A Review on Visible Light Active Perovskite-Based Photocatalysts
Abstract
:1. Introduction
2. Overview of Photocatalytic Reactions
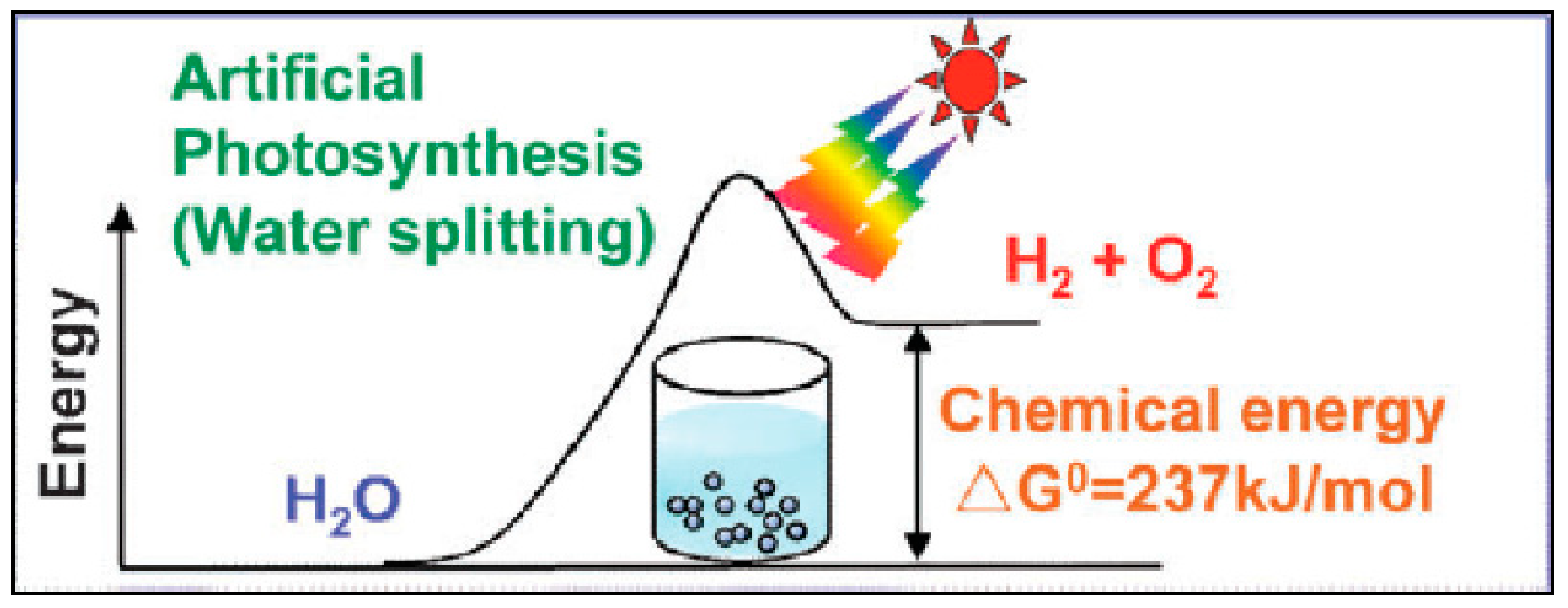
2.1. Photocatalytic Water Splitting
2.2. Photooxidation of Organic Molecules
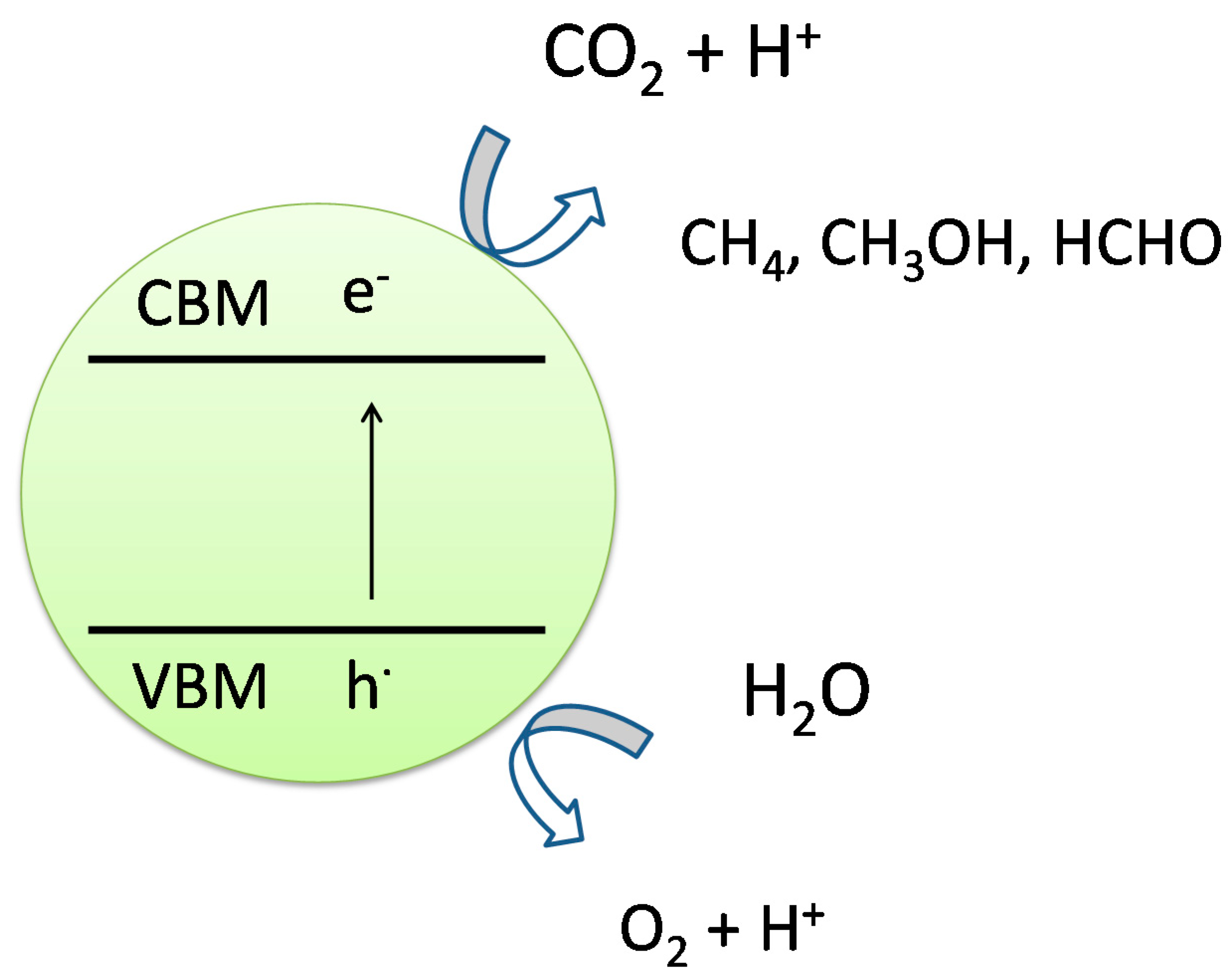
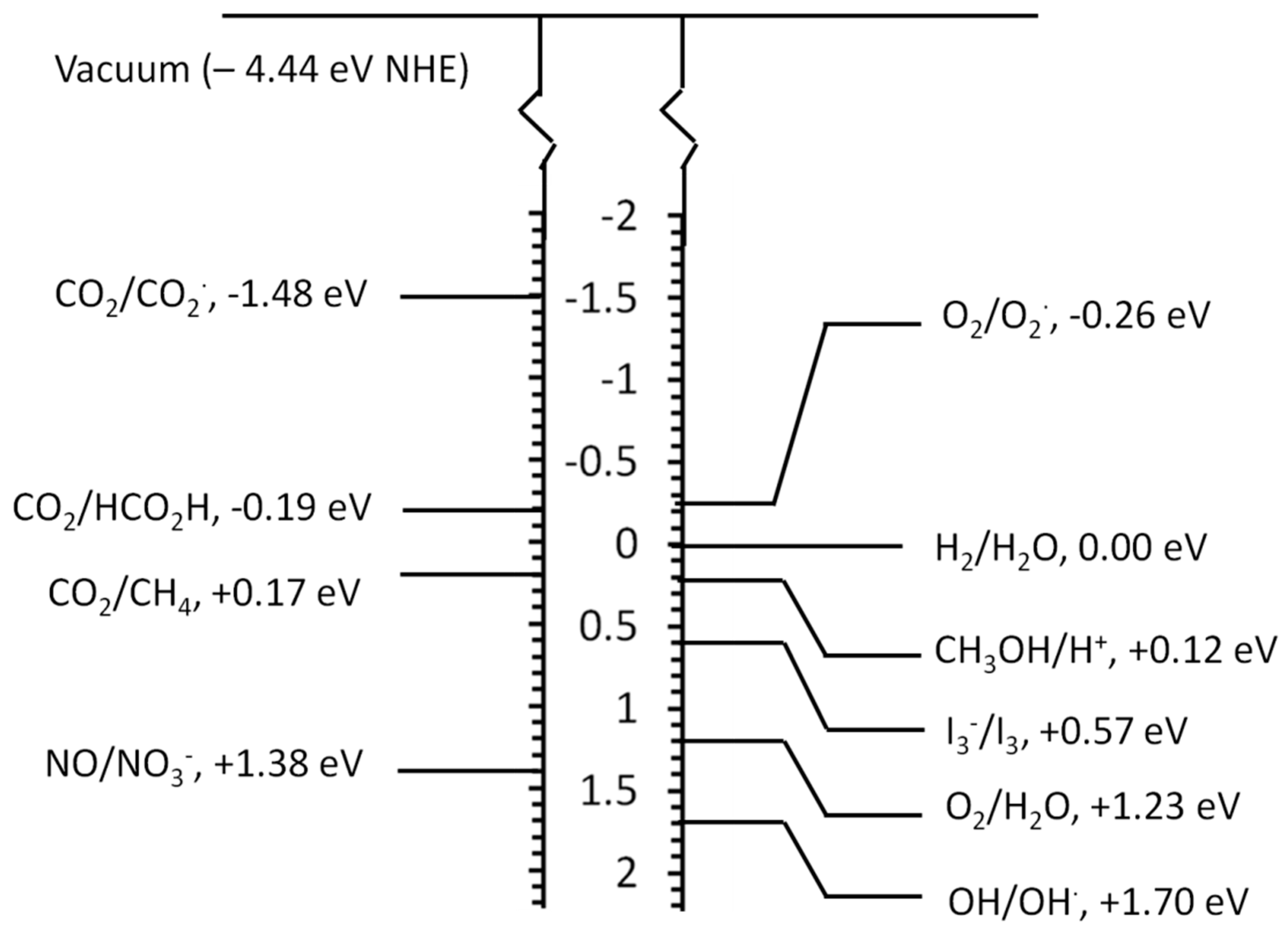
2.3. Photocatalytic Conversion of CO2 to Fuels
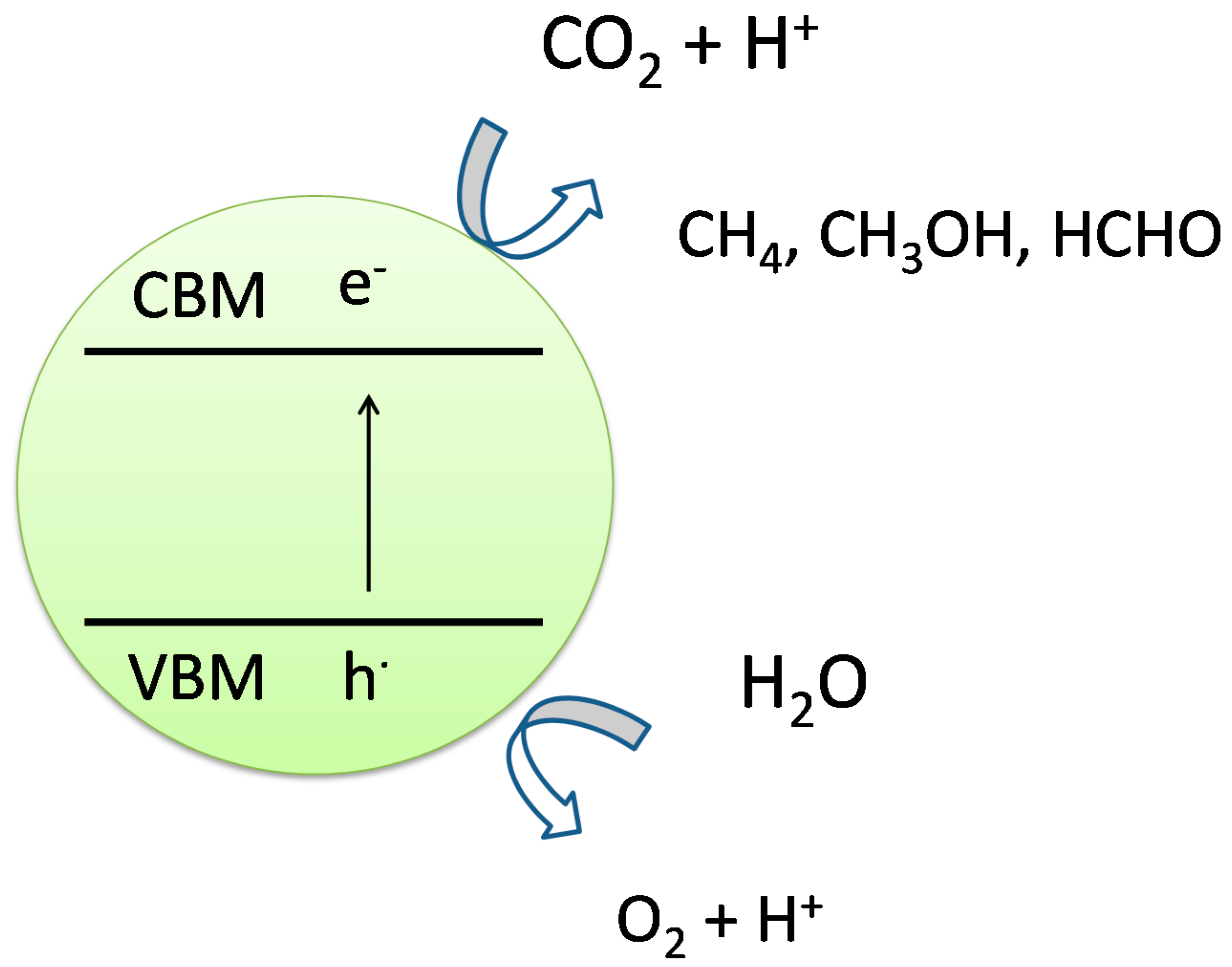
2.4. Photocatalytic Nitrogen Fixation
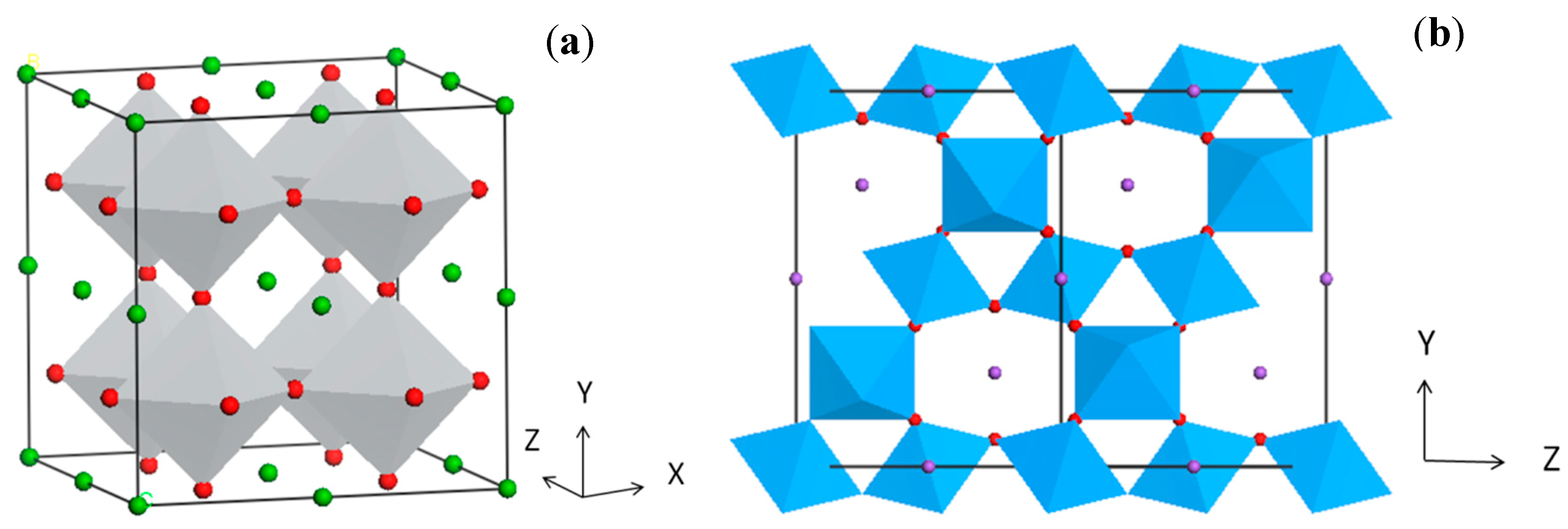
3. Simple Perovskites with Visible Light Response
3.1. Titanate Perovskites
3.1.1. SrTiO3
3.1.2. BaTiO3
3.1.3. CaTiO3
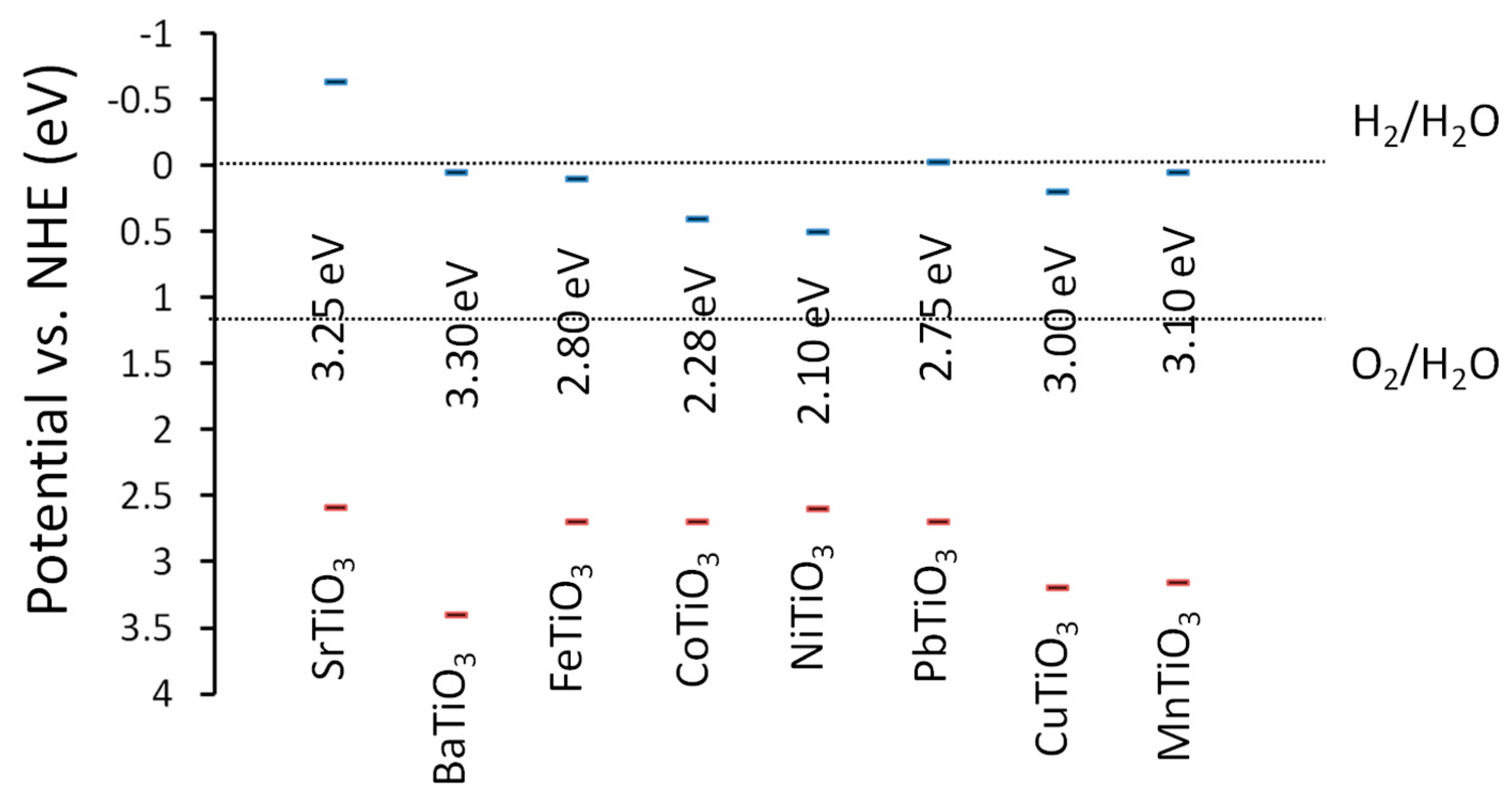
3.1.4. CoTiO3
3.1.5. NiTiO3
3.1.6. FeTiO3
3.1.7. CdTiO3
3.1.8. PbTiO3
3.1.9. MnTiO3
| Material System | Irradiation (nm) | Photocatalytic Performance | Experimental Details | Ref. |
|---|---|---|---|---|
| 1% Rh doped SrTiO3 (0.5% Pt) | 420–800 | H2 at 48.1 µmol·h−1 with sacrificial agent | 20% methanol, 50 mg in 50 mL of solution | [64] |
| Rh: SrTiO3: BiVO4 | >420 | Z scheme Water splitting. H2 at 128, O2 at 61 µmol·h−1 | 4.2% Efficiency, 50 mg 120 mL (FeCl3 shuttle) | [46] |
| Cr-Sb co-doped SrTiO3, (0.3% Pt) | >420 | H2 at 78, O2 at 0.9 µmol·h−1 with sacrificial agents | in aqueous methanol and AgNO3 solution | [65] |
| MCo1/3Nb2/3O3 (0.2% Pt) | >420 | H2 at 1.4 µmol·h−1 with sacrificial agent | 500 mg catalyst in 50 mL methanol, 220 mL water, | [66] |
| Sr1-xNbO3(1% Pt) | >420 | H2 at 44.8 µmol·h−1 with sacrificial agent | 0.025M oxalic acid, 0.1g catalyst in 200 mL, | [67] |
| AgNbO3-SrTiO3 | >420 | O2 at 162 µmol·h−1 with sacrificial agent | 0.5 g catalyst in 275 mL AgNO3 solution, | [49] |
| LaFeO3 (Pt co-catalyst) | 400–700 | H2 at 3315 µmol·h−1 with sacrificial agent | H2 = 3315, µmol·h−1,1 mg in 20 mL of ethanol | [68] |
| CaTi1_xCuxO3 (x = 0.02), NiOx co-catalyst | >400 | H2 at 22.7 µmol·h−1 with sacrificial agent | 100 mg catalyst in 420 mL methanol solution | [53] |
| PrFeO3, (Pt co-catalyst) | 200W Tungsten source | H2 at 2847 µmol·h−1 with sacrificial agent | 1 mg in 20 mL ethanol solution | [69] |
| Bi doped NaTaO3 | >400 | H2 at 59.48 µmol·h−1 with sacrificial agent | 100 mg catalyst in 210 mL of methanol solution | [70] |
| GdCrO3—Gd2Ti2O7 composite | >420 | H2 at 246.3 µmol·h−1 with sacrificial agent | 4.1% apparent quantum efficiency, methanol solution | [71] |
| CoTiO3 | >420 | O2 at 64.6 µmol·h−1 with sacrificial agent | 100 mg in 100 mL 0.04M AgNO3 and La2O3 solution, 420 nm | [57] |
3.2. Tantalate Perovskites
3.2.1. NaTaO3
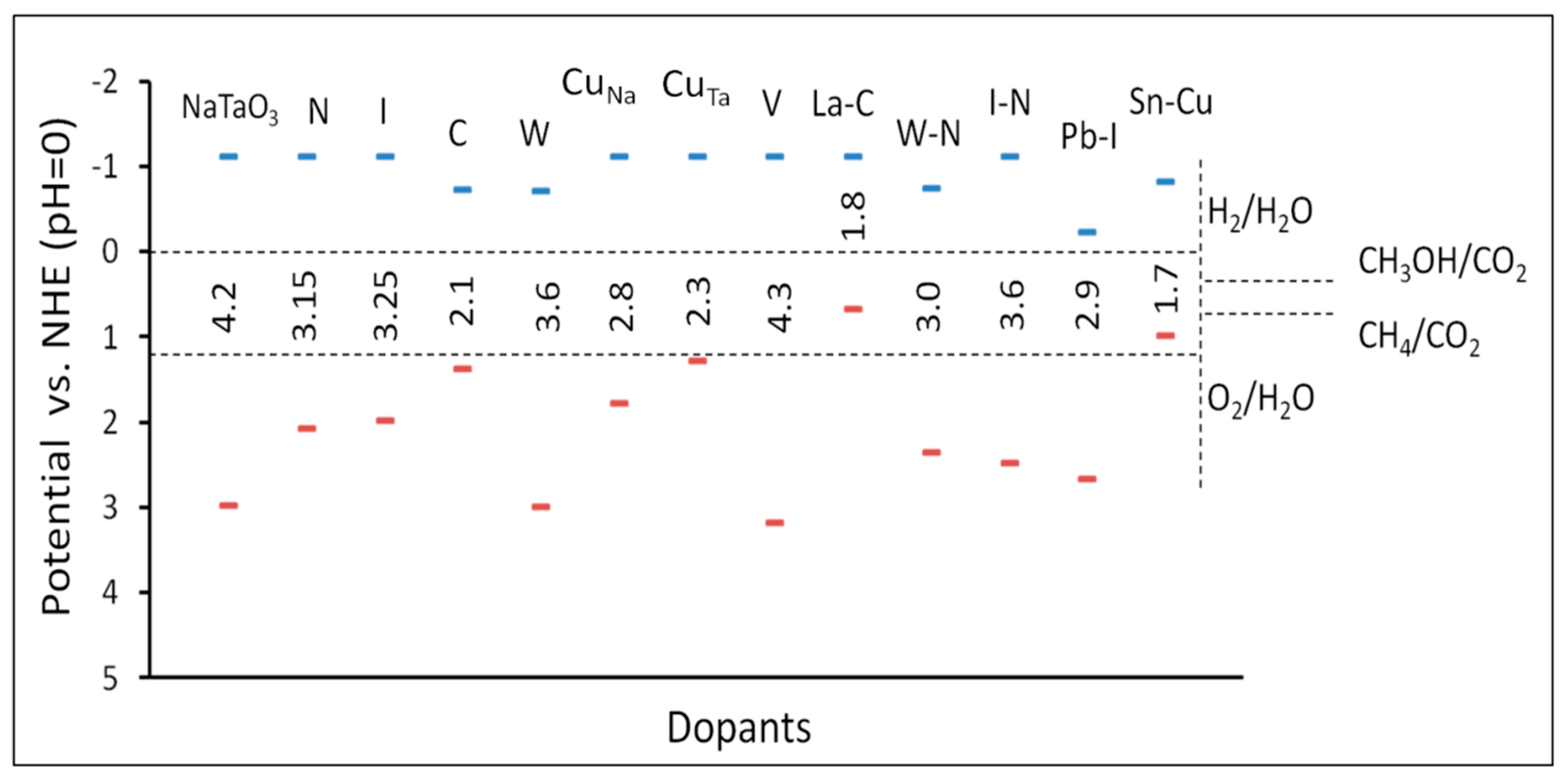
3.2.2. AgTaO3
3.2.3. KTaO3
3.3. Vanadium and Niobium Based Perovskites
3.3.1. KNbO3 and NaNbO3
3.3.2. AgNbO3
3.3.3. AgVO3
3.3.4. CuNbO3
3.4. Ferrite Perovskites
3.4.1. LaFeO3
3.4.2. BiFeO3
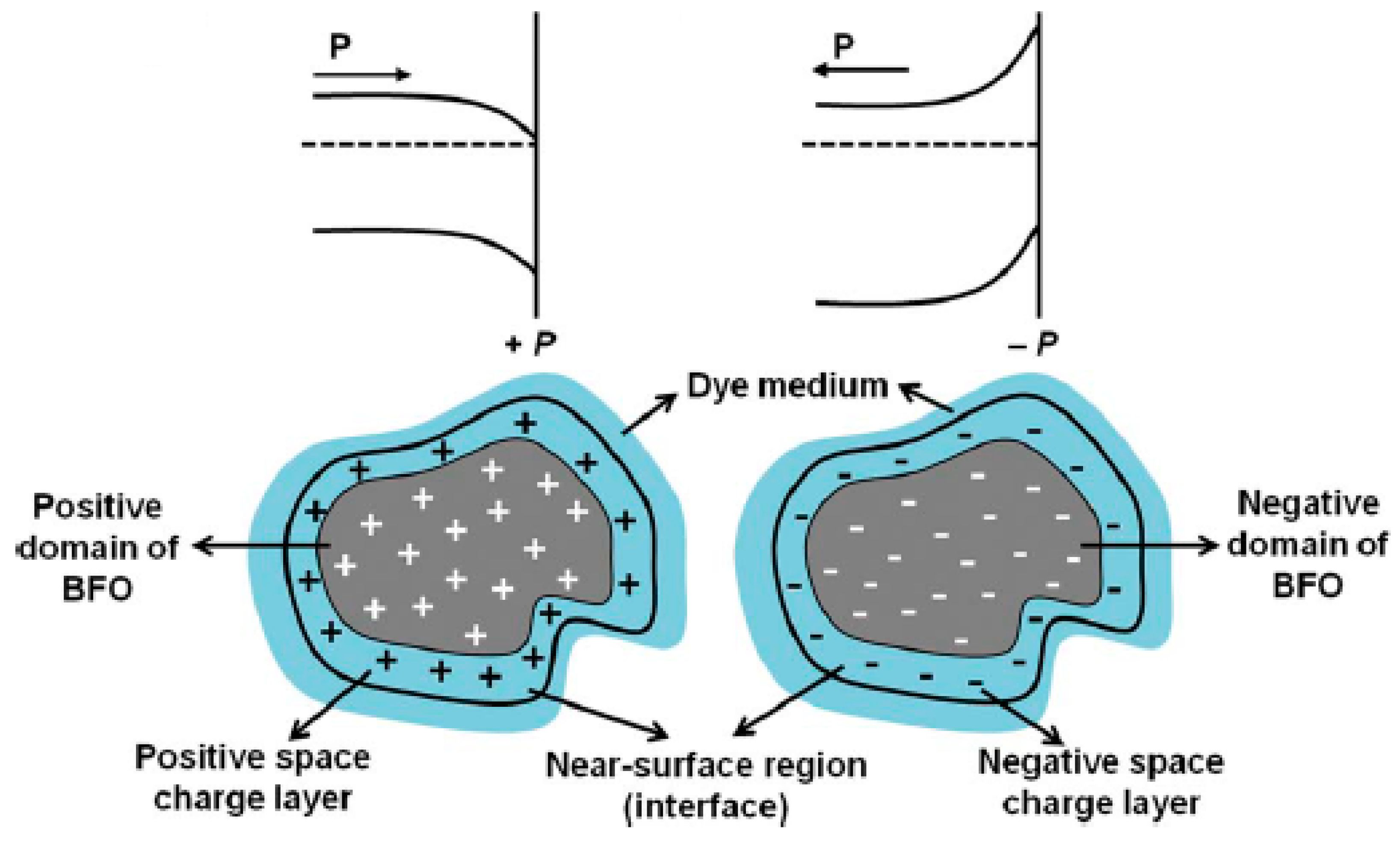
3.4.3. GaFeO3
3.4.4. YFeO3
3.4.5. PrFeO3
3.4.6. AlFeO3
3.5. Other Perovskite Systems
4. Complex Perovskite Materials
4.1. Double Perovskites
4.1.1. Sr2FeNbO6
4.1.2. La2FeTiO6
4.1.3. Other Double Perovskites
4.2. Mixed Oxides
5. Summary and Outlook
| Materials System | Band Gap (eV) | Photocatalytic Tests Reported | Ref. |
|---|---|---|---|
| Ga doped BiFeO3 | 2.18–2.50 | Enhanced degradation of rhodamine B compared to pristine BiFeO3 | [106] |
| LaFeO3 | 2.10 | Nanospheres show higher rates of rhodamine B degradation than nanocubes and nanorods | [103] |
| YFeO3 | 2.43 | Rhodamine B degradation rate higher than P25 (>400 nm) | [110] |
| NaBiO3 | 2.60 | Bleaching rate of Methylene Blue higher than N doped TiO2. (>400 nm) | [113] |
| AgSbO3 | 2.58 | Eddicient degradation of Rh B. MB, 4-chlorophenol (>420 nm) | [119,136] |
| AgBiO3 | 2.50 | Inhibition of Microcystis | [115] |
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Maeda, K. Photocatalytic water splitting using semiconductor particles: History and recent developments. J. Photchem. Photobiol. C 2011, 12, 237–268. [Google Scholar] [CrossRef]
- Qu, Y.; Duan, X. Progress, challenge and perspective of heterogeneous photocatalysts. Chem. Soc. Rev. 2013, 42, 2568–2580. [Google Scholar]
- Hou, W.; Cronin, S.B. A Review of Surface Plasmon Resonance-Enhanced Photocatalysis. Adv. Funct. Mat. 2013, 23, 1612–1619. [Google Scholar] [CrossRef]
- Osterloh, F.E. Inorganic nanostructures for photoelectrochemical and photocatalytic water splitting. Chem. Soc. Rev. 2013, 42, 2294–2320. [Google Scholar] [CrossRef] [PubMed]
- Cook, T.R.; Dogutan, D.K.; Reece, S.Y.; Surendranath, Y.; Teets, T.S.; Nocera, D.G. Solar energy supply and storage for the legacy and nonlegacy worlds. Chem. Rev. 2010, 110, 6474–6502. [Google Scholar] [CrossRef] [PubMed]
- Izumi, Y. Recent advances in the photocatalytic conversion of carbon dioxide to fuels with water and/or hydrogen using solar energy and beyond. Coord. Chem. Rev. 2013, 257, 171–186. [Google Scholar] [CrossRef]
- Tahir, M.; Amin, N.S. Recycling of carbon dioxide to renewable fuels by photocatalysis: Prospects and challenges. Renew. Sust. Energy Rev. 2013, 25, 560–579. [Google Scholar] [CrossRef]
- Tan, Y.N.; Wong, C.L.; Mohamed, A.R. An Overview on the Photocatalytic Activity of Nano-Doped-TiO2 in the Degradation of Organic Pollutants. ISRN Mat. Sci. 2011, 2011, 18. [Google Scholar]
- Pelaez, M.; Nolan, N.T.; Pillai, S.C.; Seery, M.K.; Falaras, P.; Kontos, A.G.; Dunlop, P.S.M.; Hamilton, J.W.J.; Byrne, J.A.; O’shea, K.; et al. A review on the visible light active titanium dioxide photocatalysts for environmental applications. Appl. Catal. B 2012, 125, 331–349. [Google Scholar] [CrossRef]
- Dalrymple, O.K.; Stefanakos, E.; Trotz, M.A.; Goswami, D.Y. A review of the mechanisms and modeling of photocatalytic disinfection. Appl. Catal. B 2010, 98, 27–38. [Google Scholar] [CrossRef]
- Zhang, Z.; Gamage, J. Applications of photocatalytic disinfection. Int. J. Photoenergy 2010. [Google Scholar] [CrossRef]
- Konstantinou, I.K.; Albanis, T.A. TiO2-assisted photocatalytic degradation of azo dyes in aqueous solution: Kinetic and mechanistic investigations: A review. Appl. Catal. B 2004, 49, 1–14. [Google Scholar] [CrossRef]
- Ahmed, S.; Rasul, M.G.; Martens, W.; Brown, R.; Hashib, M.A. Advances in Heterogeneous Photocatalytic Degradation of Phenols and Dyes in Wastewater: A Review. Water Air Soil Pollut. 2011, 215, 3–29. [Google Scholar] [CrossRef] [Green Version]
- Rajesh, J.T.; Praveen, K.S.; Ramchandra, G.K.; Raksh, V.J. Photocatalytic degradation of dyes and organic contaminants in water using nanocrystalline anatase and rutile TiO2. Sci. Technol. Adv. Mat. 2007, 8, 455. [Google Scholar] [CrossRef]
- Bickley, R.I.; Vishwanathan, V. Photocatalytically induced fixation of molecular nitrogen by near UV radiation. Nature 1979, 280, 306–308. [Google Scholar] [CrossRef]
- Guo, C.; Wu, X.; Yan, M.; Dong, Q.; Yin, S.; Sato, T.; Liu, S. The visible-light driven photocatalytic destruction of NOx using mesoporous TiO2 spheres synthesized via a “water-controlled release process”. Nanoscale 2013, 5, 8184–8191. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Yang, X. Photocatalytic oxidation for indoor air purification: A literature review. Build. Environ. 2003, 38, 645–654. [Google Scholar] [CrossRef]
- Lasek, J.; Yu, Y.-H.; Wu, J.C.S. Removal of NOx by photocatalytic processes. J. Photchem. Photobiol. C 2013, 14, 29–52. [Google Scholar] [CrossRef]
- Wang, H.; Wu, Z.; Zhao, W.; Guan, B. Photocatalytic oxidation of nitrogen oxides using TiO2 loading on woven glass fabric. Chemosphere 2007, 66, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Bhalla, A.S.; Guo, R.; Roy, R. The perovskite structure–a review of its role in ceramic science and technology. Mat. Res. Innov. 2000, 4, 3–26. [Google Scholar] [CrossRef]
- Damjanovic, D. Piezoelectric properties of perovskite ferroelectrics: unsolved problems and future research. Ann. Chim.-Sci. Mat. 2001, 26, 99–106. [Google Scholar] [CrossRef]
- Nuraje, N.; Su, K. Perovskite ferroelectric nanomaterials. Nanoscale 2013, 5, 8752–8780. [Google Scholar] [CrossRef] [PubMed]
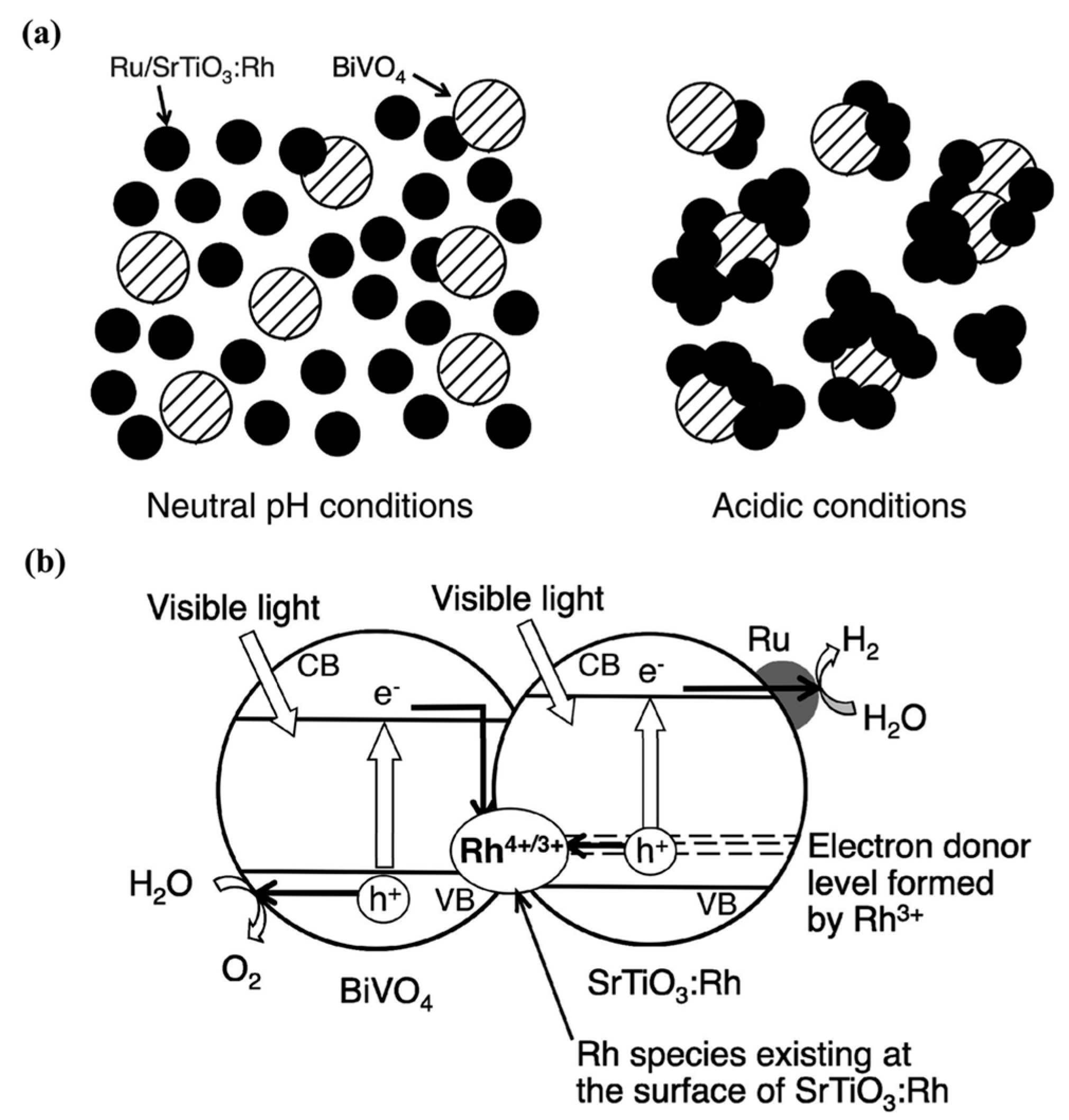
- Jia, Q.; Iwase, A.; Kudo, A. BiVO4-Ru/SrTiO3:Rh composite Z-scheme photocatalyst for solar water splitting. Chem. Sci. 2014, 5, 1513–1519. [Google Scholar] [CrossRef]
- Sayama, K.; Mukasa, K.; Abe, R.; Abe, Y.; Arakawa, H. Stoichiometric water splitting into H2 and O2 using a mixture of two different photocatalysts and an IO3−/I− shuttle redox mediator under visible light irradiation. Chem. Commun. 2001, 23, 2416–2417. [Google Scholar] [CrossRef]
- Zhang, W.F.; Tang, J.; Ye, J. Photoluminescence and photocatalytic properties of SrSnO3 perovskite. Chem. Phys. Lett. 2006, 418, 174–178. [Google Scholar] [CrossRef]
- Lin, W.H.; Cheng, C.; Hu, C.C.; Teng, H. NaTaO3 photocatalysts of different crystalline structures for water splitting into H2 and O2. Appl. Phys. Lett. 2006, 89, 211904. [Google Scholar] [CrossRef]
- Shi, J.; Guo, L. ABO3-based photocatalysts for water splitting. Prog. Nat. Sci. Mat. Int. 2012, 22, 592–615. [Google Scholar] [CrossRef]
- Wang, W.N.; Soulis, J.; Jeffrey Yang, Y.; Biswas, P. Comparison of CO2 photoreduction systems: A review. Aerosol Air Qual. Res. 2014, 14, 533–549. [Google Scholar]
- Tang, J.; Durrant, J.R.; Klug, D.R. Mechanism of Photocatalytic Water Splitting in TiO2. Reaction of Water with Photoholes, Importance of Charge Carrier Dynamics, and Evidence for Four-Hole Chemistry. J. Am. Chem. Soc. 2008, 130, 13885–13891. [Google Scholar]
- Kudo, A.; Miseki, Y. Heterogeneous photocatalyst materials for water splitting. Chem. Soc. Rev. 2009, 38, 253–278. [Google Scholar] [CrossRef] [PubMed]
- Houas, A.; Lachheb, H.; Ksibi, M.; Elaloui, E.; Guillard, C.; Herrmann, J.-M. Photocatalytic degradation pathway of methylene blue in water. Appl. Catal. B 2001, 31, 145–157. [Google Scholar] [CrossRef]
- Cho, M.; Chung, H.; Choi, W.; Yoon, J. Linear correlation between inactivation of E. coli and OH radical concentration in TiO2 photocatalytic disinfection. Water Res. 2004, 38, 1069–1077. [Google Scholar]
- Turchi, C.S.; Ollis, D.F. Photocatalytic degradation of organic water contaminants: Mechanisms involving hydroxyl radical attack. J. Catal. 1990, 122, 178–192. [Google Scholar] [CrossRef]
- Habisreutinger, S.N.; Schmidt-Mende, L.; Stolarczyk, J.K. Photocatalytic reduction of CO2 on TiO2 and other semiconductors. Angew. Chem. Int. Ed. 2013, 52, 7372–7408. [Google Scholar] [CrossRef]
- Rusina, O.; Linnik, O.; Eremenko, A.; Kisch, H. Nitrogen Photofixation on Nanostructured Iron Titanate Films. Chem. Eur. J. 2003, 9, 561–565. [Google Scholar] [CrossRef] [PubMed]
- Rusina, O.; Macyk, W.; Kisch, H. Photoelectrochemical Properties of a Dinitrogen-Fixing Iron Titanate Thin Film. J. Phys. Chem. B 2005, 109, 10858–10862. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Zhang, L.; Ruther, R.E.; Hamers, R.J. Photo-illuminated diamond as a solid-state source of solvated electrons in water for nitrogen reduction. Nat. Mater. 2013, 12, 836–841. [Google Scholar] [CrossRef] [PubMed]
- Kavan, L.; Grätzel, M.; Gilbert, S.; Klemenz, C.; Scheel, H. Electrochemical and photoelectrochemical investigation of single-crystal anatase. J. Am. Chem. Soc. 1996, 118, 6716–6723. [Google Scholar] [CrossRef]
- Van Benthem, K.; Elsässer, C.; French, R.H. Bulk electronic structure of SrTiO3: Experiment and theory. J. Appl. Phys. 2001, 90, 6156–6164. [Google Scholar] [CrossRef]
- Townsend, T.K.; Browning, N.D.; Osterloh, F.E. Overall photocatalytic water splitting with NiOx-SrTiO3—A revised mechanism. Energy Environ. Sci. 2012, 5, 9543–9550. [Google Scholar] [CrossRef]
- Konta, R.; Ishii, T.; Kato, H.; Kudo, A. Photocatalytic Activities of Noble Metal Ion Doped SrTiO3 under Visible Light Irradiation. J. Phys. Chem. B 2004, 108, 8992–8995. [Google Scholar] [CrossRef]
- Chen, H.-C.; Huang, C.-W.; Wu, J.C.S.; Lin, S.-T. Theoretical Investigation of the Metal-Doped SrTiO3 Photocatalysts for Water Splitting. J. Phys. Chem. C 2012, 116, 7897–7903. [Google Scholar] [CrossRef]
- Iwashina, K.; Kudo, A. Rh-Doped SrTiO3 Photocatalyst Electrode Showing Cathodic Photocurrent for Water Splitting under Visible-Light Irradiation. J. Am. Chem. Soc. 2011, 133, 13272–13275. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, Y.; Kato, H.; Kudo, A. [Co(bpy)3]3+/2+ and [Co(phen)3]3+/2+ Electron Mediators for Overall Water Splitting under Sunlight Irradiation Using Z-Scheme Photocatalyst System. J. Am. Chem. Soc. 2013, 135, 5441–5449. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, Y.; Nemoto, H.; Saito, K.; Kudo, A. Solar Water Splitting Using Powdered Photocatalysts Driven by Z-Schematic Interparticle Electron Transfer without an Electron Mediator. J. Phys. Chem. C 2009, 113, 17536–17542. [Google Scholar] [CrossRef]
- Kato, H.; Sasaki, Y.; Shirakura, N.; Kudo, A. Synthesis of highly active rhodium-doped SrTiO3 powders in Z-scheme systems for visible-light-driven photocatalytic overall water splitting. J. Mater. Chem. A 2013, 1, 12327–12333. [Google Scholar] [CrossRef]
- Asai, R.; Nemoto, H.; Jia, Q.; Saito, K.; Iwase, A.; Kudo, A. A visible light responsive rhodium and antimony-codoped SrTiO3 powdered photocatalyst loaded with an IrO2 cocatalyst for solar water splitting. Chem. Commun. 2014, 50, 2543–2546. [Google Scholar] [CrossRef]
- Jia, Y.; Shen, S.; Wang, D.; Wang, X.; Shi, J.; Zhang, F.; Han, H.; Li, C. Composite Sr2TiO4/SrTiO3(La,Cr) heterojunction based photocatalyst for hydrogen production under visible light irradiation. J. Mater. Chem. A 2013, 1, 7905–7912. [Google Scholar] [CrossRef]
- Wang, D.; Kako, T.; Ye, J. New Series of Solid-Solution Semiconductors (AgNbO3)1−x(SrTiO3)x with Modulated Band Structure and Enhanced Visible-Light Photocatalytic Activity. J. Phys. Chem. C 2009, 113, 3785–3792. [Google Scholar] [CrossRef]
- Fu, Q.; He, T.; Li, J.L.; Yang, G.W. Band-engineered SrTiO3 nanowires for visible light photocatalysis. J. Appl. Phys. 2012, 112, 104322. [Google Scholar] [CrossRef]
- Wunderlich, W.; Ohta, H.; Koumoto, K. Enhanced effective mass in doped SrTiO3 and related perovskites. Physica B 2009, 404, 2202–2212. [Google Scholar] [CrossRef]
- Maeda, K. Rhodium-Doped Barium Titanate Perovskite as a Stable p-Type Semiconductor Photocatalyst for Hydrogen Evolution under Visible Light. ACS App. Mater. Interfaces 2014, 6, 2167–2173. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, G.; Li, Y.; Teng, Y. Electronic structure and photocatalytic properties of copper-doped CaTiO3. Int. J. Hydrog. Energy 2010, 35, 2713–2716. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, G.; He, X.; Xu, J. Electronic structure and photocatalytic properties of Ag–La codoped CaTiO3. J. Alloys Compd. 2012, 516, 91–95. [Google Scholar] [CrossRef]
- Fu, Q.; Li, J.L.; He, T.; Yang, G.W. Band-engineered CaTiO3 nanowires for visible light photocatalysis. J. Appl. Phys. 2013, 113, 104303. [Google Scholar] [CrossRef]
- Xu, Y.; Schoonen, M.A. The absolute energy positions of conduction and valence bands of selected semiconducting minerals. Am. Miner. 2000, 85, 543–556. [Google Scholar]
- Qu, Y.; Zhou, W.; Fu, H. Porous Cobalt Titanate Nanorod: A New Candidate for Visible Light-Driven Photocatalytic Water Oxidation. ChemCatChem 2014, 6, 265–270. [Google Scholar] [CrossRef]
- Rawal, S.B.; Bera, S.; Lee, D.; Jang, D.-J.; Lee, W.I. Design of visible-light photocatalysts by coupling of narrow bandgap semiconductors and TiO2: Effect of their relative energy band positions on the photocatalytic efficiency. Catal. Sci. Technol. 2013, 3, 1822–1830. [Google Scholar] [CrossRef]
- Qu, Y.; Zhou, W.; Ren, Z.; Du, S.; Meng, X.; Tian, G.; Pan, K.; Wang, G.; Fu, H. Facile preparation of porous NiTiO3 nanorods with enhanced visible-light-driven photocatalytic performance. J. Mater. Chem. 2012, 22, 16471–16476. [Google Scholar] [CrossRef]
- Kim, Y.J.; Gao, B.; Han, S.Y.; Jung, M.H.; Chakraborty, A.K.; Ko, T.; Lee, C.; Lee, W.I. Heterojunction of FeTiO3 Nanodisc and TiO2 Nanoparticle for a Novel Visible Light Photocatalyst. J. Phys. Chem. C 2009, 113, 19179–19184. [Google Scholar] [CrossRef]
- Hassan, M.A.; Amna, T.; Khil, M.-S. Synthesis of High aspect ratio CdTiO3 nanofibers via electrospinning: characterization and photocatalytic activity. Ceram. Int. 2014, 40, 423–427. [Google Scholar] [CrossRef]
- Li, L.; Zhang, Y.; Schultz, A.M.; Liu, X.; Salvador, P.A.; Rohrer, G.S. Visible light photochemical activity of heterostructured PbTiO3-TiO2 core-shell particles. Catal. Sci. Technol. 2012, 2, 1945–1952. [Google Scholar] [CrossRef]
- Dong, W.; Wang, D.; Jiang, L.; Zhu, H.; Huang, H.; Li, J.; Zhao, H.; Li, C.; Chen, B.; Deng, G. Synthesis of F doping MnTiO3 nanodiscs and their photocatalytic property under visible light. Mater. Lett. 2013, 98, 265–268. [Google Scholar] [CrossRef]
- Shen, P.; Lofaro, J.C., Jr.; Woerner, W.R.; White, M.G.; Su, D.; Orlov, A. Photocatalytic activity of hydrogen evolution over Rh doped SrTiO3 prepared by polymerizable complex method. Chem. Eng. J. 2013, 223, 200–208. [Google Scholar] [CrossRef]
- Kato, H.; Kudo, A. Visible-Light-Response and Photocatalytic Activities of TiO2 and SrTiO3Photocatalysts Codoped with Antimony and Chromium. J. Phys. Chem. B 2002, 106, 5029–5034. [Google Scholar] [CrossRef]
- Yin, J.; Zou, Z.; Ye, J. A Novel Series of the New Visible-Light-Driven Photocatalysts MCo1/3Nb2/3O3 (M = Ca, Sr, and Ba) with Special Electronic Structures. J. Phys. Chem. B 2003, 107, 4936–4941. [Google Scholar] [CrossRef]
- Xu, X.; Randorn, C.; Efstathiou, P.; Irvine, J.T.S. A red metallic oxide photocatalyst. Nat. Mater. 2012, 11, 595–598. [Google Scholar] [CrossRef] [PubMed]
- Tijare, S.N.; Joshi, M.V.; Padole, P.S.; Mangrulkar, P.A.; Rayalu, S.S.; Labhsetwar, N.K. Photocatalytic hydrogen generation through water splitting on nano-crystalline LaFeO3 perovskite. Int. J. Hydrog. Energy 2012, 37, 10451–10456. [Google Scholar] [CrossRef]
- Tijare, S.N.; Bakardjieva, S.; Subrt, J.; Joshi, M.V.; Rayalu, S.S.; Hishita, S.; Labhsetwar, N. Synthesis and visible light photocatalytic activity of nanocrystalline PrFeO3 perovskite for hydrogen generation in ethanol–water system. J. Chem. Sci. 2014, 126, 517–525. [Google Scholar] [CrossRef]
- Li, Z.; Wang, Y.; Liu, J.; Chen, G.; Li, Y.; Zhou, C. Photocatalytic hydrogen production from aqueous methanol solutions under visible light over Na(BixTa1−x)O3 solid-solution. Int. J. Hydrog. Energy 2009, 34, 147–152. [Google Scholar] [CrossRef]
- Parida, K.M.; Nashim, A.; Mahanta, S.K. Visible-light driven Gd2Ti2O7/GdCrO3 composite for hydrogen evolution. J. Chem. Soc. Dalton Trans. 2011, 40, 12839–12845. [Google Scholar] [CrossRef]
- Kato, H.; Asakura, K.; Kudo, A. Highly efficient water splitting into H2 and O2 over lanthanum-doped NaTaO3 photocatalysts with high crystallinity and surface nanostructure. J. Am. Chem. Soc. 2003, 125, 3082–3089. [Google Scholar] [CrossRef] [PubMed]
- Yamakata, A.I.T.A.; Kato, H.; Kudo, A.; Onishi, H. Photodynamics of NaTaO3 Catalysts for Efficient Water Splitting. J. Phys. Chem. B 2003, 107, 14383–14387. [Google Scholar] [CrossRef]
- Kato, H.; Kudo, A. Water splitting into H2 and O2 on alkali tantalate photocatalysts ATaO3 (A = Li, Na, and K). J. Phys. Chem. B 2001, 105, 4285–4292. [Google Scholar] [CrossRef]
- Kanhere, P.D.; Zheng, J.; Chen, Z. Site Specific Optical and Photocatalytic Properties of Bi-Doped NaTaO3. J. Phys. Chem. C 2011, 115, 11846–11853. [Google Scholar] [CrossRef]
- Kanhere, P.; Zheng, J.; Chen, Z. Visible light driven photocatalytic hydrogen evolution and photophysical properties of Bi3+ doped NaTaO3. Int. J. Hydrog. Energy 2012, 37, 4889–4896. [Google Scholar] [CrossRef]
- Yi, Z.G.; Ye, J.H. Band gap tuning of Na1-xLaxTa1-xCoxO3 solid solutions for visible light photocatalysis. App. Phys. Lett. 2007, 91, 254108. [Google Scholar] [CrossRef]
- Yi, Z.G.; Ye, J.H. Band gap tuning of Na1-xLaxTa1-xCrxO3 for H2 generation from water under visible light irradiation. J. App. Phys. 2009, 106, 074910. [Google Scholar] [CrossRef]
- Yang, M.; Huang, X.; Yan, S.; Li, Z.; Yu, T.; Zou, Z. Improved hydrogen evolution activities under visible light irradiation over NaTaO3 codoped with lanthanum and chromium. Mater. Chem. Phys. 2010, 121, 506–510. [Google Scholar] [CrossRef]
- Iwase, A.; Saito, K.; Kudo, A. Sensitization of NaMO3 (M: Nb and Ta) photocatalysts with wide band gaps to visible light by Ir doping. Bull. Chem. Soc. Jpn. 2009, 82, 514–518. [Google Scholar] [CrossRef]
- Kanhere, P.; Nisar, J.; Tang, Y.; Pathak, B.; Ahuja, R.; Zheng, J.; Chen, Z. Electronic Structure, Optical Properties, and Photocatalytic Activities of LaFeO3–NaTaO3 Solid Solution. J. Phys. Chem. C 2012, 116, 22767–22773. [Google Scholar] [CrossRef]
- Zhao, Z.; Li, R.; Li, Z.; Zou, Z. Photocatalytic activity of La-N-codoped NaTaO3 for H2 evolution from water under visible-light irradiation. J. Phys. D Appl. Phys. 2011, 44, 165401. [Google Scholar] [CrossRef]
- Han, P.; Wang, X.; Zhao, Y.H.; Tang, C. Electronic structure and optical properties of non-metals (N, F, P, Cl,S)—Doped cubic NaTaO3 by density functional theory. Adv. Mater. Res. 2009, 79–82, 1245–1248. [Google Scholar] [CrossRef]
- Zhou, X.; Shi, J.; Li, C. Effect of metal doping on electronic structure and visible light absorption of SrTiO3 and NaTaO3 (Metal = Mn, Fe, and Co). J. Phys. Chem. C 2011, 115, 8305–8311. [Google Scholar] [CrossRef]
- Kanhere, P.; Shenai, P.; Chakraborty, S.; Ahuja, R.; Zheng, J.; Chen, Z. Mono- and co-doped NaTaO3 for visible light photocatalysis. PCCP 2014, 16, 16085–16094. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Kanhere, P.; Chen, Z.; Nisar, J.; Pathak, B.; Ahuja, R. Anion-Doped NaTaO3 for Visible Light Photocatalysis. J. Phys. Chem. C 2013, 117, 22518–22524. [Google Scholar] [CrossRef]
- Ni, L.; Tanabe, M.; Irie, H. A visible-light-induced overall water-splitting photocatalyst: conduction-band-controlled silver tantalate. Chem. Commun. 2013, 49, 10094–10096. [Google Scholar] [CrossRef]
- Li, M.; Zhang, J.; Dang, W.; Cushing, S.K.; Guo, D.; Wu, N.; Yin, P. Photocatalytic hydrogen generation enhanced by band gap narrowing and improved charge carrier mobility in AgTaO3 by compensated co-doping. PCCP 2013, 15, 16220–16226. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.W.; Chen, G.; Li, Z.H.; Zhang, Z.G. Hydrothermal synthesis and photocatalytic properties of ATaO3 and ANbO3 (A = Na and K). Int. J. Hydrog. Energy 2007, 32, 2269–2272. [Google Scholar] [CrossRef]
- Shi, H.; Li, X.; Iwai, H.; Zou, Z.; Ye, J. 2-Propanol photodegradation over nitrogen-doped NaNbO3 powders under visible-light irradiation. J. Phys. Chem. Solids 2009, 70, 931–935. [Google Scholar]
- Wang, R.; Zhu, Y.; Qiu, Y.; Leung, C.-F.; He, J.; Liu, G.; Lau, T.-C. Synthesis of nitrogen-doped KNbO3 nanocubes with high photocatalytic activity for water splitting and degradation of organic pollutants under visible light. Chem. Eng. J. 2013, 226, 123–130. [Google Scholar]
- Liu, G.; Ji, S.; Yin, L.; Xu, G.; Fei, G.; Ye, C. Visible-light-driven photocatalysts: (La/Bi + N)-codoped NaNbO3 by first principles. J. Appl. Phys. 2011, 109, 063103. [Google Scholar] [CrossRef]
- Grinberg, I.; West, D.V.; Torres, M.; Gou, G.; Stein, D.M.; Wu, L.; Chen, G.; Gallo, E.M.; Akbashev, A.R.; Davies, P.K.; et al. Perovskite oxides for visible-light-absorbing ferroelectric and photovoltaic materials. Nature 2013, 503, 509–512. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Yan, S.; Wang, Z.; Wang, X.; Li, Z.; Ye, J.; Zou, Z. Synthesis and visible light photocatalytic property of polyhedron-shaped AgNbO3. J. Chem. Soc. Dalton Trans. 2009, 40, 8519–8524. [Google Scholar] [CrossRef]
- Li, G.; Kako, T.; Wang, D.; Zou, Z.; Ye, J. Enhanced photocatalytic activity of La-doped AgNbO3 under visible light irradiation. J. Chem. Soc. Dalton Trans. 2009, 13, 2423–2427. [Google Scholar] [CrossRef]
- Konta, R.; Kato, H.; Kobayashi, H.; Kudo, A. Photophysical properties and photocatalytic activities under visible light irradiation of silver vanadates. PCCP 2003, 5, 3061–3065. [Google Scholar] [CrossRef]
- Xu, J.; Hu, C.; Xi, Y.; Wan, B.; Zhang, C.; Zhang, Y. Synthesis and visible light photocatalytic activity of β-AgVO3 nanowires. Solid State Sci. 2012, 14, 535–539. [Google Scholar] [CrossRef]
- Sang, Y.; Kuai, L.; Chen, C.; Fang, Z.; Geng, B. Fabrication of a Visible-Light-Driven Plasmonic Photocatalyst of AgVO3@AgBr@Ag Nanobelt Heterostructures. ACS App. Mater. Interfaces 2014, 6, 5061–5068. [Google Scholar] [CrossRef]
- Ju, P.; Fan, H.; Zhang, B.; Shang, K.; Liu, T.; Ai, S.; Zhang, D. Enhanced photocatalytic activity of β-AgVO3 nanowires loaded with Ag nanoparticles under visible light irradiation. Sep. Purif. Technol. 2013, 109, 107–110. [Google Scholar] [CrossRef]
- Joshi, U.A.; Palasyuk, A.M.; Maggard, P.A. Photoelectrochemical Investigation and Electronic Structure of a p-Type CuNbO3 Photocathode. J. Phys. Chem. C 2011, 115, 13534–13539. [Google Scholar]
- Lin, Y.; Yuan, G.; Sheehan, S.; Zhou, S.; Wang, D. Hematite-based solar water splitting: Challenges and opportunities. Energy Environ. Sci. 2011, 4, 4862–4869. [Google Scholar] [CrossRef]
- Parida, K.M.; Reddy, K.H.; Martha, S.; Das, D.P.; Biswal, N. Fabrication of nanocrystalline LaFeO3: An efficient sol–gel auto-combustion assisted visible light responsive photocatalyst for water decomposition. Int. J. Hydrog. Energy 2010, 35, 12161–12168. [Google Scholar] [CrossRef]
- Thirumalairajan, S.; Girija, K.; Hebalkar, N.Y.; Mangalaraj, D.; Viswanathan, C.; Ponpandian, N. Shape evolution of perovskite LaFeO3 nanostructures: A systematic investigation of growth mechanism, properties and morphology dependent photocatalytic activities. RSC Adv. 2013, 3, 7549–7561. [Google Scholar] [CrossRef]
- Wei, Z.-X.; Wang, Y.; Liu, J.-P.; Xiao, C.-M.; Zeng, W.-W.; Ye, S.-B. Synthesis, magnetization, and photocatalytic activity of LaFeO3 and LaFe0.9Mn0.1O3−δ. J. Mater. Sci. 2013, 48, 1117–1126. [Google Scholar] [CrossRef]
- Gao, F.; Chen, X.Y.; Yin, K.B.; Dong, S.; Ren, Z.F.; Yuan, F.; Yu, T.; Zou, Z.G.; Liu, J.M. Visible-Light Photocatalytic Properties of Weak Magnetic BiFeO3 Nanoparticles. Adv. Mater. 2007, 19, 2889–2892. [Google Scholar] [CrossRef]
- Mohan, S.; Subramanian, B.; Bhaumik, I.; Gupta, P.K.; Jaisankar, S.N. Nanostructured Bi(1-x)Gd(x)FeO3—A multiferroic photocatalyst on its sunlight driven photocatalytic activity. RSC Adv. 2014, 4, 16871–16878. [Google Scholar] [CrossRef]
- Feng, Y.N.; Wang, H.C.; Luo, Y.D.; Shen, Y.; Lin, Y.H. Ferromagnetic and photocatalytic behaviors observed in Ca-doped BiFeO3 nanofibres. J. App. Phys. 2013, 113, 146101. [Google Scholar] [CrossRef]
- Madhu, C.; Bellakki, M.B.; Manivannan, V. Synthesis and characterization of cation-doped BiFeO3 materials for photocatalytic applications. Indian J. Eng. Mater. Sci. 2010, 17, 131–139. [Google Scholar]
- Dhanasekaran, P.; Gupta, N.M. Factors affecting the production of H2 by water splitting over a novel visible-light-driven photocatalyst GaFeO3. Int. J. Hydrog. Energy 2012, 37, 4897–4907. [Google Scholar] [CrossRef]
- Tang, P.; Chen, H.; Cao, F.; Pan, G. Magnetically recoverable and visible-light-driven nanocrystalline YFeO3 photocatalysts. Catal. Sci. Technol. 2011, 1, 1145–1148. [Google Scholar] [CrossRef]
- Yuan, Z.; Wang, Y.; Sun, Y.; Wang, J.; Bie, L.; Duan, Y. Sunlight-activated AlFeO3/TiO2 photocatalyst. Sci. China Ser. B 2006, 49, 67–74. [Google Scholar] [CrossRef]
- Takei, T.; Haramoto, R.; Dong, Q.; Kumada, N.; Yonesaki, Y.; Kinomura, N.; Mano, T.; Nishimoto, S.; Kameshima, Y.; Miyake, M. Photocatalytic activities of various pentavalent bismuthates under visible light irradiation. J. Solid State Chem. 2011, 184, 2017–2022. [Google Scholar] [CrossRef]
- Kako, T.; Zou, Z.; Katagiri, M.; Ye, J. Decomposition of Organic Compounds over NaBiO3 under Visible Light Irradiation. Chem. Mater. 2006, 19, 198–202. [Google Scholar] [CrossRef]
- Chang, X.; Yu, G.; Huang, J.; Li, Z.; Zhu, S.; Yu, P.; Cheng, C.; Deng, S.; Ji, G. Enhancement of photocatalytic activity over NaBiO3/BiOCl composite prepared by an in situ formation strategy. Catal. Today 2010, 153, 193–199. [Google Scholar] [CrossRef]
- Yu, X.; Zhou, J.; Wang, Z.; Cai, W. Preparation of visible light-responsive AgBiO3 bactericide and its control effect on the Microcystis aeruginosa. J. Photochem. Photobiol. 2010, 101, 265–270. [Google Scholar] [CrossRef]
- Sun, M.; Jiang, Y.; Li, F.; Xia, M.; Xue, B.; Liu, D. Dye degradation activity and stability of perovskite-type LaCoO3-x (x = 0~0.075). Mater. Trans. 2010, 51, 2208–2214. [Google Scholar] [CrossRef]
- Tang, P.; Sun, H.; Cao, F.; Yang, J.; Ni, S.; Chen, H. Visible-light driven LaNiO3 nanosized photocatalysts prepared by a sol-gel process. Adv. Mater. Res. 2011, 279, 83–87. [Google Scholar] [CrossRef]
- Li, J.; Zeng, J.; Jia, L.; Fang, W. Investigations on the effect of Cu2+/Cu1+ redox couples and oxygen vacancies on photocatalytic activity of treated LaNi1−xCuxO3 (x=0.1, 0.4, 0.5). Int. J. Hydrog. Energy 2010, 35, 12733–12740. [Google Scholar] [CrossRef]
- Singh, J.; Uma, S. Efficient Photocatalytic Degradation of Organic Compounds by Ilmenite AgSbO3 under Visible and UV Light Irradiation. J. Phys. Chem. C 2009, 113, 12483–12488. [Google Scholar] [CrossRef]
- Shi, J.; Ye, J.; Zhou, Z.; Li, M.; Guo, L. Hydrothermal Synthesis of Na0.5La0.5TiO3–LaCrO3 Solid-Solution Single-Crystal Nanocubes for Visible-Light-Driven Photocatalytic H2 Evolution. Chem. Eur. J. 2011, 17, 7858–7867. [Google Scholar]
- Borse, P.H.; Cho, C.R.; Yu, S.M.; Yoon, J.H.; Hong, T.E.; Bae, J.S.; Jeong, E.D.; Kim, H.G. Improved photolysis of water from ti incorporated double perovskite Sr2FeNbO6 lattice. Bull. Korean Chem. Soc. 2012, 33, 3407–3412. [Google Scholar] [CrossRef]
- Borse, P.H.; Lim, K.T.; Yoon, J.H.; Bae, J.S.; Ha, M.G.; Chung, E.H.; Jeong, E.D.; Kim, H.G. Investigation of the physico-chemical properties of Sr2FeNb1−xWxO6 (0.0 ≤ x ≤ 0.1) for visible-light photocatalytic water-splitting applications. J. Korean Phys. Soc. 2014, 64, 295–300. [Google Scholar]
- Hu, R.; Li, C.; Wang, X.; Sun, Y.; Jia, H.; Su, H.; Zhang, Y. Photocatalytic activities of LaFeO3 and La2FeTiO6 in p-chlorophenol degradation under visible light. Catal. Commun. 2012, 29, 35–39. [Google Scholar] [CrossRef]
- Hatakeyama, T.; Takeda, S.; Ishikawa, F.; Ohmura, A.; Nakayama, A.; Yamada, Y.; Matsushita, A.; Yea, J. Photocatalytic activities of Ba2RBiO6 (R = La, Ce, Nd, Sm, Eu, Gd, Dy) under visible light irradiation. J. Ceram. Soc. Jpn. 2010, 118, 91–95. [Google Scholar] [CrossRef]
- Clark, J.H.; Dyer, M.S.; Palgrave, R.G.; Ireland, C.P.; Darwent, J.R.; Claridge, J.B.; Rosseinsky, M.J. Visible light photo-oxidation of model pollutants using CaCu3Ti4O12: An experimental and theoretical study of optical properties, electronic structure, and selectivity. J. Am. Chem. Soc. 2011, 133, 1016–1032. [Google Scholar] [CrossRef]
- Iwakura, H.; Einaga, H.; Teraoka, Y. Photocatalytic Properties of Ordered Double Perovskite Oxides. J. Novel Carbon Resour. Sci. 2011, 3, 1–5. [Google Scholar]
- Zhu, S.; Fu, H.; Zhang, S.; Zhang, L.; Zhu, Y. Two-step synthesis of a novel visible-light-driven K2Ta2O6−xNx catalyst for the pollutant decomposition. J. Photochem. Photobiol. A 2008, 193, 33–41. [Google Scholar] [CrossRef]
- Kanhere, P.; Tang, Y.; Zheng, J.; Chen, Z. Synthesis, photophysical properties, and photocatalytic applications of Bi doped NaTaO3 and Bi doped Na2Ta2O6 nanoparticles. J. Phys. Chem. Solids 2013, 74, 1708–1713. [Google Scholar] [CrossRef]
- Hara, M.T.T.; Kondo, J.N.; Domen, K. Photocatalytic reduction of water by TaON under visible light irradiation. Catal. Today 2004, 90, 313–317. [Google Scholar] [CrossRef]
- Ishikawa, A.; Takata, T.; Kondo, J.N.; Hara, M.; Kobayashi, H.; Domen, K. Oxysulfide Sm2Ti2S2O5 as a Stable Photocatalyst for Water Oxidation and Reduction under Visible Light Irradiation (λ ≤ 650 nm). J. Am. Chem. Soc. 2002, 124, 13547–13553. [Google Scholar] [CrossRef] [PubMed]
- Siritanaratkul, B.; Maeda, K.; Hisatomi, T.; Domen, K. Synthesis and Photocatalytic Activity of Perovskite Niobium Oxynitrides with Wide Visible-Light Absorption Bands. ChemSusChem 2011, 4, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K.; Higashi, M.; Siritanaratkul, B.; Abe, R.; Domen, K. SrNbO2N as a Water-Splitting Photoanode with a Wide Visible-Light Absorption Band. J. Am. Chem. Soc. 2011, 133, 12334–12337. [Google Scholar] [CrossRef] [PubMed]
- Higashi, M.; Abe, R.; Takata, T.; Domen, K. Photocatalytic Overall Water Splitting under Visible Light Using ATaO2N (A = Ca, Sr, Ba) and WO3 in a IO3−/I− Shuttle Redox Mediated System. Chem. Mater. 2009, 21, 1543–1549. [Google Scholar] [CrossRef]
- Maeda, K.; Domen, K. Preparation of BaZrO3–BaTaO2N solid solutions and the photocatalytic activities for water reduction and oxidation under visible light. J. Catal. 2014, 310, 67–74. [Google Scholar] [CrossRef]
- Le Paven-Thivet, C.; Ishikawa, A.; Ziani, A.; Le Gendre, L.; Yoshida, M.; Kubota, J.; Tessier, F.; Domen, K. Photoelectrochemical Properties of Crystalline Perovskite Lanthanum Titanium Oxynitride Films under Visible Light. J. Phys. Chem. C 2009, 113, 6156–6162. [Google Scholar] [CrossRef]
- Kako, T.; Kikugawa, N.; Ye, J. Photocatalytic activities of AgSbO3 under visible light irradiation. Catal. Today 2008, 131, 197–202. [Google Scholar] [CrossRef]
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kanhere, P.; Chen, Z. A Review on Visible Light Active Perovskite-Based Photocatalysts. Molecules 2014, 19, 19995-20022. https://doi.org/10.3390/molecules191219995
Kanhere P, Chen Z. A Review on Visible Light Active Perovskite-Based Photocatalysts. Molecules. 2014; 19(12):19995-20022. https://doi.org/10.3390/molecules191219995
Chicago/Turabian StyleKanhere, Pushkar, and Zhong Chen. 2014. "A Review on Visible Light Active Perovskite-Based Photocatalysts" Molecules 19, no. 12: 19995-20022. https://doi.org/10.3390/molecules191219995
APA StyleKanhere, P., & Chen, Z. (2014). A Review on Visible Light Active Perovskite-Based Photocatalysts. Molecules, 19(12), 19995-20022. https://doi.org/10.3390/molecules191219995