaratini, T.; Vessecchi, R.; Lopes, N.P.; Cramer, B.; Humpf, H.U.; Lopes, J.L.C. H-2 unimolecular elimination in electrospray ionization mass spectrometry from erythraline, a spirocyclic alkaloid. Eur. J. Mass Spectrom. 2013, 19, 345–350. [Google Scholar] [CrossRef]
- Rosa, D.S.; Faggion, S.A.; Gavin, A.S.; Souza, M.A.; Fachim, H.A.; Santos, W.F.; Cunha, A.O.S.; Pereira, A.M.S.; Beleboni, R.O. Erysothrine, an alkaloid extracted from flowers of Erythrina mulungu Mart. ex Benth: Evaluating its anticonvulsant and anxiolytic potential. Epilepsy Behav. 2012, 23, 205–212. [Google Scholar] [CrossRef]
- Faggion, S.A.; Cunha, A.O.S.; Fachim, H.A.; Gavin, A.S.; Santos, W.F.; Pereira, A.M.S.; Beleboni, R.O. Anticonvulsant profile of the alkaloids (+)-erythravine and (+)-11-α-hydroxy-erythravine isolated from the flowers of Erythrina mulungu Mart ex Benth (Leguminosae–Papilionaceae). Epilepsy Behav. 2011, 20, 441–446. [Google Scholar] [CrossRef]
- Iturriaga-Vásquez, P.; Carbone, A.; García-Beltrán, O.; Livingstone, P.D.; Biggin, P.C.; Cassels, B.K.; Wonnacott, S.; Zapata-Torres, G.; Bermudez, I. Molecular determinats for competitive inhibition of α4β2 nicotinic acethylcoline receptors. Mol. Pharmacol. 2010, 78, 366–375. [Google Scholar] [CrossRef]
- Setti-Perdigão, P.; Serrano, M.A.R.; Flausino, O.A., Jr.; Bolzani, V.S.; Guimarães, M.Z.P.; Castro, N.G. Erythrina mulungu alkaloids are potent inhibitors of neuronal nicotinic receptor currents in mammalian cells. PLoS One 2013, 8, 1–5. [Google Scholar]
- Wang, J.B.; Mantsch, J.R. I-Tetrahydropalamatine, a potential new medication for the treatment of cocaine addiction. Future Med. Chem. 2012, 42, 177–186. [Google Scholar] [CrossRef]
- Wangchuk, P.; Keller, P.A.; Pyne, S.G.; Willis, A.C.; Kamchonwongpaisan, S. Antimalarial alkaloids from a Bhutanese traditional medicinal plant Corydalis dubia. J. Ethnopharmacol. 2012, 143, 310–313. [Google Scholar] [CrossRef]
- Dar, G.H.; Koul, S.T.; Naqshi, A.R.; Khuroo, A.A.; Malik, A.H. A new species of Corydalis DC. (Fumariaceae) from Kashmir, North-west Himalaya, India. Taiwania 2011, 56, 305–308. [Google Scholar]
- Shrestha, R.L.S.; Adhikari, A.; Marasini, B.P.; Jha, R.N.; Choudhary, M.I. Novel inhibitors of urease from Corydalis govaniana Wall. Phytochem. Lett. 2013, 6, 228–231. [Google Scholar]
- Guaratini, T.; Silva, D.B.; Bizaro, A.C.; Sartori, L.R.; Humpf, H.U.; Lopes, N.P.; Costa-Lotufo, L.V.; Lopes, J.L.C. In vitro metabolism studies of erythraline, the major spiroalkaloid from Erythrina verna. BMC Complement. Altern. Med. 2014, 14, 61–65. [Google Scholar] [CrossRef]
- Tanaka, H.; Tanaka, T.; Etoh, H.; Goto, S.; Terada, Y. Two new erythrinan alkaloids from Erythrina x bidwillii. Heterocycles 1999, 51, 2759–2764. [Google Scholar] [CrossRef]
- Dyke, S.F.; Quessy, S.N. Erythrina and related alkaloids. In The Alkaloids, 1st ed.; Manske, R.H.F., Rodrigo, R.G.A., Eds.; Academic Press: New York, NY, USA, 1981; Volume 18, pp. 1–93. [Google Scholar]
- Singh, H.; Chawla, A.S.; Kapoor, V.K.; Kumar, N.; Piatak, D.N.; Nowicki, W. Investigation of Erythrina spp. IX. chemical constituents Erythrina stricta bark. J. Nat. Prod. 1981, 44, 526–529. [Google Scholar] [CrossRef]
- Chawla, A.S.; Chunchatprasert, S.; Jackson, A.H. Studies of Erythrina alkaloids. VII-13C NMR spectral studies of some Erytrhina alkaloids. Org. Mag. Reson. 1983, 21, 39–41. [Google Scholar] [CrossRef]
- Amer, M.E.; Shamma, M.; Freyer, A.J. The tetracyclic erythrina alkaloids. J. Nat. Prod. 1991, 54, 329–363. [Google Scholar] [CrossRef]
- Dagne, E.; Steglich, W. 8-oxoerythraline: An alkaloid from Erythrina brucei. Phytochemistry 1984, 23, 449–451. [Google Scholar]
- Barton, D.H.R.; Gunatilaka, A.A.R.; Letcher, R.M.; Lobo, A.M.F.T.; Widdowson, D.A. Phenol oxidation and biosynthesis. Part XXII. The alkaloids of Erythrina lysistemon, E. abyssinica, E. poeppigiana, E. fusca, and E. lithosperma: The structure of erythratidine. J. Chem. Sci. Perkin. Trans. 1 1973, 874–880. [Google Scholar]
- Muhammad, I.; Dunbar, D.C.; Khan, S.I.; Tekwani, B.L.; Bedir, E.; Takamatsu, S.; Ferreira, D.; Walker, L.A. Antiparasitic alkaloids from Psychotria klugii. J. Nat. Prod. 2003, 66, 962–967. [Google Scholar] [CrossRef]
- Montenegro, H.; Gutierrez, M.; Romero, L.I.; Ortega-Barria, E.; Capson, T.L.; Rios, L.C. Aporphine alkaloids from Guatteria spp. with leishmanicidal activity. Planta Med. 2003, 69, 677–679. [Google Scholar] [CrossRef]
- Feitosa, L.G.P.; Guaratini, T.; Lopes, J.L.C.; Lopes, N.P.; Bizaro, A.C.; Silva, D.B. Alicação de espectrometria de massas com ionização por elétron na análise de alcaloides do mulungu. Quim. Nova 2012, 35, 2177–2180. [Google Scholar] [CrossRef]
- Mosmann, T.J. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1–9 are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Callejon, D.R.; Riul, T.B.; Feitosa, L.G.P.; Guaratini, T.; Silva, D.B.; Adhikari, A.; Shrestha, R.L.; Marques, L.M.M.; Baruffi, M.D.; Lopes, J.L.C.; et al. Leishmanicidal Evaluation of Tetrahydroprotoberberine and Spirocyclic Erythrina-Alkaloids. Molecules 2014, 19, 5692-5703. https://doi.org/10.3390/molecules19055692
Callejon DR, Riul TB, Feitosa LGP, Guaratini T, Silva DB, Adhikari A, Shrestha RL, Marques LMM, Baruffi MD, Lopes JLC, et al. Leishmanicidal Evaluation of Tetrahydroprotoberberine and Spirocyclic Erythrina-Alkaloids. Molecules. 2014; 19(5):5692-5703. https://doi.org/10.3390/molecules19055692
Chicago/Turabian StyleCallejon, Daniel R., Thalita B. Riul, Luis G. P. Feitosa, Thais Guaratini, Denise B. Silva, Achyut Adhikari, Ram L. Shrestha, Lucas M. M. Marques, Marcelo D. Baruffi, João L. C. Lopes, and et al. 2014. "Leishmanicidal Evaluation of Tetrahydroprotoberberine and Spirocyclic Erythrina-Alkaloids" Molecules 19, no. 5: 5692-5703. https://doi.org/10.3390/molecules19055692
APA StyleCallejon, D. R., Riul, T. B., Feitosa, L. G. P., Guaratini, T., Silva, D. B., Adhikari, A., Shrestha, R. L., Marques, L. M. M., Baruffi, M. D., Lopes, J. L. C., & Lopes, N. P. (2014). Leishmanicidal Evaluation of Tetrahydroprotoberberine and Spirocyclic Erythrina-Alkaloids. Molecules, 19(5), 5692-5703. https://doi.org/10.3390/molecules19055692





