Evaluation of Antioxidant and Antimicrobial Activities and Phenolic Profile for Hyssopus officinalis, Ocimum basilicum and Teucrium chamaedrys
Abstract
:1. Introduction
2. Results and Discussion
2.1. HPLC Analysis of Polyphenols
| Polyphenolic Compounds | m/z Value | tR ± SD (min) | H. officinalis | O. basilicum | T. chamaedrys |
|---|---|---|---|---|---|
| Caftaric acid | 311 | 3.54 ± 0.05 | <0.2 | <0.2 | NF |
| Gentisic acic | 179 | 3.52 ± 0.04 | <0.2 | <0.2 | <0.2 |
| Caffeic acid | 179 | 5.60 ± 0.04 | <0.2 | <0.2 | NF |
| Chlorogenic acid | 353 | 5.62 ± 0.05 | <0.2 | <0.2 | <0.2 |
| p-Coumaric acid | 163 | 9.48 ± 0.08 | <0.2 | 21.26 ± 0.63 | 25.68 ± 0.33 |
| Ferulic acid | 193 | 12.8 ± 0.10 | 36.92 ± 1.00 | 7.09 ± 0.07 | NF |
| Isoquercitrin | 463 | 19.60 ± 0.10 | 32.78 ± 0.23 | 179.19 ± 1.93 | 524.8 ± 2.75 |
| Rutin | 609 | 20.20 ± 0.15 | 21.93 ± 0.72 | 425.71 ± 2.15 | 85.42 ± 0.9 |
| Rosmarinic acid | 360 | 20.8 ± 0.10 | <0.2 | <0.2 | <0.2 |
| Quercitrin | 447 | 23.64 ± 0.13 | 4.02 ± 0.07 | 50.39 ± 0.85 | 18.52 ± 0.49 |
| Quercetin | 301 | 26.80 ± 0.15 | 1.79 ± 0.03 | 3.39 ± 0.09 | NF |
| Luteolin | 285 | 29.10 ± 0.19 | 2.25 ± 0.03 | 6.06 ± 0.05 | 20.42 ± 0.47 |
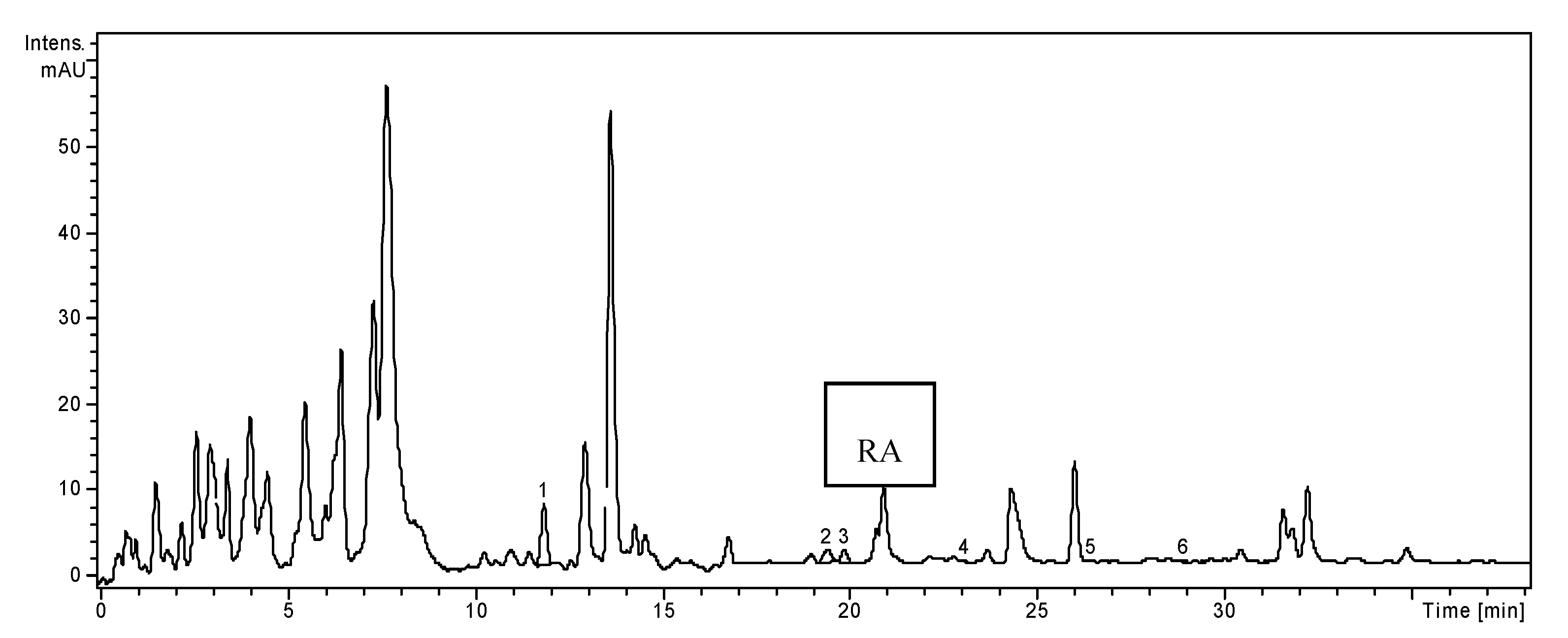
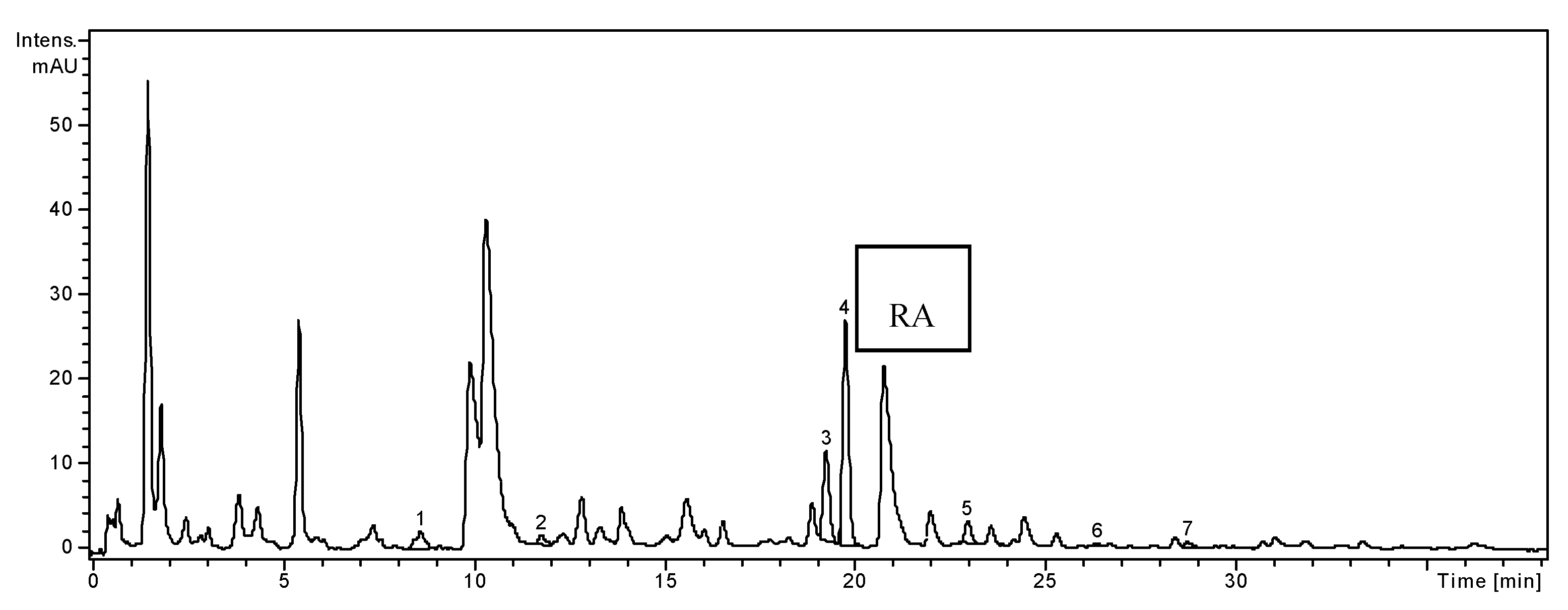
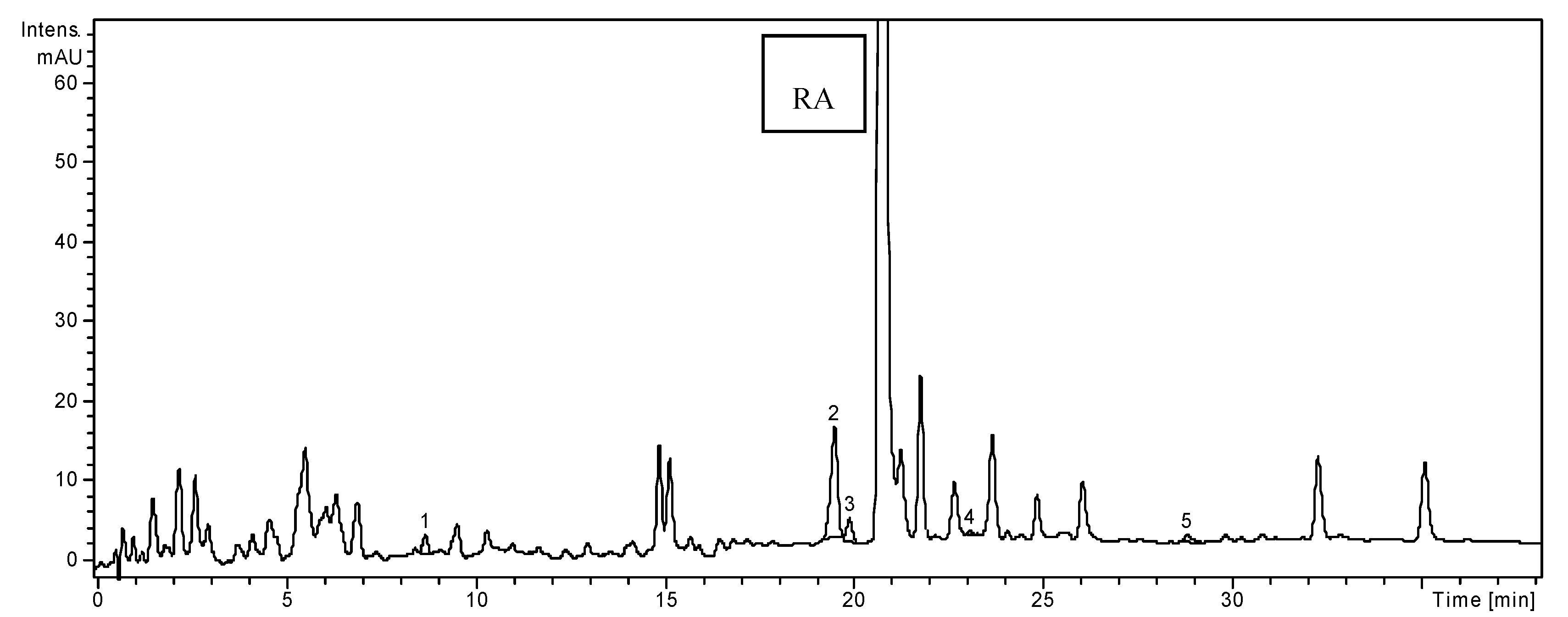
2.2. Determination of Phenolic Compounds Content
| Samples | TPC (mg GAE/g Plant Material) | Flavonoids (mg RE/g Plant Material) | Caffeic Acid Derivatives (mg CAE/g Plant Material) |
|---|---|---|---|
| H. officinalis | 77.72 ± 1.83 | 1.30 ± 0.10 | 9.25 ± 0.75 |
| O. basilicum | 175.57 ± 2.43 | 6.72 ± 0.19 | 12.11 ± 0.39 |
| T. chamaedrys | 243.65 ± 3.46 | 9.75 ± 0.25 | 12.51 ± 0.20 |
2.3. Antioxidant Activity
| Samples | IC50 (µg/mL) | TEAC (µmol Trolox/mg Plant Material) | HAPX (%) |
|---|---|---|---|
| H. officinalis | 125.44 ± 4.70 | 57.39 ± 13.68 | 16.17 ± 3.58 |
| O. basilicum | 124.95 ± 4.46 | 25.69 ± 2.96 | 18.84 ± 1.12 |
| T. chamaedrys | 26.70 ± 0.96 | 87.77 ± 0.33 | 12.87 ± 3.35 |
| Trolox | 11.20 ± 0.20 | – | – |
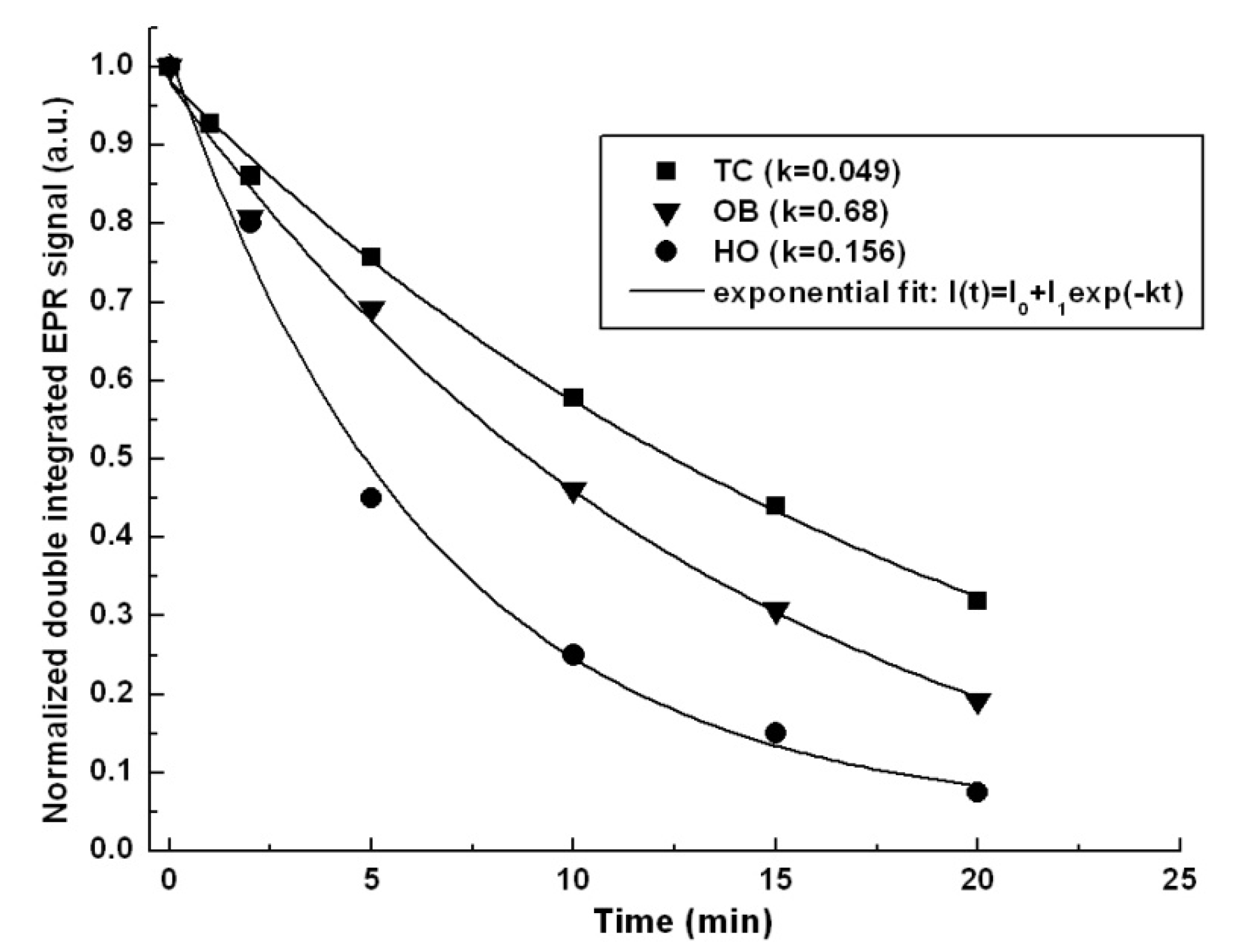
2.4. Antimicrobial Activity
| Samples | Zone of Inhibition (mm) | ||||
|---|---|---|---|---|---|
| Staphylococcus Aureus | Listeria Monocytogenes | Escherichia Coli | Salmonella Typhimurium | Candida Albicans | |
| H. officinalis | 16.0 ± 0.07 | 12.0 ± 0.00 | 10.0 ± 0.05 | 10.0 ± 0.02 | 16 ± 0.05 |
| O. basilicum | 16.0 ± 0.05 | 11.0 ± 0.1 | 10.0 ± 0.05 | 11.0 ± 0.00 | 18 ± 0.1 |
| T. chamaedrys | 20 ± 0.1 | 15 ± 0.05 | 12 ± 0.15 | 11 ± 0.05 | 22 ± 0.00 |
| Gentamicin | 19 ± 0.05 | 18 ± 0.02 | 22 ± 0.00 | 18 ± 0.01 | – |
| Fluconazole | – | – | – | – | 25 ± 0.00 |
3. Experimental
3.1. Plant Materials and Extraction Procedure
3.2. Chemicals and Instrumentation
3.3. HPLC-MS Analysis
Apparatus and Chromatographic Conditions
| Peak No. | Phenolic Compounds | m/z | RT ± SD (min) | Peak No. | Phenolic Compounds | m/z | RT ± SD (min) |
|---|---|---|---|---|---|---|---|
| 1 | Caftaric acid | 311 | 3.54 ± 0.05 | 11 | Rutin | 609 | 20.20 ± 0.15 |
| 2 | Gentisic acid | 179 | 3.52 ± 0.04 | 12 | Myricetin | 317 | 21.13 ± 0.12 |
| 3 | Caffeic acid | 179 | 5.60 ± 0.04 | 13 | Fisetin | 285 | 22.91 ± 0.15 |
| 4 | Chlorogenic acid | 353 | 5.62 ± 0.05 | 14 | Quercitrin | 447 | 23.64 ± 0.13 |
| 5 | p-Coumaric acid | 163 | 9.48 ± 0.08 | 15 | Quercetin | 301 | 26.80 ± 0.15 |
| 6 | Ferulic acid | 193 | 12.8 ± 0.10 | 16 | Patuletin | 331 | 29.41 ± 0.12 |
| 7 | Sinapic acid | 223 | 15.00 ± 0.10 | 17 | Luteolin | 285 | 29.10 ± 0.19 |
| 8 | Cichoric acid | 473 | 15.96 ± 0.13 | 18 | Kaempferol | 285 | 32.48 ± 0.17 |
| 9 | Hyperoside | 463 | 18.60 ± 0.12 | 19 | Apigenin | 279 | 33.10 ± 0.15 |
| 10 | Isoquercitrin | 463 | 19.60 ± 0.10 | Rosmarinic acid | 360 | 20.8 ± 0.10 |
3.4. Determination of Total Polyphenols, Caffeic Acid Derivatives and Flavonoids Content
3.5. In Vitro Antioxidant Activity Assays
3.5.1. DPPH Bleaching Assay
3.5.2. TEAC Assay (Trolox Equivalent Antioxidant Capacity)
3.5.3. Hemoglobin/Ascorbate Peroxidase Activity Inhibition (HAPX) Assay
3.5.4. EPR Measurements
3.6. Determination of Antimicrobial Activity
3.7. Statistical Analysis
4. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Ciocârlan, V. Illustrated Flora of Romania. Pteridophyta et Spermatophyta; Ceres Publishing House: Bucharest, Romania, 2009; pp. 644–670, 644–645, 662, 670. [Google Scholar]
- De Judicibus, M. Botanical Notebook; Warburton, Vic., Ed.; Custom Book Centre; University of Melbourne: Melbourne, Australia, 2011; p. 116. [Google Scholar]
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126. [Google Scholar] [CrossRef]
- Amiri, H. Antioxidant activity of the essential oil and methanolic extract of Teucrium orientale (L.) subsp. taylori (Boiss.) Rech. f. Iran J. Pharm. Res. 2010, 9, 417–423. [Google Scholar]
- Stankovic, S.M.; Stefanovic, O.; Comic, L.; Topuzovic, M.; Radojevic, I.; Solujic, S. Antimicrobial activity, total phenolic content and flavonoid concentrations of Teucrium species. Cent. Eur. J. Biol. 2012, 7, 664–671. [Google Scholar] [CrossRef]
- Stankovic, S.M.; Topuzovic, M.; Solujic, S.; Mihailovic, V. Antioxidant activity and concentration of phenols and flavonoids in the whole plant and plant parts of Teucrium chamaedrys L. var. glanduliferum Haussk. J. Med. Plants Res. 2010, 4, 2092–2098. [Google Scholar]
- Stankovic, S.M.; Topuzovic, M.; Markovic, A.; Pavlovic, D.; Solujic, S.; Niciforovic, N.; Mihailovic, V. Antioxidant activity, phenol and flavonoid contents of different Teucrium chamaedrys L. extracts. Biotechnol. Equip. 2010, 24, 82–86. [Google Scholar]
- Proestos, C.; Chorianopoulos, N.; Nychas, G.J.; Komaitis, M. RP-HPLC analysis of the phenolic compounds of plant extracts. Investigation of their antioxidant capacity and antimicrobial activity. J. Agric. Food Chem. 2005, 53, 190–1195. [Google Scholar]
- Bezic, N.; Vuko, E.; Dunkic, V.; Ruscic, M.; Blazevic, I.; Burcul, F. Antiphytoviral activity of sesquiterpene-rich essential oils from four croatian Teucrium species. Molecules 2011, 16, 8119–8129. [Google Scholar] [CrossRef]
- Gursoy, N.; Tepe, B. Determination of the antimicrobial and antioxidative properties and total phenolics of two ‘‘endemic’’ Lamiaceae species from Turkey: Ballota rotundifolia L. and Teucrium chamaedrys C. Koch. Plant Foods Hum. Nutr. 2009, 64, 135–140. [Google Scholar] [CrossRef]
- Kadifkova, P.T.; Kulevanova, S.; Stefova, M. In vitro antioxidant activity of some Teucrium species (Lamiaceae). Acta Pharmaceut. 2005, 55, 207–214. [Google Scholar]
- Kucuk, M.; Gulec, C.; Yasar, A.; Ucuncu, O.; Nuran, Y.; Coskuncelebi, K.; Terzioglu, S.; Nurettin, Y. Chemical composition and antimicrobial activities of the essential oils of Teucrium chamaedrys subsp. chamaedrys, T. orientale var. puberulens, and T. chamaedrys subsp. lydium. Pharm. Biol. 2006, 44, 592–599. [Google Scholar] [CrossRef]
- Fathiazad, F.; Hamedeyazdan, S. A review on Hyssopus officinalis L. Composition and biological activities. Afr. J. Pharm. Pharmacol. 2011, 5, 1959–1966. [Google Scholar]
- Ebrahimzadeh, M.A.; Nabavi, S.M.; Nabavi, S.F.; Bahramian, F.; Bekhradnia, A.R. Antioxidant and free radical scavenging activity of H. officinalis L. var. angustifolius, V. odorata, B. hyrcana and C. speciosum. Pak. J. Pharm. Sci. 2010, 23, 29–34. [Google Scholar]
- Kizil, S.; Hasimi, N.; Tolan, V.; Kilinc, E.; Karatas, H. Chemical composition, antimicrobial and antioxidant activities of hyssop (Hyssopus officinalis L.) essential oil. Not. Bot. Horti. Agrobo. 2010, 38, 99–103. [Google Scholar]
- Benedec, D.; Oniga, I.; Tiperciuc, B.; Popescu, H. Cercetări preliminare asupra unor compuşi polifenolici din Hyssopus officinalis L. (Lamiaceae). Nota 1. Farmacia 2002, 50, 54–58. [Google Scholar]
- Benedec, D.; Oniga, I.; Crişan, G.; Popescu, H. Cercetări preliminare asupra unor compuşi polifenolici din Hyssopus officinalis L. (Lamiaceae). Nota 2. Clujul Med. 2003, 76, 444–449. [Google Scholar]
- Benedec, D.; Pârvu, A.E.; Popescu, H.; Tiperciuc, B. In vitro effect of Hyssopus officinalis extract on pathogenic fungi on ornamental plants. Farmacia 2003, 51, 67–73. [Google Scholar]
- Alinezhad, H.; Azimi, R.; Zare, M.; Ebrahimzadeh, M.A.; Eslami, S.; Nabavi, S.F.; Nabavi, S.M. Antioxidant and antihemolytic activities of ethanolic extract of flowers, leaves and stems of Hyssopus officinalis L. var. angustifolius. Int. J. Food Prop. 2013, 16, 1169–1178. [Google Scholar] [CrossRef]
- Soleimani, H.; Barzegar, M.; Sahari, M.A.; Naghdi Badi, H. An investigation on the antioxidant activities of Hyssopus officinalis L. and Echinacea purpurea L. plant extracts in oil model system. J. Med. Plants 2011, 10, 61–72. [Google Scholar]
- Moro, A.; Zalacain, A.; de Hurtado, M.J.; Carmona, M. Effects of agronomic practices on volatile composition of Hyssopus officinalis L. essential oils. Molecules 2011, 16, 4131–4139. [Google Scholar] [CrossRef]
- Tomar, U.S.; Daniel, V.; Shrivastava, K.; Panwar, M.S.; Pant, P. Comparative evaluation and antimicrobial activity of Ocimum basilicum Linn. (Labiatae). J. Global Pharmacol. Technol. 2010, 2, 49–53. [Google Scholar]
- Politeo, O.; Jukic, M.; Milos, M. Chemical composition and antioxidant capacity of free volatile aglycones from basil (Ocimum basilicum L.) compared with its essential oil. Food Chem. 2007, 101, 379–385. [Google Scholar] [CrossRef]
- Benedec, D.; Pârvu, A.E.; Oniga, I.; Toiu, A.; Tiperciuc, B. Effects of Ocimum basilicum L. extract on experimental acute inflammation. Rev. Med. Chir. Soc. Med. Nat. Lasi. 2007, 3, 1065–1069. [Google Scholar]
- Benedec, D.; Oniga, I.; Toiu, A.; Tiperciuc, B.; Tămaş, M.; Vârban, I.D.; Crisan, G. GC-MS analysis of the essential oil obtained from Ocimum basilicum L. “Holland” cultivar. Farmacia 2013, 61, 448–453. [Google Scholar]
- Kaurinovic, B.; Popovic, M.; Vlaisavljevic, S.; Trivic, S. Antioxidant capacity of Ocimum basilicum L. and Origanum vulgare L. extracts. Molecules 2011, 16, 7401–7414. [Google Scholar] [CrossRef]
- Sgherri, C.; Cecconami, S.; Pinzino, C.; Navari-Izzo, F.; Izzo, R. Levels of antioxidants and nutraceuticals in basil grown in hydroponics and soil. Food Chem. 2010, 123, 416–422. [Google Scholar] [CrossRef]
- Benedec, D.; Vlase, L.; Hanganu, D.; Oniga, I. Antioxidant potential and polyphenolic content of Romanian Ocimum basilicum. Dig. J. Nanomater. Bios. 2012, 7, 1263–1270. [Google Scholar]
- Meda, R.N.T.; Vlase, L.; Lamien-Meda, A.; Lamien, C.E.; Muntean, D.; Tiperciuc, B.; Oniga, I.; Nacoulma, O.G. Identification and quantification of phenolic compounds from Balanites aegyptiaca (L) Del (Balanitaceae) galls and leaves by HPLC-MS. Nat. Prod. Res. 2011, 25, 93–99. [Google Scholar] [CrossRef]
- Vlase, L.; Parvu, M.; Parvu, E.A.; Toiu, A. hemical constituents of three Allium species from Romania. Molecules 2013, 18, 114–127. [Google Scholar]
- Benedec, D.; Vlase, L.; Oniga, I.; Mot, A.C.; Damian, G.; Hanganu, D.; Duma, M.; Silaghi-Dumitrescu, R. polyphenolic composition, antioxidant and antibacterial activities for two Romanian subspecies of Achillea distans Waldst. et Kit. ex Willd. Molecules 2013, 18, 8725–8739. [Google Scholar] [CrossRef]
- Tamas, M.; Benedec, D.; Vlase, L.; Scarlat, A. The identification and quantitative determination of rosmarinic acid by TLC and HPLC/MS from medicinal Lamiaceae species. In Proceedings of the Conference on Medicinal and Aromatic Plants of South-East European Countries, Iasi, Romania, 28–31 May 2006; Alma Mater Publishing House: Bacau, Romania, 2006; pp. 573–577. [Google Scholar]
- Ghasemzadeh, A.; Ghasemzadeh, N. Flavonoids and phenolic acids: Role and biochemical activity in plants and human. J. Med. Plants Res. 2011, 31, 6697–6703. [Google Scholar]
- Tamokou, J.D.D.; Chouna, J.R.; Fischer-Fodor, E.; Chereches, G.; Barbos, O.; Damian, G.; Benedec, D.; Duma, M.; Nkeng Efouet, P.A.; Wabo, H.K.; et al. Anticancer and antimicrobial activities of some antioxidant-rich Cameroonian medicinal plants. PloS One 2013, 8, e55880. [Google Scholar] [CrossRef]
- Reeves, D.S.; White, L.O. Principles of Methods of Assaying Antibiotics. In Pharmaceutical Microbiology, 3rd ed.; Hugo, W.B., Russel, A.D., Eds.; Blackwell Scientific Publication: Oxford, UK, 1983; pp. 140–162. [Google Scholar]
- Council of Europe. European Pharmacopoeia, 5th ed.; Council of Europe: Strasbourg Cedex, France, 2005; p. 221. [Google Scholar]
- Romanian Pharmacopoeia Commission National Medicines Agency. Romanian Pharmacopoeia, Xth ed.; Medical Publishing House: Bucharest, Romania, 1993; p. 335. [Google Scholar]
- Antonini, E.; Brunori, M. Hemoglobin and Myoglobin in Their Reaction with Ligands; North-Holland Publishing Company: Amsterdam, The Netherlands, 1971; pp. 98–134. [Google Scholar]
- Mot, A.C.; Damian, G.; Sarbu, C.; Silaghi-Dumitrescu, R. Redox reactivity in propolis: Direct detection of free radicals in basic medium and interaction with hemoglobin. Redox Rep. 2009, 14, 267–274. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar] [CrossRef]
- Slinkard, K.; Singleton, V.L. Total phenol analyses: Automation and comparison with manual methods. Am. J. Enol. Viticult. 1977, 28, 49–55. [Google Scholar]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef]
- Simirgiotis, M.J. Antioxidant capacity and HPLC-DAD-MS profiling of chilean peumo (Cryptocarya alba) fruits and comparison with german peumo (Crataegus monogyna) from Southern Chile. Molecules 2013, 18, 2061–2080. [Google Scholar] [CrossRef]
- Obon, J.M.; Castellar, M.R.; Cascales, J.A.; Fernandez-Lopez, J.A. Assessment of the TEAC method for determining the antioxidant capacity of synthetic red food colorants. Food Res. Int. 2005, 38, 843–845. [Google Scholar] [CrossRef]
- Cooper, C.E.; Silaghi-Dumitrescu, R.; Rukengwa, M.; Alayash, A.I.; Buehler, P.W. Peroxidase activity of hemoglobin towards ascorbate and urate: A synergistic protective strategy against toxicity of hemoglobin-based oxygen carriers (HBOC). Biochim. Biophys. Acta 1784, 1415–1420. [Google Scholar]
- Espinoza, M.; Olea-Azar, C.; Speisky, H.; Rodríguez, J. Determination of reactions between free radicals and selected Chilean wines and transition metals by ESR and UV–vis technique. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2009, 71, 1638–1643. [Google Scholar] [CrossRef]
- Sample Availability: Samples are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Vlase, L.; Benedec, D.; Hanganu, D.; Damian, G.; Csillag, I.; Sevastre, B.; Mot, A.C.; Silaghi-Dumitrescu, R.; Tilea, I. Evaluation of Antioxidant and Antimicrobial Activities and Phenolic Profile for Hyssopus officinalis, Ocimum basilicum and Teucrium chamaedrys. Molecules 2014, 19, 5490-5507. https://doi.org/10.3390/molecules19055490
Vlase L, Benedec D, Hanganu D, Damian G, Csillag I, Sevastre B, Mot AC, Silaghi-Dumitrescu R, Tilea I. Evaluation of Antioxidant and Antimicrobial Activities and Phenolic Profile for Hyssopus officinalis, Ocimum basilicum and Teucrium chamaedrys. Molecules. 2014; 19(5):5490-5507. https://doi.org/10.3390/molecules19055490
Chicago/Turabian StyleVlase, Laurian, Daniela Benedec, Daniela Hanganu, Grigore Damian, Ioan Csillag, Bogdan Sevastre, Augustin C. Mot, Radu Silaghi-Dumitrescu, and Ioan Tilea. 2014. "Evaluation of Antioxidant and Antimicrobial Activities and Phenolic Profile for Hyssopus officinalis, Ocimum basilicum and Teucrium chamaedrys" Molecules 19, no. 5: 5490-5507. https://doi.org/10.3390/molecules19055490
APA StyleVlase, L., Benedec, D., Hanganu, D., Damian, G., Csillag, I., Sevastre, B., Mot, A. C., Silaghi-Dumitrescu, R., & Tilea, I. (2014). Evaluation of Antioxidant and Antimicrobial Activities and Phenolic Profile for Hyssopus officinalis, Ocimum basilicum and Teucrium chamaedrys. Molecules, 19(5), 5490-5507. https://doi.org/10.3390/molecules19055490








