Inhibitory Effect of Corn Silk on Skin Pigmentation
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effects on Cell Viability and Melanin Production
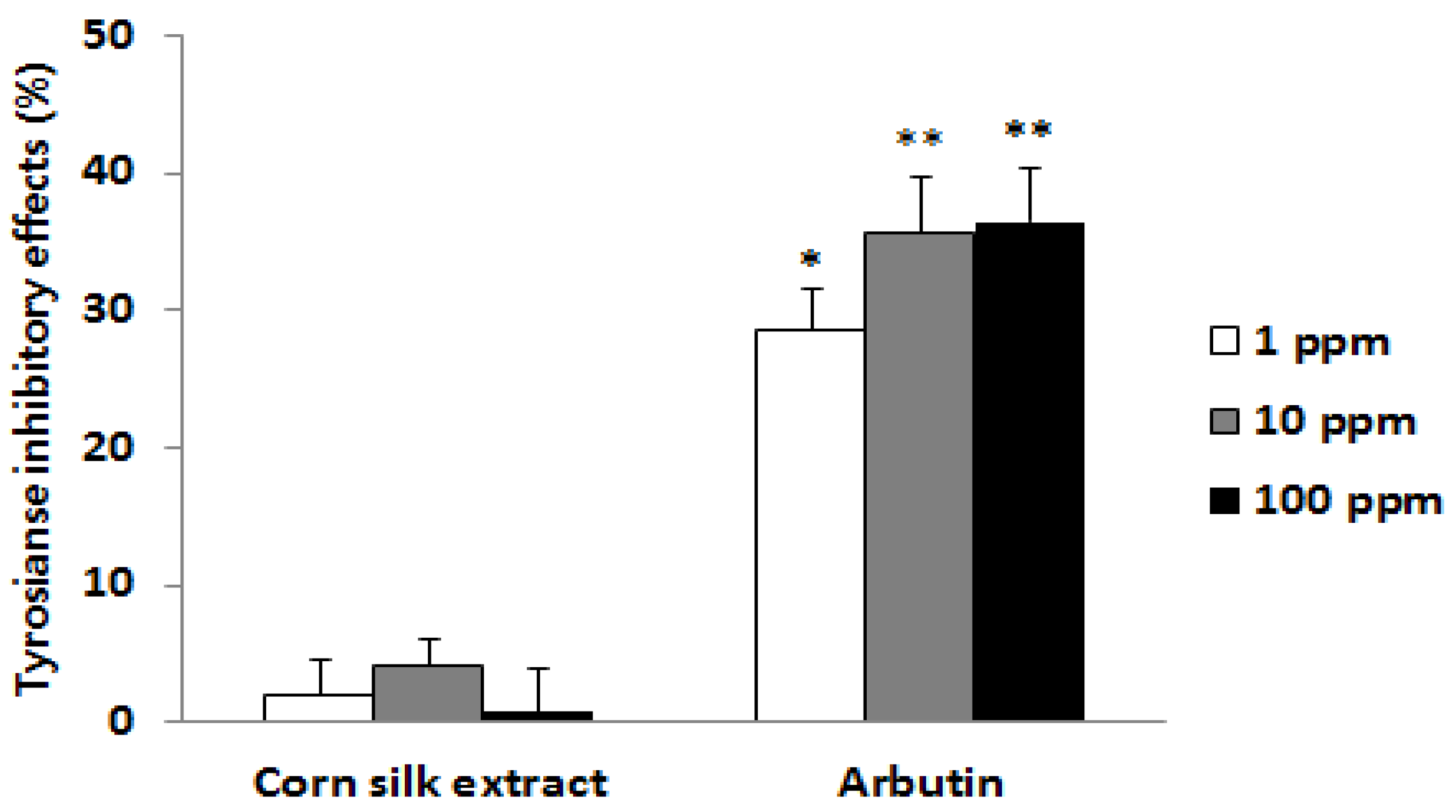
2.2. Tyrosinase Inhibitory Effect

| Concentrations (ppm) | Cell viability (%) | Melanin production (%) | |
|---|---|---|---|
| Corn silk | 1 | 97.5 ± 1.7 | 100.0 ± 0.8 |
| 10 | 95.0 ± 4.2 | 90.8 ± 4.1 | |
| 100 | 96.9 ± 4.9 | 62.8 ± 4.9 | |
| Arbutin | 1 | 97.1 ± 3.3 | 99.7 ± 2.6 |
| 10 | 95.7 ± 3.6 | 99.4 ± 5.0 | |
| 100 | 100.8 ± 2.7 | 73.2 ± 4.8 |
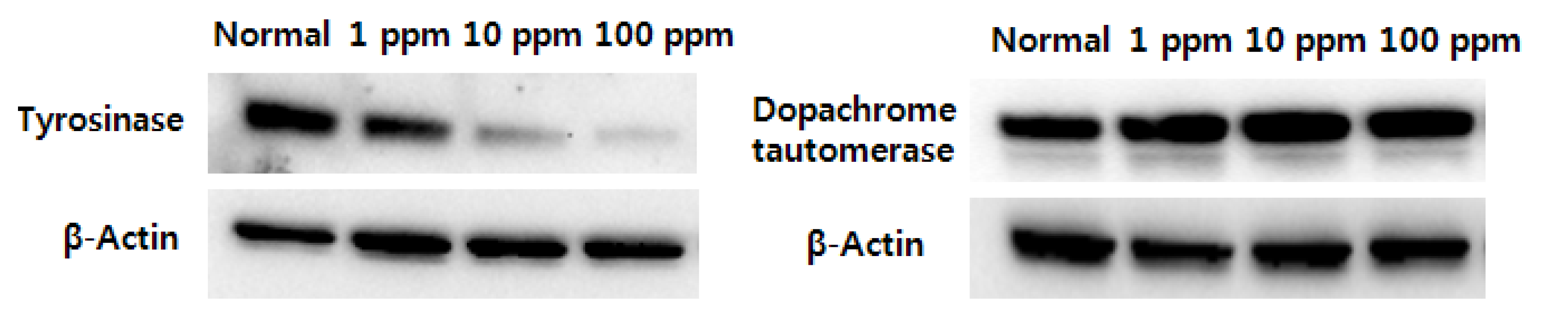
2.3. Effect on Protein Expression in the Cell
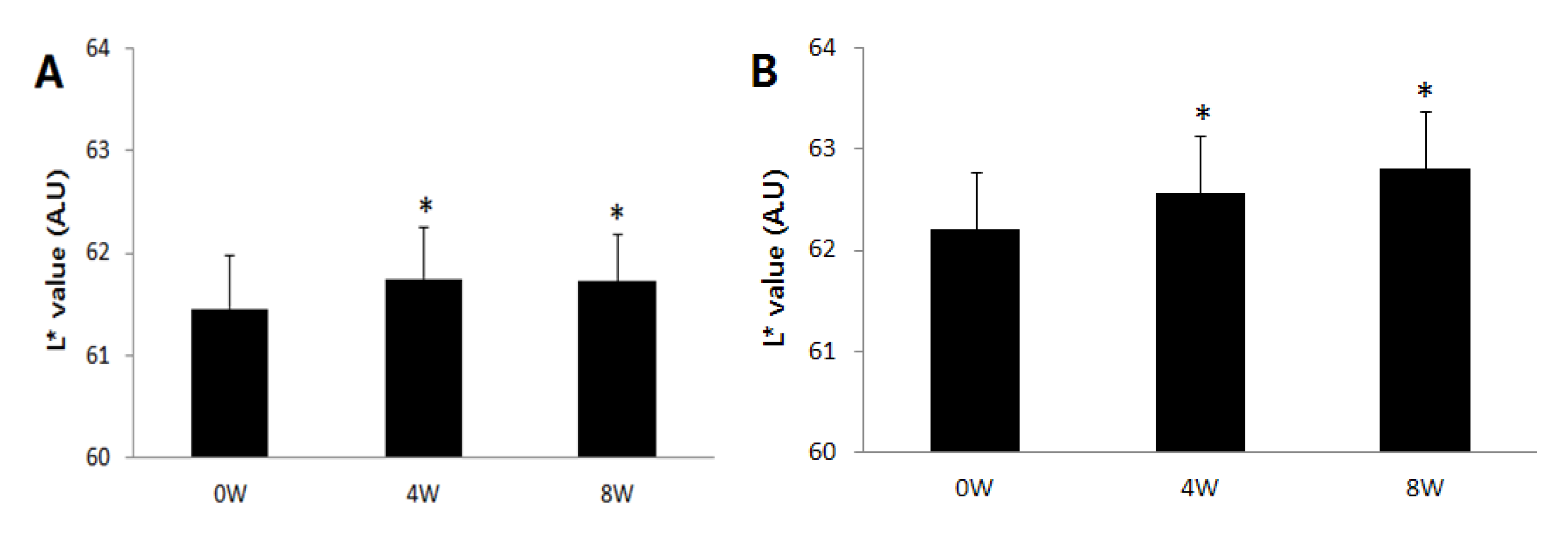
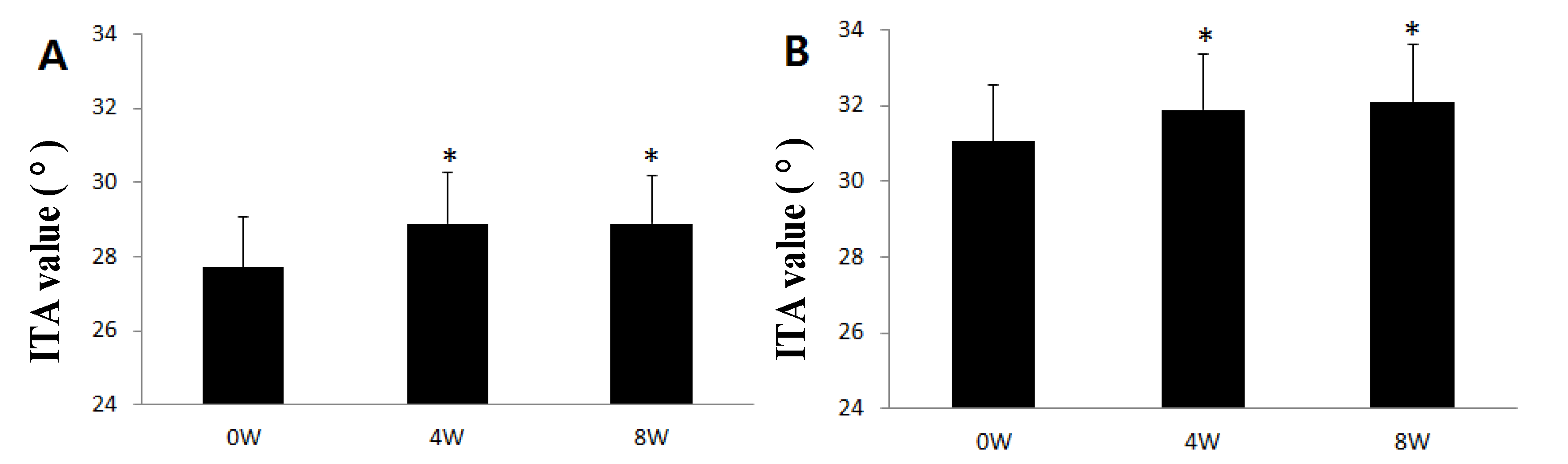
2.4. Improvement in Hyperpigmentation on Human Skin


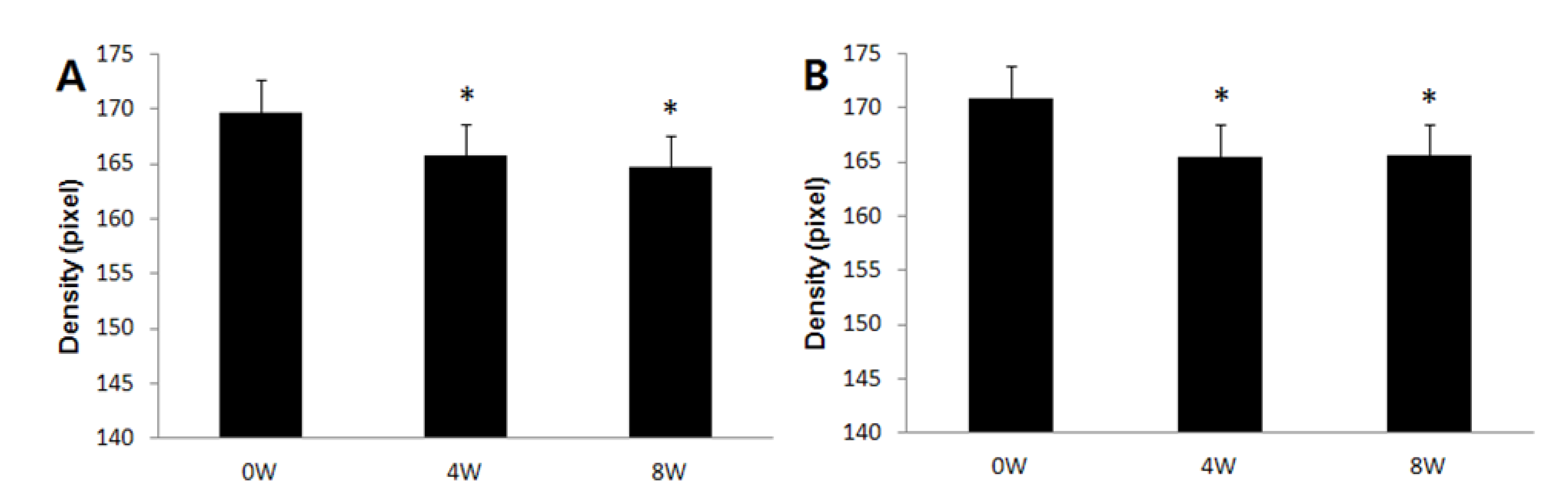
2.5. Safety on Human Skin
| Symptom | Corn silk extract 0.75% | Corn silk extract 1.5% | |||
|---|---|---|---|---|---|
| 4W | 8W | 4W | 8W | ||
| Irritation | Redness | 0 | 0 | 0 | |
| Edema | 0 | 0 | 0 | 0 | |
| Desquamation | 0 | 0 | 0 | 0 | |
| Papule | 0 | 0 | 0 | 0 | |
| Other | 0 | 0 | 0 | 0 | |
2.6. Discussion
3. Experimental
3.1. Materials
3.2. Extract Preparation
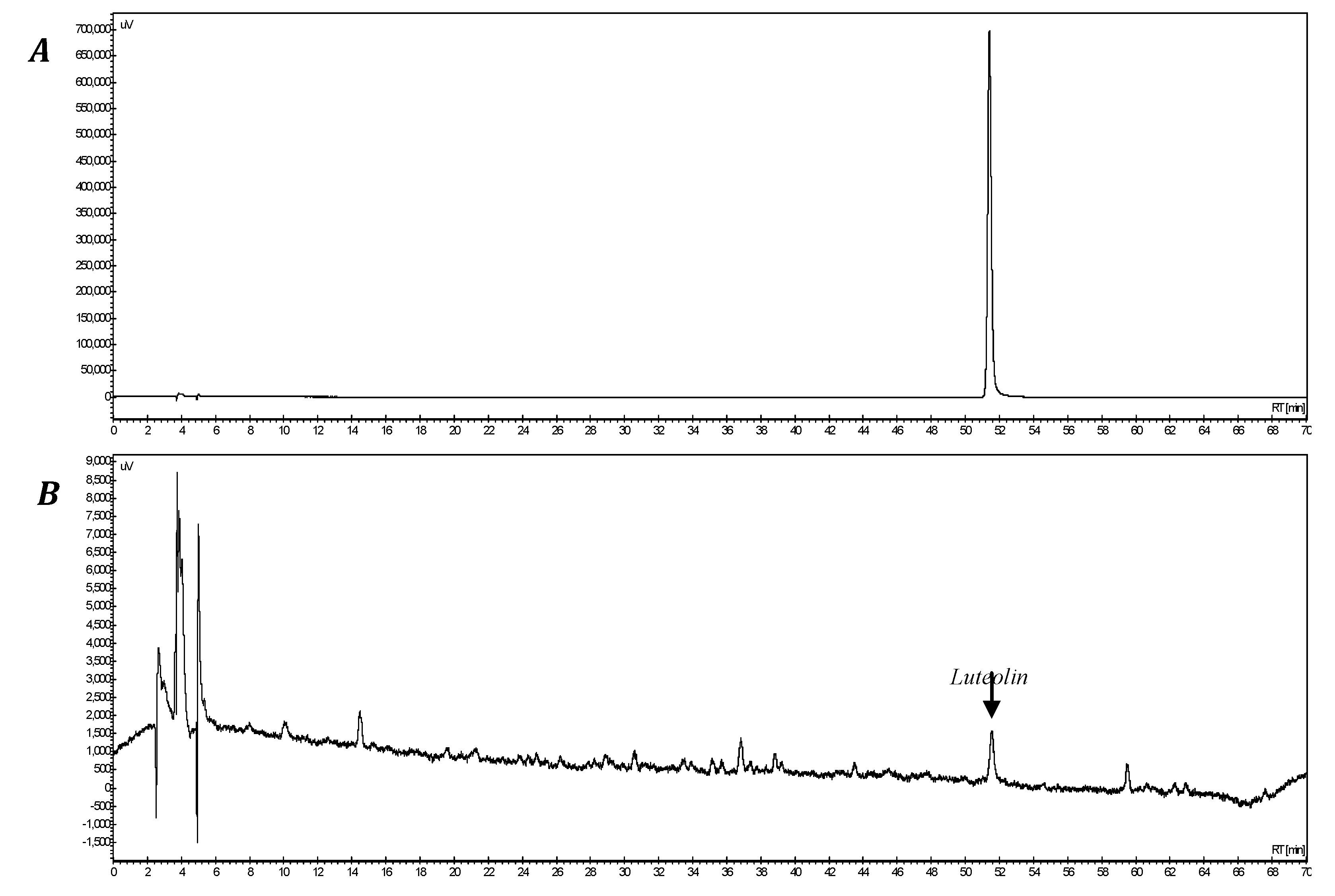
3.3. Cell Culture
3.4. Cell Viability
3.5. Melanin Production
3.6. Tyrosinase Inhibitory Effect
3.7. Protein Expression Level in the Cell
3.8. Improvement of Hyperpigmentation in Human Skin
3.9. Safety Evaluation on Human Skin
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflictts of Interest
References
- Suola, S.; Kitchell, B. The biology of melanocytes. Vet. Dermatol. 2003, 14, 57–65. [Google Scholar] [CrossRef]
- Lin, J.Y.; Fisher, D.E. Melanocyte biology and skin pigmentation. Nature 2007, 445, 843–850. [Google Scholar] [CrossRef]
- No, J.K.; Soung, D.Y.; Kim, Y.J.; Shim, K.H.; Jun, Y.S.; Rhee, S.H.; Yokozawa, T.; Chung, H.Y. Inhibition of tyrosinase by green tea components. Life Sci. 1999, 66, 241–246. [Google Scholar]
- Iozumi, K.; Hoganson, G.E.; Pemella, R.; Everett, M.A.; Fuller, B.B. Role of tyrosinase as the determinant of pigmentation in cultured human melanocytes. J. Invest. Dermatol. 1993, 66, 806–811. [Google Scholar]
- Wang, N.; Hebert, D.N. Tyrosinase maturation through the mammalian secrerory pathway: Bringing color to life. Pigment Cell Res. 2006, 19, 3–18. [Google Scholar] [CrossRef]
- Slominski, A.; Tobin, D.J.; Shibahara, S.; Wortsman, J. Melanin pigmentation in mammalian skin and its hormonal regulation. Physiol. Rev. 2004, 84, 1155–1228. [Google Scholar] [CrossRef]
- Miyamura, Y.; Coelho, S.; Wolber, R.; Miller, S.; Wakamatsu, K.; Zmudzka, B.Z.; Ito, S.; Smuda, C.; Passeron, T.; Choi, W.; et al. Regulation of human skin pigmentation and responses to ultraviolet radiation. Pigment Cell Res. 2006, 20, 2–13. [Google Scholar]
- Choi, W.; Miyamura, Y.; Wolber, R.; Smuda, C.; Reinhold, H.L.; Kolbe, L.; Hearing, V.J. Regulation of human skin pigmentation in situ by repetitive UV exposure: Molecular characterization of responses to UVA and/or UVB. J. Invest. Dermatol. 2010, 130, 1685–1696. [Google Scholar] [CrossRef]
- García-Gavín, J.; González-Vilas, D.; Fernández-Redondo, V.; Toribio, J. Pigmented contact dermatitis due to kojic acid. A paradoxical side effect of a skin lightener. Contact Dermatitis 2010, 62, 63–64. [Google Scholar] [CrossRef]
- Hasanudin, K.; Hashim, P.; Mustafa, S. Corn silk (Stigma maydis) in healthcare: A phytochemical and pharmacological review. Molecules 2012, 17, 9697–9715. [Google Scholar] [CrossRef]
- Hu, Q.I.; Deng, Z.I. Protective effects of flavonoids from corn silk on oxidative stress induced by exhaustive exercise in mice. Afr. J. Biotechnol. 2011, 10, 3163–3167. [Google Scholar]
- Wang, G.Q.; Xu, T.; Bu, X.M.; Liu, B.Y. Anti-inflammation effects of corn silk in a rat model of carrageenin-induced pleurisy. Inflammation 2012, 35, 822–827. [Google Scholar] [CrossRef]
- Zhao, W.; Yin, Y.; Yu, Z.; Liu, J.; Chen, F. Comparison of anti-diabetic effects of polysaccharides from corn silk on normal and hyperglycemia rats. Int. J. Biol. Macromol. 2012, 50, 1133–1137. [Google Scholar] [CrossRef]
- Hu, Q.L.; Zhang, L.J.; Li, Y.N.; Ding, Y.J.; Li, F.L. Purification and anti-fatigue activity of flavonoids from corn silk. Int. J. Phys Sci. 2010, 5, 321–326. [Google Scholar]
- Kan, A.; Orhan, I.; Coksari, G.; Sener, B. In-vitro neuroprotective properties of the maydis stigma extracts from four corn varieties. Int. J. Food Sci. Nutr. 2011, 63, 1–4. [Google Scholar]
- Velazquez, D.V.; Xavier, H.S.; Batista, J.E.; de Castro-Chaves, C. Zea mays L. extracts modify glomerular function and potassium urinary excretion in conscious rats. Phytomedicine 2005, 12, 363–369. [Google Scholar] [CrossRef]
- Bennett, D.; Cooper, P.; Hart, I. A line of non-tumorigenic mouse melanocytes, syngeneic with the B16 melanoma and requiring a tumor promoter for growth. Int. J. Cancer. 1987, 39, 414–418. [Google Scholar] [CrossRef]
- Lim, Y.J.; Lee, E.H.; Kang, T.H.; Ha, S.K.; Oh, M.S.; Kim, S.M.; Yoon, T.J.; Kang, C.; Park, J.H.; Kim, S.Y. Inhibitory effects of arbutin on melanin biosynthesis of alpha-melanocyte stimulating hormone-induced hyperpigmentation in cultured brownish guinea pig skin tissues. Arch. Pharm. Res. 2009, 32, 367–373. [Google Scholar] [CrossRef]
- Ando, H.; Kondoh, H.; Ichihashi, M.; Hearing, V.J. Approaches to identify inhibitors of melanin biosynthesis via the quality control of tyrosinase. J. Invest. Dermatol. 2007, 127, 751–761. [Google Scholar] [CrossRef]
- Martinez, J.; Solano, F.; Garcia, J.; Jara, J.; Lozano, J. α-MSH and other melanogenic activators mediate opposite effects on tyrosinase and dopachrome tautomerase in B16/F10 mouse melanoma cells. J. Invest. Dermatol. 1992, 99, 435–439. [Google Scholar]
- Sugumaran, M. Comparative biochemistry of eumelanogenesis and the protective roles of phenoloxidase and melanin in insects. Pigment Cell Res. 2002, 15, 2–9. [Google Scholar] [CrossRef]
- Choi, M.Y.; Song, H.S.; Hur, H.S.; Sim, S.S. Whitening activity of luteolin related to the inhibition of cAMP pathway in alpha-MSH-stimulated B16 melanoma cells. Arch. Pharm. Res. 2008, 31, 1166–1171. [Google Scholar] [CrossRef]
- Sample Availability: Sample of the corn silk extract is available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Choi, S.Y.; Lee, Y.; Kim, S.S.; Ju, H.M.; Baek, J.H.; Park, C.-S.; Lee, D.-H. Inhibitory Effect of Corn Silk on Skin Pigmentation. Molecules 2014, 19, 2808-2818. https://doi.org/10.3390/molecules19032808
Choi SY, Lee Y, Kim SS, Ju HM, Baek JH, Park C-S, Lee D-H. Inhibitory Effect of Corn Silk on Skin Pigmentation. Molecules. 2014; 19(3):2808-2818. https://doi.org/10.3390/molecules19032808
Chicago/Turabian StyleChoi, Sang Yoon, Yeonmi Lee, Sung Soo Kim, Hyun Min Ju, Ji Hwoon Baek, Chul-Soo Park, and Dong-Hyuk Lee. 2014. "Inhibitory Effect of Corn Silk on Skin Pigmentation" Molecules 19, no. 3: 2808-2818. https://doi.org/10.3390/molecules19032808
APA StyleChoi, S. Y., Lee, Y., Kim, S. S., Ju, H. M., Baek, J. H., Park, C.-S., & Lee, D.-H. (2014). Inhibitory Effect of Corn Silk on Skin Pigmentation. Molecules, 19(3), 2808-2818. https://doi.org/10.3390/molecules19032808





