A Review of Recent Research Progress on the Astragalus Genus
Abstract
:1. Introdution
2. Phytochemistry
2.1. Saponins
2.1.1. Cycloartane-Type Saponins
| Compound’s Name | Species Resource | Parts Used | Reference | ||||
|---|---|---|---|---|---|---|---|
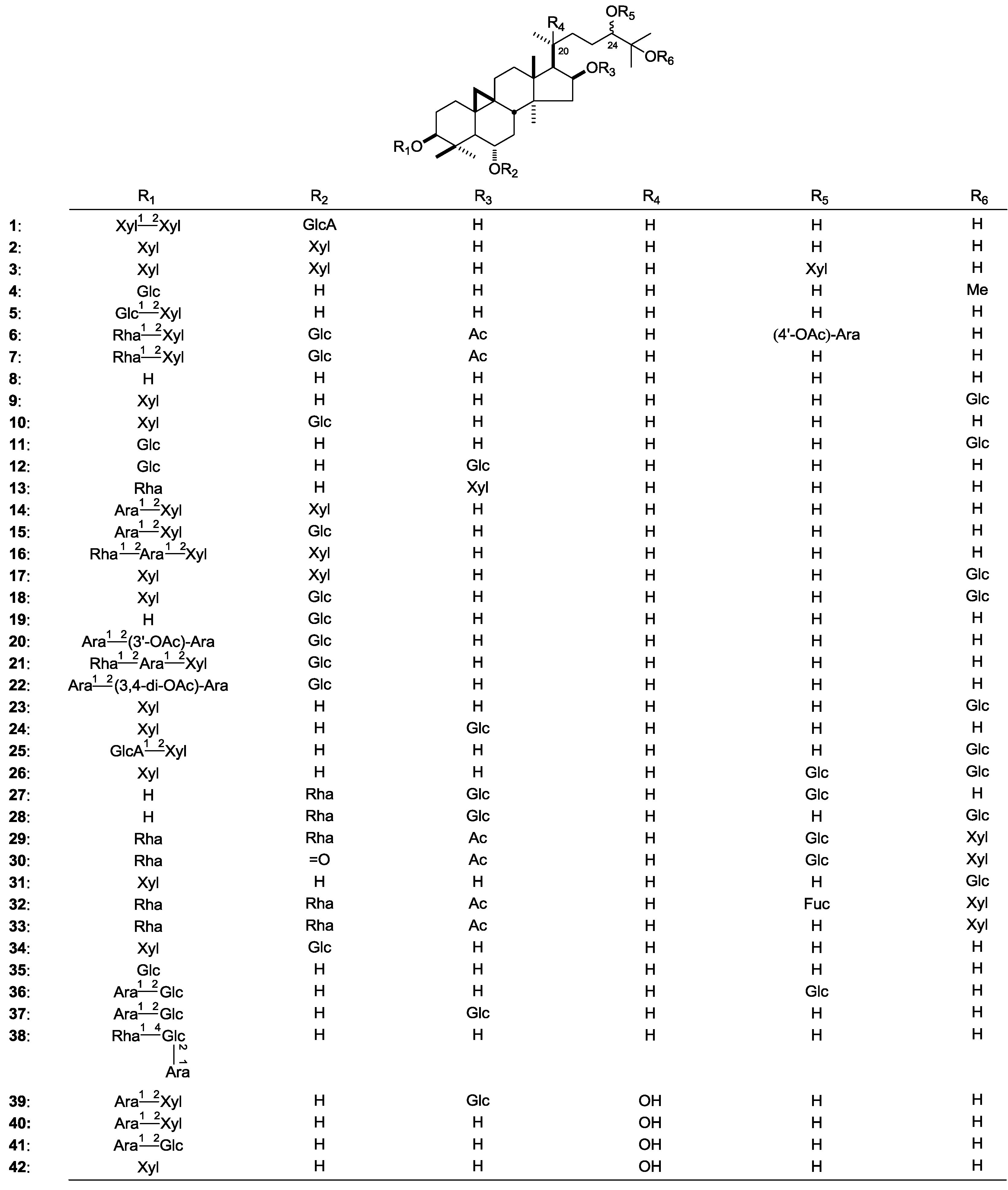
| 1 | 3-O-[β-d-Xylopyranosyl(1→2)-β-d-xylopyranosyl]-6-O-β-d-glucuronopyranosyl-3β,6α,16β,24(S),25-pentahydroxyxyxloartane | A. erinaceus | whole plant | [9] | |||
| 2 | Hareftoside A | A. erinaceus | whole plant | [9] | |||
| 3 | Hareftoside B | A. hareftae | whole plant | [10] | |||
| 4 | Cycloquivinoside A | A. chivensis | aerial parts | [11] | |||
| 5 | Astramembranosides B | A. membranaceus | roots | [12] | |||
| 6 | 3-O-[α-l-Rhamnopyranosyl(1→2)-β-d-xylopyranosyl]-6-O-β-d-glucopyranosyl-24-O-α-(4'-O-acetoxy)-l-arabinopyranosyl-16-O-acetoxy-3β,6α,16β,24S,25-pentahydroxycycloartane | A. wiedemannianus | whole plant | [13] | |||
| 7 | 3-O-[α-l-Rhamnopyranosyl(1→2)-β-d-xylopyranosyl]-6-O-β-d-glucopyranosyl-24-O-α-l-arabinopyranosyl-16-O-acetoxy-3β,6α,16β,24(S),25-pentahydroxycycloartane | A. wiedemannianus | whole plant | [13] | |||
| 8 | Cyclocanthogenin | A. unifoliolatus | epigeal parts | [14] | |||
| 9 | 3-O-β-d-Xylopyraosyl-24(S)-cycloart-3β,6α,16β,24,25-pentaol-25-O-β-d-glucopyranoside | A. ernestii | roots | [16] | |||
| 10 | Cyclocanthoside E | A. hareftae | whole plant | [10] | |||
| 11 | Cyclochivinoside B | A. chivensis | aerial parts | [15] | |||
| 12 | Cyclochivinoside C | A. chivensis | aerial parts | [20] | |||
| 13 | Caspicuside I | A. caspicus | roots | [6] | |||
| 14 | Oleifoliosides A | A. oleifolius | lower stem parts | [18] | |||
| 15 | Oleifoliosides B | A. oleifolius | lower stem parts | [18] | |||
| 16 | 3-O-[α-l-Rhamnopyranosyl(1→2)-α-l-arabinopyranosyl(1→2)-β-d-xylopyranosyl]-6-O-β-d-xylopyranosyl-3β,6α,16β,24(S),25-pentahydroxycycloartane | A. aureus | whole plant | [21] | |||
| 17 | 3,6-di-O-β-d-Xylopyranosyl-25-O-β-d-glucopyranosyl-3β,6α,16β,24(S),25-pentahydr-oxycycloartane | A. aureus | whole plant | [21] | |||
| 18 | 3-O-β-d-Xylopyranosyl-6,25-di-O-β-d-glucopyranosyl-3β,6α,16β,24(S),25-pentahydroxycycloartane | A. aureus | whole plant | [21] | |||
| 19 | 6-O-β-d-Glucopyranosyl-3β,6α,16β,24(S),25-pentahydroxycycloartane | A. aureus | whole plant | [21] | |||
| 20 | 3-O-[α-l-Arabinopyranosyl(1→2)-O-3-acetoxy-α-l-arabinopyranosyl]-6-O-β-d-glucopyranosyl-3β,6α,16β,24(S),25-pentahydroxycycloartane | A. icmadophilus | whole plant | [22] | |||
| 21 | 3-O-[α-l-Rhamnopyranosyl(1→2)-O-α-l-arabinopyranosyl(1→2)-O-β-d-xylopyranosyl]-6-O-β-d-glucopyranosyl-3β,6α,16β,24(S),25-pentahydroxycycloartane | A. icmadophilus | whole plant | [22] | |||
| 22 | 3-O-[α-l-Arabinopyranosyl(1→2)-O-3,4-diacetoxy-α-l-arabinopyranosyl]-6-O-β-d-glucopyranosyl-3β,6α,16β,24(S),25-pentahydroxycycloartane | A. icmadophilus | whole plant | [22] | |||
| 23 | 3-O-β-d-Xylopyranosyl-25-O-β-d-glucopyranosyl-3β,6α,16β,24(S),25-pentahydroxycycloartane | A. ernestii | roots | [16] | |||
| 24 | 3-O-β-d-Xylopyranosyl-16-O-β-d-glucopyranosyl-3β,6α,16β,24(S),25-pentahydroxycycloartane | A. amblolepis | roots | [17] | |||
| 25 | 3-O-[β-d-Glucuronopyranosyl(1→2)-β-d-xylopyranosyl]-25-O-β-d-glucopyranosyl-3β,6α,16β,24(S),25-pentahydroxy-cycloartane | A. amblolepis | roots | [17] | |||
| 26 | 3-O-β-d-Xylopyranosyl-24,25-di-O-β-d-glucopyranosyl-3β,6α,16β,24(S),25-pentahydr-oxy-cycloartane | A. amblolepis | roots | [17] | |||
| 27 | 6-O-α-l-Rhamnopyranosyl-16,24-di-O-β-d-glucopyranosyl-3β,6α,16β,24(S),25-pentahydroxy cycloartane | A. amblolepis | roots | [17] | |||
| 28 | 6-O-α-l-Rhamnopyranosyl-16,25-di-O-β-d-glucopyranosyl-3β,6α,16β,24(S),25-pentahydroxy cycloartane | A. amblolepis | roots | [17] | |||
| 29 | Cicerosides A | A. cicer | aerial parts | [7] | |||
| 30 | Cicerosides B | A. cicer | aerial parts | [7] | |||
| 31 | Cycloascidoside | A. ernestii | roots | [16] | |||
| 32 | Eremophiloside A | A. eremophilus | aerial parts | [24] | |||
| 33 | Eremophiloside B | A. eremophilus | aerial parts | [24] | |||
| 34 | Cycloascidoside A | A. mucidus | aerial parts | [25] | |||
| 35 | Cyclounifoliside C | A. unifoliolatus | epigeal parts | [14] | |||
| 36 | 3-O-[α-l-Arabinopyranosyl(1→2)-β-d-glucopyranosyl]-24-O-β-d-glucopyranosyl-3β,6α,16β,24(R),25-pentahydroxycycloartane | A. stereocalyx | roots | [26] | |||
| 37 | 3-O-[α-l-Arabinopyranosyl(1→2)-β-d-glucopyranosyl]-16-O-β-d-glucopyranosyl-3β,6α,16β,24(R),25-pentahydroxycycloartane | A. stereocalyx | roots | [26] | |||
| 38 | 3-O-{α-l-Rhamnopyranosyl(1→4)-[α-l-arabinopyranosyl(1→2)]-β-d-glucopyranosyl}-3β,6α,16β,24(R),25-pentahydroxycycloartane | A. stereocalyx | roots | [26] | |||
| 39 | 3-O-[α-l-Arabinopyranosyl(1→2)-β-d-xylopyranosyl]-16-O-β-d-glucopyranosyl-3β,6α,16β,20(S),24(R),25-hexahydroxycycloartane | A. stereocalyx | roots | [26] | |||
| 40 | 3-O-[α-l-Arabinopyranosyl(1→2)-β-d-xylopyranosyl]-3β,6α,16β,20(S),24(R),25-hexahydroxycycloartane | A. stereocalyx | roots | [26] | |||
| 41 | 3-O-[α-l-Arabinopyranosyl(1→2)-β-d-glucopyranosyl]-3β,6α,16β,20(S),24(R),25-hexahdroxycycloartane | A. stereocalyx | roots | [26] | |||
| 42 | 3-O-β-d-Xylopyranosyl-3β,6α,16β,20(S),24(R),25-hexahydroxycycloartane | A. schottianus | roots | [29] | |||
| 43 | Cyclomacrogenin B | A. macropus | roots | [30,31] | |||
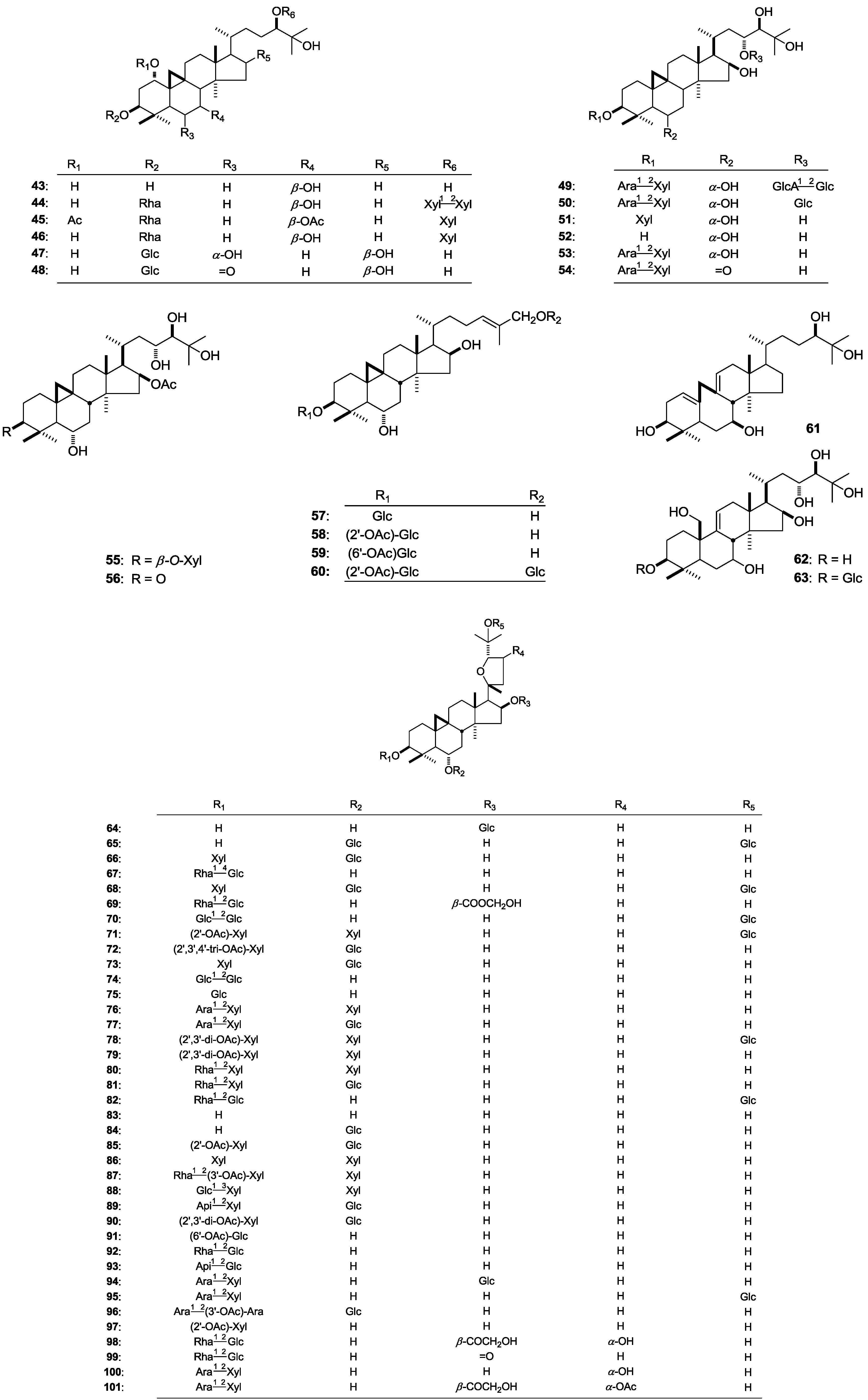
| 44 | Cyclomacroside E | A. macropus | roots | [32] | |||
| 45 | Cyclomacroside B | A. macropus | roots | [33] | |||
| 46 | Cyclomacroside D | A. macropus | roots | [31] | |||
| 47 | Mongholicoside A | A. membranace (Fisch.) Bge. var. mongholicus (Bge.) | aerial parts | [34] | |||
| 48 | Mongholicoside B | A. membranace (Fisch.) Bge. var. mongholicus (Bge.) | aerial parts | [34] | |||
| 49 | Askendoside K | A. taschkendicus | roots | [35] | |||
| 50 | Askendoside H | A. taschkendicus | roots | [36] | |||
| 51 | Cycloorbicoside D | A. orbiculatus | aerial parts | [37,38] | |||
| 52 | Cycloorbigenin C | A. taschkendicus | roots | [35,36] | |||
| A. orbiculatus | aerial parts | [37,38] | |||||
| 53 | Eremophiloside C | A. eremophilus | aerial parts | [24] | |||
| 54 | Eremophiloside D | A. eremophilus | aerial parts | [24] | |||
| 55 | Bicusposide F | A. bicuspis | whole plant | [39] | |||
| 56 | Bicusposide E | A. bicuspis | whole plant | [39] | |||
| 57 | Kahiricoside II | A. kahiricus | aerial parts | [40] | |||
| 58 | Kahiricoside III | A. kahiricus | aerial parts | [40] | |||
| 59 | Kahiricoside IV | A. kahiricus | aerial parts | [40] | |||
| 60 | Kahiricoside V | A. kahiricus | aerial parts | [40] | |||
| 61 | Secomacrogenin B | A. macropus | roots | [41] | |||
| 62 | Orbigenin | A. orbiculatus | aerial parts | [37,38] | |||
| 63 | Orbicoside | A. orbiculatus | aerial parts | [37,38] | |||
| 64 | 16-O-β-d-Glucopyranosyl-3β,6α,16β,25-tetrahydroxy-20(R),24(S)-epoxycycloartane | A. hareftae | whole plant | [10] | |||
| 65 | Astramembranosides A | A. membranaceus | roots | [12] | |||
| 66 | Cyclosiversioside F | A. oldenburgii | aerial parts | [42] | |||
| 67 | Astraverrucin IV | A. oldenburgii | aerial parts | [42] | |||
| 68 | Astragaloside VII | A. oldenburgii | aerial parts | [42] | |||
| 69 | 3-O-[α-l-Rhamnopyranosyl(1→2)-β-d-glucopyranosyl]-16-O-hydroxyacetoxy-3β,6α,16β,25-tetrahydroxy-20(R),24(S)-epoxycycloartane | A. angustifolius | whole plant | [45] | |||
| 70 | CyclolehmanosideC | A. lehmannianus | aerial parts | [46] | |||
| 71 | Armatoside II | A. armatus | roots | [47] | |||
| 72 | Acetylastragaloside I | A. baibutensis | roots | [48] | |||
| 73 | Astragaloside III | A. illyricus | roots | [49] | |||
| 74 | Cyclounifolioside B | A. illyricus | roots | [49] | |||
| 75 | Astraverrucin I | A. illyricus | roots | [49] | |||
| 76 | Trigonoside II | A. armatus | roots | [47] | |||
| 77 | Trojanoside H | A. stereocalyx | roots | [26] | |||
| 78 | Armatoside I | A. armatus | roots | [47] | |||
| 79 | Cyclosieversioside A | A. sieversianus | roots | [51] | |||
| 80 | Cyclosieversioside G | A. sieversianus | roots | [51] | |||
| 81 | Cyclosieversioside H | A. sieversianus | roots | [51] | |||
| 82 | 3-O-[α-l-Rhamnopyranosyl(1→2)-β-d-glucopyranosyl]-25-O-β-d-glucopyranosyl-20(R),24(S)-epoxy-3β,6α,16β,25-tetrahydroxycycloartane | A. wiedemannianus | whole plant | [13] | |||
| 83 | Cyclosiversigenin | A. orbiculatus | aerial parts | [52] | |||
| 84 | Brachyoside B | A. wiedemannianus | whole plant | [13] | |||
| 85 | Astragaloside II | A. hareftae A. wiedemannianus | whole plant whole plant | [10] [13] | |||
| 86 | Astrasieversianin X | A. wiedemannianus | whole plant | [13] | |||
| 87 | Astrasieversianin IX | A. sieversianus | roots | [51] | |||
| 88 | Caspicuside II | A. caspicus | roots | [6] | |||
| 89 | Baibutoside | A. baibutensis | roots | [48] | |||
| 90 | Astragalosides I | A. baibutensis | roots | [48] | |||
| 91 | Astraverrucin VII | A. verrucosus | aerial parts | [53] | |||
| 92 | Cycloaraloside D (Peregrinoside II) | A. verrucosus | aerial parts | [53] | |||
| 93 | Cycloaraloside C (Astrailienin A) | A. verrucosus | aerial parts | [53] | |||
| 94 | (20R,24S)-3-O-[α-l-Arabinopyranosyl(1→2)-β-d-xylopyranosyl]-20,24-epoxy-16-O-β-d-glucopyranosyl-3β,6α,16β,25-tetrahydroxycycloartane | A. halicacabus | whole plant | [27] | |||
| 95 | 3-O-[α-l-Arabinopyranosyl(1→2)-β-d-xylopyranosyl]-25-O-β-d-glucopyranosyl-3β,6α,16β,25-tetrahydroxy-20(R),24(S)-epoxycycloartane | A. campylosema Boiss. subsp. campylosema | roots | [28] | |||
| 96 | 3-O-[α-l-Arabinopyranosyl(1→2)-O-3-acetoxy-α-l-arabinopyranosyl]-6-O-β-d-glucopyranosyl-3β,6α,16β,25-tetrahydroxy-20(R),24(S)-epoxycycloartane | A. icmadophilus | whole plant | [22] | |||
| 97 | 20(R),24(S)-Epoxycycloartane-3β,6α,16β,25-tetraol-3-β-O-d-(2-O-acetyl)-xylopyranoside | A. bicuspis | whole plant | [39] | |||
| 98 | 3-O-[α-l-Rhamnopyranosyl(1→2)-β-d-glucopyranosyl]-16-O-hydroxyacetoxy-3β,6α,16β,23α,25-pentahydroxy-20(R),24(S)-epoxycycloartane | A. angustifolius | whole plant | [45] | |||
| 99 | 3-O-[α-l-Rhamnopyranosyl(1→2)-β-d-glucopyranosyl]-3β,6α,25-trihydroxy-20(R),24(S)-epoxycycloartane-16-one | A. angustifolius | whole plant | [45] | |||
| 100 | 3-O-[α-l-Arabinopyranosyl(1→2)-β-d-xylopyranosyl]-3β,6α,16β,23α,25-pentahydroxy-20(R),24(S)-epoxycycloartane | A. campylosema Boiss. subsp. campylosema | roots | [28] | |||
| 101 | 3-O-[α-l-Arabinopyranosyl(1→2)-β-d-xylopyranosyl]-16-O-hydroxyacetoxy-23-O-acetoxy-3β,6α,16β,23α,25-pentahydroxy-20(R),24(S)-epoxycycloartane | A. campylosema Boiss. subsp. campylosema | roots | [28] | |||
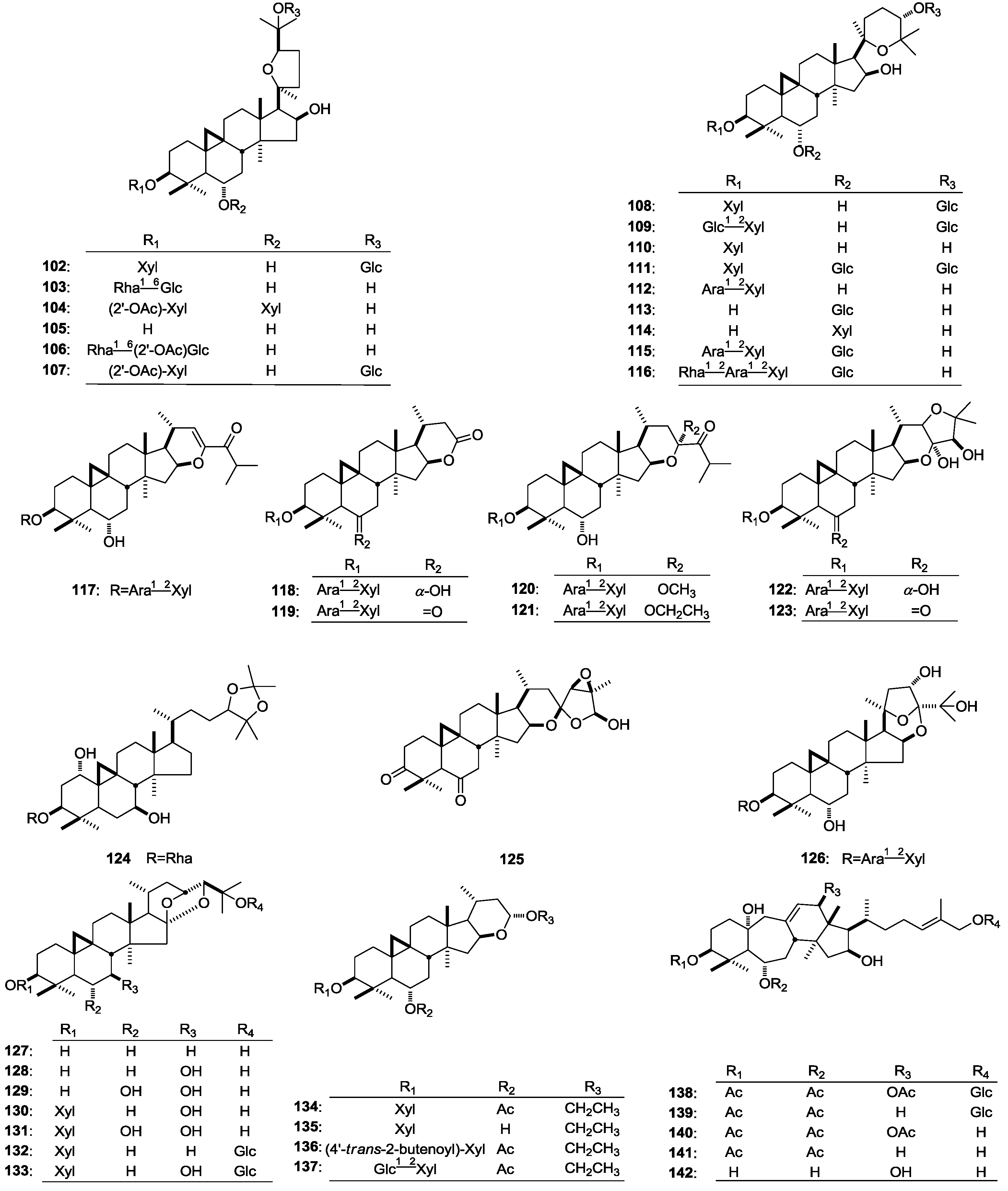
| 102 | Cyclogaleginoside E | A. galegiformis | stems | [54] | |||
| 103 | Cycloascualoside D | A. galegiformis | stems | [55] | |||
| 104 | Cyclogaleginoside C | A. galegiformis | stems | [55] | |||
| 105 | Cyclogalegigenin | A. galegiformis | stems | [54,55,57] | |||
| 106 | Cycloascauloside A | A. caucasicus | leaves | [56] | |||
| 107 | Cyclogaleginoside D | A. galegiformis | stems | [57] | |||
| 108 | 20(R),25-Epoxy-3-O-β-d-xylopyranosyl-24-O-β-d-glucopyranosyl-3β,6α,16β,24α-tetrahydroxycycloartane | A. schottianus | roots | [29] | |||
| 109 | 20(R),25-Epoxy-3-O-[-β-d-glucopyranosyl(1→2)]-β-d-xylopyranosyl-24-O-β-d-glucopyranosyl-3-β,6α,16β,24α-tetrahydroxycycloartane | A. schottianus | roots | [29] | |||
| 110 | Hareftoside C | A. hareftae | whole plant | [10] | |||
| 111 | Cylotrisectoside | A. dissectus | roots and stems | [43] | |||
| 112 | 3-O-[α-l-Arabinopyranosyl(1→2)-β-d-xylopyranosyl]-3β,6α,16β,24α-tetrahydroxy-20(R),25-epoxycycloartane | A. aureus | whole plant | [21] | |||
| 113 | 6-O-β-d-Glucopyranosyl-3β,6α,16β,24α-tetrahydroxy-20(R),25-epoxycycloartane | A. aureus | whole plant | [21] | |||
| 114 | 6-O-β-d-Xylopyranosyl-3β,6α,16β,24α-tetrahydroxy-20(R),25-epoxycycloartane | A. aureus | whole plant | [21] | |||
| 115 | 3-O-[α-l-Arabinopyranosyl(1→2)-O-β-d-xylopyranosyl]-6-O-β-d-glucopyranosyl-3β,6α,16β,24α-tetrahydroxy-20(R),25-epoxycycloartane | A. icmadophilus | whole plant | [22] | |||
| 116 | 3-O-[α-l-Rhamnopyranosyl(1→2)-O-α-l-arabinopyranosyl(1→2)-O-β-d-xylopyranosyl]-6-O-β-d-glucopyranosyl-3β,6α,16β,24α-tetrahydroxy-20(R),25-epoxycycloartane | A. icmadophilus | whole plant | [22] | |||
| 117 | Eremophiloside G | A. eremophilus | aerial parts | [24] | |||
| 118 | Eremophiloside E | A. eremophilus | aerial parts | [24] | |||
| 119 | Eremophiloside F | A. eremophilus | aerial parts | [24] | |||
| 120 | Eremophiloside H | A. eremophilus | aerial parts | [24] | |||
| 121 | Eremophiloside I | A. eremophilus | aerial parts | [24] | |||
| 122 | Eremophiloside J | A. eremophilus | aerial parts | [24] | |||
| 123 | Eremophiloside K | A. eremophilus | aerial parts | [24] | |||
| 124 | Cyclomacroside A | A. macropus | roots | [58] | |||
| 125 | Bicusposide D | A. bicuspis | whole plant | [39] | |||
| 126 | 3-O-[α-l-Arabinopyranosyl(1→2)-β-d-xylopyranosyl]-3β,6α,23α,25-tetrahydroxy-20(R),24(R)-16β,24;20,24-diepoxycycloartane | A. campylosema Boiss. subsp. campylosema | roots | [28] | |||
| 127 | Dihydrocycloorbigenin A | A. orbiculatus | aerial parts | [38] | |||
| 128 | Cycloorbigenin | A. orbiculatus | aerial parts | [38] | |||
| 129 | Cycloorbigenin B | A. orbiculatus | aerial parts | [38] | |||
| 130 | Cycloorbicoside A | A. orbiculatus | aerial parts | [38] | |||
| 131 | Cycloorbicoside B | A. orbiculatus | aerial parts | [38] | |||
| 132 | Cycloorbicoside C | A. orbiculatus | aerial parts | [38] | |||
| 133 | Cycloorbicoside G | A. orbiculatus | aerial parts | [38] | |||
| 134 | Tomentoside I | A. tomentosus | aerial parts | [59] | |||
| 135 | Deacetyltomentoside I | A. tomentosus | aerial parts | [59] | |||
| 136 | Tomentoside III | A. tomentosus | aerial parts | [59] | |||
| 137 | Tomentoside IV | A. tomentosus | aerial parts | [59] | |||
| 138 | Huangqiyenin E | A. membranaceus | leaves | [60] | |||
| 139 | Huangqiyenin F | A. membranaceus | leaves | [60] | |||
| 140 | Huangqiyegenin III | A. membranaceus | leaves | [60] | |||
| 141 | Huangqiyegenin IV | A. membranaceus | leaves | [60] | |||
| 142 | Trideacetylhuangqiyegenin III | A. membranaceus | leaves | [60] | |||
2.1.2. Oleanane-Type Saponins
| Compound’s Name | Species Resource | Parts Used | Reference | |
|---|---|---|---|---|
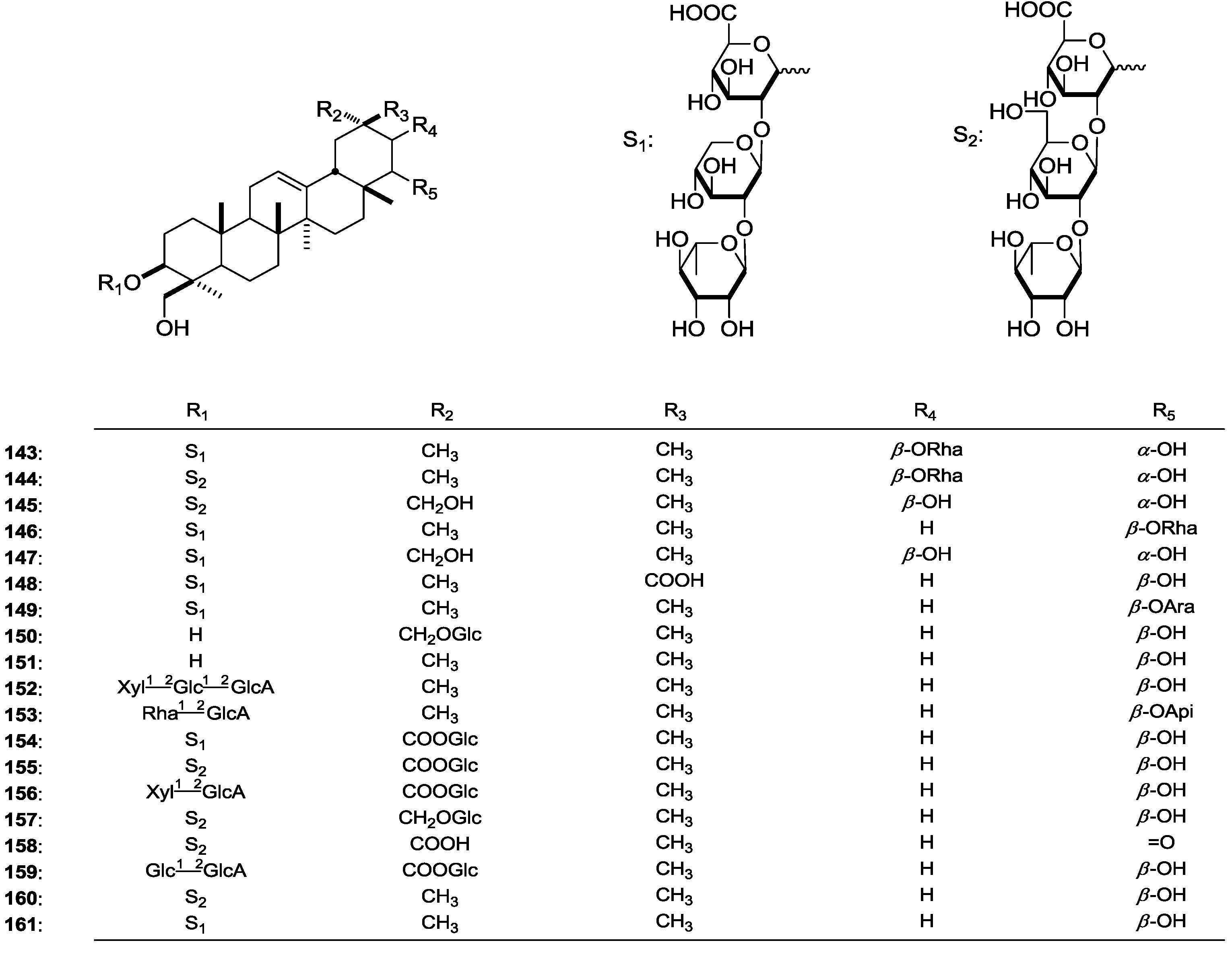
| 143 | 3-O-[α-l-Rhamnopyranosyl(1→2)-β-d-xylopyranosyl(1→2)-β-d-glucuronopyranosyl]-21-O-α-l-rhamnopyranosyl-3β,21β,22α,24-tetrahydroxyolean-12-ene | A. tauricolus | whole plant | [61] |
| 144 | 3-O-[α-l-Rhamnopyranosyl(1→2)-β-d-glucopyranosyl(1→2)-β-d-glucuronopyranosyl]-21-O-α-l-rhamnopyranosyl-3β,21β,22α,24-tetrahydroxyolean-12-ene | A. tauricolus | whole plant | [61] |
| 145 | 3-O-[α-l-Rhamnopyranosyl(1→2)-β-d-glucopyranosyl(1→2)-β-d-glucuronopyranosyl]-3β,21β,22α,24,29-pentahydroxyolean-12-ene | A. tauricolus | whole plant | [61] |
| 146 | 3-O-[α-l-Rhamnopyranosyl(1→2)-β-d-xylopyranosyl(1→2)-β-d-glucuronopyranosyl]-22-O-α-l-rhamnopyranosyl-3β,22β,24-trihydroxyolean-12-ene | A. tauricolus | whole plant | [61] |
| 147 | 3-O-[α-l-Rhamnopyranosyl(1→2)-β-d-xylopyranosyl(1→2)-β-d-glucuronopyranosyl]-3β,21β,22α,24,29-pentahydroxyolean-12-ene | A. angustifolius | whole plant | [45] |
| 148 | 3-O-[α-l-Rhamnopyranosyl(1→2)-β-d-xylopyranosyl(1→2)-β-d-glucuronopyranosyl]-3β,22β,24-trihydroxyolean-12-en-29-oic acid | A. angustifolius | whole plant | [45] |
| 149 | 3-O-[α-l-Rhamnopyranosyl(1→2)-β-d-xylopyranosyl(1→2)-β-d-glucuronopyranosyl]-22-O-α-l-arabinopyranosyl-3β,22β,24-trihydroxyolean-12-ene | A. angustifolius | whole plant | [45] |
| 150 | 29- O-β-d-Glucopyranosyl-3β,22β,24,29-tetrahydroxy-olean-12-ene | A. angustifolius | whole plant | [45] |
| 151 | Soyasapogenol B | A. caprinus | roots | [62] |
| 152 | 3-O-[β-d-Xylopyranosyl(1→2)-O-β-d-glucopyranosyl(1→2)-O-β-d-glucuronopyranosyl] soyasapogenol B | A. hareftae | whole plant | [10] |
| 153 | 3-O-α-l-Rhamnopyranosyl(1→2)-β-d-glucuronopyranosyl]-22-O-β-d-apiofuranosyl soyasapogenol B | A. caprinus | roots | [62] |
| 154 | 3-O-[α-l-Rhamnopyranosyl(1→2)-β-d-xylopyranosyl(1→2)-β-d-glucuronopyranosyl]-29-O-β-d-glucopyranosyl-3β,22β,24-trihydroxyolean-12-en-29-oic acid | A. tauricolus | whole plant | [61] |
| 155 | 3-O-[α-l-Rhamnopyranosyl(1→2)-β-d-glucopyranosyl(1→2)-β-d-glucuronopyranosyl]-29-O-β-d-glucopyranosyl-3β,22β,24,-trihydroxyolean-12-en-29-oic acid | A. tauricolus | whole plant | [61] |
| 156 | 3-O-[β-d-Xylopyranosyl(1→2)-β-d-glucuronopyranosyl]-29-O-β-d-glucopyranosyl-3β,22β,24,-trihydroxyolean-12-en-29-oic acid | A. tauricolus | whole plant | [61] |
| 157 | 3-O-[α-l-Rhamnopyranosyl-(1→2)-β-d-glucopyranosyl-(1→2)-β-d-glucuronopyranosyl]-29-O-β-d-glucopyranosyl-3β,22β,24,29-tetrahydroxyolean-12-ene | A. tauricolus | whole plant | [61] |
| 158 | 3-O-[α-l-Rhamnopyranosyl-(1→2)-β-d-glucopyranosyl-(1→2)-β-d-glucuronopyranosyl]-3β,24-dihydroxyolean-12-ene-22-oxo-29-oic acid | A. tauricolus | whole plant | [61] |
| 159 | 3-O-[β-d-Glucopyranosyl-(1→2)-β-d-glucuronopyranosyl]-29-O-β-d-glucopyranosyl-3β,22β,24,-trihydroxyolean-12-en-29-oic acid | A. tauricolus | whole plant | [61] |
| 160 | Azukisaponin V | A. cruciatus | aerial parts and roots | [2] |
| 161 | Astragaloside VIII | A. flavescens | roots | [4] |
2.2. Flavonoids
| Compound’s Name | Species Resource | Parts Used | Reference | |
|---|---|---|---|---|
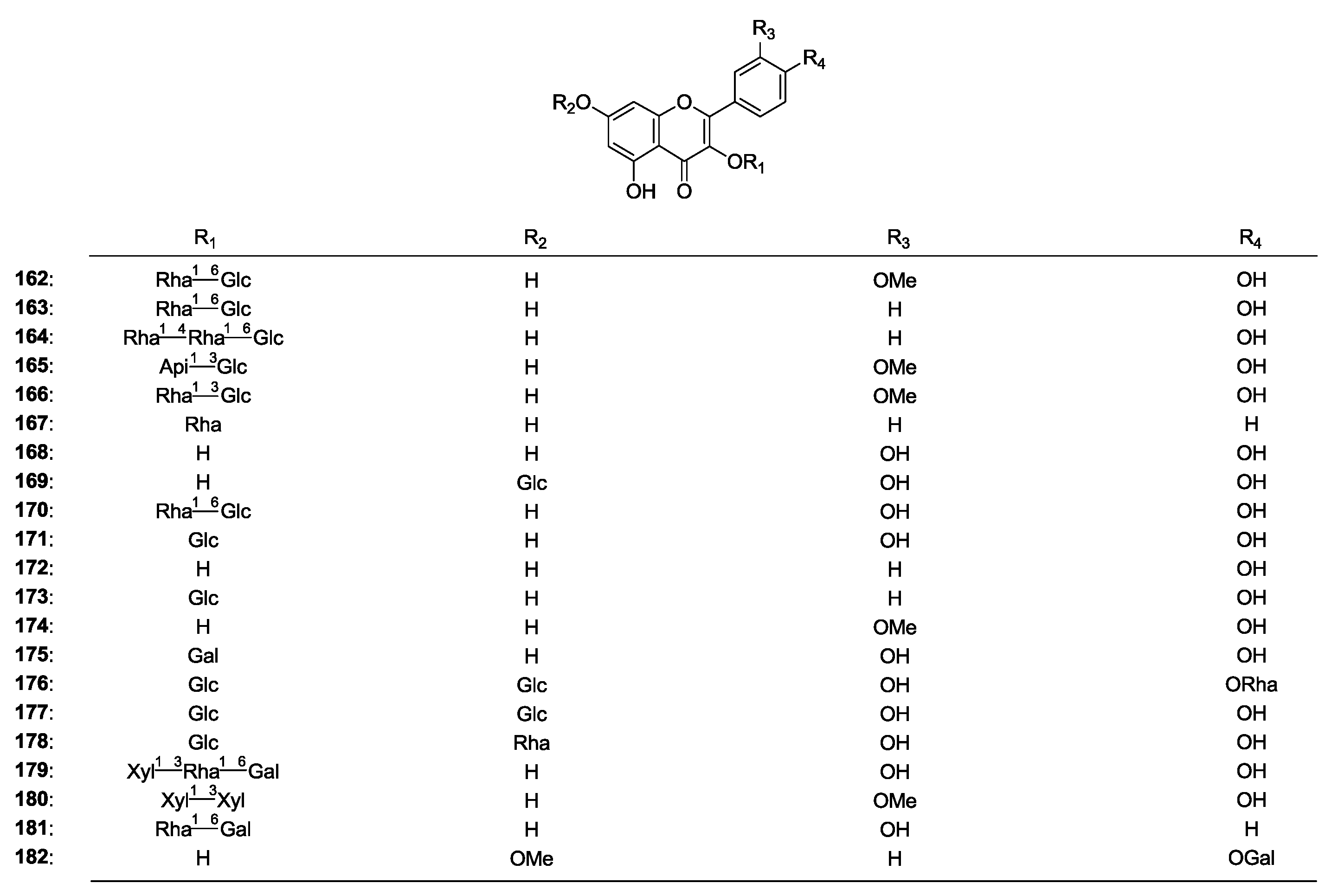
| 162 | Narcissin | A. cruciatus | aerial parts and roots | [2] |
| 163 | Nicotiflorin | A. cruciatus | aerial parts and roots | [2] |
| 164 | Kaempferol 3-O-α-l-rhamnopyranosyl(1→4)-α-l-rhamnopyranosyl(1→6)-β-d-glucopyranoside | A. cruciatus | aerial parts and roots | [2] |
| 165 | Microcephalin I | A. microcephalus | leaves | [65] |
| 166 | Microcephalin II | A. microcephalus | leaves | [65] |
| 167 | Kaempferol-3-O-α-l-rhamnoxyloside | A. microcephalus | leaves | [66] |
| 168 | Quercetin | A. asper | aerial parts | [64] |
| 169 | Quercimeritrin | A. asper | aerial parts | [64] |
| 170 | Rutin | A. cruciatus | aerial parts and roots | [2] |
| 171 | Quercetin-3-O-β-d-glucopyranoside | A. corniculatus | aerial parts | [63] |
| 172 | Kaempferol | A. corniculatus | aerial parts | [63] |
| 173 | Kaempferol-3-glucoside (Astragalin) | A. asper | aerial parts | [64] |
| 174 | Isorhamnetin | A. corniculatus | aerial parts | [63] |
| 175 | Quercetin-3-O-galactoside | A. corniculatus | aerial parts | [63] |
| 176 | Quercetin-3,7-di-β-d-glucopyranoside-4'-O-α-l-rhamnopyranoside | A. bombycinus | whole plant | [8] |
| 177 | Quercetin-3,7-di-O-β-d-glucopyranoside | A. bombycinus | whole plant | [8] |
| 178 | Quercetin 3-O-β-d-glucopyranoside-7-O-α-l-rhamnopyranoside | A. bombycinus | whole plant | [8] |
| 179 | Flagaloside C | A. galegiformis | leaves | [67] |
| 180 | Flagaloside D | A. galegiformis | leaves | [67] |
| 181 | Kaempferol 3-O-robinobioside | A. verrucosus | aerial parts | [53] |
| 182 | 7-O-Methyl-kaempferol-4'-β-d-galactopyranoside | A. hamosus | aerial parts | [68] |
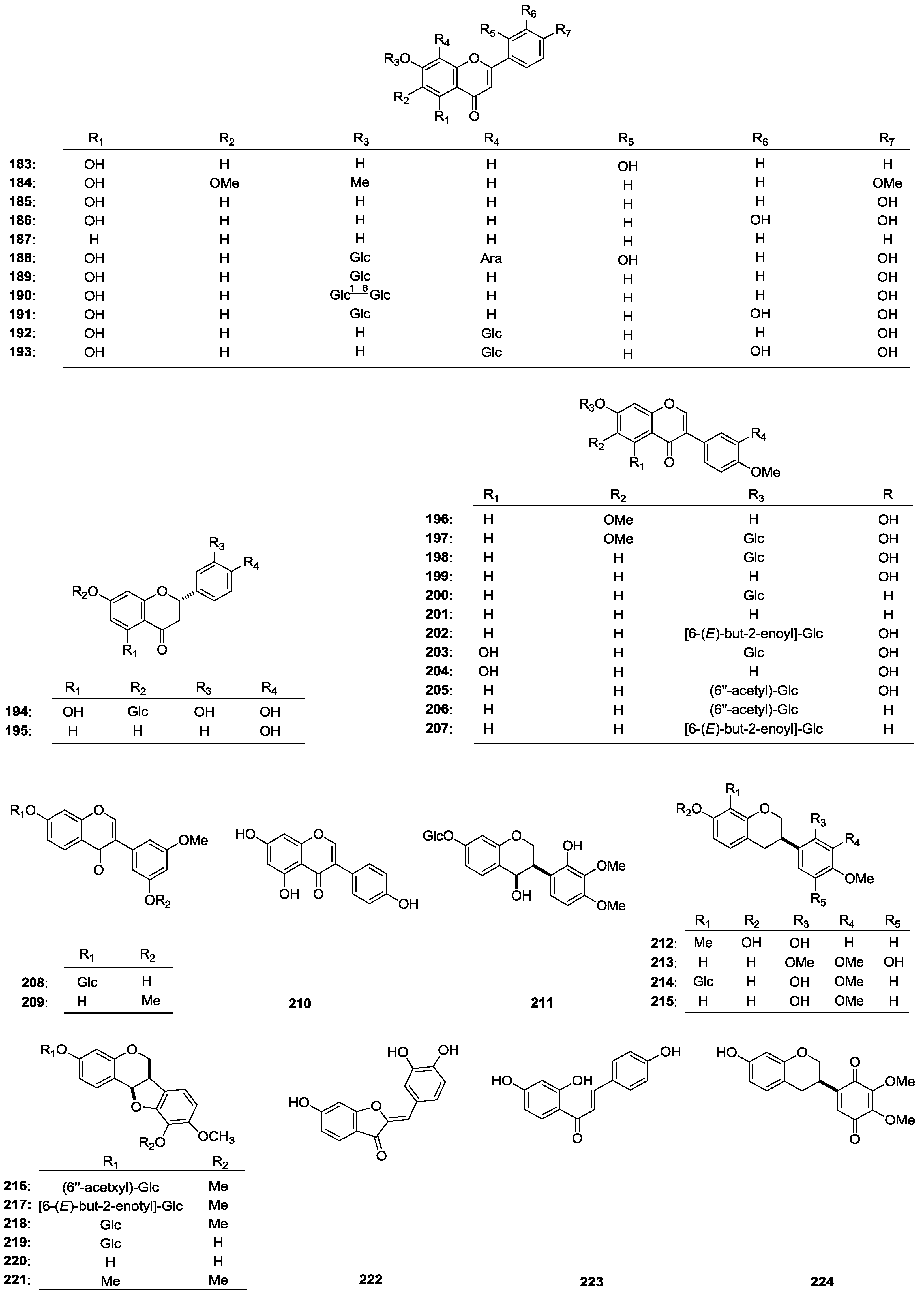
| 183 | 5,7,2'-Trihydroxyflavone | A. cruciatus | aerial parts and roots | [2] |
| 184 | Salvigenin | A. propinquus | roots | [69] |
| 185 | Apigenin | A. bombycinus | whole plant | [8] |
| 186 | Luteolin | A. bombycinus | whole plant | [8] |
| 187 | 7-Hydroxyflavone | A. microcephalus | leaves | [66] |
| 188 | 5,2',4'-Trihydroxy-flavone-8-C-l-arabinopyranoside-7-O-β-d-glucopyranoside | A. bombycinus | whole plant | [8] |
| 189 | Apigenin 7-O-β-d-glucopyranoside | A. bombycinus | whole plant | [8] |
| 190 | Apigenin 7-O-gentobioside | A. bombycinus | whole plant | [8] |
| 191 | Luteolin 7-O-β-d-glucopyranoside | A. bombycinus | whole plant | [8] |
| 192 | Apigenin-8-C-glucoside (Vitexin) | A. corniculatus | aerial parts | [63] |
| 193 | Luteolin-8-C-glucoside (Orientin) | A. corniculatus | aerial parts | [63] |
| 194 | Eriodyctiol-7-O-glucoside | A. corniculatus | aerial parts | [63] |
| 195 | Liquiritigenin | A. membranaceus | roots | [70] |
| 196 | Odoration | A. membranaceus var. mongholicus | roots | [71] |
| 197 | Odoration-7-O-β-d-glucopyranoside | A. mongholicus | aerial parts | [72] |
| 198 | Calycosin-7-O-β-d-glucopyranoside | A. ernestii | roots | [16] |
| 199 | Calycosin | A. membranaceus | roots | [70] |
| 200 | Ononin | A. membracaceus | roots | [50] |
| 201 | Formononetin | A. membranaceus | roots | [70] |
| 202 | Calycosin 7-O-β-d-{6''-[(E)-but-2-enoyl]}-glucoside | A. membracaceus | roots | [50] |
| 203 | Pratensein 7-O-β-d-glucopyranoside | A. membranaceus var. mongholicus | roots | [71] |
| 204 | Pratensein | A. verrucosus | aerial parts | [53] |
| 205 | Calycosin 7-O-β-d-(6''-acetyl)-glucoside | A. membracaceus | roots | [50] |
| 206 | 6ꞌꞌ-Acetylononin | A. membracaceus | roots | [50] |
| 207 | Ammopiptanoside A | A. membracaceus | roots | [50] |
| 208 | 7,5'-Dihydroxy-3'-methoxy-isoflavone-7-O-β-d-glucopyranoside | A. membranaceus var. mongholicus | roots | [71] |
| 209 | 7-Hydroxy-3',5'-dimethoxyisoflavone | A. peregrinus | aerial parts | [75] |
| 210 | Daidzein | A. bombycinus | whole plant | [8] |
| 211 | (3R,4R)-3-(2-Hydroxy-3,4-dimethoxy-phenyl)-chroman-4,7-diol-7-O-β-d-glucopyranoside | A. membranaceus | roots | [74] |
| 212 | (3R)-8,2'-Dihydroxy-7,4'-dimethoxyisoflavane | A. membranaceus | roots | [76] |
| 213 | (R)-3-(5-Hydroxy-2,3,4-trimethoxyphenyl)-chroman-7-ol | A. membracaceus | roots | [50] |
| 214 | Isomucronulatol 7-O-β-glucoside | A. membracaceus | roots | [50] |
| 215 | Isomucronulatol | A. membracaceus | roots | [50] |
| 216 | (–)-Methylinissolin 3-O-β-d-(6'-acetyl)-glucoside | A. membracaceus | roots | [50] |
| 217 | (–)-Methylinissolin 3-O-β-d-{6'-[(E)-but-2-enoyl]}-glucoside | A. membracaceus | roots | [50] |
| 218 | (–)-Methylinissolin 3-O-β-d-glucoside | A. membracaceus | roots | [50] |
| 219 | Licoagroside D | A. membracaceus | roots | [50] |
| 220 | Vesticarpan | A. membracaceus | roots | [50] |
| 221 | (–)-Methylinissolin | A. membracaceus | roots | [50] |
| 222 | Sulfuretin | A. microcephalus | leaves | [66] |
| 223 | Isoliquiritigenin | A. membranaceus | roots | [70] |
| 224 | Pendulone | A. membracaceus | roots | [50] |
2.3. Polysaccharides
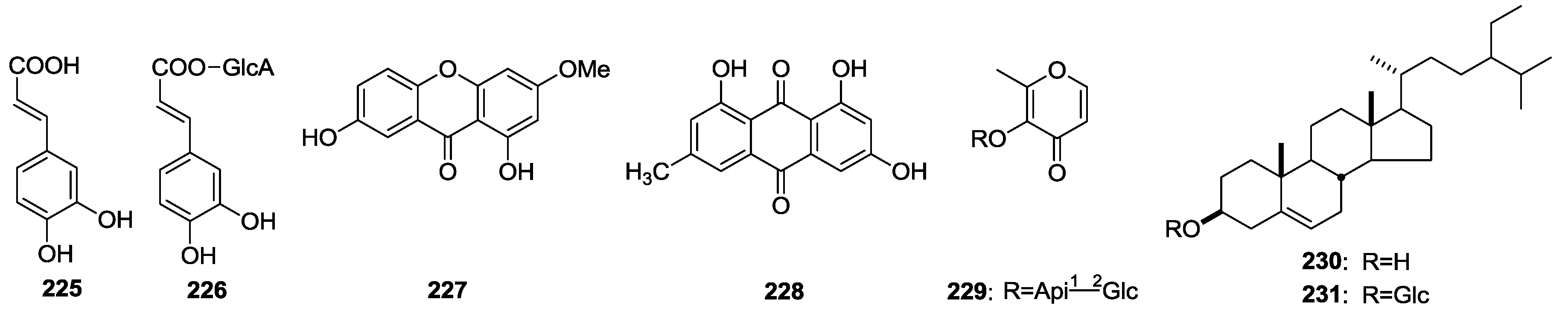
2.4. Others
3. Biological Activities of the Astragalus Genus
3.1. Anti-Inflammatory Activity
3.2. Immunoregulatory Activity
3.3. Anti-Tumor Activity
3.4. Cardioprotection Activity
3.5. Antidiabetic
3.6. Anti-Oxidative Activity
3.7. Anti-Aging
3.8. Other Biological Activities
4. Analyses
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Reference
- Podlech, D. The genus Astragalus L. (Fabaceae) in Europe with exclusion of the former Soviet Union. Feddes Repertor. 2008, 119, 310–387. [Google Scholar] [CrossRef]
- Benchadi, W.; Haba, H.; Lavaud, C.; Harakat, D.; Benkhaled, M. Secondary metabolites of Astragalus cruciatus Link. and their chemotaxonomic significance. Rec. Nat. Prod. 2013, 7, 105–113. [Google Scholar]
- Xu, L.; Podlech, D.F. Astragalus Linnaeus, Sp. Pl. 2: 755. 1753. Flora China 2010, 10, 329–333. Available online: http://www.efloras.org/florataxon.aspx?flora_id=2&taxon_id=102978 (accessed on 15 March 2014). [Google Scholar]
- Avunduk, S.; Mitaine-Offer, A.-C.; Alankus-Caliskan, O.; Miyamoto, T.; Senol, S.G.; Lacaille-Dubois, M.A. Triterpene glycosides from the roots of Astragalus flavescens. J. Nat. Prod. 2008, 71, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, M.I.; Jan, S.; Abbaskhan, A.; Musharraf, S.G.; Samreen; Sattar, S.A.; Atta-ur-Rahman. Cycloartane triterpenoids from Astragalus bicuspic. J. Nat. Prod. 2008, 71, 1557–1560. [Google Scholar] [CrossRef] [PubMed]
- Fathiazad, F.; Khosropanah, M.K.; Movafeghi, A. Cycloartane-type glycosides from the roots of Astragalus. caspicus Bieb. Nat. Prod. Res. 2010, 24, 1069–1078. [Google Scholar] [CrossRef] [PubMed]
- Linnek, J.; Mitaine-Offer, A.-C.; Miyamoto, T.; Tanaka, C.; Paululat, T.; Avunduk, S.; Alankus-Caliskan, O.; Lacaille-Dubois, M.-A. Cycloartane glycosides from three species of Astragalus (Fabaceae). Helv. Chim. Acta 2011, 94, 230–237. [Google Scholar] [CrossRef]
- Ibrahim, L.F.; Marzouk, M.M.; Hussein, S.R.; Kawashty, S.A.; Mahmoud, K.; Saleh, N.A.M. Flavonoid constituents and biological screening of Astragalus bombycinus Boiss. Nat. Prod. Res. 2013, 27, 386–393. [Google Scholar] [CrossRef] [PubMed]
- Savran, T.; Gulcemal, D.; Masullo, M.; Karayildirim, T.; Polat, E.; Piacente, S.; Alankus-Caliskan, O. Cycloartane glycosides from Astragalus erinaceus. Rec. Nat. Prod. 2012, 6, 230–236. [Google Scholar]
- Horo, I.; Bedir, E.; Masullo, M.; Piacente, S.; Ozgokce, F.; Alankus-Caliskan, O. Saponins from Astragalus hareftae (NAB.) SIRJ. Phytochemistry 2012, 84, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Naubeev, T.K.; Uteniyazov, K.K.; Isaev, M.I.; Kachala, V.V.; Shashkov, A.S. Structure of cycloquivinoside A from the aerial part of Astragalus chiwensis. Chem. Nat. Compd. 2012, 48, 810–812. [Google Scholar] [CrossRef]
- Kim, J.S.; Yean, M.-H.; Lee, E.-J.; Jung, H.S.; Lee, J.Y.; Kim, Y.J.; Kang, S.S. Two new cycloartane saponins from the roots of Astragalus membranaceus. Chem. Pharm. Bull. 2008, 56, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Polat, E.; Bedir, E.; Perrone, A.; Piacente, S.; Alankus-Caliskan, O. Triterpenoid saponins from Astragalus wiedemannianus Fischer. Phytochemistry 2010, 71, 658–662. [Google Scholar] [CrossRef] [PubMed]
- Kucherbaev, K.D.; Uteniyazov, K.K.; Kachala, V.V.; Saatov, Z.; Shashkov, A.S. Triterpene Glycosides from plants of the Astragalus Genus. III. Structure of cyclounifolioside C from Astragalus unifoliolatus. Chem. Nat. Compd. 2003, 38, 447–449. [Google Scholar] [CrossRef]
- Naubeev, T.K.; Uteniyazov, K.K.; Kachala, V.V.; Shashkov, A.S. Cyclochivinoside B from the aerial part of Astragalus chivensis. Chem. Nat. Compd. 2007, 43, 166–169. [Google Scholar] [CrossRef]
- Sun, L.-M.; Wang, X.-L.; Deng, W.-L.; Ding, L.-S.; Peng, S.-L. Chemical constituents from Astragalus ernestii. Zhongguo Tianran Yaowu 2011, 9, 38–41. [Google Scholar]
- Polat, E.; Caliskan-Alankus, O.; Perrone, A.; Piacente, S.; Bedir, E. Cycloartane-type glycosides from Astragalus amblolepis. Phytochemistry 2009, 70, 628–634. [Google Scholar] [CrossRef] [PubMed]
- Oezipek, M.; Doenmez, A.A.; Calis, I.; Brun, R.; Rueedi, P.; Tasdemir, D. Leishmanicidal cycloartane-type triterpene glycosides from Astragalus oleifolius. Phytochemistry 2005, 66, 1168–1173. [Google Scholar] [CrossRef] [PubMed]
- Alaniya, M.D.; Kavtaradze, N.S.; Gigoshvili, T.I.; Lavoi, S.; Pichette, A.; Mshvildadze, V.V. Cyclocanthoside E from Astragalus caucasicus. Chem. Nat. Compd. 2007, 43, 758–759. [Google Scholar] [CrossRef]
- Naubeev, T.K.; Uteniyazov, K.K. Structure of cyclochivinoside C from Astragalus chivensis. Chem. Nat. Compd. 2007, 43, 560–562. [Google Scholar] [CrossRef]
- Gulcemal, D.; Alankus-Caliskan, O.; Perrone, A.; Ozgokce, F.; Piacente, S.; Bedir, E. Cycloartane glycosides from Astragalus aureus. Phytochemistry 2011, 72, 761–768. [Google Scholar] [CrossRef] [PubMed]
- Horo, I.; Bedir, E.; Perrone, A.; Oezgoekce, F.; Piacente, S.; Alankus-Caliskan, O. Triterpene glycosides from Astragalus icmadophilus. Phytochemistry 2010, 71, 956–963. [Google Scholar] [CrossRef] [PubMed]
- Naubeev, T.K.; Zhanibekov, A.A.; Isaev, M.I. Triterpene glycosides from Astragalus and their genins. XCIII. Cycloascidoside from Astragalus mucidus. Chem. Nat. Comp. 2012, 48, 813–815. [Google Scholar] [CrossRef]
- Perrone, A.; Masullo, M.; Bassarello, C.; Bloise, E.; Hamed, A.; Nigro, P.; Pizza, C.; Piacente, S. Unusual cycloartane glycosides from Astragalus eremophilus. Tetrahedron 2008, 64, 5061–5071. [Google Scholar] [CrossRef]
- Naubeev, T.K.; Uteniyazov, K.K.; Isaev, M.I. Triterpene glycosides from Astragalus and their genins. LXXXVIII. Cycloascidoside A, a new bisdesmoside of cycloasgenin C. Chem. Nat. Compd. 2011, 47, 250–253. [Google Scholar] [CrossRef]
- Yalcin, F.N.; Piacente, S.; Perrone, A.; Capasso, A.; Duman, H.; Calis, I. Cycloartane glycosides from Astragalus stereocalyx Bornm. Phytochemistry 2012, 73, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Djimtombaye, B.J.; Alankus-Caliskan, O.; Gulcemal, D.; Khan, I.A; Anil, H.; Bedir, E. Unusual secondary metabolites from Astragalus halicacabus Lam. Chem. Biodivers. 2013, 10, 1328–1334. [Google Scholar] [CrossRef] [PubMed]
- Calis, I.; Doenmez, A.A.; Perrone, A.; Pizza, C.; Piacente, S. Cycloartane glycosides from Astragalus campylosema Boiss. ssp. campylosema. Phytochemistry 2008, 69, 2634–2638. [Google Scholar] [CrossRef]
- Karabey, F.; Khan, I.A.; Bedir, E. Cycloartane-type glycosides from Astragalus schottianus. Phytochem. Lett. 2012, 5, 320–324. [Google Scholar] [CrossRef]
- Iskenderov, D.A.; Isaev, I.M.; Isaev, M.I. Triterpene glycosides from Astragalus and their genins. LXXVII. Cyclomacrogenin B, a new cycloartane triterpenoid. Chem. Nat. Compd. 2008, 44, 621–624. [Google Scholar] [CrossRef]
- Iskenderov, D.A.; Isaev, I.M.; Isaev, M.I. Triterpene glycosides from Astragalus and their genins. LXXX. Cyclomacroside D, a new bisdesmoside. Chem. Nat. Compd. 2009, 45, 55–58. [Google Scholar] [CrossRef]
- Iskenderov, D.A.; Isaev, I.M.; Isaev, M.I. Triterpene glycosides from Astragalus and their genins. LXXXV. Structure of cyclomacroside E. Chem. Nat. Compd. 2010, 46, 250–253. [Google Scholar] [CrossRef]
- Iskenderov, D.A.; Isaev, I.M.; Isaev, M.I. Triterpene glycosides from Astragalus and their genins. LXXXII. Cyclomacroside B, a new glycoside. Chem. Nat. Compd. 2009, 45, 511–513. [Google Scholar] [CrossRef]
- Yu, Q.T.; Li, P.; Bi, Z.M.; Luo, J.; Gao, X.D. Two new saponins from the aerial part of Astragalus membranaceus var. mongholicus. Chin. Chem. Lett. 2007, 18, 554–556. [Google Scholar] [CrossRef]
- Isaev, I.M.; Iterpene gsaev, M.I. Trilycosides from Astragalus and their genins. XC. Askendoside K from Astragalus taschkendicus. Chem. Nat. Compd. 2011, 47, 587–591. [Google Scholar] [CrossRef]
- Isaev, I.M.; Isaev, M.I. Triterpene glycosides from Astragalus and their genins. LXXXIX. Askendoside H from Astragalus taschkendicus. Chem. Nat. Compd. 2011, 47, 411–414. [Google Scholar] [CrossRef]
- Isaev, I.M.; Mamedova, R.P.; Agzamova, M.A.; Isaev, M.I. Triterpene glycosides and their genins from Astragalus. LXXIII. Stereochemistry of C-23 and C-24 in cycloartan and lanostan-16β,23,24,25-tetraols. Chem. Nat. Compd. 2007, 43, 115–116. [Google Scholar] [CrossRef]
- Mamedova, R.P.; Agzamova, M.A.; Isaev, M.I. Triterpene glycosides of Astragalus and their genins. LXV. Cycloartane and lanostane triterpenoids of Astragalus orbiculatus. Chem. Nat. Compd. 2003, 38, 354–355. [Google Scholar] [CrossRef]
- Jan, S.; Abbaskhan, A.; Musharraf, S.G.; Sattar, S.A.; Samreen; Resayes, S.I.; Al-Othman, Z.A.; Al-Majid, A.M.; Atta-ur-Rahman, C.M.I. Three new cycloartane triterpenoids from Astragalus bicuspis. Planta Med. 2011, 77, 1829–1834. [Google Scholar] [CrossRef] [PubMed]
- Radwan, M.M.; El-Sebakhy, N.A.; Asaad, A.M.; Toaima, S.M.; Kingston, D.G.I. Kahiricosides II-V, cycloartane glycosides from an Egyptian collection of Astragalus kahiricus. Phytochemistry 2004, 65, 2909–2913. [Google Scholar] [CrossRef] [PubMed]
- Isaev, I.M.; Iskenderov, D.A.; Isaev, M.I. Triterpene glycosides and their genins from Astragalus. LXXXIV. Secomacrogenin B, a new 9,10-seco-cycloartane. Chem. Nat. Compd. 2010, 46, 36–38. [Google Scholar] [CrossRef]
- Naubeev, T.K.; Isaev, M.I. Triterpene glycosides of Astragalus and their genins. XCII. Cycloartane glycosides from Astragalus oldenburgii. Chem. Nat. Compd. 2012, 48, 704–705. [Google Scholar] [CrossRef]
- Sukhina, I.A.; Mamedova, R.P.; Agzamova, M.A.; Isaev, M.I. Triterpene glucosides of Astragalus and their genins. LXXIV. Cyclotrisectoside, the first trisdesmoside of cyclocephalogenin. Chem. Nat. Compd. 2007, 43, 159–161. [Google Scholar] [CrossRef]
- Zheng, S.; Wang, Z. Chemical constituents of the roots of Astragalus membranace (Fisch.) Bge. var. mongholicus (Bge.) hisao. Shanghai Zhongyiyao Daxue Xuebao 2011, 25, 89–94. [Google Scholar]
- Gulcemal, D.; Masullo, M.; Bedir, E.; Festa, M.; Karayildirim, T.; Alankus-Caliskan, O.; Piacente, S. Triterpene glycosides from Astragalus angustifolius. Planta Med. 2012, 78, 720–729. [Google Scholar] [CrossRef] [PubMed]
- Zhanibekov, A.A.; Naubeev, T.K.; Uteniyazov, K.K.; Bobakulov, Kh.M.; Abdullaev, N.D. Triterpene glycosides from Astragalus. Structure of cyclolehmanoside C from Astragalus lehmannianus. Chem. Nat. Compd. 2013, 49, 475–477. [Google Scholar] [CrossRef]
- Semmar, N.; Tomofumi, M.; Mrabet, Y.; Lacaille-Dubois, M.A. Two New acylated tridesmosidic saponins from Astragalus armatus. Helv. Chim. Acta 2010, 93, 870–876. [Google Scholar] [CrossRef]
- Semmar, N.; Tomofumi, M.; Mrabet, Y.; Lacaille-Dubois, M.A. Antitrypanosomal cycloartane glycosides from Astragalus baibutensis. Chem. Biodivers. 2006, 3, 923–929. [Google Scholar] [CrossRef] [PubMed]
- Barbic, M.; Macabeo, A.P.G.; Kreft, S.; Heilmann, J. Cycloastragenol glycosides from Astragalus illyricus. Biochem. Syst. Ecol. 2010, 38, 460–462. [Google Scholar] [CrossRef]
- Zhang, L.J.; Liu, H.K.; Hsiao, P.C.; Kuo, L.M.Y.; Lee, I.-J.; Wu, T.S.; Chiou, W.F.; Kuo, Y.H. New isoflavonoid glycosides and related constituents from Astragali Radix (Astragalus membranaceus) and their inhibitory activity on nitric oxide production. J. Agric. Food Chem. 2011, 59, 1131–1137. [Google Scholar] [CrossRef] [PubMed]
- Iskenderov, D.A.; Keneshov, B.M.; Isaev, M.I. Triterpene glycosides from Astragalus and their genins. LXXVI. Glycosides from Astragalus sieversianus. Chem. Nat. Compd. 2008, 44, 319–323. [Google Scholar] [CrossRef]
- Isaev, I.M.; Iskenderov, D.A.; Isaev, M.I. Triterpene glycosides from Astragalus and their genins. LXXXVII. Chemical transformation of cycloartanes. IX. Partial synthesis of cycloasalgenin. Chem. Nat. Compd. 2010, 46, 407–411. [Google Scholar] [CrossRef]
- Pistelli, L.; Giachi, I.; Lepori, E.; Bertoli, A. Further saponins and flavonoids from Astragalus verrucosus Moris. Pharm. Biol. 2003, 41, 568–572. [Google Scholar] [CrossRef]
- Gigoshvili, T.I.; Alaniya, M.D.; Tsitsishvili, V.G.; Foure, R.; Debrauver, L.; Kemertelidze, E.P. Structure of cyclogaleginoside E from Astragalus galegiformis. Chem. Nat. Compd. 2003, 39, 373–378. [Google Scholar] [CrossRef]
- Alaniya, M.D.; Gigoshvili, T.I. New cycloartane glycosides from Astragalus caucasicus and Astragalus galegiformis. Chem. Nat. Compd. 2012, 48, 914–916. [Google Scholar] [CrossRef]
- Alaniya, M.D.; Chkadua, N.F.; Gigoshvili, T.I.; Kemertelidze, E.P. Cycloascauloside A from Astragalus caucasicus leaves. Chem. Nat. Compd. 2006, 42, 445–448. [Google Scholar] [CrossRef]
- Alaniya, M.D.; Gigoshvili, T.I.; Kavtaradze, N.S. Cyclogaleginoside D from Astragalus galegiformis stems. Chem. Nat. Compd. 2006, 42, 310–312. [Google Scholar] [CrossRef]
- Iskenderov, D.A.; Isaev, I.M.; Isaev, M.I. Triterpene glycosides from Astragalus and their genins. LXXXIII. Structure of cyclomacroside A. Chem. Nat. Compd. 2009, 45, 656–659. [Google Scholar] [CrossRef]
- Radwan, M.M.; Farooq, A.; El-Sebakhy, N.A.; Asaad, A.M.; Toaima, S.M.; Kingston, D.G. Acetals of three new cycloartane-type saponins from Egyptian collections of Astragalus tomentosus. J. Nat. Prod. 2004, 67, 487–490. [Google Scholar] [CrossRef] [PubMed]
- Kuang, H.; Okada, Y.; Yang, B.; Tian, Z.; Okuyama, T. Secocycloartane triterpenoidal saponins from the leaves of Astragalus membranaceus Bunge. Helv. Chim. Acta 2009, 92, 950–958. [Google Scholar] [CrossRef]
- Gulcemal, D.; Masullo, M.; Napolitano, A.; Karayildirim, T.; Bedir, E.; Alankus-Caliskan, O.; Piacente, S. Oleanane glycosides from Astragalus tauricolus: Isolation and structural elucidation based on a preliminary liquid chromatography-electrospray ionization tandem mass spectrometry profiling. Phytochemistry 2013, 86, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Mitaine-Offer, A.C.; Miyamoto, T.; Semmar, N.; Jay, M.; Lacaille-Dubois, M.A. A new oleanane glycoside from the roots of Astragalus caprinus. Magn. Reson. Chem. 2006, 44, 713–716. [Google Scholar] [CrossRef] [PubMed]
- Krasteva, I.; Nikolov, S. Flavonoids in Astragalus corniculatus. Quim. Nova 2008, 31, 59–60. [Google Scholar] [CrossRef]
- Guzhva, N.N. Flavonoids and hydroxycinnamic acids from Astragalus asper. Chem. Nat. Compd. 2010, 46, 303–304. [Google Scholar] [CrossRef]
- Fathiazad, F.; Movafeghi, A.; Khosropanah, M.K. Flavonol glycosides from the leaves of Astragalus microcephalus. Int. J. Biosci. 2012, 2, 23–28. [Google Scholar]
- Kavtaradze, N.S.; Alaniya, M.D.; Mshvildadze, V.D.; Skhirtladze, A.V.; Lavoie, S.; Pichette, A. Flavonoids from Astragalus microcephalus. Chem. Nat. Compd. 2011, 46, 971–973. [Google Scholar] [CrossRef]
- Alaniya, M.D.; Kavtaradze, N.S.; Bassarello, C.; Skhirtladze, A.V.; Pizza, C.; Kutateladze, I. Flavonoid glycosides from Astragalus galegiformis leaves. Chem. Nat. Compd. 2006, 42, 681–685. [Google Scholar] [CrossRef]
- Alaniya, M.D.; Kavtaradze, N.S.; Bassarello, C.; Skhirtladze, A.V.; Pizza, C.; Kutateladze, I. Flavonoids from Astragalus hamosus. Nat. Prod. Res. 2007, 21, 392–395. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedula, V.S.P.; Prakash, I. Flavonoids from Astragalus propinquus. J. Chem. Pharm. Res. 2013, 5, 261–265. [Google Scholar]
- Li, W.; Sun, Y.N.; Yan, X.T.; Yang, S.Y.; Kim, S.; Lee, Y.M.; Koh, Y.S.; Kim, Y.H. Flavonoids from Astragalus membranaceus and their inhibitory effects on LPS-stimulated pro-inflammatory cytokine production in bone marrow-derived dendritic cells. Arch. Pharm. Res. 2014, 37, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Bai, Y.; Liang, H.; Wang, Z.; Zhao, Y.; Zhang, Q.; Huang, L. Solvent effect in 1H-NMR spectra of 3'-hydroxy-4'-methoxy isoflavonoids from Astragalus membranaceus var. mongholicus. Magn. Reson. Chem. 2006, 44, 708–712. [Google Scholar] [CrossRef]
- Bi, Z.M.; Yu, Q.T.; Li, P.; Lin, Y.; Gao, X.D. Flavonoids from aerial parts of Astragalus membranaceus. Zhongguo Tianran Yaowu 2007, 5, 263–265. [Google Scholar]
- Yu, D.H.; Bao, Y.M.; Wei, C.; An, L.J. Studies of chemical constituents and their antioxidant activities from Astragalus mongholicus Bunge. Biomed. Environ. Sci. 2005, 18, 297–301. [Google Scholar] [PubMed]
- Liu, W.; Chen, J.; Zuo, W.J.; Li, X.; Wang, J.H. A new isoflavane from processed Astragalus membranaceus. Chin. Chem. Lett. 2007, 18, 1092–1094. [Google Scholar] [CrossRef]
- Abd El-Latif, R.R.; Shabana, M.H.; El-Gandour, A.H.; Mansour, R.M.; Sharaf, M. A new isoflavone from Astragalus peregrinus. Chem. Nat. Compd. 2003, 39, 536–537. [Google Scholar] [CrossRef]
- Li, R.; Zhou, Y; Qiao, Li; Fu, H.; Pei, Y. Separation and identification of chemical constituents from Astragalus membranaceus. Shenyang Yaoke Daxue Xuebao 2007, 24, 20–22. [Google Scholar]
- Yao, D.; Wang, H. Monosaccharide composition in Radix Astragali polysaccharides by gas chromatography. Med. Plant 2012, 3, 36–38. [Google Scholar]
- Xu, D.J.; Xia, Q.; Wang, J.J.; Wang, P.P. Molecular weight and monosaccharide composition of Astragalus polysaccharides. Molecules 2008, 13, 2408–2415. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Liu, X. Determination of eight metal elements in Astragalus by microwave digestion-flame atomic absorption spectrometry. Fenxi Kexue Xuebao 2009, 25, 605–608. [Google Scholar]
- Kim, J.H.; Kim, M.H.; Yang, G.; Huh, Y.; Kim, S.H.; Yang, W.M. Effects of topical application of Astragalus membranaceus on allergic dermatitis. Immunopharmacol. Immunotoxicol. 2013, 35, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Ryu, M.; Kim, E.H.; Chun, M.; Kang, S.; Shim, B.; Yu, Y.B.; Jeong, G.; Lee, J.S. Astragali Radix elicits anti-inflammation via activation of MKP-1, concomitant with attenuation of p38 and Erk. J. Ethnopharmacol. 2008, 115, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Chen, X.; Zhang, Y.; Xu, J.; Zhang, L.; Li, Z.; Liu, W.; Ouyang, J.; Han, S.; He, X. Astragaluspolysaccharide induces anti-inflammatory effects dependent on AMPK activity in palmitate-treated RAW264.7 cells. Int. J. Mol. Med. 2013, 31, 1463–1470. [Google Scholar] [PubMed]
- Wang, X.; Li, Y.; Yang, X.; Yao, J. Astragalus polysaccharide reduces inflammatory response by decreasing permeability of LPS-infected Caco2 cells. Int. J. Biol. Macromol. 2013, 61, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.Y.; Noh, H.J.; Choi, J.; Lee, K.H.; Lee, M.H.; Lee, J.H.; Hong, Y.; Lee, S.E.; Kim, S.Y.; Kim, G.S. Anti-inflammatory cycloartane-type saponins of Astragalus membranaceus. Molecules 2013, 18, 3725–3732. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Peng, L.H.; Li, N.; Li, Q.M.; Li, P.; Fung, K.P.; Leung, P.C.; Gao, J.Q. The healing and anti-scar effects of astragaloside IV on the wound repair in vitro and in vivo. J. Ethnopharmacol. 2012, 139, 721–727. [Google Scholar] [CrossRef] [PubMed]
- Gui, D.; Huang, J.; Guo, Y.; Chen, J.; Chen, Y.; Xiao, W.; Liu, X.; Wang, N. Astragaloside IV ameliorates renal injury in streptozotocin-induced diabetic rats through inhibiting NF-κB-mediated inflammatory genes expression. Cytokine 2013, 61, 970–977. [Google Scholar] [CrossRef] [PubMed]
- Qin, Q.; Niu, J.; Wang, Z.; Xu, W.; Qiao, Z.; Gu, Y. Astragalus membranaceus inhibits inflammation via phospho-p38 mitogen-activated protein kinase (MAPK) and nuclear factor (NF)-κB pathways in advanced glycation end product-stimulated macrophages. Int. J. Mol. Sci. 2012, 13, 8379–8387. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Chen, X.; Zhao, B.; Lv, Y.; Zhang, H.; Liu, H.; Chen, Z.; Chen, Y.; Zeng, X. Astragalus polysaccharides enhance the humoral and cellular immune responses of hepatitis B surface antigen vaccination through inhibiting the expression of transforming growth factor β and the frequency of regulatory T cells. FEMS Immunol. Med. Microbiol. 2011, 63, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Nalbantsoy, A.; Nesil, T.; Yilmaz-Dilsiz, O.; Aksu, G.; Khan, S.; Bedir, E. Evaluation of the immunomodulatory properties in mice and in vitro anti-inflammatory activity of cycloartane type saponins from Astragalus species. J. Ethnopharmcol. 2012, 139, 574–581. [Google Scholar] [CrossRef]
- Huang, L.F.; Yao, Y.M.; Li, J.F.; Zhang, S.W.; Li, W.X.; Dong, N.; Yu, Y.; Sheng, Z.Y. The effect of Astragaloside IV on immune function of regulatory T cell mediated by high mobility group box 1 protein in vitro. Fitoterapia 2012, 83, 1514–1522. [Google Scholar] [CrossRef] [PubMed]
- Tian, Q.E.; Li, H.D.; Yan, M.; Cai, H.L.; Tan, Q.Y.; Zhang, W.Y. Astragaluspolysaccharides can regulate cytokine and P-glycoprotein expression in H22 tumor-bearing mice. World J. Gastroentero. 2012, 18, 7079–7086. [Google Scholar] [CrossRef]
- Qi, H.; Wei, L.; Han, Y.; Zhang, Q.; Lau, A.S.; Rong, J. Proteomic characterization of the cellular response to chemopreventive triterpenoid astragaloside IV in human hepatocellular carcinoma cell line HepG2. Int. J. Oncol. 2010, 36, 725–735. [Google Scholar] [PubMed]
- Huang, C.; Xu, D.; Xia, Q.; Wang, P.; Rong, C.; Su, Y. Reversal of P-glycoprotein-mediated multidrug resistance of human hepatic cancer cells by Astragaloside II. J. Pharm. Pharmacol. 2012, 64, 1741–1750. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Zhuang, Y.; Pan, J.; Wang, H.; Li, H.; Yu, Y.; Wang, D. Investigation of effects and mechanisms of total flavonoids of Astragalus and calycosin on human erythroleukemia cells. Oxid. Med. Cell Longev. 2012, 2012, 209843. [Google Scholar] [PubMed]
- Ma, X.; Zhang, K.; Li, H.; Han, S.; Ma, Z.; Tu, P. Extracts from Astragalus membranaceus limit myocardial cell death and improve cardiac function in a rat model of myocardial ischemia. J. Ethnopharmacol. 2013, 149, 720–728. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Zhuang, Y.; Tian, Y.; Thomas, G.N.; Ying, M.; Tomlinson, B. Study of the effects of total flavonoids of Astragalus on atherosclerosis formation and potential mechanisms. Oxid. Med. Cell. Longev. 2012, 2012. doi:org/10.1155/2012/282383. [Google Scholar]
- Bai, F.; Makino, T.; Kono, K.; Nagatsu, A.; Ono, T.; Mizukami, H. Calycosin and formononetin from astragalus root enhance dimethylarginine dimethylaminohydrolase 2 and nitric oxide synthase expressions in Madin Darby Canine Kidney II cells. J. Nat. Med. 2013, 67, 782–789. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Zhao, J.; He, G.; Sun, X.; Huang, X.; Hao, L. Effects of astragaloside IV on action potentials and ionic currents in guinea-pig ventricular myocytes. Biol. Pharm. Bull. 2013, 36, 515–521. [Google Scholar] [CrossRef]
- Liu, M; Wu, K; Mao, X; Wu, Y; Ouyang, J. Astragalus polysaccharide improves insulin sensitivity in KKAy mice: Regulation of PKB/GLUT4 signaling in skeletal muscle. J. Ethnopharmacol. 2009, 127, 32–37. [Google Scholar]
- Zhou, X.; Xu, Y.; Yang, G.; Li, F. Increased galectin-1 expression in muscle of Astragalus polysaccharide-treated Type 1 diabetic mice. J. Nat. Med. 2011, 65, 500–507. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Zhang, Y.; Sun, S.; Shen, J.; Qiu, J.; Yin, X.; Yin, H.; Jiang, S. Inhibitory effects of astragaloside IV on diabetic peripheral neuropathy in rats. Can. J. Physiol. Pharmacol. 2006, 84, 579–587. [Google Scholar] [CrossRef] [PubMed]
- Motomura, K.; Fujiwara, Y.; Kiyota, N.; Tsurushima, K.; Takeya, M.; Nohara, T.; Nagai, R.; Ikeda, T. Astragalosides isolated from the root of astragalus radix inhibit the formation of advanced glycation end products. J. Agric. Food Chem. 2009, 57, 7666–7672. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.J.; Yang, K.S. Antilipidperoxidative activity of Astragalus membranaceus. Yakhak. Hoechi. 2005, 49, 11–19. [Google Scholar]
- Li, X.; Wang, X.; Han, C.; Wang, X.; Xing, G.; Zhou, L.; Li, G; Niu, Y. Astragaloside IV suppresses collagen production of activated hepatic stellate cells via oxidative stress-mediated p38 MAPK pathway. Free Radic. Biol. Med. 2013, 60, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Lei, H.; Wang, B.; Li, W.P.; Yang, Y.; Zhou, A.W.; Chen, M.Z. Anti-aging effect of astragalosides and its mechanism of action. Acta Pharmacol. Sin. 2003, 24, 230–234. [Google Scholar] [PubMed]
- Gao, X.; Li, L.; Liu, B. Effect of Astragalus polysaccharides on stress response ability and regulation of free radicals in mice. Zhongguo Yufang Yixue Zazhi 2010, 11, 120–121. [Google Scholar]
- Han, L.; Yu, C.; Lin, H. Ultra-performance liquid chromatography for quantification of flavone in Astragalus membranaceus and its preparation. Zhongguo Shiyan Fangjixue Zazhi 2012, 18, 115–118. [Google Scholar]
- Zhang, Y.; Liu, N.; Liu, S.; Yang, S.; Zhang, S. Comparative study on flavonoids extracted from Astragalus membranaceus of different growth year. Yanbian Daxue Yixue Xuebao 2011, 34, 34–37. [Google Scholar]
- Ye, G.; Tang, Y.H.; Xia, G.X.; Sun, Z.L.; Li, Z.X.; Huang, C.G. Characterization of anti-coxsackie virus B3 constituents of Radix Astragali by high-performance liquid chromatography coupled with electrospray ionization tandem mass spectrometry. Biomed. Chromatogr. 2010, 24, 1147–1151. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Xu, Y.; Chen, X. Analysis of Radix Astragali and its water extract by infrared spectroscopy. Guangpu Shiyanshi 2012, 29, 2823–2826. [Google Scholar]
- Movafeghi, A.; Djozan, D.; Razeghi, J.A.; Baheri, T. Identification of volatile organic compounds in leaves, roots and gum of Astragalus compactus Lam. using solid phase microextraction followed by GC-MS analysis. Nat. Prod. Res. 2010, 24, 703–709. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Lu, H.; Zhao, B.; Huo, X.; Wan, X.; Liu, Z.; Wang, Z. Preliminary chemical test of Astragalus hamiensis S.B.Ho and alkaloids analysis. Zhongguo Shouyi Xuebao 2009, 29, 1217–1221. [Google Scholar]
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, X.; Qu, L.; Dong, Y.; Han, L.; Liu, E.; Fang, S.; Zhang, Y.; Wang, T. A Review of Recent Research Progress on the Astragalus Genus. Molecules 2014, 19, 18850-18880. https://doi.org/10.3390/molecules191118850
Li X, Qu L, Dong Y, Han L, Liu E, Fang S, Zhang Y, Wang T. A Review of Recent Research Progress on the Astragalus Genus. Molecules. 2014; 19(11):18850-18880. https://doi.org/10.3390/molecules191118850
Chicago/Turabian StyleLi, Xiaoxia, Lu Qu, Yongzhe Dong, Lifeng Han, Erwei Liu, Shiming Fang, Yi Zhang, and Tao Wang. 2014. "A Review of Recent Research Progress on the Astragalus Genus" Molecules 19, no. 11: 18850-18880. https://doi.org/10.3390/molecules191118850
APA StyleLi, X., Qu, L., Dong, Y., Han, L., Liu, E., Fang, S., Zhang, Y., & Wang, T. (2014). A Review of Recent Research Progress on the Astragalus Genus. Molecules, 19(11), 18850-18880. https://doi.org/10.3390/molecules191118850




