Syzygium jambos and Solanum guaraniticum Show Similar Antioxidant Properties but Induce Different Enzymatic Activities in the Brain of Rats
Abstract
:1. Introduction
2. Results and Discussion
2.1. Pytochemical Screening of Aqueous Extract of Syzygium jambos and Solanum guaraniticum
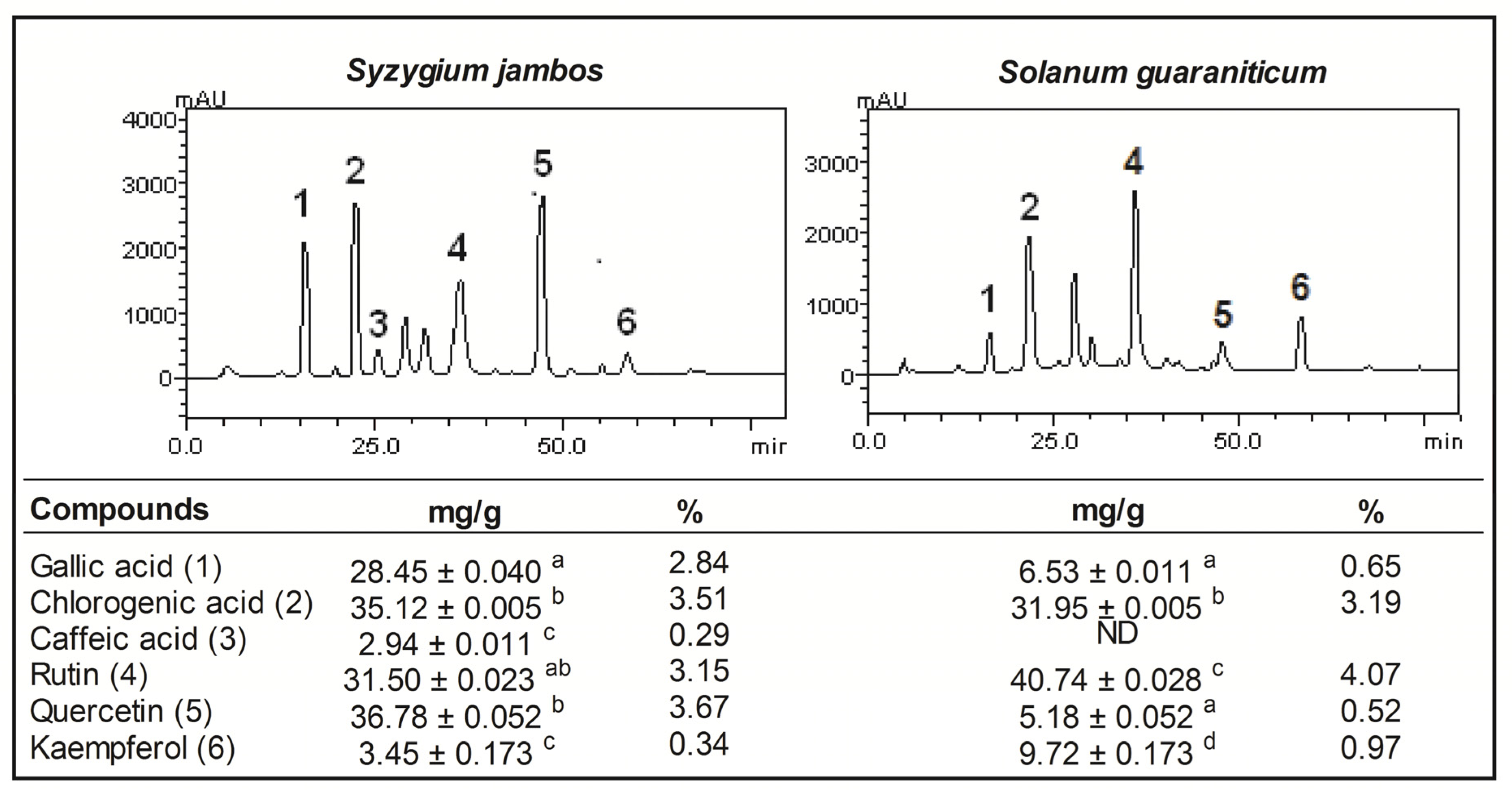
| Extract content | Syzygium jambos | Solanum guaraniticum | ||
|---|---|---|---|---|
| Total phenolic (mg GAE/g) | 108.2 ± 3.34 | 58.76 ± 1.72 ** | ||
| Total flavonoid (mg QE/g) | 85.55 ± 2.54 | 237.90 ± 7.12 *** | ||
| Vitamin C (mg VIT C/g) | 21.07 ± 0.64 | 58.01 ± 4.21 ** | ||
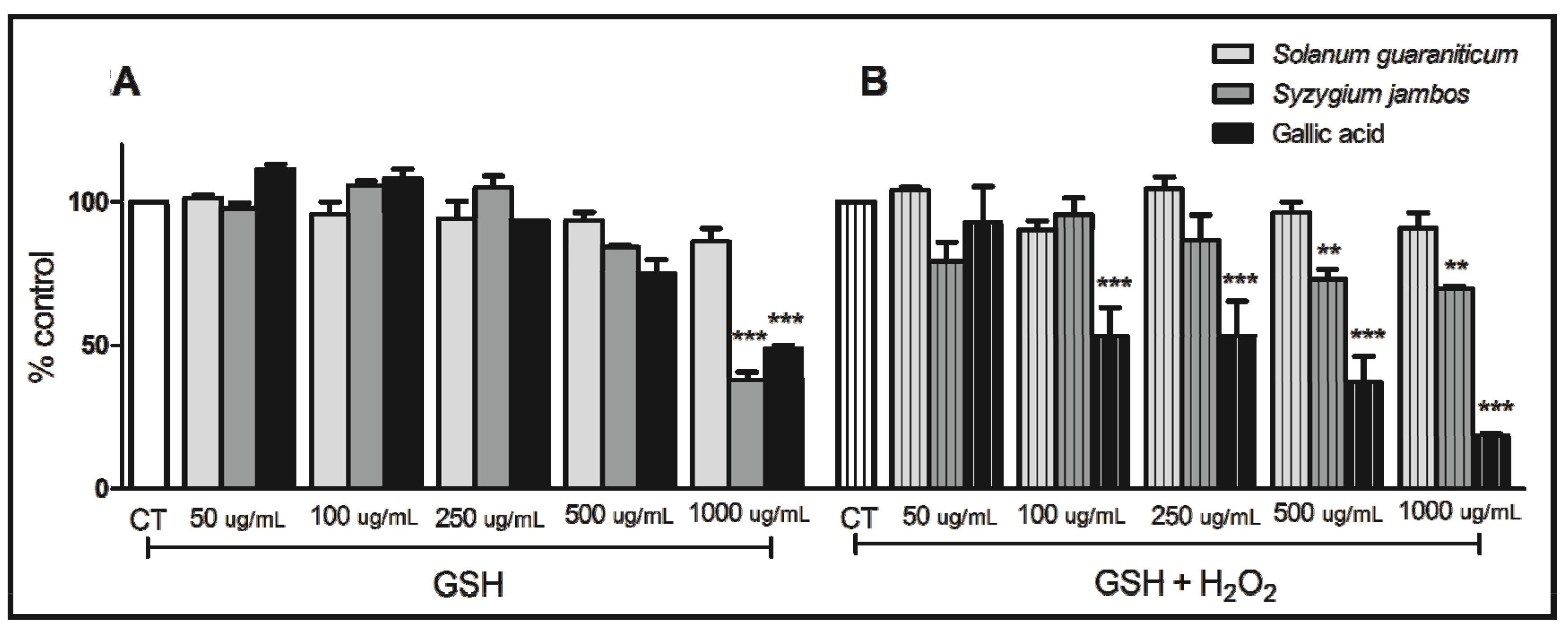
2.2. Thiol Peroxidase-Like Activity of Both Extracts Evaluated
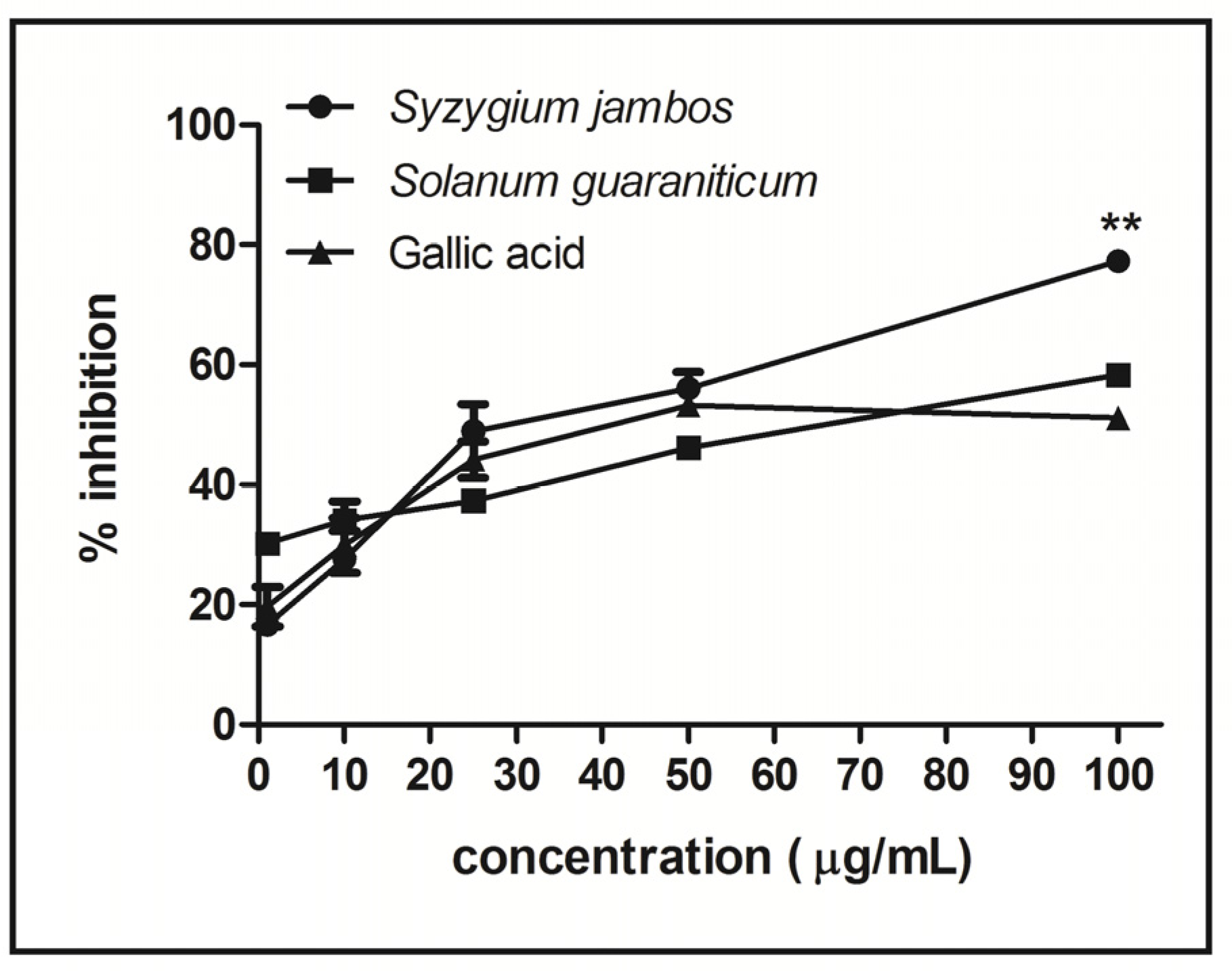
2.3. Nitric Oxide-Scavenging Activity Assay of Extracts
2.4. Effect of Extracts on δ-ALA-D and AChE Activity in Rat Tissues
| Treatment | δ-ALA-D | AChE | ||
|---|---|---|---|---|
| Liver | Brain | Kidney | ||
| Control (PBS) | 4.33 ± 0.48 | 1.23 ± 0.18 | 1.49 ± 0.43 | 2.78 ± 0.14 |
| Lead acetate 10 µM | 2.92 ± 0.23 | 0.95 ± 0.02 | 0.99 ± 0.43 | - |
| Paraoxon 1 µM | - | - | - | 1.26 ± 0.07 *** |
| Syzygium jambos 100 µg/ mL | 4.85 ± 0.71 | 1.10 ± 0.06 | 1.89 ± 0.49 | 2.65 ± 0.14 |
| Syzygium jambos 250 µg/mL | 4.09 ± 0.46 | 1.38 ± 0.28 | 1.97 ± 0.53 | 2.75 ± 0.14 |
| Solanum guaraniticum 500 µg/mL | 4.24 ± 0.52 | 0.91 ± 0.11 | 1.66 ± 0.32 | 2.73 ± 0.14 |
| Solanum guaraniticum 1000 µg/mL | 4.31 ± 0.72 | 0.66 ± 0.14 * | 1.35 ± 0.18 | 2.79 ± 0.14 |
2.5. Effects of Syzygium jambos and Solanum guaraniticum on Lipid Peroxidation, NPSH Content and Catalase Activity of Sodium Nitroprusside (SNP)-Induced Tissues
| Treatments | Liver | Inhibition (%) | Brain | Inhibition (%) | Kidney | Inhibition (%) |
|---|---|---|---|---|---|---|
| Control | 2.23 ± 0.54 | ------- | 3.36 ± 0.93 | ------- | 1.62 ± 0.57 | ------- |
| Gallic acid 25 µg/mL | 1.26 ± 0.42 | 43.49 | 1.57 ± 0.51 | 53.27 | 1.29 ± 0.13 | 20.37 |
| Syzygium jambos 100 µg/mL | 1.98 ± 0.52 | 11.21 | 1.53 ± 0.18 * | 54.46 | 0.93 ± 0.25 | 42.59 |
| Syzygium jambos 250 µg/mL | 2.01 ± 0.51 | 9.86 | 1.19 ± 0.26 ** | 64.58 | 0.73 ± 0.18 | 54.93 |
| Solanum guaraniticum 500 µg/mL | 2.36 ± 0.53 | ------- | 1.49 ± 0.26 * | 55.65 | 0.81 ± 0.15 | 50.00 |
| Solanum guaraniticum 1000 µg/mL | 2.41 ± 0.40 | ------- | 1.89 ± 0.35 * | 43.75 | 0.84 ± 0.14 | 48.14 |
| Induced (SNP) | 10.81 ± 1.45 *** | ------- | 30.61 ± 3.41 *** | ------- | 4.78 ± 1.47 * | ------- |
| Gallic acid 25 µg/mL | 1.08 ± 0.09 ### | 90.00 | 27.88 ± 1.15 | 8.91 | 1.30 ± 0.22 ### | 74.26 |
| Syzygium jambos 100 µg/mL + SNP | 2.39 ± 0.51 ### | 77.89 | 2.97 ± 0.61 ### | 90.29 | 1.09 ± 0.30 ## | 77.19 |
| Syzygium jambos 250 µg/mL + SNP | 2.33 ± 0.47 ### | 78.44 | 1.95 ± 0.78 ### | 93.62 | 0.86 ± 0.10 ## | 82.00 |
| Solanum guaraniticum 500 µg/mL + SNP | 2.58 ± 0.65 ### | 76.13 | 1.86 ± 0.80 ### | 93.92 | 1.00 ± 0.20 ## | 79.07 |
| Solanum guaraniticum 1000 µg/mL + SNP | 2.75 ± 0.59 ## | 74.56 | 2.77 ± 0.74 ### | 90.95 | 1.18 ± 0.17 ## | 75.31 |
| Treatments | Thiol content | CAT | ||||
|---|---|---|---|---|---|---|
| Liver | Brain | Kidney | Liver | Brain | Kidney | |
| Data are reported as mean ± SEM (n = 6) and assessed by one way ANOVA followed by Duncan Multiple Comparison post hoc test. Results are presented as mmol NPSH (non-protein thiol)/mg protein and units of catalase (CAT)/mg protein. (*) and (**) denotes p < 0.05 and p < 0.001, respectively, as compared to the respective control samples. (#), (##) and (###) denotes p < 0.05, p < 0.001 and p < 0.0001, respectively, as compared to the respective induced samples. | ||||||
| Control | 11.16 ± 2.16 | 7.57 ± 1.01 | 5.66 ± 1.19 | 38.14 ± 6.49 | 6.36 ± 1,49 | 17.71 ± 2.22 |
| Gallic acid 25 µg/mL | 11.44 ± 2.6 | 9.85 ± 0.29 | 7.31 ± 0.78 | 40.28 ± 9.87 | 6.34 ± 0.34 | 26.53 ± 1.53 * |
| Syzygium jambos 100 µg/mL | 7.41 ± 0.84 | 9.12 ± 0.50 | 6.21 ± 1.81 | 37.65 ± 7.81 | 4.16 ± 1.25 | 15.41 ± 2.75 |
| Syzygium jambos 250 µg/mL | 7.32 ± 1.89 | 10.66 ± 0.73 | 9.73 ± 1.36 | 28.72 ± 6.54 | 2.80 ± 1.10 | 13.84 ± 2.33 |
| Solanum guaraniticum 500 µg/mL | 6.42 ± 1.82 | 9.20 ± 1.00 | 10.52 ± 1.76 * | 41.98 ± 6.18 | 3.43 ± 1.07 | 20.80 ± 2.03 |
| Solanum guaraniticum 1000 µg/mL | 10.94 ± 2.35 | 10.91 ± 1.69 | 8.84 ± 0.20 | 49.10 ± 6.45 | 3.37 ± 1.17 | 18.82 ± 1.65 |
| Induced (SNP) | 4.84 ± 1.69 * | 2.64 ± 0.57 ** | 5.42 ± 1.70 | 46.78 ± 9.98 | 4.04 ± 1.17 | 16.74 ± 1.01 |
| Gallic acid 25 µg/mL | 5.51 ± 0.57 | 3,01 ± 0.20 | 5.43 ± 0.54 | 53.47 ± 10.44 | 2.95 ± 0.56 | 26.52 ± 3.08 # |
| Syzygium jambos 100 µg/mL + SNP | 5.80 ± 1.46 | 7.52 ± 0.79 ## | 6.13 ± 1.00 | 48.04 ± 7.70 | 4.13 ± 1.42 | 17.92 ± 1.52 |
| Syzygium jambos 250 µg/mL + SNP | 6.66 ± 0.90 | 9.40 ± 0.65 ### | 10.56 ± 1.96 # | 40.04 ± 9.82 | 2.37 ± 1.37 | 19.47 ± 3.23 |
| Solanum guaraniticum 500 µg/mL + SNP | 8.45 ± 1.87 | 7.30 ± 0.52 ## | 6.95 ± 0.62 | 47.69 ± 13.39 | 4.17 ± 1.01 | 19.18 ± 1.17 |
| Solanum guaraniticum 1000 µg/mL + SNP | 7.96 ± 1.67 | 9.03 ± 0.86 ### | 9.50 ± 1.51 | 41.60 ± 11.14 | 3.52 ± 1.06 | 19.18 ± 1.69 |
3. Experimental
3.1. Chemicals and Apparatus
3.2. Plant Material and Preparation of Extracts
3.3. Phytochemical Analysis
3.3.1. High-Performance Liquid Chromatography (HPLC) Characterization
3.3.2. Total Polyphenols Content
3.3.3. Total Flavonoid Content
3.3.4. Determination of Vitamin C Content
3.4. Nitric Oxide-Scavenging Assay of Extracts
3.5. Thiol Peroxidase-Like Activity of Extracts
3.6. Animals
3.6.2. Acetylcholinesterase Activity Assay for Brain
3.6.3. Sodium Nitroprusside (SNP) induced Oxidative Stress
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Nordberg, J.; Arner, E.S.J. Reactive oxygen species, antioxidants, and the mammalian thioredoxin system. Free Radic. Biol. Med. 2001, 3, 1287–1312. [Google Scholar] [CrossRef]
- Halliwell, B. Free radicals and antioxidants, and human disease: Curiosity, cause, or consequence. Lancet 1994, 344, 721–724. [Google Scholar] [CrossRef]
- Cho, E.J.; Lee, Y.A.; Yoo, H.H.; Yokozawa, T. Protective effects of broccoli (Brassica oleracea) against oxidative damage in vitro and in vivo. J. Nutr. Sci. Vitaminol. 2006, 52, 437–444. [Google Scholar] [CrossRef]
- Bahramikia, S.; Yazdanparast, R. Antioxidant efficacy of nasturtium officinale extracts using various in vitro assay systems. J. Acupunct. Meridian Stud. 2010, 3, 283–290. [Google Scholar] [CrossRef]
- Pereira, R.P.; Fachinetto, R.; de Souza Prestes, A.; Puntel, R.L.; Santos da Silva, G.N.; Heinzmann, B.M.; Boschetti, T.K.; Athayde, M.L.; Bürger, M.E.; Morel, A.F.; et al. Antioxidant effects of different extracts from Melissa officinalis, Matricaria recutita and Cymbopogon citratus. Neurochem. Res. 2009, 34, 973–983. [Google Scholar] [CrossRef]
- Morton, J.F. Rose Apple. USA: Fruits of Warm Climates; Florida Flair Books: Miami, FL, USA, 1987; pp. 383–386. [Google Scholar]
- Slowing, K.; Carretero, E.; Villar, A. Anti-inflammatory activity of leaf extracts of Eugenia jambos in rats. J. Ethnopharmacol. 1994, 43, 9–11. [Google Scholar] [CrossRef]
- Slowing, K.; Carretero, E.; Villar, A. Anti-inflammatory compounds of Eugenia jambos. Phytother. Res. 1996, 10, 8126–8127. [Google Scholar]
- Teixeira, C.C.; Fuchs, F.D.; Blotta, R.M.; Knijnik, J.; Delcado, I.C.; Netto, M.S.; Ferreira, E.; Costa, A.P.; Mussnich, D.G.; Ranquetat, G.G.; et al. Effect of tea prepared from leaves of Syzygium jambos on glucose tolerance in nondiabetic subjects. Diabetes Care 1990, 13, 907–908. [Google Scholar]
- Costa, O.A. Jurubeba. Revista Brasileira De Farmácia 1940, 21, 404–416. [Google Scholar]
- Costa, O.F. Farmacognosia, 2nd ed.; Ed Lisboa-Fundacao Calouste-Gulbenkian: Lisboa, Portugal, 1975. [Google Scholar]
- Penna, M. Dicionario Brasileiro de Plantas Medicinais; Kosmos: Rio de Janeiro, Brazil, 1964. [Google Scholar]
- Paulovich, F.B.; Portiansky, E.L.; Gimeno, E.J.; Schild, A.L.; Mendez, M.C.; Riet-Correa, F. Lectin histochemical study of lipopigments present in the cerebellum of Solanum fastigiatum var. fastigiatum intoxicated cattle. J. Vet. Med. A Physiol. Pathol Clin. Med. 2002, 49, 473–477. [Google Scholar] [CrossRef]
- Rech, R.R.; Rissi, D.R.; Rodrigues, A.; Pierezan, F.; Piazer, J.V.M.; Kommers, G.D.; Barros, C.S.L. Intoxicação por Solanum fastigiatum (Solanaceae) em bovinos: epidemiologia, sinais clínicos e morfometria das lesões cerebelares. Pesq. Vet. Bras. 2006, 26, 183–189. [Google Scholar]
- Sabir, S.M.; Rocha, J.B.T. Antioxidant and hepatoprotective activity of aqueous extract of Solanum fastigiatum (false “Jurubeba”) against paracetamol-induced liver damage in mice. J. Ethnopharmacol. 2008, 120, 226–232. [Google Scholar] [CrossRef]
- Rosidah; Yam, M.F.; Sadikun, A.; Ahmad, M.; Akowuah, G.A.; Asmawi, M.Z. Toxicology evaluation of standardized methanol extract of Gynura procumbens. J. Ethnopharmacol. 2009, 123, 244–249. [Google Scholar] [CrossRef]
- Bevan, D.R.; Bodlaender, P.; Shemin, D. Mechanism of porphobilinogen synthase. Requirement of Zn2+ for enzyme activity. J. Biol. Chem. 1980, 255, 2030–2035. [Google Scholar]
- Brito, V.B.; Folmer, V.; Soares, J.C.M.; Silveira, I.D. Long-term sucrose and glucose consumption decreases the δ-aminolevulinate dehydratase activity in mice. Nutrition 2007, 23, 818–826. [Google Scholar] [CrossRef]
- Nogueira, C.W.; Borges, V.C.; Zeni, G.; Rocha, J.B.T. Organochalogens effects on δ-aminolevulinate dehydratase activity from human erythrocytic cells in vitro. Toxicology 2003, 191, 169–178. [Google Scholar] [CrossRef]
- Souza, A.C.G.; Luchese, C.; Santos Neto, J.S.; Nogueira, C.W. Antioxidant effect of a novel class of telluroacetilene compounds: Studies in vitro and in vivo. Life Sci. 2009, 84, 351–357. [Google Scholar] [CrossRef]
- Silver, A. The Biology of Cholinesterases; North-Holland Publishing Company: Amsterdam, The Netherlands, 1974; p. 596. [Google Scholar]
- Pohanka, M. Cholinesterases, a target of pharmacology and toxicology. Biomed. Papers 2011, 155, 219–223. [Google Scholar] [CrossRef]
- Shenouda, J.; Green, P.; Sultatos, L. An evaluation of the inhibition of human butyrylcholinesterase and acetylcholinesterase by the organophosphate chlorpyrifos oxon. Toxicol Appl. Pharmacol. 2009, 241, 135–142. [Google Scholar] [CrossRef]
- Zadra, M.; Piana, M.; Brum, T.F.; Boligon, A.A.; de Freitas, R.B.; Machado, M.M.; Stefanello, S.T.; Soares, F.A.A.; Athayde, M.L. Antioxidant activity and phytochemical composition of the leaves of Solanum guaraniticum A. St.-Hil. Molecules 2012, 17, 12560–12574. [Google Scholar] [CrossRef]
- Banerjee, A.; Nabasree, D.; De, B. In vitro study of antioxidant activity of Syzygium cumini fruit. Food Chem. 2005, 90, 727–733. [Google Scholar] [CrossRef]
- Padayatty, S.J.; Katz, A.; Wang, Y.; Eck, P.; Kwon, O.; Lee, J.H.; Chen, S.; Corpe, C.; Dutta, A.; Dutta, S.K.; et al. Vitamin C as an antioxidant: Evaluation of its role in disease prevention. J. Am. Coll. Nutr. 2003, 22, 18–35. [Google Scholar] [CrossRef]
- Oboh, G. Effect of blanching on the antioxidant properties of some tropical green leafy G vegetables. Food Sci. Technol. 2005, 38, 513–517. [Google Scholar]
- Materska, M.; Perucka, I. Antioxidant activity of the main phenolic compounds isolated from hot pepper fruit (Capsicum annuum L.). J. Agric. Food Chem. 2005, 53, 1750–1756. [Google Scholar] [CrossRef]
- Amic, D.; Davidovic-Amic, D.; Beslo, D.; Trinajstic, N. Structure-radical scavenging activity relationship of flavonoids. Croat. Chem. Acta 2003, 76, 55–61. [Google Scholar]
- Flohe, L.; Gunzler, W.A.; Schock, H.H. Glutathione peroxidase: A selenoenzyme. FEBS Lett. 1973, 32, 132–134. [Google Scholar] [CrossRef]
- Sakihama, Y.; Cohen, M.F.; Grace, S.C.; Yamasaki, H. Plant phenolic antioxidant and prooxidant activities: Phenolics-induced oxidative damage mediated by metals in plants. Toxicology 2002, 177, 67–80. [Google Scholar] [CrossRef]
- Riet-Correa, F.; Méndez, M.C.; Schild, A.L.; Summers, B.A.; Oliveira, J.A. Intoxication by Solanum fastigiatum var. fastigiatum as a cause of cerebellar degeneration of cattle. Cornell Vet. 1983, 73, 240–256. [Google Scholar]
- Prigol, M.; Wilhelm, E.A.; Schneider, C.C.; Rocha, J.B.T.; Nogueira, C.W.; Zeni, G. Involvement of oxidative stress in seizures induced by diphenyl diselenide in rat pups. Brain Res. 2007, 1147, 226–232. [Google Scholar] [CrossRef]
- Emanuelli, T.; Prauchner, C.A.; Dacanal, J.; Zeni, A.; Reis, E.C.; de Mello, C.F.; de Souza, D.O. Intrastriatal administration of 5-aminolevulinic acid induces convulsions and body asymmetry through glutamatergic mechanisms. Brain Res. 2000, 868, 88–94. [Google Scholar] [CrossRef]
- Hermes-Lima, M.; Pereira, B.; Bechara, E.J. Are free radicals involved in lead poisoning? Xenobiotica 1991, 21, 1085–1090. [Google Scholar] [CrossRef]
- Floyd, R.A.; Zaleska, M.M.; Harmon, J. Free Radicals in Molecular Biology, Aging and Disease; Armstrong, D., Sohal, R.S., Cutler, R.G., Slater, T.F., Eds.; Raven Press: New York, NY, USA, 1984; pp. 143–161. [Google Scholar]
- Freitas, R.M. Lipoic acid alters d-aminolevulinic dehydratase, glutathione peroxidase and Na+, K+-ATPase activities and glutathione reduced levels in rat hippocampus after pilocarpineinduced seizures. Cell. Mol. Neurobiol. 2010, 30, 381–387. [Google Scholar] [CrossRef]
- Oboh, G.; Rocha, J.B.T. Water extractable phytochemicals from Capsicu pubescens (tree pepper) inhibit lipid peroxidation induced by different pro-oxidant agents in brain: In vitro. Eur. Food Res. Technol. 2008, 226, 707–713. [Google Scholar] [CrossRef]
- Manian, R.; Anusuya, N.; Siddhuraju, P.; Manian, S. The antioxidant activity and free radical scavenging potential of two different solvent extracts of Camellia sinensis (L.) O. Kuntz, Ficus bengalensis L. and Ficus racemosa L. Food Chem 2008, 107, 1000–1007. [Google Scholar] [CrossRef]
- Awaha, F.M.; Uzoegwua, P.N.; Ifeonua, P.; Oyugib, J.O.; Rutherfordb, J.; Yao, X.; Fehrmannb, F.; Fowkeb, K.R.; Eze, M.O. Free radical scavenging activity, phenolic contents and cytotoxicity of selected Nigerian medicinal plants. Food Chem. 2012, 131, 1279–1286. [Google Scholar]
- Giugliano, D.; Ceriello, A.; Paolisso, G. Oxidative stress and diabetic vascular complications. Diabetes Care 1996, 19, 257–267. [Google Scholar]
- Zhu, R.; Wang, Y.; Zhang, L.; Guo, Q. Oxidative stress and liver disease. Hepatol. Res. 2012, 42, 741–749. [Google Scholar] [CrossRef]
- Vega-Naredo, I.; Poeggeler, B.; Sierra-Sánchez, V.; Caballero, B.; Tomás-Zapico, C.; Alvarez-García, O.; Tolivia., D.; Rodríguez-Colunga, M.J.; Coto-Montes, A. Melatonin neutralizes neurotoxicity induced by quinolinic acid in brain tissue culture. J. Pineal Res. 2005, 39, 266–275. [Google Scholar] [CrossRef]
- Sies, H. Glutathione and its role in cellular functions. Free Radic. Biol. Med. 1999, 27, 916–921. [Google Scholar] [CrossRef]
- Calabrese, V.; Cornelius, C.; Dinkova-Kostova, A.T.; Iavicoli, I.; di Paola, R.; Koverech, A.; Cuzzocrea, S.; Rizzarelli, E.; Calabrese, E.J. Cellular stress responses, hormetic phytochemicals and vitagenes in aging and longevity. Biochim. Biophys. Acta 2012, 1822, 753–783. [Google Scholar] [CrossRef]
- Posser, T.; Moretto, M.B.; Dafre, A.L.; Farina, M.; da Rocha, J.B.; Nogueira, C.W.; Zeni, G.; Ferreira, J.S.; Leal, R.B.; France, J.L. Antioxidant effect of diphenyl diselenide against sodium nitroprusside (SNP) induced lipid peroxidation in human platelets and erythrocyte membranes: An in vitro evaluation. Chem. Biol. Interact. 2006, 164, 126–135. [Google Scholar] [CrossRef]
- Li, S.; Yan, T.; Yang, J.Q.; Oberley, T.D.; Oberley, L.W. The role of cellular glutathione peroxidase redox regulation in the suppression of tumor cell growth by manganese superoxide dismutase. Cancer Res. 2000, 60, 3927–3939. [Google Scholar]
- Islam, M.R.; Parvin, M.S.; Islam, M.E. Antioxidant and hepatoprotective activity of an ethanol extract of Syzygium jambos (L.) leaves. Drug Discov. Ther. 2012, 6, 205–211. [Google Scholar]
- Subramanian, K.N.; Padmanaban, G.; Sarma, P.S. Folin-Ciocalteu reagent for the estimation of siderochromes. Anal. Biochem. 1965, 12, 106–112. [Google Scholar] [CrossRef]
- Zhishen, J.; Mengcheng, T.W.J. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Benderitter, M.; Maupoil, V.; Vergely, C.; Dalloz, F.; Briot, F.; Rochette, L. Studies by electron paramagnetic resonance of the importance of iron in the hydroxyl scavenging properties of ascorbic acid in plasma: Effects of iron chelators. Fundam. Clin. Pharmacol. 1998, 12, 510–516. [Google Scholar] [CrossRef]
- Sreejayan, N.; Rao, M.N.A. Nitric oxide scavenging by curcuminoids. J. Pharm. Pharmacol. 1997, 49, 105–107. [Google Scholar] [CrossRef]
- Ellman, G.L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 1959, 82, 70–77. [Google Scholar] [CrossRef]
- Peterson, G.L. A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal. Biochem. 1977, 83, 346–356. [Google Scholar] [CrossRef]
- Sassa, S. Delta-aminolevulinic acid dehydratase assay. Enzyme 1982, 28, 133–145. [Google Scholar]
- Peixoto, N.C.; Roza, T.; Pereira, M.E. Sensitivity of d-ALA-D (E.C. 4.2.1.24) of rats to metals in vitro depends on the stage of postnatal growth and tissue. Toxicol. In Vitro 2004, 18, 805–809. [Google Scholar] [CrossRef]
- Ellman, G.L.; Courtney, K.D.; Andres, V. JR.; Feather-stone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Rocha, J.B.; Emanuelli, T.; Pereira, M.E. Effects of early undernutrition on kinetic parameters of brain acetylcholinesterase from adult rats. Acta Neurobiol. Exp. 1993, 53, 431–437. [Google Scholar]
- Herkerta, N.M.; Freudeb, G.; Kunzb, U.; Thiermanna, H.; Woreka, F. Comparative kinetics of organophosphates and oximes with erythrocyte, muscle and brain acetylcholinesterase. Toxicol. Lett. 2012, 209, 173–178. [Google Scholar] [CrossRef]
- Niehaus, W.G.J.; Samuelsson, B. Formation of malonaldehyde from phospholipid arachidonate during microsomal lipid peroxidation. Eur. J. Biochem. 1968, 6, 126–130. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. Methods Enzymol. 1984, 105, 121–125. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bonfanti, G.; Bitencourt, P.R.; Bona, K.S.d.; Silva, P.S.d.; Jantsch, L.B.; Pigatto, A.S.; Boligon, A.; Athayde, M.L.; Gonçalves, T.L.; Moretto, M.B. Syzygium jambos and Solanum guaraniticum Show Similar Antioxidant Properties but Induce Different Enzymatic Activities in the Brain of Rats. Molecules 2013, 18, 9179-9194. https://doi.org/10.3390/molecules18089179
Bonfanti G, Bitencourt PR, Bona KSd, Silva PSd, Jantsch LB, Pigatto AS, Boligon A, Athayde ML, Gonçalves TL, Moretto MB. Syzygium jambos and Solanum guaraniticum Show Similar Antioxidant Properties but Induce Different Enzymatic Activities in the Brain of Rats. Molecules. 2013; 18(8):9179-9194. https://doi.org/10.3390/molecules18089179
Chicago/Turabian StyleBonfanti, Gabriela, Paula Rodrigues Bitencourt, Karine Santos de Bona, Priscila Sabino da Silva, Letícia B. Jantsch, Aline S. Pigatto, Aline Boligon, Margareth L. Athayde, Thissiane L. Gonçalves, and Maria Beatriz Moretto. 2013. "Syzygium jambos and Solanum guaraniticum Show Similar Antioxidant Properties but Induce Different Enzymatic Activities in the Brain of Rats" Molecules 18, no. 8: 9179-9194. https://doi.org/10.3390/molecules18089179
APA StyleBonfanti, G., Bitencourt, P. R., Bona, K. S. d., Silva, P. S. d., Jantsch, L. B., Pigatto, A. S., Boligon, A., Athayde, M. L., Gonçalves, T. L., & Moretto, M. B. (2013). Syzygium jambos and Solanum guaraniticum Show Similar Antioxidant Properties but Induce Different Enzymatic Activities in the Brain of Rats. Molecules, 18(8), 9179-9194. https://doi.org/10.3390/molecules18089179




