Antimicrobial Activities against Periodontopathic Bacteria of Pittosporum tobira and Its Active Compound
Abstract
:1. Introduction
2. Results and Discussion
2.1. Antibacterial Activity of the Plant Extracts against Periodontopathogens
| Family | Scientific Name | Part | Activity a | ||
|---|---|---|---|---|---|
| Pg | Pi | Fn | |||
| Primulaceae | Lysimachia mauritiana Lam. | Whole plant | - | + | +++ |
| Betulaceae | Alnus firma S. et Z. | Leaf | ++ | - | - |
| Betulaceae | Carpinus laxiflora Bl. | Leaf | + | - | - |
| Taxaceae | Torreya nucifera S. et Z. | Fruit | - | - | +++ |
| Fabaceae | Albizzia julibrissin Durazz | Leaf | - | - | ++ |
| Fabaceae | Albizzia julibrissin Durazz | Fruit | ++ | - | +++ |
| Euphorbiaceae | Sapium japonicum Pax et Hoffm | Leaf | + | - | - |
| Pittosporaceae | Pittosporum tobira Ait. | Leaf | + | + | ++ |
| Lardizabalaceae | Akebia quinata Decne. | Fruit | ++ | - | - |
| Lauraceae | Litsea japonica Juss. | Fruit | - | - | +++ |
| Erythromycin | +++ | +++ | +++ | ||
2.2. Antibacterial Effect of P. tobira against Periodontopathogens
| Bacterial strains | MICs (μg/mL) | |||||||
|---|---|---|---|---|---|---|---|---|
| EtOH80% | n-Hexane | MC | EtOAc | n-BuOH | DW | Erythromycin | ||
| Pg | >800 | 400 | 800 | 200 | >800 | 800 | <0.02 | |
| Pi | >800 | 200 | 800 | 200 | >800 | 400 | <0.02 | |
| Fn | >800 | 200 | 800 | 200 | >800 | 400 | <0.02 | |
2.3. Chemistry
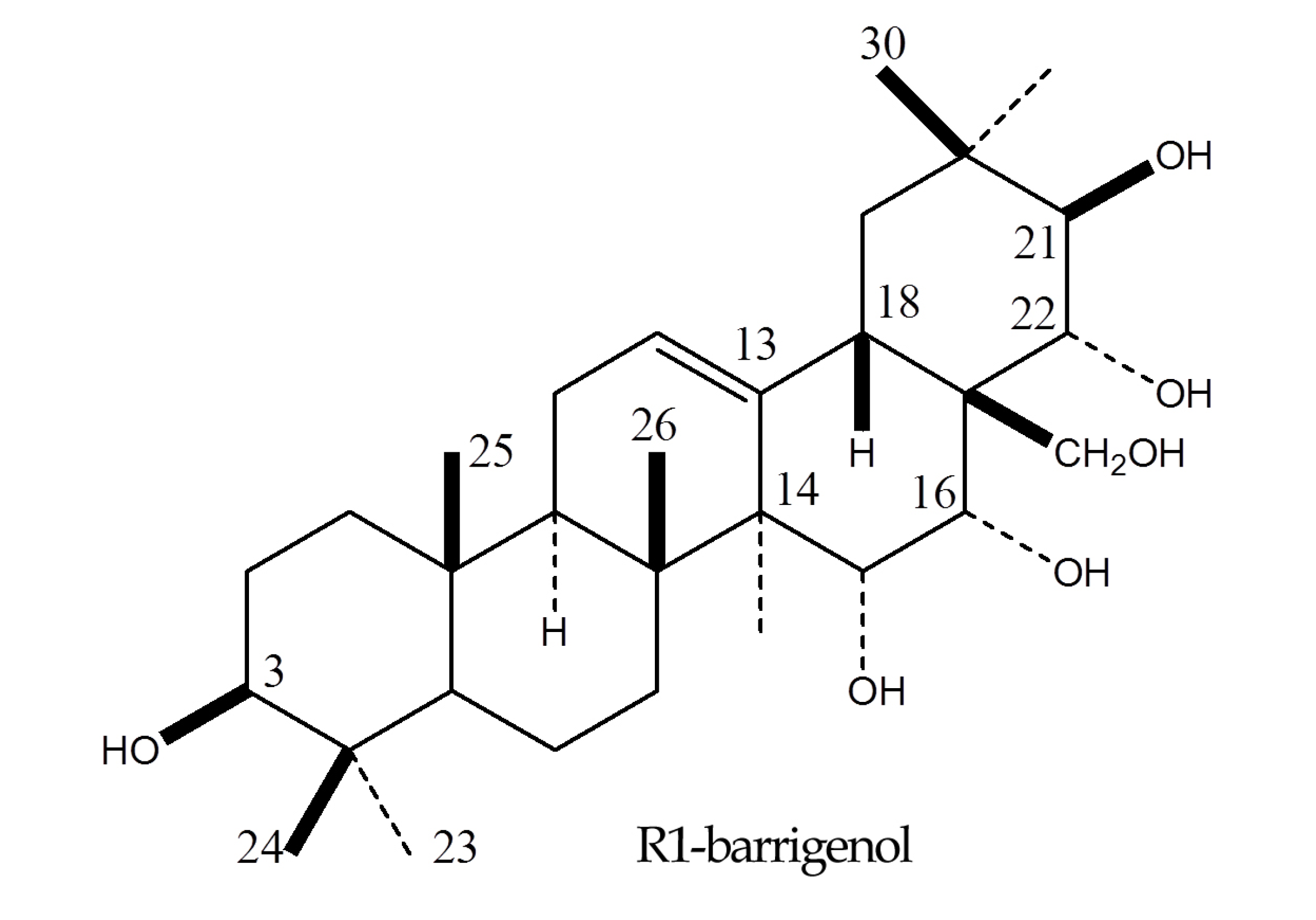
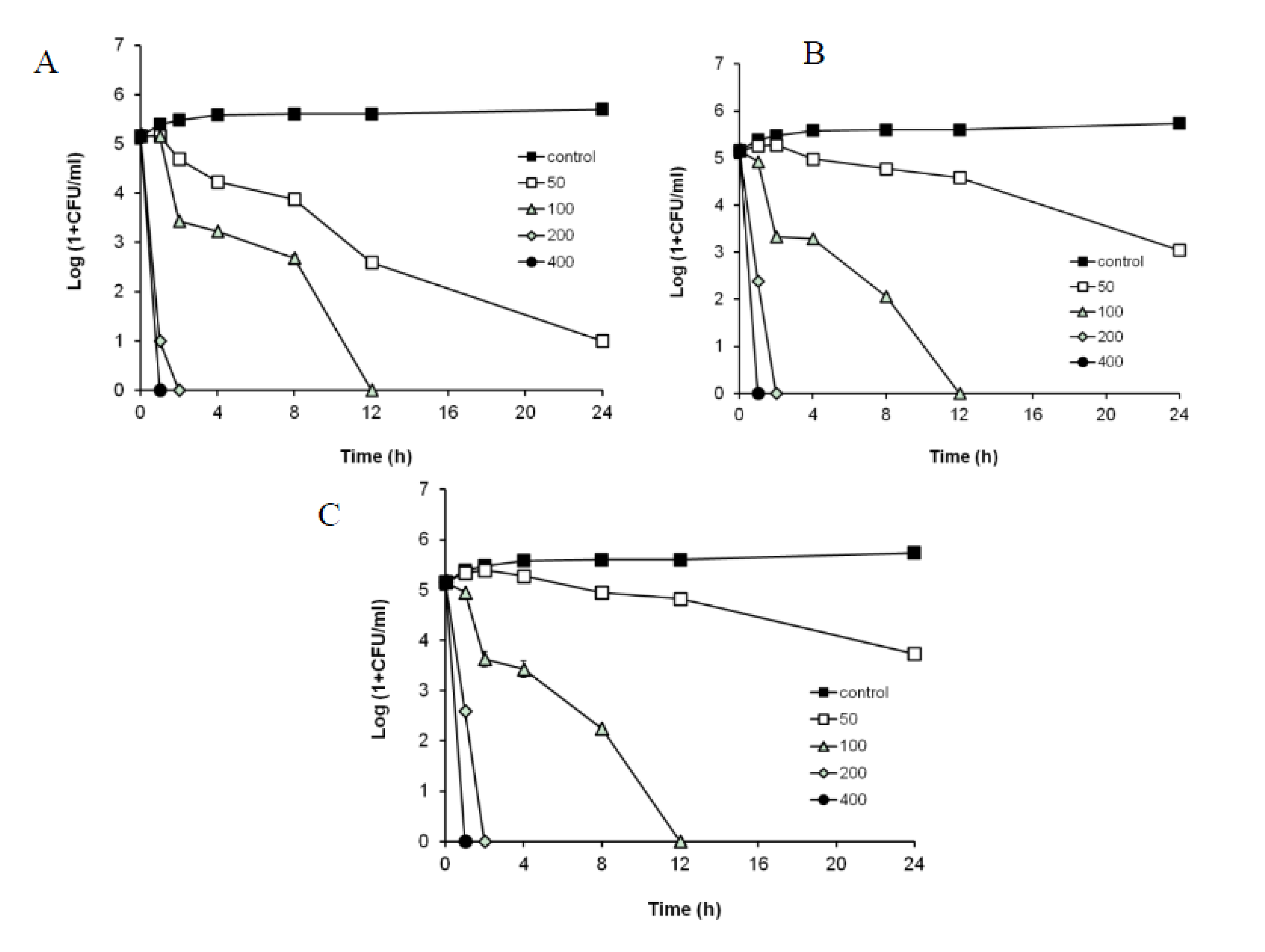
2.4. Antibacterial Effect of R1-Barrigenol against Periodontopathogens
3. Experimental
3.1. Plant Materials
3.2. Antibacterial Activity Screening: Disc Diffusion Test
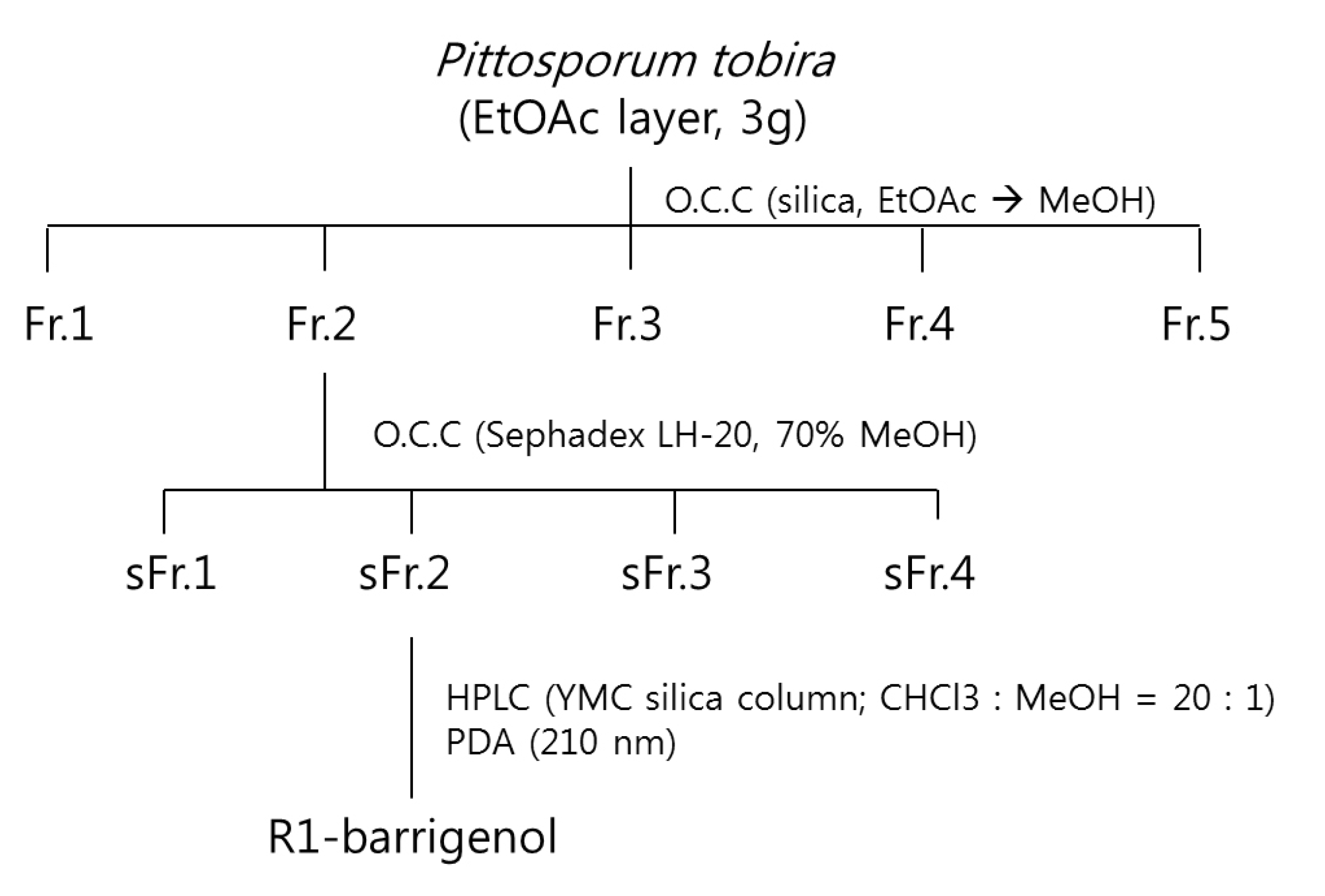
3.3. Extraction and Solvent Partitions of P. tobira

3.4. Determination of the Minimal Inhibitory Concentrations (MIC) and Minimal Bactericidal Concentration (MBC)
3.5. Cell Culture and Cellular Toxicity Assay
3.6. Isolation of the Compounds
4. Conclusions
Acknowledgments
Author Contributions
Conflictts of Interest
References
- Miyazaki, H.; Pilot, T.; Leclercq, M.H.; Barmes, D.E. Profiles of periodontal conditions in adults measured by CPITN. Int. Dent. J. 1991, 41, 74–80. [Google Scholar]
- Komerik, N.; Nakanishi, H.; MacRobert, A.J.; Henderson, B.; Speight, P.; Wilson, M. In vivo killing of Porphyromonas gingivalis by toluidine blue-mediated photosensitization in an animal model. Antimicrob. Agents Chemother. 2003, 47, 932–940. [Google Scholar] [CrossRef]
- Pihlstrom, B.L.; Michalowicz, B.S.; Johnson, N.W. Periodontal diseases. Lancet 2005, 366, 1809–1820. [Google Scholar] [CrossRef]
- Haraszthy, V.I.; Zambon, J.J.; Trevisan, M.; Zeid, M.; Genco, R.J. Identification of periodontal pathogens in atheromatous plaques. J. Periodontol. 2000, 71, 1554–1560. [Google Scholar] [CrossRef]
- Walker, C.B. The acquisition of antibiotic resistance in the periodontal microflora. Periodontology 2000 1996, 10, 79–88. [Google Scholar] [CrossRef]
- Chaves, E.S.; Jeffcoat, M.K.; Ryerson, C.C.; Snyder, B. Persistent bacterial colonization of Porphyromonas gingivalis, Prevotella intermedia, and Actinobacillus actinomycetemcomitans in periodontitis and its association with alveolar bone loss after 6 months of therapy. J. Clin. Periodontol. 2000, 27, 897–903. [Google Scholar]
- Feres, M.; Haffajee, A.D.; Goncalves, C.; Allard, K.A.; Som, S.; Smith, C.; Goodson, J.M.; Socransky, S.S. Systemic doxycycline administration in the treatment of periodontal infections (II). Effect on antibiotic resistance of subgingival species. J. Clin. Periodontol. 1999, 26, 784–792. [Google Scholar] [CrossRef]
- Loesche, W.J.; Syed, S.A.; Morrison, E.C.; Kerry, G.A.; Higgins, T.; Stoll, J. Metronidazole in periodontitis. I. Clinical and bacteriological results after 15 to 30 weeks. J. Periodontol. 1984, 55, 325–335. [Google Scholar] [CrossRef]
- Olsvik, B.; Tenover, F.C. Tetracycline resistance in periodontal pathogens. Clin. Infect. Dis. 1993, 16 (Suppl. 4), S310–S313. [Google Scholar] [CrossRef]
- Renvert, S.; Dahlen, G.; Wikstrom, M. Treatment of periodontal disease based on microbiological diagnosis. Relation between microbiological and clinical parameters during 5 years. J. Periodontol. 1996, 67, 562–571. [Google Scholar] [CrossRef]
- Andres, M.T.; Chung, W.O.; Roberts, M.C.; Fierro, J.F. Antimicrobial susceptibilities of Porphyromonas gingivalis, Prevotella intermedia, and Prevotella nigrescens spp. isolated in Spain. Antimicrob. Agents Chemother. 1998, 42, 3022–3023. [Google Scholar]
- Fosse, T.; Madinier, I.; Hannoun, L.; Giraud-Morin, C.; Hitzig, C.; Charbit, Y.; Ourang, S. High prevalence of cfxA beta-lactamase in aminopenicillin-resistant Prevotella. strains isolated from periodontal pockets. Oral Microbiol. Immunol. 2002, 17, 85–88. [Google Scholar] [CrossRef]
- Betoni, J.E.; Mantovani, R.P.; Barbosa, L.N.; di Stasi, L.C.; Fernandes Junior, A. Synergism between plant extract and antimicrobial drugs used on Staphylococcus aureus diseases. Mem. Inst. Oswaldo. Cruz 2006, 101, 387–390. [Google Scholar] [CrossRef]
- Lee, S.B.; Cha, K.H.; Kim, S.N.; Altantsetseg, S.; Shatar, S.; Sarangerel, O.; Nho, C.W. The Antimicrobial activity of essential Oil from Dracocephalum foetidum against pathogenic microorganisms. J. Microbiol. 2007, 45, 53–57. [Google Scholar]
- Lewis, K.; Ausubel, F.M. Prospects of plant derived antibacterials. Nat. Biotechnol. 2006, 24, 1504–1507. [Google Scholar] [CrossRef]
- Sharma, A.; Chandraker, S.; Patel, V.K.; Ramteke, P. Antibacterial activity of medicinal plants against pathogens causing complicated urinary tract infections. Indian J. Pharm. Sci. 2009, 71, 136–139. [Google Scholar] [CrossRef]
- Khan, R.; Islam, B.; Akram, M.; Shakil, S.; Ahmad, A.; Ali, S.M.; Siddiqui, M.; Khan, A.U. Antimicrobial activity of five herbal extracts against multi drug resistant (MDR) strains of bacteria and fungus of clinical origin. Molecules 2009, 14, 586–597. [Google Scholar] [CrossRef]
- Santos, P.R.V.; Oliveira, A.C.X.; Tomassini, T.C.B. Controle Microbiogico De Productous Fitoterapicos. Rev. Farm. Bioquim. 1995, 31, 35–38. [Google Scholar]
- Havyarimana, L.; Ndendoung, S.T.; Tamokou Jde, D.; Atchade Ade, T.; Tanyi, J.M. Chemical constituents of Millettia barteri and their antimicrobial and antioxidant activities. Pharm. Biol. 2012, 50, 141–146. [Google Scholar] [CrossRef]
- Kuorwel, K.K.; Cran, M.J.; Sonneveld, K.; Miltz, J.; Bigger, S.W. Essential oils and their principal constituents as antimicrobial agents for synthetic packaging films. J. Food Sci. 2011, 76, R164–R177. [Google Scholar] [CrossRef]
- Saddiqe, Z.; Naeem, I.; Maimoona, A. A review of the antibacterial activity of Hypericum perforatum L. J. Ethnopharmacol. 2010, 131, 511–521. [Google Scholar] [CrossRef]
- Mahady, G.B. Medicinal plants for the prevention and treatment of bacterial infections. Curr. Pharm. Des. 2005, 11, 2405–2427. [Google Scholar] [CrossRef]
- Radhika, L.G.; Meena, C.V.; Peter, S.; Rajesh, K.S.; Rosamma, M.P. Phytochemical and antimicrobial study of Oraxylum indicum. Anc. Sci. Life 2011, 30, 114–120. [Google Scholar]
- D’Acquarica, I.; di Giovanni, M.C.; Gasparrini, F.; Misiti, D.; D’Arrigo, C.; Fagnano, N.; Guarnieri, D.; Iacono, G.; Bifulco, G.; Riccio, R. Isolation and structure elucidation of four new triterpenoid estersaponins from fruits of Pittosporum tobira ait. Tetrahedron 2002, 58, 10127–10136. [Google Scholar] [CrossRef]
- Moon, H.I.; Park, W.H. Four carotenoids from Pittosporum tobira protect primary cultured rat cortical cells from glutamate-induced toxicity. Phytother. Res. 2010, 24, 625–628. [Google Scholar]
- Moon, J.H.; Park, J.H.; Lee, J.Y. Antibacterial action of polyphosphate on Porphyromonas gingivalis. Antimicrob. Agents Chemother. 2011, 55, 806–812. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute (CLSI). Methods for Determining Bactericidal Activity of Antimicrobial Agents; In Approved Guideline, Document M26-A; CLSI: Wayne, PA, USA, 1999. [Google Scholar]
- Bauer, A.W.; Kirby, W.M.; Sherris, J.C.; Turck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol. 1966, 45, 493–496. [Google Scholar]
- Sample Availability: Not available.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Oh, J.-H.; Jeong, Y.J.; Koo, H.J.; Park, D.W.; Kang, S.C.; Khoa, H.V.B.; Le, L.B.; Cho, J.H.; Lee, J.-Y. Antimicrobial Activities against Periodontopathic Bacteria of Pittosporum tobira and Its Active Compound. Molecules 2014, 19, 3607-3616. https://doi.org/10.3390/molecules19033607
Oh J-H, Jeong YJ, Koo HJ, Park DW, Kang SC, Khoa HVB, Le LB, Cho JH, Lee J-Y. Antimicrobial Activities against Periodontopathic Bacteria of Pittosporum tobira and Its Active Compound. Molecules. 2014; 19(3):3607-3616. https://doi.org/10.3390/molecules19033607
Chicago/Turabian StyleOh, Jung-Hyun, Yong Joon Jeong, Hyun Jung Koo, Dae Won Park, Se Chan Kang, Hoang Viet Bach Khoa, Le Ba Le, Joon Hyeong Cho, and Jin-Yong Lee. 2014. "Antimicrobial Activities against Periodontopathic Bacteria of Pittosporum tobira and Its Active Compound" Molecules 19, no. 3: 3607-3616. https://doi.org/10.3390/molecules19033607
APA StyleOh, J.-H., Jeong, Y. J., Koo, H. J., Park, D. W., Kang, S. C., Khoa, H. V. B., Le, L. B., Cho, J. H., & Lee, J.-Y. (2014). Antimicrobial Activities against Periodontopathic Bacteria of Pittosporum tobira and Its Active Compound. Molecules, 19(3), 3607-3616. https://doi.org/10.3390/molecules19033607




