Chemical Composition and Biological Activity of the Essential Oil from Leaves of Moringa oleifera Lam. Cultivated in Mozambique
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemical Composition of the Essential Oils
| Component | Ri a | Ri b | Identification c | % |
|---|---|---|---|---|
| Oxygenated monoterpenes | ||||
| Linalool | 1099 | 1553 | 1,2,3 | t |
| α-Terpineol | 1189 | 1706 | 1,2,3 | t |
| Phenolic compounds | ||||
| p-Vinylguaiacol | 1311 | 1937 | 1,2 | t |
| Oxygenated sesquiterpenes | 0.7 | |||
| cis-Dihydroagarofuran | 1518 | 1,2 | 0.1 | |
| Eudesm-11-en-4-α,6α-diol | 1807 | 1,2 | 0.6 | |
| Hydrocarbons | 91.1 | |||
| 1-Octadecene | 1783 | 1,2 | 0.3 | |
| Octadecane | 1800 | 1,2,3 | 0.1 | |
| 5-Octadecin | 1844 | 1,2 | 0.3 | |
| n-Hexadecanol | 1889 | 1,2 | 0.1 | |
| Nonadecane | 1896 | 1,2,3 | 0.8 | |
| 1-Eicosene | 1990 | 1,2 | 0.3 | |
| Eicosane | 1998 | 1,2,3 | 1.2 | |
| n-Octadecanol | 2091 | 1,2 | 0.2 | |
| Heneicosane | 2100 | 1,2,3 | 1.9 | |
| Cyclopentadecanol | 2119 | 1,2 | 0.4 | |
| 1-Docosene | 2191 | 1,2 | 0.4 | |
| Docosane | 2200 | 1,2,3 | 6.8 | |
| cis-9-Eicosen-1-ol | 2224 | 1,2 | 0.3 | |
| Tricosane | 2297 | 1,2,3 | 8.1 | |
| Tetracosane | 2405 | 2400 | 1,2,3 | 9.7 |
| Pentacosane | 2499 | 2500 | 1,2,3 | 13.3 |
| Hexacosane | 2601 | 2600 | 1,2,3 | 13.9 |
| Heptacosane | 2698 | 2700 | 1,2,3 | 11.4 |
| Octacosane | 2821 | 2800 | 1,2,3 | 10.0 |
| Nonacosane | 2930 | 2900 | 1,2,3 | 10.5 |
| Triacontane | 3008 | 3000 | 1,2,3 | 1.1 |
| Others | 0.5 | |||
| Hexenyl propanoate | 1101 | 1,2 | t | |
| Phenylethyl alcohol | 1110 | 1,2 | t | |
| Pseudo Phytol | 2016 | 1,2 | 0.5 | |
| Total identified | 92.3 | |||
2.2. UPLC-DAD Analyses
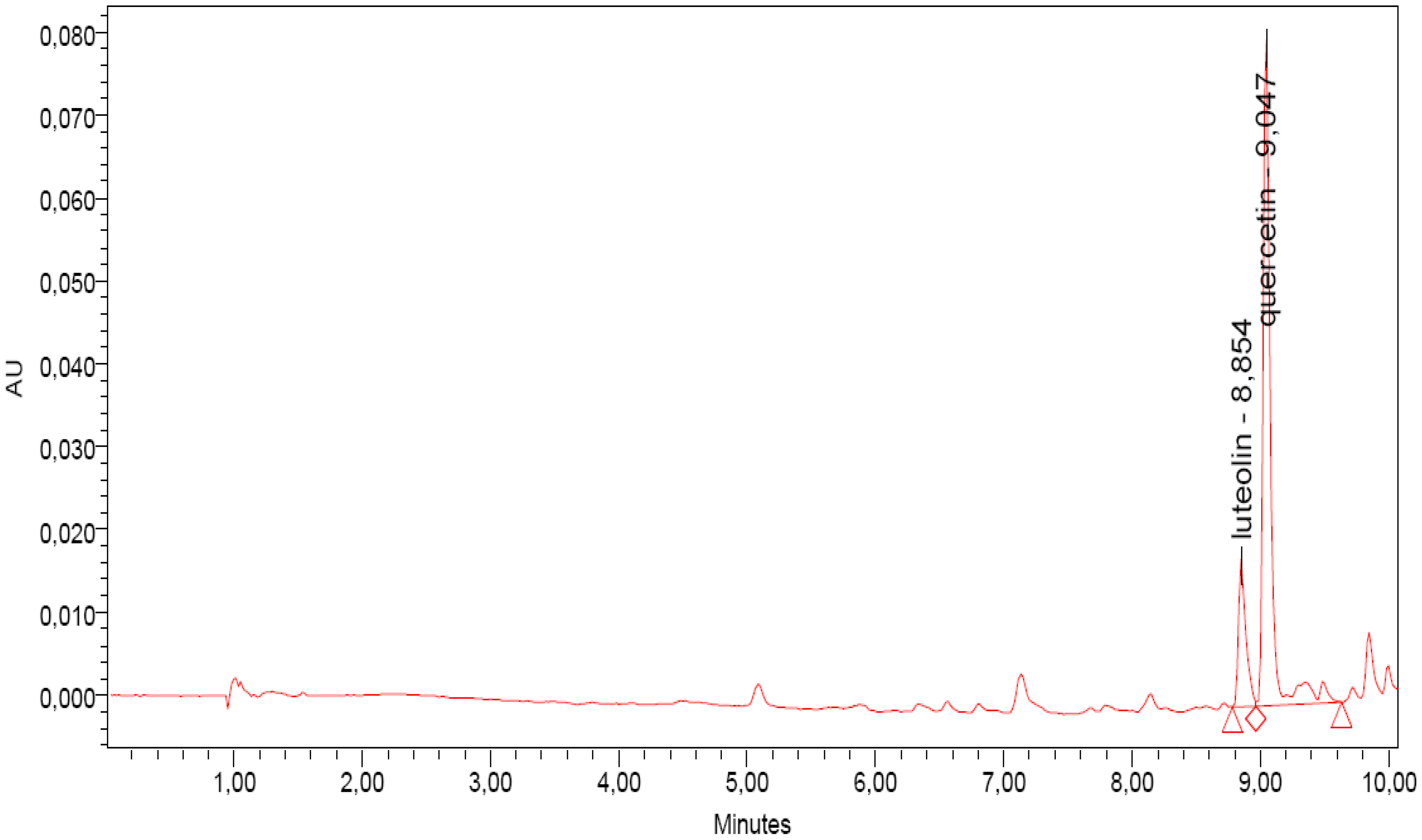
2.3. Antioxidant Activity
2.4. Antibacterial and Antifungal Activity
| Organism | Doses | ||
|---|---|---|---|
| 2 µg | 5 µg | 10 µg | |
| Bacteria | |||
| Gram-positive strains | |||
| Bacillus cereus | 5.7 ± 1.2 ** | 9 ± 1.7 *** | 10.7 ± 1.2 *** |
| Sthaphylococcus aureus | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| Gram-negative strains | |||
| Escherichia coli | 0.0 ± 0.0 | 0.0 ± 0.0 | 6.7 ± 0.6 *** |
| Pseudomonas aeruginosa | 0.0 ± 0.0 | 5.0 ± 0.0 *** | 8.0 ± 1.3 *** |
| Fungal strains | |||
| Penicillium aurantiogriseum | 0.0 ± 0.0 | 4.7 ± 0.6 *** | 9.3 ± 0.6 *** |
| Penicillium expansum | 5.0 ± 0.0 | 5.3 ± 0.3 *** | 7.3 ± 0.6 *** |
| Penicillium citrinum | 4.8 ± 0.3 *** | 5.0 ± 0.0 *** | 8.3 ± 1.2 *** |
| Penicillium digitatum | 4.7 ± 0.6 *** | 5.2 ± 0.3 *** | 9.3 ± 0.3 *** |
| Aspergillus niger | 3.7 ± 0.3 *** | 5.3 ± 0.6 *** | 8.8 ± 0.7 *** |
3. Experimental Section
3.1. Plant Material
3.2. Isolation of the Volatile Oil
3.3. GC-FID Analysis
3.4. GC/MS Analysis
3.5. Identification of the Essential Oil Components
3.6. UPLC Analyses
3.7. Free Radical-Scavenging Capacity
3.8. Antibacterial and Antifungal Activity
4. Conclusions
Conflicts of Interest
References
- Steinitz, B.; Tabib, Y.; Gaba, V.; Gefen, T.; Vaknin, Y. Vegetative micro-cloning to sustain biodiversity of threatened Moringa species. In Vitro Cell. Dev. Biol.-Plant 2009, 45, 65–71. [Google Scholar] [CrossRef]
- Lalas, S.; Gortzi, O.; Athanasiadis, V.; Tsaknis, J.; Chinou, I. Determination of antimicrobial activity and resistance to oxidation of Moringa peregrina seed oil. Molecules 2012, 17, 2330–2334. [Google Scholar] [CrossRef]
- Fahey, J.W. Moringa oleifera: A Review of the Medical Evidence for Its Nutritional, Therapeutic, and Prophylactic Propertie. Part 1. Trees for Life J. 2005, 1, 1–33. [Google Scholar]
- Mbikay, M. Therapeutic potential of Moringa oleifera leaves in chronic hyperglycemia and dyslipidemia: A review. Front. Pharmacol. 2012, 3, 1–12. [Google Scholar] [CrossRef]
- Mukunzi, D.; Nsor-Atindana, J.; Xiaoming, Z.; Gahungu, A.; Karangwa, E.; Mukamurezi, G. Comparison of volatile profile of Moringa oleifera leaves from Rwanda and China using HS-SPME. Pakistan J. Nutr. 2011, 10, 602–608. [Google Scholar] [CrossRef]
- Asare, G.A.; Gyan, B.; Bugyei, K.; Adjei, S.; Mahama, R.; Addo, P.; Otu-Nyarko, L.; Wiredu, E.K.; Nyarko, A. Toxicity potentials of the nutraceutical Moringa oleifera at supra-supplementation levels. J. Ethnopharmacol. 2012, 139, 265–272. [Google Scholar] [CrossRef]
- Chuang, P.H.; Lee, C.W.; Chou, J.Y.; Murugan, M.; Shieh, B.J.; Chen, H.M. Anti-fungal activity of crude extracts and essential oil of Moringa oleifera Lam. Bioresource Technol. 2007, 98, 232–236. [Google Scholar] [CrossRef]
- Barreto, M.B.; Freitas, J.V.B.D.; Silveira, E.R.; Bezerra, A.M.E.; Nunes, E.P.; Gramosa, N.V. Volatile and non-volatile chemical constituents of Moringa oleifera Lam., Moringaceae. Rev. Bras. Farmacogn. 2009, 19, 893–897. [Google Scholar] [CrossRef]
- Zhao, S.; Zhang, D. Supercritical fluid extraction and characterisation of essential oil from Moringa. oleifera leaves. In Chemeca 2011–Engineering a Better World, Sydney, Australia, 18–21 September 2011.
- Siddhuraju, P.; Becker, K. Antioxidant Properties of Various Solvent Extracts of Total Phenolic Constituents from Three Different Agroclimatic Origins of Drumstick Tree (Moringa oleifera Lam.) Leaves. J. Agric. Food Chem. 2003, 51, 2144–2155. [Google Scholar] [CrossRef]
- Vongsak, B.; Sithisarn, P.; Gritsanapan, W. HPLC quantitative analysis of three major antioxidative components of Moringa oleifera leaf extracts. Planta Med. 2012, 78, PJ15. [Google Scholar]
- Li, S.Y.; Yu, Y.; Li, S.P. Identification of antioxidants in essential oil of radix Angelicae. sinensis using HPLC coupled with DAD-MS and ABTS-based assay. J. Agric. Food Chem. 2007, 55, 3358–3362. [Google Scholar] [CrossRef]
- Mateos, R.; Espartero, J.L.; Trujillo, M.; Ríos, J.J.; León-Camacho, M.; Alcudia, F.; Cert, A. Determination of Phenols, Flavones and Lignans in Virgin Olive Oils by Solid-Phase Extraction and High-Performance Liquid Chromatography with Diode Array Ultraviolet Detection. J. Agric. Food Chem. 2001, 49, 2185–2192. [Google Scholar] [CrossRef]
- Anwar, F.; Ashraf, M.; Bhanger, M.I. Interprovenance variation in the composition of Moringa oleifera oilseeds from Pakistan. J. Am. Oil Chem. Soc. 2005, 82, 45–51. [Google Scholar] [CrossRef]
- Makkar, H.P.S.; Becker, K. Nutritional value and antinutritional components of whole and ethanol extracted Moringa oleifera leaves. Anim. Feed Sci. Technol. 1996, 63, 211–228. [Google Scholar] [CrossRef]
- Hertog, M.G.L.; Hollman, P.C.H.; Venema, D.P. Optimization of a quantitative HPLC determination of potentially anticarcinogenic flavonoids in vegetables and fruits. J. Agric. Food Chem. 1992, 40, 1591–1598. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Vasilopoulou, E.; Hollman, P.; Chamalides, C.; Foufa, E.; Kaloudis, T.; Kromhout, D.; Miskaki, P.; Petrochilou, I.; Poulima, E.; et al. Nutritional composition and flavonoid content of edible wild greens and green pies: A potential rich source of antioxidant nutrients in the Mediterranean diet. Food Chem. 2000, 70, 319–323. [Google Scholar] [CrossRef]
- Pace-Asciak, C.R.; Hahn, S.; Diamandis, E.P.; Soleas, G.; Goldberg, D.M. The red wine phenolics trans-resveratrol and quercetin block human platelet aggregation and eicosanoid synthesis: Implications for protection against coronary heart disease. Clin. Chim. Acta 1995, 235, 207–219. [Google Scholar] [CrossRef]
- Kawada, N.; Seki, S.; Inoue, M.; Kuroki, T. Effect of antioxidants, resveratrol, quercetin, and N-acetylcysteine, on the functions of cultured rat hepatic stellate cells and Kupffer cells. Hepatology 1998, 27, 1265–1274. [Google Scholar] [CrossRef]
- Baghel, S.S.; Shrirastava, N.; Baghel, R.S.; Agrawal, P.; Rajput, S. A review of quercetin: Antioxidant and anticancer properties. World J. Pharm. Pharmaceut. Sci. 2012, 1, 146–160. [Google Scholar]
- Romanová, D.; Vachálková, A.; Cipák, L.; Ovesná, Z.; Rauko, P. Study of antioxidant effect of apigenin, luteolin and quercetin by DNA protective method. Neoplasma 2001, 48, 104–107. [Google Scholar]
- El-Hawary, S.S.; El-Sofany, R.H.; Abdel-Monem, A.R.; Ashour, R.S.; Sleem, A.A. Polyphenolics content and biological activity of Plectranthus amboinicus (Lour.) spreng growing in Egypt (Lamiaceae). Phcog. J. 2012, 4, 45–54. [Google Scholar]
- Yassa, N.; Masoomi, F.; Rohani Rankouhi, S.E.; Hadjiakhoondi, A. Chemical composition and antioxidant activity of the extract and essential oil of Rosa damascena from Iran, Population of Guilan. DARU J. Pharm. Sci. 2009, 17, 175–180. [Google Scholar]
- Shafaghat, A. Antioxidant, antimicrobial activities and fatty acid components of flower, Leaf, Stem and seed of Hypericum scabrum. Nat. Prod. Commun. 2011, 6, 1739–1742. [Google Scholar]
- Faleiro, M.L. The mode of antibacterial action of essential oils. In Science against microbial pathogens: communicating current research and technological advances; Microbiology Series N 3; Méndez-Vilas, A., Ed.; FORMATEX: Badajoz, Spain, 2011; Volume 2, pp. 1143–1156. [Google Scholar]
- Tiwari, B.K.; Valdramidis, V.P.; O’Donnell, C.P.; Muthukumarappan, K.; Bourke, P.; Cullen, P.J. Application of natural antimicrobials for food preservation. J. Agric. Food Chem. 2009, 57, 5987–6000. [Google Scholar]
- Sikkema, J.; De Bont, J.A.M.; Poolman, B. Mechanisms of membrane toxicity of hydrocarbons. Microbiol. Rev. 1995, 59, 201–222. [Google Scholar]
- Gill, A.O.; Holley, R.A. Mechanisms of bactericidal action of cinnamaldehyde against Listeria. monocytogenes and of eugenol against L. monocytogenes and Lactobacillus sakei. Appl. Environm. Microbiol. 2004, 70, 5750–5755. [Google Scholar]
- Cushnie, T.P.T.; Lamb, A.J. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agents 2005, 5, 343–356. [Google Scholar] [CrossRef]
- Caillet, S.; Ursachi, L.; Shareck, F.; Lacroix, M. Effect of gamma radiation and oregano essential oil on murein and ATP concentration of Staphylococcus aureus. J. Food Sci. 2009, 74, M499–M508. [Google Scholar] [CrossRef]
- Oluduro, A.O. Evaluation of antimicrobial properties and nutritional potentials of Moringa oleifera Lam. leaf in South-Western Nigeria. Malaysian J. Microbiol. 2012, 8, 59–67. [Google Scholar]
- Bukar, A.; Uba, A.; Oyeyi, T.I. Antimicrobial profile of Moringa oleifera Lam. Extracts against some food-borne microorganisms. Bayero. J. Pure Applied Sci. 2010, 3, 43–48. [Google Scholar]
- Rahman, M.M.; Rahman, M.M.; Akhter, S.; Jamal, M.A.; Pandeya, D.R.; Haque, M.A.; Alam, M.F.; Rahman, A. Control of coliform bacteria detected from diarrhea associated patients by extracts of Moringa oleifera. Nepal Med. Coll. J. 2010, 12, 12–19. [Google Scholar]
- Bhatnagar, S.S.; Santapau, H.; Desai, J.D.H.; Yellore, S.; Rao, T.N.S. Biological activity of Indian medicinal plants. Part 1. Antibacterial, Antitubercular and antifungal action. Indian J. Med. Res. 1961, 49, 799–805. [Google Scholar]
- Kalemba, D.; Kunicka, A. Antibacterial and Antifungal Properties of Essential Oils. Curr. Med. Chem. 2003, 10, 813–829. [Google Scholar] [CrossRef]
- European Pharmacopoeia, 5th ed.; Council of Europe: Strasbourg Cedex, France, 2004; Volume I, pp. 217–218.
- Jennings, W.; Shibamoto, T. Qualitative Analysis of Flavour and Fragrance Volatiles by Glasscapillary Gas Chromatography; Academic Press: New York, NY, USA, 1980. [Google Scholar]
- Davies, N.W. Gas chromatographic retention indices of monoterpenes and sesquiterpenes on methyl silicone and Carbowax 20M phases. J. Chromatogr. A 1990, 503, 1–24. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/MassSpectroscopy, 4th ed.; Allured Publishing: Carol Stream, IL, USA, 2007. [Google Scholar]
- Goodner, K.L. Practical retention index models of OV-101, DB-1, DB-5, and DB-Wax for flavor and fragrance compounds. LWT-Food Sci. Technol. 2008, 41, 951–958. [Google Scholar] [CrossRef]
- Wiley Registry of Mass Spectral Data, with NIST Spectral Data CD Rom, 7th ed.; John Wiley & Sons: New York, NY, USA, 1998.
- Fratianni, F.; Nazzaro, F.; Marandino, A.; Fusco, M.D.R.; Coppola, R.; De Feo, V.; De Martino, L. Biochemical composition, Antimicrobial activities, and Anti-quorum-sensing activities of ethanol and ethyl acetate extracts from Hypericum connatum Lam. (Guttiferae). J. Med. Food 2013, 16, 454–459. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of free radical method to evaluate antioxidant activity. Food Sci. Technol. 1995, 28, 25–30. [Google Scholar]
- De Martino, L.; De Feo, V.; Fratianni, F.; Nazzaro, F. Chemistry, antioxidant, antibacterial and antifungal activities of volatile oils and their components. Nat. Prod. Commun. 2009, 4, 1741–1750. [Google Scholar]
- Sample Availability: Samples of the essential oil are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Marrufo, T.; Nazzaro, F.; Mancini, E.; Fratianni, F.; Coppola, R.; De Martino, L.; Agostinho, A.B.; De Feo, V. Chemical Composition and Biological Activity of the Essential Oil from Leaves of Moringa oleifera Lam. Cultivated in Mozambique. Molecules 2013, 18, 10989-11000. https://doi.org/10.3390/molecules180910989
Marrufo T, Nazzaro F, Mancini E, Fratianni F, Coppola R, De Martino L, Agostinho AB, De Feo V. Chemical Composition and Biological Activity of the Essential Oil from Leaves of Moringa oleifera Lam. Cultivated in Mozambique. Molecules. 2013; 18(9):10989-11000. https://doi.org/10.3390/molecules180910989
Chicago/Turabian StyleMarrufo, Tatiana, Filomena Nazzaro, Emilia Mancini, Florinda Fratianni, Raffaele Coppola, Laura De Martino, Adelaide Bela Agostinho, and Vincenzo De Feo. 2013. "Chemical Composition and Biological Activity of the Essential Oil from Leaves of Moringa oleifera Lam. Cultivated in Mozambique" Molecules 18, no. 9: 10989-11000. https://doi.org/10.3390/molecules180910989
APA StyleMarrufo, T., Nazzaro, F., Mancini, E., Fratianni, F., Coppola, R., De Martino, L., Agostinho, A. B., & De Feo, V. (2013). Chemical Composition and Biological Activity of the Essential Oil from Leaves of Moringa oleifera Lam. Cultivated in Mozambique. Molecules, 18(9), 10989-11000. https://doi.org/10.3390/molecules180910989









