3. Experimental
3.1. General
All reagents were of commercial quality. Solvents were dried and purified by standard methods. Analytical TLC was performed on aluminum sheets coated with a 0.2 mm layer of silica gel 60 F254. Silica gel 60 (230–400 mesh) was used for flash chromatography. Analytical HPLC was performed on a Sunfire C18 (4.6 × 150 mm, 3.5 μm) column, with a flow rate of 1 mL/min, and using a tunable UV detector set at 214 nm. 10%–100% gradient of CH3CN (solvent A) in 0.05% of TFA in H2O (solvent B) in 30 min was used as mobile phase. 1H-NMR spectra were recorded at 300 or 400 MHz, using TMS as reference, and 13C-NMR spectra were recorded at 75 or 100 MHz. The NMR spectra assignment was based on COSY, HSQC, and HMBC spectra. ESI-MS spectra were performed, in positive mode, using MeOH as solvent. MW experiments were carried out in a EmrysTM Synthesizer MW reactor (Biotage AB, surface IR sensor). Elemental analyses were obtained on a CH-O-RAOID apparatus. Optical rotations were determined in a Perkin Elmer 141 polarimeter.
3.2. Synthesis of Benzyl 2-[(2RS)-[(1S)-(3-benzylureido)-2-phenylethyl]-5-oxopiperazin-1-yl]acetate (2) and (1S,9aRS)- 1-benzyl-3,7-dioxooctahydro-1H-pyrazino[1,2-a]pyrazine (3)
The epimeric mixture of piperazinones
1 [
23] [(
R:
S) = (3:1)] (500 mg, 1.07 mmol) was dissolved in a solution of HCl in EtOAc (3.4 N, 20 mL) and the mixture was stirred at room temperature for 30 min. Afterwards, the solvent was evaporated to dryness, the residue was dissolved in CH
3CN/H
2O (1:3, 8 mL) and the solution was lyophilized. Benzyl isocyanate (199 μL, 1.61 mmol) and Et
3N (224 μL, 1.61 mmol) were added to a solution of the lyophilized powder in THF (40 mL) and the mixture was stirred for 1 h. Afterwards, the solvent was removed under low pressure and the residue was dissolved in CH
2Cl
2 (60 mL). The solution was washed with H
2O (2 × 10 mL), brine (10 mL), dried over Na
2SO
4 and evaporated to dryness. The residue was purified by flash chromatography, with 0%–5% MeOH gradient in EtOAc as mobile phase, to afford the epimeric mixture of ureas
2 [(
R:
S) = (3:1)] as a foam (215 mg, 40%), along with the 1
H-pyrazino[1,2-
a]pyrazines
3 [
23] (83 mg, 30%).
Benzyl 2-[(2RS)-[(1S)-(3-benzylureido)-2-phenylethyl]-5-oxopiperazin-1-yl]acetate (2). HPLC tR: 20.02 min [(R)-2] and 21.24 min [(S)-2]; 1H-NMR (300 MHz, CDCl3). (R)-2 δ (ppm): 2.56 (dd, 1H, J = 10.5 and 13.5 Hz, CH2-Ph), 2.82 (dd, 1H, J = 4 and 13.5 Hz, CH2-Ph), 3.06 (dt, 1H, J = 4.5 and 9 Hz, 2-H), 3.24 (m, 1H, 3-H), 3.26 (d, J = 18 Hz, 6-H), 3.43 (s, 2H, CH2CO2Bn), 3.46 (d, 1H, J = 18 Hz, 6-H), 3.60 (m, 1H, 3-H), 3.89 (m, 1H, 2-CH), 4.32 [d, 2H, J = 5.5 Hz, CH2 (NHBn)], 5.04 [m, 1H, NHBn], 5.10 [s, 2H, CH2 (CO2Bn)], 5.45 (m, 1H, 4-H), 5.70 (m, 1H, 2-CHNH), 7.14–7.35 (m, 15H, Ar). (S)-2 δ (ppm): 2.56 (m, 1H, CH2-Ph), 2.82 (m, 1H, CH2-Ph), 3.06 (m, 1H, 2-H), 3.24 (m, 1H, 3-H), 3.50 (d, J = 17.5 Hz, 6-H), 3.43 (s, 2H, CH2CO2Bn), 3.58 (d, 1H, J = 17.5 Hz, 6-H), 3.60 (m, 1H, 3-H), 3.89 (m, 1H, 2-CH), 4.32 [m, 2H, CH2 (NHBn)], 5.04 (m, 1H, NHBn), 5.10 [s, 2H, CH2 (CO2Bn)], 5.45 (m, 1H, 4-H), 5.70 (m, 1H, 2-CHNH), 7.14–7.35 (m, 15H, Ar); 13C-NMR (75 MHz, CDCl3). (R)-2 δ (ppm): 36.7 [C3], 37.4 [CH2-Ph], 44.5 [CH2 (NHBn)], 51.3 [C6 and CH2CO2Bn], 53.0 [C2-CH], 58.0 [C2], 66.9 [CH2 (CO2Bn)], 127.3, 127.4, 127.6, 128.4, 128.5, 128.6, 128.7, 129.0, 129.2 [15CH (Ar)], 135.2 [C (CO2Bn)], 136.0 [C (Ph)], 139.3 [C (NHBn)], 158.2 [CO (Urea)], 169.2 [C5], 171.0 [CO2]. (S)-2 δ (ppm): 36.7 [C3], 37.4 [CH2-Ph], 44.5 [CH2 (NHBn)], 51.3 [CH2CO2Bn], 53.0 [C2-CH], 55.6 [C6], 58.0 [C2], 66.9 [CH2 (CO2Bn)], 127.3, 127.4, 127.6, 128.4, 128.5, 128.6, 128.7, 129.0, 129.2 [CH (Ar)], 135.2 [C (CO2Bn)], 136.0 [C (Ph)], 139.3 [C (NHBn)], 158.2 [CO (Urea)], 169.2 [C5], 171.0 [CO2]; ES-MS m/z 501.2 [M+1]+; C29H32N4O5 (%): C: 69.58, H: 6.44, N: 11.19. Found (%): C: 69.73, H: 6.32, N: 11.45.
3.3. General Procedure for the Synthesis of the Piperazinone-Derived Acids 4 and 14
Pd(C) (10%) was added to a solution of the corresponding epimeric mixture of piperazinones
1 [
23] or
13 [
23] [(
R:
S) = (3:1)] (1.00 mmol) in MeOH (50 mL) and the mixture was hydrogenated at 1 atm of H
2 at room temperature for 1 h. Afterwards, the reaction mixture was filtered and the solvent was evaporated under reduced pressure to obtain the epimeric mixture of the corresponding acids
4 or
14 [(
R:
S) = (3:1)].
2-[(2RS)-[(1S)-((tert-Butoxycarbonyl)amino)-2-phenyl-ethyl]-5-oxopiperazin-1-yl] acetic acid (4). Foam (377.4 mg, 100%); HPLC tR: 13.99 min [(R)-4] and 13.39 min [(S)-4]; 1H-NMR (500 MHz, DMSO-d6). (R)-4 δ (ppm): 1.24 (s, 9H, Boc), 2.56 (dd, 1H, J = 10 and 10.5 Hz, CH2-Ph), 2.88 (m, 1H, 2-H), 2.97 (dd, 1H, J = 3.5 and 10.5 Hz, CH2-Ph), 3.19 (m, 2H, 3-H), 3.30 (m, 1H, 6-H), 3.47 (d, 3H, J = 17 Hz, 6-H and CH2CO2H), 3.80 (m, 1H, 2-CH), 6.80 (d, 1H, J = 9.5 Hz, NHBoc), 7.02–7.36 (m, 5H, Ph), 7.76 (s, 1H, 4-H). (S)-4 δ (ppm): 1.25 (s, 9H, Boc), 2.47 (m, 1H, CH2-Ph), 2.82 (dd, 1H, J = 2 and 13.5 Hz, CH2-Ph), 2.92 (m, 1H, 2-H), 3.19 (m, 2H, 3-H), 3.31 (m, 1H, 6-H), 3.42 (m, 1H, 6-H), 3.47 (m, 2H, CH2CO2H), 3.80 (m, 1H, 2-CH), 6.89 (d, 1H, J = 9.5 Hz, NHBoc), 7.02–7.36 (m, 5H, Ph), 7.75 (s, 1H, 4-H); 13C-NMR (125 MHz, DMSO-d6). (R)-4 δ (ppm): 28.6 [3CH3 (Boc)], 37.9 [CH2-Ph], 38.9 [C3], 51.8 [C2-CH], 53.3 [CH2CO2H], 54.0 [C6], 58.4 [C2], 78.1 [C (Boc)], 126.2, 128.4, 129.7 [5CH (Ph)], 139.6 [C (Ph)], 155.8 [CO (Boc)], 168.3 [C5], 172.6 [CO2]. (S)-4 δ (ppm): 28.6 [3CH3 (Boc)], 35.7 [CH2-Ph], 38.8 [C3], 52.0 [C2-CH], 53.0[C6], 54.1 [CH2CO2H], 59.0 [C2], 78.0 [C (Boc)], 126.3, 128.4, 129.4 [5CH (Ph)], 139.8 [C (Ph)], 155.5 [CO (Boc)], 169.2 [C5], 172.4 [CO2]; ES-MS m/z 378.0 [M+1]+; C19H27N3O5 (%): C: 60.46, H: 7.21, N: 11.13. Found (%): C: 60.60, H: 7.02, N: 11.25.
3.4. General Procedure for the Synthesis of the Piperazinone-Derived Pseudotripeptides 7a,b
HOBt (136 mg, 1.00 mmol), DIC (309 μL, 2.00 mmol) and a solution of the corresponding benzylamides H-Orn(Boc)-NHBn (
6a) [
26] and H-Lys(Boc)-NHBn (
6b) [
27] (1.50 mmol) in dry DMF (4 mL) were added to a solution of the epimeric mixture of the piperazinone-derived acid
4 (1.00 mmol) in dry CH
2Cl
2 (16 mL) and stirred for 24 h. Afterwards, the solvent was removed under reduced pressure and the residue was dissolved in EtOAc (100 mL). This solution was washed with a solution of 10% citric acid (2 × 20 mL), a saturated solution of NaHCO
3 (2 × 20 mL) and brine (20 mL), dried over Na
2SO
4, and evaporated to dryness. The residue was purified by flash chromatography, with 1%–10% MeOH gradient in CH
2Cl
2 as mobile phase to afford the corresponding epimeric mixture of piperazinone derivatives
7a,
b [(
R:
S) = (3:1)].
N-[2-[(2RS)-[(1S)-((tert-Butoxycarbonyl)amino)-2-phenylethyl]-5-oxopiperazin-1-yl]acetyl]-Orn(Z)-NHBn (7a). Foam (429 mg, 60%); HPLC tR: 21.07 min; 1H-NMR (400 MHz, CDCl3) (R)-7a δ (ppm): 1.31 (s, 9H, Boc), 1.52 (m, 2H, γ-H), 1.67 (m, 1H, β-H), 1.84 (m, 1H, β-H), 2.82 (m, 1H, CH2-Ph), 2.86 (m, 1H, 2-H), 3.02 (m, 1H, CH2-Ph), 3.12 (m, 1H, δ-H), 3.30 (m, 1H, CH2CO), 3.34 (m, 1H, CH2CO), 3.36 (m, 3H, 3-H and 6-H), 3.42 (m, 1H, δ-H), 3.44 (m, 1H, 6-H), 4.04 (m, 1H, 2-CH), 4.34 [dd, 1H, J = 5.5 and 15 Hz, CH2 (NHBn)], 4.42 [dd, 1H, J = 5.5 and 15 Hz, CH2 (NHBn)], 4.70 (m, 3H, α-H and NHBoc), 4.83 [d, 1H, J = 12 Hz, CH2 (Z)], 4.93 [d, 1H, J = 12 Hz, CH2 (Z)], 5.12 (t, 1H, J = 6 Hz, NHZ), 6.38 (m, 1H, 4-H), 7.11–7.39 (m, 16H, Ar and NHBn), 7.79 (d, 1H, J = 8 Hz, α-NH). (S)-7a δ (ppm): 1.31 (s, 9H, Boc), 1.64(m, 1H, β-H), 1.86 (m, 1H, β-H), 3.26 (m, 1H, CH2CO), 3.32 (m, 1H, 6-H), 3.38 (m, 1H, CH2CO), 3.46 (m, 1H, 6-H), 3.94 (m, 1H, 2-CH), 4.35, 4.47 [m, 2H, CH2 (NHBn)], 4.82 [m, 1H, CH2 (Z)], 4.95 [m, 1H, CH2 (Z)], 5.04 (m, 1H, NHZ), 6.62 (m, 1H, 4-H), 7.11–7.39 (m, 16H, Ar and NHBn), 7.79 (d, 1H, J = 8 Hz, α-NH); 13C-NMR (100 MHz, CDCl3) (R)-7a δ (ppm): 26.3 [Cγ], 28.2 [3CH3 (Boc)], 30.3 [Cβ], 37.6 [CH2-Ph], 39.4 [C3], 39.7 [Cδ], 43.5 [CH2 (NHBn)], 51.3 [C2-CH], 51.5 [Cα], 54.0 [C6], 55.7 [CH2CO], 58.8 [C2], 66.6 [CH2 (Z)], 79.9 [C (Boc)], 126.7, 127.4, 127.7, 127.9, 128.1, 128.4, 128.6, 129.3 [15CH (Ar)], 136.4 [C (Ph)], 137.0 [C (Z)], 138.0 [C (NHBn)], 155.6 [CO (Boc)], 157.1 [CO (Z)], 168.9 [C5], 169.7 [CO], 171.5 [α-CONH]. (S)-7a δ (ppm): 28.2 [3CH3 (Boc)], 43.5 [CH2 (NHBn)], 66.6 [CH2 (Z)], 79.9 [C (Boc)], 126.6, 127.4, 127.6, 127.9, 128.0, 128.4, 128.6, 129.3 [15CH (Ar)], 136.4 [C (Ph)], 137.0 [C (Z)], 138.0 [C (NHBn)], 155.6 [CO (Boc)], 157.1 [CO (Z)], 171.5 [α-CONH]; ES-MS m/z 715.6 [M+1]+; C39H50N6O7 (%): C: 65.53, H: 7.05, N: 11.76. Found (%): C: 65.71, H: 6.98, N: 11.89.
N-[2-[(2RS)-[(1S)-((tert-Butoxycarbonyl)amino)-2-phenylethyl]-5-oxopiperazin-1-yl]acetyl]-Lys(Z)-NHBn (7b). Foam (481 mg, 66%); HPLC tR: 21.56 min [(R)-7b] and 21.41 min [(S)-7b]; 1H-NMR (400 MHz, CDCl3) (R)-7b δ (ppm): 1.27 (s, 9H, Boc), 1.29 (m, 2H, γ-H), 1.44 (m, 2H, δ-H), 1.65 (m, 1H, β-H), 1.83 (m, 1H, β-H), 2.77 (m, 1H, CH2-Ph), 2.78 (m, 1H, 2-H), 2.95 (d, 1H, J = 10 Hz, CH2-Ph), 3.05 (m, 2H, ε-H), 3.20 (m, 1H, 6-H), 3.22 (m, 2H, CH2CO), 3.23 (m, 2H, 3-H), 3.35 (m, 1H, 6-H), 4.00 (m, 1H, 2-CH), 4.34 [dd, 1H, J = 8 and 15 Hz, CH2 (NHBn)], 4.40 [dd, 1H, J = 8 and 15 Hz, CH2 (NHBn)], 4.48 (m, 1H, α-H), 4.81 (d, 1H, J = 8 Hz, NHBoc), 5.03 [m, 2H, CH2 (Z)], 5.25 (m, 1H, NHZ), 6.85 (m, 1H, 4-H), 7.08–7.40 (m, 16H, Ar and NHBn), 7.79 (d, 1H, J = 8 Hz, α-NH). (S)-7b δ (ppm): 1.27 (s, 9H, Boc), 1.29 (m, 2H, γ-H), 1.44 (m, 2H, δ-H), 1.65 (m, 1H, β-H), 1.83 (m, 1H, β-H), 2.78 (m, 1H, 2-H), 3.10 (m, 1H, 6-H), 3.15 (m, 2H, CH2CO), 3.23 (m, 2H, 3-H), 3.36 (m, 1H, 6-H), 3.90 (m, 1H, 2-CH), 4.28, 4.42 [m, 2H, CH2 (NHBn)], 4.46 (m, 1H, α-H), 5.03 [m, 2H, CH2 (Z)], 5.25 (m, 1H, NHZ), 6.77 (m, 1H, 4-H), 7.08–7.40 (m, 16H, Ar and NHBn), 7.73 (d, 1H, J = 8 Hz, α-NH); 13C-NMR (100 MHz, CDCl3) (R)-7b δ (ppm): 22.6 [Cγ], 28.1 [3CH3 (Boc)], 29.2 [Cδ], 32.1 [Cβ], 37.6 [CH2-Ph], 39.5 [C3], 40.5 [Cε], 43.4 [CH2 (NHBn)], 51.6 [C2-CH], 52.7 [Cα], 53.9 [C6], 55.7 [CH2CO], 58.7 [C2], 66.4 [CH2 (Z)], 79.6 [C (Boc)], 126.7, 127.3, 127.6, 128.0, 128.5, 128.6, 129.2 [15CH (Ar)], 136.6 [C (Ph)], 137.1 [C (Z)], 138.1 [C (NHBn)], 155.6 [CO (Boc)], 156.5 [CO (Z)], 169.4 [C5], 169.8 [CO], 171.5 [α-CONH]. (S)-7b δ (ppm): 22.6 [Cγ], 28.1 [3CH3 (Boc)], 29.6 [Cδ], 31.8 [Cβ], 39.4 [C6], 43.4 [CH2 (NHBn)], 51.9 [C2-CH], 52.7 [Cα], 53.9 [C6], 55.7 [CH2CO], 59.0 [C2], 66.4 [CH2 (Z)], 79.7 [C (Boc)], 126.6, 127.3, 127.6, 128.0, 128.5, 128.6, 129.2 [15CH (Ar)], 136.6 [C (Ph)], 137.1 [C (Z)], 138.1 [C (NHBn)], 155.8 [CO (Boc)], 156.5 [CO (Z)], 169.4 [C5], 170.3 [CO], 171.6 [α-CONH]; ES-MS m/z 729.3 [M+1]+; C40H52N6O7 (%): C: 65.91, H: 7.19, N: 11.53. Found (%): C: 65.72, H: 7.40, N: 11.68.
3.5. General Procedure for the N-Boc Removal in 7a,b. Synthesis of the Hydrochlorides 8a,b
The epimeric corresponding epimeric mixture of piperazine derivatives 7a,b [(R:S) = (3:1)] (0.60 mmol) was dissolved in 3.4 N HCl in EtOAc (15 mL) and the mixture was stirred at room temperature for 30 min. Afterwards, the solvent was evaporated to dryness, the residue was dissolved in CH3CN/H2O (1:3, 5 mL), and the solution was lyophilized. The desired epimeric mixture of hydrochlorides [(R:S) = (3:1)] was obtained quantitatively.
N-[2-[(2RS)-[(1S)-Amino-2-phenylethyl]-5-oxopiperazin-1-yl]acetyl]-Orn(Z)-NHBn hydrochloride (8a). Amorphous solid (391 mg, 100%); HPLC tR: 14.86 min; 1H-NMR (400 MHz, DMSO-d6) (R)-8a δ (ppm): 1.30 (m, 2H, γ-H), 1.57 (m, 1H, β-H), 1.72 (m, 1H, β-H), 2.59 (dd, 1H, J = 9 and 14 Hz, CH2-Ph), 2.86 (dd, 1H, J = 6.5 and 14 Hz, CH2-Ph and 2-H), 2.95 (m, 4H, δ-H and 3-H), 3.02 (d, 1H, J = 18 Hz, 6-H), 3.23 (d, 1H, J = 16.5 Hz, CH2CO), 3.33 (d, 1H, J = 16.5 Hz, CH2CO), 3.52 (d, 1H, J = 18 Hz, 3-H), 4.16 (m, 1H, 2-CH), 4.28 (m, 1H, α-H), 4.40 [m, 2H, CH2 (NHBn)], 4.97 [m, 2H, CH2 (Z)], 7.15–7.40 (m, 16H, Ar and NHZ), 7.63 (m, 1H, 4-H), 8.03 (m, 3H, NH2·HCl), 8.14 (d, 1H, J = 8.5 Hz, α-NH), 8.51 (t, 1H, J = 6 Hz, NHBn). (S)-8a δ (ppm): 1.30 (m, 2H, γ-H), 1.57 (m, 1H, β-H), 1.72 (m, 1H, β-H), 4.32 (m, 1H, α-H), 4.40 [m, 2H, CH2 (NHBn)], 4.98 [m, 2H, CH2 (Z)], 7.15–7.40 (m, 16H, Ar and NHZ), 8.03 (m, 3H, NH2·HCl), 8.20 (m, 1H, α-NH), 8.57 (m, 1H, NHBn); 13C-NMR (100 MHz, DMSO-d6) (R)-8a δ (ppm): 26.1 [Cγ], 28.8 [Cβ], 34.9 [C3], 36.0 [CH2-Ph], 40.5 [Cδ], 42.0 [CH2 (NHBn)], 49.7 [C6], 50.2 [C2-CH], 52.4 [Cα], 55.9 [C2], 58.5 [CH2CO], 65.1 [CH2 (Z)], 126.7, 127.0, 127.7, 128.3, 128.4, 128.6, 128.8 [15CH (Ar)], 136.6 [C (Ph)], 137.2 [C (Z)], 139.4 [C (NHBn)], 156.1 [CO (Z)], 168.2 [C5], 169.7 [CO], 171.6 [α-CONH]. (S)-8a δ (ppm): 26.0 [Cγ], 28.8 [Cβ], 42.0 [CH2 (NHBn)], 52.3 [Cα], 65.1 [CH2 (Z)], 126.6, 127.1, 127.8, 128.3, 128.4, 128.6, 128.8 [15CH (Ar)], 136.6 [C (Ph)], 137.2 [C (Z)], 139.4 [C (NHBn)], 156.1 [CO (Z)], 171.6 [α-CONH]; ES-MS m/z 615.8 [M−Cl]+; C34H42N6O5·HCl (%): C: 62.71, H: 6.66, N: 12.91. Found (%): C: 62.53, H: 6.78, N: 12.98.
N-[2-[(2RS)-[(1S)-Amino-2-phenylethyl]-5-oxopiperazin-1-yl]acetyl]-Lys(Z)-NHBn hydrochloride (8b). Amorphous solid (399 mg, 100%); HPLC tR: 15.25 min; 1H-NMR (400 MHz, DMSO-d6) (R)-8b δ (ppm): 1.22 (m, 2H, γ-H), 1.35 (m, 2H, δ-H), 1.66 (m, 2H, β-H), 2.59 (dd, 1H, J = 9 and 14 Hz, CH2-Ph), 2.84 (m, 1H, CH2-Ph), 2.88 (m, 1H, 2-H), 3.94 (m, 2H, ε-H), 3.01 (m, 2H, 3-H), 3.02 (d, 1H, J = 18 Hz, 6-H), 3.23 (d, 1H, J = 16.5 Hz, CH2CO), 3.37 (d, 1H, J = 16.5 Hz, CH2CO), 3.54 (d, 1H, J = 18 Hz, 6-H), 4.18 (m, 1H, 2-CH), 4.24 [m, 2H, CH2 (NHBn)], 4.26 (m, 1H, α-H), 4.98 [m, 2H, CH2 (Z)], 7.14–7.41 (m, 16H, Ar and NHZ), 7.63 (m, 1H, 4-H), 8.07 (m, 3H, NH2·HCl), 8.15 (d, 1H, J = 8.5 Hz, α-NH), 8.55 (t, 1H, J = 6 Hz, NHBn). (S)-8b δ (ppm): 1.22 (m, 2H, γ-H), 1.35 (m, 2H, δ-H), 1.66 (m, 2H, β-H), 3.01 (m, 1H, 3-H), 3.37 (m, 1H, CH2CO), 3.38 (m, 1H, CH2CO), 3.55 (m, 1H, 6-H), 4.24 [m, 2H, CH2 (NHBn)], 4.26 (m, 1H, α-H), 4.98 [m, 2H, CH2 (Z)], 7.14–7.41 (m, 16H, Ar and NHZ), 8.07 (m, 3H, NH2·HCl), 8.23 (m, 1H, α-NH), 8.58 (m, 1H, NHBn); 13C-NMR (100 MHz, DMSO-d6) (R)-8b δ (ppm): 22.9 [Cγ], 29.0 [Cδ], 31.2 [Cβ], 34.8 [C3], 36.0 [CH2-Ph], 39.5 [Cε], 42.0 [CH2 (NHBn)], 49.6 [C6], 51.6 [C2-CH], 52.8 [Cα], 55.9 [C2], 58.5 [CH2CO], 65.1 [CH2 (Z)], 126.6, 127.0, 127.7, 128.2, 128.3, 128.6, 128.8 [15CH (Ar)], 136.6 [C (Ph)], 137.3 [C (Z)], 139.4 [C (NHBn)], 156.1 [CO (Z)], 168.2 [C5], 169.6 [CO], 171.7 [α-CONH]. (S)-8b δ (ppm): 22.9 [Cγ], 29.0 [Cδ], 31.2 [Cβ], 42.0 [CH2 (NHBn)], 49.6 [C6], 58.5 [CH2CO], 65.1 [CH2 (Z)], 126.6, 127.0, 127.7, 128.2, 128.3, 128.6, 128.8 [15CH (Ar)], 136.6 [C (Ph)], 137.3 [C (Z)], 139.4 [C (NHBn)], 156.1 [CO (Z)], 171.7 [α-CONH]; ES-MS m/z 629.7 [M-Cl]+; C35H44N6O5·HCl (%): C: 63.19, H: 6.82, N: 12.63. Found (%): C: 63.02, H: 6.94, N: 12.74.
3.6. General Procedure for the Synthesis of the Piperazinone-Derived Ureas 9a,b and 10a,b
Et3N (168 μL, 1.20 mmol) and the corresponding isocyanate (phenyl or benzyl isocyanate) (1.20 mmol) were added to a solution of the corresponding hydrochloride 8a,b (0.60 mmol) in dry THF (30 mL). After stirring for 1 h at room temperature, the solvent was removed under reduced pressure and the residue was dissolved in CH2Cl2 (100 mL). The solution was washed with H2O (2 × 20 mL), brine (20 mL), dried over Na2SO4, and evaporated to dryness. The residue was purified by flash chromatography using 1%–8% MeOH gradient in EtOAc as mobile phase. The respective (R)- and (S)-epimers were resolved in this purification. The purified compounds were dissolved in CH3CN/H2O (1:2, 2 mL) and the solution was lyophilized, to afford the desired ureas 9a,b and 10a,b.
N
-[2-[5-Oxo-(2R
)-[2-phenyl-(1S
)-(3-phenylureido)ethyl]piperazin-1-yl]acetyl]-Orn(Z)-NHBn [(
R)-
9a]. Amorphous solid (176 mg, 46%);
![Molecules 19 04814 i001]()
= −0.1 (
c 1, MeOH); HPLC
tR: 20.30 min;
1H-NMR (400 MHz, CDCl
3)
δ (ppm): 1.30 (m, 2H, γ-H), 1.50 (m, 1H, β-H), 1.70 (m, 1H, β-H), 2.82 (m, 1H,
CH2-Ph), 2.83 (m, 1H, δ-H), 2.84 (m, 1H, 2-H), 2.88 (m, 1H,
CH2-Ph), 3.03 (m, 1H,
CH2CO), 3.14 (m, 1H, 6-H), 3.35 (m, 1H, 6-H), 3.37 (m, 1H, 3-H), 3.24 (m, 1H, δ-H), 3.44 (m, 1H,
CH2CO), 4.20 (m, 1H, 3-H), 4.25 (m, 1H, 2-
CH), 4.26 [m, 1H, CH
2 (NH
Bn)], 4.36 [m, 1H, CH
2 (NH
Bn)], 4.60 (m, 1H, α-H), 4.82 [d, 1H,
J = 12.5 Hz, CH
2 (Z)], 4.91 [d, 1H,
J = 12.5 Hz, CH
2 (Z)], 5.25 (m, 1H,
NHZ), 5.97 (m, 1H, 4-H), 6.12 (m, 1H, 2-CH
NH), 6.91–7.35 (m, 20H, Ar), 7.46 (m, 1H,
NHBn), 7.65 [m, 1H,
NHPh], 7.88 (m, 1H, α-NH);
13C-NMR (100 MHz, CDCl
3)
δ (ppm): 26.7 [C
γ], 29.9 [C
β], 37.9 [
CH2-Ph], 39.7 [C
δ], 40.1 [C
3], 43.7 [CH
2 (NH
Bn)], 51.6 [C
2-
CH], 52.0 [C
α], 54.9 [C
6], 57.8 [
CH2CO], 59.6 [C
2], 67.0 [CH
2 (Z)], 116.7, 119.7, 123.0, 127.0, 127.9, 120.4, 128.8, 129.0, 129.3 [20CH (Ar)], 134.3 [C (Ph)], 136.1 [C (Z)], 137.9 [C (NH
Bn)], 139.5 [C (NH
Ph)], 157.2 [CO (Z) and CO (Urea)], 168.7 [C
5], 170.0 [CO], 171.1 [α-CONH]; ES-MS
m/z 734.4 [M+1]
+; C
41H
47N
7O
6 (%): C: 67.10, H: 6.46, N: 13.36. Found (%): C: 67.28, H: 6.59, N: 13.19.
N
-[2-[5-Oxo-(2S
)-[2-phenyl-(1S
)-(3-phenylureido)ethyl]piperazin-1-yl]acetyl]-Orn(Z)-NHBn [(
S)-
9a]. Amorphous solid (79 mg, 18%);
![Molecules 19 04814 i001]()
= +9.2 (
c 1.5, MeOH);
tR: 21.41 min;
1H-NMR (500 MHz, CDCl
3)
δ (ppm): 1.32 (m, 1H, γ-H), 1.40 (m, 1H, γ-H), 1.53 (m, 1H, β-H), 1.72 (m, 1H, β-H), 2.54 (dd, 1H,
J = 11 and 13.5 Hz,
CH2-Ph), 2.90 (dd, 1H,
J = 4 and 13.5 Hz,
CH2-Ph), 2.92 (m, 1H, δ-H), 3.08 (m, 1H, 5-H), 3.10 (m, 1H, 3-H), 3.14 (m, 1H, 6-H), 3.32 (m, 2H,
CH2CO), 3.35 (m, 1H, δ-H), 3.54 (d, 1H,
J = 18 Hz, 6-H), 3.92 (m, 1H, 2-
CH), 3.95 (m, 1H, 3-H), 4.28 [dd, 1H,
J = 5 and 15 Hz, CH
2 (NH
Bn)], 4.44 [dd, 1H,
J = 6 and 15 Hz, CH
2 (NH
Bn)], 4.60 [d, 1H,
J = 13 Hz, CH
2 (Z)], 4.71 (m, 1H, α-H), 4.83 [d, 1H,
J = 13 Hz, CH
2 (Z)], 4.95 (m, 1H,
NHZ), 5.67 (m, 1H, 4-H), 5.94 (d, 1H,
J = 6 Hz, 2-CH
NH), 6.83–7.35 (m, 20H, Ar), 7.53 (m, 1H,
NHBn), 7.93 [m, 1H,
NHPh], 7.97 (d, 1H,
J = 8.5 Hz, α-NH);
13C-NMR (125 MHz, CDCl
3)
δ (ppm): 26.4 [C
γ], 31.1 [C
β], 35.8 [C
3], 37.8 [
CH2-Ph], 38.9 [C
δ], 43.8 [CH
2 (NH
Bn)], 50.8 [C
α], 51.9 [C
6], 52.3 [C
2-
CH], 57.7 [
CH2CO], 58.6 [C
2], 66.7 [CH
2 (Z)], 118.3, 122.2, 127.5, 127.8, 128.2, 128.5, 128.8, 128.9, 129.0, 129.3 [20CH (Ar)], 135.6 [C (Ph)], 136.2 [C (Z)], 137.3 [C (NH
Bn)], 139.6 [C (NH
Ph)], 155.6 [CO (Z)], 157.6 [CO (Urea)], 168.8 [C
5], 169.2 [CO], 172.9 [α-CONH]; ES-MS
m/z 734.5 [M+1]
+; C
41H
47N
7O
6 (%): C: 67.10, H: 6.46, N: 13.36. Found (%): C: 67.21, H: 6.30, N: 13.49.
N
-[2-[5-Oxo-(2R
)-[2-phenyl-(1S
)-(3-phenylureido)ethyl]piperazin-1-yl]acetyl]-Lys(Z)-NHBn [(
R)-
9b]. Amorphous solid (206 mg, 46%);
![Molecules 19 04814 i001]()
= −3.7 (
c 1.5, MeOH); HPLC
tR: 20.09 min;
1H-NMR (400 MHz, CDCl
3)
δ (ppm): 1.30 (m, 2H, γ-H), 1.40 (m, 2H, δ-H), 1.50 (m, 1H, β-H), 1.73 (m, 1H, β-H), 2.80 (m, 1H,
CH2-Ph), 2.87 (m, 1H,
CH2-Ph), 2.85 (m, 1H, ε-H), 2.92 (m, 1H, 2-H), 3.03 (m, 1H,
CH2CO), 3.16 (m, 1H, 6-H), 3.20 (m, 2H, 3-H and ε-H), 3.38 (m, 1H, 6-H), 3.44 (m, 1H,
CH2CO), 4.25 (m, 1H, 3-H), 4.28 (m, 1H, 2-
CH), 4.30 [m, 1H, CH
2 (NH
Bn)], 4.38 [m, 1H, CH
2 (NH
Bn)], 4.50 (m, 1H, α-H), 5.01 [m, 2H, CH
2 (Z)], 5.27 (m, 1H,
NHZ), 5.98 (m, 1H, 2-CH
NH), 6.23 (m, 1H, 4-H), 6.78–7.59 (m, 21H, Ar and
NHBn), 7.64 (m, 1H,
NHPh), 7.90 (m, 1H, α-NH);
13C-NMR (100 MHz, CDCl
3)
δ (ppm): 23.1 [C
γ], 29.3 [C
δ], 31.9 [C
β], 38.3 [
CH2-Ph], 39.6 [C
3], 40.7 [C
ε], 43.6 [CH
2 (NH
Bn)], 51.7 [C
6], 53.9 [C
2-
CH], 54.8 [C
α], 59.2 [
CH2CO], 59.8 [C
2], 66.8 [CH
2 (Z)], 119.2, 119.9, 123.1, 127.1, 127.5, 127.6, 127.7, 128.1, 128.4, 128.8, 128.9, 129.0, 129.1 [20CH (Ar)], 136.8 [C (Ph)], 137.5 [C (Z)], 138.2 [C (NH
Bn)], 139.2 [C (NH
Ph)], 156.1 [CO (Z)], 156.9 [CO (Urea)], 170.5 [C
5 and CO], 172.4 [α-CONH]; ES-MS
m/z 748.6 [M+1]
+; C
42H
49N
7O
6 (%): C: 67.45, H: 6.60, N: 13.11. Found (%): C: 67.62, H: 6.74, N: 13.02.
N
-[2-[5-Oxo-(2S
)-[2-phenyl-(1S
)-(3-phenylureido)ethyl]piperazin-1-yl]acetyl]-Lys(Z)-NHBn [(
S)-
9b]. Amorphous solid (67 mg, 15%);
![Molecules 19 04814 i001]()
= +6.7 (
c 0.9, MeOH); HPLC
tR: 21.76 min;
1H-NMR (500 MHz, CDCl
3)
δ (ppm): 1.23 (m, 2H, γ-H), 1.34 (m, 2H, δ-H), 1.67 (m, 1H, β-H), 1.82 (m, 1H, β-H), 2.58 (t, 1H,
J = 12.5 Hz,
CH2-Ph), 2.87 (m, 1H,
CH2-Ph), 2.90 (m, 1H, ε-H), 2.98 (m, 1H, 2-H), 3.05 (m, 1H, ε-H), 3.10 (m, 1H, 3-H), 3.18 (m, 1H, 6-H), 3.30 (m, 1H,
CH2CO), 3.42 (m, 1H,
CH2CO), 3.62 (m, 1H, 6-H), 3.95 (m, 1H, 2-
CH), 4.05 (m, 1H, 3-H), 4.35 [m, 1H, CH
2 (NH
Bn)], 4.50 [m, 2H, CH
2 (NH
Bn) and α-H], 4.98 [m, 2H, CH
2 (Z)], 5.02 (m, 1H,
NHZ), 5.69 (m, 1H, 2-CH
NH), 5.81 (m, 1H, 4-H), 6.94 (t, 1H,
J = 7.5 Hz,
NHBn), 6.98–7.14 (m, 20H, Ar), 7.98 (m, 1H,
NHPh), 8.09 (m, 1H, α-NH);
13C-NMR (125 MHz, CDCl
3)
δ (ppm): 22.2 [C
γ], 29.1 [C
δ], 32.6 [C
β], 35.7 [C
3], 37.7 [
CH2-Ph], 40.2 [C
ε], 43.8 [CH
2 (NH
Bn)], 52.0 [C
6 and C
2-
CH], 52.8 [C
α], 57.8 [
CH2CO], 58.7 [C
2], 66.5 [CH
2 (Z)], 118.4, 122.3, 127.7, 127.8, 128.1, 128.5, 128.9, 129.0, 129.3 [20CH (Ar)], 135.6 [C (Ph)], 136.5 [C (Z)], 137.1 [C (NH
Bn)], 139.6 [C (NH
Ph)], 155.6 [CO (Z)], 156.7 [CO (Urea)], 168.6 [C
5], 169.9 [CO], 172.0 [α-CONH]; ES-MS
m/z 748.7 [M+1]
+; C
42H
49N
7O
6 (%): C: 67.45, H: 6.60, N: 13.11. Found (%): C: 67.31, H: 6.81, N: 13.25.
N
-[2-[-(2R
)-[(1S
)-(3-Benzylureido)-2-phenylethyl]-5-oxo-piperazin-1-yl]acetyl]-Orn(Z)-NHBn [(
R)-
10a]. Amorphous solid (170 mg, 38%);
![Molecules 19 04814 i001]()
= −3.8 (
c 1.2, MeOH); HPLC
tR: 20.79 min;
1H-NMR (400 MHz, CDCl
3)
δ (ppm): 1.40 (m, 2H, γ-H), 1.55 (m, 1H, β-H), 1.70 (m, 1H, β-H), 2.57 (dd, 1H,
J = 11 and 13.5,
CH2-Ph), 2.86 (dd, 1H,
J = 3.5 and 13.5,
CH2-Ph), 3.00 (m, 1H, 2-H), 3.04 (m, 1H, δ-H), 3.09 (m, 1H, 3-H), 3.15 (m, 1H, 3-H), 3.25 (m, 2H,
CH2CO), 3.32 (m, 1H, δ-H), 3.50 (d, 1H,
J = 18 Hz, 6-H), 3.85 (m, 1H, 3-H), 3.94 (m, 1H, 2-
CH), 4.17 [dd, 1H,
J = 5 and 15 Hz, CH
2 (NH
Bn)], 4.23 [dd, 1H,
J = 6 and 15 Hz, CH
2 (NH
Bn)], 4.27 [dd, 1H,
J = 5 and 15 Hz, CH
2 (NH
Bn, Urea)], 4.36 [dd, 1H,
J = 5.5 and 15 Hz, CH
2 (NH
Bn, Urea)], 4.66 (m, 1H, α-H), 4.79 [d, 1H,
J = 13 Hz, CH
2 (Z)], 4.89 [d, 1H,
J = 13 Hz, CH
2 (Z)], 5.30 (t, 1H,
J = 6 Hz,
NHZ), 5.75 (m, 1H, 2-CH
NH), 5.90 (m, 1H, 4-H), 6.07 [t, 1H,
J = 5.5 Hz,
NHBn (Urea)], 7.08–7.39 (m, 20H, Ar), 7.50 (t, 1H,
J = 5.5 Hz,
NHBn), 7.99 [d, 1H,
J = 8.5, α-NH);
13C-NMR (100 MHz, CDCl
3)
δ (ppm): 26.1 [C
γ], 30.6 [C
β], 35.8 [C
3], 37.5 [
CH2-Ph], 39.3 [C
δ], 43.5 [CH
2 (NH
Bn)], 44.4 [CH
2 (NH
Bn, Urea)], 51.0 [C
α], 51.8 [C
6], 52.5 [C
2-
CH], 57.5 [
CH2CO], 58.3 [C
2], 66.5 [CH
2 (Z)], 127.0, 127.4, 127.5, 127.8, 128.1, 128.4, 128.5, 128.7, 129.0, 129.2 [20CH (Ar)], 135.8 [C (Ph)], 136.5 [C (Z)], 137.5 [C (NH
Bn)], 139.6 [C (NH
Bn, Urea)], 157.2 [CO (Z)], 158.5 [CO (Urea)], 168.9 [C
5], 170.0 [CO], 172.2 [α-CONH]; ES-MS
m/z 748.6 [M+1]
+; C
42H
49N
7O
6 (%): C: 67.45, H: 6.60, N: 13.11. Found (%): C: 67.28, H: 6.82, N: 13.20.
N
-[2-[-(2S
)-[(1S
)-(3-Benzylureido)-2-phenylethyl]-5-oxo-piperazin-1-yl]acetyl]-Orn(Z)-NHBn [(
S)-
10a]. Amorphous solid (76 mg, 17%);
![Molecules 19 04814 i001]()
= −8.2 (
c 1.0, MeOH); HPLC
tR: 20.09 min;
1H-NMR (500 MHz, (CD
3)
2CO)
δ (ppm): 1.55 (m, 2H, γ-H), 1.71 (m, 1H, β-H), 1.87 (m, 1H, β-H), 2.80 (dd, 1H,
J = 10 and 14,
CH2-Ph), 2.98 (dd, 1H,
J = 6 and 13, 2-H), 3.06 (dd, 1H,
J = 4 and 14,
CH2-Ph), 3.12 (m, 1H, δ-H), 3.16 (d, 1H,
J = 16.5 Hz, 6-H), 3.35 (s, 2H,
CH2CO), 3.40 (d, 1H,
J = 16.5 Hz, 6-H), 3.42 (m, 1H, 3-H), 3.51 (ddd, 1H,
J = 4, 13 and 15 Hz, 3-H), 4.16 [dd, 1H,
J = 6 and 15 Hz, CH
2 (NH
Bn, Urea)], 4.27 [dd, 1H,
J = 5.5 and 15 Hz, CH
2 (NH
Bn, Urea)], 4.38 (m, 1H, 2-
CH), 4.33 [d, 2H,
J = 6 Hz, CH
2 (NH
Bn)], 4.56 (dt, 1H,
J = 5 and 9 Hz, α-H), 5.00 [d, 1H,
J = 4.5 Hz, CH
2 (Z)], 4.89 [d, 1H,
J = 13 Hz, CH
2 (Z)], 6.00 [t, 1H,
J = 6 Hz,
NHBn (Urea)], 5.89 (d, 1H,
J = 9 Hz, 2-CH
NH), 5.90 (m, 1H, 4-H), 6.46 (t, 1H,
J = 5.5 Hz,
NHZ), 7.07–7.35 (m, 21H, Ar and 1-H), 8.03 (t, 1H,
J = 6 Hz,
NHBn), 8.14 [d, 1H,
J = 8.5, α-NH);
13C-NMR (125 MHz, (CD
3)
2CO)
δ (ppm): 27.8 [C
γ], 31.5 [C
β], 39.4 [
CH2-Ph], 40.9 [C
3], 41.6 [C
δ], 44.1 [CH
2 (NH
Bn)], 44.8 [CH
2 (NH
Bn, Urea)], 53.5 [C
2-
CH], 54.1 [C
α], 55.9 [C
6], 59.6 [
CH2CO], 62.5 [C
2], 67.1 [CH
2 (Z)], 127.6, 128.0, 128.3, 128.4, 128.8, 129.2, 129.3, 129.7, 129.8, 130.9 [20CH (Ar)], 139.2 [C (Z)], 140.5 [C (Ph)], 140.9 [C (NH
Bn)], 142.3 [C (NH
Bn, Urea)], 158.1 [CO (Z)], 159.8 [CO (Urea)], 170.7 [C
5], 171.5 [CO], 173.5 [α-CONH]; ES-MS
m/z 748.4 [M+1]
+; C
42H
49N
7O
6 (%): C: 67.45, H: 6.60, N: 13.11. Found (%): C: 67.60, H: 6.85, N: 13.01.
N
-[2-[(2R
)-[(1S
)-(3-Benzylureido)-2-phenylethyl]-5-oxo-piperazin-1-yl]acetyl]-Lys(Z)-NHBn [(
R)-
10b]. Amorphous solid (165 mg, 36%);
![Molecules 19 04814 i001]()
= −5.6 (
c 0.8, MeOH); HPLC
tR: 21.05 min;
1H-NMR (400 MHz, CDCl
3)
δ (ppm): 1.25 (m, 2H, γ-H), 1.42 (m, 2H, δ-H), 1.59 (dt, 1H,
J = 7.5 and 14 Hz, β-H), 1.75 (m, 1H, β-H), 2.54 (dd, 1H,
J = 11 and 13 Hz,
CH2-Ph), 2.86 (dd, 1H,
J = 2.5 and 13 Hz,
CH2-Ph), 3.01 (m, 1H, 2-H), 3.04 (m, 1H, ε-H), 3.10 (m, 1H, ε-H), 3.12 (m, 1H, 6-H), 3.16 (m, 1H, 3-H), 3.22 (m, 1H,
CH2CO), 3.28 (m, 1H,
CH2CO), 3.53 (d, 1H,
J = 18 Hz, 6-H), 3.86 (m, 1H, 3-H), 3.94 (m, 1H, 2-
CH), 4.24 [d, 2H,
J = 5.5 Hz, CH
2 (NH
Bn)], 4.27 [d, 1H,
J = 4.5 Hz, CH
2 (NH
Bn, Urea)], 4.37 [d, 1H,
J = 4.5 Hz, CH
2 (NH
Bn, Urea)], 4.43 (m, 1H, α-H), 5.00 [d, 2H,
J = 7 Hz, CH
2 (Z)], 5.24 (t, 1H,
J = 5 Hz,
NHZ), 5.66 (m, 1H, 2-CH
NH), 5.77 (m, 1H, 4-H), 6.04 [m, 1H, (
NHBn, Urea)], 7.09–7.41 (m, 21H, Ar and
NHBn), 7.97 (d, 1H,
J = 8 Hz, α-NH);
13C-NMR (100 MHz, CDCl
3)
δ (ppm): 22.4 [C
γ], 29.2 [C
δ], 32.5 [C
β], 35.9 [C
3], 37.5 [
CH2-Ph], 40.4 [C
ε], 43.6 [CH
2 (NH
Bn)], 44.2 [CH
2 (NH
Bn, Urea)], 51.7 [C
6], 52.4 [C
2-
CH], 52.6 [C
α], 57.7 [
CH2CO], 58.5 [C
2], 66.6 [CH
2 (Z)], 127.1, 127.5, 127.6, 127.7, 127.9, 128.1, 128.5, 128.7, 129.0, 129.2 [20CH (Ar)], 135.8 [C (Ph)], 136.5 [C (Z)], 137.4 [C (NH
Bn)], 139.6 [C (NH
Bn, Urea)], 156.7 [CO (Z)], 158.3 [CO (Urea)], 168.6 [C
5], 169.9 [CO], 172.0 [α-CONH]; ES-MS
m/z 763.2 [M+1]
+; C
43H
51N
7O
6 (%): C: 67.79, H: 6.75, N: 12.87. Found (%): C: 67.60, H: 7.01, N: 12.69.
N
-[2-[(2S
)-[(1S
)-(3-Benzylureido)-2-phenylethyl]-5-oxo-piperazin-1-yl]acetyl]-Lys(Z)-NHBn [(
S)-
10b]. Amorphous solid (59 mg, 13%);
![Molecules 19 04814 i001]()
= −11.2 (
c 0.9, MeOH); HPLC
tR: 20.47 min;
1H-NMR (500 MHz, (CD
3)
2CO)
δ (ppm): 1.30 (m, 2H, δ-H), 1.56 (m, 2H, γ-H), 1.68 (m, 1H, β-H), 1.83 (m, 1H, β-H), 2.82 (m, 1H,
CH2-Ph), 3.02 (m, 1H, 2-H), 3.05 (m, 1H,
CH2-Ph), 3.18 (m, 3H, ε-H and 6-H), 3.37 (m, 2H,
CH2CO), 3.16 (m, 1H, 3-H), 3.40 (m, 1H, 6-H), 3.55 (m, 2H, 3-H), 4.15 [m, 1H, CH
2 (NH
Bn, Urea)], 4.25 [m, 1H, CH
2 (NH
Bn, Urea)], 4.28 [m, 2H, CH
2 (NH
Bn)], 4.40 (m, 1H, 2-
CH), 4.51 (m, 1H, α-H), 5.00 [m, 2H, CH
2 (Z)], 5.98 (d, 1H,
J = 7 Hz, 2-CH
NH), 6.04 [m, 1H, (
NHBn, Urea)], 6.45 (m, 1H,
NHZ), 7.03 (m, 1H, 4-H), 7.08–7.41 (m, 20H, Ar), 8.04 (m, 1H, NH
Bn), 8.15 (d, 1H,
J = 8.5 Hz, α-NH);
13C-NMR (125 MHz, (CD
3)
2CO)
δ (ppm): 26.2 [C
γ], 29.7 [C
δ], 29.9 [C
β], 37.9 [
CH2-Ph], 39.2 [C
3], 39.9 [C
ε], 42.5 [CH
2 (NH
Bn)], 43.2 [CH
2 (NH
Bn, Urea)], 52.0 [C
2-
CH], 52.6 [C
α], 54.4 [C
6], 57.6 [
CH2CO], 61.0 [C
2], 65.5 [CH
2 (Z)], 126.1, 126.5, 126.6, 126.8, 127.3, 127.7, 127.8, 128.3, 128.4, 129.3, 129.6 [20CH (Ar)], 137.5 [C (Z)], 138.9 [C (Ph)], 139.4 [C (NH
Bn)], 140.7 [C (NH
Bn, Urea)], 156.5 [CO (Z)], 158.3 [CO (Urea)], 169.0 [C
5], 169.7 [CO], 171.9 [α-CONH]; ES-MS
m/z 763.3 [M+1]
+; C
43H
51N
7O
6 (%): C: 67.79, H: 6.75, N: 12.87. Found (%): C: 67.96, H: 6.93, N: 12.70.
3.7. General Procedure for the N-Z Removal in 9a,b and 10a,b. Synthesis of the Hydrochlorides (R)-(11a,b and 12a,b) and (S)-(11a,b and 12a,b)
Pd(C) (10%) and a 3.4 N solution of HCl in EtOAc (134 μL, 0.40 mmol) were added to a solution of (R)-(9a,b and -10a,b) and (S)-(9a,b and 10a,b) (0.20 mmol) in MeOH (5 mL), and the mixture was hydrogenated at 1 atm of H2 and room temperature for 1 h. Afterwards, the reaction mixture was filtered through celite and the solvent was evaporated under reduced pressure. The residue was dissolved in CH3CN/H2O (1:3, 2 mL) and the solution was lyophilized. (R)-(11a,b and 12a,b) and (S)-(11a,b and 12a,b) were obtained quantitatively.
N
-[2-[5-Oxo-(2R
)-[2-phenyl-(1S
)-(3-phenylureido)ethyl]piperazin-1-yl]acetyl]-Orn-NHBn hydrochloride [(
R)-
11a]. Amorphous solid (127 mg, 100%);
![Molecules 19 04814 i001]()
= +1.7 (
c 0.7, MeOH); HPLC
tR: 14.65 min;
1H-NMR (500 MHz, DMSO-
d6)
δ (ppm): 1.58 (m, 2H, γ-H), 1.63 (m, 1H, β-H), 1.80 (m, 1H, β-H), 2.62 (m, 1H,
CH2-Ph), 2.63 (m, 1H, 2-H), 2.73 (m, 1H,
CH2-Ph), 2.75 (m, 2H, δ-H), 3.05 (m, 1H, 3-H), 3.38 (m, 1H, 6-H), 3.40 (m, 2H,
CH2CO and 3-H), 3.50 (m, 1H,
CH2CO), 3.55 (m, 1H, 6-H), 4.05 (m, 1H, 2-
CH), 4.24 [dd, 1H,
J = 6 and 15 Hz, CH
2 (NH
Bn)], 4.31 [dd, 1H,
J = 6 and 15 Hz, CH
2 (NH
Bn)], 4.39 (dd, 1H,
J = 5 and 8 Hz, α-H), 6.58 (m, 1H, 2-CH
NH), 6.80–7.35 (m, 15H, Ar), 7.86 (m, 4H, 4-H and NH
2·HCl), ], 8.20 (m, 1H, α-NH), 8.65 (m, 1H,
NHBn), 8.74 [m, 1H,
NHPh];
13C-NMR (125 MHz, DMSO-
d6)
δ (ppm): 23.5 [C
γ], 29.2 [C
β], 38.2 [C
δ], 42.0 [CH
2 (NH
Bn)], 51.5 [C
α], 117.6, 121.1, 126.1, 126.8, 127.1, 128.1, 128.3, 128.6, 129.3 [15CH (Ar)], 139.1 [C (NH
Bn)], 140.2 [C (NH
Ph)], 155.1 [CO (Urea)], 171.0 [α-CONH]; ES-MS
m/z [M]
+ calculated for C
33H
41N
7O
4: 600.2; found: 600.5.
N
-[2-[5-Oxo-(2S
)-[2-phenyl-(1S
)-(3-phenylureido)ethyl]piperazin-1-yl]acetyl]-Orn-NHBn hydrochloride [(
S)-
11a]. Amorphous solid (127 mg, 100%);
![Molecules 19 04814 i001]()
= −1.6 (
c 1.1, MeOH); HPLC
tR: 15.07 min;
1H-NMR (500 MHz, DMSO-
d6)
δ (ppm): 1.48 (m, 2H, γ-H), 1.58 (m, 1H, β-H), 1.74 (m, 1H, β-H), 2.62 (m, 2H, δ-H), 2.79 (m, 1H,
CH2-Ph), 2.83 (m, 1H, 2-H), 2.89 (m, 1H,
CH2-Ph), 3.00 (m, 1H, 3-H), 3.35 (m, 1H,
CH2CO), 3.40 (m, 1H, 6-H), 3.42 (m, 1H, 3-H), 3.50 (m, 1H,
CH2CO), 3.65 (m, 1H, 6-H), 4.04 (m, 1H, 2-
CH), 4.25 [d, 2H,
J = 6, CH
2 (NH
Bn)], 4.31 (dd, 1H,
J = 5 and 8 Hz, α-H), 6.70 (m, 1H, 2-CH
NH), 6.79–7.49 (m, 15H, Ar), 7.81 (m, 4H, 4-H and NH
2·HCl), ], 8.21 (m, 1H, α-NH), 8.65 (t, 1H,
J = 6 Hz,
NHBn), 8.92 [m, 1H,
NHPh];
13C-NMR (125 MHz, DMSO-
d6)
δ (ppm): 23.4 [C
γ], 28.9 [C
β], 38.1 [C
δ], 42.0 [CH
2 (NH
Bn)], 51.5 [C
α], 117.6, 121.1, 126.4, 126.7, 127.0, 128.2, 128.4, 128.6, 129.0 [15CH (Ar)], 137.0 [C (Ph)], 139.1 [C (NH
Bn)], 140.3 [C (NH
Ph)], 155.3 [CO (Urea)], 170.9 [α-CONH]; ES-MS
m/z [M]
+ calculated for C
33H
41N
7O
4: 600.2; found: 600.5.
N
-[2-[5-Oxo-(2R
)-[2-phenyl-(1S
)-(3-phenylureido)ethyl]piperazin-1-yl]acetyl]-Lys-NHBn hydrochloride [(
R)-
11b]. Amorphous solid (130 mg, 100%);
![Molecules 19 04814 i001]()
= −3.0 (
c 1.7, MeOH); HPLC
tR: 14.97 min;
1H-NMR (500 MHz, DMSO-
d6)
δ (ppm): 1.27 (m, 2H, γ-H), 1.52 (m, 2H, δ-H), 1.58 (m, 1H, β-H), 1.70 (m, 1H, β-H), 2.61 (m, 1H,
CH2-Ph), 2.72 (m, 1H, ε-H), 2.84 (m, 1H, 2-H), 2.92 (m, 1H, ε-H), 2.95 (m, 1H,
CH2-Ph), 3.00 (m, 2H, 3-H), 3.38 (m, 1H, 3-H), 3.40 (m, 1H,
CH2CO), 3.42 (m, 1H, 6-H), 3.50 (m, 1H,
CH2CO), 3.54 (m, 1H, 3-H), 4.22 [m, 1H, CH
2 (NH
Bn)], 4.23 (m, 1H, 2-
CH), 4.31 [m, 1H, CH
2 (NH
Bn)], 4.32 (m, 1H, α-H), 6.80–7.37 (m, 16H, Ar and 2-CH
NH), 7.88 (m, 4H, NH
2·HCl and 4-H), 8.17 (m, 1H, α-NH), 8.61 (t, 1H,
J = 6 Hz,
NHBn), 8.90 (m, 1H,
NHPh);
13C-NMR (125 MHz, DMSO-
d6)
δ (ppm): 22.2 [C
γ], 26.5 [C
δ], 30.7 [C
β], 38.4 [C
ε], 42.0 [CH
2 (NH
Bn)], 52.5 [C
α], 117.7, 121.3, 126.3, 126.7, 127.0, 128.2, 128.3, 128.5, 129.2 [15CH (Ar)], 138.1 [C (Ph)], 139.2 [C (NH
Bn)], 140.0 [C (NH
Ph)], 155.3 [CO (Urea)], 171.3 [α-CONH]; ES-MS
m/z [M]
+ calculated for C
34H
43N
7O
4: 614.2; found: 614.5.
N
-[2-[5-Oxo-(2S
)-[2-phenyl-(1S
)-(3-phenylureido)ethyl]piperazin-1-yl]acetyl]-Lys-NHBn hydrochloride [(
S)-
11b]. Amorphous solid (130 mg, 100%);
![Molecules 19 04814 i001]()
= −4.2 (
c 0.4, MeOH); HPLC
tR: 15.21 min;
1H-NMR (500 MHz, DMSO-
d6)
δ (ppm): 1.29 (m, 2H, γ-H), 1.50 (m, 2H, δ-H), 1.55 (m, 1H, β-H), 1.73 (m, 1H, β-H), 2.72 (m, 1H,
CH2-Ph), 2.72 (m, 1H, ε-H), 2.74 (m, 2H, 3-H), 2.80 (m, 1H, ε-H), 2.89 (m, 1H,
CH2-Ph), 2.90 (m, 1H, 2-H), 3.13 (m, 1H,
CH2CO), 3.29 (m, 1H, 3-H), 3.33 (m, 1H, 6-H), 3.42 (m, 1H,
CH2CO), 3.63 (m, 1H, 6-H), 4.10 (m, 1H, 2-
CH), 4.18 [m, 1H, CH
2 (NH
Bn)], 4.20 (m, 1H, α-H), 4.35 [m, 1H, CH
2 (NH
Bn)], 6.55 (m, 1H, 2-CH
NH), 6.80–7.40 (m, 16H, Ar), 7.69 (m, 4H, NH
2·HCl and 4-H), 7.92 (d, 1H,
J = 9 Hz, α-NH), 8.56 (m, 1H,
NHBn), 8.70 (m, 1H,
NHPh);
13C-NMR (125 MHz, DMSO-
d6)
δ (ppm): 22.1 [C
γ], 26.8 [C
δ], 30.7 [C
β], 38.5 [C
ε], 42.0 [CH
2 (NH
Bn)], 51.6 [C
α], 116.5, 118.0, 127.0, 127.2, 127.5, 128.7, 128.9, 129.1, 129.5 [15CH (Ar)], 138.2 [C (Ph)], 139.4 [C (NH
Bn)], 140.0 [C (NH
Ph)], 171.2 [α-CONH]; ES-MS
m/z [M]
+ calculated for C
34H
43N
7O
4: 614.2; found: 614.5.
N
-[2-[(2R
)-[(1S
)-(3-Benzylureido)-2-phenylethyl]-5-oxo-piperazin-1-yl]acetyl]-Orn-NHBn hydrochloride [(
R)-
12a]. Amorphous solid (130 mg, 100%);
![Molecules 19 04814 i001]()
= −7.9 (
c 1.3, MeOH); HPLC
tR: 14.67 min;
1H-NMR (500 MHz, DMSO-
d6)
δ (ppm): 1.56 (m, 3H, γ-H and β-H), 1.78 (m, 1H, β-H), 2.60–3.16 (m, 6H,
CH2-Ph, 2-H, δ-H and 3-H), 3.20–3.86 (m, 5H, 3-H, 6-H and
CH2CO), 4.04 (m, 1H, 2-
CH), 4.20 [m, 2H, CH
2 (NH
Bn, Urea)], 4.26 [d, 2H,
J = 6 Hz, CH
2 (NH
Bn)], 4.34 (dd, 1H,
J = 5 and 8 Hz, α-H), 6.53 (m, 1H, 2-CH
NH), 6.74 [m, 1H,
NHBn (Urea)], 7.18–7.33 (m, 15H, Ar), 7.92 (m, 4H, NH
2·HCl and 4-H), 8.34 [m, 1H, α-NH), 8.70 (t, 1H,
J = 6 Hz,
NHBn);
13C-NMR (125 MHz, DMSO-
d6)
δ (ppm): 23.8 [C
γ], 29.5 [C
β], 39.5 [C
δ], 42.5 [CH
2 (NH
Bn)], 43.4 [CH
2 (NH
Bn, Urea)], 52.0 [C
α], 126.9, 127.0, 127.2, 127.4, 127.5, 128.6, 128.7, 128.9, 129.3 [15CH (Ar)], 137.5 [C (Ph)], 139.6 [C (NH
Bn)], 141.0 [C (NH
Bn, Urea)], 158.7 [CO (Urea)], 171.4 [α-CONH]; ES-MS
m/z [M]
+ calculated for C
34H
43N
7O
4: 614.2; found: 614.5.
N
-[2-[(2S
)-[(1S
)-(3-Benzylureido)-2-phenylethyl]-5-oxo-piperazin-1-yl]acetyl]-Orn-NHBn hydrochloride [(
S)-
12a]. Amorphous solid (130 mg, 100%);
![Molecules 19 04814 i001]()
= −3.2 (
c 1.2, MeOH); HPLC
tR: 15.01 min;
1H-NMR (500 MHz, DMSO-
d6)
δ (ppm): 1.58 (m, 3H, γ-H), 1.60 (m, 1H, β-H) 1.88 (m, 1H, β-H), 2.52–3.97 (m, 6H,
CH2-Ph, 2-H, δ-H and 3-H), 3.24–3.69 (m, 5H, 3-H, 6-H and
CH2CO), 4.03 (m, 1H, 2-
CH), 4.05 [m, 1H, CH
2 (NH
Bn, Urea)], 4.20 [m, 1H, CH
2 (NH
Bn, Urea)], 4.25 [m, 2H, CH
2 (NH
Bn)], 4.39 (m, 1H, α-H), 6.48 (m, 1H, 2-CH
NH), 6.96–7.36 [m, 16H, Ar and
NHBn (Urea)], 7.90 (m, 4H, NH
2·HCl and 4-H), 8.30 [m, 1H, α-NH), 8.70 (m, 1H,
NHBn);
13C-NMR (125 MHz, DMSO-
d6)
δ (ppm): 23.9 [C
γ], 29.6 [C
β], 38.7 [C
δ], 42.5 [CH
2 (NH
Bn)], 43.1 [CH
2 (NH
Bn, Urea)], 52.1 [C
α], 126.5, 126.8, 127.0, 127.1, 127.5, 128.5, 128.7, 129.7 [15CH (Ar)], 139.6 [C (NH
Bn)], 141.0 [C (NH
Bn, Urea)], 158.5 [CO (Urea)], 171.3 [α-CONH]; ES-MS
m/z [M]
+ calculated for C
34H
43N
7O
4: 614.2; found: 614.5.
N
-[2-[(2R
)-[(1S
)-(3-Benzylureido)-2-phenylethyl]-5-oxo-piperazin-1-yl]acetyl]-Lys-NHBn hydrochloride [(
R)-
12b]. Amorphous solid (133 mg, 100%);
![Molecules 19 04814 i001]()
= −4.3 (c 0.6, MeOH); HPLC
tR: 14.80 min;
1H-NMR (500 MHz, DMSO-
d6)
δ (ppm): 1.21 (m, 2H, γ-H), 1.49 (m, 2H, δ-H), 1.57 (m, 1H, β-H), 1.69 (m, 1H, β-H), 2.68 (m, 2H, ε-H), 2.75 (m, 1H,
CH2-Ph), 2.80 (m, 1H, 2-H), 2.82 (m, 1H,
CH2-Ph), 3.02 (m, 1H, 3-H), 3.25 (m, 1H,
CH2CO), 3.37 (m, 1H, 6-H), 3.40 (m, 2H,
CH2CO and 3-H), 3.60 (m, 1H, 6-H), 3.98 (m, 1H, 2-
CH), 4.20 [m, 2H, CH
2 (NH
Bn, Urea)], 4.24 (m, 1H, α-H), 4.25 [m, 2H, CH
2 (NH
Bn)], 6.35 (m, 1H, 2-CH
NH), 6.59 [m, 1H, (
NHBn, Urea)], 7.08–7.35 (m, 15H, Ar), 7.82 (m, 4H, NH
2·HCl and 4-H), 8.08 (m, 1H, α-NH), 8.57 (t, 1H,
J = 6 Hz,
NHBn);
13C-NMR (125 MHz, DMSO-
d6)
δ (ppm): 22.1 [C
γ], 26.5 [C
δ], 31.4 [C
β], 38.5 [C
ε], 42.0 [CH
2 (NH
Bn)], 42.9 [CH
2 (NH
Bn, Urea)], 52.0 [C
α], 126.4, 126.5, 126.7, 126.9, 127.0, 128.2, 128.4, 129.1 [15CH (Ar)], 137.3 [C (Ph)], 139.3 [C (NH
Bn)], 140.7 [C (NH
Bn, Urea)], 158.0 [CO (Urea)], 171.2 [α-CONH]; ES-MS
m/z [M]
+ calculated for C
35H
45N
7O
4: 628.2; found: 628.5.
N
-[2-[(2S
)-[(1S
)-(3-Benzylureido)-2-phenylethyl]-5-oxo-piperazin-1-yl]acetyl]-Lys-NHBn hydrochloride [(
S)-
12b]. Amorphous solid (133 mg, 100%);
![Molecules 19 04814 i001]()
= −1.6 (c 0.6, MeOH); HPLC
tR: 15.34 min;
1H-NMR (500 MHz, DMSO-
d6)
δ (ppm): 1.28 (m, 2H, γ-H), 1.47 (m, 1H, β-H), 1.50 (m, 2H, δ-H), 1.70 (m, 1H, β-H), 2.57–3.01 (m, 7H,
CH2-Ph, ε-H, 2-H and 3-H), 3.38 (m, 1H, 6-H), 3.42 (m, 1H,
CH2CO), 3.53 (m, 1H,
CH2CO), 3.62 (m, 1H, 6-H), 4.00 [m, 1H, CH
2 (NH
Bn, Urea)], 4.17 [m, 1H, CH
2 (NH
Bn, Urea)], 4.20 (m, 1H, 2-
CH), 4.26 [m, 2H, CH
2 (NH
Bn)], 4.30 (m, 1H, α-H), 6.45 (m, 1H, 2-CH
NH), 6.96–7.34 [m, 16H, Ar and (
NHBn, Urea)], 7.84 (m, 4H, NH
2·HCl and 4-H), 8.05 (m, 1H, α-NH), 8.60 (m, 1H,
NHBn);
13C-NMR (125 MHz, DMSO-
d6)
δ (ppm): 22.2 [C
γ], 26.5 [C
δ], 31.4 [C
β], 38.5 [C
ε], 42.0 [CH
2 (NH
Bn)], 42.6 [CH
2 (NH
Bn, Urea)], 52.5 [C
α], 126.4, 126.6, 126.7, 127.0, 128.1, 128.2, 129.3 [15CH (Ar)], 137.4 [C (Ph)], 139.6 [C (NH
Bn)], 141.0 [C (NH
Bn, Urea)], 159.0 [CO (Urea)], 171.5 [α-CONH]; ES-MS
m/z [M]
+ calculated for C
35H
45N
7O
4: 628.2; found: 628.5.
3.8. Synthesis of 2-[4-Benzyl-(2RS)-[(1S)-((tert-butoxycarbonyl)amino)-2-phenylethyl]-5-oxopiperazin-1-yl]acetic Acid (14)
This compound was obtained from the benzyl ester
13 [
23] by applying the general procedure of benzyl ester hydrogenolysis above indicated for the synthesis of
4. Foam (467.6 mg, 100%); HPLC
tR: 19.72 min [(
R)-
14] and 19.00 min [(
S)-
14];
1H-NMR (500 MHz, CDCl
3). (
R)-
14 δ (ppm): 1.35 (s, 9H, Boc), 2.73 (m, 2H,
CH2-Ph), 2.87 (m, 1H, 2-H), 3.23 (d, 1H,
J = 7.5 and 13 Hz, 3-H), 3.33 (d, 1H,
J = 5 and 13 Hz, 3-H), 3.38 (d, 1H,
J = 17 Hz,
CH2CO
2H), 3.49 (d, 2H,
J = 17 Hz, 6-H and
CH2CO
2H), 3.62 (d, 1H,
J = 17 Hz, 6-H), 4.00 (m, 1H, 2-
CH), 4.30 (d, 1H,
J = 9 Hz,
NHBoc), 4.52 [d, 1H,
J = 14.5 Hz, 4-CH
2 (Bn)], 4.68 [d, 1H,
J = 14.5 Hz, 4-CH
2 (Bn)], 6.93–7.40 (m, 10H, Ar). (
S)-
14 δ (ppm): 1.35 (s, 9H, Boc), 2.73 (m, 2H,
CH2-Ph), 2.97 (m, 1H, 2-H), 3.17 (m, 1H, 3-H), 3.33 (m, 1H, 3-H), 3.23 (m, 1H,
CH2CO
2H), 3.38 (m, 1H, 3-H), 3.49 (m, 1H,
CH2CO
2H), 3.63 (d, 1H,
J = 17.5 Hz, 6-H), 3.82 (m, 1H, 2-
CH), 4.30 (d, 1H,
J = 9 Hz,
NHBoc), 4.57 [m, 1H, 4-CH
2 (Bn)], 4.82 [m, 1H, 4-CH
2 (Bn)], 6.93–7.40 (m, 10H, Ar).
13C-NMR (125 MHz, DMSO-
d6).
(R)-
14 δ (ppm): 28.2 [3CH
3 (Boc)], 37.5 [
CH2-Ph], 44.1 [C
3], 49.7 [4-CH
2 (Bn)], 51.6 [C
2-
CH], 54.1 [C
6], 54.4 [
CH2CO
2H], 58.7 [C
2], 80.2 [C (Boc)], 126.7, 127.9, 128.6, 128.9 [10CH (Ar)], 136.2 [C (Bn)], 136.8 [C (Ph)], 155.7 [CO (Boc)], 167.8 [C
5], 172.2 [CO
2].
(S)-
14 δ (ppm): 28.2 [3CH
3 (Boc)], 37.5 [
CH2-Ph], 44.1 [C
3], 49.6 [4-CH
2 (Bn)], 51.6 [C
2-
CH], 54.1 [C
6], 54.4 [
CH2CO
2H], 58.7 [C
2], 80.2 [C (Boc)], 128.0, 128.3, 128.4, 129.1 [10CH (Ar)], 136.2 [C (Bn)], 136.8 [C (Ph)], 155.7 [CO (Boc)], 167.8 [C
5], 172.2 [CO
2]; ES-MS
m/z 468.2 [M+1]
+; C
26H
33N
3O
5 (%): C: 66.79, H: 7.11, N: 8.99. Found (%): C: 66.58, H: 7.25, N: 9.14.
3.9. General Procedure for the Synthesis of the Piperazinone-Derived Pseudotripeptides 15a–c
These compounds were prepared by applying the general procedure described for the synthesis of 7a,b.
N-[2-[4-Benzyl-(2RS)-[(1S)-((tert-butoxycarbonyl)-amino)-2-phenylethyl]-5-oxopiperazin-1-yl]acetyl]-Orn(Z)-NHBn (15a). Foam (523 mg, 65%); HPLC tR: 25.24 min; 1H-NMR (500 MHz, CDCl3) (R)-15a δ (ppm): 1.34 (s, 9H, Boc), 1.43 (m, 2H, γ-H), 1.67 (m, 1H, β-H), 1.86 (m, 1H, β-H), 2.76 (m, 1H, CH2-Ph), 2.83 (m, 1H, CH2-Ph), 2.86 (m, 1H, 2-H), 3.08 (m, 1H, 3-H), 3.23 (m, 2H, δ-H), 3.30 (m, 1H, 3-H), 3.35 (m, 1H, 6-H), 3.44 (m, 2H, CH2CO), 3.55 (d, 1H, J = 18 Hz, 6-H), 4.00 (m, 1H, 2-CH), 4.34 [dd, 1H, J = 6 and 15 Hz, CH2 (NHBn)], 4.42 (m, 1H, α-H), 4.39 [m, 1H, CH2 (NBn)], 4.44 [m, 1H, CH2 (NHBn)], 4.70 (m, 1H, NHBoc), 4.75 [m, 1H, CH2 (NBn)], 4.86 [m, 2H, CH2 (Z)], 5.06 (m, 1H, NHZ), 6.70 (m, 1H, NHBn), 7.02–7.46 (m, 20H, Ar), 7.74 (m, 1H, α-NH). (S)-15a δ (ppm): 1.34 (s, 9H, Boc), 1.67 (m, 1H, β-H), 1.86 (m, 1H, β-H), 2.56 (m, 1H, CH2-Ph), 2.74 (m, 1H, CH2-Ph), 3.08 (m, 1H, 3-H), 3.23 (m, 2H, δ-H), 3.30 (m, 1H, 3-H), 3.85 (m, 1H, 2-CH), 4.34 [m, 1H, CH2 (NHBn)], 4.42 (m, 1H, α-H), 4.44 [m, 1H, CH2 (NHBn)], 4.44 [m, 1H, CH2 (NHBn)], 4.50 [m, 1H, CH2 (NBn)], 4.64 (m, 1H, NHBoc), 4.80 [m, 1H, CH2 (NBn)], 4.86 [m, 2H, CH2 (Z)], 5.06 (m, 1H, NHZ), 6.70 (m, 1H, NHBn), 7.02–7.46 (m, 20H, Ar), 7.74 (m, 1H, α-NH); 13C-NMR (125 MHz, CDCl3) (R)-15a δ (ppm): 26.7 [Cγ], 28.4 [3CH3 (Boc)], 31.1 [Cβ], 37.7 [CH2-Ph], 39.5 [Cδ], 43.7 [C3 and CH2 (NHBn)], 50.1 [CH2 (NBn)], 51.4 [C2-CH], 51.7 [Cα], 54.6 [C6], 56.3 [CH2CO], 59.5 [C2], 66.8 [CH2 (Z)], 80.1 [C (Boc)], 126.9, 127.9, 128.0, 128.3, 128.5, 128.8, 129.0, 129.3 [20CH (Ar)], 136.5 [C (Ph) and C (NBn)], 136.9 [C (Z)], 138.1 [C (NHBn)], 155.6 [CO (Boc)], 157.3 [CO (Z)], 166.9 [C5], 169.6 [CO], 171.5 [α-CONH]. (S)-15a δ (ppm): 28.4 [3CH3 (Boc)], 30.5 [Cβ], 37.7 [CH2-Ph], 39.6 [Cδ], 43.7 [C3 and CH2 (NHBn)], 49.8 [CH2 (NBn)], 51.4 [C2-CH], 51.7 [Cα], 66.8 [CH2 (Z)], 80.1 [C (Boc)], 126.8, 127.8, 128.0, 128.6, 128.8, 129.1, 129.3 [20CH (Ar)], 136.5 [C (Ph) and C (NBn)], 136.9 [C (Z)], 138.1 [C (NHBn)], 155.6 [CO (Boc)], 157.3 [CO (Z)], 166.9 [C2], 169.6 [CO], 171.5 [α-CONH]; ES-MS m/z 806.6 [M+1]+; C46H56N6O7 (%): C: 68.63, H: 7.01, N: 10.44. Found (%): C: 68.50, H: 7.19, N: 10.62.
N-[2-[4-Benzyl-(2RS)-[(1S)-((tert-butoxycarbonyl)-amino)-2-phenylethyl]-5-oxopiperazin-1-yl]acetyl]-Lys(Z)-NHBn (15b). Foam (639 mg, 78%); HPLC tR: 25.42 min; 1H-NMR (500 MHz, CDCl3) (R)-15b δ (ppm): 1.31 (m, 11H, Boc and γ-H), 1.47 (m, 2H, δ-H), 1.65 (m, 1H, β-H), 1.86 (m, 1H, β-H), 2.69 (dd, 1H, J = 8 and 13 Hz, CH2-Ph), 2.78 (m, 1H, 2-H), 2.82 (m, 1H, CH2-Ph), 3.12 (m, 2H, ε-H), 3.19 (m, 1H, 6-H), 3.20 (m, 1H, CH2CO), 3.25 (m, 1H, 3-H), 3.35 (m, 1H, 6-H), 3.37 (m, 1H, CH2CO), 3.50 (d, 1H, J = 17 Hz, 3-H), 3.95 (m, 1H, 2-CH), 4.29 (d, 1H, J = 9 Hz, NHBoc), 4.39 [m, 1H, CH2 (NBn], 4.42 [m, 1H, CH2 (NHBn)], 4.44 (m, 1H, α-H), 4.45[m, 1H, CH2 (NHBn)], 4.75 [d, 1H, J = 14.5 Hz, CH2 (NBn)], 5.04 [m, 3H, CH2 (Z) and NHZ], 6.74 (m, 1H, NHBn), 7.01–7.38 (m, 20H, Ar), 7.67 (d, 1H, J = 8 Hz, α-NH). (S)-15b δ (ppm): 1.31 (m, 9H, Boc), 1.65 (m, 1H, β-H), 1.86 (m, 1H, β-H), 3.12 (m, 1H, CH2CO), 3.38 (m, 1H, 6-H), 3.39 (m, 1H, CH2CO), 3.54 (m, 1H, 6-H), 3.82 (m, 1H, 2-CH), 4.33 (d, 1H, J = 9 Hz, NHBoc), 4.42 [m, 1H, CH2 (NHBn)], 4.43 (m, 1H, α-H), 4.45[m, 1H, CH2 (NHBn)], 4.72 [m, 1H, CH2 (NBn], 4.79 [m, 1H, CH2 (NBn], 5.04 [m, 3H, CH2 (Z) and NHZ], 6.96 (m, 1H, NHBn), 7.01–7.38 (m, 20H, Ar), 7.55 (m, 1H, α-NH); 13C-NMR (125 MHz, CDCl3) (R)-15b δ (ppm): 22.7 [Cγ], 28.2 [3CH3 (Boc)], 29.4 [Cδ], 31.9 [Cβ], 37.7 [CH2-Ph], 40.5 [Cε], 43.6 [C3 and CH2 (NHBn)], 49.8 [CH2 (NBn)], 51.7 [C2-CH], 52.7 [Cα], 54.4 [C6], 56.1 [CH2CO], 59.4 [C2], 66.5 [CH2 (Z)], 79.9 [C (Boc)], 127.0, 127.7, 127.8, 128.0, 128.2, 128.5, 128.6, 128.8, 128.9, 129.0, 129.3 [20CH (Ar)], 136.2 [C (NBn)], 136.6 [C (Ph)], 136.7 [C (Z)], 138.0 [C (NHBn)], 155.4 [CO (Boc)], 156.4 [CO (Z)], 166.9 [C5], 169.6 [CO], 171.2 [α-CONH]. (S)-15b δ (ppm): 28.2 [3CH3 (Boc)], 31.6 [Cβ], 43.6 [C3 and CH2 (NHBn)], 49.6 [CH2 (NBn)], 51.7 [C2-CH], 54.4 [C6], 56.1 [CH2CO], 66.5 [CH2 (Z)], 79.9 [C (Boc)], 126.8, 127.7, 127.8, 128.1, 128.2, 128.5, 128.7, 128.8, 128.9, 129.0, 129.2 [20CH (Ar)], 136.2 [C (NBn)], 136.6 [C (Ph)], 136.7 [C (Z)], 138.0 [C (NHBn)], 155.4 [CO (Boc)], 156.4 [CO (Z)], 169.6 [CO], 171.2 [α-CONH]; ES-MS m/z 819.7 [M+1]+; C47H58N6O7 (%): C: 68.93, H: 7.14, N: 10.26. Found (%): C: 68.67, H: 7.36, N: 10.20.
N-[2-[4-Benzyl-(2RS)-[(1S)-((tert-butoxycarbonyl)-amino)-2-phenylethyl]-5-oxopiperazin-1-yl]acetyl]-Arg(Pbf)-NHBn (15c). Foam (705 mg, 73%); HPLC tR: 26.88 min; 1H-NMR (500 MHz, CDCl3) (R)-15c δ (ppm): 1.28 (s, 9H, Boc), 1.45 [s, 6H, 2CH3 (Pbf)], 1.53 (m, 2H, γ-H), 1.67 (m, 1H, β-H), 1.90 (m, 1H, β-H), 2.07 [s, 3H, CH3 (Pbf)], 2.48 [s, 3H, CH3 (Pbf)], 2.55 [s, 3H, CH3 (Pbf)], 2.73 (d, 1H, J = 8.5 and 13.5 Hz, CH2-Ph), 2.82 (m, 1H, 5-H), 2.84 (m, 1H, CH2-Ph), 2.93 [m, 2H, CH2 (Pbf)], 3.24 (m, 5H, 3-H, CH2CO and δ-H), 3.30 (m, 1H, 3-H), 3.32 (m, 1H, 6-H), 3.49 (d, 1H, J = 16.5 Hz, 6-H), 3.95 (m, 1H, 2-CH), 4.31 [dd, 1H, J = 6 and 15 Hz, CH2 (NHBn)], 4.38 [d, 1H, J = 14.5 Hz, CH2 (NBn)], 4.41 [dd, 1H, J = 5.5 and 15 Hz, CH2 (NHBn)], 4.50 (d, 1H, J = 9 Hz, NHBoc), 4.57 (dt, 1H, J = 4.5 and 9 Hz, α-H), 4.76 [d, 1H, J = 14.5 Hz, CH2 (NBn)], 6.41 [m, 3H, NHC(NH2) = N], 6.81–7.24 (m, 15H, Ar), 7.60 (m, 1H, NHBn), 7.74 (d, 1H, J = 8 Hz, α-NH). (S)-15c δ (ppm): 1.28 (s, 9H, Boc), 1.45 [s, 6H, 2CH3 (Pbf)], 1.67 (m, 1H, β-H), 1.90 (m, 1H, β-H), 2.07 [s, 3H, CH3 (Pbf)], 2.48 [s, 3H, CH3 (Pbf)], 2.55 [s, 3H, CH3 (Pbf)], 2.73 (m, 1H, CH2-Ph), 2.84 (m, 1H, CH2-Ph), 2.93 [m, 2H, CH2 (Pbf)], 3.20 (m, 1H, 3-H), 3.35 (m, 1H, 3-H), 3.83 (m, 1H, 2-CH), 4.31 [m, 1H, CH2 (NBn)], 4.41 [m, 1H, CH2 (NBn)], 4.50 (d, 1H, J = 9 Hz, NHBoc), 4.57 (m, 1H, α-H), 4.70 [m, 1H, CH2 (NBn)], 4.86 [m, 1H, CH2 (NBn)], 6.41 [m, 3H, NHC(NH2) = N], 6.81–7.24 (m, 15H, Ar), 7.62 (m, 1H, NHBn), 7.83 (d, 1H, J = 8 Hz, α-NH); 13C-NMR (125 MHz, CDCl3) (R)-15c δ (ppm): 12.4, 18.0, 19.3 [3CH3 (Pbf)], 25.4 [Cγ], 28.2 [3CH3 (Boc)], 28.6 [2CH3 (Pbf)], 31.0 [Cβ], 37.6 [CH2-Ph], 40.4 [Cδ], 43.2 [CH2 (Pbf)], 43.4 [CH2 (NHBn)], 44.0 [C3], 49.8 [CH2 (NBn)], 51.7 [C2-CH], 52.2 [Cα], 54.4 [C6], 56.5 [CH2CO], 59.4 [C2], 79.9 [C (Boc)], 86.4, 117.5, 124.6 [3C (Pbf)], 126.7, 127.2, 127.7, 127.9, 128.3, 128.5, 128.9, 129.1 [15CH (Ar)], 132.3 [2C (Pbf)], 136.1 [C (NBn)], 136.9 [C (Ph)], 138.2 [C (NHBn)], 138.4 [C (Pbf)], 155.5 [CO (Boc)], 156.3 [C (NHC(NH2) = N)], 158.8 [C (Pbf)], 167.3 [C5], 169.9 [CO], 171.3 [α-CONH]. (S)-15c δ (ppm): 12.4, 18.0, 19.3 [3CH3 (Pbf)], 28.2 [3CH3 (Boc)], 28.6 [2CH3 (Pbf)], 30.9 [Cβ], 37.6 [CH2-Ph], 43.2 [CH2 (Pbf)], 43.3 [CH2 (NHBn)], 44.0 [C3], 49.6 [CH2 (NBn)], 51.7 [C2-CH], 52.2 [Cα], 79.9 [C (Boc)], 86.4, 117.5, 124.6 [3C (Pbf)], 126.7, 127.2, 127.6, 127.9, 128.3, 128.6, 128.9, 129.1 [15CH (Ar)], 132.3 [2C (Pbf)], 136.1 [C (NBn)], 136.9 [C (Ph)], 138.2 [C (NHBn)], 138.4 [C (Pbf)], 155.5 [CO (Boc)], 156.3 [C (NHC(NH2) = N)], 158.8 [C (Pbf)], 171.3 [α-CONH]; ES-MS m/z 966.8 [M+1]+; C52H68N8O8S (%): C: 64.71, H: 7.10, N: 11.61. Found (%): C: 64.58, H: 7.26, N: 11.81.
3.10. Synthesis of the Hydrochlorides 16a–c
These compounds were obtained by applying the above indicated method of N-Boc removal.
N-[2-[4-Benzyl-(2RS)-[(1S)-amino-2-phenylethyl]-5-oxo-piperazin-1-yl]acetyl]-Orn(Z)-NHBn hydrochloride (16a). Amorphous solid (445 mg, 100%); HPLC tR: 16.76 min; 1H-NMR (500 MHz, DMSO-d6) (R)-16a δ (ppm): 1.40 (m, 1H, γ-H), 1.47 (m, 1H, γ-H), 1.57 (m, 1H, β-H), 1.70 (m, 1H, β-H), 2.85 (m, 1H, CH2-Ph), 2.90 (m, 1H, CH2-Ph), 2.98 (m, 1H, 2-H and δ-H), 3.21 (d, 1H, J = 17 Hz, 6-H), 3.30 (m, 2H, CH2CO), 3.46 (m, 2H, 3-H), 3.56 (d, 1H, J = 17 Hz, 6-H), 3.70 (m, 1H, 2-CH), 4.28 [m, 2H, CH2 (NHBn)], 4.30 (m, 1H, α-H), 4.45 [d, 1H, J = 15 Hz, CH2 (NBn)], 4.62 [d, 1H, J = 15 Hz, CH2 (NBn)], 4.97 [m, 2H, CH2 (Z)], 7.11–7.37 (m, 21H, Ar and NHZ), 8.11 (m, 3H, NH2·HCl), 8.19 (d, 1H, J = 8 Hz, α-NH), 8.54 (m, 1H, NHBn). (S)-16a δ (ppm): 1.40 (m, 1H, γ-H), 1.47 (m, 1H, γ-H), 1.57 (m, 1H, β-H), 1.70 (m, 1H, β-H), 3.15 (m, 1H, 2-H), 3.45 (m, 1H, 6-H), 3.46 (m, 2H, 3-H), 3.56 (m, 1H, 6-H), 3.60 (m, 1H, 2-CH), 4.28 [m, 3H, CH2 (NHBn) and CH2 (NBn)], 4.30 (m, 1H, α-H), 4.38 [d, 1H, J = 15 Hz, CH2 (NBn)], 4.97 [m, 2H, CH2 (Z)], 7.11–7.37 (m, 21H, Ar and NHZ), 8.11 (m, 3H, NH2·HCl), 8.43 (d, 1H, J = 8 Hz, α-NH), 8.52 (m, 1H, NHBn); 13C-NMR (125 MHz, DMSO-d6) (R)-16a δ (ppm): 26.0 [Cγ], 29.5 [Cβ], 34.6 [CH2-Ph], 40.5 [Cδ], 42.0 [CH2 (NHBn)], 43.5 [C3], 48.8 [CH2 (NBn)], 51.7 [C2-CH], 52.3 [Cα], 54.7 [C6], 56.1 [CH2CO], 57.3 [C2], 65.1 [CH2 (Z)], 126.8, 127.0, 127.7, 127.8, 128.3, 127.4, 128.6, 129.2 [20CH (Ar)], 135.8 [C (Ph)], 137.0 [C (NBn)], 137.2 [C (Z)], 139.2 [C (NHBn)], 156.1 [CO (Z)], 167.1 [C5], 169.4 [CO], 171.4 [α-CONH]. (S)-16a δ (ppm): 26.2 [Cγ], 29.3 [Cβ], 42.0 [CH2 (NHBn)], 43.4 [C3], 48.9 [CH2 (NBn)], 51.3 [C2-CH], 52.5 [Cα], 54.7 [C6], 56.1 [CH2CO], 58.6 [C2], 65.1 [CH2 (Z)], 126.9, 127.4, 127.7, 127.8, 128.3, 127.4, 128.6, 129.3 [20CH (Ar)], 135.9 [C (Ph)], 136.9 [C (NBn)], 137.2 [C (Z)], 139.2 [C (NHBn)], 156.1 [CO (Z)], 167.1 [C5], 169.4 [CO], 171.4 [α-CONH]; ES-MS m/z [M+1]+ calculated for C41H48N6O5: 706.3; found: 706.5.
N-[2-[4-Benzyl-(2RS)-[(1S)-amino-2-phenylethyl]-5-oxo-piperazin-1-yl]acetyl]-Lys(Z)-NHBn hydrochloride (16b). Amorphous solid (453 mg, 100%); HPLC tR: 16.93 min; 1H-NMR (500 MHz, DMSO-d6) (R)-16b δ (ppm): 1.22 (m, 1H, γ-H), 1.27 (m, 1H, γ-H), 1.41 (m, 2H, δ-H), 1.58 (m, 1H, β-H), 1.70 (m, 1H, β-H), 2.83 (dd, 1H, J = 8 and 14 Hz, CH2-Ph), 2.94 (m, 2H, ε-H), 2.96 (m, 2H, 2-H and CH2-Ph), 3.20 (d, 1H, J = 17 Hz, 6-H), 3.24 (d, 1H, J = 16.5 Hz, CH2CO), 3.29 (d, 1H, J = 16.5 Hz, CH2CO), 3.38 (m, 1H, 3-H), 3.46 (m, 1H, 3-H), 3.53 (d, 1H, J = 17 Hz, 6-H), 3.72 (m, 1H, 2-CH), 4.26 [m, 2H, CH2 (NHBn)], 4.28 (m, 1H, α-H), 4.45 [1d, 1H, J = 15 Hz, CH2 (NBn)], 4.63 [1d, 1H, J = 15 Hz, CH2 (NBn)], 4.98 [m, 2H, CH2 (Z)], 7.16–7.37 (m, 21H, Ar and NHZ), 8.19 (m, 3H, NH2·HCl), 8.21 (m, 1H, α-NH), 8.57 (t, 1H, J = 6 Hz, NHBn). (S)-16b δ (ppm): 1.22 (m, 1H, γ-H), 1.27 (m, 1H, γ-H), 1.41 (m, 2H, δ-H), 1.58 (m, 1H, β-H), 1.70 (m, 1H, β-H), 3.15 (m, 1H, 2-H), 3.38 (m, 1H, 3-H), 3.46 (m, 1H, 3-H), 3.55 (m, 1H, 2-CH), 4.26 [m, 2H, CH2 (NHBn)], 4.28 [m, 2H, α-H and CH2 (NBn)], 4.40 [d, 1H, J = 15 Hz, CH2 (NBn)], 4.98 [m, 2H, CH2 (Z)], 7.16–7.37 (m, 21H, Ar and NHZ), 8.19 (m, 3H, NH2·HCl), 8.43 (d, 1H, J = 8 Hz, α-NH), 8.57 (m, 1H, NHBn); 13C-NMR (125 MHz, DMSO-d6) (R)-16b δ (ppm): 23.2 [Cγ], 29.5 [Cδ], 32.0 [Cβ], 35.0 [CH2-Ph], 41.1 [Cε], 42.4 [CH2 (NHBn)], 44.0 [C3], 49.2 [CH2 (NBn)], 52.1 [C2-CH], 53.1 [Cα], 55.0 [C6], 56.6 [CH2CO], 57.7 [C2], 65.5 [CH2 (Z)], 127.1, 127.5, 128.1, 128.2, 128.7, 128.8, 129.0, 129.7 [20CH (Ar)], 136.2 [C (Ph)], 137.4 [C (NBn)], 137.7 [C (Z)], 139.8 [C (NHBn)], 156.5 [CO (Z)], 167.5 [C5], 169.8 [CO], 172.0 [α-CONH]. (S)-16b δ (ppm): 23.3 [Cγ], 31.9 [Cβ], 42.4 [CH2 (NHBn)], 43.8 [C3], 49.3 [CH2 (NBn)], 51.7 [C2-CH], 53.3 [Cα], 59.1 [C2], 65.5 [CH2 (Z)], 127.3, 127.5, 127.8, 128.2, 128.7, 129.0, 129.1, 129.7 [20CH (Ar)], 136.3 [C (Ph)], 137.3 [C (NBn)], 137.7 [C (Z)], 139.2 [C (NHBn)], 162.3 [CO (Z)], 172.0 [α-CONH]; ES-MS m/z [M+1]+ calculated for C42H50N6O5: 720.5; found: 720.8.
N-[2-[4-Benzyl-(2RS)-[(1S)-amino-2-phenylethyl]-5-oxo-piperazin-1-yl]acetyl]-Arg(Pbf)-NHBn hydrochloride (16c). Amorphous solid (541 mg, 100%); HPLC tR: 14.80 min [(R)-16c] and 19.68 min [(S)-16c]; 1H-NMR (500 MHz, DMSO-d6) (R)-16c δ (ppm): 1.38 [s, 6H, 2CH3 (Pbf)], 1.44 (m, 2H, γ-H), 1.56 (m, 1H, β-H), 1.70 (m, 1H, β-H), 1.98 [s, 3H, CH3 (Pbf)], 2.40 [s, 3H, CH3 (Pbf)], 2.46 [s, 3H, CH3 (Pbf)], 2.83 (d, 1H, J = 6.5 and 14 Hz, CH2-Ph), 2.94 (m, 1H, CH2-Ph), 2.95 (m, 1H, 2-H), 2.96 [m, 2H, CH2 (Pbf)], 3.02 (dd, 2H, J = 6.5 and 12 Hz, δ-H), 3.19 (d, 1H, J = 16.5 Hz, 6-H), 3.29 (m, 2H, CH2CO), 3.39 (m, 1H, 3-H), 3.44 (m, 1H, 3-H), 3.55 (d, 1H, J = 16.5 Hz, 6-H), 3.65 (m, 1H, 2-CH), 4.23 [m, 2H, CH2 (NHBn)], 4.30 (m, 1H, α-H), 4.45 [d, 1H, J = 15 Hz, CH2 (NBn)], 4.63 [d, 1H, J = 15 Hz, CH2 (NBn)], 6.45 [m, 3H, NHC(NH2) = N], 6.91–7.37 (m, 15H, Ar), 8.18 (m, 3H, NH2·HCl), 8.23 (d, 1H, J = 8 Hz, α-NH), 8.59 (t, 1H, J = 6Hz, NHBn). (S)-16c δ (ppm): 1.38 [s, 6H, 2CH3 (Pbf)], 1.98 [s, 3H, CH3 (Pbf)], 2.40 [s, 3H, CH3 (Pbf)], 2.46 [s, 3H, CH3 (Pbf)], 2.96 [m, 2H, CH2 (Pbf)], 3.15 (m, 1H, 2-H), 3.39 (m, 1H, 3-H), 3.44 (m, 1H, 3-H), 3.55 (m, 1H, 2-CH), 4.23 [m, 2H, CH2 (NHBn)], 4.30 (m, 2H, α-H and CH2 (NBn)), 4.40 [d, 2H, J = 15 Hz, CH2 (NBn)], 6.45 [m, 3H, NHC(NH2) = N], 6.91–7.37 (m, 15H, Ar), 8.18 (m, 3H, NH2·HCl), 8.46 (d, 1H, J = 8 Hz, α-NH), 8.56 (m, 1H, NHBn); 13C-NMR (125 MHz, DMSO-d6) (R)-16c δ (ppm): 12.7, 18.1, 19.4 [3CH3 (Pbf)], 26.1 [Cγ], 28.2 [2CH3 (Pbf)], 29.9 [Cβ], 35.0 [CH2-Ph], 40.3 [Cδ], 42.4 [CH2 (NHBn)], 42.9 [CH2 (Pbf)], 44.0 [C3], 49.2 [CH2 (NBn)], 52.1 [C2-CH], 52.8 [Cα], 55.0 [C6], 56.7 [CH2CO], 57.8 [C2], 86.8, 116.8, 124.8 [4C (Pbf)], 127.1, 127.5, 127.8, 128.2, 128.7, 129.0, 129.7 [15CH (Ar)], 131.9, 134.5 [2C (Pbf)], 136.2 [C (Ph)], 137.7 [C (NBn)], 137.8 [C (Pbf)], 139.7 [C (NHBn)], 156.5 [C (NHC(NH2) = N)], 158.0 [C (Pbf)], 167.6 [C5], 169.9 [CO], 171.8 [α-CONH]. (S)-16c δ (ppm): 12.7, 18.1, 19.4 [3CH3 (Pbf)], 28.2 [2CH3 (Pbf)], 42.4 [CH2 (NHBn)], 42.9 [CH2 (Pbf)], 43.9 [C3], 49.1 [CH2 (NBn)], 51.8 [C2-CH], 53.0 [Cα], 59.1 [C2], 86.8, 116.8, 124.8 [4C (Pbf)], 127.3, 127.5, 127.8, 128.2, 128.7, 129.0, 129.1, 129.7 [15CH (Ar)], 131.9, 134.5 [2C (Pbf)], 136.3 [C (Ph)], 137.4 [C (NBn)], 137.8 [C (Pbf)], 139.7 [C (NHBn)], 156.5 [C (NHC(NH2) = N)], 158.0 [C (Pbf)], 167.5 [C5], 171.8 [α-CONH]; ES-MS m/z [M+1]+ calculated for C47H60N8O6S: 866.6; found: 866.0.
3.11. General Procedure for the Synthesis of the Piperazinone-Derived Ureas 17a–c and 18a,b
These compounds were obtained by applying the already indicated procedure for the synthesis of the urea analogues 9a,b and 10a,b.
N-[2-[4-Benzyl-5-oxo-(2RS)-[2-phenyl-(1S)-(3-phenyl-ureido)ethyl]-piperazin-1-yl]acetyl]-Orn(Z)-NHBn (17a). Amorphous solid (R:S) = (3:1)] (346 mg, 70%); HPLC tR: 23.73 min [(R)-17a] and 24.44 min [(S)-17a]; 1H-NMR (500 MHz, CDCl3) (R)-17a δ (ppm): 1.50 (m, 2H, γ-H), 1.70 (m, 1H, β-H), 1.80 (m, 1H, β-H), 2.69 (dd, 1H, J = 6 and 14 Hz, CH2-Ph), 2.88 (m, 1H, CH2-Ph), 2.94 (m, 1H, 2-H), 3.10 (m, 1H, δ-H), 3.20 (m, 2H, 3-H and 6-H), 3.35 (m, 1H, CH2CO), 3.38 (m, 1H, 3-H), 3.40 (m, 1H, CH2CO), 3.42 (m, 1H, δ-H), 3.49 (d, 1H, J = 17 Hz, 6-H), 4.07 (m, 1H, 2-CH), 4.18 [m, 1H, CH2 (NHBn)], 4.32 [m, 1H, CH2 (NBn)], 4.40 [m, 1H, CH2 (NHBn)], 4.78 [m, 2H, α-H and CH2 (Z)], 4.82 [m, 1H, CH2 (NBn)], 4.87 [m, 1H, CH2 (Z)], 5.09 (t, 1H, J = 6 Hz, NHZ), 5.57 (m, 1H, 2-CHNH), 6.84–7.30 (m, 26H, Ar and NHPh), 7.34 (m, 1H, NHBn), 7.79 (m, 1H, α-NH). (S)-17a δ (ppm): 1.40 (m, 2H, γ-H), 1.70 (m, 1H, β-H), 1.80 (m, 1H, β-H), 2.81 (m, 1H, CH2-Ph), 2.94 (m, 1H, CH2-Ph), 3.05 (m, 1H, δ-H), 3.08 (m, 1H, 3-H), 3.35 (m, 1H, CH2CO), 3.39 (m, 1H, 3-H), 3.40 (m, 1H, CH2CO), 3.43 (m, 1H, δ-H), 4.07 (m, 1H, 2-CH), 4.16 [dd, 1H, J = 5 and 15 Hz, CH2 (NHBn)], 4.38 [m, 1H, CH2 (NHBn)], 4.50 [m, 2H, CH2 (NBn)], 4.72 (m, 1H, α-H), 4.76 [m, 1H, CH2 (Z)], 4.85 [m, 1H, CH2 (Z)], 4.96 (m, 1H, NHZ), 5.39 (d, 1H, J = 6.5 Hz, 5-CHNH), 6.84–7.30 (m, 26H, Ar and NHPh), 7.41 (m, 1H, NHBn), 7.73 (m, 1H, α-NH); 13C-NMR (125 MHz, CDCl3) (R)-17a δ (ppm): 26.6 [Cγ], 30.1 [Cβ], 36.9 [CH2-Ph], 39.1 [Cδ], 42.0 [CH2 (NHBn)], 44.8 [C3], 49.5 [CH2 (NBn)], 51.2 [Cα], 52.3 [C2-CH], 55.8 [C6], 59.0 [CH2CO], 61.2 [C2], 66.7 [CH2 (Z)], 120.0, 123.1, 126.7, 127.5, 127.8, 127.9, 128.1, 128.3, 128.5, 128.8 [25CH (Ar)], 136.1 [C (NBn)], 136.2 [C (Z)], 137.2 [C (Ph)], 137.6 [C (NHBn)], 138.7 [C (NHPh)], 155.1 [CO (Z)], 157.5 [CO (Urea)], 167.9 [C5], 170.4 [CO], 172.7 [α-CONH]. (S)-17a δ (ppm): 26.5 [Cγ], 30.6 [Cβ], 37.6 [CH2-Ph], 38.8 [Cδ], 42.1 [CH2 (NHBn)], 44.8 [C3], 49.9 [CH2 (NBn)], 50.6 [Cα], 51.9 [C2-CH], 66.7 [CH2 (Z)], 118.9, 122.3, 126.7, 127.5, 127.8, 127.9, 128.2, 128.5, 128.6, 128.7, 128.9, 129.4 [25CH (Ar)], 136.1 [C (NBn)], 136.2 [C (Z)], 137.2 [C (Ph)], 137.4 [C (NHBn)], 139.5 [C (NHPh)], 155.0 [CO (Z)], 157.6 [CO (Urea)], 166.9 [C5], 169.7 [CO], 172.9 [α-CONH]; ES-MS m/z 825.7 [M+1]+; C48H53N7O6 (%): C: 69.97, H: 6.48, N: 11.90. Found (%): C: 69.75, H: 6.65, N: 12.02.
N-[2-[4-Benzyl-5-oxo-(2RS)-[2-phenyl-(1S)-(3-phenyl-ureido)ethyl]-piperazin-1-yl]acetyl]-Lys(Z)-NHBn (17b). Amorphous solid [(R:S) = (3:1)] (327 mg, 65%); HPLC tR: 24.06 min; 1H-NMR (500 MHz, CDCl3) (R)-17b δ (ppm): 1.33 (m, 2H, γ-H), 1.46 (m, 2H, δ-H), 1.67 (m, 1H, β-H), 1.85 (m, 1H, β-H), 2.68 (m, 1H, CH2-Ph), 2.82 (m, 1H, CH2-Ph), 2.89 (m, 1H, 2-H), 3.05 (m, 2H, ε-H), 3.15 (m, 1H, 6-H), 3.25 (m, 2H, 3-H), 3.31 (m, 2H, CH2CO), 3.53 (d, 1H, J = 16.5 Hz, 6-H), 4.20 (m, 1H, 2-CH), 4.25 [m, 3H, CH2 (NHBn and NBn)], 4.44 (m, 1H, α-H), 4.77 [d, 1H, J = 14.5 Hz, CH2 (NBn], 5.00 [s, 2H, CH2 (Z)], 5.23 (m, 1H, NHZ), 5.45 (m, 1H, 2-CHNH), 6.84–7.52 (m, 27H, Ar, NHBn and NHPh), 7.79 (m, 1H, α-NH). (S)-17b δ (ppm): 2.78 (m, 1H, CH2-Ph), 2.89 (m, 1H, CH2-Ph), 2.94 (m, 1H, 2-H), 4.20 (m, 1H, 2-CH), 4.25 [m, 2H, CH2 (NHBn)], 4.52 [d, 1H, J = 14.5 Hz, CH2 (NBn], 4.58 [d, 1H, J = 14.5 Hz, CH2 (NBn], 5.03 [m, 2H, CH2 (Z)], 5.09 (m, 1H, NHZ), 5.55 (m, 1H, 2-CHNH), 6.84–7.52 (m, 27H, Ar, NHBn and NHPh), 7.89 (m, 1H, α-NH); 13C-NMR (125 MHz, CDCl3) (R)-17b δ (ppm): 22.7 [Cγ], 29.1 [Cδ], 31.9 [Cβ], 37.2 [CH2-Ph], 40.3 [Cε], 43.6 [CH2 (NHBn)], 44.6 [C3], 49.5 [CH2 (NBn)], 52.0 [C2-CH], 53.0 [Cα], 55.6 [C6], 58.8 [CH2CO], 60.9 [C2], 66.0 [CH2 (Z)], 120.1, 123.2, 126.8, 127.5, 128.0, 128.1, 128.4, 128.5, 128.7, 128.8, 128.9 [25CH (Ar)], 136.2 [C (NBn)], 136.5 [C (Z)], 137.0 [C (Ph)], 137.7 [C (NHBn)], 138.6 [C (NHPh)], 155.3 [CO (Z)], 156.6 [CO (Urea)], 167.6 [C5], 170.1 [CO], 172.3 [α-CONH]. (S)-17b δ (ppm): 43.6 [CH2 (NHBn)], 44.6 [C3], 52.1 [C2-CH], 60.8 [C2], 66.0 [CH2 (Z)], 120.1, 123.2, 126.8, 127.5, 128.0, 128.1, 128.4, 128.5, 128.7, 128.8, 129.0 [25CH (Ar)], 136.5 [C (Z)], 156.4 [CO (Z)], 172.3 [α-CONH]; ES-MS m/z 839.7 [M+1]+; C49H55N7O6 (%): C: 70.23, H: 6.62, N: 11.70. Found (%): C: 70.46, H: 6.75, N: 11.54.
N-[2-[4-Benzyl-5-oxo-(2RS)-[2-phenyl-(1S)-(3-phenyl-ureido)ethyl]-piperazin-1-yl]acetyl]-Arg(Pbf)-NHBn (17c). Amorphous solid [(R:S) = (3:1)] (443 mg, 75%); HPLC tR: 25.80 min [(R)-17c] and 23.82 min [(S)-17c]; 1H-NMR (500 MHz, CDCl3) (R)-17c δ (ppm): 1.45 [s, 3H, CH3 (Pbf)], 1.46 [s, 3H, CH3 (Pbf)], 1.40 (m, 2H, γ-H), 1.52 (m, 1H, β-H), 1.68 (m, 1H, β-H), 2.10 [s, 3H, CH3 (Pbf)], 2.50 [s, 3H, CH3 (Pbf)], 2.58 [s, 3H, CH3 (Pbf)], 2.64 (m, 1H, 2-H), 2.68 (m, 1H, CH2-Ph), 2.77 (m, 1H, CH2-Ph), 2.94 [s, 2H, CH2 (Pbf)], 2.98 (m, 1H, CH2CO), 3.06 (m, 1H, 6-H), 3.25 (m, 1H, δ-H), 3.28 (dd, 1H, J = 5 and 13 Hz, 3-H), 3.29 (m, 1H, δ-H), 3.50 (d, 1H, J = 15.5 Hz, CH2CO), 3.58 (m, 1H, 3-H), 3.64 (d, 1H, J = 16.5 Hz, 6-H), 4.13 [d, 1H, J = 14.5 Hz, CH2 (NBn)], 4.30 (m, 1H, 2-CH), 4.32 (m, 1H, α-H), 4.36 [m, 2H, CH2 (NHBn)], 5.00 [d, 1H, J = 14.5 Hz, CH2 (NBn)], 5.98 (m, 1H, 2-CHNH), 6.18 [m, 2H, NHC(NH2) = N], 6.36 [m, 1H, NHC(NH2) = N], 6.86–7.37 (m, 21H, Ar and NHPh), 7.64 (m, 1H, NHBn), 7.88 (d, 1H, J = 8 Hz, α-NH). (S)-17c δ (ppm): 1.45 [s, 3H, CH3 (Pbf)], 1.46 [s, 3H, CH3 (Pbf)], 2.10 [s, 3H, CH3 (Pbf)], 2.50 [s, 3H, CH3 (Pbf)], 2.58 [s, 3H, CH3 (Pbf)], 2.68 (m, 1H, CH2-Ph), 2.77 (m, 1H, CH2-Ph), 2.94 [s, 2H, CH2 (Pbf)], 3.23 (m, 1H, δ-H), 3.29 (m, 1H, δ-H), 3.28 (m, 1H, 3-H), 3.58 (m, 1H, 3-H), 4.05 [m, 1H, CH2 (NHBn)], 4.29 (m, 1H, α-H), 4.30 [m, 1H, CH2 (NHBn)], 4.46 [d, 1H, J = 14 Hz, CH2 (NBn)], 4.59 [d, 1H, J = 14 Hz, CH2 (NBn)], 5.98 (m, 1H, 2-CHNH), 6.18 [m, 2H, NHC(NH2) = N], 6.36 [m, 1H, NHC(NH2) = N], 6.86–7.37 (m, 21H, Ar and NHPh), 7.64 (m, 1H, NHBn), 7.78 (d, 1H, J = 8 Hz, α-NH); 13C-NMR (125 MHz, CDCl3) (R)-17c δ (ppm): 12.5, 18.0, 19.4 [3CH3 (Pbf)], 25.4 [Cγ], 28.6 [2CH3 (Pbf)], 29.3 [Cβ], 38.1 [CH2-Ph], 40.3 [Cδ], 43.2 [2CH2 (Pbf and NHBn)], 44.3 [C3], 49.2 [CH2 (NBn)], 51.2 [C2-CH], 53.1 [Cα], 55.4 [C6], 59.5 [CH2CO], 60.2 [C2], 86.6, 117.8, 124.9 [3C (Pbf)], 119.5, 122.7, 126.7, 127.1, 127.3, 127.9, 128.1, 128.5, 128.6, 129.0, 129.3 [20CH (Ar)], 132.2 [2C (Pbf)], 136.2 [C (NBn)], 137.2 [C (Ph)], 138.1 [C (NHBn)], 138.3 [C (Pbf)], 139.0 [C (NHPh)], 156.2 [CO (Urea)], 156.4 [C (NHC(NH2) = N)], 159.0 [C (Pbf)], 168.3 [C5], 171.0 [CO], 172.0 [α-CONH]. (S)-17c δ (ppm): 12.5, 18.0, 19.4 [3CH3 (Pbf)], 28.6 [2CH3 (Pbf)], 38.4 [CH2-Ph], 40.3 [Cδ], 43.2 [2CH2 (Pbf and NHBn)], 44.2 [C3], 49.2 [CH2 (NBn)], 53.1 [Cα], 86.6, 117.8, 124.9 [3C (Pbf)], 118.9, 122.2, 126.6, 127.1, 127.2, 127.9, 128.1, 128.4, 128.7, 129.0, 129.3 [20CH (Ar)], 132.2 [2C (Pbf)], 136.1 [C (NBn)], 137.1 [C (Ph)], 138.0 [C (NHBn)], 138.3 [C (Pbf)], 139.4 [C (NHPh)], 156.4 [C (NHC(NH2) = N)], 159.0 [C (Pbf)], 172.0 [α-CONH]; ES-MS m/z 985.1 [M+1]+; C54H65N9O7S (%): C: 65.90, H: 6.66, N: 12.81. Found (%): C: 65.72, H: 6.90, N: 12.63.
N-[2-[4-Benzyl-(2RS)-[(1S)-(3-benzylureido)-2-phenyl-ethyl]-5-oxopiperazin-1-yl]acetyl]-Orn(Z)-NHBn (18a). Amorphous solid [(R:S) = (3:1)] (375 mg, 65%); HPLC tR: 23.30 min [(R)-18a] and 23.82 min [(S)-18a]; 1H-NMR (500 MHz, CDCl3) (R)-18a δ (ppm): 1.52 (m, 2H, γ-H), 1.65 (m, 1H, β-H), 1.82 (m, 1H, β-H), 2.69 (m, 1H, CH2-Ph), 2.88 (m, 1H, CH2-Ph), 2.90 (m, 1H, 2-H), 3.11 (m, 1H, δ-H), 3.20 (m, 1H, CH2CO), 3.23 (m, 1H, 3-H), 3.32 (m, 2H, CH2CO and 6-H), 3.36 (m, 1H, 3-H), 3.42 (m, 1H, δ-H), 3.55 (m, 1H, 6-H), 4.05 [m, 1H, CH2 (NHBn)], 4.08 [m, 1H, CH2 (NHBn, Urea)], 4.15 [m, 1H, CH2 (NHBn, Urea)], 4.18 (m, 1H, 2-CH), 4.25 [m, 1H, CH2 (NHBn)], 4.32 [m, 1H, CH2 (NBn)], 4.68 (m, 1H, α-H), 4.75 [m, 1H, CH2 (Z)], 4.79 [m, 1H, CH2 (NBn)], 4.88 [d, 1H, J = 12.5 Hz, CH2 (Z)], 5.08 (m, 1H, 2-CHNH), 5.20 (m, 1H, NHZ), 5.95 [m, 1H, NHBn (Urea)], 6.95–7.40 (m, 25H, Ar), 7.40 (m, 1H, NHBn), 7.86 (d, 1H, J = 9 Hz, α-NH). (S)-18a δ (ppm): 1.46 (m, 2H, γ-H), 1.63 (m, 1H, β-H), 1.80 (m, 1H, β-H), 2.57 (m, 1H, CH2-Ph), 2.85 (m, 1H, CH2-Ph), 3.04 (m, 1H, 3-H), 3.07 (m, 1H, δ-H), 3.34 (m, 1H, 3-H), 3.38 (m, 1H, 6-H), 3.44 (m, 1H, δ-H), 3.57 (m, 1H, 6-H), 3.96 [dd, 1H, J = 5 and 15 Hz, CH2 (NHBn)], 4.02 (m, 1H, 2-CH), 4.08 [m, 1H, CH2 (NHBn, Urea)], 4.15 [m, 1H, CH2 (NHBn, Urea)], 4.20 [m, 1H, CH2 (NHBn)], 4.50 [m, 1H, CH2 (NBn)], 4.66 [m, 1H, CH2 (Z)], 4.68 (m, 1H, α-H), 4.79 [m, 1H, CH2 (NBn)], 4.82 [m, 1H, CH2 (Z)], 5.02 (t, 1H, J = 6 Hz, NHZ), 5.08 (m, 1H, 5-CHNH), 5.95 [m, 1H, NHBn (Urea)], 6.95–7.40 (m, 25H, Ar), 7.40 (m, 1H, NHBn), 7.78 (d, 1H, J = 8.5 Hz, α-NH); 13C-NMR (125 MHz, CDCl3) (R)-18a δ (ppm): 26.6 [Cγ], 30.2 [Cβ], 37.3 [CH2-Ph], 39.0 [Cδ], 43.5 [CH2 (NHBn)], 44.0 [CH2 (NHBn, Urea)], 44.6 [C3], 49.5 [CH2 (NBn)], 51.1 [Cα], 52.2 [C2-CH], 55.6 [C6 and CH2CO], 60.8 [C2], 66.7 [CH2 (Z)], 126.6, 127.0, 127.4, 127.6, 127.9, 128.1, 128.4, 128.5, 128.7, 128.9 [25CH (Ar)], 136.3 [C (NBn) and C (Z)], 137.2 [C (Ph)], 137.7 [C (NHBn)], 139.3 [C (NHBn, Urea)], 155.4 [CO (Z)], 157.7 [CO (Urea)], 167.6 [C5], 170.0 [CO], 172.4 [α-CONH]. (S)-18a δ (ppm): 26.6 [Cγ], 30.7 [Cβ], 38.2 [CH2-Ph], 38.7 [Cδ], 43.5 [CH2 (NHBn)], 43.8 [CH2 (NHBn, Urea)], 45.0 [C3], 49.9 [CH2 (NBn)], 51.4 [Cα], 52.2 [C2-CH], 55.6 [CH2CO], 66.7 [CH2 (Z)], 126.9, 127.1, 127.4, 127.5, 127.8, 127.9, 128.2, 128.5, 128.6, 128.9, 129.6 [25CH (Ar)], 136.1 [C (NBn) and C (Z)], 137.1 [C (Ph)], 137.7 [C (NHBn)], 139.8 [C (NHBn, Urea)], 155.2 [CO (Z)], 157.9 [CO (Urea)], 167.6 [C5], 170.0 [CO], 172.6 [α-CONH]; ES-MS m/z 839.6 [M+1]+; C49H55N7O6 (%): C: 70.23, H: 6.22, N: 11.70. Found (%): C: 70.01, H: 6.46, N: 11.59.
N-[2-[4-Benzyl-(2RS)-[(1S)-(3-benzylureido)-2-phenyl-ethyl]-5-oxopiperazin-1-yl]acetyl]-Lys(Z)-NHBn (18b). Amorphous solid [(R:S) = (3:1)] (317 mg, 62%); HPLC tR: 23.69 min [(R)-18b] and 24.16 min [(S)-19b]; 1H-NMR (500 MHz, CDCl3) (R)-18b δ (ppm): 1.27 (m, 2H, γ-H), 1.40 (m, 2H, δ-H), 1.60 (m, 1H, β-H), 1.78(m, 1H, β-H), 2.64 (m, 2H, CH2-Ph), 2.76 (m, 1H, 2-H), 3.08 (m, 1H, ε-H), 3.12 (m, 2H, CH2CO and 6-H), 3.14 (m, 1H, 3-H), 3.15 (m, 1H, ε-H), 3.24 (m, 1H, CH2CO), 3.25 (m, 1H, 3-H), 3.41 (d, 1H, J = 16.5 Hz, 6-H), 4.10 (m, 1H, 2-CH), 3.95 [dd, 1H, J = 5.5 and 15, CH2 (NHBn, Urea)], 4.04 [m, 1H, CH2 (NHBn, Urea)], 4.18 [m, 2H, CH2 (NHBn)], 4.32 [m, 1H, CH2 (NBn], 4.38 (m, 1H, α-H), 4.62 [d, 1H, J = 14.5 Hz, CH2 (NBn], 4.92 (m, 1H, 2-CHNH), 5.00 [s, 2H, CH2 (Z)], 5.20 (m, 1H, NHZ), 5.70 [m, 1H, NHBn (Urea)], 6.81–7.33 (m, 26H, Ar and NHBn), 7.71 (d, 1H, J = 8 Hz, α-NH). (S)-18b δ (ppm): 1.27 (m, 2H, γ-H), 1.40 (m, 2H, δ-H), 1.60 (m, 1H, β-H), 1.78(m, 1H, β-H), 2.68 (m, 2H, CH2-Ph), 2.76 (m, 1H, 2-H), 2.98 (m, 1H, 3-H), 3.10 (m, 1H, CH2CO), 3.18 (m, 1H, 3-H), 3.22 (m, 1H, CH2CO), 3.76 (m, 1H, 2-CH), 3.95 [m, 1H, CH2 (NHBn, Urea)], 4.04 [m, 1H, CH2 (NHBn, Urea)], 4.10 [m, 1H, CH2 (NHBn)], 4.18 [m, 1H, CH2 (NHBn)], 4.34 (m, 1H, α-H), 4.43 [d, 1H, J = 14.5 Hz, CH2 (NBn], 4.52 [d, 1H, J = 14.5 Hz, CH2 (NBn], 4.92 (m, 1H, 2-CHNH), 4.96 [s, 2H, CH2 (Z)], 5.10 (m, 1H, NHZ), 5.70 [m, 1H, NHBn (Urea)], 6.81–7.33 (m, 26H, Ar and NHBn), 7.64 (d, 1H, J = 7.5 Hz, α-NH); 13C-NMR (125 MHz, CDCl3) (R)-18b δ (ppm): 22.7 [Cγ], 29.2 [Cδ], 31.9 [Cβ], 37.5 [CH2-Ph], 40.4 [Cε], 43.4 [CH2 (NHBn)], 44.0 [CH2 (NHBn, Urea)], 44.3 [C3], 49.5 [CH2 (NBn)], 52.0 [C2-CH], 53.0 [Cα], 55.3 [C6], 58.6 [CH2CO], 60.1 [C2], 66.6 [CH2 (Z)], 126.7, 127.0, 127.4, 127.5, 128.0, 128.2, 128.4, 128.5, 128.6, 128.7, 128.9 [25CH (Ar)], 136.4 [C (NBn)], 136.6 [C (Z)], 137.1 [C (Ph)], 137.8 [C (NHBn)], 139.1 [C (NHBn, Urea)], 156.6 [CO (Z)], 157.7 [CO (Urea)], 167.8 [C5], 170.1 [CO], 172.1 [α-CONH]. (S)-18b δ (ppm): 22.4 [Cγ], 29.7 [Cδ], 30.9 [Cβ], 38.2 [CH2-Ph], 43.5 [CH2 (NHBn)], 43.9 [CH2 (NHBn, Urea)], 44.3 [C3], 49.7 [CH2 (NBn)], 51.2 [C2-CH], 52.8 [Cα], 58.6 [CH2CO], 60.0 [C2], 66.7 [CH2 (Z)], 126.7, 127.2, 127.4, 127.5, 128.0, 128.1, 128.4, 128.5, 128.6, 128.7, 128.9, 129.5 [25CH (Ar)], 136.1 [C (NBn)], 136.6 [C (Z)], 137.1 [C (Ph)], 137.8 [C (NHBn)], 139.6 [C (NHBn, Urea)], 156.7 [CO (Z)], 158.0 [CO (Urea)], 170.1 [CO], 172.1 [α-CONH]; ES-MS m/z 853.7 [M+1]+; C50H57N7O6 (%): C: 70.48, H: 6.74, N: 11.51. Found (%): C: 70.31, H: 6.95, N: 11.69.
3.12. General Procedure for the Synthesis of the Hydrochlorides 19a,b and 20a,b
These compounds were prepared following the general procedure for the removal of the N-Z protecting group, already indicated for the synthesis of 11a,b and 12a,b.
N-[2-[4-Benzyl-5-oxo-(2RS)-[2-phenyl-(1S)-(3-phenylureido)ethyl]-piperazin-1-yl]acetyl]-Orn-NHBn hydrochloride (19a). Amorphous solid [(R:S) = (3:1)] (145 mg, 100%); HPLC tR: 16.27 min [(R)-19a] and 16.52 min [(S)-19a]; 1H-NMR (500 MHz, DMSO-d6) (R)-19a δ (ppm): 1.62 (m, 3H, γ-H and β-H), 1.82 (m, 1H, β-H), 2.70 (m, 1H, CH2-Ph), 2.75 (m, 3H, δ-H and 2-H), 2.91 (d, 1H, J = 11 Hz, CH2-Ph), 3.33–4.11 (m, 6H, 3-H, 6-H and CH2CO), 4.22 [m, 1H, CH2 (NHBn)], 4.34 [m, 1H, CH2 (NHBn)], 4.38 (m, 1H, 2-CH), 4.39 (m, 1H, α-H), 4.48 [m, 1H, CH2 (NBn)], 4.62 [m, 1H, CH2 (NBn)], 6.80 (m, 1H, 2-CHNH), 6.76–6.95 (m, 2H, Ar), 7.08–7.38 (m, 18H, Ar), 7.96 (m, 3H, NH2·HCl), 8.60 (m, 1H, α-NH), 8.72 (m, 1H, NHBn), 8.85 (m, 1H, NHPh). (S)-19a δ (ppm): 1.58 (m, 3H, γ-H and β-H), 1.78 (m, 1H, β-H), 2.55 (dd, 1H, J = 6 and 14 Hz, CH2-Ph), 2.75 (m, 3H, δ-H and 2-H), 2.85 (m, 1H, CH2-Ph), 3.33–4.11 (m, 7H, 3-H, 6-H, CH2CO and 2-CH), 4.22 [m, 1H, CH2 (NHBn)], 4.34 [m, 1H, CH2 (NHBn)], 4.51 [m, 1H, CH2 (NBn)], 4.64 [m, 1H, CH2 (NBn)], 6.80 (m, 1H, 2-CHNH), 6.76–6.95 (m, 2H, Ar), 7.08–7.38 (m, 18H, Ar), 7.96 (m, 3H, NH2·HCl), 8.60 (m, 1H, α-NH), 8.70 (m, 1H, NHBn), 8.81 (m, 1H, NHPh); 13C-NMR (125 MHz, DMSO-d6) (R)-19a δ (ppm): 23.4 [Cγ], 28.9 [Cβ], 37.8 [CH2-Ph], 38.1 [Cδ], 42.0 [CH2 (NHBn)], 43.7 [C3], 49.2 [CH2 (NBn)], 49.8 [C2-CH], 51.9 [Cα], 53.6 [C6], 53.9 [CH2CO], 60.5 [C2], 117.7, 121.2, 126.2, 126.6, 127.0, 127.2, 127.5, 128.2, 128.4, 128.5, 129.1 [20CH (Ar)], 136.3 [C (NBn)], 137.8 [C (Ph)], 139.1 [C (NHBn)], 139.9 [C (NHPh)], 155.2 [CO (Urea)], 170.7 [α-CONH]. (S)-19a δ (ppm): 23.3 [Cγ], 29.0 [Cβ], 37.8 [CH2-Ph], 38.2 [Cδ], 42.0 [CH2 (NHBn)], 43.7 [C3], 49.2 [CH2 (NBn)], 49.8 [C2-CH], 53.5 [C6], 53.9 [CH2CO], 60.2 [C2], 117.5, 121.0, 126.1, 126.6, 127.0, 127.3, 127.7, 127.9, 128.1, 128.4, 128.5, 129.1 [20CH (Ar)], 136.3 [C (NBn)], 138.0 [C (Ph)], 139.1 [C (NHBn)], 140.1 [C (NHPh)], 155.0 [CO (Urea)], 170.8 [α-CONH]; ES-MS m/z [M+2]+ calculated for C40H47N7O4: 690.3; found: 690.6.
N-[2-[4-Benzyl-5-oxo-(2RS)-[2-phenyl-(1S)-(3-phenylureido)ethyl]-piperazin-1-yl}acetyl}-Lys-NHBn hydrochloride (19b). Amorphous solid [(R:S) = (3:1)] (148 mg, 100%); HPLC tR: 16.44 min; 1H-NMR (500 MHz, DMSO-d6) (R)-19b δ (ppm): 1.30 (m, 2H, γ-H), 1.50 (m, 2H, δ-H), 1.60 (m, 1H, β-H), 1.72 (m, 1H, β-H), 2.70 (m, 3H, ε-H and 2-H), 2.72 (m, 1H, CH2-Ph), 2.92 (m, 1H, CH2-Ph), 3.26–4.20 (m, 6H, 3-H, 6-H and CH2CO), 4.32 (m, 1H, 2-CH), 4.24 [dd, 1H, J = 6 and 15 Hz, CH2 (NHBn)], 4.30 [m, 2H, α-H and CH2 (NHBn)], 4.50 [m, 1H, CH2 (NBn)], 4.70 [m, 1H, CH2 (NBn)], 6.55 (m, 1H, 2-CHNH), 6.86 (t, 1H, J = 7 Hz, Ar), 6.97–7.41 (m, 19H, Ar), 7.84 (m, 3H, NH2·HCl), 8.51 (m, 1H, α-NH), 8.62 (m, 1H, NHBn), 8.80 (m, 1H, NHPh). (S)-19b δ (ppm): 1.60 (m, 1H, β-H), 1.72 (m, 1H, β-H), 2.55 (m, 1H, CH2-Ph), 2.88 (m, 1H, CH2-Ph), 4.24 [m, 1H, CH2 (NHBn)], 4.30 [m, 1H, CH2 (NHBn)], 6.55 (m, 1H, 2-CHNH), 6.86 (t, 1H, J = 7 Hz, Ar), 6.97–7.41 (m, 19H, Ar), 7.84 (m, 3H, NH2·HCl), 8.58 (m, 1H, NHBn), 8.83 (m, 1H, NHPh); 13C-NMR (125 MHz, DMSO-d6) (R)-19b δ (ppm): 22.2 [Cγ], 26.5 [Cδ], 31.3 [Cβ], 37.8 [CH2-Ph], 38.4 [Cε], 42.0 [CH2 (NHBn)], 43.8 [C3], 49.2 [CH2 (NBn) and C2-CH], 52.5 [Cα], 53.7 [C6], 60.4 [C2], 117.8, 121.3, 126.7, 127.0, 127.3, 127.6, 128.2, 128.5, 128.6, 129.2 [20CH (Ar)], 136.5 [C (NBn)], 137.9 [C (Ph)], 139.2 [C (NHBn)], 139.9 [C (NHPh)], 155.3 [CO (Urea)], 171.1 [α-CONH]. (S)-19b δ (ppm): 31.5 [Cβ], 37.8 [CH2-Ph], 42.0 [CH2 (NHBn)], 117.8, 121.3, 126.3, 127.0, 127.3, 127.6, 128.2, 128.5, 128.6, 129.2 [20CH (Ar)], 136.5 [C (NBn)], 137.9 [C (Ph)], 139.2 [C (NHBn)], 139.9 [C (NHPh)], 155.5 [CO (Urea)], 171.1 [α-CONH]; ES-MS m/z [M+2]+ calculated for C41H49N7O4: 705.3; found: 705.6.
N-[2-[4-Benzyl-(2RS)-[(1S)-(3-benzylureido)-2-phenylethyl]-5-oxopiperazin-1-yl]acetyl]-Orn-NHBn hydrochloride (20a). Amorphous solid [(R:S) = (3:1)] (148 mg, 100%); HPLC tR: 16.32 min [(R)-20a] and 16.78 min [(S)-20a]; 1H-NMR (500 MHz, DMSO-d6) (R)-20a δ (ppm): 1.60 (m, 2H, γ-H), 1.65 (m, 1H, β-H), 1.75 (m, 1H, β-H), 2.65 (dd, 1H, J = 10 and 14 Hz, CH2-Ph), 2.74 (m, 1H, 2-H), 2.78 (m, 1H, δ-H), 2.93 (m, 1H, CH2-Ph), 3.35–3.82 (m, 6H, 3-H, 6-H and CH2CO), 4.03 [d, 1H, J = 15 Hz, CH2 (NHBn, Urea)], 4.15 [d, 1H, J = 15 Hz, CH2 (NHBn, Urea)], 4.25 [m, 1H, CH2 (NHBn)], 4.30 [m, 1H, CH2 (NHBn)], 4.36 (m, 1H, 2-CH), 4.38 (m, 1H, α-H), 4.47 [d, 1H, J = 15 Hz, CH2 (NBn)], 4.62 [d, 1H, J = 15 Hz, CH2 (NBn)], 6.49 (m, 1H, 2-CHNH), 6.90–7.40 [m, 21H, Ar and NHBn (Urea)], 7.92 (m, 3H, NH2·HCl), 8.56 (m, 1H, α-NH), 8.72 (t, 1H, J = 6 Hz, NHBn). (S)-20a δ (ppm): 1.58 (m, 1H, β-H), 1.72 (m, 1H, β-H), 2.50 (m, 1H, CH2-Ph), 2.78 (m, 1H, δ-H), 2.79 (m, 1H, CH2-Ph), 3.97 [d, 1H, J = 15 Hz, CH2 (NHBn, Urea) and 5-CH], 4.14 [m, 1H, CH2 (NHBn, Urea)], 4.25 [m, 1H, CH2 (NHBn)], 4.30 [m, 1H, CH2 (NHBn)], 4.44 [m, 1H, CH2 (NBn)], 4.58 [m, 1H, CH2 (NBn)], 6.42 (m, 1H, 2-CHNH), 6.90–7.40 [m, 21H, Ar and NHBn (Urea)], 7.92 (m, 3H, NH2·HCl), 8.74 (t, 1H, J = 6 Hz, NHBn); 13C-NMR (125 MHz, DMSO-d6) (R)-20a δ (ppm): 23.4 [Cγ], 29.2 [Cβ], 37.8 [CH2-Ph], 38.1 [Cδ], 42.0 [CH2 (NHBn)], 42.7 [CH2 (NHBn, Urea)], 43.7 [C3], 49.1 [CH2 (NBn)], 51.9 [Cα], 49.7 [C2-CH], 53.7 [C6], 53.9 [CH2CO], 60.8 [C2], 126.4, 126.6, 126.7, 127.1, 127.6, 128.1, 128.3, 128.5, 129.2 [20CH (Ar)], 136.4 [C (NBn)], 137.1 [C (Ph)], 139.0 [C (NHBn)], 140.4 [C (NHBn, Urea)], 158.1 [CO (Urea)], 170.9 [α-CONH]. (S)-20a δ (ppm): 29.4 [Cβ], 37.8 [CH2-Ph], 38.2 [Cδ], 42.0 [CH2 (NHBn)], 42.7 [CH2 (NHBn, Urea)], 49.1 [CH2 (NBn)], 49.7 [C2-CH], 126.4, 126.5, 126.7, 127.3, 127.8, 128.0, 128.3, 128.6, 129.2 [20CH (Ar)], 136.4 [C (NBn)], 137.1 [C (Ph)], 139.0 [C (NHBn)], 140.4 [C (NHBn, Urea)], 157.8 [CO (Urea)], 170.9 [α-CONH]; ES-MS m/z [M+2]+ calculated for C41H49N7O4: 705.3; found: 705.6.
N-[2-[4-Benzyl-(2RS)-[(1S)-(3-benzylureido)-2-phenylethyl]-5-oxopiperazin-1-yl]acetyl]-Lys-NHBn hydrochloride (20b). Amorphous solid [(R:S) = (3:1)] (151 mg, 100%); HPLC tR: 15.83 min [(R)-20b] and 16.25 min [(S)-20b]; 1H-NMR (500 MHz, DMSO-d6) (R)-20b δ (ppm): 1.29 (m, 2H, γ-H), 1.52 (m, 2H, δ-H), 1.54 (m, 1H, β-H), 1.72 (m, 1H, β-H), 2.67 (m, 1H, 2-H), 2.68 (m, 2H, ε-H), 2.69 (m, 1H, CH2-Ph), 2.87 (m, 1H, CH2-Ph), 3.34–3.87 (m, 6H, 3-H, 6-H and CH2CO), 4.03 [d, 1H, J = 15 Hz, CH2 (NHBn, Urea)], 4.14 [d, 1H, J = 15 Hz, CH2 (NHBn, Urea)], 4.20 [m, 1H, CH2 (NHBn)], 4.29 [m, 1H, CH2 (NHBn)], 4.30 (m, 1H, α-H), 4.33 (m, 1H, 2-CH), 4.50 [m, 1H, CH2 (NBn], 4.64 [m, 1H, CH2 (NBn], 6.48 (m, 1H, 2-CHNH), 6.94–7.41 [m, 21H, Ar and NHBn (Urea)], 7.87 (m, 3H, NH2·HCl), 8.50 (m, 1H, α-NH), 8.60 (m, 1H, NHBn). (S)-20b δ (ppm): 1.25 (m, 1H, γ-H), 1.35 (m, 1H, γ-H), 1.54 (m, 1H, β-H), 1.72 (m, 1H, β-H), 2.52 (m, 1H, CH2-Ph), 2.80 (m, 1H, CH2-Ph), 3.98 [d, 1H, J = 15 Hz, CH2 (NHBn, Urea)], 4.14 [m, 1H, CH2 (NHBn, Urea)], 4.20 [m, 1H, CH2 (NHBn)], 4.29 [m, 1H, CH2 (NHBn)], 6.40 (m, 1H, 2-CHNH), 6.94–7.41 [m, 21H, Ar and NHBn (Urea)], 7.87 (m, 3H, NH2·HCl), 8.62 (m, 1H, NHBn); 13C-NMR (125 MHz, DMSO-d6) (R)-20b δ (ppm): 22.7 [Cγ], 26.9 [Cδ], 31.7 [Cβ], 38.2 [CH2-Ph], 38.9 [Cε], 42.5 [CH2 (NHBn)], 43.0 [CH2 (NHBn, Urea)], 44.2 [C3], 49.5 [C2-CH], 49.7 [CH2 (NBn)], 53.0 [Cα], 54.0 [C6], 60.1 [C2], 126.7, 126.8, 127.0, 127.2, 127.5, 128.0, 128.5, 128.6, 129.0, 129.6 [20CH (Ar)], 136.9 [C (NBn)], 138.3 [C (Ph)], 139.7 [C (NHBn)], 140.8 [C (NHBn, Urea)], 158.6 [CO (Urea)], 171.5 [α-CONH]. (S)-20b δ (ppm): 22.7 [Cγ], 31.5 [Cβ], 38.0 [CH2-Ph], 42.5 [CH2 (NHBn)], 43.0 [CH2 (NHBn, Urea)], 126.7, 126.8, 127.0, 127.2, 127.7, 128.2, 128.5, 128.6, 129.0, 129.6 [20CH (Ar)], 136.9 [C (NBn)], 138.3 [C (Ph)], 139.7 [C (NHBn)], 141.0 [C (NHBn, Urea)], 158.2 [CO (Urea)], 171.5 [α-CONH]; ES-MS m/z [M+2]+ calculated for C42H51N7O4: 719.4; found: 719.9.
3.13. General Procedure for Removal of the N-Pbf Protecting Group. Synthesis of N-[2-[4-benzyl-5-oxo-(2RS)-[2-phenyl-(1S)-(3-phenyl-ureido)ethyl]piperazin-1-yl]acetyl]-Arg-NHBn Trifluoroacetate (19c)
The epimeric mixture of the Arg(Pbf) -derived phehylureido-piperazine 17c [(R:S) = (3:1)] (295 mg, 0.30 mmol) was dissolved in TFA/H2O/TIS mixture (90:5:5; 5 mL) and the mixture was stirred at room temperature for 5 h. Afterwards, the TFA was evaporated under stream of argon and the residue was centrifuged three times in diethyl ether (10 mL) at 5000 rpm and −15 °C for 15 min. The residue was dissolved in CH3CN/H2O (1:3, 2 mL) and the solution was lyophilized. The epimeric mixture of trifluoroacetate salts 19c [(R:S) = (3:1)] was obtained quantitatively (254 mg, 100%). HPLC tR: 16.60 min [(R)-19c] and 16.87 min [(S)-19c]; 1H-NMR (500 MHz, DMSO-d6) (R)-19c δ (ppm): 1.44 (m, 2H, γ-H), 1.63 (m, 1H, β-H), 1.72 (m, 1H, β-H), 2.71 (dd, 1H, J = 9 and 13.5 Hz, CH2-Ph), 2.95 (m, 1H, 2-H), 2.96 (m, 1H, CH2-Ph), 3.03 (m, 2H, δ-H), 3.20 (m, 1H, 3-H), 3.28 (m, 1H, 6-H), 3.30 (m, 1H, 3-H), 3.31 (m, 2H, CH2CO), 3.48 (d, 1H, J = 17 Hz, 6-H), 4.10 (m, 1H, 2-CH), 4.23 [m, 2H, CH2 (NHBn)], 4.30 [m, 1H, CH2 (NBn)], 4.32 (m, 1H, α-H), 4.72 [d, 1H, J = 15 Hz, CH2 (NBn)], 6.49 (m, 1H, 2-CHNH), 6.73–7.36 (m, 20H, Ar), 7.76 [m, 1H, NHC(NH2·CF3CO2H) = NH], 8.23 (d, 1H, J = 7 Hz, α-NH), 8.57 (t, 1H, J = 6 Hz, NHBn), 9.00 (m, 1H, NHPh). (S)-19c δ (ppm): 2.97 (m, 1H, δ-H), 3.07 (m, 1H, δ-H), 3.12 (m, 1H, CH2CO), 3.14 (m, 1H, 3-H), 3.23 (m, 1H, 6-H), 3.30 (m, 1H, 3-H), 3.42 (m, 1H, CH2CO), 3.77 (d, 1H, J = 16.5 Hz, 6-H), 3.91 (m, 1H, 2-CH), 4.04 [m, 1H, CH2 (NHBn)], 4.30 [m, 1H, CH2 (NHBn)], 4.43 [d, 1H, J = 15 Hz, CH2 (NBn)], 4.54 [d, 1H, J = 15 Hz, CH2 (NBn)], 6.73–7.36 (m, 20H, Ar), 7.76 [m, 1H, NHC(NH2·CF3CO2H) = NH], 8.59 (m, 1H, NHBn), 9.00 (m, 1H, NHPh); 13C-NMR (125 MHz, DMSO-d6) (R)-19c δ (ppm): 25.4 [Cγ], 29.7 [Cβ], 37.6 [CH2-Ph], 40.8 [Cδ], 42.5 [CH2 (NHBn)], 45.3 [C3], 49.5 [CH2 (NBn)], 50.8 [C2-CH], 52.6 [Cα], 54.9 [C6], 55.4 [CH2CO], 59.9 [C2], 118.2, 121.5, 126.4, 127.2, 217.3, 127.5, 128.1, 128.6, 128.7, 129.0, 129.7 [20CH (Ar)], 137.6 [C (NBn)], 139.2 [C (Ph)], 139.7 [C (NHBn)], 140.9 [C (NHPh)], 157.1 [CO (Urea)], 155.4 [C (NHC(NH2) = N)], 167.1 [C5], 170.0 [CO], 171.8 [α-CONH]. (S)-19c δ (ppm): 40.8 [Cδ], 42.3 [CH2 (NHBn)], 46.1 [C3], 48.9 [CH2 (NBn)], 51.6 [C2-CH], 54.9 [C6], 55.4 [CH2CO], 118.0, 121.5, 126.4, 127.2, 217.3, 127.6, 128.2, 128.4, 128.6, 129.0, 129.7 [20CH (Ar)], 138.0 [C (NBn)], 139.2 [C (Ph)], 139.7 [C (NHBn)], 140.9 [C (NHPh)], 157.5 [CO (Urea)], 155.8 [C (NHC(NH2) = N)], 170.6 [CO], 173.0 [α-CONH]; ES-MS m/z [M+1]+ calculated for C41H49N9O4: 732.4; found: 732.7.
3.14. General Procedure for the Synthesis of the Indazole-Derived Ureas 23b,c.
Propylene oxide (19 μL, 0.27 mmol) was added to a 0 °C cooled solution of 1-(2,6-dichlorobenzyl)-6-amino-3-(pyrrolidin-1-ylmethyl)-1
H-indazol [
24] (83 mg, 0.22 mmol) in dry THF (4 mL). Then, a solution of bis(trichloromethyl)carbonate (24 mg, 0.082 mmol) in dry THF (1 mL) was added dropwise and stirring was maintened at 0 °C for 15 min. Afterwards, the mixture was added dropwise to a 0 °C cooled solution of the corresponding epimeric mixture of hydrochlorides
16b,
c (0.22 mmol) and Et
3N (17 μL, 0.48 mmol) in dry THF (5 mL) and stirred for 2h. Then, the solvent was removed under reduced pressure and the residue was dissolved in CH
2Cl
2 (50 mL). The solution was washed with H
2O (2 × 10 mL), brine (10 mL), dried over Na
2SO
4, and evaporated to dryness. The residue was purified by reverse phase chromatography, using 10%–100% CH
3CN gradient in 0.05% TFA solution in H
2O as mobile phase, to afford the desired compounds
23b,
c.N-[2-[4-Benzyl-(2RS)-[(1S)-[3-(1-(2,6-dichlorobenzyl)-3-(pyrrolidin-1-ylmethyl)-1H-indazol-6-yl)ureido]-2-phenylethyl]-5-oxopiperazin-1-yl]acetyl]-Lys(Z)-NHBn (23b). Amorphous solid [(R:S) = (3:1)] (74 mg, 30%); HPLC tR: 19.99 min; 1H-NMR (500 MHz, CDCl3) (R)-23b δ (ppm): 1.26 (m, 2H, γ-H), 1.33 (m, 2H, δ-H), 1.70 (m, 1H, β-H), 1.72 (m, 2H, pyrrolidine), 1.82 (m, 1H, β-H), 1.88 (m, 2H, pyrrolidine), 2.68 (dd, 1H, J = 6.5 and 14 Hz, CH2-Ph), 2.90 (m, 1H, J = 8 and 14 Hz, CH2-Ph), 2.96 (m, 1H, 2-H), 2.98 (m, 2H, ε-H), 3.00 (m, 2H, pyrrolidine), 3.27 (m, 1H, 6-H), 3.42 (m, 1H, 3-H), 3.45 (m, 4H, pyrrolidine and CH2CO), 3.47 (m, 1H, 3-H), 3.53 (d, 1H, J = 16 Hz, 6-H), 4.18 [m, 1H, J = 14.5 Hz, CH2 (NBn)], 4.28 [m, 1H, CH2 (NHBn), 4.30 (m, 1H, 2-CH), 4.32 [m, 1H, CH2 (NHBn), 4.35 (s, 2H, CH2-pyrrolidine), 4.40 (m, 1H, α-H), 4.95 [m, 1H, CH2 (NBn], 4.98 [s, 2H, CH2 (Z)], 5.36 (m, 1H, NHZ), 5.50 (s, 2H, CH2-diClPh), 6.83 (d, 2H, J = 8 Hz, Ar), 7.02 (d, 2H, J = 7 Hz, Ar), 7.12–7.35 (m, 22H, Ar and NHBn), 7.91 (s, 1H, Ar), 8.06 (m, 1H, α-NH), 8.58 (m, 1H, 2-CHNH), 11.67 [m, 1H, Indz-NH (Urea)]. (S)-23b δ (ppm): 1.35 (m, 2H, δ-H), 1.72 (m, 2H, pyrrolidine), 1.88 (m, 2H, pyrrolidine), 3.00 (m, 2H, pyrrolidine), 3.24 (m, 1H, 3-H), 3.43 (m, 2H, pyrrolidine), 3.45 (m, 1H, 3-H), 4.35 (s, 2H, CH2-Pyrrolidine), 4.38 (m, 1H, α-H), 4.94 [s, 2H, CH2 (Z)], 5.36 (m, 1H, NHZ), 5.50 (s, 2H, CH2-diClPh), 6.76 (m, 2H, Ar), 7.02 (m, 2H, Ar), 7.12–7.35 (m, 22H, Ar and NHBn), 7.91 (s, 1H, Ar), 8.10 (m, 1H, α-NH), 8.70 (m, 1H, 2-CHNH), 11.67 [m, 1H, Indz-NH (Urea)]; 13C-NMR (125 MHz, CDCl3) (R)-23b δ (ppm): 22.8 [Cγ], 23.4 [2CH2 (pyrrolidine)], 29.1 [Cδ], 31.5 [Cβ], 37.7 [CH2-Ph], 40.3 [Cε], 43.5 [CH2 (NHBn)], 44.2 [C3], 47.6 [CH2-diClPh], 48.2 [CH2-pyrrolidine], 49.5 [CH2 (NBn)], 51.6 [C2-CH], 52.4 [2CH2 (pyrrolidine)], 53.4 [Cα], 55.2 [C6], 57.5 [CH2CO], 60.7 [C2], 66.6 [CH2 (Z)], 98.0, 116.0, 118.6 [3CH (Ar)], 119.2 [C (Ar)], 126.8, 127.3, 127.9, 128.0, 128.2, 128.4, 128.5, 128.6, 128.7, 128.9, 130.1 [23CH (Ar)], 131.3, 133.7 [2C (Ar)], 135.9 [C (NBn)], 136.5 [C (Z)], 136.8 [3C (Ph and Ar)], 137.7 [C (NHBn)], 139.3, 137.7 [2C (Ar)], 155.8 [CO (Z)], 156.8 [CO (Urea)], 166.9 [C5], 169.6 [CO], 172.5 [α-CONH]. (S)-23b δ (ppm): 23.4 [2CH2 (pyrrolidine)], 29.1 [Cδ], 44.2 [C3], 47.6 [CH2-diClPh], 48.2 [CH2-pyrrolidine], 52.4 [2CH2 (pyrrolidine)], 53.4 [Cα], 66.6 [CH2 (Z)], 98.0, 115.2, 117.5 [3CH (Ar)], 119.2 [C (Ar)], 126.8, 127.4, 127.9, 128.0, 128.2, 128.4, 128.5, 128.6, 128.7, 128.9, 130.1 [23CH (Ar)], 131.3, 133.7 [2C (Ar)], 135.9 [C (NBn)], 136.5 [C (Z)], 136.8 [3C (Ph and Ar)], 137.7 [C (NHBn)], 139.3, 137.7 [2C (Ar)], 155.8 [CO (Z)], 156.8 [CO (urea)], 172.5 [α-CONH]; ES-MS m/z 1120.9 [M+1]+; C62H68Cl2N10O6 (%): C: 66.48, H: 6.12, N: 12.50. Found (%): C: 66.79, H: 6.38, N: 12.34.
N-[2-[4-Benzyl-(2RS)-[(1S)-[3-(1-(2,6-dichlorobenzyl)-3-(pyrrolidin-1-ylmethyl)-1H-indazol-6-yl)ureido]-2-phenylethyl]-5-oxopiperazin-1-yl]acetyl]-Arg(Pbf)-NHBn (23c). Amorphous solid [(R:S) = (3:1)] (106 mg, 38%); HPLC tR: 20.38 min [(R)-23c] and 21.40 min [(S)-23c]; 1H-NMR (500 MHz, (CD3)2CO) (R)-23c δ (ppm): 1.30 [s, 6H, 2CH3 (Pbf)], 1.40 (m, 2H, γ-H), 1.60 (m, 1H, β-H), 1.73 (m, 1H, β-H), 1.86 [s, 3H, CH3 (Pbf)], 1.88 (m, 2H, pyrrolidine), 1.97 (m, 2H, pyrrolidine), 2.36 [s, 3H, CH3 (Pbf)], 2.44 [s, 3H, CH3 (Pbf)], 2.72 (m, 2H, CH2-Ph), 2.86 [s, 2H, CH2 (Pbf)], 3.00 (m, 1H, δ-H), 3.08 (m, 1H, 2-H), 3.10 (m, 1H, δ-H), 3.26 (m, 1H, CH2CO), 3.32 (m, 1H, 6-H), 3.34 (m, 2H, pyrrolidine), 3.52 (m, 1H, CH2CO), 3.57 (m, 1H, 6-H), 3.58 (m, 1H, 3-H), 3.62 (m, 1H, 3-H), 3.68 (m, 2H, pyrrolidine), 4.20 [m, 1H, CH2 (NBn)], 4.34 [m, 1H, CH2 (NHBn)], 4.38 [m, 2H, 2-CH and CH2 (NHBn)], 4.39 (m, 1H, α-H), 4.66 (s, 2H, CH2-pyrrolidine), 4.88 [d, 1H, J = 15 Hz, CH2 (NBn)], 5.55 (d, 2H, J = 6 Hz, CH2-diClPh), 6.39 [m, 3H, NHC(NH2) = N], 6.95–7.34 (m, 21H, Ar), 7.86 (m, 1H, NHBn), 8.26 (d, 1H, J = 6 Hz, α-NH), 9.26 (m, 1H, 2-CHNH), 10.32 [m, 1H, Indz-NH (Urea)]. (S)-23c δ (ppm): 1.30 [s, 6H, 2CH3 (Pbf)], 1.86 [s, 3H, CH3 (Pbf)], 1.88 (m, 2H, pyrrolidine), 1.97 (m, 2H, pyrrolidine), 2.36 [s, 3H, CH3 (Pbf)], 2.44 [s, 3H, CH3 (Pbf)], 2.68 (m, 2H, CH2-Ph), 2.80 (m, 2H, CH2-Ph), 2.86 [s, 2H, CH2 (Pbf)], 3.34 (m, 2H, pyrrolidine), 3.68 (m, 2H, pyrrolidine), 4.10 [dd, 1H, J = 6 and 15 Hz, CH2 (NHBn)], 4.37 (m, 1H, α-H), 4.40 [m, 1H, CH2 (NHBn)], 4.43 [d, 1H, J = 14 Hz, CH2 (NBn)], 4.58 [m, 1H, CH2 (NBn)], 4.66 (m, 2H, CH2-pyrrolidine), 5.55 (m, 2H, J = 6 Hz, CH2-diClPh), 6.50 [m, 3H, NHC(NH2) = N], 6.95–7.34 (m, 21H, Ar), 7.91 (m, 1H, NHBn), 8.26 (m, 1H, α-NH), 9.26 (m, 1H, 2-CHNH), 10.32 [m, 1H, Indz-NH (urea)]; 13C-NMR (125 MHz, (CD3)2CO) (R)-23c δ (ppm): 13.2, 19.0, 20.2 [3CH3 (Pbf)], 24.5 [2CH2 (pyrrolidine)], 27.5 [Cγ], 28.6 [2CH3 (Pbf)], 31.3 [Cβ], 39.2 [CH2-Ph], 41.4 [Cδ], 44.0 [CH2 (NHBn)], 44.3 [CH2 (Pbf)], 45.7 [C3], 49.0 [CH2-diClPh], 50.5 [CH2 (NBn)], 50.7 [CH2-pyrrolidine], 54.1 [C2-CH and Cα], 55.1 [2CH2 (pyrrolidine)], 56.0 [C6], 59.5 [CH2CO], 63.2 [C2], 87.5 [C (Pbf)], 98.9, 117.2 [2CH (Ar)], 118.4 [C (Pbf)], 118.6 [CH (Ar)], 119.7 [C (Ar)], 121.4 [CH (Ar)], 126.2 [C (Pbf)], 127.7, 128.2, 128.7, 129.6, 129.8, 130.1, 130.2, 130.8, 132.1 [17CH (Ar)], 133.5, 135.7 [2C (Pbf)], 136.7, 138.3 [3C (Ar)], 139.0 [C (NBn)], 139.5 [C (Pbf)], 140.3 [C (Ph)], 140.9 [C (NHBn)], 141.9, 143.5 [3C (Ar)], 157.6 [CO (Urea)], 160.9 [C (NHC(NH2) = N)], 159.7 [C (Pbf)], 168.3 [C5], 171.1 [CO], 173.2 [α-CONH]. (S)-23c δ (ppm): 13.2, 19.0, 20.2 [3CH3 (Pbf)], 24.5 [2CH2 (pyrrolidine)], 28.6 [2CH3 (Pbf)], 39.2 [CH2-Ph], 44.0 [CH2 (NHBn)], 44.3 [CH2 (Pbf)], 49.0 [CH2-diClPh], 50.4 [CH2 (NBn)], 50.7 [CH2-pyrrolidine], 54.1 [Cα], 55.1 [2CH2 (pyrrolidine)], 87.5 [C (Pbf)], 98.9, 117.2 [2CH (Ar)], 118.4 [C (Pbf)], 118.6 [CH (Ar)], 119.1 [C (Ar)], 121.4 [CH (Ar)], 126.2 [C (Pbf)], 127.7, 128.2, 128.8, 129.6, 129.8, 130.1, 130.2, 131.0, 132.1 [17CH (Ar)], 133.5, 135.7 [2C (Pbf)], 136.7, 138.3 [3C (Ar)], 139.1 [C (NBn)], 139.5 [C (Pbf)], 140.3 [C (Ph)], 140.9 [C (NHBn)], 141.9, 143.5 [3C (Ar)], 158.1 [CO (Urea)], 160.9 [C (NHC(NH2) = N)], 159.7 [C (Pbf)]; ES-MS m/z 1267.7 [M+1]+; C67H78Cl2N12O7S (%): C: 63.54, H: 6.21, N: 13.27. Found (%): C: 63.78, H: 6.02, N: 13.39.
3.15. N-Z Removal in 23b. Synthesis of N-[2-[4-Benzyl-(2RS)-[(1S)-[3-(1-(2,6-dichlorobenzyl)-3-(pyrrolidin-1-ylmethyl)-1H-indazol-6-yl)ureido]-2-phenylethyl]-5-oxopiperazin-1-yl]acetyl]-Lys-NHBn hydrochloride (24b)
It was carried out by applying the general methodology for N-Z removal above described for the synthesis of (11–12)a,b. Amorphous solid [(R:S) = (3:1)] (70 mg, 100%); HPLC tR: 14.99 min; 1H-NMR (500 MHz, DMSO-d6) (R)-24b δ (ppm): 1.30 (m, 2H, γ-H), 1.50 (m, 2H, δ-H), 1.62 (m, 1H, β-H), 1.72 (m, 1H, β-H), 1.79 (m, 2H, pyrrolidine), 1.85 (m, 2H, pyrrolidine), 2.74 (dd, 1H, J = 10 and 14 Hz, CH2-Ph), 2.96 (m, 1H, CH2-Ph), 2.65 (m, 3H, 5-H and ε-H), 3.05 (m, 2H, pyrrolidine), 3.35 (m, 2H, pyrrolidine), 3.44–3.98 (m, 6H, 3-H, 6-H and CH2CO), 4.25 [d, 1H, J = 6 Hz, CH2 (NHBn), 4.27 [d, 1H, J = 6 Hz, CH2 (NHBn), 4.33 (m, 2H, 2-CH and α-H), 4.46 [m, 1H, CH2 (NBn], 4.57 (d, 2H, J = 5 Hz, CH2-pyrrolidine), 4.72 [d, 1H, J = 15 Hz, CH2 (NBn], 5.59 (s, 2H, CH2-diClPh), 7.01 (d, 1H, J = 8 Hz, Ar), 6.96–7.46 (m, 16H, Ar), 7.52 (d, 2H, J = 8 Hz, Ar), 7.83 (d, 1H, J = 9 Hz, Ar), 7.86 (s, 1H, Ar), 7.95 (m, 3H, NH2·HCl), 8.45 (m, 1H, α-NH), 8.63 (t, 1H, J = 6 Hz, NHBn), 9.35 (s, 1H, 2-CHNH), 11.68 [m, 2H, Indz-NH (urea) and N·HCl (pyrrolidine)]. (S)-24b δ (ppm): 1.79 (m, 2H, pyrrolidine), 1.85 (m, 2H, pyrrolidine), 2.74 (m, 1H, CH2-Ph), 2.96 (m, 1H, CH2-Ph), 3.05 (m, 2H, pyrrolidine), 3.35 (m, 2H, pyrrolidine), 4.22 [m, 1H, CH2 (NHBn), 4.28 [m, 1H, CH2 (NHBn), 4.57 (d, 2H, J = 5 Hz, CH2-pyrrolidine), 4.72 [d, 1H, J = 15 Hz, CH2 (NBn], 5.59 (s, 2H, CH2-diClPh), 7.01 (d, 1H, J = 8 Hz, Ar), 6.96–7.46 (m, 16H, Ar), 7.52 (d, 2H, J = 8 Hz, Ar), 7.83 (d, 1H, J = 9 Hz, Ar), 7.86 (s, 1H, Ar), 7.95 (m, 3H, NH2·HCl), 8.45 (m, 1H, α-NH), 8.63 (m, 1H, NHBn), 9.35 (s, 1H, 2-CHNH), 11.68 [m, 2H, Indz-NH (Urea) and N·HCl (pyrrolidine)]; 13C-NMR (125 MHz, DMSO-d6) (R)-24b δ (ppm): 22.3 [Cγ], 22.6 [2CH2 (pyrrolidine)], 26.4 [Cδ], 31.4 [Cβ], 37.9 [CH2-Ph], 38.4 [Cε], 42.0 [CH2 (NHBn)], 44.1 [C3], 47.2 [CH2-diClPh], 47.7 [CH2-pyrrolidine], 49.2 [CH2 (NBn)], 49.6 [C2-CH], 52.5 [2CH2 (pyrrolidine), Cα and CH2CO], 53.9 [C6], 60.3 [C2], 96.1, 114.5 [2CH (Ar)], 117.7 [C (Ar)], 120.6, 126.7, 127.0, 127.2, 127.6, 128.2, 128.5, 128.7, 129.2 [19CH (Ar)], 130.8, 131.4 [4C (Ar)], 135.4 [C (NBn)], 136.0 [C (Ph)], 139.2 [C (NHBn)], 141.2 [2C (Ar)], 155.2 [CO (urea)], 171.1 [α-CONH]. (S)-24b δ (ppm): 22.6 [2CH2 (pyrrolidine)], 37.8 [CH2-Ph], 42.0 [CH2 (NHBn)], 47.2 [CH2-diClPh], 47.7 [CH2-pyrrolidine], 52.5 [2CH2 (pyrrolidine)], 96.1, 114.5 [2CH (Ar)], 117.7 [C (Ar)], 120.6, 126.7, 127.0, 127.2, 127.6, 128.1, 128.5, 128.7, 129.2 [19CH (Ar)], 130.8, 131.4 [4C (Ar)], 135.4 [C (NBn)], 136.0 [C (Ph)], 139.6 [C (NHBn)], 141.2 [2C (Ar)], 155.2 [CO (urea)], 171.1 [α-CONH]; ES-MS m/z [(M+2)/2]+ calculated for C54H62Cl2N10O4: 493.2; found: 493.6.
3.16. N-Pbf Removal in 23c. Synthesis of N-[2-[4-Benzyl-(2RS)-[(1S)-[3-(1-(2,6-dichlorobenzyl)-3-(pyrrolidin-1-ylmethyl)-1H-indazol-6-yl)ureido]-2-phenylethyl]-5-oxopiperazin-1-yl]acetyl]-Arg-NHBn Trifluoroacetate (24c)
It was carried out by applying the above described methodology for N-Pbf removal in the Arg derivative 19c. Amorphous solid [(R:S) = (3:1)] (104 mg, 100%); HPLC tR: 15.14 min [(R)-24c] and 15.72 min [(S)-24c]; 1H-NMR (500 MHz, (CD3)2CO) (R)-24c δ (ppm): 1.44 (m, 2H, γ-H), 1.60 (m, 1H, β-H), 1.84 (m, 1H, β-H), 1.83 (m, 4H, pyrrolidine), 2.75 (dd, 1H, J = 9.5 and 14 Hz, CH2-Ph), 3.00 (m, 1H, 2-H), 3.04 (m, 2H, δ-H), 3.06 (m, 1H, CH2-Ph), 3.20 (m, 1H, CH2CO), 3.26 (m, 1H, 3-H), 3.32 (m, 1H, 6-H), 3.35 (m, 4H, pyrrolidine), 3.39 (m, 1H, 3-H), 3.42 (m, 1H, CH2CO), 3.47 (m, 1H, 6-H), 4.21 (m, 1H, 2-CH), 4.27 [d, 2H, J = 6 Hz, CH2 (NHBn)], 4.33 [d, 1H, J = 15 Hz, CH2 (NBn)], 4.40 (td, 1H, J = 5.5 and 8 Hz, α-H), 4.58 (s, 2H, CH2-pyrrolidine), 4.74 [d, 1H, J = 15 Hz, CH2 (NBn)], 5.60 (s, 2H, CH2-diClPh), 6.37 (m, 1H, 2-CHNH), 6.90–7.44 (m, 18H, Ar), 7.52 [m, 2H, NHC(NH2·CF3CO2H) = NH and Ar], 7.72 (d, 1H, J = 9 Hz, Ar), 7.92 (s, 1H, Ar), 8.09 (d, 1H, J = 8 Hz, α-NH), 8.58 (t, 1H, J = 6 Hz, NHBn), 8.77 [m, 1H, Indz-NH (Urea)], 9.95 [m, 1H, N·CF3CO2H (Pyrrolidine)]. (S)-24c δ (ppm): 1.83 (m, 4H, pyrrolidine), 2.63 (m, 1H, CH2-Ph), 2.82 (m, 1H, CH2-Ph), 3.30 (m, 1H, 6-H), 3.35 (m, 4H, pyrrolidine), 3.50 (m, 1H, 6-H), 4.04 (m, 1H, 2-CH), 4.14 [m, 1H, CH2 (NHBn)], 4.28 [m, 1H, CH2 (NHBn)], 4.58 (s, 2H, CH2-pyrrolidine), 5.57 (s, 2H, CH2-diClPh), 6.90–7.44 (m, 18H, Ar), 7.52 [m, 2H, NHC(NH2·CF3CO2H) = NH and Ar], 7.70 (d, 1H, J = 9 Hz, Ar), 7.95 (s, 1H, Ar), 8.57 (m, 1H, NHBn), 9.95 [m, 1H, N·CF3CO2H (pyrrolidine)]; 13C-NMR (125 MHz, (CD3)2CO) (R)-24c δ (ppm): 23.0 [2CH2 (pyrrolidine)], 25.5 [Cγ], 30.0 [Cβ], 37.8 [CH2-Ph], 40.4 [Cδ], 42.5 [CH2 (NHBn)], 44.9 [C3], 47.7 [CH2-diClPh], 48.6 [CH2-pyrrolidine], 49.6 [CH2 (NBn)], 50.5 [C2-CH], 52.2 [Cα], 53.5 [2CH2 (pyrrolidine)], 54.4 [C6], 55.2 [CH2CO], 59.7 [C2], 96.8, 115.1 [2CH (Ar)], 118.0 [C (Ar)], 120.7 [CH (Ar)], 126.5, 127.2, 127.5, 128.0, 128.5, 128.7, 129.0, 129.2, 129.7 [18CH (Ar)], 131.4, 131.9, 136.5 [4C (Ar)], 137.4 [C (NBn)], 139.0 [C (Ph)], 139.6 [C (NHBn)], 140.2, 141.9 [2C (Ar)], 157.0 [CO (urea)], 155.3 [C (NHC(NH2) = N)], 167.0 [CO], 169.8 [C5], 171.6 [α-CONH]. (S)-24c δ (ppm): 23.0 [2CH2 (pyrrolidine)], 37.8 [CH2-Ph], 42.5 [CH2 (NHBn)], 47.7 [CH2-diClPh], 48.6 [CH2-pyrrolidine], 50.4 [C2-CH], 53.5 [2CH2 (pyrrolidine)], 54.4 [C6], 96.8, 115.1 [2CH (Ar)], 118.0 [C (Ar)], 120.7 [CH (Ar)], 126.5, 127.4, 127.6, 128.3, 128.5, 128.7, 129.0, 129.2, 129.6 [18CH (Ar)], 131.4, 131.9, 136.5 [4C (Ar)], 137.4 [C (NBn)], 139.0 [C (Ph)], 139.6 [C (NHBn)], 140.2, 141.9 [2C (Ar)], 155.3 [C (NHC(NH2) = N)]; ES-MS m/z [(M+2)/2]+ calculated for C54H62Cl2N12O4: 507.2; found: 507.
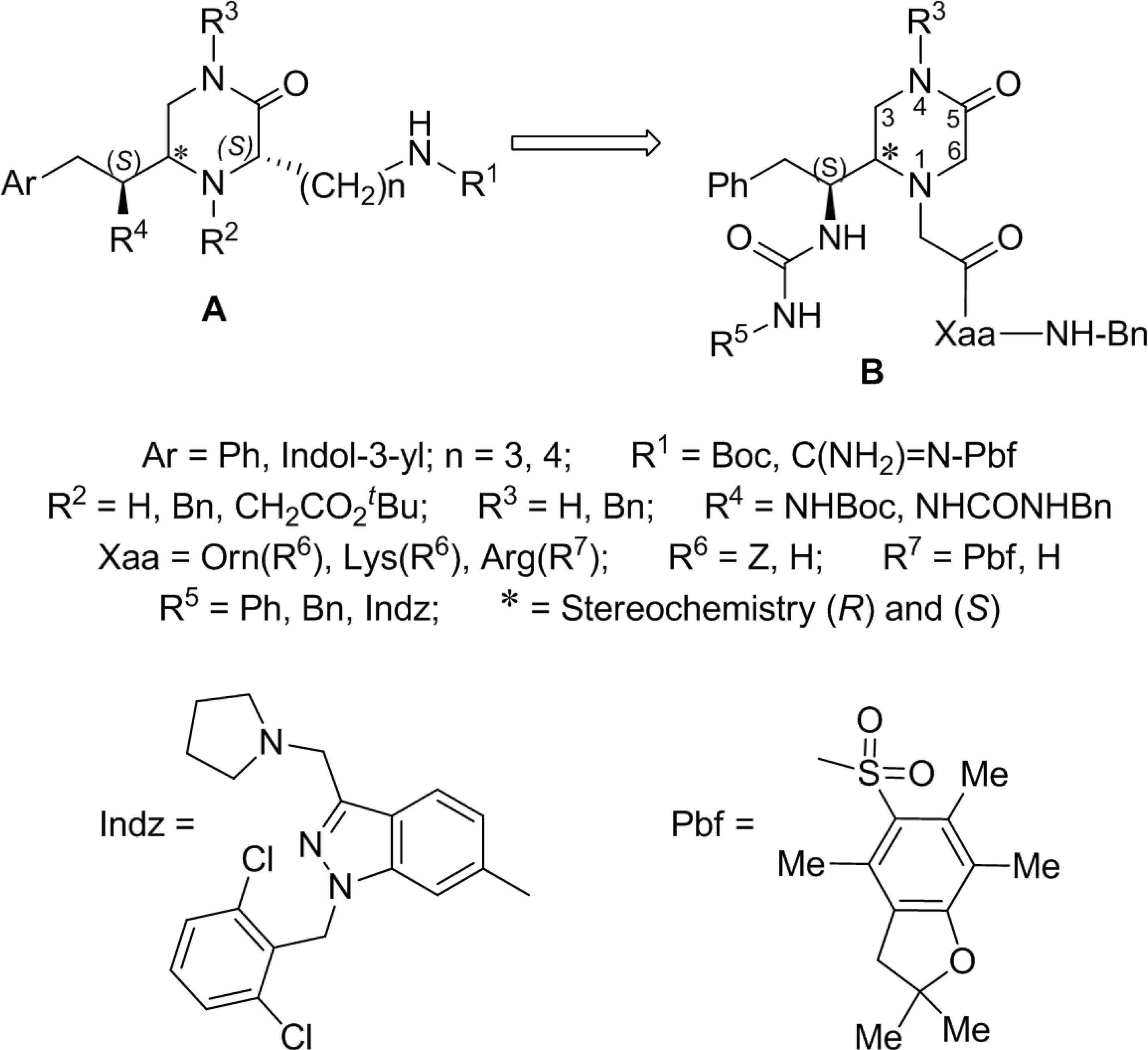


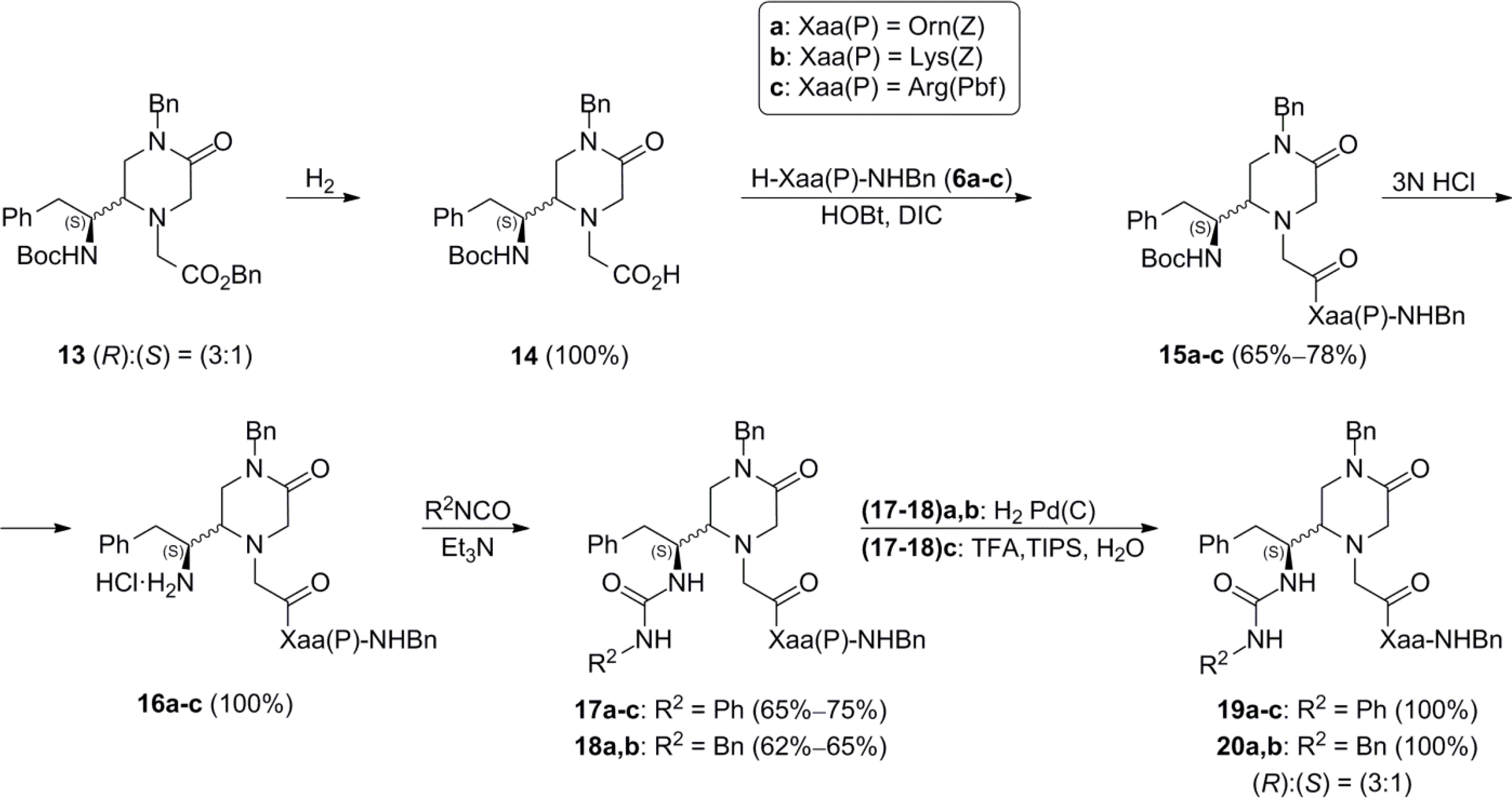
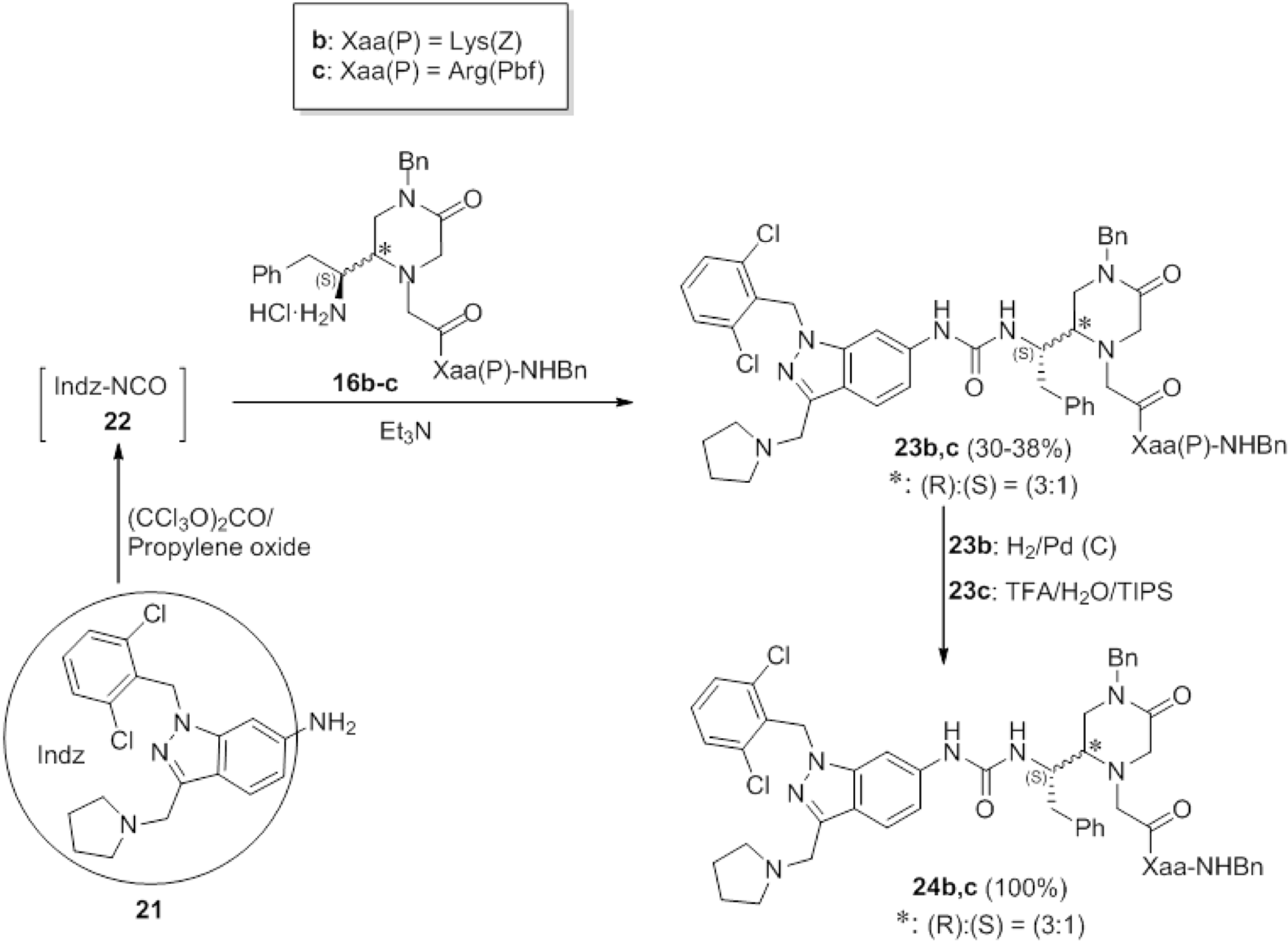
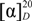 = −0.1 (c 1, MeOH); HPLC tR: 20.30 min; 1H-NMR (400 MHz, CDCl3) δ (ppm): 1.30 (m, 2H, γ-H), 1.50 (m, 1H, β-H), 1.70 (m, 1H, β-H), 2.82 (m, 1H, CH2-Ph), 2.83 (m, 1H, δ-H), 2.84 (m, 1H, 2-H), 2.88 (m, 1H, CH2-Ph), 3.03 (m, 1H, CH2CO), 3.14 (m, 1H, 6-H), 3.35 (m, 1H, 6-H), 3.37 (m, 1H, 3-H), 3.24 (m, 1H, δ-H), 3.44 (m, 1H, CH2CO), 4.20 (m, 1H, 3-H), 4.25 (m, 1H, 2-CH), 4.26 [m, 1H, CH2 (NHBn)], 4.36 [m, 1H, CH2 (NHBn)], 4.60 (m, 1H, α-H), 4.82 [d, 1H, J = 12.5 Hz, CH2 (Z)], 4.91 [d, 1H, J = 12.5 Hz, CH2 (Z)], 5.25 (m, 1H, NHZ), 5.97 (m, 1H, 4-H), 6.12 (m, 1H, 2-CHNH), 6.91–7.35 (m, 20H, Ar), 7.46 (m, 1H, NHBn), 7.65 [m, 1H, NHPh], 7.88 (m, 1H, α-NH); 13C-NMR (100 MHz, CDCl3) δ (ppm): 26.7 [Cγ], 29.9 [Cβ], 37.9 [CH2-Ph], 39.7 [Cδ], 40.1 [C3], 43.7 [CH2 (NHBn)], 51.6 [C2-CH], 52.0 [Cα], 54.9 [C6], 57.8 [CH2CO], 59.6 [C2], 67.0 [CH2 (Z)], 116.7, 119.7, 123.0, 127.0, 127.9, 120.4, 128.8, 129.0, 129.3 [20CH (Ar)], 134.3 [C (Ph)], 136.1 [C (Z)], 137.9 [C (NHBn)], 139.5 [C (NHPh)], 157.2 [CO (Z) and CO (Urea)], 168.7 [C5], 170.0 [CO], 171.1 [α-CONH]; ES-MS m/z 734.4 [M+1]+; C41H47N7O6 (%): C: 67.10, H: 6.46, N: 13.36. Found (%): C: 67.28, H: 6.59, N: 13.19.
= −0.1 (c 1, MeOH); HPLC tR: 20.30 min; 1H-NMR (400 MHz, CDCl3) δ (ppm): 1.30 (m, 2H, γ-H), 1.50 (m, 1H, β-H), 1.70 (m, 1H, β-H), 2.82 (m, 1H, CH2-Ph), 2.83 (m, 1H, δ-H), 2.84 (m, 1H, 2-H), 2.88 (m, 1H, CH2-Ph), 3.03 (m, 1H, CH2CO), 3.14 (m, 1H, 6-H), 3.35 (m, 1H, 6-H), 3.37 (m, 1H, 3-H), 3.24 (m, 1H, δ-H), 3.44 (m, 1H, CH2CO), 4.20 (m, 1H, 3-H), 4.25 (m, 1H, 2-CH), 4.26 [m, 1H, CH2 (NHBn)], 4.36 [m, 1H, CH2 (NHBn)], 4.60 (m, 1H, α-H), 4.82 [d, 1H, J = 12.5 Hz, CH2 (Z)], 4.91 [d, 1H, J = 12.5 Hz, CH2 (Z)], 5.25 (m, 1H, NHZ), 5.97 (m, 1H, 4-H), 6.12 (m, 1H, 2-CHNH), 6.91–7.35 (m, 20H, Ar), 7.46 (m, 1H, NHBn), 7.65 [m, 1H, NHPh], 7.88 (m, 1H, α-NH); 13C-NMR (100 MHz, CDCl3) δ (ppm): 26.7 [Cγ], 29.9 [Cβ], 37.9 [CH2-Ph], 39.7 [Cδ], 40.1 [C3], 43.7 [CH2 (NHBn)], 51.6 [C2-CH], 52.0 [Cα], 54.9 [C6], 57.8 [CH2CO], 59.6 [C2], 67.0 [CH2 (Z)], 116.7, 119.7, 123.0, 127.0, 127.9, 120.4, 128.8, 129.0, 129.3 [20CH (Ar)], 134.3 [C (Ph)], 136.1 [C (Z)], 137.9 [C (NHBn)], 139.5 [C (NHPh)], 157.2 [CO (Z) and CO (Urea)], 168.7 [C5], 170.0 [CO], 171.1 [α-CONH]; ES-MS m/z 734.4 [M+1]+; C41H47N7O6 (%): C: 67.10, H: 6.46, N: 13.36. Found (%): C: 67.28, H: 6.59, N: 13.19.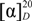 = +9.2 (c 1.5, MeOH); tR: 21.41 min; 1H-NMR (500 MHz, CDCl3) δ (ppm): 1.32 (m, 1H, γ-H), 1.40 (m, 1H, γ-H), 1.53 (m, 1H, β-H), 1.72 (m, 1H, β-H), 2.54 (dd, 1H, J = 11 and 13.5 Hz, CH2-Ph), 2.90 (dd, 1H, J = 4 and 13.5 Hz, CH2-Ph), 2.92 (m, 1H, δ-H), 3.08 (m, 1H, 5-H), 3.10 (m, 1H, 3-H), 3.14 (m, 1H, 6-H), 3.32 (m, 2H, CH2CO), 3.35 (m, 1H, δ-H), 3.54 (d, 1H, J = 18 Hz, 6-H), 3.92 (m, 1H, 2-CH), 3.95 (m, 1H, 3-H), 4.28 [dd, 1H, J = 5 and 15 Hz, CH2 (NHBn)], 4.44 [dd, 1H, J = 6 and 15 Hz, CH2 (NHBn)], 4.60 [d, 1H, J = 13 Hz, CH2 (Z)], 4.71 (m, 1H, α-H), 4.83 [d, 1H, J = 13 Hz, CH2 (Z)], 4.95 (m, 1H, NHZ), 5.67 (m, 1H, 4-H), 5.94 (d, 1H, J = 6 Hz, 2-CHNH), 6.83–7.35 (m, 20H, Ar), 7.53 (m, 1H, NHBn), 7.93 [m, 1H, NHPh], 7.97 (d, 1H, J = 8.5 Hz, α-NH); 13C-NMR (125 MHz, CDCl3) δ (ppm): 26.4 [Cγ], 31.1 [Cβ], 35.8 [C3], 37.8 [CH2-Ph], 38.9 [Cδ], 43.8 [CH2 (NHBn)], 50.8 [Cα], 51.9 [C6], 52.3 [C2-CH], 57.7 [CH2CO], 58.6 [C2], 66.7 [CH2 (Z)], 118.3, 122.2, 127.5, 127.8, 128.2, 128.5, 128.8, 128.9, 129.0, 129.3 [20CH (Ar)], 135.6 [C (Ph)], 136.2 [C (Z)], 137.3 [C (NHBn)], 139.6 [C (NHPh)], 155.6 [CO (Z)], 157.6 [CO (Urea)], 168.8 [C5], 169.2 [CO], 172.9 [α-CONH]; ES-MS m/z 734.5 [M+1]+; C41H47N7O6 (%): C: 67.10, H: 6.46, N: 13.36. Found (%): C: 67.21, H: 6.30, N: 13.49.
= +9.2 (c 1.5, MeOH); tR: 21.41 min; 1H-NMR (500 MHz, CDCl3) δ (ppm): 1.32 (m, 1H, γ-H), 1.40 (m, 1H, γ-H), 1.53 (m, 1H, β-H), 1.72 (m, 1H, β-H), 2.54 (dd, 1H, J = 11 and 13.5 Hz, CH2-Ph), 2.90 (dd, 1H, J = 4 and 13.5 Hz, CH2-Ph), 2.92 (m, 1H, δ-H), 3.08 (m, 1H, 5-H), 3.10 (m, 1H, 3-H), 3.14 (m, 1H, 6-H), 3.32 (m, 2H, CH2CO), 3.35 (m, 1H, δ-H), 3.54 (d, 1H, J = 18 Hz, 6-H), 3.92 (m, 1H, 2-CH), 3.95 (m, 1H, 3-H), 4.28 [dd, 1H, J = 5 and 15 Hz, CH2 (NHBn)], 4.44 [dd, 1H, J = 6 and 15 Hz, CH2 (NHBn)], 4.60 [d, 1H, J = 13 Hz, CH2 (Z)], 4.71 (m, 1H, α-H), 4.83 [d, 1H, J = 13 Hz, CH2 (Z)], 4.95 (m, 1H, NHZ), 5.67 (m, 1H, 4-H), 5.94 (d, 1H, J = 6 Hz, 2-CHNH), 6.83–7.35 (m, 20H, Ar), 7.53 (m, 1H, NHBn), 7.93 [m, 1H, NHPh], 7.97 (d, 1H, J = 8.5 Hz, α-NH); 13C-NMR (125 MHz, CDCl3) δ (ppm): 26.4 [Cγ], 31.1 [Cβ], 35.8 [C3], 37.8 [CH2-Ph], 38.9 [Cδ], 43.8 [CH2 (NHBn)], 50.8 [Cα], 51.9 [C6], 52.3 [C2-CH], 57.7 [CH2CO], 58.6 [C2], 66.7 [CH2 (Z)], 118.3, 122.2, 127.5, 127.8, 128.2, 128.5, 128.8, 128.9, 129.0, 129.3 [20CH (Ar)], 135.6 [C (Ph)], 136.2 [C (Z)], 137.3 [C (NHBn)], 139.6 [C (NHPh)], 155.6 [CO (Z)], 157.6 [CO (Urea)], 168.8 [C5], 169.2 [CO], 172.9 [α-CONH]; ES-MS m/z 734.5 [M+1]+; C41H47N7O6 (%): C: 67.10, H: 6.46, N: 13.36. Found (%): C: 67.21, H: 6.30, N: 13.49.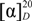 = −3.7 (c 1.5, MeOH); HPLC tR: 20.09 min; 1H-NMR (400 MHz, CDCl3) δ (ppm): 1.30 (m, 2H, γ-H), 1.40 (m, 2H, δ-H), 1.50 (m, 1H, β-H), 1.73 (m, 1H, β-H), 2.80 (m, 1H, CH2-Ph), 2.87 (m, 1H, CH2-Ph), 2.85 (m, 1H, ε-H), 2.92 (m, 1H, 2-H), 3.03 (m, 1H, CH2CO), 3.16 (m, 1H, 6-H), 3.20 (m, 2H, 3-H and ε-H), 3.38 (m, 1H, 6-H), 3.44 (m, 1H, CH2CO), 4.25 (m, 1H, 3-H), 4.28 (m, 1H, 2-CH), 4.30 [m, 1H, CH2 (NHBn)], 4.38 [m, 1H, CH2 (NHBn)], 4.50 (m, 1H, α-H), 5.01 [m, 2H, CH2 (Z)], 5.27 (m, 1H, NHZ), 5.98 (m, 1H, 2-CHNH), 6.23 (m, 1H, 4-H), 6.78–7.59 (m, 21H, Ar and NHBn), 7.64 (m, 1H, NHPh), 7.90 (m, 1H, α-NH); 13C-NMR (100 MHz, CDCl3) δ (ppm): 23.1 [Cγ], 29.3 [Cδ], 31.9 [Cβ], 38.3 [CH2-Ph], 39.6 [C3], 40.7 [Cε], 43.6 [CH2 (NHBn)], 51.7 [C6], 53.9 [C2-CH], 54.8 [Cα], 59.2 [CH2CO], 59.8 [C2], 66.8 [CH2 (Z)], 119.2, 119.9, 123.1, 127.1, 127.5, 127.6, 127.7, 128.1, 128.4, 128.8, 128.9, 129.0, 129.1 [20CH (Ar)], 136.8 [C (Ph)], 137.5 [C (Z)], 138.2 [C (NHBn)], 139.2 [C (NHPh)], 156.1 [CO (Z)], 156.9 [CO (Urea)], 170.5 [C5 and CO], 172.4 [α-CONH]; ES-MS m/z 748.6 [M+1]+; C42H49N7O6 (%): C: 67.45, H: 6.60, N: 13.11. Found (%): C: 67.62, H: 6.74, N: 13.02.
= −3.7 (c 1.5, MeOH); HPLC tR: 20.09 min; 1H-NMR (400 MHz, CDCl3) δ (ppm): 1.30 (m, 2H, γ-H), 1.40 (m, 2H, δ-H), 1.50 (m, 1H, β-H), 1.73 (m, 1H, β-H), 2.80 (m, 1H, CH2-Ph), 2.87 (m, 1H, CH2-Ph), 2.85 (m, 1H, ε-H), 2.92 (m, 1H, 2-H), 3.03 (m, 1H, CH2CO), 3.16 (m, 1H, 6-H), 3.20 (m, 2H, 3-H and ε-H), 3.38 (m, 1H, 6-H), 3.44 (m, 1H, CH2CO), 4.25 (m, 1H, 3-H), 4.28 (m, 1H, 2-CH), 4.30 [m, 1H, CH2 (NHBn)], 4.38 [m, 1H, CH2 (NHBn)], 4.50 (m, 1H, α-H), 5.01 [m, 2H, CH2 (Z)], 5.27 (m, 1H, NHZ), 5.98 (m, 1H, 2-CHNH), 6.23 (m, 1H, 4-H), 6.78–7.59 (m, 21H, Ar and NHBn), 7.64 (m, 1H, NHPh), 7.90 (m, 1H, α-NH); 13C-NMR (100 MHz, CDCl3) δ (ppm): 23.1 [Cγ], 29.3 [Cδ], 31.9 [Cβ], 38.3 [CH2-Ph], 39.6 [C3], 40.7 [Cε], 43.6 [CH2 (NHBn)], 51.7 [C6], 53.9 [C2-CH], 54.8 [Cα], 59.2 [CH2CO], 59.8 [C2], 66.8 [CH2 (Z)], 119.2, 119.9, 123.1, 127.1, 127.5, 127.6, 127.7, 128.1, 128.4, 128.8, 128.9, 129.0, 129.1 [20CH (Ar)], 136.8 [C (Ph)], 137.5 [C (Z)], 138.2 [C (NHBn)], 139.2 [C (NHPh)], 156.1 [CO (Z)], 156.9 [CO (Urea)], 170.5 [C5 and CO], 172.4 [α-CONH]; ES-MS m/z 748.6 [M+1]+; C42H49N7O6 (%): C: 67.45, H: 6.60, N: 13.11. Found (%): C: 67.62, H: 6.74, N: 13.02.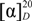 = +6.7 (c 0.9, MeOH); HPLC tR: 21.76 min; 1H-NMR (500 MHz, CDCl3) δ (ppm): 1.23 (m, 2H, γ-H), 1.34 (m, 2H, δ-H), 1.67 (m, 1H, β-H), 1.82 (m, 1H, β-H), 2.58 (t, 1H, J = 12.5 Hz, CH2-Ph), 2.87 (m, 1H, CH2-Ph), 2.90 (m, 1H, ε-H), 2.98 (m, 1H, 2-H), 3.05 (m, 1H, ε-H), 3.10 (m, 1H, 3-H), 3.18 (m, 1H, 6-H), 3.30 (m, 1H, CH2CO), 3.42 (m, 1H, CH2CO), 3.62 (m, 1H, 6-H), 3.95 (m, 1H, 2-CH), 4.05 (m, 1H, 3-H), 4.35 [m, 1H, CH2 (NHBn)], 4.50 [m, 2H, CH2 (NHBn) and α-H], 4.98 [m, 2H, CH2 (Z)], 5.02 (m, 1H, NHZ), 5.69 (m, 1H, 2-CHNH), 5.81 (m, 1H, 4-H), 6.94 (t, 1H, J = 7.5 Hz, NHBn), 6.98–7.14 (m, 20H, Ar), 7.98 (m, 1H, NHPh), 8.09 (m, 1H, α-NH); 13C-NMR (125 MHz, CDCl3) δ (ppm): 22.2 [Cγ], 29.1 [Cδ], 32.6 [Cβ], 35.7 [C3], 37.7 [CH2-Ph], 40.2 [Cε], 43.8 [CH2 (NHBn)], 52.0 [C6 and C2-CH], 52.8 [Cα], 57.8 [CH2CO], 58.7 [C2], 66.5 [CH2 (Z)], 118.4, 122.3, 127.7, 127.8, 128.1, 128.5, 128.9, 129.0, 129.3 [20CH (Ar)], 135.6 [C (Ph)], 136.5 [C (Z)], 137.1 [C (NHBn)], 139.6 [C (NHPh)], 155.6 [CO (Z)], 156.7 [CO (Urea)], 168.6 [C5], 169.9 [CO], 172.0 [α-CONH]; ES-MS m/z 748.7 [M+1]+; C42H49N7O6 (%): C: 67.45, H: 6.60, N: 13.11. Found (%): C: 67.31, H: 6.81, N: 13.25.
= +6.7 (c 0.9, MeOH); HPLC tR: 21.76 min; 1H-NMR (500 MHz, CDCl3) δ (ppm): 1.23 (m, 2H, γ-H), 1.34 (m, 2H, δ-H), 1.67 (m, 1H, β-H), 1.82 (m, 1H, β-H), 2.58 (t, 1H, J = 12.5 Hz, CH2-Ph), 2.87 (m, 1H, CH2-Ph), 2.90 (m, 1H, ε-H), 2.98 (m, 1H, 2-H), 3.05 (m, 1H, ε-H), 3.10 (m, 1H, 3-H), 3.18 (m, 1H, 6-H), 3.30 (m, 1H, CH2CO), 3.42 (m, 1H, CH2CO), 3.62 (m, 1H, 6-H), 3.95 (m, 1H, 2-CH), 4.05 (m, 1H, 3-H), 4.35 [m, 1H, CH2 (NHBn)], 4.50 [m, 2H, CH2 (NHBn) and α-H], 4.98 [m, 2H, CH2 (Z)], 5.02 (m, 1H, NHZ), 5.69 (m, 1H, 2-CHNH), 5.81 (m, 1H, 4-H), 6.94 (t, 1H, J = 7.5 Hz, NHBn), 6.98–7.14 (m, 20H, Ar), 7.98 (m, 1H, NHPh), 8.09 (m, 1H, α-NH); 13C-NMR (125 MHz, CDCl3) δ (ppm): 22.2 [Cγ], 29.1 [Cδ], 32.6 [Cβ], 35.7 [C3], 37.7 [CH2-Ph], 40.2 [Cε], 43.8 [CH2 (NHBn)], 52.0 [C6 and C2-CH], 52.8 [Cα], 57.8 [CH2CO], 58.7 [C2], 66.5 [CH2 (Z)], 118.4, 122.3, 127.7, 127.8, 128.1, 128.5, 128.9, 129.0, 129.3 [20CH (Ar)], 135.6 [C (Ph)], 136.5 [C (Z)], 137.1 [C (NHBn)], 139.6 [C (NHPh)], 155.6 [CO (Z)], 156.7 [CO (Urea)], 168.6 [C5], 169.9 [CO], 172.0 [α-CONH]; ES-MS m/z 748.7 [M+1]+; C42H49N7O6 (%): C: 67.45, H: 6.60, N: 13.11. Found (%): C: 67.31, H: 6.81, N: 13.25.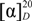 = −3.8 (c 1.2, MeOH); HPLC tR: 20.79 min; 1H-NMR (400 MHz, CDCl3) δ (ppm): 1.40 (m, 2H, γ-H), 1.55 (m, 1H, β-H), 1.70 (m, 1H, β-H), 2.57 (dd, 1H, J = 11 and 13.5, CH2-Ph), 2.86 (dd, 1H, J = 3.5 and 13.5, CH2-Ph), 3.00 (m, 1H, 2-H), 3.04 (m, 1H, δ-H), 3.09 (m, 1H, 3-H), 3.15 (m, 1H, 3-H), 3.25 (m, 2H, CH2CO), 3.32 (m, 1H, δ-H), 3.50 (d, 1H, J = 18 Hz, 6-H), 3.85 (m, 1H, 3-H), 3.94 (m, 1H, 2-CH), 4.17 [dd, 1H, J = 5 and 15 Hz, CH2 (NHBn)], 4.23 [dd, 1H, J = 6 and 15 Hz, CH2 (NHBn)], 4.27 [dd, 1H, J = 5 and 15 Hz, CH2 (NHBn, Urea)], 4.36 [dd, 1H, J = 5.5 and 15 Hz, CH2 (NHBn, Urea)], 4.66 (m, 1H, α-H), 4.79 [d, 1H, J = 13 Hz, CH2 (Z)], 4.89 [d, 1H, J = 13 Hz, CH2 (Z)], 5.30 (t, 1H, J = 6 Hz, NHZ), 5.75 (m, 1H, 2-CHNH), 5.90 (m, 1H, 4-H), 6.07 [t, 1H, J = 5.5 Hz, NHBn (Urea)], 7.08–7.39 (m, 20H, Ar), 7.50 (t, 1H, J = 5.5 Hz, NHBn), 7.99 [d, 1H, J = 8.5, α-NH); 13C-NMR (100 MHz, CDCl3) δ (ppm): 26.1 [Cγ], 30.6 [Cβ], 35.8 [C3], 37.5 [CH2-Ph], 39.3 [Cδ], 43.5 [CH2 (NHBn)], 44.4 [CH2 (NHBn, Urea)], 51.0 [Cα], 51.8 [C6], 52.5 [C2-CH], 57.5 [CH2CO], 58.3 [C2], 66.5 [CH2 (Z)], 127.0, 127.4, 127.5, 127.8, 128.1, 128.4, 128.5, 128.7, 129.0, 129.2 [20CH (Ar)], 135.8 [C (Ph)], 136.5 [C (Z)], 137.5 [C (NHBn)], 139.6 [C (NHBn, Urea)], 157.2 [CO (Z)], 158.5 [CO (Urea)], 168.9 [C5], 170.0 [CO], 172.2 [α-CONH]; ES-MS m/z 748.6 [M+1]+; C42H49N7O6 (%): C: 67.45, H: 6.60, N: 13.11. Found (%): C: 67.28, H: 6.82, N: 13.20.
= −3.8 (c 1.2, MeOH); HPLC tR: 20.79 min; 1H-NMR (400 MHz, CDCl3) δ (ppm): 1.40 (m, 2H, γ-H), 1.55 (m, 1H, β-H), 1.70 (m, 1H, β-H), 2.57 (dd, 1H, J = 11 and 13.5, CH2-Ph), 2.86 (dd, 1H, J = 3.5 and 13.5, CH2-Ph), 3.00 (m, 1H, 2-H), 3.04 (m, 1H, δ-H), 3.09 (m, 1H, 3-H), 3.15 (m, 1H, 3-H), 3.25 (m, 2H, CH2CO), 3.32 (m, 1H, δ-H), 3.50 (d, 1H, J = 18 Hz, 6-H), 3.85 (m, 1H, 3-H), 3.94 (m, 1H, 2-CH), 4.17 [dd, 1H, J = 5 and 15 Hz, CH2 (NHBn)], 4.23 [dd, 1H, J = 6 and 15 Hz, CH2 (NHBn)], 4.27 [dd, 1H, J = 5 and 15 Hz, CH2 (NHBn, Urea)], 4.36 [dd, 1H, J = 5.5 and 15 Hz, CH2 (NHBn, Urea)], 4.66 (m, 1H, α-H), 4.79 [d, 1H, J = 13 Hz, CH2 (Z)], 4.89 [d, 1H, J = 13 Hz, CH2 (Z)], 5.30 (t, 1H, J = 6 Hz, NHZ), 5.75 (m, 1H, 2-CHNH), 5.90 (m, 1H, 4-H), 6.07 [t, 1H, J = 5.5 Hz, NHBn (Urea)], 7.08–7.39 (m, 20H, Ar), 7.50 (t, 1H, J = 5.5 Hz, NHBn), 7.99 [d, 1H, J = 8.5, α-NH); 13C-NMR (100 MHz, CDCl3) δ (ppm): 26.1 [Cγ], 30.6 [Cβ], 35.8 [C3], 37.5 [CH2-Ph], 39.3 [Cδ], 43.5 [CH2 (NHBn)], 44.4 [CH2 (NHBn, Urea)], 51.0 [Cα], 51.8 [C6], 52.5 [C2-CH], 57.5 [CH2CO], 58.3 [C2], 66.5 [CH2 (Z)], 127.0, 127.4, 127.5, 127.8, 128.1, 128.4, 128.5, 128.7, 129.0, 129.2 [20CH (Ar)], 135.8 [C (Ph)], 136.5 [C (Z)], 137.5 [C (NHBn)], 139.6 [C (NHBn, Urea)], 157.2 [CO (Z)], 158.5 [CO (Urea)], 168.9 [C5], 170.0 [CO], 172.2 [α-CONH]; ES-MS m/z 748.6 [M+1]+; C42H49N7O6 (%): C: 67.45, H: 6.60, N: 13.11. Found (%): C: 67.28, H: 6.82, N: 13.20.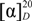 = −8.2 (c 1.0, MeOH); HPLC tR: 20.09 min; 1H-NMR (500 MHz, (CD3)2CO) δ (ppm): 1.55 (m, 2H, γ-H), 1.71 (m, 1H, β-H), 1.87 (m, 1H, β-H), 2.80 (dd, 1H, J = 10 and 14, CH2-Ph), 2.98 (dd, 1H, J = 6 and 13, 2-H), 3.06 (dd, 1H, J = 4 and 14, CH2-Ph), 3.12 (m, 1H, δ-H), 3.16 (d, 1H, J = 16.5 Hz, 6-H), 3.35 (s, 2H, CH2CO), 3.40 (d, 1H, J = 16.5 Hz, 6-H), 3.42 (m, 1H, 3-H), 3.51 (ddd, 1H, J = 4, 13 and 15 Hz, 3-H), 4.16 [dd, 1H, J = 6 and 15 Hz, CH2 (NHBn, Urea)], 4.27 [dd, 1H, J = 5.5 and 15 Hz, CH2 (NHBn, Urea)], 4.38 (m, 1H, 2-CH), 4.33 [d, 2H, J = 6 Hz, CH2 (NHBn)], 4.56 (dt, 1H, J = 5 and 9 Hz, α-H), 5.00 [d, 1H, J = 4.5 Hz, CH2 (Z)], 4.89 [d, 1H, J = 13 Hz, CH2 (Z)], 6.00 [t, 1H, J = 6 Hz, NHBn (Urea)], 5.89 (d, 1H, J = 9 Hz, 2-CHNH), 5.90 (m, 1H, 4-H), 6.46 (t, 1H, J = 5.5 Hz, NHZ), 7.07–7.35 (m, 21H, Ar and 1-H), 8.03 (t, 1H, J = 6 Hz, NHBn), 8.14 [d, 1H, J = 8.5, α-NH); 13C-NMR (125 MHz, (CD3)2CO) δ (ppm): 27.8 [Cγ], 31.5 [Cβ], 39.4 [CH2-Ph], 40.9 [C3], 41.6 [Cδ], 44.1 [CH2 (NHBn)], 44.8 [CH2 (NHBn, Urea)], 53.5 [C2-CH], 54.1 [Cα], 55.9 [C6], 59.6 [CH2CO], 62.5 [C2], 67.1 [CH2 (Z)], 127.6, 128.0, 128.3, 128.4, 128.8, 129.2, 129.3, 129.7, 129.8, 130.9 [20CH (Ar)], 139.2 [C (Z)], 140.5 [C (Ph)], 140.9 [C (NHBn)], 142.3 [C (NHBn, Urea)], 158.1 [CO (Z)], 159.8 [CO (Urea)], 170.7 [C5], 171.5 [CO], 173.5 [α-CONH]; ES-MS m/z 748.4 [M+1]+; C42H49N7O6 (%): C: 67.45, H: 6.60, N: 13.11. Found (%): C: 67.60, H: 6.85, N: 13.01.
= −8.2 (c 1.0, MeOH); HPLC tR: 20.09 min; 1H-NMR (500 MHz, (CD3)2CO) δ (ppm): 1.55 (m, 2H, γ-H), 1.71 (m, 1H, β-H), 1.87 (m, 1H, β-H), 2.80 (dd, 1H, J = 10 and 14, CH2-Ph), 2.98 (dd, 1H, J = 6 and 13, 2-H), 3.06 (dd, 1H, J = 4 and 14, CH2-Ph), 3.12 (m, 1H, δ-H), 3.16 (d, 1H, J = 16.5 Hz, 6-H), 3.35 (s, 2H, CH2CO), 3.40 (d, 1H, J = 16.5 Hz, 6-H), 3.42 (m, 1H, 3-H), 3.51 (ddd, 1H, J = 4, 13 and 15 Hz, 3-H), 4.16 [dd, 1H, J = 6 and 15 Hz, CH2 (NHBn, Urea)], 4.27 [dd, 1H, J = 5.5 and 15 Hz, CH2 (NHBn, Urea)], 4.38 (m, 1H, 2-CH), 4.33 [d, 2H, J = 6 Hz, CH2 (NHBn)], 4.56 (dt, 1H, J = 5 and 9 Hz, α-H), 5.00 [d, 1H, J = 4.5 Hz, CH2 (Z)], 4.89 [d, 1H, J = 13 Hz, CH2 (Z)], 6.00 [t, 1H, J = 6 Hz, NHBn (Urea)], 5.89 (d, 1H, J = 9 Hz, 2-CHNH), 5.90 (m, 1H, 4-H), 6.46 (t, 1H, J = 5.5 Hz, NHZ), 7.07–7.35 (m, 21H, Ar and 1-H), 8.03 (t, 1H, J = 6 Hz, NHBn), 8.14 [d, 1H, J = 8.5, α-NH); 13C-NMR (125 MHz, (CD3)2CO) δ (ppm): 27.8 [Cγ], 31.5 [Cβ], 39.4 [CH2-Ph], 40.9 [C3], 41.6 [Cδ], 44.1 [CH2 (NHBn)], 44.8 [CH2 (NHBn, Urea)], 53.5 [C2-CH], 54.1 [Cα], 55.9 [C6], 59.6 [CH2CO], 62.5 [C2], 67.1 [CH2 (Z)], 127.6, 128.0, 128.3, 128.4, 128.8, 129.2, 129.3, 129.7, 129.8, 130.9 [20CH (Ar)], 139.2 [C (Z)], 140.5 [C (Ph)], 140.9 [C (NHBn)], 142.3 [C (NHBn, Urea)], 158.1 [CO (Z)], 159.8 [CO (Urea)], 170.7 [C5], 171.5 [CO], 173.5 [α-CONH]; ES-MS m/z 748.4 [M+1]+; C42H49N7O6 (%): C: 67.45, H: 6.60, N: 13.11. Found (%): C: 67.60, H: 6.85, N: 13.01.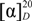 = −5.6 (c 0.8, MeOH); HPLC tR: 21.05 min; 1H-NMR (400 MHz, CDCl3) δ (ppm): 1.25 (m, 2H, γ-H), 1.42 (m, 2H, δ-H), 1.59 (dt, 1H, J = 7.5 and 14 Hz, β-H), 1.75 (m, 1H, β-H), 2.54 (dd, 1H, J = 11 and 13 Hz, CH2-Ph), 2.86 (dd, 1H, J = 2.5 and 13 Hz, CH2-Ph), 3.01 (m, 1H, 2-H), 3.04 (m, 1H, ε-H), 3.10 (m, 1H, ε-H), 3.12 (m, 1H, 6-H), 3.16 (m, 1H, 3-H), 3.22 (m, 1H, CH2CO), 3.28 (m, 1H, CH2CO), 3.53 (d, 1H, J = 18 Hz, 6-H), 3.86 (m, 1H, 3-H), 3.94 (m, 1H, 2-CH), 4.24 [d, 2H, J = 5.5 Hz, CH2 (NHBn)], 4.27 [d, 1H, J = 4.5 Hz, CH2 (NHBn, Urea)], 4.37 [d, 1H, J = 4.5 Hz, CH2 (NHBn, Urea)], 4.43 (m, 1H, α-H), 5.00 [d, 2H, J = 7 Hz, CH2 (Z)], 5.24 (t, 1H, J = 5 Hz, NHZ), 5.66 (m, 1H, 2-CHNH), 5.77 (m, 1H, 4-H), 6.04 [m, 1H, (NHBn, Urea)], 7.09–7.41 (m, 21H, Ar and NHBn), 7.97 (d, 1H, J = 8 Hz, α-NH); 13C-NMR (100 MHz, CDCl3) δ (ppm): 22.4 [Cγ], 29.2 [Cδ], 32.5 [Cβ], 35.9 [C3], 37.5 [CH2-Ph], 40.4 [Cε], 43.6 [CH2 (NHBn)], 44.2 [CH2 (NHBn, Urea)], 51.7 [C6], 52.4 [C2-CH], 52.6 [Cα], 57.7 [CH2CO], 58.5 [C2], 66.6 [CH2 (Z)], 127.1, 127.5, 127.6, 127.7, 127.9, 128.1, 128.5, 128.7, 129.0, 129.2 [20CH (Ar)], 135.8 [C (Ph)], 136.5 [C (Z)], 137.4 [C (NHBn)], 139.6 [C (NHBn, Urea)], 156.7 [CO (Z)], 158.3 [CO (Urea)], 168.6 [C5], 169.9 [CO], 172.0 [α-CONH]; ES-MS m/z 763.2 [M+1]+; C43H51N7O6 (%): C: 67.79, H: 6.75, N: 12.87. Found (%): C: 67.60, H: 7.01, N: 12.69.
= −5.6 (c 0.8, MeOH); HPLC tR: 21.05 min; 1H-NMR (400 MHz, CDCl3) δ (ppm): 1.25 (m, 2H, γ-H), 1.42 (m, 2H, δ-H), 1.59 (dt, 1H, J = 7.5 and 14 Hz, β-H), 1.75 (m, 1H, β-H), 2.54 (dd, 1H, J = 11 and 13 Hz, CH2-Ph), 2.86 (dd, 1H, J = 2.5 and 13 Hz, CH2-Ph), 3.01 (m, 1H, 2-H), 3.04 (m, 1H, ε-H), 3.10 (m, 1H, ε-H), 3.12 (m, 1H, 6-H), 3.16 (m, 1H, 3-H), 3.22 (m, 1H, CH2CO), 3.28 (m, 1H, CH2CO), 3.53 (d, 1H, J = 18 Hz, 6-H), 3.86 (m, 1H, 3-H), 3.94 (m, 1H, 2-CH), 4.24 [d, 2H, J = 5.5 Hz, CH2 (NHBn)], 4.27 [d, 1H, J = 4.5 Hz, CH2 (NHBn, Urea)], 4.37 [d, 1H, J = 4.5 Hz, CH2 (NHBn, Urea)], 4.43 (m, 1H, α-H), 5.00 [d, 2H, J = 7 Hz, CH2 (Z)], 5.24 (t, 1H, J = 5 Hz, NHZ), 5.66 (m, 1H, 2-CHNH), 5.77 (m, 1H, 4-H), 6.04 [m, 1H, (NHBn, Urea)], 7.09–7.41 (m, 21H, Ar and NHBn), 7.97 (d, 1H, J = 8 Hz, α-NH); 13C-NMR (100 MHz, CDCl3) δ (ppm): 22.4 [Cγ], 29.2 [Cδ], 32.5 [Cβ], 35.9 [C3], 37.5 [CH2-Ph], 40.4 [Cε], 43.6 [CH2 (NHBn)], 44.2 [CH2 (NHBn, Urea)], 51.7 [C6], 52.4 [C2-CH], 52.6 [Cα], 57.7 [CH2CO], 58.5 [C2], 66.6 [CH2 (Z)], 127.1, 127.5, 127.6, 127.7, 127.9, 128.1, 128.5, 128.7, 129.0, 129.2 [20CH (Ar)], 135.8 [C (Ph)], 136.5 [C (Z)], 137.4 [C (NHBn)], 139.6 [C (NHBn, Urea)], 156.7 [CO (Z)], 158.3 [CO (Urea)], 168.6 [C5], 169.9 [CO], 172.0 [α-CONH]; ES-MS m/z 763.2 [M+1]+; C43H51N7O6 (%): C: 67.79, H: 6.75, N: 12.87. Found (%): C: 67.60, H: 7.01, N: 12.69.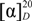 = −11.2 (c 0.9, MeOH); HPLC tR: 20.47 min; 1H-NMR (500 MHz, (CD3)2CO) δ (ppm): 1.30 (m, 2H, δ-H), 1.56 (m, 2H, γ-H), 1.68 (m, 1H, β-H), 1.83 (m, 1H, β-H), 2.82 (m, 1H, CH2-Ph), 3.02 (m, 1H, 2-H), 3.05 (m, 1H, CH2-Ph), 3.18 (m, 3H, ε-H and 6-H), 3.37 (m, 2H, CH2CO), 3.16 (m, 1H, 3-H), 3.40 (m, 1H, 6-H), 3.55 (m, 2H, 3-H), 4.15 [m, 1H, CH2 (NHBn, Urea)], 4.25 [m, 1H, CH2 (NHBn, Urea)], 4.28 [m, 2H, CH2 (NHBn)], 4.40 (m, 1H, 2-CH), 4.51 (m, 1H, α-H), 5.00 [m, 2H, CH2 (Z)], 5.98 (d, 1H, J = 7 Hz, 2-CHNH), 6.04 [m, 1H, (NHBn, Urea)], 6.45 (m, 1H, NHZ), 7.03 (m, 1H, 4-H), 7.08–7.41 (m, 20H, Ar), 8.04 (m, 1H, NHBn), 8.15 (d, 1H, J = 8.5 Hz, α-NH); 13C-NMR (125 MHz, (CD3)2CO) δ (ppm): 26.2 [Cγ], 29.7 [Cδ], 29.9 [Cβ], 37.9 [CH2-Ph], 39.2 [C3], 39.9 [Cε], 42.5 [CH2 (NHBn)], 43.2 [CH2 (NHBn, Urea)], 52.0 [C2-CH], 52.6 [Cα], 54.4 [C6], 57.6 [CH2CO], 61.0 [C2], 65.5 [CH2 (Z)], 126.1, 126.5, 126.6, 126.8, 127.3, 127.7, 127.8, 128.3, 128.4, 129.3, 129.6 [20CH (Ar)], 137.5 [C (Z)], 138.9 [C (Ph)], 139.4 [C (NHBn)], 140.7 [C (NHBn, Urea)], 156.5 [CO (Z)], 158.3 [CO (Urea)], 169.0 [C5], 169.7 [CO], 171.9 [α-CONH]; ES-MS m/z 763.3 [M+1]+; C43H51N7O6 (%): C: 67.79, H: 6.75, N: 12.87. Found (%): C: 67.96, H: 6.93, N: 12.70.
= −11.2 (c 0.9, MeOH); HPLC tR: 20.47 min; 1H-NMR (500 MHz, (CD3)2CO) δ (ppm): 1.30 (m, 2H, δ-H), 1.56 (m, 2H, γ-H), 1.68 (m, 1H, β-H), 1.83 (m, 1H, β-H), 2.82 (m, 1H, CH2-Ph), 3.02 (m, 1H, 2-H), 3.05 (m, 1H, CH2-Ph), 3.18 (m, 3H, ε-H and 6-H), 3.37 (m, 2H, CH2CO), 3.16 (m, 1H, 3-H), 3.40 (m, 1H, 6-H), 3.55 (m, 2H, 3-H), 4.15 [m, 1H, CH2 (NHBn, Urea)], 4.25 [m, 1H, CH2 (NHBn, Urea)], 4.28 [m, 2H, CH2 (NHBn)], 4.40 (m, 1H, 2-CH), 4.51 (m, 1H, α-H), 5.00 [m, 2H, CH2 (Z)], 5.98 (d, 1H, J = 7 Hz, 2-CHNH), 6.04 [m, 1H, (NHBn, Urea)], 6.45 (m, 1H, NHZ), 7.03 (m, 1H, 4-H), 7.08–7.41 (m, 20H, Ar), 8.04 (m, 1H, NHBn), 8.15 (d, 1H, J = 8.5 Hz, α-NH); 13C-NMR (125 MHz, (CD3)2CO) δ (ppm): 26.2 [Cγ], 29.7 [Cδ], 29.9 [Cβ], 37.9 [CH2-Ph], 39.2 [C3], 39.9 [Cε], 42.5 [CH2 (NHBn)], 43.2 [CH2 (NHBn, Urea)], 52.0 [C2-CH], 52.6 [Cα], 54.4 [C6], 57.6 [CH2CO], 61.0 [C2], 65.5 [CH2 (Z)], 126.1, 126.5, 126.6, 126.8, 127.3, 127.7, 127.8, 128.3, 128.4, 129.3, 129.6 [20CH (Ar)], 137.5 [C (Z)], 138.9 [C (Ph)], 139.4 [C (NHBn)], 140.7 [C (NHBn, Urea)], 156.5 [CO (Z)], 158.3 [CO (Urea)], 169.0 [C5], 169.7 [CO], 171.9 [α-CONH]; ES-MS m/z 763.3 [M+1]+; C43H51N7O6 (%): C: 67.79, H: 6.75, N: 12.87. Found (%): C: 67.96, H: 6.93, N: 12.70.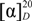 = +1.7 (c 0.7, MeOH); HPLC tR: 14.65 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.58 (m, 2H, γ-H), 1.63 (m, 1H, β-H), 1.80 (m, 1H, β-H), 2.62 (m, 1H, CH2-Ph), 2.63 (m, 1H, 2-H), 2.73 (m, 1H, CH2-Ph), 2.75 (m, 2H, δ-H), 3.05 (m, 1H, 3-H), 3.38 (m, 1H, 6-H), 3.40 (m, 2H, CH2CO and 3-H), 3.50 (m, 1H, CH2CO), 3.55 (m, 1H, 6-H), 4.05 (m, 1H, 2-CH), 4.24 [dd, 1H, J = 6 and 15 Hz, CH2 (NHBn)], 4.31 [dd, 1H, J = 6 and 15 Hz, CH2 (NHBn)], 4.39 (dd, 1H, J = 5 and 8 Hz, α-H), 6.58 (m, 1H, 2-CHNH), 6.80–7.35 (m, 15H, Ar), 7.86 (m, 4H, 4-H and NH2·HCl), ], 8.20 (m, 1H, α-NH), 8.65 (m, 1H, NHBn), 8.74 [m, 1H, NHPh]; 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 23.5 [Cγ], 29.2 [Cβ], 38.2 [Cδ], 42.0 [CH2 (NHBn)], 51.5 [Cα], 117.6, 121.1, 126.1, 126.8, 127.1, 128.1, 128.3, 128.6, 129.3 [15CH (Ar)], 139.1 [C (NHBn)], 140.2 [C (NHPh)], 155.1 [CO (Urea)], 171.0 [α-CONH]; ES-MS m/z [M]+ calculated for C33H41N7O4: 600.2; found: 600.5.
= +1.7 (c 0.7, MeOH); HPLC tR: 14.65 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.58 (m, 2H, γ-H), 1.63 (m, 1H, β-H), 1.80 (m, 1H, β-H), 2.62 (m, 1H, CH2-Ph), 2.63 (m, 1H, 2-H), 2.73 (m, 1H, CH2-Ph), 2.75 (m, 2H, δ-H), 3.05 (m, 1H, 3-H), 3.38 (m, 1H, 6-H), 3.40 (m, 2H, CH2CO and 3-H), 3.50 (m, 1H, CH2CO), 3.55 (m, 1H, 6-H), 4.05 (m, 1H, 2-CH), 4.24 [dd, 1H, J = 6 and 15 Hz, CH2 (NHBn)], 4.31 [dd, 1H, J = 6 and 15 Hz, CH2 (NHBn)], 4.39 (dd, 1H, J = 5 and 8 Hz, α-H), 6.58 (m, 1H, 2-CHNH), 6.80–7.35 (m, 15H, Ar), 7.86 (m, 4H, 4-H and NH2·HCl), ], 8.20 (m, 1H, α-NH), 8.65 (m, 1H, NHBn), 8.74 [m, 1H, NHPh]; 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 23.5 [Cγ], 29.2 [Cβ], 38.2 [Cδ], 42.0 [CH2 (NHBn)], 51.5 [Cα], 117.6, 121.1, 126.1, 126.8, 127.1, 128.1, 128.3, 128.6, 129.3 [15CH (Ar)], 139.1 [C (NHBn)], 140.2 [C (NHPh)], 155.1 [CO (Urea)], 171.0 [α-CONH]; ES-MS m/z [M]+ calculated for C33H41N7O4: 600.2; found: 600.5.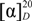 = −1.6 (c 1.1, MeOH); HPLC tR: 15.07 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.48 (m, 2H, γ-H), 1.58 (m, 1H, β-H), 1.74 (m, 1H, β-H), 2.62 (m, 2H, δ-H), 2.79 (m, 1H, CH2-Ph), 2.83 (m, 1H, 2-H), 2.89 (m, 1H, CH2-Ph), 3.00 (m, 1H, 3-H), 3.35 (m, 1H, CH2CO), 3.40 (m, 1H, 6-H), 3.42 (m, 1H, 3-H), 3.50 (m, 1H, CH2CO), 3.65 (m, 1H, 6-H), 4.04 (m, 1H, 2-CH), 4.25 [d, 2H, J = 6, CH2 (NHBn)], 4.31 (dd, 1H, J = 5 and 8 Hz, α-H), 6.70 (m, 1H, 2-CHNH), 6.79–7.49 (m, 15H, Ar), 7.81 (m, 4H, 4-H and NH2·HCl), ], 8.21 (m, 1H, α-NH), 8.65 (t, 1H, J = 6 Hz, NHBn), 8.92 [m, 1H, NHPh]; 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 23.4 [Cγ], 28.9 [Cβ], 38.1 [Cδ], 42.0 [CH2 (NHBn)], 51.5 [Cα], 117.6, 121.1, 126.4, 126.7, 127.0, 128.2, 128.4, 128.6, 129.0 [15CH (Ar)], 137.0 [C (Ph)], 139.1 [C (NHBn)], 140.3 [C (NHPh)], 155.3 [CO (Urea)], 170.9 [α-CONH]; ES-MS m/z [M]+ calculated for C33H41N7O4: 600.2; found: 600.5.
= −1.6 (c 1.1, MeOH); HPLC tR: 15.07 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.48 (m, 2H, γ-H), 1.58 (m, 1H, β-H), 1.74 (m, 1H, β-H), 2.62 (m, 2H, δ-H), 2.79 (m, 1H, CH2-Ph), 2.83 (m, 1H, 2-H), 2.89 (m, 1H, CH2-Ph), 3.00 (m, 1H, 3-H), 3.35 (m, 1H, CH2CO), 3.40 (m, 1H, 6-H), 3.42 (m, 1H, 3-H), 3.50 (m, 1H, CH2CO), 3.65 (m, 1H, 6-H), 4.04 (m, 1H, 2-CH), 4.25 [d, 2H, J = 6, CH2 (NHBn)], 4.31 (dd, 1H, J = 5 and 8 Hz, α-H), 6.70 (m, 1H, 2-CHNH), 6.79–7.49 (m, 15H, Ar), 7.81 (m, 4H, 4-H and NH2·HCl), ], 8.21 (m, 1H, α-NH), 8.65 (t, 1H, J = 6 Hz, NHBn), 8.92 [m, 1H, NHPh]; 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 23.4 [Cγ], 28.9 [Cβ], 38.1 [Cδ], 42.0 [CH2 (NHBn)], 51.5 [Cα], 117.6, 121.1, 126.4, 126.7, 127.0, 128.2, 128.4, 128.6, 129.0 [15CH (Ar)], 137.0 [C (Ph)], 139.1 [C (NHBn)], 140.3 [C (NHPh)], 155.3 [CO (Urea)], 170.9 [α-CONH]; ES-MS m/z [M]+ calculated for C33H41N7O4: 600.2; found: 600.5.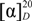 = −3.0 (c 1.7, MeOH); HPLC tR: 14.97 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.27 (m, 2H, γ-H), 1.52 (m, 2H, δ-H), 1.58 (m, 1H, β-H), 1.70 (m, 1H, β-H), 2.61 (m, 1H, CH2-Ph), 2.72 (m, 1H, ε-H), 2.84 (m, 1H, 2-H), 2.92 (m, 1H, ε-H), 2.95 (m, 1H, CH2-Ph), 3.00 (m, 2H, 3-H), 3.38 (m, 1H, 3-H), 3.40 (m, 1H, CH2CO), 3.42 (m, 1H, 6-H), 3.50 (m, 1H, CH2CO), 3.54 (m, 1H, 3-H), 4.22 [m, 1H, CH2 (NHBn)], 4.23 (m, 1H, 2-CH), 4.31 [m, 1H, CH2 (NHBn)], 4.32 (m, 1H, α-H), 6.80–7.37 (m, 16H, Ar and 2-CHNH), 7.88 (m, 4H, NH2·HCl and 4-H), 8.17 (m, 1H, α-NH), 8.61 (t, 1H, J = 6 Hz, NHBn), 8.90 (m, 1H, NHPh); 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 22.2 [Cγ], 26.5 [Cδ], 30.7 [Cβ], 38.4 [Cε], 42.0 [CH2 (NHBn)], 52.5 [Cα], 117.7, 121.3, 126.3, 126.7, 127.0, 128.2, 128.3, 128.5, 129.2 [15CH (Ar)], 138.1 [C (Ph)], 139.2 [C (NHBn)], 140.0 [C (NHPh)], 155.3 [CO (Urea)], 171.3 [α-CONH]; ES-MS m/z [M]+ calculated for C34H43N7O4: 614.2; found: 614.5.
= −3.0 (c 1.7, MeOH); HPLC tR: 14.97 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.27 (m, 2H, γ-H), 1.52 (m, 2H, δ-H), 1.58 (m, 1H, β-H), 1.70 (m, 1H, β-H), 2.61 (m, 1H, CH2-Ph), 2.72 (m, 1H, ε-H), 2.84 (m, 1H, 2-H), 2.92 (m, 1H, ε-H), 2.95 (m, 1H, CH2-Ph), 3.00 (m, 2H, 3-H), 3.38 (m, 1H, 3-H), 3.40 (m, 1H, CH2CO), 3.42 (m, 1H, 6-H), 3.50 (m, 1H, CH2CO), 3.54 (m, 1H, 3-H), 4.22 [m, 1H, CH2 (NHBn)], 4.23 (m, 1H, 2-CH), 4.31 [m, 1H, CH2 (NHBn)], 4.32 (m, 1H, α-H), 6.80–7.37 (m, 16H, Ar and 2-CHNH), 7.88 (m, 4H, NH2·HCl and 4-H), 8.17 (m, 1H, α-NH), 8.61 (t, 1H, J = 6 Hz, NHBn), 8.90 (m, 1H, NHPh); 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 22.2 [Cγ], 26.5 [Cδ], 30.7 [Cβ], 38.4 [Cε], 42.0 [CH2 (NHBn)], 52.5 [Cα], 117.7, 121.3, 126.3, 126.7, 127.0, 128.2, 128.3, 128.5, 129.2 [15CH (Ar)], 138.1 [C (Ph)], 139.2 [C (NHBn)], 140.0 [C (NHPh)], 155.3 [CO (Urea)], 171.3 [α-CONH]; ES-MS m/z [M]+ calculated for C34H43N7O4: 614.2; found: 614.5.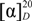 = −4.2 (c 0.4, MeOH); HPLC tR: 15.21 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.29 (m, 2H, γ-H), 1.50 (m, 2H, δ-H), 1.55 (m, 1H, β-H), 1.73 (m, 1H, β-H), 2.72 (m, 1H, CH2-Ph), 2.72 (m, 1H, ε-H), 2.74 (m, 2H, 3-H), 2.80 (m, 1H, ε-H), 2.89 (m, 1H, CH2-Ph), 2.90 (m, 1H, 2-H), 3.13 (m, 1H, CH2CO), 3.29 (m, 1H, 3-H), 3.33 (m, 1H, 6-H), 3.42 (m, 1H, CH2CO), 3.63 (m, 1H, 6-H), 4.10 (m, 1H, 2-CH), 4.18 [m, 1H, CH2 (NHBn)], 4.20 (m, 1H, α-H), 4.35 [m, 1H, CH2 (NHBn)], 6.55 (m, 1H, 2-CHNH), 6.80–7.40 (m, 16H, Ar), 7.69 (m, 4H, NH2·HCl and 4-H), 7.92 (d, 1H, J = 9 Hz, α-NH), 8.56 (m, 1H, NHBn), 8.70 (m, 1H, NHPh); 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 22.1 [Cγ], 26.8 [Cδ], 30.7 [Cβ], 38.5 [Cε], 42.0 [CH2 (NHBn)], 51.6 [Cα], 116.5, 118.0, 127.0, 127.2, 127.5, 128.7, 128.9, 129.1, 129.5 [15CH (Ar)], 138.2 [C (Ph)], 139.4 [C (NHBn)], 140.0 [C (NHPh)], 171.2 [α-CONH]; ES-MS m/z [M]+ calculated for C34H43N7O4: 614.2; found: 614.5.
= −4.2 (c 0.4, MeOH); HPLC tR: 15.21 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.29 (m, 2H, γ-H), 1.50 (m, 2H, δ-H), 1.55 (m, 1H, β-H), 1.73 (m, 1H, β-H), 2.72 (m, 1H, CH2-Ph), 2.72 (m, 1H, ε-H), 2.74 (m, 2H, 3-H), 2.80 (m, 1H, ε-H), 2.89 (m, 1H, CH2-Ph), 2.90 (m, 1H, 2-H), 3.13 (m, 1H, CH2CO), 3.29 (m, 1H, 3-H), 3.33 (m, 1H, 6-H), 3.42 (m, 1H, CH2CO), 3.63 (m, 1H, 6-H), 4.10 (m, 1H, 2-CH), 4.18 [m, 1H, CH2 (NHBn)], 4.20 (m, 1H, α-H), 4.35 [m, 1H, CH2 (NHBn)], 6.55 (m, 1H, 2-CHNH), 6.80–7.40 (m, 16H, Ar), 7.69 (m, 4H, NH2·HCl and 4-H), 7.92 (d, 1H, J = 9 Hz, α-NH), 8.56 (m, 1H, NHBn), 8.70 (m, 1H, NHPh); 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 22.1 [Cγ], 26.8 [Cδ], 30.7 [Cβ], 38.5 [Cε], 42.0 [CH2 (NHBn)], 51.6 [Cα], 116.5, 118.0, 127.0, 127.2, 127.5, 128.7, 128.9, 129.1, 129.5 [15CH (Ar)], 138.2 [C (Ph)], 139.4 [C (NHBn)], 140.0 [C (NHPh)], 171.2 [α-CONH]; ES-MS m/z [M]+ calculated for C34H43N7O4: 614.2; found: 614.5.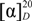 = −7.9 (c 1.3, MeOH); HPLC tR: 14.67 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.56 (m, 3H, γ-H and β-H), 1.78 (m, 1H, β-H), 2.60–3.16 (m, 6H, CH2-Ph, 2-H, δ-H and 3-H), 3.20–3.86 (m, 5H, 3-H, 6-H and CH2CO), 4.04 (m, 1H, 2-CH), 4.20 [m, 2H, CH2 (NHBn, Urea)], 4.26 [d, 2H, J = 6 Hz, CH2 (NHBn)], 4.34 (dd, 1H, J = 5 and 8 Hz, α-H), 6.53 (m, 1H, 2-CHNH), 6.74 [m, 1H, NHBn (Urea)], 7.18–7.33 (m, 15H, Ar), 7.92 (m, 4H, NH2·HCl and 4-H), 8.34 [m, 1H, α-NH), 8.70 (t, 1H, J = 6 Hz, NHBn); 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 23.8 [Cγ], 29.5 [Cβ], 39.5 [Cδ], 42.5 [CH2 (NHBn)], 43.4 [CH2 (NHBn, Urea)], 52.0 [Cα], 126.9, 127.0, 127.2, 127.4, 127.5, 128.6, 128.7, 128.9, 129.3 [15CH (Ar)], 137.5 [C (Ph)], 139.6 [C (NHBn)], 141.0 [C (NHBn, Urea)], 158.7 [CO (Urea)], 171.4 [α-CONH]; ES-MS m/z [M]+ calculated for C34H43N7O4: 614.2; found: 614.5.
= −7.9 (c 1.3, MeOH); HPLC tR: 14.67 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.56 (m, 3H, γ-H and β-H), 1.78 (m, 1H, β-H), 2.60–3.16 (m, 6H, CH2-Ph, 2-H, δ-H and 3-H), 3.20–3.86 (m, 5H, 3-H, 6-H and CH2CO), 4.04 (m, 1H, 2-CH), 4.20 [m, 2H, CH2 (NHBn, Urea)], 4.26 [d, 2H, J = 6 Hz, CH2 (NHBn)], 4.34 (dd, 1H, J = 5 and 8 Hz, α-H), 6.53 (m, 1H, 2-CHNH), 6.74 [m, 1H, NHBn (Urea)], 7.18–7.33 (m, 15H, Ar), 7.92 (m, 4H, NH2·HCl and 4-H), 8.34 [m, 1H, α-NH), 8.70 (t, 1H, J = 6 Hz, NHBn); 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 23.8 [Cγ], 29.5 [Cβ], 39.5 [Cδ], 42.5 [CH2 (NHBn)], 43.4 [CH2 (NHBn, Urea)], 52.0 [Cα], 126.9, 127.0, 127.2, 127.4, 127.5, 128.6, 128.7, 128.9, 129.3 [15CH (Ar)], 137.5 [C (Ph)], 139.6 [C (NHBn)], 141.0 [C (NHBn, Urea)], 158.7 [CO (Urea)], 171.4 [α-CONH]; ES-MS m/z [M]+ calculated for C34H43N7O4: 614.2; found: 614.5.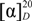 = −3.2 (c 1.2, MeOH); HPLC tR: 15.01 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.58 (m, 3H, γ-H), 1.60 (m, 1H, β-H) 1.88 (m, 1H, β-H), 2.52–3.97 (m, 6H, CH2-Ph, 2-H, δ-H and 3-H), 3.24–3.69 (m, 5H, 3-H, 6-H and CH2CO), 4.03 (m, 1H, 2-CH), 4.05 [m, 1H, CH2 (NHBn, Urea)], 4.20 [m, 1H, CH2 (NHBn, Urea)], 4.25 [m, 2H, CH2 (NHBn)], 4.39 (m, 1H, α-H), 6.48 (m, 1H, 2-CHNH), 6.96–7.36 [m, 16H, Ar and NHBn (Urea)], 7.90 (m, 4H, NH2·HCl and 4-H), 8.30 [m, 1H, α-NH), 8.70 (m, 1H, NHBn); 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 23.9 [Cγ], 29.6 [Cβ], 38.7 [Cδ], 42.5 [CH2 (NHBn)], 43.1 [CH2 (NHBn, Urea)], 52.1 [Cα], 126.5, 126.8, 127.0, 127.1, 127.5, 128.5, 128.7, 129.7 [15CH (Ar)], 139.6 [C (NHBn)], 141.0 [C (NHBn, Urea)], 158.5 [CO (Urea)], 171.3 [α-CONH]; ES-MS m/z [M]+ calculated for C34H43N7O4: 614.2; found: 614.5.
= −3.2 (c 1.2, MeOH); HPLC tR: 15.01 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.58 (m, 3H, γ-H), 1.60 (m, 1H, β-H) 1.88 (m, 1H, β-H), 2.52–3.97 (m, 6H, CH2-Ph, 2-H, δ-H and 3-H), 3.24–3.69 (m, 5H, 3-H, 6-H and CH2CO), 4.03 (m, 1H, 2-CH), 4.05 [m, 1H, CH2 (NHBn, Urea)], 4.20 [m, 1H, CH2 (NHBn, Urea)], 4.25 [m, 2H, CH2 (NHBn)], 4.39 (m, 1H, α-H), 6.48 (m, 1H, 2-CHNH), 6.96–7.36 [m, 16H, Ar and NHBn (Urea)], 7.90 (m, 4H, NH2·HCl and 4-H), 8.30 [m, 1H, α-NH), 8.70 (m, 1H, NHBn); 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 23.9 [Cγ], 29.6 [Cβ], 38.7 [Cδ], 42.5 [CH2 (NHBn)], 43.1 [CH2 (NHBn, Urea)], 52.1 [Cα], 126.5, 126.8, 127.0, 127.1, 127.5, 128.5, 128.7, 129.7 [15CH (Ar)], 139.6 [C (NHBn)], 141.0 [C (NHBn, Urea)], 158.5 [CO (Urea)], 171.3 [α-CONH]; ES-MS m/z [M]+ calculated for C34H43N7O4: 614.2; found: 614.5.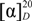 = −4.3 (c 0.6, MeOH); HPLC tR: 14.80 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.21 (m, 2H, γ-H), 1.49 (m, 2H, δ-H), 1.57 (m, 1H, β-H), 1.69 (m, 1H, β-H), 2.68 (m, 2H, ε-H), 2.75 (m, 1H, CH2-Ph), 2.80 (m, 1H, 2-H), 2.82 (m, 1H, CH2-Ph), 3.02 (m, 1H, 3-H), 3.25 (m, 1H, CH2CO), 3.37 (m, 1H, 6-H), 3.40 (m, 2H, CH2CO and 3-H), 3.60 (m, 1H, 6-H), 3.98 (m, 1H, 2-CH), 4.20 [m, 2H, CH2 (NHBn, Urea)], 4.24 (m, 1H, α-H), 4.25 [m, 2H, CH2 (NHBn)], 6.35 (m, 1H, 2-CHNH), 6.59 [m, 1H, (NHBn, Urea)], 7.08–7.35 (m, 15H, Ar), 7.82 (m, 4H, NH2·HCl and 4-H), 8.08 (m, 1H, α-NH), 8.57 (t, 1H, J = 6 Hz, NHBn); 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 22.1 [Cγ], 26.5 [Cδ], 31.4 [Cβ], 38.5 [Cε], 42.0 [CH2 (NHBn)], 42.9 [CH2 (NHBn, Urea)], 52.0 [Cα], 126.4, 126.5, 126.7, 126.9, 127.0, 128.2, 128.4, 129.1 [15CH (Ar)], 137.3 [C (Ph)], 139.3 [C (NHBn)], 140.7 [C (NHBn, Urea)], 158.0 [CO (Urea)], 171.2 [α-CONH]; ES-MS m/z [M]+ calculated for C35H45N7O4: 628.2; found: 628.5.
= −4.3 (c 0.6, MeOH); HPLC tR: 14.80 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.21 (m, 2H, γ-H), 1.49 (m, 2H, δ-H), 1.57 (m, 1H, β-H), 1.69 (m, 1H, β-H), 2.68 (m, 2H, ε-H), 2.75 (m, 1H, CH2-Ph), 2.80 (m, 1H, 2-H), 2.82 (m, 1H, CH2-Ph), 3.02 (m, 1H, 3-H), 3.25 (m, 1H, CH2CO), 3.37 (m, 1H, 6-H), 3.40 (m, 2H, CH2CO and 3-H), 3.60 (m, 1H, 6-H), 3.98 (m, 1H, 2-CH), 4.20 [m, 2H, CH2 (NHBn, Urea)], 4.24 (m, 1H, α-H), 4.25 [m, 2H, CH2 (NHBn)], 6.35 (m, 1H, 2-CHNH), 6.59 [m, 1H, (NHBn, Urea)], 7.08–7.35 (m, 15H, Ar), 7.82 (m, 4H, NH2·HCl and 4-H), 8.08 (m, 1H, α-NH), 8.57 (t, 1H, J = 6 Hz, NHBn); 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 22.1 [Cγ], 26.5 [Cδ], 31.4 [Cβ], 38.5 [Cε], 42.0 [CH2 (NHBn)], 42.9 [CH2 (NHBn, Urea)], 52.0 [Cα], 126.4, 126.5, 126.7, 126.9, 127.0, 128.2, 128.4, 129.1 [15CH (Ar)], 137.3 [C (Ph)], 139.3 [C (NHBn)], 140.7 [C (NHBn, Urea)], 158.0 [CO (Urea)], 171.2 [α-CONH]; ES-MS m/z [M]+ calculated for C35H45N7O4: 628.2; found: 628.5.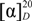 = −1.6 (c 0.6, MeOH); HPLC tR: 15.34 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.28 (m, 2H, γ-H), 1.47 (m, 1H, β-H), 1.50 (m, 2H, δ-H), 1.70 (m, 1H, β-H), 2.57–3.01 (m, 7H, CH2-Ph, ε-H, 2-H and 3-H), 3.38 (m, 1H, 6-H), 3.42 (m, 1H, CH2CO), 3.53 (m, 1H, CH2CO), 3.62 (m, 1H, 6-H), 4.00 [m, 1H, CH2 (NHBn, Urea)], 4.17 [m, 1H, CH2 (NHBn, Urea)], 4.20 (m, 1H, 2-CH), 4.26 [m, 2H, CH2 (NHBn)], 4.30 (m, 1H, α-H), 6.45 (m, 1H, 2-CHNH), 6.96–7.34 [m, 16H, Ar and (NHBn, Urea)], 7.84 (m, 4H, NH2·HCl and 4-H), 8.05 (m, 1H, α-NH), 8.60 (m, 1H, NHBn); 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 22.2 [Cγ], 26.5 [Cδ], 31.4 [Cβ], 38.5 [Cε], 42.0 [CH2 (NHBn)], 42.6 [CH2 (NHBn, Urea)], 52.5 [Cα], 126.4, 126.6, 126.7, 127.0, 128.1, 128.2, 129.3 [15CH (Ar)], 137.4 [C (Ph)], 139.6 [C (NHBn)], 141.0 [C (NHBn, Urea)], 159.0 [CO (Urea)], 171.5 [α-CONH]; ES-MS m/z [M]+ calculated for C35H45N7O4: 628.2; found: 628.5.
= −1.6 (c 0.6, MeOH); HPLC tR: 15.34 min; 1H-NMR (500 MHz, DMSO-d6) δ (ppm): 1.28 (m, 2H, γ-H), 1.47 (m, 1H, β-H), 1.50 (m, 2H, δ-H), 1.70 (m, 1H, β-H), 2.57–3.01 (m, 7H, CH2-Ph, ε-H, 2-H and 3-H), 3.38 (m, 1H, 6-H), 3.42 (m, 1H, CH2CO), 3.53 (m, 1H, CH2CO), 3.62 (m, 1H, 6-H), 4.00 [m, 1H, CH2 (NHBn, Urea)], 4.17 [m, 1H, CH2 (NHBn, Urea)], 4.20 (m, 1H, 2-CH), 4.26 [m, 2H, CH2 (NHBn)], 4.30 (m, 1H, α-H), 6.45 (m, 1H, 2-CHNH), 6.96–7.34 [m, 16H, Ar and (NHBn, Urea)], 7.84 (m, 4H, NH2·HCl and 4-H), 8.05 (m, 1H, α-NH), 8.60 (m, 1H, NHBn); 13C-NMR (125 MHz, DMSO-d6) δ (ppm): 22.2 [Cγ], 26.5 [Cδ], 31.4 [Cβ], 38.5 [Cε], 42.0 [CH2 (NHBn)], 42.6 [CH2 (NHBn, Urea)], 52.5 [Cα], 126.4, 126.6, 126.7, 127.0, 128.1, 128.2, 129.3 [15CH (Ar)], 137.4 [C (Ph)], 139.6 [C (NHBn)], 141.0 [C (NHBn, Urea)], 159.0 [CO (Urea)], 171.5 [α-CONH]; ES-MS m/z [M]+ calculated for C35H45N7O4: 628.2; found: 628.5.