The Biological Activities of Cinnamon, Geranium and Lavender Essential Oils
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemical Composition of the Cinnamon, Geranium and Lavender Essential Oils
2.2. Susceptibility Testing of Clinical Acinetobacter Baumannii Strains to Antibiotics
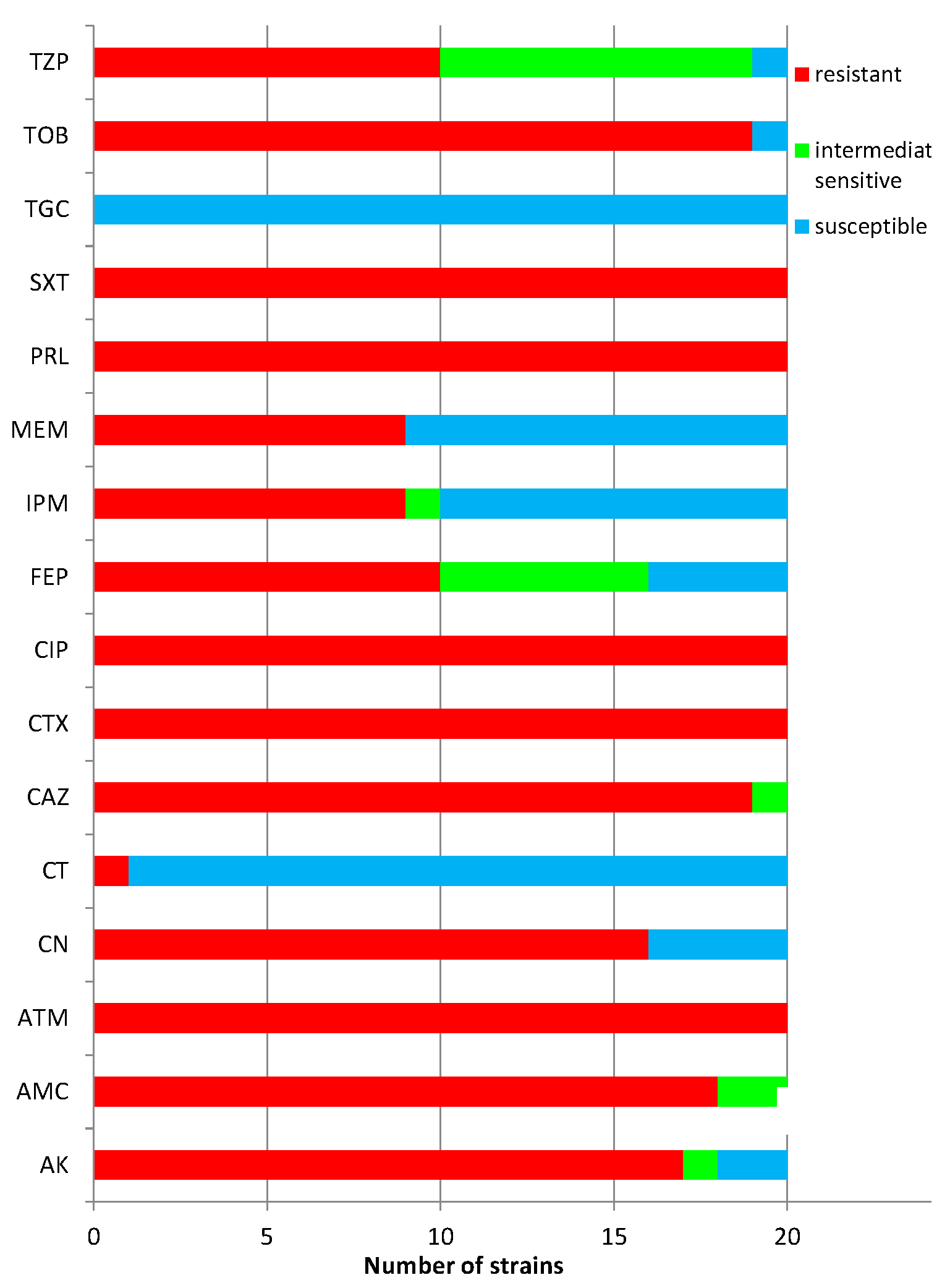
2.3. The Susceptibility of Acinetobacter Baumannii Bacterial Strains to Cinnamon Bark Oil

2.4. The Susceptibility of Acinetobacter Baumannii Bacterial Strains to Geranium Oil
2.5. The Susceptibility of Acinetobacter Baumannii Bacterial Strains to Lavender Oil
2.6. The Effects of Essential Oils on the Viability of Human Microvascular Endothelial Cells (HMEC-1) and Glioblastoma Cell Line (T98G)
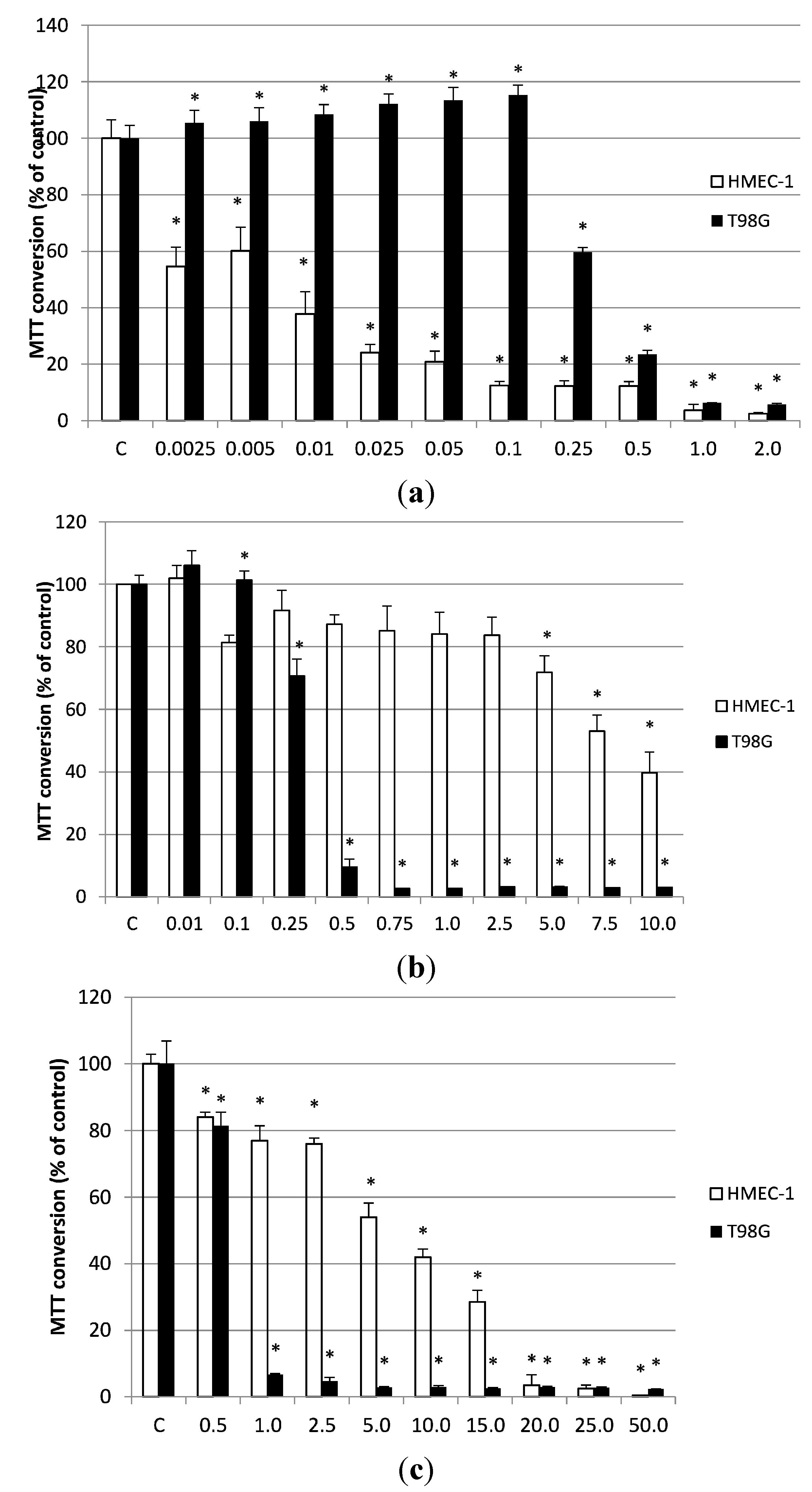
2.7. Discussion
3. Experimental Section
3.1. Bacterial Strains
3.2. Bacterial Strain Identification
3.3. Essential Oil Analysis
3.4. Antibacterial Tests
3.5. Susceptibility Testing
3.6. Cell Cultures
3.7. MTT Conversion
3.8. Statistical Analysis
4. Conclusions
- The obtained results show that the cinnamon, geranium and lavender essential oils demonstrate inhibitory activity against resistant clinical and environmental strains of Acinetobacter baumannii.
- Cinnamon bark oil was the most active against all Acinetobacter baumannii bacteria.
- Despite the fact that the bacteria are characterized by a high degree of antibiotic resistance, the tested essential oils have strong antibacterial action.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Maragakis, L.L.; Perl, T.M. Acinetobacter baumannii: Epidemiology, Antimicrobial Resistance, and Treatment Options. Clin. Infect. Dis. 2008, 46, 1254–1263. [Google Scholar]
- Fournier, P.E.; Richet, H.; Weinstein, R.A. The Epidemiology and Control of Acinetobacter baumannii in Health Care Facilities. Clin. Infect. Dis. 2006, 42, 692–699. [Google Scholar]
- Cusri, S.; Chongsuvivatwong, V.; Riviera, J.I.; Silpapojakul, K.; McNeil, E.; Doi, Y. Clinical Outcomes of Hospital-Acquired Infection with Acinetobacter nosocomialis and Acinetobacter pitii. Antimicrob. Agents Chemother. 2014, 58, 4172–4179. [Google Scholar]
- Inouye, S.; Takizawa, T.; Yamaguchi, H. Antibacterial activity of essential oils and their major constituents against respiratory tract pathogens by gaseous contact. J. Antimicrob. Chemother. 2001, 47, 565–573. [Google Scholar]
- Inouye, S.; Yamaguchi, H.; Takizawa, T. Screening of the antibacterial effects of a variety of essential oils on respiratory tract pathogens, using a modified dilution assay method. J. Infect. Chemother. 2001, 7, 251–254. [Google Scholar]
- Sivamani, P.; Hameed, A.S.S. In vitro antibacterial activity of essential oils of selected herbals against isolates from HIV/AIDS patients. J. Pharm. Res. 2010, 3, 672–678. [Google Scholar]
- Prabuseenivasan, S.; Jayakumar, M.; Ignacimuthu, S. In vitro antibacterial activity of some plant essential oils. BMC Complement. Altern. Med. 2006, 6. [Google Scholar] [CrossRef]
- Cavanagh, H.MA.; Wilkinson, J.M. Lavender essential oil: A review. Aust. Infect. Control. 2005, 10, 35–37. [Google Scholar]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Antibacterial activity of essential oils and other plant extracts. J. Appl. Microbiol. 1999, 86, 985–990. [Google Scholar]
- Manosroi, J.; Dhumtanom, P.; Manosroi, A. Antiproliferative activity of essential oil extracted from Thai medicinal plants on KB and P388 cell lines. Cancer Lett. 2006, 235, 114–120. [Google Scholar]
- Tsuneki, H.; Ma, E.L.; Kobayashi, S.; Sekizaki, N.; Maekawa, K.; Sasaoka, T.; Wang, M.W.; Kimura, I. Antiangiogenic activity of beta-eudesmol in vitro and in vivo. Eur. J. Pharmacol. 2005, 512, 105–115. [Google Scholar]
- Guba, R. Toxicity myths essential oils and their carcinogenic potential. Int. J. Aromather. 2001, 11, 76–83. [Google Scholar]
- Stammati, A.; Bonsi, P.; Zucco, F.; Moezelaar, R.; Alakomi, H.I.; von Wright, A. Toxicity of selected plant volatiles in microbial and mammalian short-term assays. Food Chem. Toxicol. 1999, 37, 813–823. [Google Scholar]
- Lahlou, S.; Leal-Cardoso, J.; Duarte, G. Antihypertensive effects of the essential oil of Alpinia zerumbet and its main constituent, terpinen-4-ol, in DOCA-salt hypertensive conscious rats. Fundam. Clin. Pharmacol. 2003, 17, 323–330. [Google Scholar]
- Dijoux, N.; Guingand, Y.; Bourgeois, C.; Durand, S.; Fromageot, C.; Combe, C.; Ferret, P.J. Assessment of the phototoxic hazard of some essential oils using modified 3T3 neutral red uptake assay. Toxicol. In Vitro 2006, 20, 480–489. [Google Scholar]
- Bleasel, N.; Tate, B.; Rademaker, M. Allergic contact dermatitis following exposure to essential oils. Australas. J. Dermatol. 2002, 43, 211–213. [Google Scholar]
- European Pharmacopoeia,, 6th ed; Council of Europe: Strasbourg, France, 2008.
- Polish Pharmacopeia VIII, 8th ed; Polish Pharmaceutical Society: Warsaw, Poland, 2008.
- Prakasam, G.; Bhashini, M.; Lakshmipriya, N.; Ramesh, S.S. In-vitro antibacterial activity of some essential oils against clinical isolates of Acinetobacter baumannie. Indian J. Med. Microbiol. 2014, 32, 90–91. [Google Scholar]
- Prashar, A.; Locke, I.C.; Evans, C.S. Cytotoxicity of lavender oil and its major components to human skin cells. Cell Prolif. 2004, 37, 221–229. [Google Scholar]
- Yap, P.S.X.; Yiap, B.C.; Ping, H.C.; Lim, S.H.E. Essential Oils, A New Horizon in Combating Bacterial Antibiotic Resistance. Open Microbiol. J. 2014, 8, 6–14. [Google Scholar]
- Mayaud, L.; Carricajo, A.; Zhiri, A.; Aubert, G. Comparison of bacteriostatic and bactericidal activity of 13 essential oils against strains with varying sensitivity to antibiotics. Lett. Appl. Microbiol. 2008, 47, 167–173. [Google Scholar]
- Fani, M.M.; Kohanteb, J. Inhibitory activity of Cinnamon zeylanicum and Eucalyptus globulus oils on Streptococcus mutans, Staphylococcus aureus, and Candida species isolated from patients with oral infections. Shiraz Univ. Dent. J. 2011, 11, 14–22. [Google Scholar]
- Pinto, V.; Barbosa, C.; Magalhães, P.; Coelho, C.; Fontenelle, J.; Cristino-Filho, G.; Chaves, H.; Silva, A.; Teixeira, A.; Bezerra, M. Antimicrobial activity of the trans-cinnamaldehyde on nosocomial enteric bacilli producers of extended spectrum β-lactamase (ESBL). BMC Proc. 2014, 8 (Suppl. 4). [Google Scholar] [CrossRef]
- Sienkiewicz, M.; Poznańska-Kurowska, K.; Kaszuba, A.; Kowalczyk, E. The antibacterial activity of geranium oil against Gram-negative bacteria isolated from difficult-to-heal wounds. Burns 2013, 40, 1046–1051. [Google Scholar]
- Guerra, F.Q.; Mendez, J.M.; Sousa, J.P.; Moralis-Braga, M.F.; Santos, B.H.; Melo Coutinho, H.D.; lima Ede, O. Increasing antibiotic activity against a multidrug-resistant Acinetobacter spp by essential oils of Citrus limon and Cinnamomum zeylanicum. Nat. Prod. Res. 2012, 26, 2235–2238. [Google Scholar]
- Khanuja, S.P.S.; Srivastava, S.; Shasney, A.K.; Darokar, M.; Kumar, T.R.S.; Agarwal, K.K.; Ahmed, A.; Patra, N.K.; Sinha, P.; Dhawan, S.; et al. Formulation Comprising Thymol Useful in the Treatment of Drug Resistant Bacterial Infections. U.S. Patent 6,824,795, 30 November 2004. [Google Scholar]
- Johnson, E.A.; Brehm-Stecher, B.F. Method of Sensitizing Microbial Cells to Antimicrobial Compound. U.S. Patent 6,319,958, 20 November 2001. [Google Scholar]
- NIST 98.1 NIST/EPA/NIH Mass Spectral Library; Mass Spectrometry Data Center National Institute of Standards and Technology: Gaithersburg, MD, USA, 1998.
- Wiley Registry of Mass Spectral Data, 8th ed.; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2008.
- MassFinder 3.1 Mass Spectral Library “Terpenoids and Related Constituents of Essential oils”; Hochmuth Scientific Consulting: Hamburg, Germany, 2007.
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy, 4th ed; Allured Publishing Corporation: Carol Stream, IL, USA, 2007. [Google Scholar]
- Joulain, D.; Konig, W.A. The Atlas of Spectral Data of Sesquiterpene Hydrocarbons; E.B.-Verlag: Hamburg, Germany, 1998. [Google Scholar]
- European Committee on Antimicrobial Susceptibility Testing (EUCAST). Breakpoint Tables for Interpretation of MICs and Zone Diameters, version 2.0; EUCAST Laboratory for Antimicrobial Susceptibility Testing: Växjö, Sweden, 1 January 2012. Available online: http://www.eucast.org (accessed on 16 August 2012).
- Ades, E.W.; Candal, F.J.; Swerlick, R.A.; George, V.G.; Summers, S.; Bosse, D.C.; Lawley, T.J. HMEC-1: Establishment of an immortalized human microvascular endothelial cell line. J. Investig. Dermatol. 1992, 99, 683–690. [Google Scholar]
- Wiktorowska-Owczarek, A. The effect of diclofenac on proliferation and production of growth factors by endothelial cells (HMEC-1) under hypoxia and inflammatory conditions. Acta Pharm. 2014, 64, 131–138. [Google Scholar]
- Sample Availability: Samples of the cinnamon, geranium and lavender essential oils (Pollena-Aroma, Warsaw, Poland) are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sienkiewicz, M.; Głowacka, A.; Kowalczyk, E.; Wiktorowska-Owczarek, A.; Jóźwiak-Bębenista, M.; Łysakowska, M. The Biological Activities of Cinnamon, Geranium and Lavender Essential Oils. Molecules 2014, 19, 20929-20940. https://doi.org/10.3390/molecules191220929
Sienkiewicz M, Głowacka A, Kowalczyk E, Wiktorowska-Owczarek A, Jóźwiak-Bębenista M, Łysakowska M. The Biological Activities of Cinnamon, Geranium and Lavender Essential Oils. Molecules. 2014; 19(12):20929-20940. https://doi.org/10.3390/molecules191220929
Chicago/Turabian StyleSienkiewicz, Monika, Anna Głowacka, Edward Kowalczyk, Anna Wiktorowska-Owczarek, Marta Jóźwiak-Bębenista, and Monika Łysakowska. 2014. "The Biological Activities of Cinnamon, Geranium and Lavender Essential Oils" Molecules 19, no. 12: 20929-20940. https://doi.org/10.3390/molecules191220929
APA StyleSienkiewicz, M., Głowacka, A., Kowalczyk, E., Wiktorowska-Owczarek, A., Jóźwiak-Bębenista, M., & Łysakowska, M. (2014). The Biological Activities of Cinnamon, Geranium and Lavender Essential Oils. Molecules, 19(12), 20929-20940. https://doi.org/10.3390/molecules191220929





