Abstract
An NMR titration method has been used to simultaneously measure the acid dissociation constant (pKa) and the intramolecular NHO prototropic constant ΔKNHO on a set of Schiff bases. The model compounds were synthesized from benzylamine and substituted ortho-hydroxyaldehydes, appropriately substituted with electron-donating and electron-withdrawing groups to modulate the acidity of the intramolecular NHO hydrogen bond. The structure in solution was established by 1H-, 13C- and 15N-NMR spectroscopy. The physicochemical parameters of the intramolecular NHO hydrogen bond (pKa, ΔKNHO and ΔΔG°) were obtained from 1H-NMR titration data and pH measurements. The Henderson–Hasselbalch data analysis indicated that the systems are weakly acidic, and the predominant NHO equilibrium was established using Polster–Lachmann δ-diagram analysis and Perrin model data linearization.
1. Introduction
Schiff bases are a great topic of basic research, that to date have an important place in organic chemistry and they have a great versatility in different fields of study. They have different biologic applications as antitumor agents [1,2,3,4,5], in the strengthening of immune response for cancer, in leukemia, in HIV, as anticonvulsant, antibacterials, antifungal, antiinflammatory, as prodrugs [6,7,8,9,10,11,12,13,14,15] and as study models in the intramolecular hydrogen bond from cofactor pyridoxal-5-phosphate [16,17,18,19,20]. They are also of interest because of their solvatochromic, thermochromic and photochromic properties with applications in optical recording technology, molecular electronics and photonics [21,22,23,24,25,26,27,28,29,30].
The Schiff bases derived from ortho-hydroxyaromatic aldehydes that are pentaconjugated non-symmetric systems [31] in which proton transfer from the oxygen hydroxyl to the nitrogen of imine, through the NHO hydrogen bond is observed (Scheme 1) have been extensively studied in recent years [32,33,34,35,36,37,38,39,40,41,42,43,44,45,46].
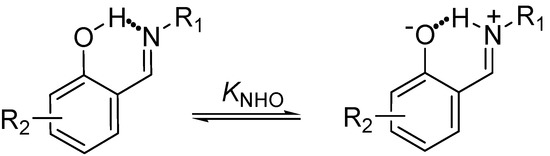
Scheme 1.
Prototropic 1,5 rearrangement in Schiff bases.
Scheme 1.
Prototropic 1,5 rearrangement in Schiff bases.
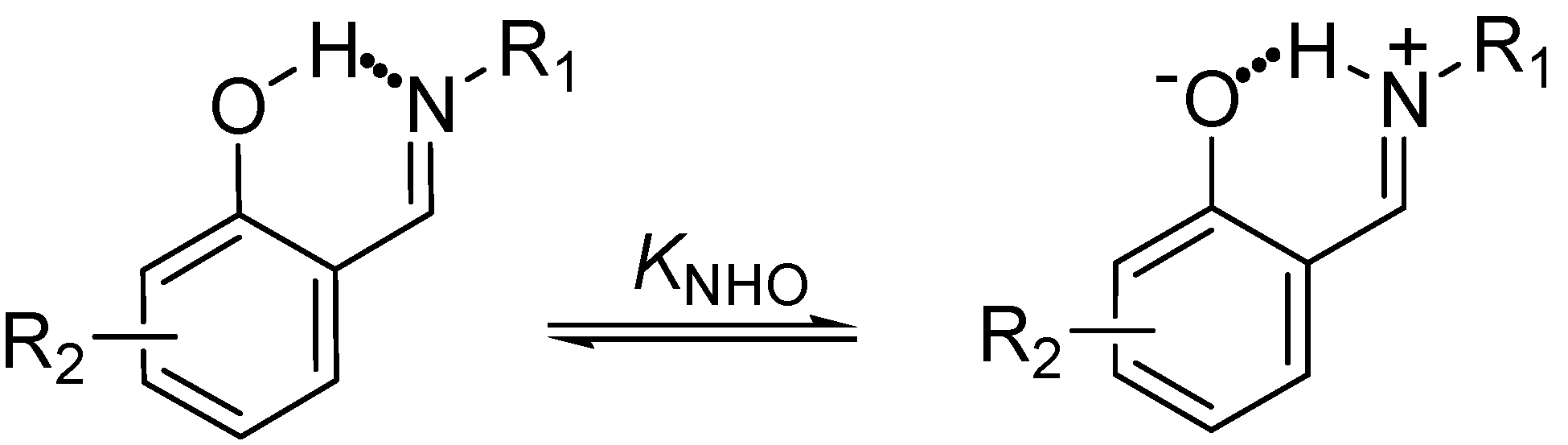
In these investigations several analytical methods for determining the prototropic equilibrium have been applied, such as FT-IR spectroscopy and X-ray diffraction in the solid state [45,46,47,48], as well as solution 1H-, 13C- and 15N-NMR [33,34,49,50,51]. This 1,5 tautomeric equilibrium is directly affected by the substituents [52,53,54,55,56] attached to both the phenyl group and the imine nitrogen which exert a strong influence on the acidity of the OH group, the basicity of the nitrogen atom and thus the NHO bond strength. Substituents also greatly increase the stability of the compounds by the effect of hydrogen bonding assisted by resonance (RAHB) [57,58,59]; preferences have been found in the position of the hydrogen either linked to oxygen (N∙∙∙H-O) or nitrogen (N-H∙∙∙O) [32,33,46,60] atoms and even being in the middle of both (O−∙∙∙H∙∙∙N+) [60,61].
For a prototropic acid-base-system HA in equilibrium, its equilibrium constant Ka is expressed by Equation (1), which after logarithms becomes the Henderson-Hasselbalch Equation (2):
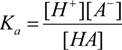
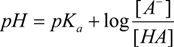
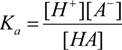
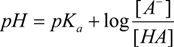
Equation (2) is directly related to the chemical shifts of active nuclei in NMR, which are dependent on pH changes, this leads to Equation (3):
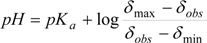
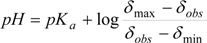
The pKa is experimentally obtained, using the tabulation of log[(δmax − δobs)/(δobs − δmin)] against pH, where δmin and δmax are the chemical shifts in the inflection points in the titration curve, while δobs is the observed chemical shift during the course of the titration, so the equilibrium point is at point zero, which corresponds to pH = pKa [44,62,63,64,65,66,67]. This method has been extensively used because of its simplicity, however is limited by variability in pH readings and accuracy in measuring the volumes of the titrant.
Polster and Lachmann postulated the Gibbs triangle method, which later emerged as the absorbance diagram (A-diagram) or chemical shift diagram (δ-diagram), depending on the spectrometry used for the analysis of data from a titration, for the study of acid-base systems [62,68]. This method allows the evaluation of the quotient of acidity constants (ΔKa) of two or more compounds, mainly in diprotic and polyprotic acid-base systems [68], on the bases of a ratio of distances from the Gibbs triangle which is independent of pH readings [68].
Later, Perrin et al. [69,70,71,72] also developed a mathematical model for the determination of ΔKa for mixtures of isomers in equilibrium with independency from the pH readings by drawing δ-diagrams also, so this model can be applied to the analysis of acid-base equilibrium mainly in monoprotic systems. Then for two acids HA and HB, the quotient of their acidity constants ΔKa, can be measured by the variation in chemical shifts due to changes in the acidity of the systems:
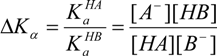
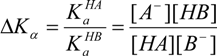
Equation (4), written in terms of chemical shifts when ΔKa ≠ 1, allows the evaluation of ΔKa as the slope of a straight line, Equation (5):
where δA°, δB° are the chemical shifts from species at the start of the titration, δa, δb are the chemical shifts observed during the titration, andδHA, δHB are the chemical shifts from species at the end of the titration.
(δb − δB°)(δAH − δa) = ∆Kα(δa − δA°)(δBH − δb)
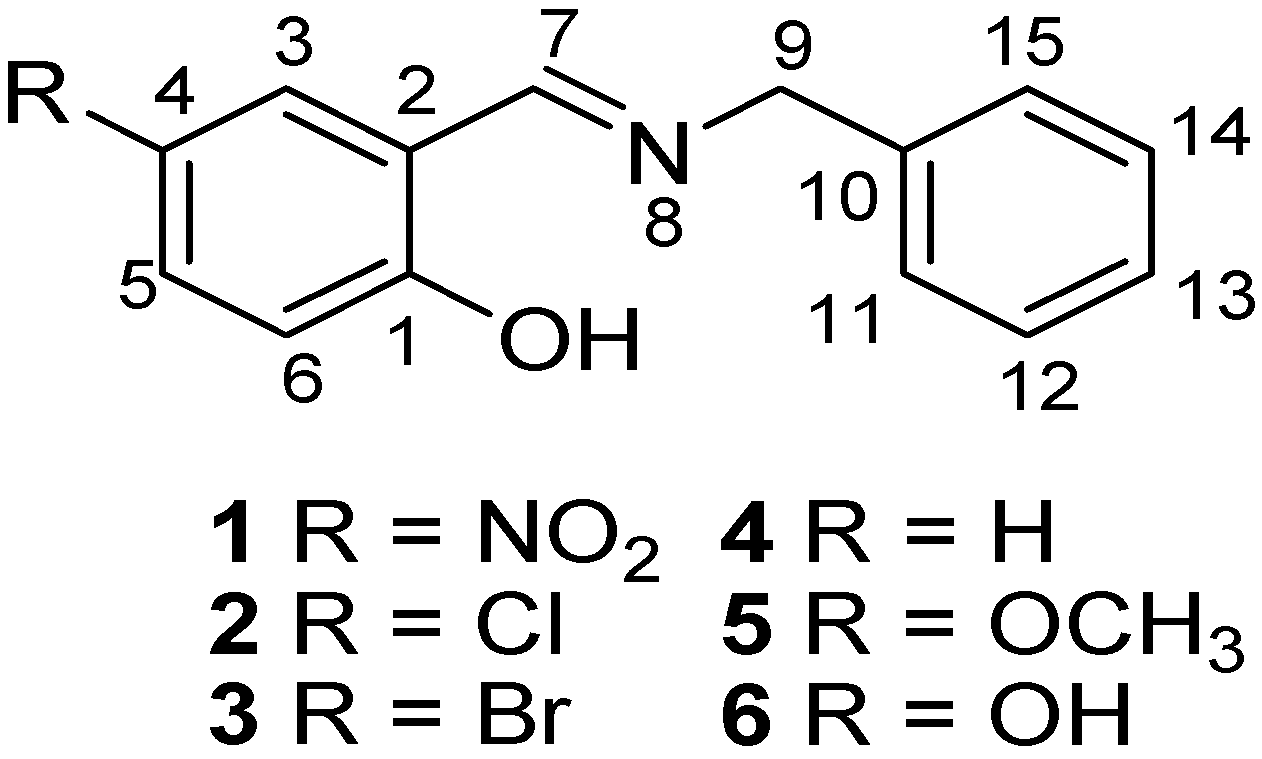
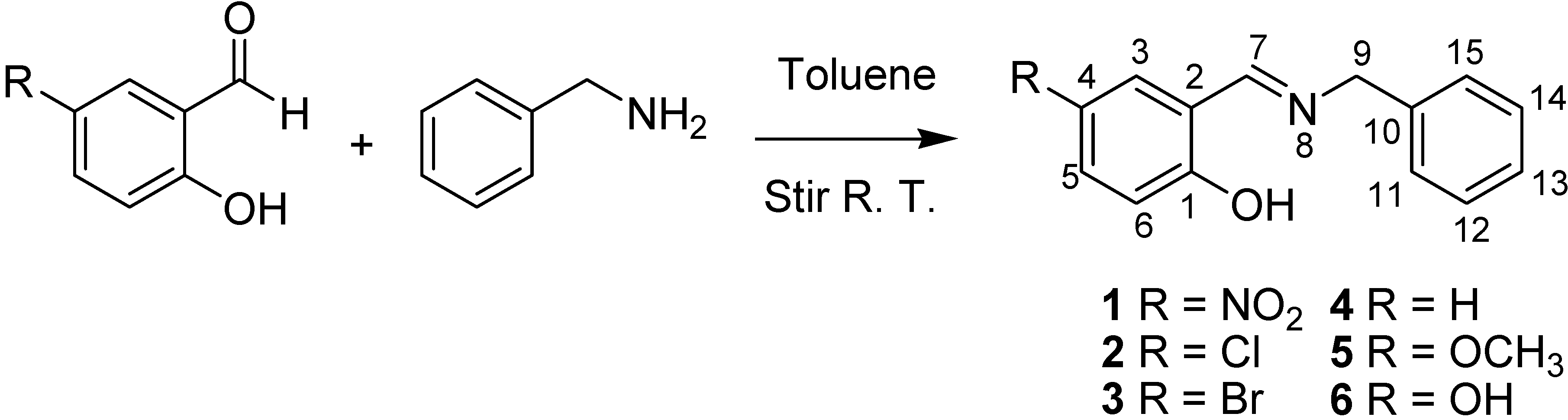
In this contribution, both the Perrin and Polster-Lachmann models are applied to the study of intramolecular hydrogen bonds that involve prototropic equilibrium with the aim to find with accuracy and selectivity the position of the proton on the oxygen or nitrogen atoms. The model compounds were a set of Schiff base derivatives of 5-nitrosalicylaldehyde, 5-chlorosalicylaldehyde, 5-bromo-salicylaldehyde, salicylaldehyde, 5-methoxysalicyladehyde and 5-hydroxysalicyladehyde with benzylamine (compounds 1–6, Figure 1). The substituents were selected in order to cover a broad range of both electrodonating (ED) and electrowithdrawing (EW) groups whose electronic effects could modulate the NHO hydrogen bonding scheme. 1H-NMR spectrometry was used as the titration method.
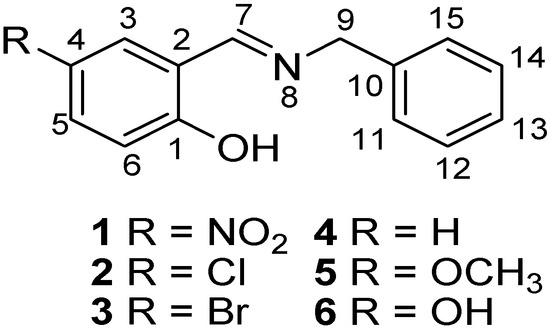
Figure 1.
Schiff base derivatives of 1–6.
Figure 1.
Schiff base derivatives of 1–6.
2. Results and Discussion
2.1. NMR Spectra
Synthesized compounds were identified by 1H-, 13C- and 15N-NMR. The 1H-NMR spectra of compounds 1–6 in DMSO-d6 solution showed remarkable changes in the chemical shift of the acidic proton NHO in the range of 12.53–14.34 ppm, in response to the electronic character of the substituent R. Since a larger value in the chemical shift indicates a greater acidity of the proton, compound 1 has the largest acidity and compound 6 has the lowest acidity. Simultaneously the chemical shift of protons H3, H5, H6 and H7 were affected too.
The 13C spectra of all compounds showed clear shielding and deshielding effects, according to the substituent, mainly from C1 to C7. The chemical shifts of compound 1 were more affected than those of compounds 2–6, especially the carbon atoms C1 and C4. Compound 1, the NO2 derivative, showed a chemical shift of 175.8 and 136.9 ppm for C1 and C4, respectively, where C1 is in the range of carbonyl chemical shifts (170 to 200 ppm) while C4 is in the range of nitro Schiff base compounds (130 to 150 ppm). The 15N chemical shift of compound 1 was −162.1 ppm, indicating an average between imine-enamine forms, therefore in this last compound the zwitterionic structure (Scheme 2a) is favored and the hydrogen H8 is localized with the nitrogen atom (+N–H∙∙∙O).
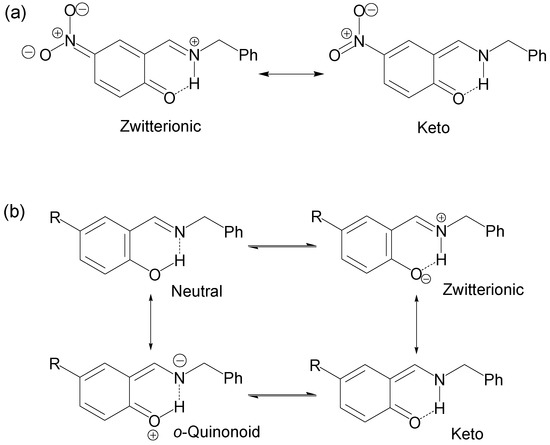
Scheme 2.
Possible resonance and equilibrium structures for compounds 1–6.
Scheme 2.
Possible resonance and equilibrium structures for compounds 1–6.
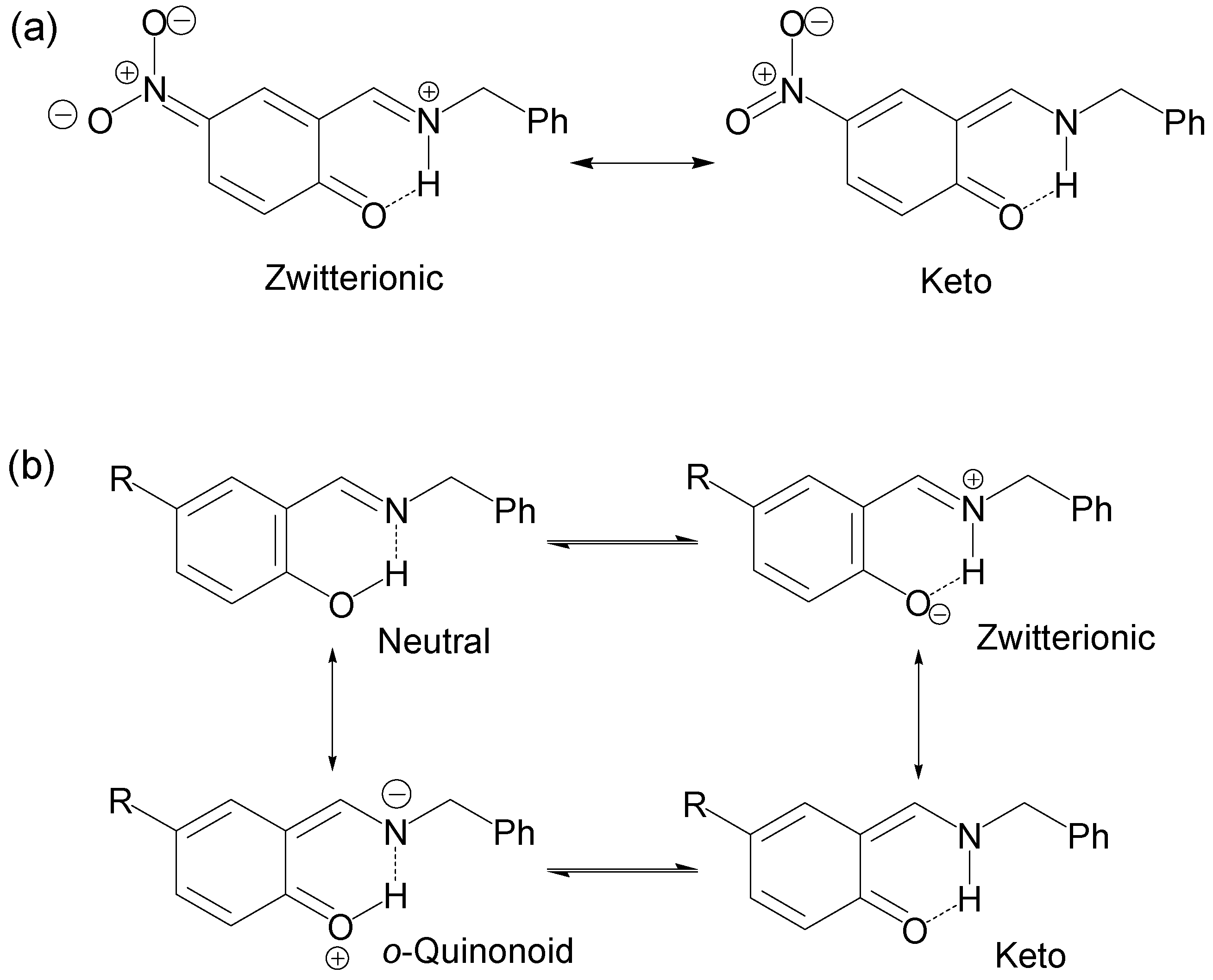
In the case of compounds 2–6 the chemical shifts of C1 appear at lower frequencies from 160.4 to 153.5 ppm, a region characteristic of OH structures (150–160 ppm) and the chemical shifts of the imine C7=N appear from 165.8 to 167.4 ppm, a less significant variation. The 15N chemical shifts for compounds 2–6 were in the range of −79.7 to −81.8 ppm (−50 to −90 ppm for imine), in agreement with a neutral N∙∙∙H–O tautomeric form with the hydrogen H8 is localized with the oxygen atom (Scheme 2b). The NMR chemical shifts of compounds 1, 3, 4 and 5 have already been reported [73] and are in agreement with the above mentioned results, except for the nitro derivative 1 for which the authors conclude that the N–H tautomer is present in solution instead of the zwitterion form proposed herein.
2.2. NMR Titration
All compounds were titrated in CD3OD solution with NaOD, and only compound 2 was further titrated with DCl. 1H-NMR spectra were recorded after each aliquot of titrant and simultaneously the pH was measured following each recorded spectrum. The resonances of H6 and H9 were used to plot pH vs. δ1H, because these protons were most affected by deprotonation of the labile hydrogen.
Compound 2 was initially titrated with DCl, however hydrolysis occured with the acid titrant and only five 1H spectra and their corresponding pH readings could be recorded, so subsequently all compounds were titrated with NaOD (Figure 2A).
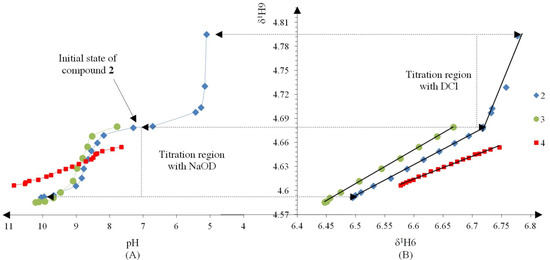
Figure 2.
(A) Titration curve of compounds 2–4; compound 2 was titrated with DCl; only the titration region with NaOD was used to calculate the pKa values with the Henderson-Hasselbalch equation; (B) δ-Diagram of δ1H6 vs. δ1H9 of compounds 2–4; the initial data obtained was not linearized but after using the Perrin model the data became for linearized compounds 2 (Cl), 3 (Br) and 4 (H) to obtain the slope ΔKNHO as shown in Scheme 3.
Figure 2.
(A) Titration curve of compounds 2–4; compound 2 was titrated with DCl; only the titration region with NaOD was used to calculate the pKa values with the Henderson-Hasselbalch equation; (B) δ-Diagram of δ1H6 vs. δ1H9 of compounds 2–4; the initial data obtained was not linearized but after using the Perrin model the data became for linearized compounds 2 (Cl), 3 (Br) and 4 (H) to obtain the slope ΔKNHO as shown in Scheme 3.
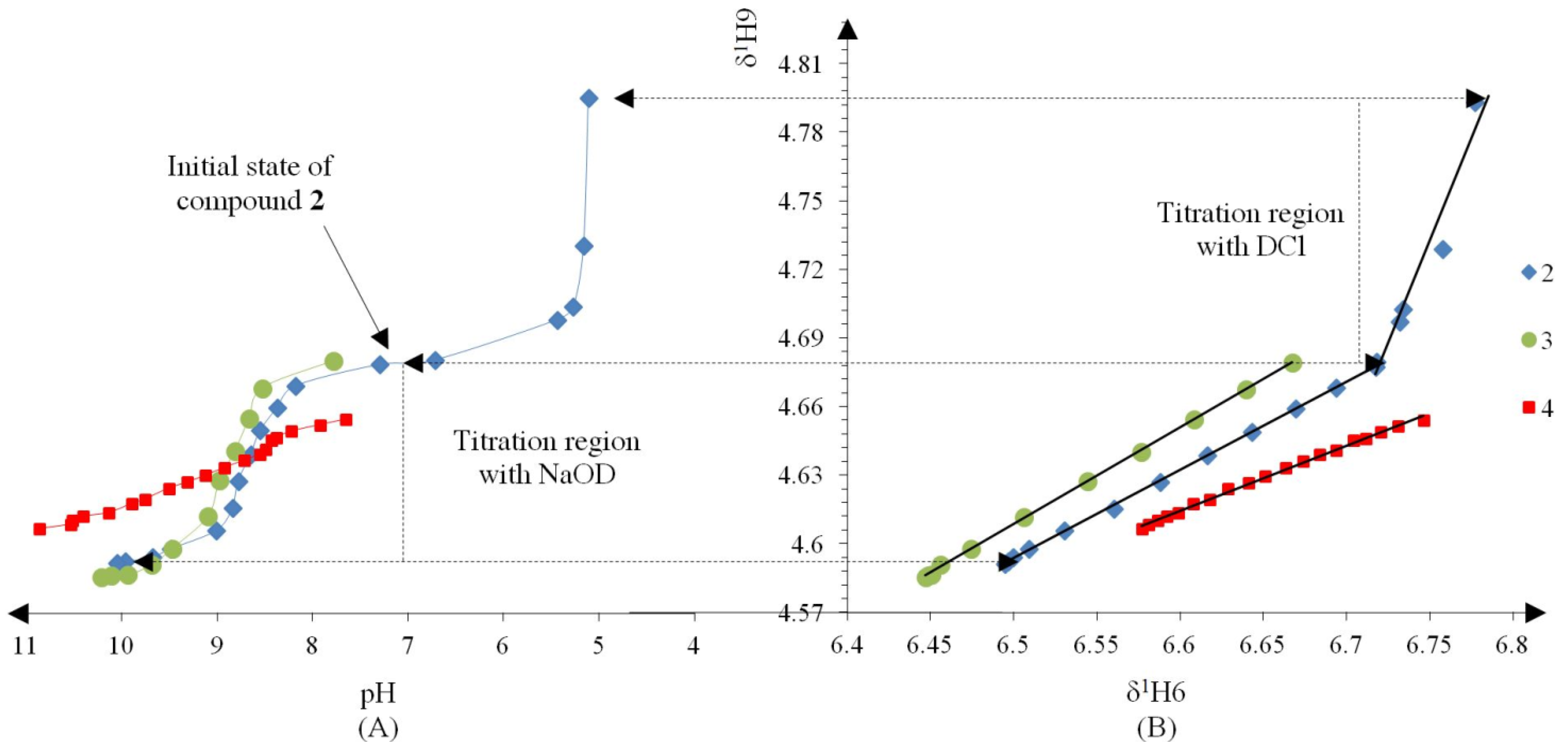
The Henderson-Hasselbalch equation was used to measure pKa values of the compounds by a graphical method with plots of pH against log[(δH9max − δH9obs)/(δH9obs − δH9min)] (Figure 3) from the titration curve, while ΔKNHO was obtained from the δ-diagram (Figure 2B) using the Perrin model linearization [(δH9 − δH9°)(δH6e− δH6) vs. (δH6 − δH6°)(δH9e− δH9)] for compounds 2, 3, 4 and by Polster and Lachmann analysis for compounds 1, 5 and 6. Table 1 summarizes the physicochemical parameters obtained by the graphical methods mentioned above.
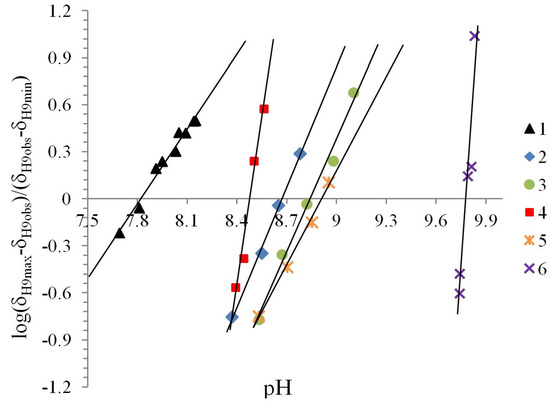
Figure 3.
The experimental pKa were found using the plot of the Henderson-Hasselbalch equation, when log[(δH9max − δH9obs)/(δH9obs − δH9min)] = 0 then the pH intercept is the pKa value.
Figure 3.
The experimental pKa were found using the plot of the Henderson-Hasselbalch equation, when log[(δH9max − δH9obs)/(δH9obs − δH9min)] = 0 then the pH intercept is the pKa value.
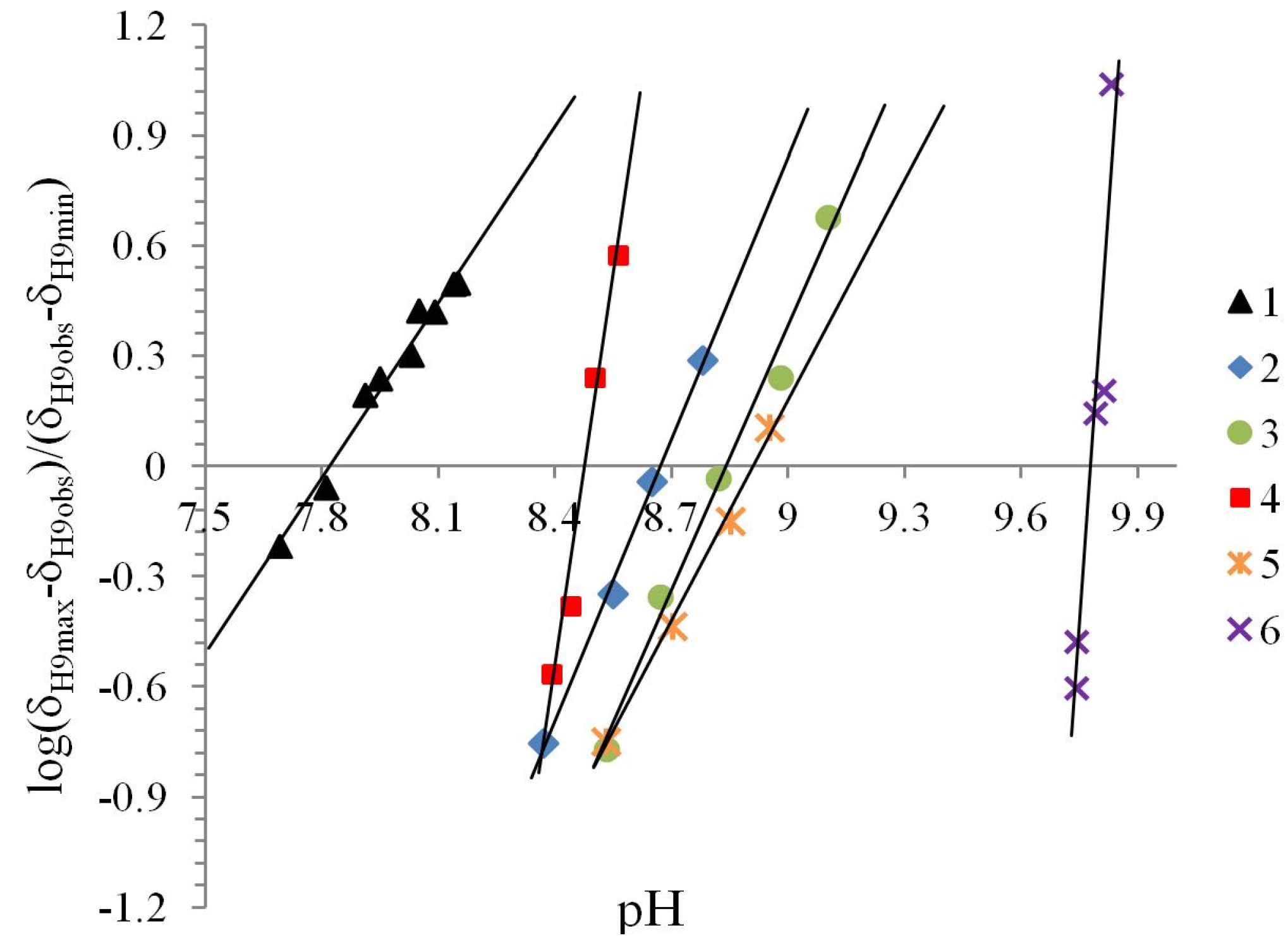

Table 1.
Physicochemical parameters of compounds 1–6 at 296.15 ± 1 K in CD3OD solution.
| Compound | Ka/10−9 | pKa | ΔKNHO | ΔpKNHO | ΔΔG° [a] |
|---|---|---|---|---|---|
| 1 | 15.1 | 7.8 | 1.04(±0.05) | −0.017 | −0.097 |
| 2 | 2.13 | 8.6 | 1.031(±0.002) | −0.0133 | −0.075 |
| 3 | 1.44 | 8.8 | 0.986(±0.002) | 0.006 | 0.036 |
| 4 | 3.33 | 8.4 | 0.841(±0.005) | 0.0754 | 0.426 |
| 5 | 1.23 | 8.9 | 1.021(±0.014) | −0.01 | −0.052 |
| 6 | 0.17 | 9.7 | 1.02(±0.02) | −0.001 | −0.004 |
[a] ΔΔG° = −RTlnΔKNHO (kJ mol−1 K−1).
The pKa values obtained by the Henderson-Hasselbalch equation for all compounds were greater than 7 and less than 11, showing that these compounds are weak acids, the pKa value increases in the order NO2 < H < Cl < Br < OMe < OH. Only compound 6 showed two pKa values, the value of 9.7 belongs to NHO and the second value of 9.8 to the phenolic hydroxyl group C4-OH. On the other hand the ΔΔG° values, associated with the prototropic NHO equilibrium, are favored in the order NO2 > Cl > OMe ≈ OH > Br > H.
Figure 2 shows the titration curve with full pH scale (A) and the δ-diagram (B) of compounds 2 to 4; only the data region titrated with NaOD was taken for the pKa value calculation. All compounds should show the same shape of δ-diagram as they were titrated with NaOD. However, the titration curves of compounds 2, 3 and 4 showed an almost linear behavior, whereas those of compounds 1 and 5 showed one inflection point and those of compound 6 two inflection points (see Supporting Information). These results indicate that the initial structure of compounds 1–6, at the beginning of the titration, was not the same in agreement with the NMR data discussed above.
On the other hand, the prototropic 1,5-rearrangement (Scheme 3) can be envisaged as composed by two equilibria as depicted by in Figure 2. The quotient of the equilibrium constants KHN and KHO is defined as ΔKNHO, corresponding to the equilibrium constant of the prototropic 1,5-tautomerism. The chemical shifts of H6 and H9 are the most sensitive to changes in the equilibrium positions, thus they were used as probes for KHO and KHN measurements, respectively.
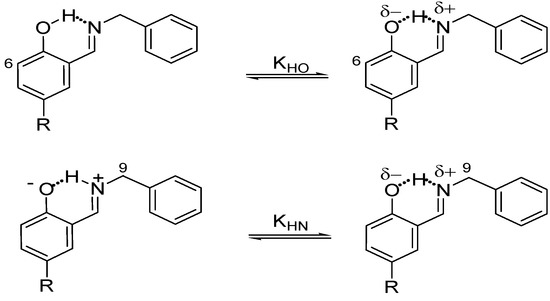
Scheme 3.
Equilibria variation in the prototropic 1,5-tautomeric equilibrium. The quotient of the equilibrium constants KHN and KHO is ΔKNHO.
Scheme 3.
Equilibria variation in the prototropic 1,5-tautomeric equilibrium. The quotient of the equilibrium constants KHN and KHO is ΔKNHO.
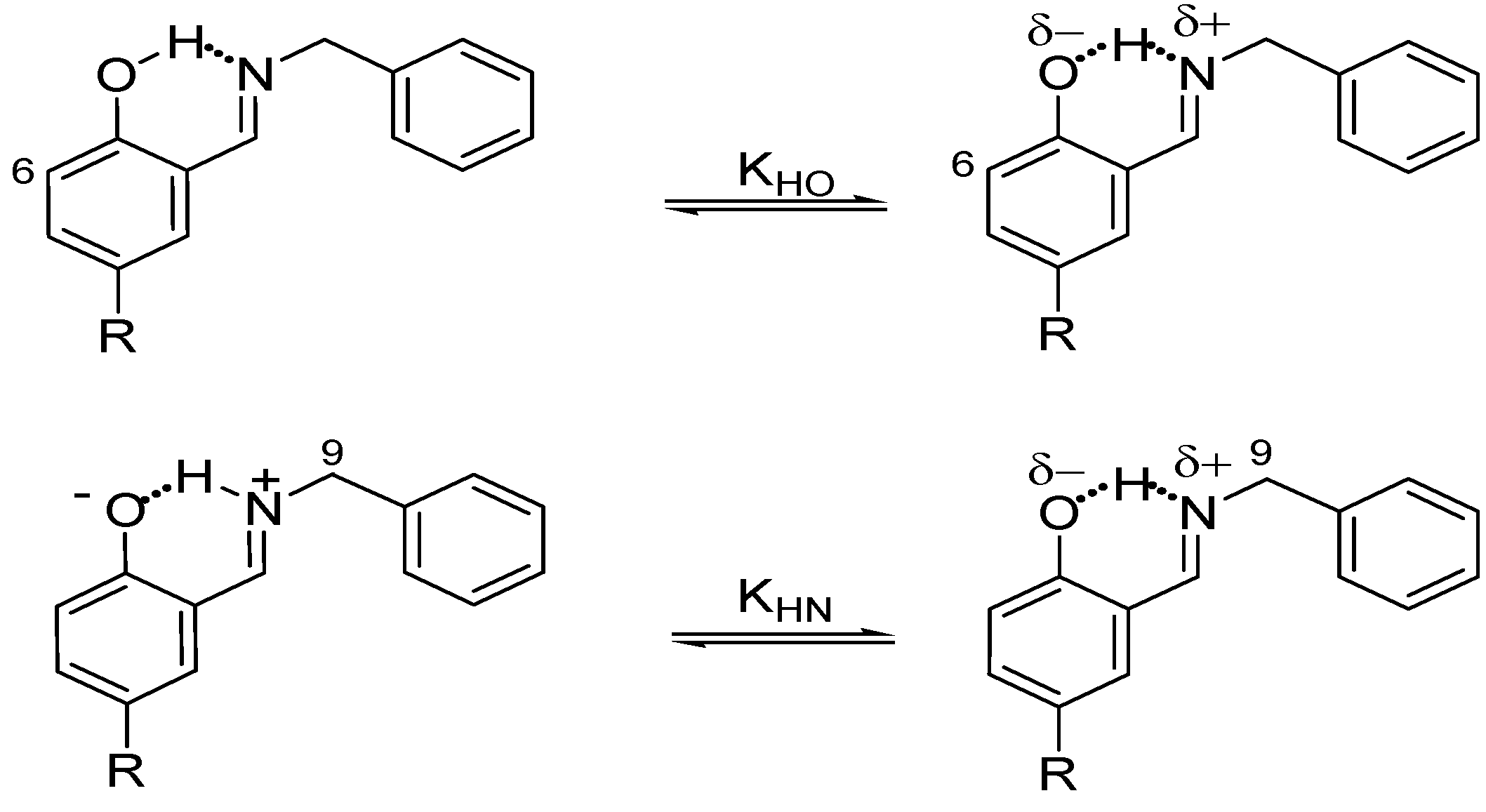
Thus, the ΔKNHO value allows one to establish the position of the NHO equilibrium. Therefore, if the ΔKNHO value is equal to 1 then the system is in equilibrium Nδ+∙∙∙H∙∙∙Oδ− and both ΔpKNHO and ΔΔG° are equal to zero; if the value of ΔKNHO is higher than 1, both ΔpKNHO and ΔΔG° are less than zero and labile hydrogen is predominantly positioned on the N atom, +N–H∙∙∙O; and finally if ΔKNHO is less than 1 then ΔpKNHO and ΔΔG° are greater than zero and therefore the labile hydrogen is predominantly positioned on the O atom, N∙∙∙H–O.
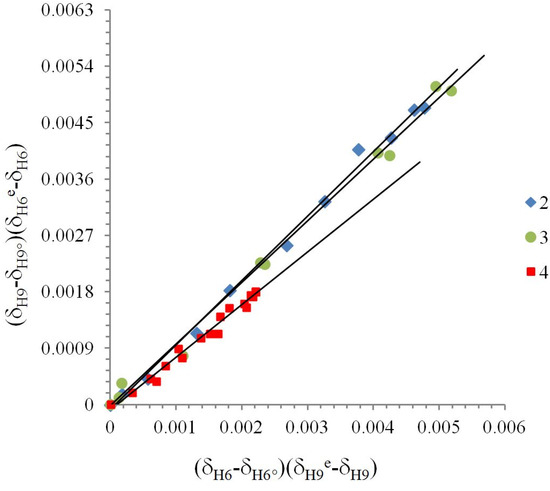
Figure 4.
Linearization of 1H chemical shifts from the δ-diagram for compounds 2, 3 and 4 by Perrin model. (δH9 − δH9°)(δH6e − δH6) = ΔKNHO (δH − δH6°)(δH9e − δH9), ΔKNHO is the slope of the straight line. Compounds 2 (Cl) y = 1.031(±0.002)x + 8*10−5, R = 0.996; 3 (Br) y = 0.986(±0.002)x − 2*10−5, R = 0.994; 4 (H) y = 0.841(±0.005)x − 9*10−5, R = 0.983.
Figure 4.
Linearization of 1H chemical shifts from the δ-diagram for compounds 2, 3 and 4 by Perrin model. (δH9 − δH9°)(δH6e − δH6) = ΔKNHO (δH − δH6°)(δH9e − δH9), ΔKNHO is the slope of the straight line. Compounds 2 (Cl) y = 1.031(±0.002)x + 8*10−5, R = 0.996; 3 (Br) y = 0.986(±0.002)x − 2*10−5, R = 0.994; 4 (H) y = 0.841(±0.005)x − 9*10−5, R = 0.983.
Compounds 2, 3 and 4 were treated with the Perrin model (Figure 4) for the nonlinear behavior in the δ-diagram. In the case of compounds 1, 5 and 6 the Polster-Lachmann analysis seems to be more appropriate, because of the shape of the Gibbs triangle taken in the δ-diagram (Figure 5).
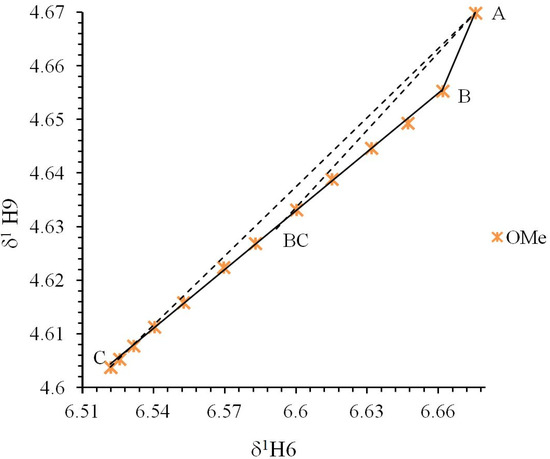
Figure 5.
Polster-Lachmann analysis of the δ-diagram from compound 5. Point “A” shows the initial state, point “B” indicates a change on the compound (neutralization point), point “C” the final state and point “BC” is the experimental ΔKNHO of the system. The dotted line A to C shows the shape of a triangle; the dotted line from vertex A to point BC shows the intercept in an ΔKNHO equilibrium point.
Figure 5.
Polster-Lachmann analysis of the δ-diagram from compound 5. Point “A” shows the initial state, point “B” indicates a change on the compound (neutralization point), point “C” the final state and point “BC” is the experimental ΔKNHO of the system. The dotted line A to C shows the shape of a triangle; the dotted line from vertex A to point BC shows the intercept in an ΔKNHO equilibrium point.
The Polster-Lachmann analysis is based on a ratio of distances established by the Gibbs triangle and the ΔKNHO values are obtained from the chemical shifts of the titration data, hence from points A, B, C and BC in the δ-diagram, in agreement with Equation (6):
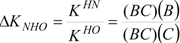
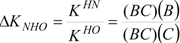
From δ-diagrams, the mechanism occurring in the course of the titration with NaOD, can be proposed (Scheme 4).
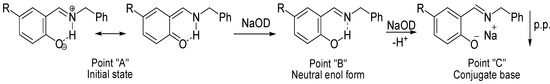
Scheme 4.
Mechanism proposed during the titration with NaOD; the scheme is according to points in the δ-diagram of Figure 5.
Scheme 4.
Mechanism proposed during the titration with NaOD; the scheme is according to points in the δ-diagram of Figure 5.
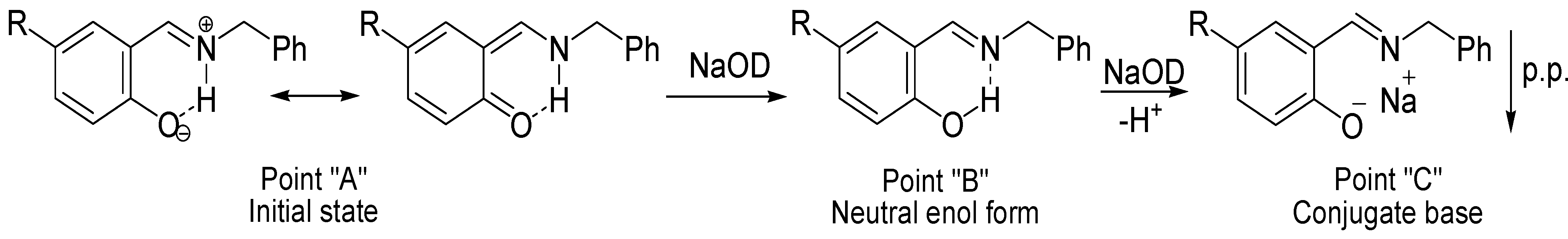
Compounds 1, 2, 5 and 6 begin at an initial state as acidic species (Point “A”), with a small increase of pH, the intramolecular hydrogen bond equilibrium is shifted from +N-H∙∙∙O− to N∙∙∙H-O, reaching the neutralization point of the solution (Point “B”); then, as long as the pH is increased compounds are deprotonated to become into the conjugated bases that precipitate as a salt (Point “C”). In the case of compounds 3 and 4, the initial state is at point “B” with the intramolecular hydrogen bond in the N∙∙∙H-O form, the addition of NaOD aliquots only shift the equilibrium to point “C” the conjugate base.
The δ-diagrams show the initial state in all compounds and indicate the most stable species in a methanol solution, so the stability of the NHO intramolecular hydrogen bond is affected by the electronic nature of the substituent as well as solvation of methanol; therefore, the structure of compounds 1, 5 and 6 with NO2, OMe and OH substituents, respectively, stabilizes and direct the NHO equilibrium position by both mesomeric and inductive effects, although they have different ΔKNHO values, whereas the halogen substituent in compounds 2 and 3 exert both electronegative and inductive effects; none of such effects are present in compound 4. Thus from the obtained ΔKNHO values, the predominant NHO equilibrium in compounds 3 and 4 are the neutral N∙∙∙H–O form, while for the rest of the compounds the zwitterionic +N–H∙∙∙O− form is present (Scheme 2). In the particular case of compound 1, it is as an imine-enamine tautomeric form in agreement with 1H, 13C and 15N pfg-HMQC spectroscopy mentioned above.
Finally, the obtained ΔKNHO values (Table 1) are very close to the equilibrium point Nδ−∙∙∙H+∙∙∙Oδ− (ΔKNHO = 1), which indicate a fast interchange of intramolecular hydrogen bond and the effect produced by both the substituent and the solvent that stabilize the systems in a preferred tautomeric form.
3. Experimental
3.1. General Remarks
Schiff bases 1 to 6 were obtained by condensation of the appropriate aromatic ortho-hydroxyaldehyde with benzylamine in toluene at 25 °C (Scheme 5). Solids products were filtered and dried under a vacuum. Compound 4 was a liquid and the excess of toluene was eliminated under vacuum. 1H and 13C-NMR spectra were recorded in DMSO-d6 on a JEOL ECA-500 spectrometer (1H, 500.1 MHz; 13C, 125.7 MHz; 15N, 50.7 MHz) and the 15N chemical shifts were obtained by correlation of 1H, 15N pfg-HMQC (see Supporting Information).
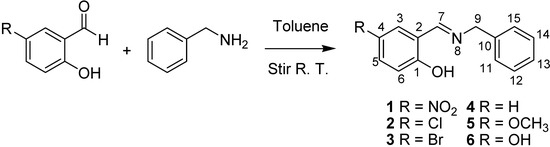
Scheme 5.
Synthesis of Schiff bases.
Scheme 5.
Synthesis of Schiff bases.
(E)-2-((Benzylimino)methyl)-4-nitrophenol (1). Compound 1 was prepared as reported [73] by condensation of 5-nitrosalicylaldehyde (0.5 g, 2.99 mmol) with benzylamine (0.32 g, 0.32 mL, 2.99 mmol) in toluene at room temperature (25 °C) and with a stirring time of 5 min. 1H-NMR (DMSO-d6): δ = 8.42 (d, 4JH,H = 2.9 Hz, 1H, H3), 8.02 (dd, 3JH,H = 9.5, 4JH,H = 3.0 Hz, 1H, H5), 6.65 (d, 3JH,H = 9.5 Hz, 1H, H6), 8.86 (s, 1H, H7), 4.84 (s, 2H, H9), 7.28–7.37 (m, 5H, H11-H15), 14.3 (broad signal, 1H, NHO) ppm. 13C-NMR (DMSO-d6): δ = 175.8 (s, 1C, C1), 115.0 (s, 1C, C2), 132.1 (s, 1C, C3), 136.9 (s, 1C, C4), 129.5 (s, 1C, C5), 122.2 (s, 1C, C6), 167.4 (s, 1C, C7), 57.2 (s, 1C, C9), 135.5 (s, 1C, C10), 129.3 (s, 2C, C11, C15), 128.6 (s, 2C, C12, C14), 128.5 (s, 1C, C13) ppm. 15N-NMR (DMSO-d6): δ = −162.1 (s, 1N, N8), −9.5 (s, 1N, N4) ppm.
(E)-2-((Benzylimino)methyl)-4-chlorophenol (2). Compound 2 was prepared by condensation of 5-chlorosalicylaldehyde (0.5 g, 3.19 mmol) with benzylamine (0.34 g, 0.34 mL, 3.19 mmol) in toluene at room temperature (25 °C) and with a stirring time of 5 min. Yield 0.73 g (93%). m.p. 359–360 K. FT-IR (ATR, cm−1): 1628 (C=N), 1573 (asymmetrical C=C-O-H stretch), 1479 (symmetrical C=C-O-H stretch), 3069 (intramolecular hydrogen bonding N∙∙∙H-O, as a weak broad band). LC-MS-TOF in HPLC-methanol solution, m/z (%) calculated: 246.0686 (100); found: 246.0680 (100) [M+H]+, empirical formula C14H13NOCl. 1H-NMR (DMSO-d6): δ = 7.53 (d, 4JH,H = 2.7 Hz, 1H, H3), 7.31 (dd, 3JH,H=8.7, 4JH,H = 2.7 Hz, 1H, H5), 6.87 (d, 3JH,H = 8.8 Hz, 1H, H6), 8.64 (s, 1H, H7), 4.76 (s, 2H, H9), 7.23–7.34 (m, 5H, H11-H15), 13.4 (broad signal, 1H, NHO) ppm. 13C-NMR (DMSO-d6): δ = 160.0 (s, 1C, C1), 120.2 (s, 1C, C2), 131.1 (s, 1C, C3), 122.5 (s, 1C, C4), 132.5 (s, 1C, C5), 119.0 (s, 1C, C6), 165.8 (s, 1C, C7), 62.4 (s, 1C, C9), 138.8 (s, 1C, C10), 129.1 (s, 2C, C11, C15), 128.3 (s, 2C, C12, C14), 127.7 (s, 1C, C13) ppm. 15N-NMR (DMSO-d6): δ = −80.5 (s, 1N, N8) ppm.
(E)-2-((Benzylimino)methyl)-4-bromophenol (3). Compound 3 was prepared as reported [73] by condensation of 5-bromosalicylaldehyde (0.5 g, 2.48 mmol) with benzylamine (0.26 g, 0.27 mL, 2.48 mmol) in toluene at room temperature of 25 °C and with a stirring time of 5 min. 1H-NMR (DMSO-d6): δ = 7.64 (d, 4JH,H = 2.5 Hz, 1H, H3), 7.41 (dd, 3JH,H = 8.7 and 4JH,H = 2.7 Hz, 1H, H5), 6.82 (d, 3JH,H = 8.7 Hz, 1H, H6), 8.62 (s, 1H, H7), 4.75 (s, 2H, H9), 7.22–7.33 (m, 5H, H11-H15), 13.5 (broad signal, 1H, NHO) ppm. 13C-NMR (DMSO-d6): δ = 160.4 (s, 1C, C1), 120.9 (s, 1C, C2), 134.0 (s, 1C, C3), 109.8 (s, 1C, C4), 135.3 (s, 1C, C5), 119.5 (s, 1C, C6), 165.7 (s, 1C, C7), 62.4 (s, 1C, C9), 138.8 (s, 1C, C10), 129.1 (s, 2C, C11, C15), 128.3 (s, 2C, C12, C14), 127.8 (s, 1C, C13) ppm. 15N-NMR (DMSO-d6): δ = −81.8 (s, 1N, N8) ppm.
(E)-2-((Benzylimino)methyl)phenol (4). Compound 4 was prepared as reported [73] by condensation of salicylaldehyde (0.5 g, 0.42 mL, 4.09 mmol) with benzylamine (0.43 g, 0.44 mL, 4.09 mmol) in toluene at room temperature (25 °C) and with a stirring time of 5 min. 1H-NMR (DMSO-d6): δ = 7.44 (dd, 3JH,H = 7.5, 4JH,H = 1.7 Hz, 1H, H3), 6.87 (td, 3JH,H = 7.4, 3JH,H = 8.3 and 4JH,H = 0.9, 1H, H4), 7.30 (t, d, 3JH,H = 7.4, 3JH,H = 8.2 and 4JH,H = 1.7 Hz, 1H, H5), 6.85 (dd, 3JH,H = 8.2 and 4JH,H = 1.0 Hz, 1H, H6), 8.67 (s, 1H, H7), 4.76 (s, 2H, H9), 7.23–7.34 (m, 5H, H11-H15), 13.4 (s, 1H, NHO) ppm. 13C-NMR (DMSO-d6): δ = 161.2 (s, 1C, C1), 119.2 (s, 1C, C2), 132.3 (s, 1C, C3), 119.1 (s, 1C, C4), 132.9 (s, 1C, C5), 117.0 (s, 1C, C6), 166.8 (s, 1C, C7), 62.7 (s, 1C, C9), 139.0 (s, 1C, C10), 129.0 (s, 2C, C11, C15), 128.2 (s, 2C, C12, C14), 127.6 (s, 1C, C13) ppm. 15N-NMR (DMSO-d6): δ = −81.7 (s, 1N, N8) ppm.
S1-5 (E)-2-((Benzylimino)methyl)-4-methoxyphenol (5). Compound 5 was prepared as reported [73] by condensation of 5-methoxysalicylaldehyde (0.5 g, 0.40 mL, 3.28 mmol) with benzylamine (0.35 g, 0.35 mL, 3.28 mmol) in toluene at room temperature (25 °C) and with a stirring time of 5 min. 1H-NMR (DMSO-d6): δ = 7.04 (d, 4JH,H = 3.1 Hz, 1H, H3), 6.95 (dd, 3JH,H = 9.0 and 4JH,H = 3.1 Hz, 1H, H5), 6.87 (d, 3JH,H = 9.0, 1H, H6), 8.58 (s, 1H, H7), 4.74 (s, 2H, H9), 7.22–7.34 (m, 5H, H11-H15), 3.70 (s, 3H, H16), 12.8 (s, 1H, NHO) ppm. 13C-NMR (DMSO-d6): δ= 155.0 (s, 1C, C1), 119.0 (s, 1C, C2), 115.4 (s, 1C, C3), 152.2 (s, 1C, C4), 119.8 (s, 1C, C5), 117.7 (s, 1C, C6), 166.6 (s, 1C, C7), 62.9 (s, 1C, C9), 139.1 (s, 1C, C10), 129.0 (s, 2C, C11, C15), 128.2 (s, 2C, C12, C14), 127.6 (s, 1C, C13), 55.9 (s, 1C, C16) ppm. 15N-NMR (DMSO-d6): δ = −79.8 (s, 1N, N8) ppm.
(E)-2-((Benzylimino)methyl)benzene-1,4-diol (6). Compound 6 was prepared by condensation of 5-hydroxysalicylaldehyde (0.5 g, 3.62 mmol) with benzylamine (0.38 g, 0.39 mL, 3.62 mmol) in toluene at room temperature of 25 °C and with a stirring time of 5 min. Yield 0.73 g (89%). m.p. 397–399 K. FT-IR (ATR, cm−1): 1641 (C=N), 1601 (asymmetrical C=C-O-H stretch), 1496 (symmetrical C=C-O-H stretch), 3311 (free phenolic O-H medium broad band), 3054 (intramolecular hydrogen bonding N∙∙∙H-O, as a weak broad band). LC-MS-TOF in HPLC-grade methanol, m/z (%) calculated: 228.1025 (100); found: 228.1022 (100) [M+H]+, empirical formula C14H14NO2. 1H-NMR (DMSO-d6): δ= 6.83 (d, 4JH,H = 3.0 Hz, 1H, H3), 6.76 (dd, 3JH,H = 8.8, 4JH,H = 3.0 Hz, 1H, H5), 6.68 (d, 3JH,H = 8.9, 1H, H6), 8.58 (s, 1H, H7), 4.74 (s, 1H, H9), 7.23–7.34 (m, 5H, H11-H15), 9.00 (s, 1H, C4-OH), 12.5 (s, 1H, NHO) ppm. 13C-NMR (DMSO-d6): δ= 153.5 (s, 1C, C1), 119.1 (s, 1C, C2), 117.0 (s, 1C, C3), 149.9 (s, 1C, C4), 120.5 (s, 1C, C5), 117.4 (s, 1C, C6), 166.8 (s, 1C, C7), 62.8 (s, 1C, C9), 139.3 (s, 1C, C10), 129.1 (s, 2C, C11, C15), 128.3 (s, 2C, C12, C14), 127.6 (s, 1C, C13) ppm. 15N-NMR (DMSO-d6): δ= −79.7 (s, 1N, N8) ppm.
3.2. Sample Preparation, Titrant Solution and pH Meter
Solutions of compounds 1–6 (0.06–0.10 M) in CD3OD (0.4–0.5 mL) and 1,4-dioxane as internal reference (0.5–1.5 μL, δH 3.53), were prepared in resonance tubes. The NaOD titrant solution, was prepared to 1.4 and 4.8% (v/v) from NaOD/D2O (40%) in CD3OD, while the DCl solution was prepared to 5% from DCl/D2O (70%) in CD3OD. The glass electrode was filled with a KCl standard solution and calibrated with phosphate buffer pH 7.0 and 4.0.
3.3. NMR Spectrometric Titration
The 1H-NMR spectra were recorded in CD3OD on a JEOL ECA-500 spectrometer at room temperature of 295.15 ± 1 K (22 ± 1 °C). An initial 1H-NMR spectrum of the solutions was recorded and assigned as initial value for the titration. Subsequently the solutions were titrated with aliquots of the NaOD/D2O solution base (3.0 μL), until invariant changes in the chemical shifts were observed; each 1H-NMR spectrum as well as the corresponding pH reading were recorded simultaneously, after the addition of the base. Only compound 2 was further titrated with DCl (5%), to observe the behavior of the system at acidic pH.
3.4. NMR Titration Graphics (Figure 6, Figure 7, Figure 8, Figure 9, Figure 10 and Figure 11)
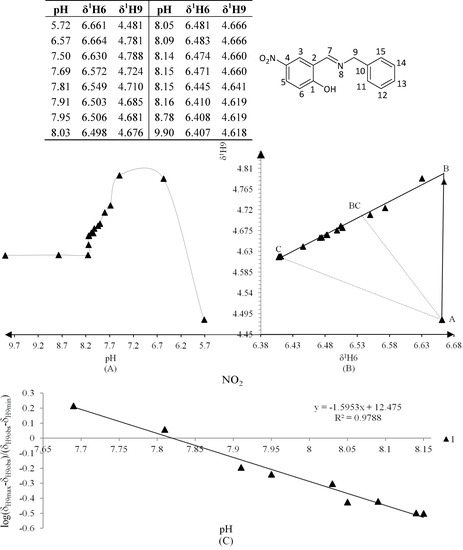
Figure 6.
(A) titration curve (pH vs. δ1H9); (B) δ-diagram with Polster-Lachmann analysis (δ1H6 vs. δ1H9) and (C) plot of the semilogarithmic Henderson-Hasselbalch equation (pH vs. log[(δH9max − δH9obs)/(δH9obs − δH9min)]) of compound 1 (R = NO2).
Figure 6.
(A) titration curve (pH vs. δ1H9); (B) δ-diagram with Polster-Lachmann analysis (δ1H6 vs. δ1H9) and (C) plot of the semilogarithmic Henderson-Hasselbalch equation (pH vs. log[(δH9max − δH9obs)/(δH9obs − δH9min)]) of compound 1 (R = NO2).
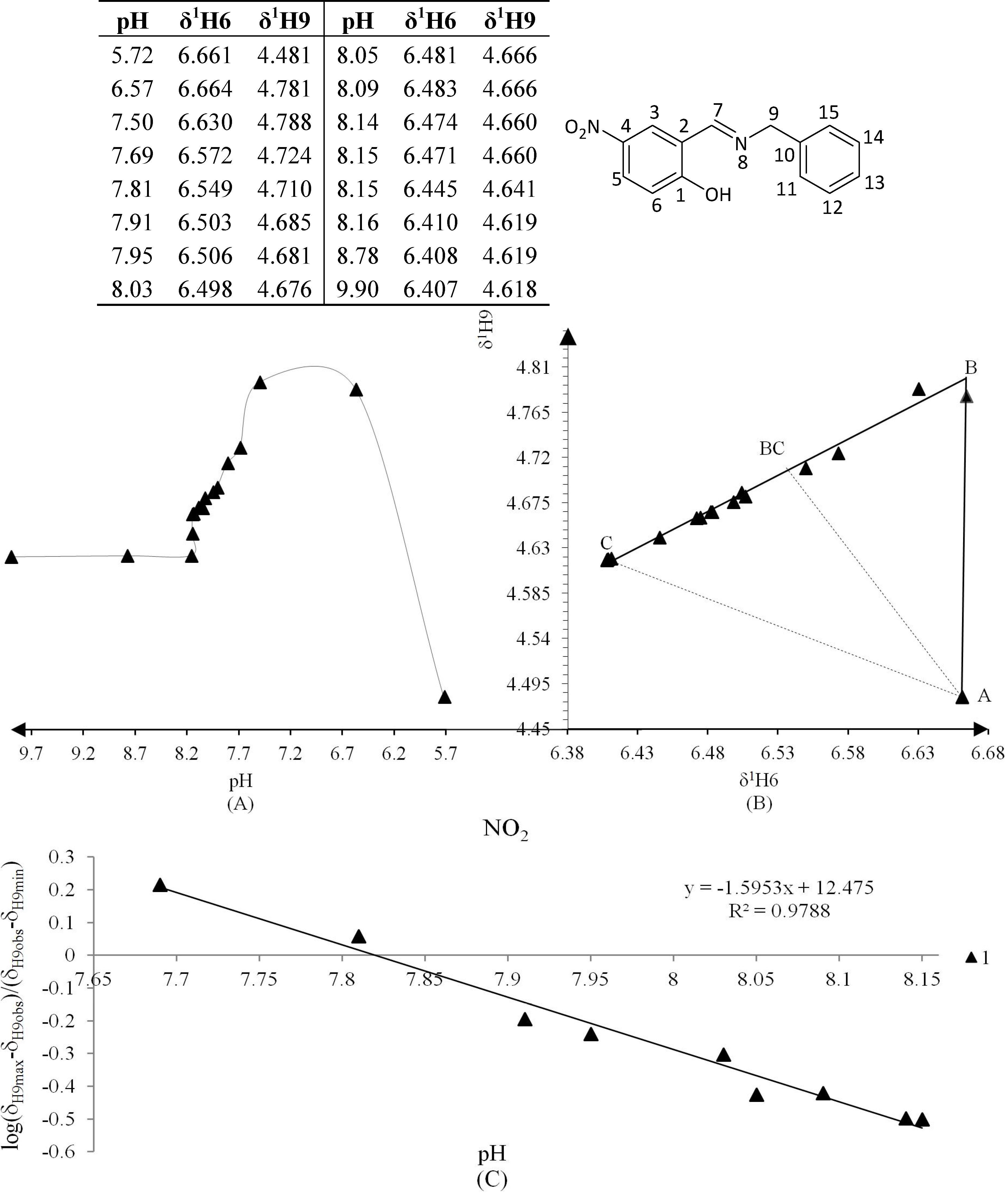
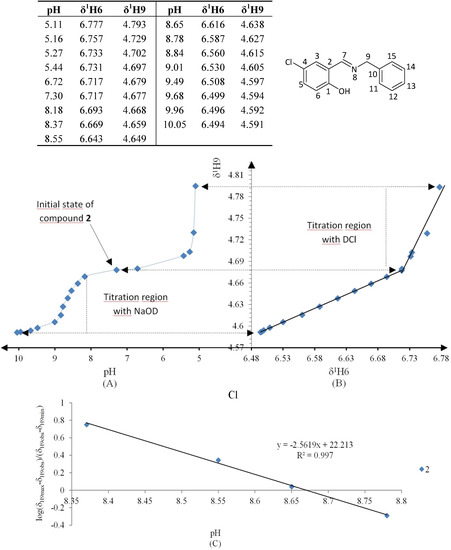
Figure 7.
(A) titration curve (pH vs. δ1H9); (B) δ-diagram with Perrin model analysis (δ1H6 vs. δ1H9) and (C) plot of the semilogarithmic Henderson-Hasselbalch equation (pH vs. log[(δH9max − δH9obs)/(δH9obs − δH9min)]) of compound 2 (R = Cl). Only compound 2 was titrated with DCl solution.
Figure 7.
(A) titration curve (pH vs. δ1H9); (B) δ-diagram with Perrin model analysis (δ1H6 vs. δ1H9) and (C) plot of the semilogarithmic Henderson-Hasselbalch equation (pH vs. log[(δH9max − δH9obs)/(δH9obs − δH9min)]) of compound 2 (R = Cl). Only compound 2 was titrated with DCl solution.
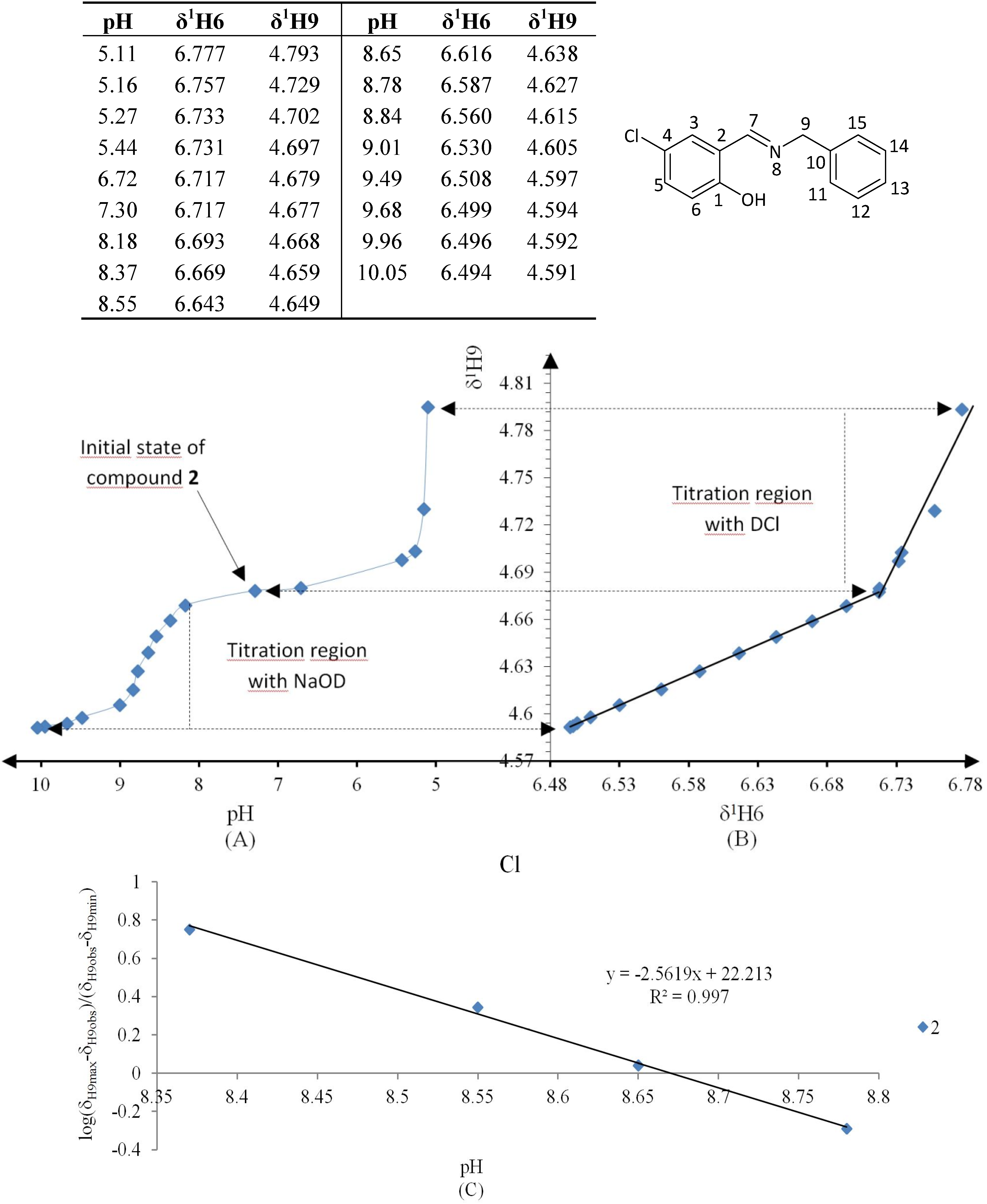
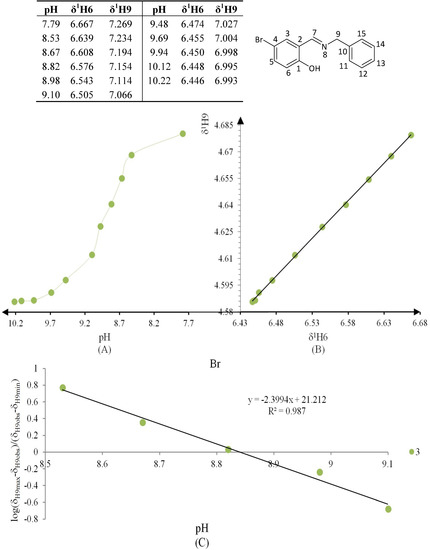
Figure 8.
(A) titration curve (pH vs. δ1H9); (B) δ-diagram with Perrin model analysis (δ1H6 vs. δ1H9) and (C) plot of the semilogarithmic Henderson-Hasselbalch equation (pH vs. log[(δH9max − δH9obs)/(δH9obs − δH9min)]) of compound 3 (R = Br).
Figure 8.
(A) titration curve (pH vs. δ1H9); (B) δ-diagram with Perrin model analysis (δ1H6 vs. δ1H9) and (C) plot of the semilogarithmic Henderson-Hasselbalch equation (pH vs. log[(δH9max − δH9obs)/(δH9obs − δH9min)]) of compound 3 (R = Br).
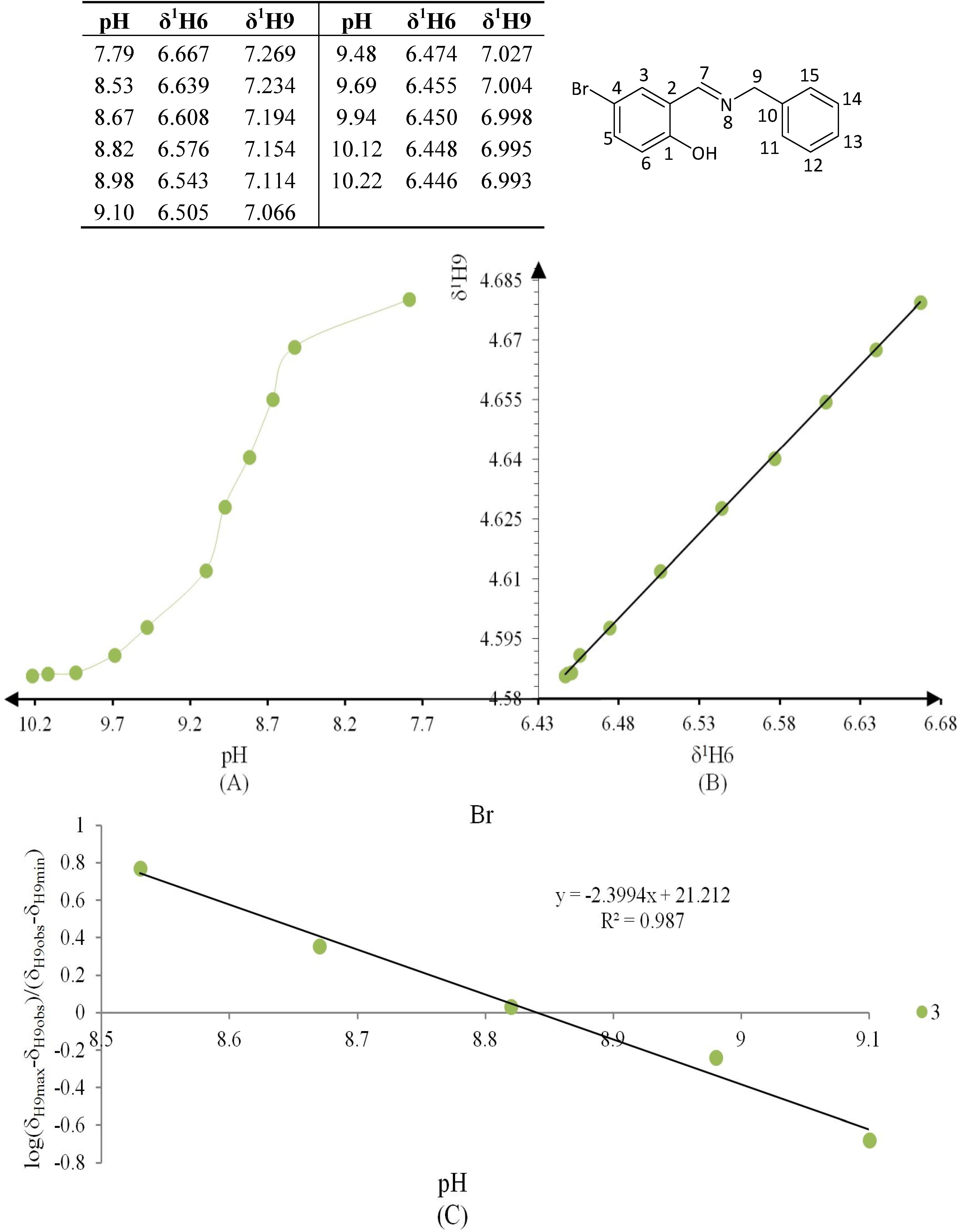
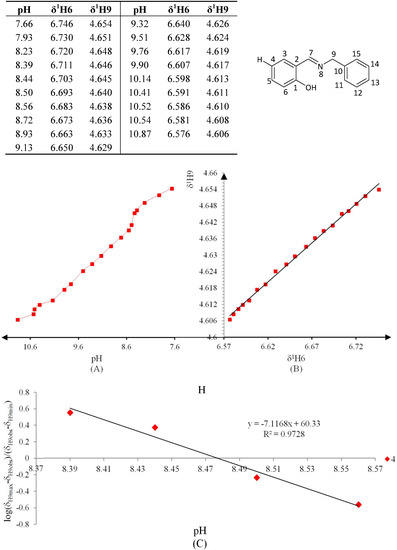
Figure 9.
(A) titration curve (pH vs. δ1H9); (B) δ-diagram with Perrin model analysis (1H6 vs. 1H9) and (C) plot of the semilogarithmic Henderson-Hasselbalch equation (pH vs. log[(δH9max − δH9obs)/(δH9obs − δH9min)])of compound 4 (R = H).
Figure 9.
(A) titration curve (pH vs. δ1H9); (B) δ-diagram with Perrin model analysis (1H6 vs. 1H9) and (C) plot of the semilogarithmic Henderson-Hasselbalch equation (pH vs. log[(δH9max − δH9obs)/(δH9obs − δH9min)])of compound 4 (R = H).
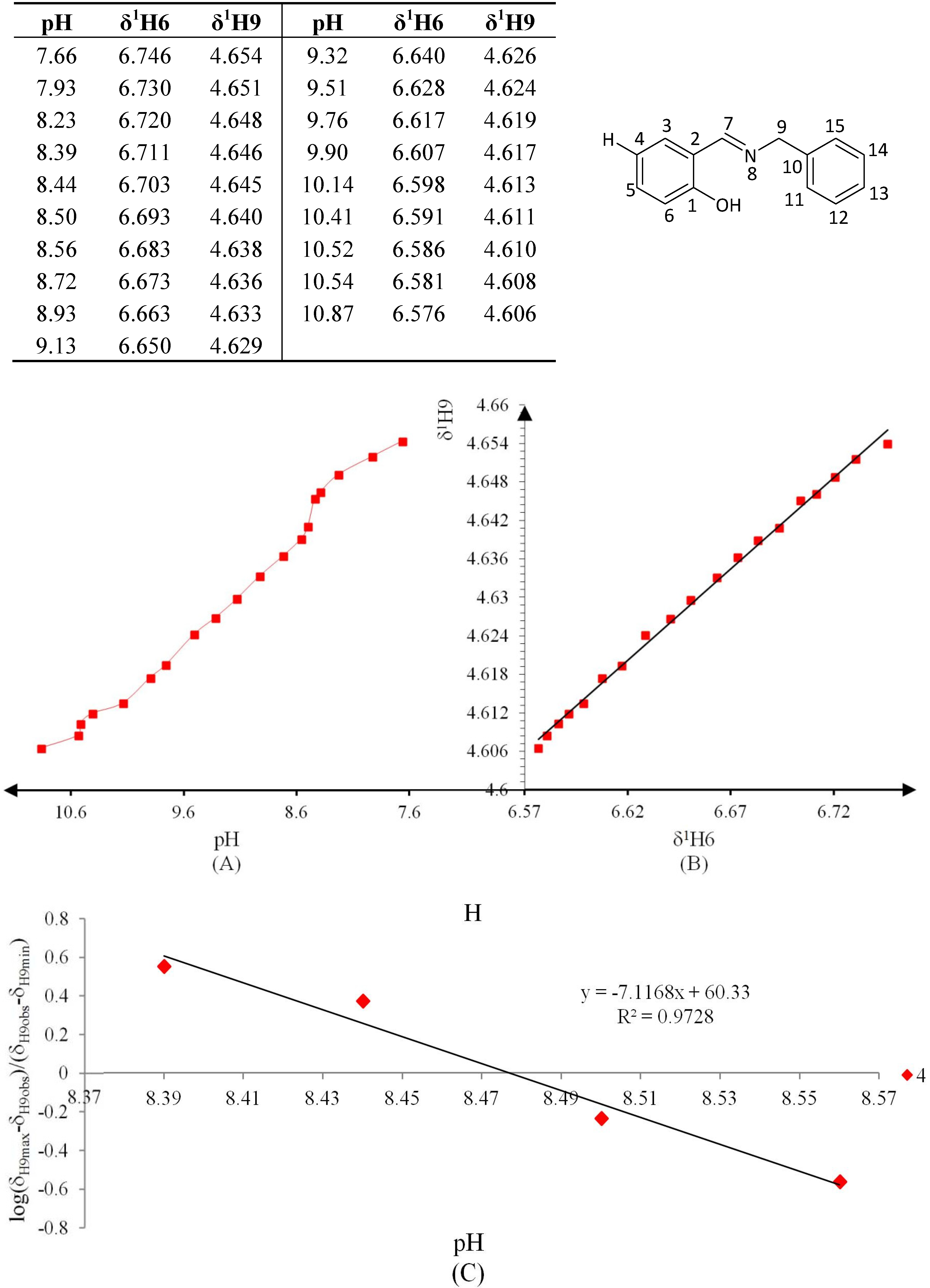
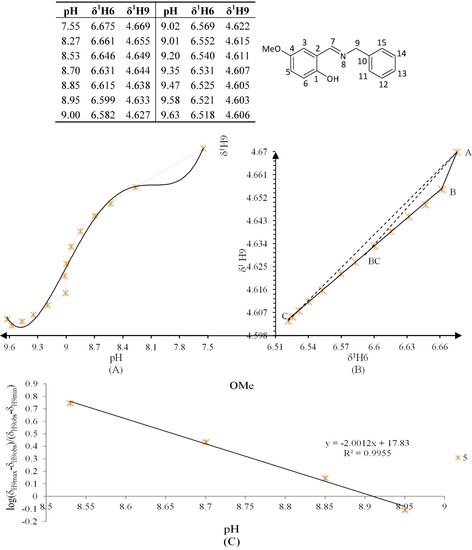
Figure 10.
(A) titration curve (pH vs. δ1H9); (B) δ-diagram with Polster-Lachmann analysis (δ1H6 vs. δ1H9) and (C) plot of the semilogarithmic Henderson-Hasselbalch equation (pH vs. log[(δH9max − δH9obs)/(δH9obs − δH9min)]) of compound 5 (R = OMe).
Figure 10.
(A) titration curve (pH vs. δ1H9); (B) δ-diagram with Polster-Lachmann analysis (δ1H6 vs. δ1H9) and (C) plot of the semilogarithmic Henderson-Hasselbalch equation (pH vs. log[(δH9max − δH9obs)/(δH9obs − δH9min)]) of compound 5 (R = OMe).
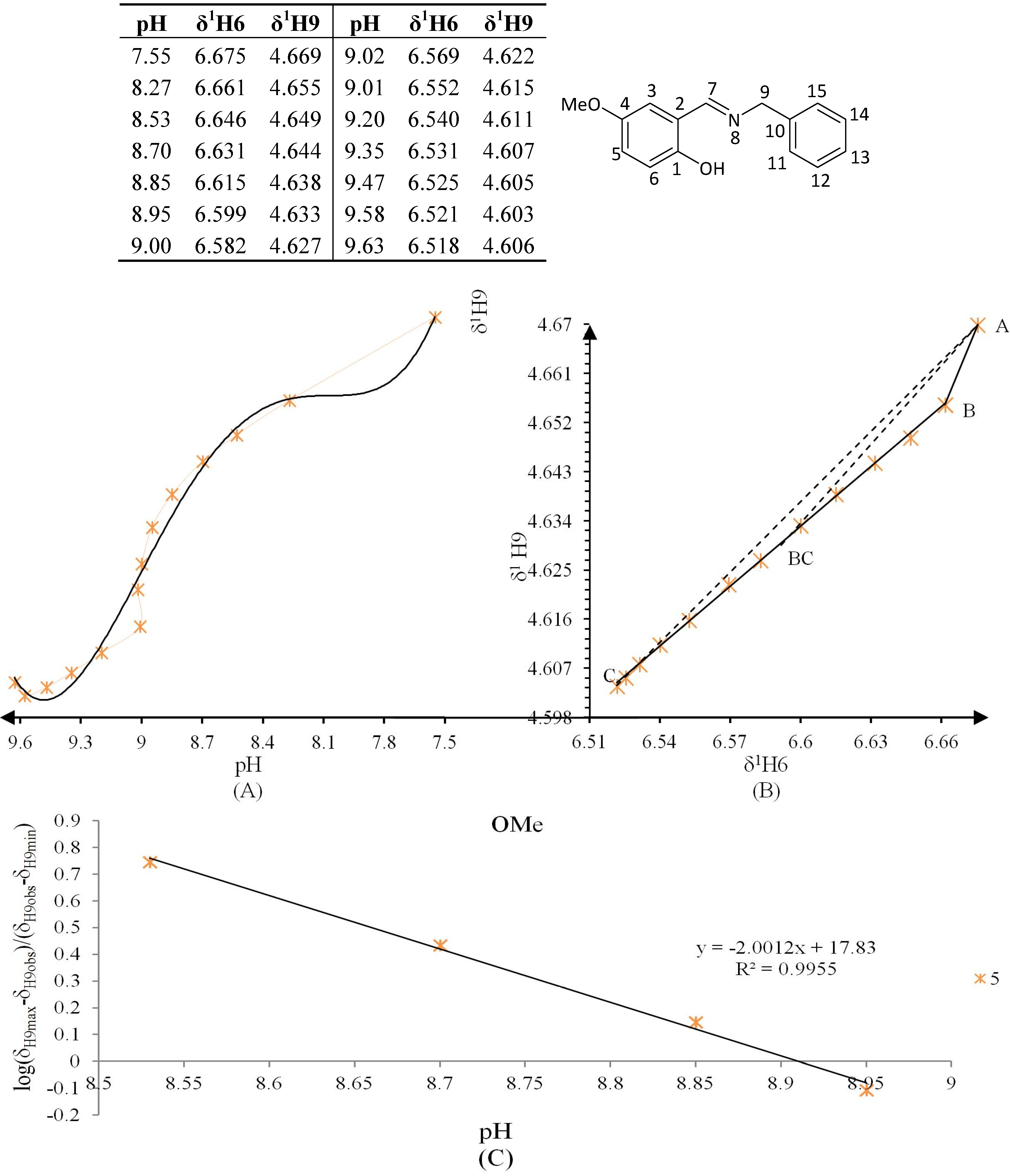
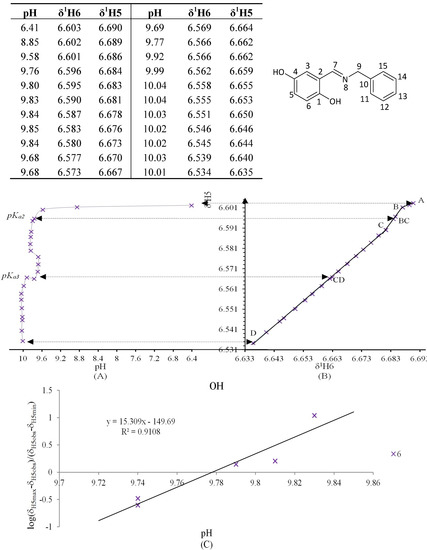
Figure 11.
(A) titration curve (pH vs. δ1H5); (B) δ-diagram with Polster-Lachmann analysis (δ1H6 vs. δ1H5) and (C) plot of the semilogarithmic Henderson-Hasselbalch equation (pH vs. log[(δH5max – δH5obs)/(δH5obs – δH5min)])of compound 6 (R = OH) calculating the pKa2. The pKa2 value belong to deprotonation of intramolecular hydrogen bond N∙∙∙H-O, whereas pKa3 correspond to free phenolic C4-OH. For this compound the plot of δ-diagram was done with 1H6 vs. 1H5 chemical shifts, since only this correlation showed a system with three slope changes similar to polyprotic system.
Figure 11.
(A) titration curve (pH vs. δ1H5); (B) δ-diagram with Polster-Lachmann analysis (δ1H6 vs. δ1H5) and (C) plot of the semilogarithmic Henderson-Hasselbalch equation (pH vs. log[(δH5max – δH5obs)/(δH5obs – δH5min)])of compound 6 (R = OH) calculating the pKa2. The pKa2 value belong to deprotonation of intramolecular hydrogen bond N∙∙∙H-O, whereas pKa3 correspond to free phenolic C4-OH. For this compound the plot of δ-diagram was done with 1H6 vs. 1H5 chemical shifts, since only this correlation showed a system with three slope changes similar to polyprotic system.
3.5. Data Analysis
The calculations were performed on a Microsoft Excel worksheet. The data analysis of the semilogarithmic Henderson-Hasselbalch (Equation (3)) was applied to data that have adapted to this analysis method, using a dependent variable log[(δHmax − δHobs)/(δHobs − δHmin)]; H9 for compounds 1–5 and H5 for compound 6) (HX = H5 or H9) and pH as an independent variable for titration curves to find the pKa values.
Data analysis for the δ-diagram by both Perrin and Polster-Lachmann analysis used H6 and H9 proton chemical shifts for the analysis in δ-diagram, since they are the proton chemical shifts adjacent to NHO intramolecular hydrogen bond and the most affected by deprotonation. Thus in the Perrin Analysis Equation (5) can be written as follows (Equation (7)):
where δH9° and δH6° are the chemical shifts from the species at the beginning of the titration, δH9 and δH6 the chemical shifts observed in the course of the titration, δH9e and δH6e are the chemical shifts from species at the end of the titration. Finally in the Polster-Lachmann analysis, the ratio of distances to calculate the ΔKNHO value is established by the graphic method described by the Gibbs triangle [62,68].
(δH9 − δH9°)(δH6e − δH6) = ∆KNHO(δH6 − δH6°)(δH9e − δH9)
4. Conclusions
The study of compounds 1–6 by NMR titration in methanol solution, confirmed the predominant tautomeric forms in solution, noting that the NHO prototropic equilibrium is dependent of the substituent and the solvent. The pKa values obtained using the Henderson-Hasselbalch analysis showed that all compounds are weak acids. The strength and lability of the NHO intramolecular hydrogen bond are consequently affected by the mesomeric and inductive effects exerted by the substituents. The values of the KNHO equilibrium constant indicate that the equilibrium is slightly shifted to the nitrogen atom when the substituent in the phenyl ring exerts a strong electronic effect, either ED or EW (R = NO2, Cl, OMe and OH), and to the oxygen atom when Br or H in CD3OD solutions. Nevertheless the ΔKNHO values close to the unit, highlight that the proton is in the middle of both basic sites (O−∙∙∙H∙∙∙N+), in contrast to what is found in DMSO-d6 solutions, where NMR data is in agreement with the neutral N∙∙∙H–O tautomer for most of the compounds except for the nitro derivative which is in the zwitterion +N–H∙∙∙O form. Finally, we have demonstrated the simplicity, accuracy and versatility of both the Perrin and Polster-Lachmann analysis applied to the study of intramolecular hydrogen bonds.
Acknowledgments
The authors are grateful for financial support from Conacyt (Grants 204648), FRABA Universidad de Colima (797/12) and PROMEP. ORD is greatfully acknowledged to Conacyt grant number 209273 for PhD formation.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Hodnett, E.M.; Mooney, P.D. Antitumor activities of some Schiff bases. J. Med. Chem. 1970, 13, 786. [Google Scholar] [CrossRef]
- Hodnett, E.M.; Dunn, W.J. Cobalt derivatives of Schiff bases of aliphatic amines as antitumor agents. J. Med. Chem. 1972, 15, 339. [Google Scholar] [CrossRef]
- Huang, S.C. New Schiff Bases of Aminohydroxyguanidine as Inhibitors of Tumor Cells and QSAR Analysis. M.S. Thesis, University of the Southern California, Los Angeles, CA, USA, 2001. [Google Scholar]
- Ren, S. Design, Synthesis, Biological Testing and QSAR Analysis of new Schiff bases of N-Hydroxysemicarbazide as Inhibitors of Tumor Cells. Ph.D. Thesis, University of Southern California, Los Angeles, CA, USA, 2001. [Google Scholar]
- Gacche, R.N.; Gond, D.S.; Dhole, N.A.; Dawane, B.S. Coumarin Schiff-bases: As antioxidant and possibly anti-inflammatory agents. J. Enzym. Inhib. Med. Chem. 2006, 21, 157–161. [Google Scholar] [CrossRef]
- Krause, M.; Rouleau, A.; Stark, H.; Luger, P.; Lipp, R.; Garbarg, M.; Schwartz, J.-C.; Schunack, W. Synthesis, X-ray crystallography, and pharmacokinetics of novel azomethine prodrugs of (r)-α-methylhistamine: Highly potent and selective histamine h3 receptor agonists. J. Med. Chem. 1995, 38, 4070–4079. [Google Scholar] [CrossRef]
- Kaplan, J.P.; Raizon, B.M.; Desarmenien, M.; Feltz, P.; Headley, P.M.; Worms, P.; Lloyd, K.G.; Bartholini, G. New anticonvulsants: Schiff bases of γ-aminobutyric acid and γ-aminobutyramide. J. Med. Chem. 1980, 23, 702–704. [Google Scholar] [CrossRef]
- Koneru, P.B.; Lien, E.J.; Avramis, V.I. Synthesis and testing of new antileukemic Schiff bases of N-hydroxy-N'-aminoguanidine against CCRF-CEM/0 human leukemia cells in vitro and synergism studies with cytarabine (Ara-C). Pharm. Res. 1993, 10, 515–520. [Google Scholar] [CrossRef]
- Gangani, B.J.; Parsania, P.H. Microwave-irradiated and classical syntheses of symmetric double Schiff bases of 1,1'-bis(4-aminophenyl)cyclohexane and their physicochemical characterization. Spectrosc. Lett. 2007, 40, 97–112. [Google Scholar] [CrossRef]
- Parekh, J.; Inamdhar, P.; Nair, R.; Baluja, S.; Chanda, S. Synthesis and antibacterial activity of some Schiff bases derived from 4-aminobenzoic acid. J. Serb. Chem. Soc. 2005, 70, 1155–1161. [Google Scholar] [CrossRef]
- Shi, L.; Fang, R.; Xue, J.; Xiao, Z.; Tan, S.; Zhu, H. Synthesis, characterization, and antibacterial and cytotoxic study of metal complexes with Schiff base ligands. Aust. J. Chem. 2008, 61, 288–296. [Google Scholar] [CrossRef]
- Shi, L.; Ge, H.M.; Tan, S.H.; Li, H.Q.; Song, Y.C.; Zhu, H.L.; Tan, R.X. Synthesis and antimicrobial activities of Schiff bases derived from 5-chloro-salicylaldehyde. Eur. J. Med. Chem. 2007, 42, 558–564. [Google Scholar] [CrossRef]
- Venugopala, K.N.; Jayashree, B.S. Microwave-induced synthesis of Schiff bases of aminothiazolyl bromocoumarins as antibacterials. Indian J. Pharm. Sci. 2008, 70, 88–91. [Google Scholar] [CrossRef]
- Zheng, B.; Brett, S.J.; Tite, J.P.; Lifely, M.R.; Brodie, T.A.; Rhodes, J. Galactose oxidation in the design of immunogenic vaccines. Science 1992, 256, 1560–1563. [Google Scholar]
- Vazzana, I.; Terranova, E.; Mattioli, F.; Sparatore, F. Aromatic Schiff bases and 2,3-disubstituted-1,3-thiazolidin-4-one derivatives as antiinflammatory agents. Arkivoc 2004, 364–374. [Google Scholar]
- Sharif, S.; Powell, D.R.; Schagen, D.; Steiner, T.; Toney, M.D.; Fogle, E.; Limbach, H.H. X-ray crystallographic structures of enamine and amine Schiff bases of pyridoxal and its 1:1 hydrogen-bonded complexes with benzoic acid derivatives: Evidence for coupled inter- and intramolecular proton transfer. Acta Crystallogr. 2006, B62, 480–487. [Google Scholar]
- Golubev, N.S.; Smirnov, S.N.; Tolstoy, P.M.; Sharif, S.; Toney, M.D.; Denisov, G.S.; Limbach, H.H. Observation by NMR of the tautomerism of an intramolecular OHOHN-charge relay chain in a model Schiff base. J. Mol. Struct. 2007, 319–327. [Google Scholar]
- Sharif, S.; Denisov, G.S.; Toney, M.D.; Limbach, H.H. NMR studies of coupled low- and high-barrier hydrogen bonds in pyridoxal-5'-phosphate model systems in polar solution. J. Am. Chem. Soc. 2007, 129, 6313–6327. [Google Scholar] [CrossRef]
- Sharif, S.; Huot, M.C.; Tolstoy, P.M.; Toney, M.D.; Jonsson, K.H.M.; Limbach, H.H. 15N nuclear magnetic resonance studies of acid-base properties of pyridoxal-5'-phosphate aldimines in aqueous solution. J. Phys. Chem. B 2007, 111, 3869–3876. [Google Scholar] [CrossRef]
- Lin, Y.L.; Gao, J. Internal proton transfer in the external pyridoxal 5'-phosphate Schiff base in dopa decarboxylase. Biochemistry 2010, 49, 84–94. [Google Scholar] [CrossRef]
- Day, J.H. Thermochromism. Chem. Rev. 1962, 63, 65–80. [Google Scholar] [CrossRef]
- Minkin, V.I. Photo-, Thermo-, Solvato-, and Electrochromic spiroheterocyclic compounds. Chem. Rev. 2004, 104, 2751–2776. [Google Scholar] [CrossRef]
- Hoshino, N.; Inabe, T.; Mitani, T.; Maruyama, Y. Structure and optical properties of a thermochromic Schiff base. Thermally induced intramolecular proton transfer in the N,N'-bis(salicylidene)-p-phenylenediamine crystals. Bull. Chem. Soc. Jpn. 1988, 61, 4207–4214. [Google Scholar]
- Takeda, S.; Chihara, H.; Inabe, T.; Mitani, T.; Maruyama, Y. NMR study of proton dynamics in the NHO hydrogen in the thermochromic crystals of N-salicylideneanilines. Chem. Phys. Lett. 1992, 189, 13–17. [Google Scholar] [CrossRef]
- Lambi, E.; Gegiou, D.; Hadjoudis, E. Thermochromism and photochromism of N-salicylidene-benzylamines and N-salicylidene-2-aminomethylpyridine. J. Photochem. Photobiol. A Chem. 1995, 86, 241–246. [Google Scholar] [CrossRef]
- Hadjoudis, E.; Rontoyianni, A.; Ambroziak, K.; Dziembowska, T.; Mavridis, I.M. Photochromism and thermochromism of solid trans-N.,N'-bis-(salicylidene)-1,2-cyclohexane-diamines and trans-N.,N'-bis-(2-hydroxynaphylidene)-1,2-cyclohexanediamine. J. Photochem. Photobiol. A Chem. 2004, 162, 521–530. [Google Scholar] [CrossRef]
- Amimoto, K.; Kawato, T. Photochromism of organic compounds in the crystal state. J. Photochem. Photobiol. C 2005, 6, 207–226. [Google Scholar] [CrossRef]
- Akitsu, T.; Einaga, Y. A chiral photochromic Schiff base: (R)-4-Bromo-2-[(1-phenylethyl)-iminomethyl]phenol. Acta Crystallogr. 2006, E62, o4315–o4317. [Google Scholar]
- Bolz, I.; May, C.; Spange, S. Solvatochromic properties of Schiff bases derived from 5-amino-barbituric acid: Chromophores with hydrogen bonding patterns as components for coupled structures. New J. Chem. 2007, 31, 1568–1571. [Google Scholar] [CrossRef]
- Gegiou, D.; Lambi, E.; Hadjoudis, E. Solvatochromism in N-(2-Hydroxybenzylidene)aniline, N-(2-Hydroxybenzylidene)benzylamine, and N-(2-Hydroxybenzylidene)-2-phenylethylamine. J. Phys. Chem. 1996, 100, 17762–17765. [Google Scholar] [CrossRef]
- Raczynska, E.D.; Kosinska, W.; Osmiałowski, B.; Gawinecki, R. Tautomeric equilibria in relation to pi-electron delocalization. Chem. Rev. 2005, 105, 3561–3612. [Google Scholar] [CrossRef]
- Schilf, W. Intramolecular hydrogen bond investigations in some Schiff bases using C-C and N-C coupling constants. J. Mol. Struct. 2004, 689, 245–249. [Google Scholar] [CrossRef]
- Schilf, W.; Kamienski, B.; Szady-Chelmieniecka, A.; Grech, E.; Makal, A.; Wozniak, K. NMR and X-ray studies of 2,6-bis(alkylimino)phenol Schiff bases. J. Mol. Struct. 2007, 94–101. [Google Scholar]
- Schilf, W.; Cmoch, P.; Szady-Chelmieniecka, A.; Grech, E. Deprotonation of hydrogen bonded Schiff bases by three strong nitrogen bases. J. Mol. Struct. 2009, 921, 34–37. [Google Scholar] [CrossRef]
- Claramunt, R.M.; López, C.; Santa María, M.D.; Sanz, D.; Elguero, J. The use of NMR spectroscopy to study tautomerism. Prog. Nucl. Magn. Reson. Spectrosc. 2006, 49, 169–206. [Google Scholar] [CrossRef]
- Dziembowska, T.; Ambroziak, K.; Majerz, I. Analysis of the vibrational spectra of trans-N.,N'-bis-salicylidene-1',2'-cyclohexanediamine tautomers. J. Mol. Struct. 2005, 738, 15–24. [Google Scholar] [CrossRef]
- Filarowski, A. Intramolecular hydrogen bonding in o-hydroxyaryl Schiff bases. J. Phys. Org. Chem. 2005, 18, 686–698. [Google Scholar] [CrossRef]
- Filarowski, A.; Koll, A.; Rospenk, M.; Krol-Starzomska, I.; Hansen, P.E. Tautomerism of sterically hindered Schiff bases. Deuterium isotope effects on 13C chemical shifts. J. Phys. Chem. A 2005, 109, 4464–4473. [Google Scholar] [CrossRef]
- Raczynska, E.D.; Krygowski, T.M.; Zachara, J.E.; Osmiałowski, B.; Gawinecki, R. Tautomeric equilibria, H-bonding and π-electron delocalization in o-nitrosophenol. A B3LYP/6–311+G(2df,2p) study. J. Phys. Org. Chem. 2005, 18, 892–897. [Google Scholar] [CrossRef]
- Osmiałowski, B.; Raczynska, E.D.; Krygowski, T.M. Tautomeric equilibria and pi electron delocalization for some monohydroxyarenes-quantum chemical studies. J. Org. Chem. 2006, 71, 3727–3736. [Google Scholar] [CrossRef]
- Filarowski, A.; Kochel, A.; Kluba, M.; Kamounah, F.S. Structural and aromatic aspects of tautomeric equilibrium in hydroxyaryl Schiff bases. J. Phys. Org. Chem. 2008, 21, 939–944. [Google Scholar] [CrossRef]
- Kluba, M.; Lipkowski, P.; Filarowski, A. Theoretical investigation of tautomeric equilibrium in ortho-hydroxy phenyl Schiff bases. Chem. Phys. Lett. 2008, 463, 426–430. [Google Scholar] [CrossRef]
- Salman, S.R.; Lindon, J.C.; Farrant, R.D.; Carpenter, T.A. Tautomerism in 2-hydroxy-1-naphthaldehyde Schiff bases in solution and the solid state investigated using 13C-NMR spectroscopy. Magn. Reson. Chem. 1993, 31, 991–994. [Google Scholar] [CrossRef]
- Salman, S.R.; Kamounah, F.S. Tautomerism in 1-hydroxy-2-naphthaldehyde Schiff bases: Calculation of tautomeric isomers using carbon-13 NMR. Spectrosc.-Int. J. 2003, 17, 747–752. [Google Scholar] [CrossRef]
- Santos-Contreras, R.J.; Ramos-Organillo, A.; Garcia-Baez, E.V.; Padilla-Martinez, I.I.; Martinez-Martinez, F.J. The zwitterion of 4-nitro-2-{(E)-[2-(piperidin-1-yl)ethyl]iminomethyl}phenol. Acta Crystallogr. 2009, C65, o8–o10. [Google Scholar]
- Pyta, K.; Przybylski, P.; Schilf, W.; Kolodziej, B.; Szady-Chelmieniecka, A.; Grech, E.; Brzezinski, B. Spectroscopic and theoretical studies of the protonation of N-(5-nitrosalicylidene)-ethylamine. J. Mol. Struct. 2010, 967, 140–146. [Google Scholar] [CrossRef]
- Filarowski, A.; Glowiaka, T.; Koll, A. Strengthening of the intramolecular O…H…N hydrogen bonds in Schiff bases as a result of steric repulsion. J. Mol. Struct. 1999, 484, 75–89. [Google Scholar] [CrossRef]
- Przybylski, P.; Schroeder, G.; Brzezinski, B.; Bartl, F. 1H-NMR, FT-IR and MS studies and PM5 semiempirical calculations of complexes between the Schiff base of gossypol with 2-(aminomethyl)-15-crown-5 and Ca2+, Pb2+ and Ba2+ cations. J. Phys. Org. Chem. 2003, 16, 289–297. [Google Scholar] [CrossRef]
- Dziembowska, T.; Rozwadowski, Z.; Filarowski, A.; Hansen, P.E. NMR study of proton transfer equilibrium in Schiff bases derived from 2-hydroxy-1-naphthaldehyde and 1-hydroxy-2-acetonaphthone. Deuterium isotope effects on 13C and 15N chemical shifts. Magn. Reson. Chem. 2001, 39, S67–S80. [Google Scholar] [CrossRef]
- Sharif, S.; Denisov, G.S.; Toney, M.D.; Limbach, H.H. NMR studies of solvent-assisted proton transfer in a biologically relevant Schiff base: Toward a distinction of geometric and equilibrium H-bond isotope effects. J. Am. Chem. Soc. 2006, 128, 3375–3387. [Google Scholar] [CrossRef]
- Rozwadowski, Z.; Nowak-Wydra, B. Chiral recognition of Schiff bases by 15N-NMR spectroscopy in the presence of a dirhodium complex. Deuterium isotope effect on 15N chemical shift of the optically active Schiff bases and their dirhodium tetracarboxylate adducts. Magn. Reson. Chem. 2008, 46, 974–978. [Google Scholar] [CrossRef]
- Kamounah, F.S.; Salman, S.R.; Mahmoud, A.A.K. Substitution and solvent effect of some substituted hydroxy schiff bases. Spectrosc. Lett. 1998, 31, 1557–1567. [Google Scholar] [CrossRef]
- Irle, S.; Krygowski, T.M.; Niu, J.E.; Schwarz, W.H.E. Substituent effects of -NO and -NO2 groups in aromatic systems. J. Org. Chem. 1995, 60, 6744–6755. [Google Scholar]
- Krygowski, T.M.; Stepien, B.T. Sigma- and pi-electron delocalization: Focus on substituent effects. Chem. Rev. 2005, 105, 3482–3512. [Google Scholar] [CrossRef]
- Otto, E.; Stanislav, B. Substituent effects of the alkyl groups: Polarity vs. Polarizability. Eur. J. Org. Chem. 2007, 2007, 2870–2876. [Google Scholar]
- Ozeryanskii, V.A.; Pozharskii, A.F.; Schilf, W.; Kamieński, B.; Sawka-Dobrowolska, W.; Sobczyk, L.; Grech, E. Novel polyfunctional tautomeric systems containing salicylideneamino and proton sponge moieties. Eur. J. Org. Chem. 2006, 2006, 782–790. [Google Scholar] [CrossRef]
- Gilli, G.; Bellucci, F.; Ferretti, V.; Bertolasi, V. Evidence for resonance-assisted hydrogen bonding from crystal-structure correlations on the enol form of the β-diketone fragment. J. Am. Chem. Soc. 1989, 111, 1023–1028. [Google Scholar]
- Gilli, P.; Bertolasi, V.; Ferretti, V.; Gilli, G. Towards an unified hydrogen-bond theory. J. Am. Chem. Soc. 2000, 122, 10405–10417. [Google Scholar] [CrossRef]
- Gilli, P.; Bertolasi, V.; Pretto, L.; Antonov, L.; Gilli, G. Variable-temperature X-ray crystallographic and DFT computational study of the N-H···O/N···H-O tautomeric competition in 1-(Arylazo)-2-naphthols. Outline of a transiton-state hydrogen-bond theory. J. Am. Chem. Soc. 2005, 127, 4943–4953. [Google Scholar] [CrossRef]
- Dominiak, P.M.; Grech, E.; Barr, G.; Teat, S.; Mallinson, P.; Wozniak, K. Neutral and ionic hydrogen bonding in Schiff bases. Chem. Eur. J. 2003, 9, 963–970. [Google Scholar] [CrossRef]
- Krygowski, T.M.; Wozniak, K.; Anulewicz, R.; Pawlak, D.; Kolodziejski, W.; Grech, E.; Szady, A. Through-resonance assisted ionic hydrogen bonding in 5-Nitro-N-salicylideneethylamine. J. Phys. Chem. A 1997, 101, 9399–9404. [Google Scholar] [CrossRef]
- Polster, J.; Lachmann, H. Spectrometric Titration: Analysis of Chemical Equilibria; VCH Verlagsgesellschaft: Weinheim, Germany, 1989; pp. 33–132. [Google Scholar]
- Macomber, R.S. An introduction to NMR titration for studying rapid reversible complexation. J. Chem. Educ. 1992, 69, 375. [Google Scholar] [CrossRef]
- Breitmaier, E.; Spohn, K.-H. PH-abhängigkeit der 13C-chemischen verschiebungen sechsgliedriger stickstoff-heteroaromaten. Tetrahedron 1973, 29, 1145. [Google Scholar] [CrossRef]
- Breitmaier, E.; Voelter, W. Carbon-13 NMR Spectroscopy: High Resolution Methods and Applications in Organic Chemistry and Biochemistry, 3rd ed.; VCH Verlagsgesellschaft GbBH: New York, NY, USA, 1990; pp. 107–123. [Google Scholar]
- Szakács, Z.; Hägele, G. Accurate determination of low pK values by 1H-NMR titration. Talanta 2004, 62, 819–825. [Google Scholar] [CrossRef]
- Berger, S. A 13C magnetic resonance study. Tetrahedron 1977, 33, 1587–1589. [Google Scholar]
- Gobber, F.; Polster, J. Determination of relative pK values of dibasic protolytes by regression analysis of absorbance diagrams. Anal. Chem. 1976, 48, 1546–1550. [Google Scholar] [CrossRef]
- Perrin, C.L.; Thoburn, J.D. Evidence for a double-minimum potential for intramolecular hydrogen bonds of aqueous hydrogen maleate and hydrogen phthalate anions. J. Am. Chem. Soc. 1989, 111, 8010–8012. [Google Scholar] [CrossRef]
- Miles, A.F.; Perrin, C.L.; Sinnott, M.L. Absence of reverse anomeric effect: Conformational analysis of glucosylimidazolium and glucosylimidazole. J. Am. Chem. Soc. 1994, 116, 8398–8399. [Google Scholar] [CrossRef]
- Perrin, C.L.; Fabian, M.A.; Armstrong, K.B. Solvation effect on steric bulk of ionic substituents: Imidazolium vs. imidazole. J. Org. Chem. 1994, 59, 5246–5253. [Google Scholar] [CrossRef]
- Perrin, C.L.; Fabian, M.A. Multicomponent NMR titration for simultaneous measurement of relative pKas. Anal. Chem. 1996, 68, 2127–2134. [Google Scholar] [CrossRef]
- Makal, A.; Schilf, W.; Kamienski, B.; Szady-Chelmieniecka, A.; Grech, E.; Wozniak, K. Hydrogen bonding in Schiff bases-NMR, structural and experimental charge density studies. Dalton Trans. 2011, 40, 421–430. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1–6 are available from the authors.
© 2013 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
